Abstract
Imaging the heart is one of the most technically challenging applications of Computed Tomography (CT) due to the presence of cardiac motion limiting optimal visualization of small structures such as the coronary arteries. Electrocardiographic gating during CT data acquisition facilitates motion free imaging of the coronary arteries. Since publishing the first version of the Canadian Association of Radiologists (CAR) cardiac CT guidelines, many technological advances in CT hardware and software have emerged necessitating an update. The goal of these cardiac CT practice guidelines is to present an overview of the current evidence supporting the use of cardiac CT in various clinical scenarios and to outline standards of practice for patient safety and quality of care when establishing a cardiac CT program in Canada.
These Practice Guidelines have been reviewed and endorsed by the North American Society for Cardiovascular Imaging (NASCI).
Introduction
Many technological advances in computed tomography (CT) hardware and software have emerged since the first version of the Canadian Association of Radiologists (CAR) cardiac CT practice guidelines. 1 This has prompted a revision that incorporates technological improvements and changes in clinical practice to align with recently published international guidelines. The goal of these cardiac CT practice guidelines is to present an overview of the current evidence supporting the use of cardiac CT in various clinical scenarios and to outline standards of practice for patient safety and quality of care when establishing a cardiac CT program in Canada. Coronary computed tomography angiography (CCTA) is a major component of cardiac CT volume that also includes valvular and pericardial imaging, tumour and mass evaluation, pulmonary vein imaging to guide ablation procedures, and imaging of congenital heart disease for diagnosis and monitoring.
Recommendations were written by a working group of Canadian cardiac imaging experts informed by a current literature review that prioritized systematic reviews and randomized controlled trials, where available, or based on consensus expert opinion in the absence of high-quality studies. These guidelines are intended to be relevant for community hospitals, as well as large academic centres that conduct cardiac CT. Part I focuses on CCTA and Part II outlines the use of cardiac CT for non-coronary indications.
Standards for the Performance of Cardiac CT
CT Requirements
Imaging the heart is one of the most technically challenging applications of CT due to the presence of cardiac motion limiting optimal visualization of small structures such as the coronary arteries. Electrocardiographic gating during CT data acquisition facilitates motion free imaging of the coronary arteries and their sub-millimetre branches. The ideal cardiac imaging platform combines high spatial and temporal resolution with high X-ray tube capacity while maintaining low radiation dose.
When the previous CAR guidelines were published, the mainstay of cardiac imaging consisted of 64-detector row CT scanners. 1 Since then, some CT scanner manufacturers have increased the number of detector rows facilitating complete coverage of the heart in one gantry rotation (ie, 320-detector row). Other CT scan manufacturers optimized dual X-ray tube design for greater temporal resolution in the range of 66 ms that further reduced cardiac motion blurring, even at higher heart rates, thereby reducing the need for beta-blockers.
Advances in noise reducing reconstruction algorithms were also introduced, facilitating more effective radiation dose reduction.2,3
For CCTA, the minimum CT scanner specifications should meet or exceed criteria outlined by the American College of Radiology, North American Society for Cardiovascular Imaging, and the Society for Pediatric Radiology practice parameters as listed below 4 :
• 64-detector row or greater
• Axial resolution ≤0.5 × 0.5 mm
• Z-axis resolution ≤1 mm
• Temporal resolution ≤250 ms
Volume scanners (128-detector rows or greater) or dual-source scanners are much more robust than 64-detector row scanners and are generally preferred for cardiac imaging.
Radiation Safety
Depending on the type of CT scanner and specific technique used, ECG-gated cardiac CT can result in higher radiation dose exposure to the patient compared to helical non-gated CT. Published dose estimates are often erroneous due to the use of dose conversion factors that do not account for patient size or characteristics, or CT technique.5,6 Current CT scanners offer a wide variety of radiation dose reduction strategies.
ECG-gated cardiac CT synchronizes data acquisition to the cardiac cycle, usually in diastole when there is less motion while the ventricles are filling, to minimize cardiac motion blurring. Retrospective ECG-gating is required for ventricular functional assessment and may also be needed when heart rates are high. However, it results in higher radiation dose compared to prospective ECG-triggered or helical non-gated techniques. ECG-based tube current modulation, whereby the mAs is reduced to 20% or 40% during part of the cardiac cycle, typically systole and utilizing the lowest kVp possible, can significantly reduce radiation exposure. 7 At higher heart rates systolic phase imaging may be optimal due to decreased diastolic filling time and relatively less motion at the end of systole at higher heart rates.8,9
Prospective ECG-triggering is most commonly used for CCTA (Table 1). The X-ray tube output and data acquisition are timed to diastole when there is less cardiac motion. Depending on the number of detector rows and scan coverage per gantry rotation, multiple heart beats may be required to image the entire heart. In contrast, CT scanners that have 320 detector rows can cover the entire heart with a single gantry rotation and thus are less susceptible to arrhythmias such as atrial fibrillation.
Common Indications and Scan Techniques.
A high pitch helical scan (pitch of 3.2-3.4) is available on dual source CT scanners that enables imaging of the entire heart during a single beat, which reduces radiation exposure 7 and facilitates sub-millisievert imaging. 10 Use of this scanning mode may require more aggressive beta-blockade to achieve heart rates of <65 beats per minute for third-generation scanners. 11
Higher tube potential or kVp contributes to higher radiation dose in a non-linear relationship. Imaging with a lower kVp means that a greater proportion of X-ray is near the K-edge of iodine, resulting in improved contrast enhancement per volume of contrast used due to increased photoelectric interactions. By reducing the tube potential from 120 to 100 kVp, it is possible to reduce the radiation dose while improving vascular enhancement. The ability to reduce kVp is often guided by the patient’s weight. Larger patients require a higher kVp for optimal image quality as higher energy X-ray photons are needed to penetrate the soft tissues and reach the detectors. 9
The use of automatic exposure control, or tube current modulation along the z-axis, adjusts the tube current to the attenuation of the region being scanned. In this way, the image noise is kept at a constant level and the tube current is reduced when scanning less attenuating anatomy.
Filtered back projection was the standard method of image reconstruction for several decades after the introduction of CT technology. Due to increased computational power, more advanced noise reduction methods such as iterative reconstruction were introduced in 2008 as an effective radiation dose reduction strategy.12-14 Iterative reconstruction reduces image noise and permits imaging with lower radiation doses. Recently introduced methods of deep learning reconstruction further reduce radiation dose while maintaining image quality.15,16
Average radiation dose for CCTA will vary depending on patient body habitus and heart rate at the time of image acquisition and CT scanner hardware and scanning parameters. However, sub-millisievert scanning is achievable on most CT scanner platforms. The CCTA radiation dose ranges between 3 and 5 mSv and is lower than typical radiation doses for nuclear perfusion scanning or invasive coronary angiography (ICA). 17
Contrast Medium Administration
Excluding coronary Agatston scoring, most cardiac CT examinations require the administration of intravenous contrast. For CCTA, the optimal vascular enhancement is between 250 and 300 HU.9,18 In adult patients, an injection rate of 5 to 7 mL/s using an 18G size needle is recommended in an antecubital vein, preferably on the right to avoid dense contrast in the left innominate vein causing beam-hardening (streak) artifact and obscuring vascular structures such as the aorta.4,9,18 The timing of contrast administration should be determined by using a test injection or bolus tracking technique. Higher contrast injection rates are required in larger patients and pregnant women, due to increased intravascular blood volume. 9
With the use of a dual injector, biphasic (contrast followed by saline) and triphasic (contrast followed by mix of diluted contrast and then saline) protocols are possible. Triphasic injection allows maximum enhancement of the structure of interest and reduced right heart enhancement. Beam-hardening artifacts related to contrast in the right atrium are reduced, while maintaining ability to assess the right heart.
Patient Preparation
CCTA image quality is maximized at low and regular heart rates. Typically, beta-blockers are administered to reduce heart rate to a target level depending on the type of CT scanner (Table 2). Most current CT scanners can acquire satisfactory images at higher heart rates (>80 beats per minute [bpm]), but this is typically accomplished at the expense of using higher radiation doses compared to imaging at lower heart rates (<60 bpm). Dual source CT scanners however, can produce high quality images at relatively higher heart rates due to superior temporal resolution.
Medications Commonly Used for CCTA.
Metoprolol is often used due to wide availability, safety, and low cost; esmolol can be used as an alternative to metoprolol for patients with relative contra-indications to beta blockers and given at dose of 0.8 mg/kg (can be repeated to achieve target heart rate) and has benefits of rapid onset of action and shorter half-life than metoprolol.
Nitroglycerin patch may also be administered 30 to 45 minutes prior to imaging as an alternative. 23
Sublingual nitroglycerine is often administered as either a tablet or spray 5 minutes prior to the scan acquisition. Nitroglycerine works as a smooth muscle relaxant resulting in dilatation of coronary arteries. This improves visualization, especially of the smaller coronary artery branches.9,19,20
Acquisition Protocols
Cardiac CT scan modes are reviewed in the radiation safety section above. Whenever feasible, the lowest radiation mode available should be used. Retrospective ECG-gating should only be used when functional data is required, or the patient’s heart rate cannot be reduced sufficiently (Table 1).
Z-axis coverage should be limited to the minimum clinically required. For coronary imaging, scanning from the carina to just beyond the bottom of the heart is generally sufficient. If there is a history of coronary artery bypass surgery, the coverage should begin at the lung apices to include the entire course of internal mammary grafts.
Images should be acquired with the thinnest slice reconstruction possible. For coronary imaging, the reconstructed field of view should be limited to the heart to maximize the spatial resolution for coronary assessment. If desired, larger reconstructed field of view with thicker slices may be obtained for assessment of incidental findings.
If possible, images should be reviewed by a cardiac imager for diagnostic quality immediately after the scan. If a diagnostic quality study has not been obtained, the examination can be repeated.
Post-Processing
In addition to the axial data set, more advanced techniques may be required to assess cardiac anatomy. These include multiplanar reformats, curved multiplanar reformats, volume-rendered three-dimensional images, and cinematic rendering. For coronary arteries, curved multiplanar reformats are particularly useful. Reformatted images should always be correlated with the original axial data set to ensure that the anatomy is correctly displayed.
If functional information is obtained, then specialized software can be used to navigate the four-dimensional data set and generate dynamic multiplanar or dynamic volume-rendered images. Segmentation of the ventricular cavities can be used to quantify ventricular volumes and systolic function. Artificial intelligence (AI) has been progressively integrated into routine image processing of cardiac CT studies and has been used for automated segmentation of ventricular volumes, atherosclerotic plaque characterization, and epicardial coronary artery segmentation and analysis, as well as epicardial fat volume assessment.24-29 Use of AI is rapidly evolving, as are consensus discussions on standards of practice and how AI should be used in routine clinical practice. As such, we have currently refrained from issuing a statement on AI use for cardiac CT until more is known about standards and incremental benefits to workflow, accuracy and prognostic value of information provided.
Reporting Standards
The CCTA report should contain the relevant imaging findings, their interpretation, an overall summary/conclusion, and recommendations for further management, if appropriate. The components of a comprehensive report have previously been described in the literature.30-32
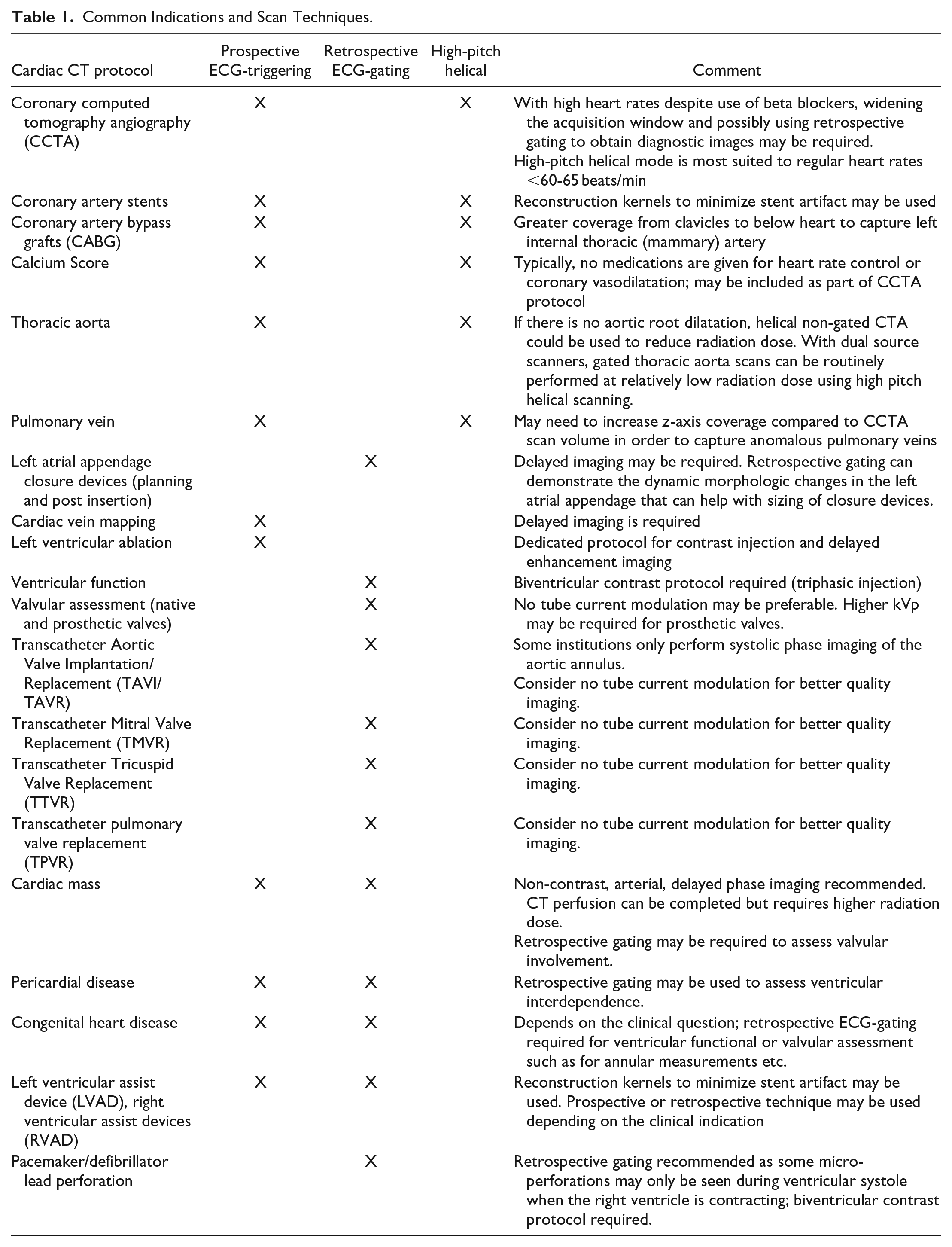
For CCTA, the Coronary Artery Disease-Reporting and Data System (CAD-RADS 2.0) was created to standardize reporting and interpretation (Figure 1). 33 It includes management recommendations based on the most severe coronary plaque stenosis and the overall burden of coronary atherosclerosis. 33 A sample structured report is included in Appendix A.
CAD-RADS 2.0 categories (A) CAD-RADS 0. No plaque or stenosis. (B) CAD-RADS 1/P1. Minimal stenosis (1%-24%). Mild plaque burden. (C) CAD-RADS 2/P2/HRP. Mild stenosis (25%-49%). Moderate plaque burden. High-risk plaque in the proximal LAD suggested by positive remodelling and spotty calcification. (D) CAD-RADS 3/P3. Moderate stenosis (50%-69%). Severe plaque burden. (E) CAD-RADS 4A/P3. Severe stenosis (70%-99%) without left main ≥50% or 3-vessel obstructive (≥70%) disease. Severe plaque burden. (F) CAD-RADS 5/P4. Total occlusion. Extensive plaque burden. Most severe stenosis depicted by arrows in B-F.
Cardiac CT Consensus Recommendations
Coronary CTA
Calcium Score
Coronary artery calcium CT informs atherosclerotic cardiovascular disease (ASCVD) risk stratification and guides preventive measures in asymptomatic individuals. 34 Coronary artery calcium (CAC) is an independent predictor of cardiovascular risk and multiple studies that incorporate CAC in risk models result in improved prediction of ASCVD events.35,36 A calcium score of 0 has high negative predictive value for ASCVD events.37-39
The primary indication for coronary artery calcium CT is to assess ASCVD risk in asymptomatic, intermediate-risk individuals in whom the presence or absence of CAC would result in a reclassification into low or high-risk groups. The Canadian Cardiovascular Society Guidelines provide a strong recommendation to consider coronary artery calcium CT for asymptomatic adults 40 years of age or older who are at intermediate risk (Framingham Risk Score 10%-20%) and for whom treatment decisions are uncertain. 34 The guideline also provides a strong recommendation that coronary artery calcium CT is not indicated for high-risk individuals, patients already receiving statin treatment, or most asymptomatic, low-risk adults. The guideline provides a weak recommendation for CAC screening in a subset of low-risk individuals 40 years of age or older with a family history of premature ASCVD (specifically, men 55 years or younger and women 65 years or younger). 34
The Agatston score is the best validated method for quantifying CAC. 40 Each calcified lesion with a peak intensity of 130 HU or more is assigned a factor from 1 to 4 based on its peak intensity (1 for 130-199 HU, 2 for 200-299 HU, 3 for 300-399 HU, and 4 for 400 HU or more). This factor is then multiplied by the area of the lesion to obtain the lesion score. The total Agatston score is calculated by summing the scores of all individual lesions. CAC is commonly categorized as no CAC (Agatston score 0), mild (1-100), moderate (101-400), or severe (>400). 40 In addition to reporting the Agatston score, the age, sex, and race-specific CAC percentile, the number of vessels with CAC, and the presence of CAC in the left main coronary artery, is also recommended.
The Coronary Artery Calcium Data and Reporting System (CAC-DRS), proposed by the Society of Cardiovascular Computed Tomography, aims to standardize the reporting and communication of CAC scan results and guide patient management. 41 CAC-DRS is a structured reporting system that classifies patients into 4 categories—CAC-DRS 0, 1, 2, and 3—based on the Agatston score. Each category reflects an increasing burden of coronary calcification and escalating risk of future ASCVD events. CAC-DRS also includes modifiers indicating the number of vessels with CAC and whether scoring was based on qualitative visual estimation or quantitative Agatston score, the former to be used only for non-contrast, non-gated CT (Figure 2).
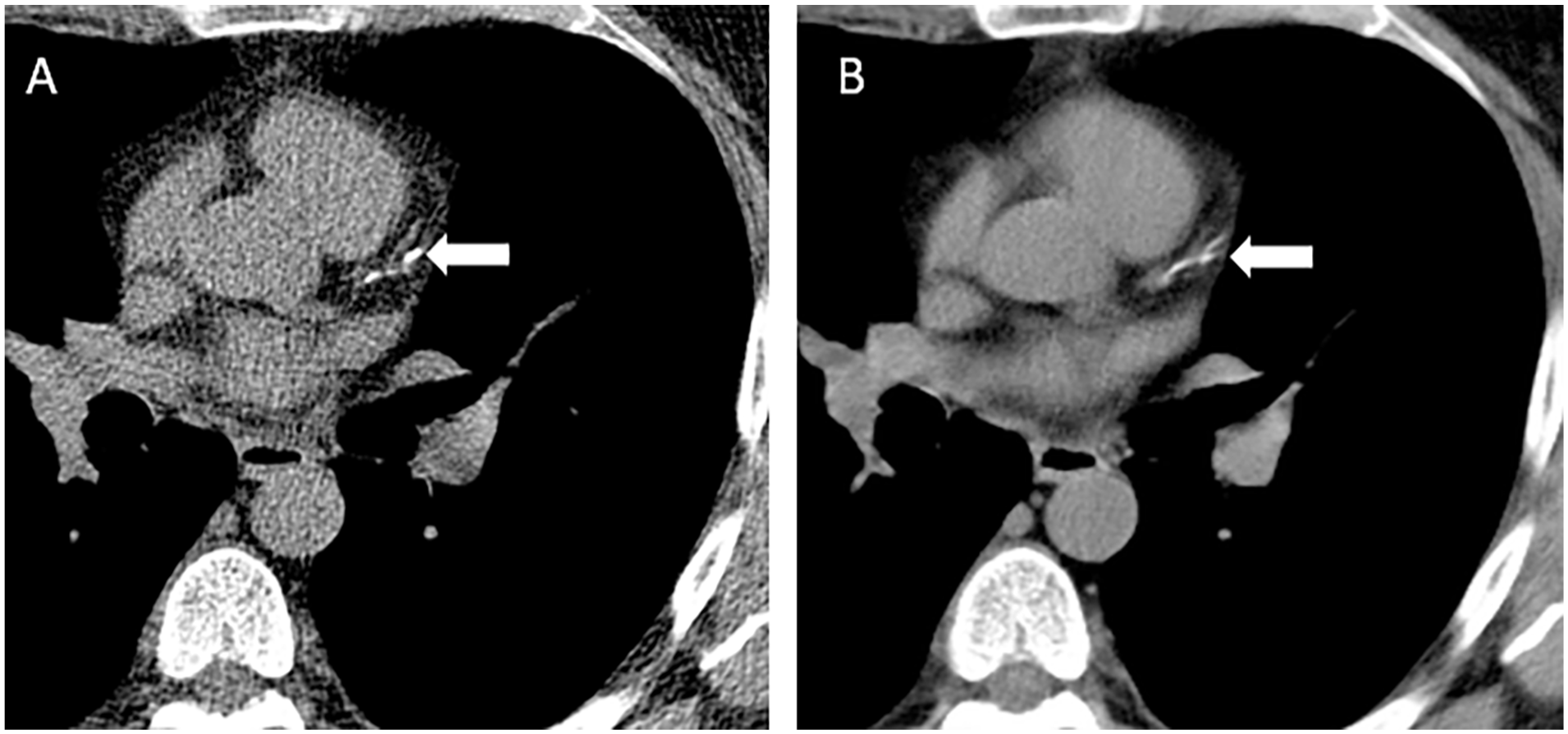
(A) Calcium score CT was <100 in a 62-year-old asymptomatic male who was intermediate risk by Framingham risk scoring. All the calcium was found within the proximal left anterior descending coronary artery (arrow). It is not necessary to repeat a calcium score CT after an initial non-gated chest CT is completed, as shown in (B), as multiple studies have shown excellent correlation between non-gated chest CT qualitative scoring (mild, shown in B, arrow) and quantitative Agatston score.
Lipid-lowering therapies may increase Agatston scores despite regression of non-calcific plaques. Serial CAC scans are not recommended, 34 but in patients with a CAC score of 0 on the index scan, repeat CAC testing can be considered in 5 to 7 years for low-risk individuals, 3 to 5 years for intermediate-risk individuals, and in approximately 3 years for high-risk individuals or for patients with diabetes only if a reassessment would impact clinical management. 42
Native Coronary Arteries
CCTA has evolved as an effective, non-invasive imaging modality for the evaluation of patients with stable typical or atypical chest pain or anginal equivalent, assessment of coronary artery bypass graft patency, and assessment of coronary anomalies. CCTA’s ability to assess coronary artery stenosis and plaque characteristics can guide immediate and long-term treatment strategies based on risk prognostication arising from the likelihood of future cardiovascular events.
A large number of comparative effectiveness studies and meta-analyses have validated the diagnostic accuracy of CCTA for detection of obstructive and non-obstructive CAD compared to invasive coronary angiography (ICA) and, more recently, invasive fractional flow reserve (FFR) as reference standards.43,44 CCTA, stress myocardial perfusion imaging with MRI, and PET each have higher sensitivity than SPECT and stress echocardiography for detecting >50% diameter stenosis (with ICA as the reference standard). They also have higher sensitivity than SPECT and stress echocardiography for detecting lesions with FFR ≤0.80 (with invasive FFR as the reference standard). 44 However, among these modalities, CCTA has the lowest specificity for lesions with FFR ≤0.80. The addition of CT-Fractional Flow Reserve (CT-FFR) and CT perfusion to CCTA anatomic imaging can increase the specificity of CT to that of MRI and PET. 43
Several studies have demonstrated the high sensitivity and negative predictive value of CCTA for acute coronary syndrome (ACS). CCTA is now recognized as a first-line imaging test for patients with acute chest pain and low-to-intermediate pretest probability for ACS. 45 Other guidelines do not recommend routine imaging investigations for low risk patients presenting to emergency departments with ACS symptoms. 20 However, subsequent outpatient testing with calcium score CT can be considered in select patient populations for risk prognostication. Randomized controlled trials have demonstrated that the use of CCTA can reduce time to diagnosis in acute chest pain, decrease length of hospital stay, and reduce costs by minimizing the need for further downstream testing.46,47 In patients with a high pretest probability for ACS, CCTA may also be a reasonable diagnostic strategy compared to using early ICA due to high bleeding risk or patient preference. CCTA can add increased specificity to high sensitivity troponin assays, that can have false positives or borderline results. 48
CAD-RADS provides a structured framework to interpret and report CCTA findings and aims to improve communication between cardiac imagers and referring physicians, standardize reporting practices, provide a framework for patient management decisions, and facilitate research in CAD. CAD-RADS categorizes CAD based on the highest grade coronary artery stenosis identified by CCTA (based on a reduction in luminal diameter as seen on multiplanar reformats),49-51 with 6 categories: CAD-RADS 0 (absence of CAD; no coronary stenosis), CAD-RADS 1 (minimal non-obstructive CAD; 1%-24% maximal coronary stenosis), CAD-RADS 2 (mild non-obstructive CAD; maximal coronary stenosis 25%-49%), CAD-RADS 3 (moderate stenosis; maximal coronary stenosis 50%-69%), CAD-RADS 4 (severe stenosis; 70%-99% stenosis or left main ≥50% or 3-vessel obstructive [≥70%] disease), and CAD-RADS 5 (total coronary occlusion; 100% stenosis) (Figure 1A-E).
CAD-RADS 2.0, published in 2022, introduces additional parameters including plaque burden and ischemia. 33 Like the original version, CAD-RADS 2.0 maintains coronary artery luminal stenosis as an estimate of reduction in luminal diameter, defined on a per-patient basis, as the central component of assessment upon which the numeric category is based. However, it goes a step further by introducing methods to estimate, quantify, and report overall plaque burden, aligning with growing evidence that plaque characteristics and burden play an important role in cardiovascular risk stratification. The update incorporates new P1 to P4 descriptors, denoting increasing categories of plaque burden. The modifier HRP (high risk plaque, previously V to indicate vulnerable plaque) should be used when 2 or more high risk features are present, including spotty calcifications, low attenuation plaque (less than 30 Hounsfield Units), positive remodelling, and the napkin ring sign (defined as low attenuation plaque representing a “necrotic core” that is surrounded by a rim of higher attenuation representing fibroatheroma or the thin fibrous cap that is prone to rupture). CAD-RADS 2.0 also includes an I modifier (ischemia) to indicate whether CT-FFR or myocardial CT perfusion demonstrates lesion-specific ischemia or a reversible perfusion defect, and an E modifier (exceptions) to indicate any non-atherosclerotic narrowing of the coronary arteries such as spontaneous coronary artery dissection, vasculitis or extrinsic compression. 33 Other modifiers include S (presence of a coronary artery stent), G (presence of a coronary artery bypass graft), and N (non-diagnostic study). CAD-RADS 2.0 also provides post-test recommendations based on the CAD-RADS classification, such as whether further diagnostic testing, therapeutic changes, or cardiology consultation is advised. 33
Coronary Artery Stents
Coronary artery stent implantation is a standard therapeutic strategy for CAD. Although efficacious, stents are subject to complications such as in-stent re-stenosis and stent thrombosis. The assessment of coronary stents using CCTA was historically fraught with challenges, primarily due to the metallic stent causing beam-hardening and partial volume averaging (blooming artifacts) that limit visualization of the stent lumen. Stent size, stent composition, and the patient’s heart rate during imaging substantially influence CCTA’s performance for stent evaluation. As such, functional ischemic testing has generally been preferred over CTA in the assessment of symptomatic patients with coronary stents. However, advancements in CT technology, including increased detector rows and iterative reconstruction algorithms, have substantially improved CCTA’s ability to evaluate stent patency. As a result, CCTA is a reasonable test for evaluating patients with coronary stents >3 mm, particularly with current generation drug-eluting stents that have struts <100 µm. 52 Stents with smaller diameters (<3 mm) are more challenging to assess, but CCTA may still be a reasonable test for assessing proximal, non-bifurcation thin strut stents that are <3 mm. 53
Coronary Artery Bypass Grafts
Coronary artery bypass grafting (CABG) remains a cornerstone in the treatment of multivessel and complex CAD. CTA offers a non-invasive and reliable method to assess the patency of coronary artery bypass grafts, including both arterial and venous grafts. CTA can accurately depict graft patency, anastomotic sites, and stenosis, and can help in presurgical planning of redo sternotomies to assess left internal mammary artery graft location and retrosternal adhesions. While CTA has excellent diagnostic accuracy for assessing graft stenosis and patency, assessment of the native coronary arteries is more limited in patients with prior CABG, often due to severe coronary artery calcification, 54 and a functional test may be most appropriate to identify protected and unprotected territories in these patients.
Advanced Techniques
Computed Tomography-Fractional Flow Reserve (CT-FFR)
FFR is the gold standard for identifying hemodynamically significant CAD and guiding revascularization strategies. 55 Traditionally, FFR measurement has been an invasive procedure, requiring cardiac catheterization and specialized pressure wires to determine hemodynamically important reductions in blood flow within the coronary arteries. Technological advancements have facilitated the calculation of FFR from standard CCTA, termed CT-FFR paving the way for non-invasive functional evaluation of CAD. 56
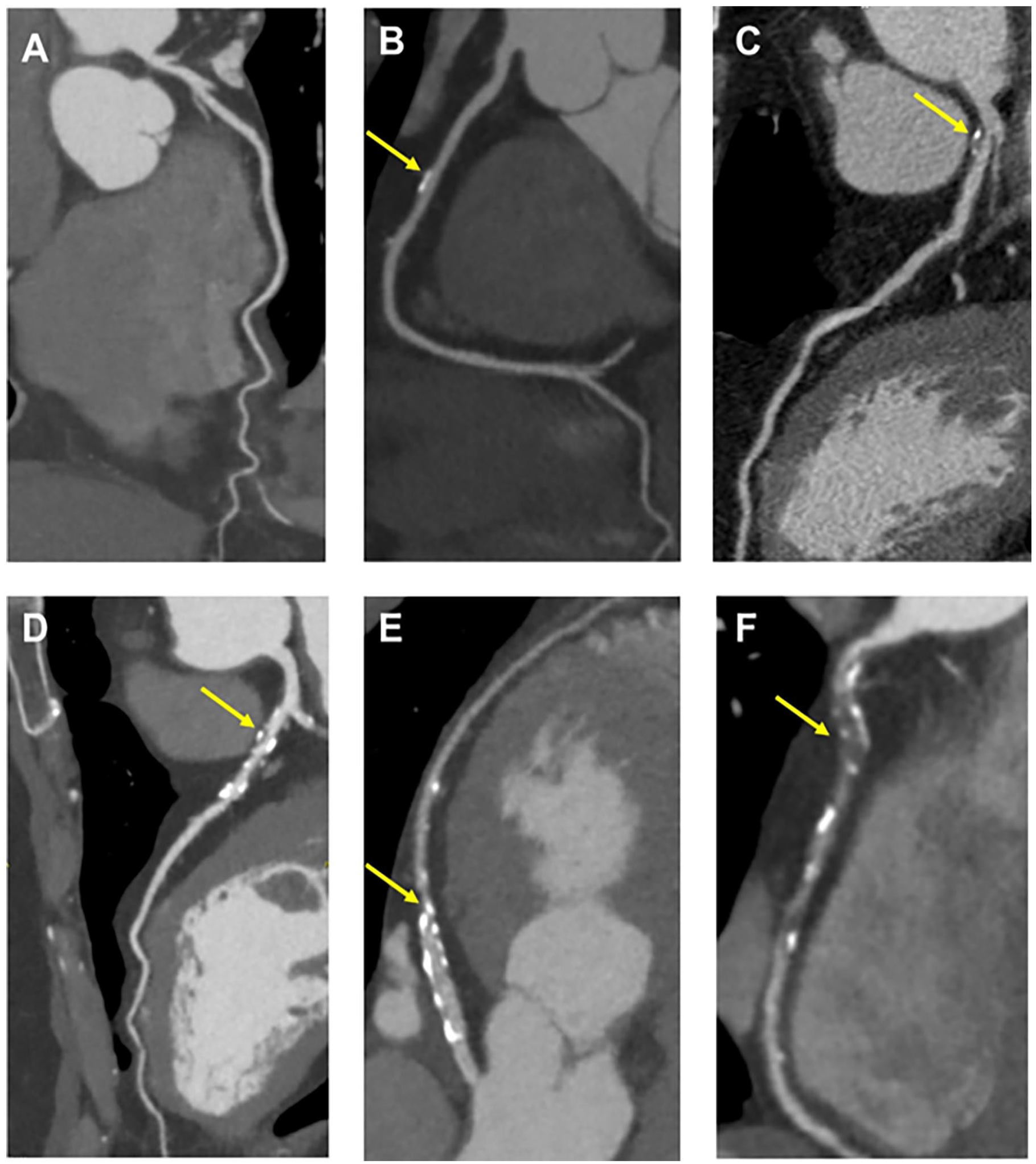
CT-FFR is based on computational fluid dynamics to simulate blood flow and pressure in the coronary arteries from anatomical data derived from CCTA based on vessel attenuation. 56 This computational approach estimates the pressure drop across a stenosis under simulated hyperemic conditions, providing a ratio indicative of lesion-specific ischemia. CT-FFR values >0.80, mirroring invasive FFR, are considered normal (Figure 3), while values between 0.76 and 0.8 are borderline and values ≤0.75 are indicative of a hemodynamically significant lesion (Figure 4). 57
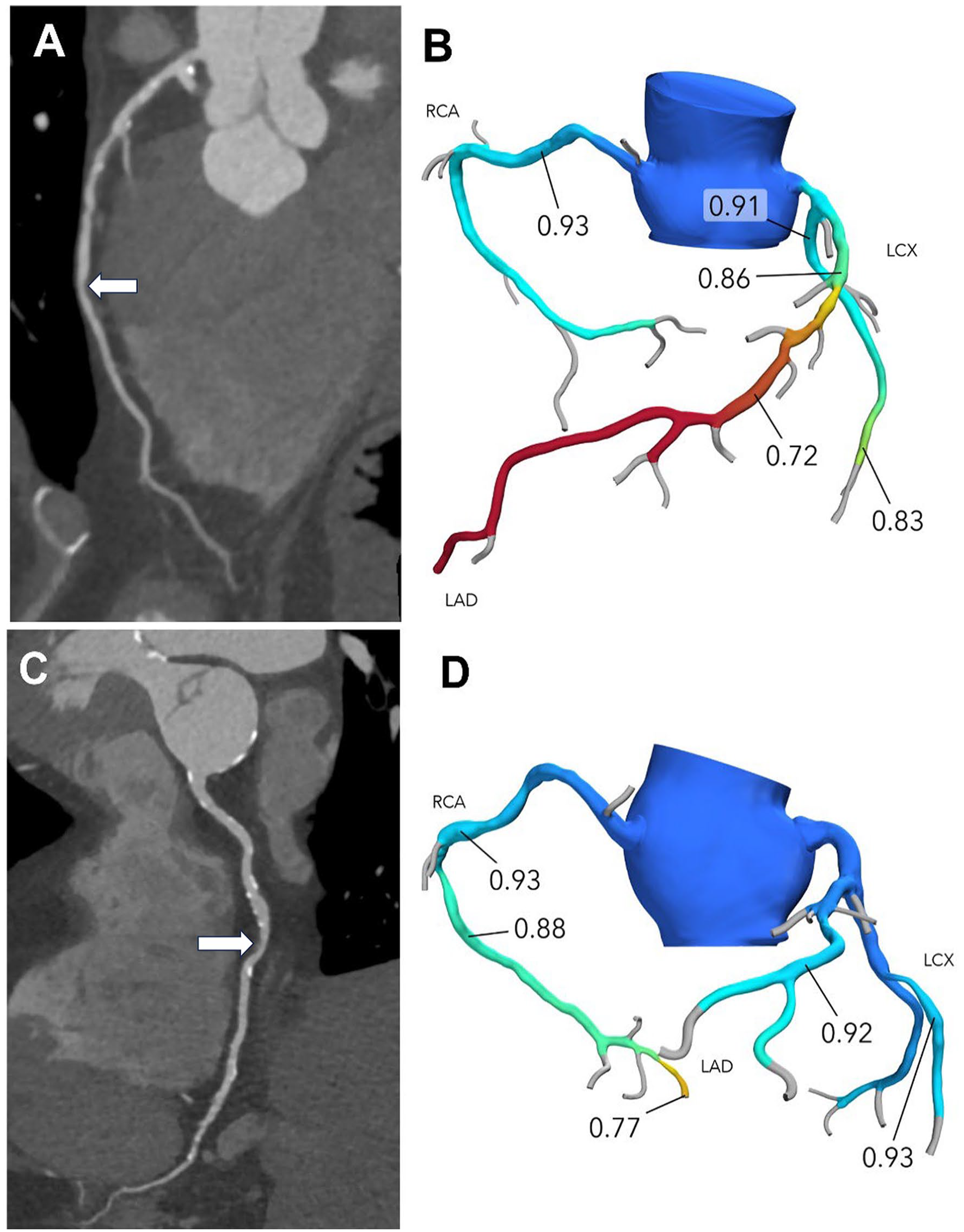
(A) Moderate stenosis of the mid LAD (CAD-RADS 3) with (B) a CT-FFR value of 0.72, corresponding to a hemodynamically significant lesion (arrow). (C) Moderate stenosis of the mid RCA (CAD-RADS 3) with (D) a CT-FFR value of 0.88, suggesting that the lesion is not hemodynamically significant (arrow). Images courtesy of Brian Pogatchnik, MD, Stanford University.
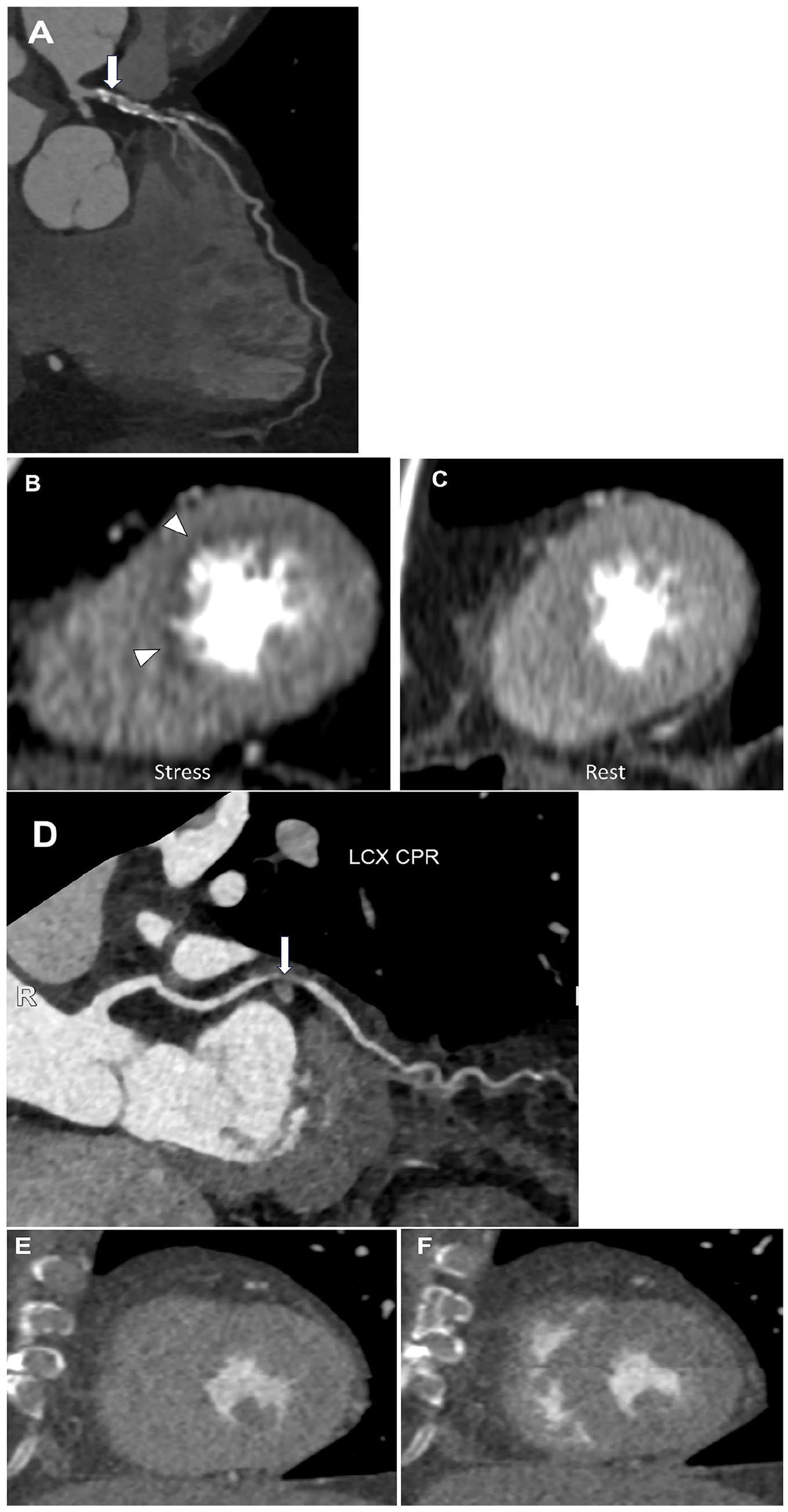
(A) Severe stenosis of the proximal LAD (CAD-RADS 4A, arrow), with (B) a reversible perfusion defect in the LAD territory seen on stress imaging, (C) but not on rest imaging, corresponding to myocardial ischemia. (D) Severe stenosis of the mid LCx (CAD-RADS 4A, arrow) with (E) no perfusion defect seen on stress imaging or (F) rest imaging. Images courtesy of Brian Pogatchnik, MD, Stanford University.
Multiple studies have demonstrated the efficacy of CT-FFR in the assessment of CAD and the ability for CT-FFR to increase the specificity of CCTA.58,59 Initial work has been dedicated to determining the role of CT-FFR in reducing the proportion of patients referred for ICA with anatomical stenoses on CCTA that are not hemodynamically significant. This reduces the number of patients referred for ICA with non-hemodynamically significant lesions and increases the number of patients appropriately referred for ICA for percutaneous coronary intervention (PCI) of obstructive CAD. The ADVANCE registry demonstrated that the addition of CT-FFR resulted in a change in management strategy in 66.9% of patients. 60 Improved specificity by CT-FFR may be particularly important as CCTA is increasingly used for patients with higher pre-test probabilities and higher calcium burden that limits accurate anatomic assessment of stenosis. In addition to its role in reducing ICA utilization, there is an increasing role for CT-FFR to guide revascularization strategies to ensure full revascularization in complex CAD. 61
CT-FFR has not yet been validated for ACS, recent myocardial infarction (within 30 days), coronary artery bypass grafts, coronary anomalies, coronary dissection, or transcatheter aortic valve replacement. 62 In addition, CT-FFR has not been validated in the presence of coronary stents, but can still be performed in other coronary arteries if there are no stents in the left main coronary artery or in 2 or more systems. 62
Despite its demonstrated utility, CT-FFR is not without challenges. It requires high-quality CTA images and significant computational resources, often leading to off-site analyses, increased cost of testing and consequently time delays. CT-FFR is currently not widely used in Canada due to lack of availability and reimbursement by provincial health care plans.
Myocardial Perfusion and Viability
Myocardial computed tomography perfusion (CTP) imaging offers insight into myocardial blood supply complementing the anatomical data provided by traditional CCTA. The primary role of myocardial CTP is to evaluate the presence and extent of myocardial ischemia by visualizing myocardial distribution of iodinated contrast material at rest and after pharmacologically induced stress.62,63 It is based on the principle that areas of myocardium supplied by stenosed coronary arteries will exhibit a relative decrease in contrast enhancement, particularly during stress conditions, compared to areas supplied by non-diseased arteries. By integrating functional information about myocardial perfusion with anatomical data on CCTA, myocardial CTP can enhance diagnostic accuracy and prognostic evaluation compared to CCTA alone (Figure 4). This is specific to a select patient population in which CTA demonstrates coronary artery stenosis of unknown hemodynamic significance, severe coronary artery calcification, or coronary stents. Despite the capabilities of myocardial CTP, it is also important to consider the associated challenges, including the need for pharmacological stress agents and additional radiation exposure, making careful patient selection critical.
Although MRI remains the predominant technique for assessing myocardial viability in patients with previous myocardial infarction, an additional CT acquisition 5 to 10 minutes after vascular contrast washout can assess myocardial scar. 64 While CT has a lower contrast to noise ratio compared to MRI, late enhancement CT imaging may be used as an alternative modality to evaluate myocardial viability in patients with MRI contraindications.
Mapping and ECV
Extracellular volume fraction (ECV) is an important imaging parameter to estimate expansion of the myocardial extracellular space secondary to myocardial fibrosis or other infiltrative myocardial diseases. 65 While ECV measurements originated from MRI, recent advancements have enabled these measurements using cardiac CT. CT-based ECV measurements involve the acquisition of pre- and post-contrast images (or post-contrast images and virtual unenhanced images if acquired using dual-energy CT), allowing for the calculation of hematocrit-corrected ECV. The quantification of ECV using cardiac CT correlates well with MRI-derived ECV measurements 66 and histologic findings. 67 While preliminary studies have shown promising results, further research is needed to validate the clinical utility and prognostic significance of CT-derived ECV measurements in different patient populations.
Conclusion
These practice guidelines have been revised to reflect technological advancements and changes in the use of cardiac CT since the first iteration published in 2009. As CT technology is constantly evolving and the use of AI will undoubtedly expand in future, these guidelines will undergo future revisions to include ongoing CT scanner innovations that shape and enhance the use of cardiac CT in clinical practice.
Footnotes
Appendix
Sample structured report for coronary CTA. The structure may require modification depending on coronary anatomy, the presence of bypass grafts, etc. The CAD-RADS score may be included but should not replace the conclusion.

| CTA CORONARY STUDY INDICATION: [ ] COMPARISON: [ ] TECHNIQUE: Coronary CTA performed with [description of ECG gating, eg, prospective ECG gating] MEDICATION: [ ] QUALITY: [ ] FINDINGS: Normal coronary origins. Coronary Dominance: [ ] Left main: [ ] Left anterior descending and branches: [ ] Left circumflex and branches: [ ] Right coronary and branches: [ ] Incidental findings: [ ] Conclusion: [Summary including most severe stenosis, extent of plaque, recommendations, significant incidental findings if any.] |
Acknowledgements
The authors would like to thank the members of the Canadian Association of Radiologists and the Canadian Association of Radiologists who took the time to provide their feedback and peer review during the drafting of these guidelines.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Carole Dennie reported receiving payments for consultant fees from AstraZeneca and speaker honorarium from Boehringer-Ingelheim. Dr. Kate Hanneman reported receiving honorarium from Sanofi. No other authors declared potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
