Abstract
Gastrointestinal stromal tumours (GISTs) are defined as mesenchymal tumours of the gastrointestinal tract that express positivity for CD117, which is a c-KIT proto-oncogene antigen. Expression of the c-KIT protein, a tyrosine kinase growth factor receptor, allows the distinction between GISTs and other mesenchymal tumours such as leiomyoma, leiomyosarcoma, schwannoma and neurofibroma. GISTs can develop anywhere in the gastrointestinal tract, as well as in the mesentery and omentum. Over the years, the management of GISTs has improved due to a better knowledge of their behaviors and risk or recurrence, the identification of specific mutations and the use of targeted therapies. This has resulted in a better prognosis for patients with GISTs. In parallel, imaging of GISTs has been revolutionized by tremendous progress in the field of detection, characterization, survival prediction and monitoring during therapy. Recently, a particular attention has been given to radiomics for the characterization of GISTs using analysis of quantitative imaging features. In addition, radiomics has currently many applications that are developed in conjunction with artificial intelligence with the aim of better characterizing GISTs and providing a more precise assessment of tumour burden. This article sums up recent advances in computed tomography and magnetic resonance imaging of GISTs in the field of image/data acquisition, tumour detection, tumour characterization, treatment response evaluation, and preoperative planning.
Introduction
Gastrointestinal stromal tumours (GISTs) are the most common mesenchymal tumours of the gastrointestinal tract. Although they can develop anywhere in the gastrointestinal tract, the stomach is the most frequently affected organ (50-60% of GISTs) followed by the small bowel (20-30%) and the duodenum (10%). 1-3 GISTs most frequently arise from the muscularis propria of a given gastrointestinal organ but frequently involve the outer muscular layer, resulting in an exophytic growth. 3 However, GISTs can be submucosal, subserosal or intraluminal, thus explaining great variations in imaging presentation. 3 Clinically, GISTs are frequently revealed by acute gastrointestinal bleeding due to mucosal ulceration, anemia caused by chronic bleeding, spontaneous rupture, or intussusception. 4,5
Another specific characteristic of GISTs is the presence of gene mutations. 6,7 Eighty to 85% of GISTs harbor activating mutations in the KIT protooncogene and 5-10% harbor mutations in platelet-derived growth factor receptor alpha (PDGFRA) genes, with the 2 mutations being mutually exclusive. 6,8 Wild-type GISTs represent a small proportion of GISTs with no KIT or PDGFRA mutations. 1 In addition, the affected exon responsible for KIT mutation has a major impact on tumour response to therapy, imatinib in particular. 8 For these reasons, molecular evaluation of GIST is now crucial to select the most appropriate therapy. 8
Since the introduction of helical technology, computed tomography (CT) is the most used imaging technique for the assessment of GISTs. 2,3,9,10 CT provides crucial information that can suggest the diagnosis of GIST and be used for the follow-up of patients with GIST. 3,4,10 Magnetic resonance imaging (MRI) may contribute additional information in selected patients. 11 However, considering the major advances in knowledge of the behavior of GIST, the concept of risk of recurrence, the identification of specific mutations and the use of targeted therapies, more demand has been placed on imaging. In general, CT and MRI supply information such as size, contour patterns, attenuation value or signal intensity, homogeneity and degree of enhancement. 2,4,10 Although these features may help predict high risk GIST, 12 they have limited capabilities to provide crucial data on gene mutation and response to therapy. 13 To overcome these limitations, the capabilities of radiomics have been evaluated in recent studies with encouraging results that have led to the development of radiomics models to predict risk of recurrence, gene mutation, prognosis, and response to targeted therapy. 12 In addition, radiomics has currently many applications that are developed in conjunction with deep learning with the aim of better characterizing GISTs and providing more precise assessment of tumour aggressiveness. 14,15
The objective of this review was to sum up recent advances in CT and MRI of GIST in the field of image/data acquisition, tumour detection, tumour characterization, treatment response evaluation and preoperative planning.
Image and Data Acquisition
Regarding image acquisition, the most remarkable advances in imaging of GISTs relate to the use of dual-energy CT. Dual-energy CT is used to calculate iodine concentration in a given tissue and has several advantages over single-energy CT.
16,17
In this regard, dual-energy CT conveys greater stability of attenuation measurements and iodine quantification, which are important issues for identifying mild enhancement in GISTs. The benefit of dual-energy CT has been assessed in patients with GISTs.
18-20
Schramm et al. reported that dual-energy CT (80-140 kV) allowed to discriminate between stable or progressive disease in patients with GISTs receiving tyrosine kinase inhibitor (TKI) using a cut-off value of 15 Hounsfield units (HU) for defining stable, increased, or decreased lesion iodine uptake.
18
Meyer et al. found that dual-energy CT findings surpassed RECIST 1.1 and mChoi criteria for the assessment of treatment response to TKI.
19
Another study found that noise-optimized virtual monoenergetic images obtained at low keV (40-50-60 keV) provided best image quality, best tumour delineation, and lower image noise (Figure 1).
20
Finally, dual-energy CT allows to differentiate between high-risk and non-high-risk GISTs due to greater CT attenuation values at 70 KeV in high-risk GISTs during arterial and portal phases of enhancement (85.03 ± 14.46 [standard deviation (SD)] HU and 88.41 ± 14.66 [SD] HU for arterial and portal phases, respectively) compared to intermediate (62.37 ± 10.74 [SD] HU and 63.90 ± 10.79 [SD] HU for arterial and portal phases, respectively) and low-risk GISTs (65.10 ± 22.43 [SD] HU and 66.22 ± 16.92 [SD] HU for arterial and portal phases, respectively) (P < .001).
21
45-year-old man with a gastric gastrointestinal tumour (GIST) who underwent dual-energy computed tomography (CT) examination. Virtual unenhanced CT image in the axial plane reveals no gastric tumour. Multi-energy CT image in the axial plane obtained after intravenous administration of iodinated material shows enhancing gastric tumour (arrow). Iodine only image allows calculating iodine uptake by the gastric tumour (arrow). Mono-energetic CT image in the axial plane obtained during the portal venous phase of enhancement at low voltage (55 KeV) shows better contrast and better tumour conspicuity (arrow) by comparison with (B).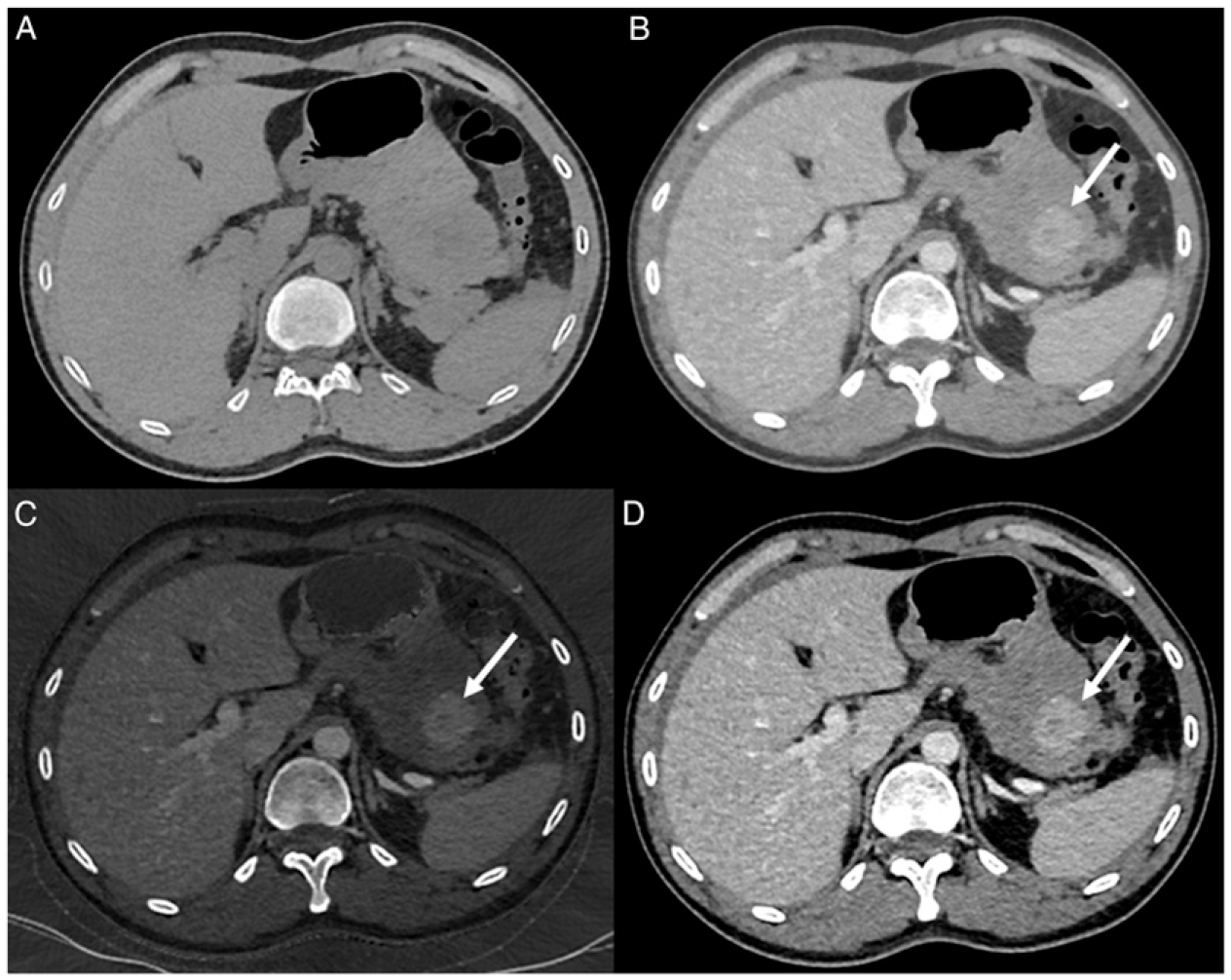
Studies have reported that the use of iterative reconstruction algorithms in conjunction with dual-energy CT results in greater quality of abdominal images. 22,23 However, further research is needed to analyze the benefit of iterative reconstruction in the specific field of GIST.
One recent advance in MRI of the abdomen is the implementation of deep learning reconstruction. 24,25 In general, deep learning image reconstruction results in better image quality, greater signal-to-noise ratio, and better sharpness of abdominal organs. However, to date, no studies have specifically investigated the potential and added value of deep learning reconstruction in the elective field of MRI of GIST. One study revealed improved detection of GISTs when adding diffusion-weighted imaging (DWI) to conventional imaging for MR-enterography and a better interobserver agreement for image interpretation. 26 In this study, the use of DWI allowed the detection of 3 additional GISTs by comparison with standard MR-enterography, yielding a sensitivity similar to that of gadolinium-chelate enhanced sequences. 26
Multiple studies have investigated the capabilities of radiomics for the assessment of GIST.
12
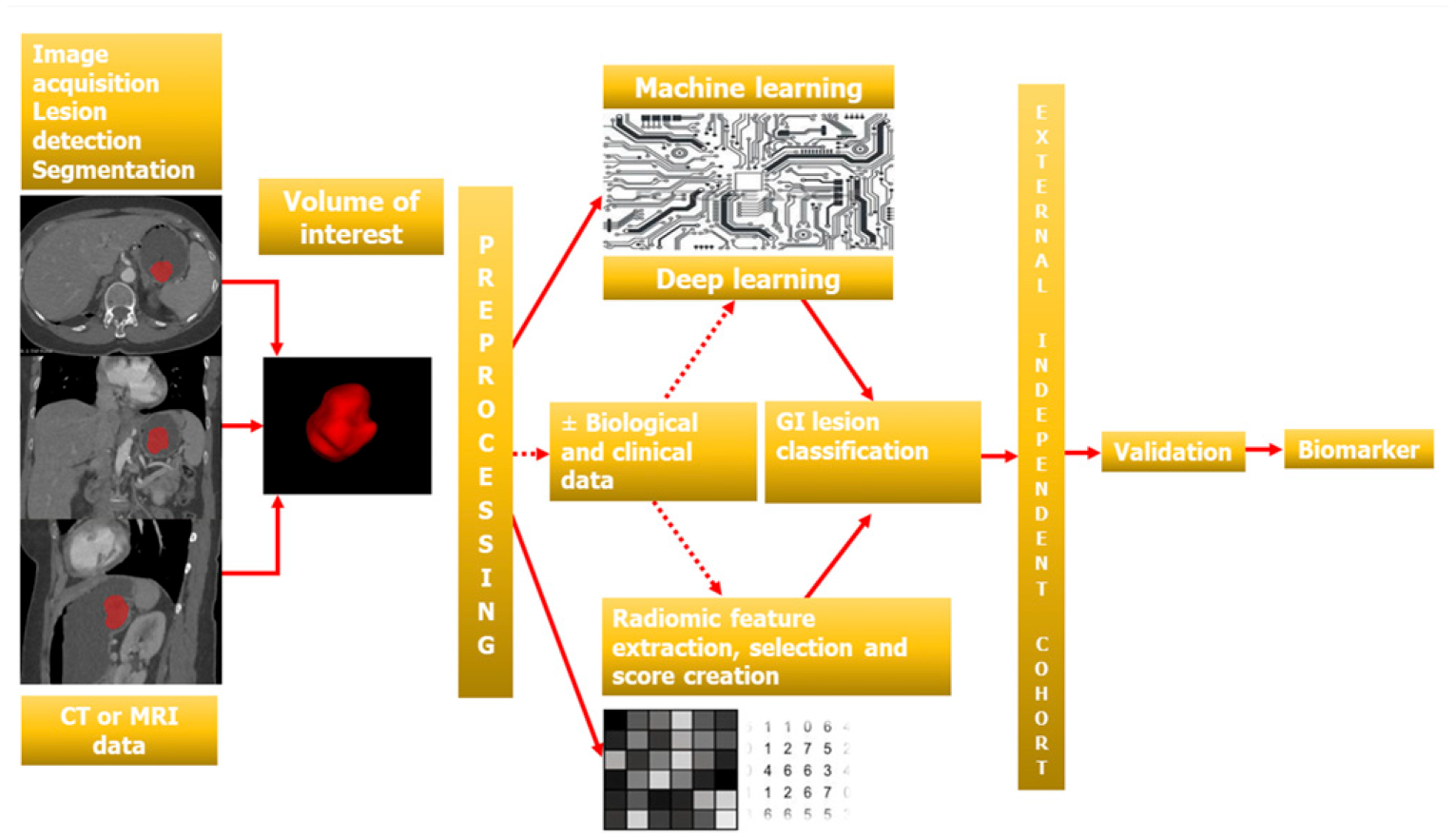
Similar to other conditions, radiomics of GIST requires robust methods for tumour segmentation and further radiomics feature extraction and analysis (Figures 2 and 3). Ren et al. reported excellent inter-observer agreement between 2 observers for 16 tumour radiomics feature extraction in a series of 440 patients (intraclass correlation coefficients [ICC]>.85).
27
In addition, Xu et al. found that manual segmentation using a single region of interest (ROI) yields high intra- and inter-reader reproducibility of radiomics features (ICC >.98) than multiple ROIs on the whole tumour.
28
67-year-old woman with gastric polypoid gastrointestinal tumour (GIST) who underwent computed tomography (CT) examination. (A) CT image in the axial plane obtained after intravenous administration of iodinated material shows polypoid tumour (arrow). Figures B, C,D show lesion segmentation using a texture analysis software (3DSlicer) on axial (B), coronal (C) and sagittal (D) images. Figure E shows final threedimensional representation after tumour segmentation for further radiomics analysis. Diagram shows the different steps of the process of building a radiomics or an artificial intelligence model to analyze a gastrointestinal stromal tumour (GIST). In this gastric GIST (same as in Figure 2), computed tomography images obtained during the arterial dominant phase of enhancement were used for the first step. CT indicates computed tomography; MRI indicates magnetic resonance imaging.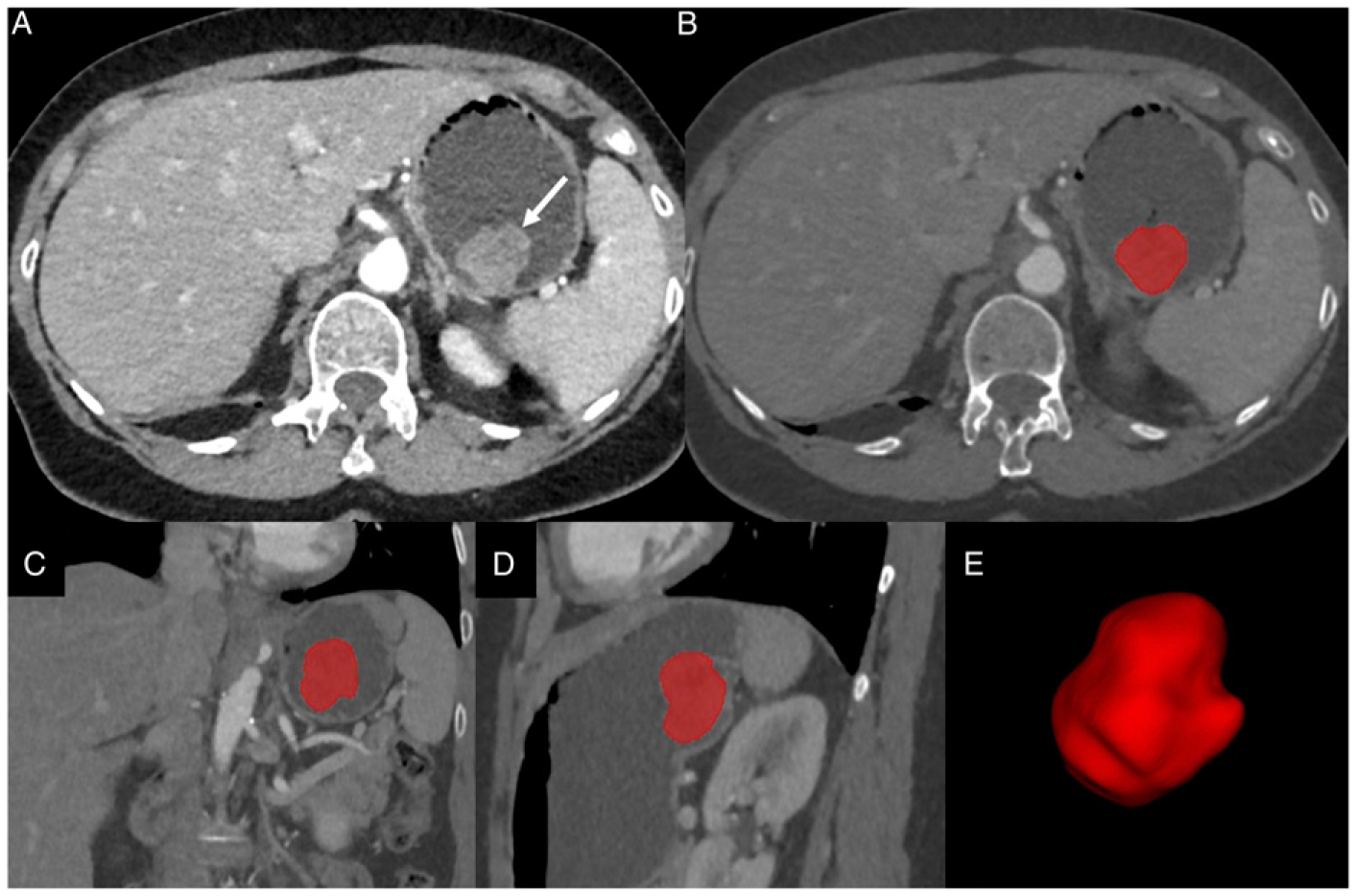
Another advance in the assessment of GIST is the use of cinematic rendering. Cinematic rendering is a three-dimensional display technique that uses a complex global lighting model to provide realistic images.
29
Cinematic rendering has multiple applications in the abdomen
30
and may serve as an important adjunct to standard CT images in the evaluation of gastric and small bowel masses (Figure 4).
29
Cinematic rendering helps demonstrate the submucosal location of GIST, the intramural depth, and its relationship to overlying mucosa, serosa and vessels, which has potential for further surgical or endoscopic treatment planning.
31
66-year-old man with gastrointestinal tumour (GIST) of the duodenal-jejunal junction who underwent computed tomography (CT) examination. (A) CT image in the axial plane obtained during the arterial phase of enhancement shows heterogeneous tumour (arrow) with marked peripheral enhancement. (B) T2-weighted MR image in the axial plane demonstrates hyperintense areas within the tumour (arrow) consistent with internal necrosis. (C) On maximum intensity projection reconstructed CT image in the axial plane, the tumour (arrow) harbors multiple internal vessels. A large draining vessel (arrowhead) is present. (D) Cinematic rendering image shows heterogeneous tumour (arrows) with internal vessels and enlarged draining vessels (arrowhead). Cinematic rendering allows realistic representation of internal texture of the tumour.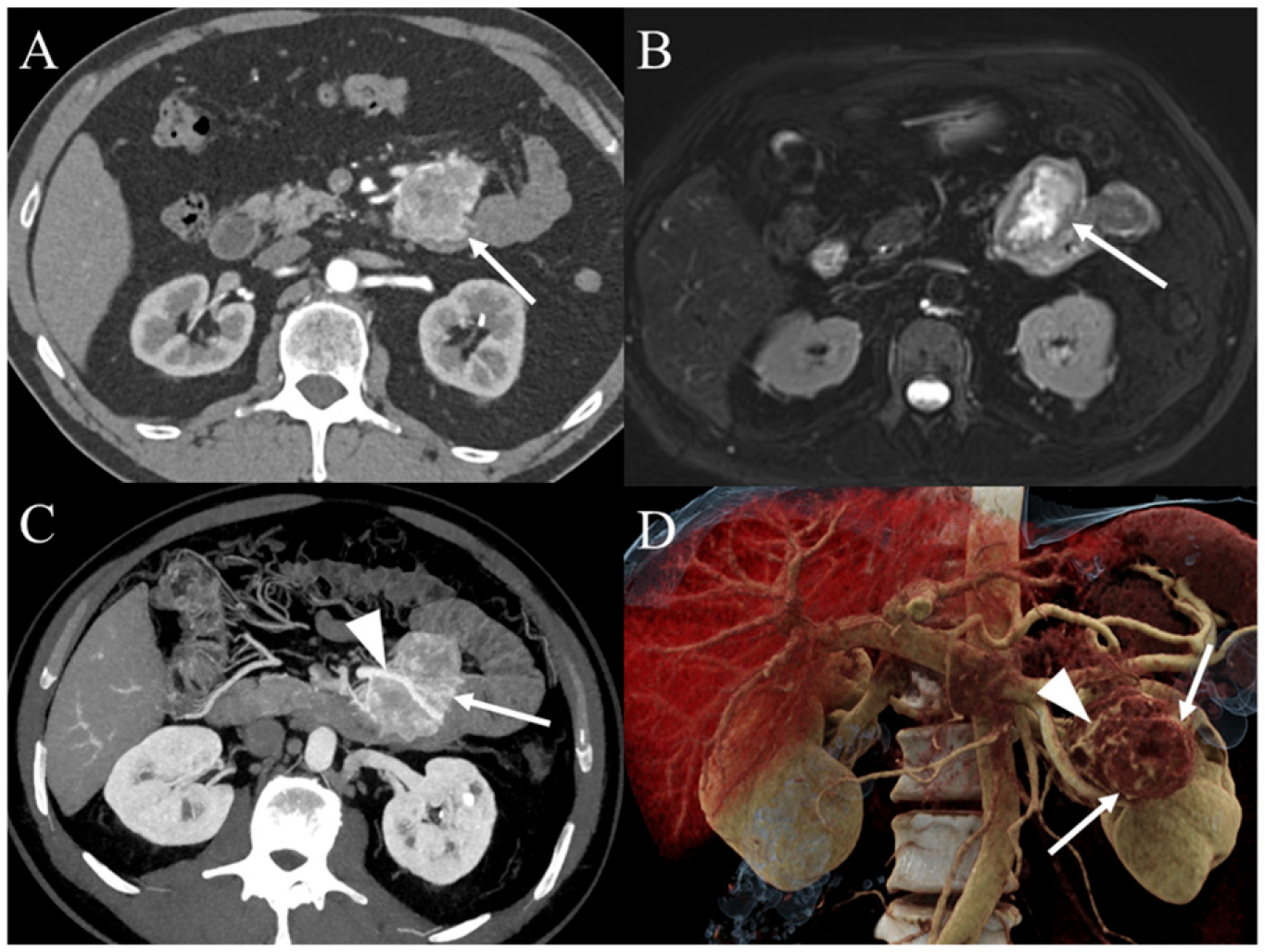
The analysis of recent literature shows a large amount of technical innovations in imaging techniques that result in more refined image and data acquisition. However, some of them require further investigation to fully determine their clinical utility in patients with GISTs.
Lesion Detection
The detection of GISTs depends on tumour location, tumour size and symptoms at presentation. Approximately one third of GISTs are asymptomatic and identified incidentally during endoscopy or imaging examination. 32 Gastrointestinal bleeding is the most common manifestation, with obscure gastrointestinal bleeding being a diagnostic challenge. 33 There are limited data regarding the detection rate of GISTs with CT. One study found that CT has limited capabilities for the detection of small GISTs, with 75% sensitivity for GISTs between 10 and 20 mm and 15% for those ≤10 mm. 34 Although videocapsule endoscopy (VCE) has multiple benefits in the evaluation of small bowel disease, its use for the detection of GISTs is limited. Indeed, GISTs have a submucosal origin and produce little mass effect or displacement so that they are hardly visible by VCE. 35 In this regard, one study found that CT-enterography showed more GISTs than VCE (17/21, 81% vs. 15/21, 71%, respectively), although the difference was not significant. 36 More generally, the performance of VCE for the detection of mesenteric small bowel tumours is low with a false-negative rate of up to 18.9%. 37
CT-enteroclysis, which is another option for the study of the small bowel, has demonstrated high capabilities in identifying GISTs of the small bowel. In a series of 219 patients with 65 small-bowel tumours, none of the 5 GISTs were missed by CT-enteroclysis. 38
MR-enterography, which combines dedicated MRI sequences and small bowel distension, is routinely used in patients with small bowel diseases. However, no studies have specifically focused on its performance for GIST detection. In one study including 7 GISTs in 3 patients, MR-enterography yielded 100% sensitivity on a per-patient basis, although the sensitivity was 43% only on a per-tumour basis. 39 Missed GISTs were all less than 7 mm in size. 39
Although artificial intelligence (AI) has now multiple applications in abdominal imaging, 40,41 limited data in terms of tumour detection is currently available for GIST imaging and most studies refer to the detection of GIST with endoscopic ultrasound. 42
Currently, there is a limited data in the literature, so that GIST detection with imaging remains not fully elucidated. Only one study reports limited sensitivity for GIST detection using CT, which is not the most effective technique in the absence of specific gastrointestinal tract distention. 34 Studies on dedicated imaging techniques such as CT-enterography or MR-enterography are scarce. 38,39 In addition, the application of AI to imaging data of GIST has received little attention to date and only relates to endoscopic ultrasound. 42
Lesion Characterization
GIST characterization with imaging is a critical issue because various conditions may involve the gastrointestinal tract, including benign and malignant lesions, with different management and prognosis. Depending on the specific location, GISTs may mimic other tumours, so that lesion characterization is an important issue. GISTs can be differentiated from other tumours using CT or MRI on the basis of morphologic and dynamic features but also using radiomics. 12 Several CT or MRI features are suggestive for the diagnosis of GIST; of these, extraluminal growth, marked enhancement on arterial phase images, early venous drainage and submucosal origin of the tumour, which is best depicted using CT-enteroclysis, are highly suggestive features. 2,43-45 One study involving 124 patients (64 with GISTs and 60 with benign gastric polyp) who underwent CT examination, found that extraluminal growth (50%; 32/64), irregular margins (83%; 53/64) and tumour ulceration (28%; 18/64) were more frequently observed in gastric GISTs than in benign gastric polyps (10 [6/60]; 33% [20/60]; and 7% [4/60], respectively) (P < .001, <.001 and .002, respectively). 46 Similarly, GISTs showed greater degrees of enhancement during the arterial phase compared to benign gastric polyps. 46 In this study, the best capabilities for tumour discrimination were obtained using semantic features (area under the curve [AUC], .784) rather than using attenuation values on arterial phase images (AUC, .710) or peak attenuation values (AUC, .710). 46 Liu et al found that dual-energy CT helped discriminate between GISTs and gastric schwanommas due to significant difference in iodine concentration between gastric schwanommas and gastric GISTs on both arterial and portal venous phases. 47 Another study involving 50 gastric leiomyomas and 110 gastric GISTs revealed that CT features such as cardia location, endophytic growth and homogeneous gradual enhancement helped differentiate between gastric leiomyomas and gastric GISTs, with these 3 variables being independently and significantly associated with the diagnosis of gastric leiomyoma at logistic regression analysis. 9
One difficulty with GIST is sometimes to ascertain the origin of the tumour. This is especially true for GISTs arising in the periampulla, where other types of pancreatic or duodenal tumour often arise. 48 Lu et al. addressed this issue by using whole-lesion histogram analysis from CT data. 49 These researchers compared the histogram features of 28 duodenal GISTs to those of 26 duodenal adenocarcinomas and 20 pancreatic ductal adenocarcinomas. They found that 90th percentile of arterial phase images yielded best capabilities for discriminating between these 3 entities with AUCs of .809 (duodenal adenocarcinomas vs. GIST) and .936 for (pancreatic ductal adenocarcinomas vs. GIST). 49
Presently, lesion characterization is one of the most prolific areas of research in GIST imaging, with multiple articles showing potential for radiomics in this task.
Gene Mutation Status
Gene mutation status is of paramount importance in GISTs because it has a direct impact on management and therapy. GISTs with KIT protooncogene mutation (representing 80−85% of all GISTs) and those with PDGFRA mutation (representing 5−7% of all GISTs) favorably respond to imatinib mesylate, a selective TKI of the KIT and PDGFRA receptors. 8,12 Another important point is the identification of the exon involved in the mutation. In this regard, approximately 70% of KIT mutations in GISTs affect exon 11, which encodes the intracellular juxtamembrane domain of the receptor. Mutations in exon 9 affecting the extracellular ligand-binding domain are detected in 12-15% of GISTs. 8 This identification is of major importance because GISTs with KIT exon 11 mutation are more responsive to imatinib, while GISTs with KIT exon 9 mutation are more responsive to sunitinib or may require a higher dose of imatinib. 12
Several studies have addressed these important points and identified imaging features that can be used to anticipate the mutational status of GIST. In a series of 88 patients with GISTs, Cannella et al. found that hyperenhancement on CT before treatment was more frequent in PDGFRA-mutated/wild-type GISTs (59.3%) compared to GISTs with KIT mutations (23.0%) (P = .004). 50 One study including 35 patients found that GISTs with KIT exon 9 mutation were >10 cm, had greater enhancement ratio and greater area of tumour necrosis by comparison with those with KIT exon 11 mutation. 51 Of interest, in multivariable analysis, tumour necrosis was significantly associated with KIT exon 9 mutation. 51 Xu et al. have suggested a potential for texture analysis in identifying GIST with KIT exon 11 mutation. 28 These researchers found that non-gastric location, lower CD34 stain level and greater standard deviations of texture analysis parameters were associated with the absence of KIT exon 11 mutation in a series of 86 GISTs (59 with KIT exon 11 mutation). In this study, texture analysis performed better than visual assessment of tumour heterogeneity. 28 Starmans et al. developed a CT-based radiomics model to predict KIT mutation. 52 The model yielded AUCs of .52 for KIT and of .56 for KIT exon 11. 52 Although all these studies were retrospective and based on relatively small populations, they provide interesting information regarding the capabilities of imaging to inform on the mutational status of GIST and anticipate the most appropriate treatment.
Tumour mutation status is an active field of research in GIST imaging and current studies suggest a high potential for detecting GIST mutational status. 28,50-52 This should encourage further studies to investigate the impact of anticipating the mutational status in the real-life setting. Because 3.9% of wild-type GISTs harbor BRAF mutation, BRAF inhibition may represent a therapeutic option for this specific category. 6 However, no studies have identified imaging features associated with BRAF mutation and this should warrant further evaluation.
Risk Stratification and Survival Prediction
Risk Stratification
Risk assessment of postoperative recurrence of GISTs is based on tumour location, tumour size, mitotic count, and tumour rupture. This allows categorizing GISTs into very-low-risk, low-risk, intermediate-risk, and high-risk groups (modified National Institutes of Health [NIH] Joensuu classification). 53 Several studies have identified CT features suggestive of high-risk GISTs. Of these, tumour size, exophytic tumour growth pattern, and enlarged feeding vessels are significantly associated with high-risk GISTs (Figure 4). 50,54-57 Cannella et al. found that GIST size ≥5 cm (odds ratio [OR], 10.52; P = .009) and enlarged feeding vessels (OR, 12.08; P = .040) on CT images were independently associated with high-risk GISTs in multivariable analysis. 50 In a series of 113 patients with GISTs, Jung et al. found that non-gastric location (OR, 5.12; P = .029), ill-defined tumour margin (OR, 4.93, P = .023), and prominent vessels (OR, 6.78; P = .007) were significant variables associated with recurrence after surgery. 58 They further develop a preoperative, predictive model that achieved an AUC of .863 for identifying patients with postoperative recurrence. 58
In the study by Li et al. based on 64 gastric GISTs (41 low-intermediate risk GISTs and 23 high-risk GISTs), irregular lesion margins and tumour ulceration were more frequently found in high-risk GISTs (96% [22/23] and 43% [10/23], respectively) than in low-intermediate-risk GISTs 76% [76% [31/41] and 20% [8/41], respectively) (P = .041 for both). In addition, mean attenuation values on arterial phase images and peak attenuation values were greater in high-risk GISTs (78.72 ± 12.66 [SD] HU and 80.79 ± 12.37 [SD] HU, respectively) compared to the low-intermediate-risk GISTs (55.71 ± 13.77 [SD] HU, P < .001 and 72.66 ± 12.46 [SD] HU, P = .015, respectively).
46
Grazzini et al. confirmed the high value of enlarged feeding or draining vessels but also that of tumour necrosis in a series of 54 patients with 56 GISTs.
57
They found that these 2 features on contrast-enhanced CT images had 89.3% accuracy (50/56 GISTs) for the diagnosis of high-risk GISTs.
57
Tumour necrosis on CT was also identified as an indicator of high-risk GIST by Mazzei et al. who found significant correlation between percentage of tumour necrosis and mitotic index.
59
Peng et al. have identified overlying enhancing gastric mucosa on CT as an indicator of high-risk GISTs, with incomplete overlying enhancing gastric mucosa being the best independent variable (AUC, .835 [95% CI: .766-.904]; OR, 21.944 [95% CI: 4.344-110.863]) for the diagnosis of high-risk gastric GISTs according to the modified NIH criteria (Figure 5).
60
(A) 59-year-old man with 120-mm large, exophytic, gastric gastrointestinal tumour (GIST). Computed tomography (CT) image in the axial plane obtained after intravenous administration of iodinated material shows large, heterogenous, exophytic gastric tumour (arrows) with incomplete overlying gastric mucosa (arrowheads), consistent with high-risk GIST. (B) 34-year-old woman with 45-mm large, gastric GIST (arrow). CT image in the axial plane obtained after intravenous administration of iodinated material reveals complete overlying gastric mucosa (arrowheads), consistent with low-risk GIST.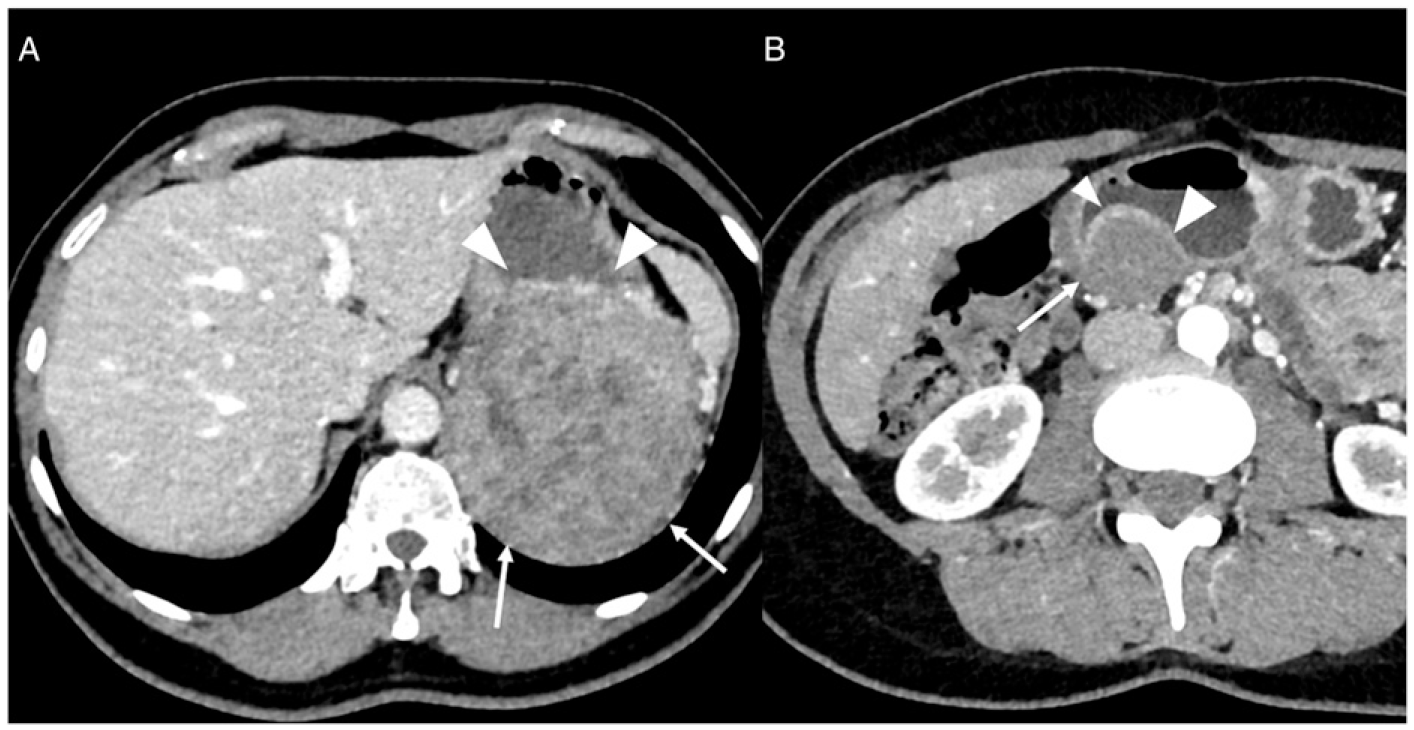
Li et al. have developed a CT-based nomogram for predicting the malignant potential of gastric GISTs. They used 4 variables identified in multivariable analysis as significantly associated with high-risk GISTs (tumour size >5 cm, cardia location, enlarged feeding or draining vessels, and mesenteric fat infiltration) to build a nomogram. 61 The nomogram yielded 86.5% sensitivity, 91.5% specificity, and 78% accuracy for the diagnosis of high-risk GIST. 61
Of interest, one study tried to identify CT variables that could help discriminate between very-low and intermediate risk among 151 small gastric GISTs (with a size between 1 and 2 cm). 62 This study found that radiomics features showed better performances (AUCs of .866, .812, and .766, in the training, validation, and testing cohorts, respectively) than visual evaluation using morphological high-risk features, such as calcification, necrosis, intratumoural heterogeneity, irregular border, or surface ulceration. 62 In the same line, Song et al. built a radiomics nomogram derived from CT-based texture analysis features that achieved an AUC of .905 in the validation cohort to discriminate between very low-risk and intermediate-high risk GIST. 63 Similarly, Palatresi et al. found significant association between radiomics features from CT data and Miettinen classification. 64 The superiority of radiomics for predicting recurrence-free survival of patients with GISTs has been reported by Wang et al. 65 In a series of 220 patients with intermediate or high-risk GISTs treated with imatinib, these researchers found that a model using 8 radiomics features extracted from portal venous phase CT data yielded AUCs of .80, .84, .76 for predicting 3-, 5-, and 7-year recurrence-free survival in the training cohort and AUCs of .78, .80, .76, respectively in the validation cohort. 65 One interesting result of this study is that the radiomics model outperformed a clinicopathological nomogram for the prediction of recurrence-free survival. 65 In addition, patients with high radiomics score had a greater benefit from imatinib used as adjuvant therapy. 65
One study reported the use of a predictive model to discriminate between GISTs with high mitotic count (>5/50 mitoses on high-power fields) and those with low mitotic count (≤5/50 mitoses). 66 The model was based on portal venous phase CT images of 108 patients with GISTs that were used to train a deep learning algorithm. The AI model yielded 100% sensitivity (95% CI: 65.6-100) and 90% accuracy (95% CI: 75.6-100) in the test cohort for the diagnosis of high mitotic count. 66 Although the test cohort included only 20 patients with a 1:1 ratio of high and low mitotic count GISTs, this study shows a potential for AI in risk assessment of GIST. 66
Survival Prediction
Some morphological and dynamic features have been associated with survival. GISTs with ill-defined margins (HR, 9.66) on CT are associated with shorter progression-free survival in multivariable analysis, whereas those with ill-defined margins (HR, 44.41) and internal hemorrhage (HR, 30.22) are independently associated with shorter overall survival. 50 In a series of 132 patients with gastric GISTs who underwent surgical resection, Chen et al. have identified that serosal invasion (HR, 5.277; P < .001) and volume of tumour viable component as estimated by enhancing tissue volume with attenuation >30 HU (HR, 1.447; P = .005) on portal venous phase CT were independent variables associated with shorter disease-free survival after curative resection. 67
Chen et al. developed and validated a prognostic ResNet nomogram for recurrence-free survival at 3- and 5-years after complete surgical resection of local primary GIST based on deep learning. 15 In an external validation cohort, the nomogram yielded AUC of .722 (95% CI: .453-.991) for predicting recurrence-free survival at 3-years and AUC of .923 (95% CI: .812-.999) for predicting recurrence-free survival at 5-years, with performances that outperfomed those of clinical models and modified NIH criteria. 15
Response to Therapy
Assessment of tumour response in GISTs remains challenging. This is because size alone does not provide accurate information relative to response to therapy and additional information is needed. 13 In this regard, RECIST 1.1. Criteria that are based on tumour size only are limited for GISTs. 13 The assessment of tumour response to therapy has been studied using dual-energy CT. An early study involving 18 patients with GISTs, found that iodine uptake by GISTs on dual-energy CT was more marked in progressive GISTs under TKI. 18 Another study involving 17 patients found that iodine attenuation used in conjunction with change in tumour size at dual-energy CT outperformed RECIST 1.1 and mChoi criteria for the assessment of treatment response to TKI. 19
Tang et al. prospectively investigated the potential of DWI as a predictor of early tumour response in 32 patients with 91 GISTs who received imatinib. 68 They found that baseline ADC value was lower in the 56 GISTs that showed good-response (1.06 ± .27 [SD] 10−3 mm2/s) compared to that in the 35 GISTs with poor-response (1.24 ± .32 [SD] 10−3 mm2/s) (P = .005). 68 Similarly, these researchers observed that a marked increase in tumour ADC at one week after the start of therapy was associated with good tumour response. 68 In a later study, involving 62 patients with GISTs, the same group found that ADC surpassed T2-weighted images and change in tumour size to differentiate between good and poor response to targeted therapy (either imatinib in 44 patients or sunitinib in 18 patients). 69 These 2 studies by Tang et al. suggest a potential for DWI but because of technical issues and variability in ADC measurement, DWI has not now a well-established role for the follow-up of GISTs.
Ekert et al. identified CT texture features associated with disease progression in a series of 25 patients with metastatic GISTs who underwent first-line imatinib therapy and other TKI as second line therapy. 70 Two second order features (grey-level co-occurrence matrix (Glcm) inverse difference, grey-level dependence matrix (Gldm) inverse difference normalized) and 2 high order features (grey-level run length matrix (Glrlm) non-uniformity normalized and neighboring grey-tone difference matrix (Ngtdm) coarseness) were significantly greater in patients with progressive disease compared to those with stable disease. 70 These 4 features are indicators of tumour homogeneity, thus confirming results of prior observations showing that homogeneity is an indicator of favorable tumour response to TKI. 71
Conclusion and Future Developments
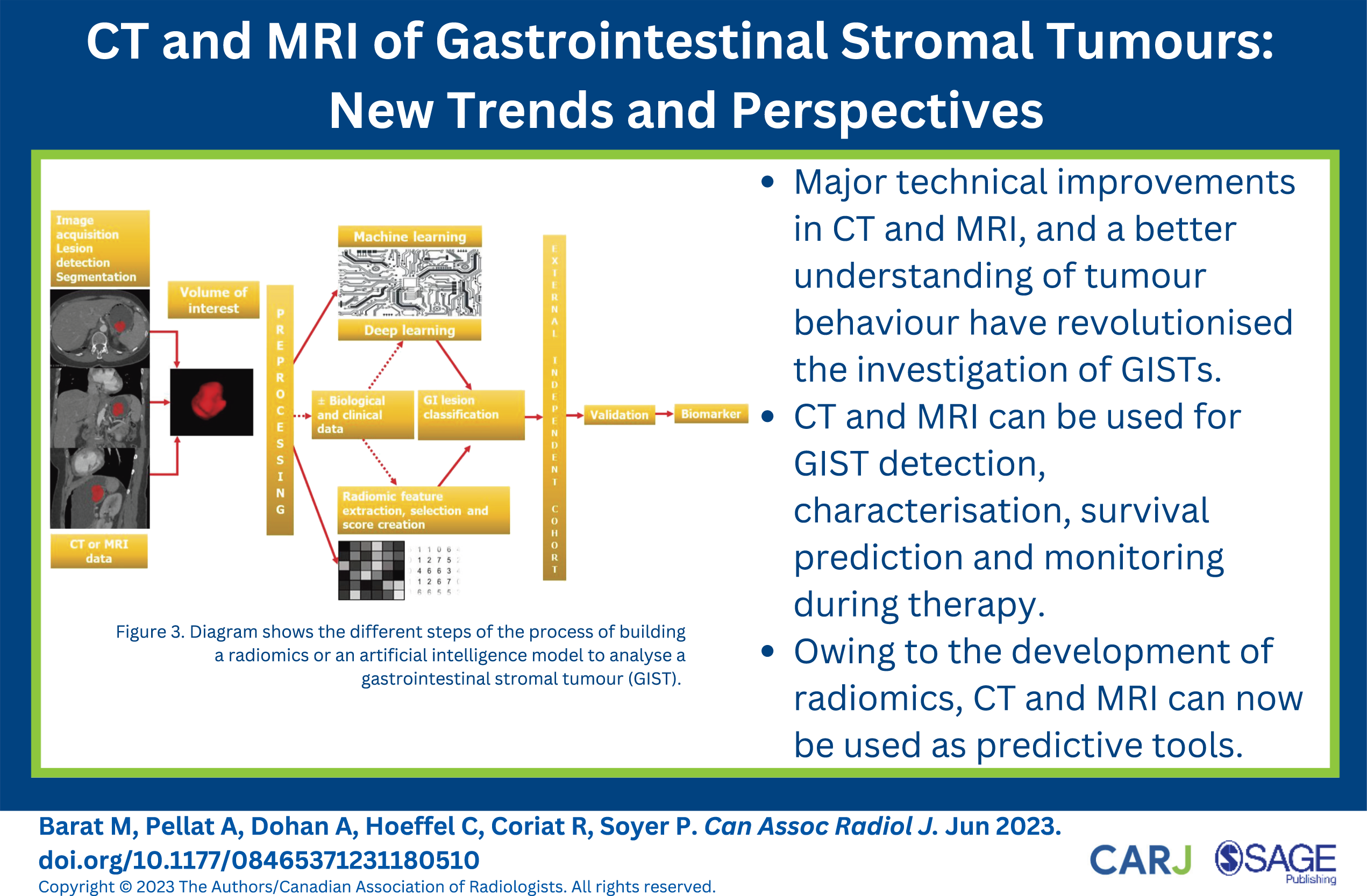
Major technical improvements in CT and MRI in conjunction with a better understanding of tumour behavior have revolutionized the investigation of GISTs. As a counterpart, more demand has been placed on imaging, which may be used not only for the initial assessment of GISTs but also to provide information relative to potential risk and genetic mutation. Owing to the development of radiomics, CT and MRI can now be used as predictive tools. Although tremendous advances have been made in GIST imaging and promising results have been published, these results are still preliminary. The next step should be to investigate how all these advances can be implemented in the real-life setting and how they can positively influence patient care and outcome.
Footnotes
Author Contributions
All authors attest that they meet the current International Committee of Medical Journal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
