Abstract
Several articles in the literature have demonstrated a promising role for breast MRI techniques that are more economic in total exam time than others when used as supplement to mammography for detection and diagnosis of breast cancer. There are many technical factors that must be considered in the shortened breast MRI protocols to cut down time of standard ones, including using optimal fat suppression, gadolinium-chelates intravascular contrast administrations for dynamic imaging with post processing subtractions and maximum intensity projections (MIP) high spatial and temporal resolution among others. Multiparametric breast MRI that includes both gadolinium-dependent, i.e., dynamic contrast-enhanced (DCE-MRI) and gadolinium-free techniques, i.e., diffusion-weighted/diffusion-tensor magnetic resonance imaging (DWI/DTI) are shown by several investigators that can provide extremely high sensitivity and specificity for detection of breast cancer. This article provides an overview of the proven indications for breast MRI including breast cancer screening for higher than average risk, determining chemotherapy induced tumor response, detecting residual tumor after incomplete surgical excision, detecting occult cancer in patients presenting with axillary node metastasis, detecting residual tumor after incomplete breast cancer surgical excision, detecting cancer when results of conventional imaging are equivocal, as well patients suspicious of having breast implant rupture. Despite having the highest sensitivity for breast cancer detection, there are pitfalls, however, secondary to false positive and false negative contrast enhancement and contrast-free MRI techniques. Awareness of the strengths and limitations of different approaches to obtain state of the art MR images of the breast will facilitate the work-up of patients with suspicious breast lesions.
Breast magnetic resonance imaging (MRI) has been subject to significant quality improvement since surface coils were used to provide higher spatial resolution.1,2 Dedicated breast coils are mandatory for state-of-the-art exams.3-5 Intravenous gadolinium-chelates based contrast agents (GBCA) are used to detect angiogenesis and form the basis of dynamic-contrast enhanced (DCE)-MRI.1,6-8 Unfortunately, the need for i.v. GBCA results in brain, bone and soft tissue retention in patients with a normal kidney function, which is accentuated in patients with poor renal function.9-11 It is unknown whether these GBCA deposits are harmful or can lead to adverse health effects. Studies12,13 have shown that the stability of GBCA is associated with the degree of retention. For this reason, linear nonionic contrasts retain more gadolinium than linear ionic, and linear ionic more than macrocyclic contrasts.14-16 A recent trend to utilize gadolinium-free MRI techniques in clinical scenarios where use of GBCA is a challenge, like pregnancy, severe contrast allergies, and lactational status17-19 rely on Diffusion-(GBCA-free)-MRI techniques (DWI/DTI-MRI) that are based on the intrinsic cellular contrast.20-26 Certain breast Diffusion-MRI protocols have also been used for high-risk screening and as a problem-solving tool in selected cases,27,28 however it should not be used as stand-alone technique yet, as clinical protocols are still under evaluation.29,30
This review provides considerations that may be helpful for clinical practice of both general MRI and breast subspecialty radiologists. The improvements in the protocols which allow decreased magnet time and potentially its costs will be discussed.
Indications for Breast MRI
A summary of the current indications for Breast MRI is presented in the Table 1.
Indications for Breast Magnetic Resonance Imaging (MRI) Associated to the MRI Protocol.
Abbreviations: DCE-MRI = dynamic contrast-enhanced MRI; DWI/DTI = diffusion MR, either diffusion-weighted or diffusion-tensor.
* Not performed as stand-alone method.
** If the inconclusive findings are related to determine the integrity of silicone breast implant(s).
Diagnostic Breast MRI for Silicone Implant Evaluation
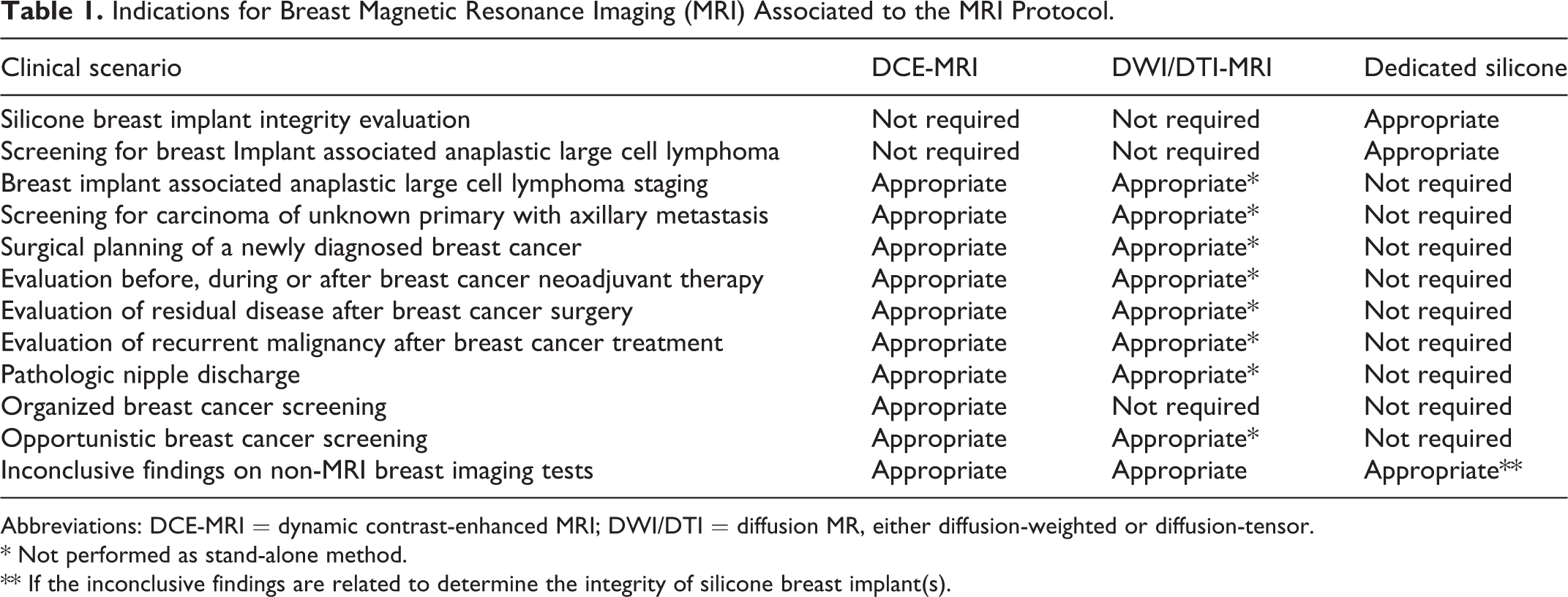
Breast MRI shown to be superior in detecting implant rupture31-33 with sensitivity and specificity ranging from 78-99% compared to ultrasound and mammography of respectively, 59-81% and 28-89%. Classical MRI findings for intracapsular rupture include the linguine (Figure 1) and keyhole signs. 34 The MRI protocol for detection of silicone leak is different from the protocol for breast cancer. It is a GBCA-free breast MRI using protocols mainly based on long relaxation time and selective silicone-specific sequences that allow separation of the water and silicone in the presence of inhomogeneous magnetic fields and do not require diffusion/DCE MRI techniques. Hence, it is important for the referring physician to clearly define the purpose of the breast MRI if the concern is a silicone leak. For the increasingly recognized risk of Breast Implant-associated Anaplastic Large Cell Lymphoma (BIA-ALCL), a T-cell lymphoma typically associated with textured implants occurring at a median of 10-years after inclusion, features of peri-implant effusion and less frequently masses may be image detected. 35 For clinically suspected BIA-ALCL, breast ultrasound is elected the first imaging test of choice. 36
A 54-year-old female with bilateral silicone breast implants included 5 years ago. Breast MRI performed using a 1.5 T scanner (TR = 5050 ms/TE = 76 ms) showing a sagittal view of the right breast demonstrating presence of the linguine sign (arrow) in the inferior aspect of the breast implant and presence of the extracapsular free silicone in the pre-pectoral region (arrowhead).
Diagnostic Breast MRI for Carcinoma of Unknown Primary
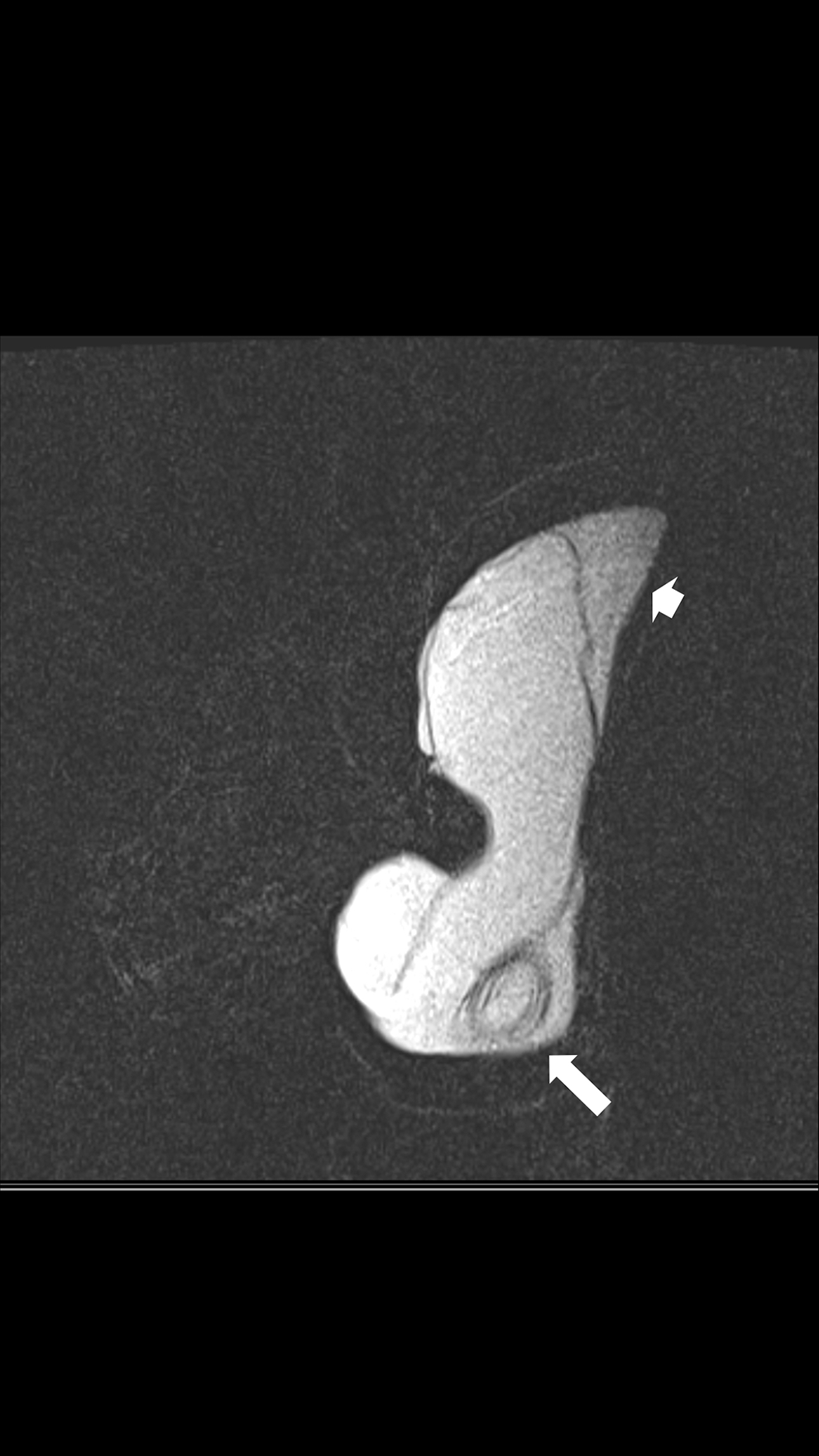
The American Society of Breast Surgeons 2017 guideline supports the use of breast MRI to search for occult breast cancer in patients with Paget’s disease of the nipple when clinical examination and conventional breast imaging fail to detect the primary breast cancer. 37 Occult breast carcinoma may present with axillary lymphadenopathy that is histologically consistent with metastatic breast cancer (Figure 2). Less than 2% of patients present with clinically palpable axillary nodes and negative mammography/breast ultrasound. In such a clinical scenario, breast DCE-MRI can detect 75% of the initially occult primary breast carcinoma in the conventional breast imaging.38,39 Patients with a negative MRI who underwent breast conservation treatment instead of mastectomy had 96% breast cancer-free survival in a multicenter study with a small risk of ipsilateral recurrence. 40
Carcinoma of unknown primary in a 42-year-old female with right axillary metastasis T0N1 treated with neoadjuvant chemotherapy, followed by axillary node dissection and whole breast plus axillary radiation. The arrow shows the enlarged level I axillary lymph node in the initial breast magnetic resonance imaging 3D maximum intensity projection (MIP) images during the 2nd minute post gadolinium-based contrast agent without suspicious findings in the breast. No primary breast cancer was identified. Patient is well after 3 years post treatment.
Diagnostic Breast MRI for Preoperative Assessment of Breast Cancer
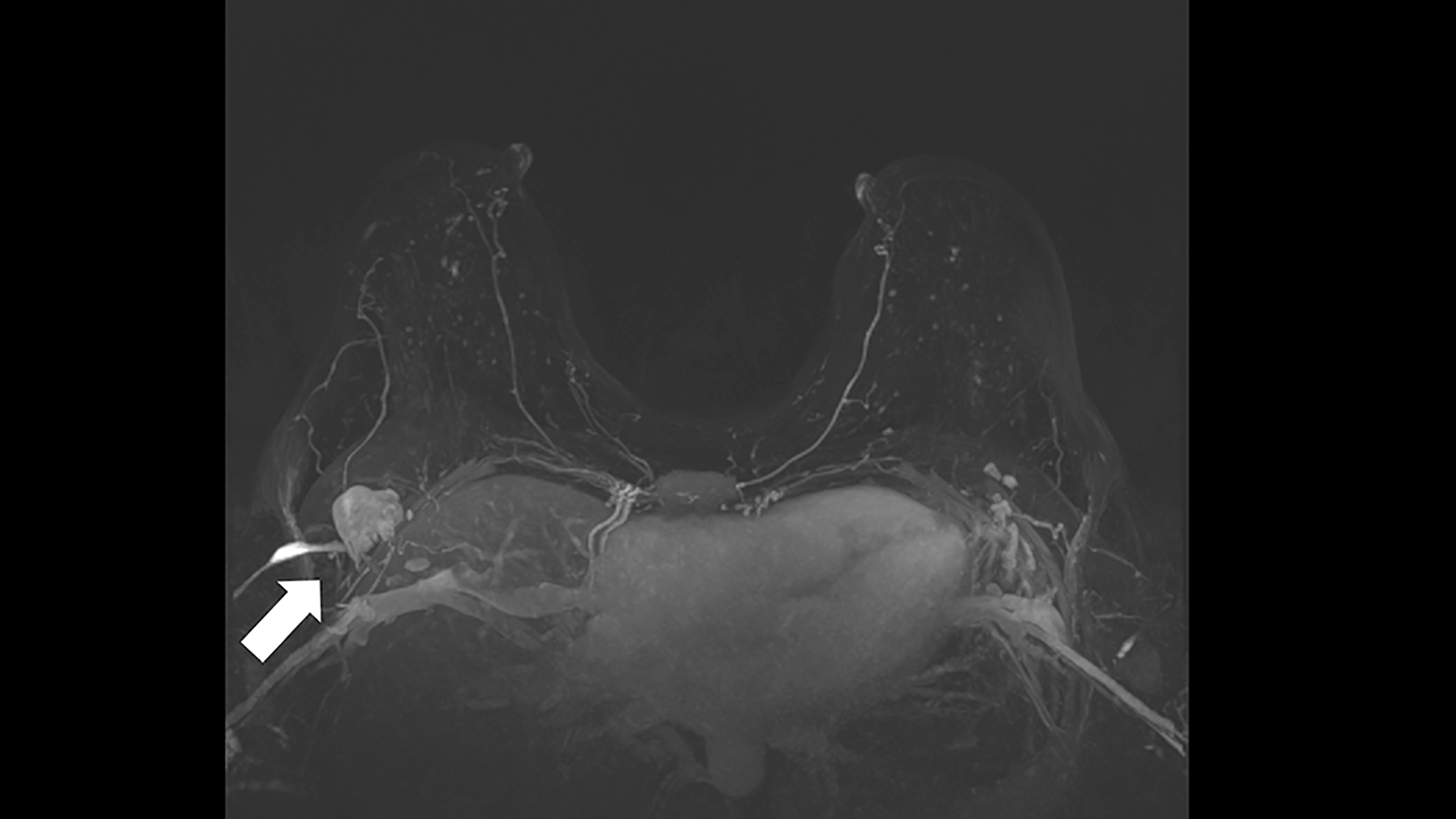
The initial studies have shown that breast DCE-MRI can detect additional cancer (not identified on mammography/or ultrasound) in up to 33% of patients with newly diagnosed carcinoma.41-43 Breast MRI for evaluation of extent of disease, as a routine test, remains controversial among surgeons. 37 It is unclear if MRI improves surgical outcomes, impacts survival or is cost effective. 44 A multicentric randomized controlled trial (RCT) found surgical re-excisions to be significantly lower, 5% vs 15% (p < 0.0001) with MRI. 45 A Canadian population-based study demonstrated that just 14.8% of breast cancer patients treated 2003-2012 received pre-operative breast MRI. This test increased across all cancer stages by 8-fold, from 3% to 24% (p < 0.001). 46 For suspected multicentric or contralateral disease, needle biopsy is required before surgical management to avoid unnecessary surgery for lesions that could be proven benign in up to 8%. 47 For local staging, MRI is highly accurate in suspected pectoral (Figure 3) muscle invasion and may also help to assess nipple to tumor distance providing useful information about the ability to perform nipple sparing mastectomy if >1 cm from the nipple. 48 Overall, breast MRI is quite useful in patients in whom the disease extent is uncertain from physical exam, mammography/ultrasound and several breast surgeons rely upon this imaging method for tailored surgical plan.49,50
A 68-year-old “survivor” patient with a personal history of a left lumpectomy for invasive ductal carcinoma 7 years ago presents with a self-detected lump not demonstrated by mammography. Sonographic assessment show a suspicious mass lesion (A) in a very deep position in the breast and muscle invasion was suspected. Ultrasound-guided core biopsy shows invasive ductal carcinoma, NOS. Breast MRI requested by the attending surgeon for optimal surgical plan shows 2 distinct (C) regions of the recurrent cancer associated to the post contrast enhancements. The arrow shows the palpable lump in the axial 2nd min DCE (B) and in the reformatted isotropic axial image in the sagittal plane with subtraction (C). The arrowhead shows the second mass lesion, more inferior that was solely MRI detected. Metastatic level I axillary nodes are evident (C).
The best established indications for preoperative MRI are for patients with dense breasts, those with invasive lobular carcinoma, or for those younger than 50 years of age.
Diagnostic Breast MRI Related to Neoadjuvant Therapy (NAT)
Breast MRI can be done before starting the pre surgical treatment with a wide variety of drugs that are indicated based on molecular pathology. 51 The overall survival and disease progression in patients receiving neoadjuvant versus adjuvant chemotherapy are not considerably different, 52 however women receiving NAT are more likely to be treated with breast conservation. This may be related to the substantial role that breast MRI plays in detecting suspicious lesions and providing accurate cancer mapping. Breast MRI has the capacity to provide information on responders versus non-responders to NAT and to detect residual disease (Figure 4). Breast MRI is more accurate than clinical exam, mammography/sonography to correctly identify the presence of residual tumor in 92% of cases and to correctly identify the imaging findings of a pathologic complete response (pCR) in 90% of cases.53-57
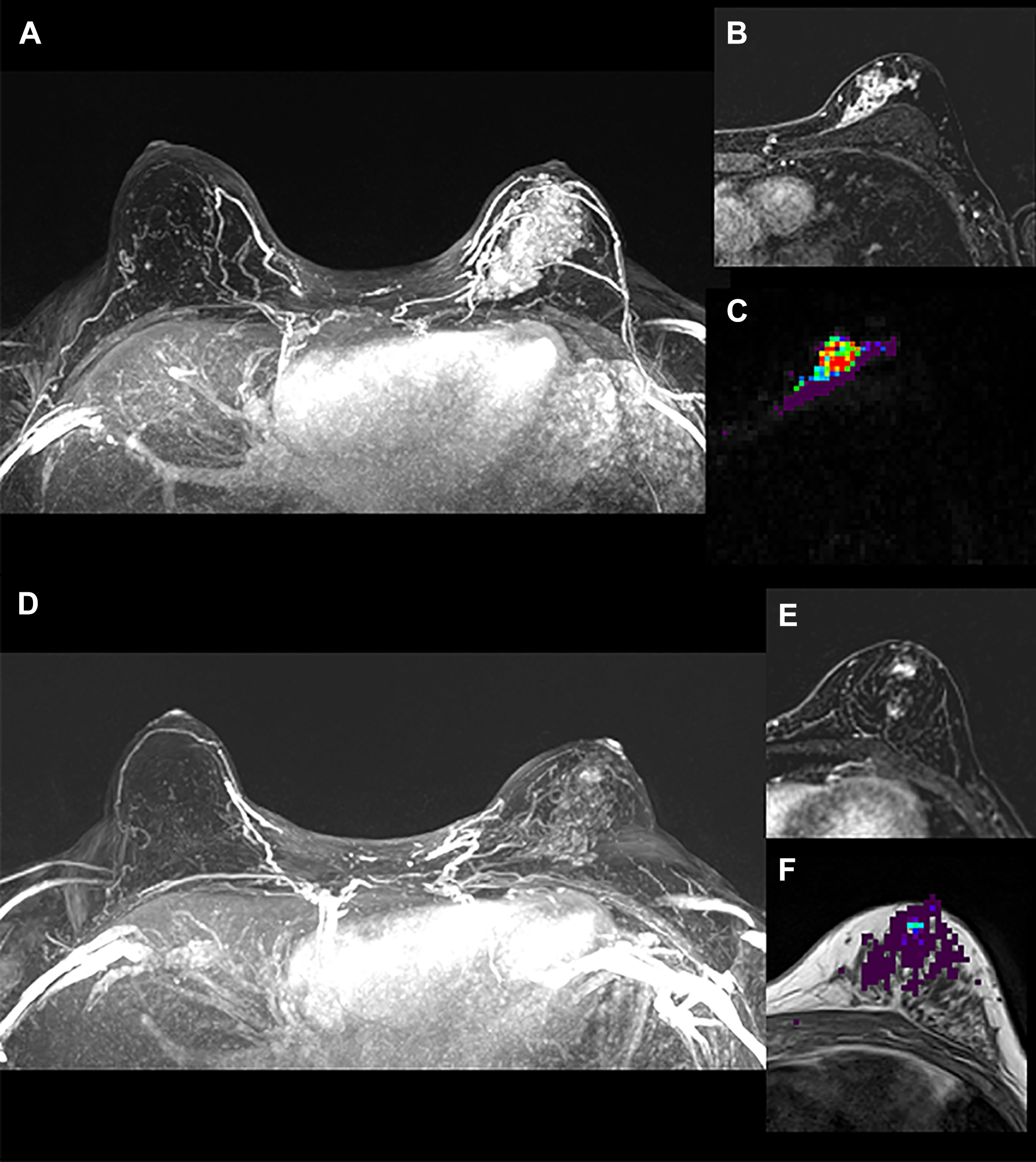
Invasive ductal carcinoma, grade 3, ER positive, PR positive, HER2-neu negative before starting the neoadjuvant chemotherapy with multiparametric diagnostic breast MRI showing a large enhancing cancer in the left breast on (A) the axial maximum intensity projection (MIP) images during the 2nd minute post gadolinium-based contrast agent, (B) and (C) are the same location slice taken in the upper breast showing the clumped internal non mass enhancement pattern during the 2nd minute post contrast of the dynamic contrast enhancement (DCE)-MRI and the color coded lambda-1 mapping of diffusion tensor MRI. The same patient underwent another multiparametric diagnostic breast MRI after receiving the 6 cycles of 21 days of chemotherapy with 3 drugs (fluoracil, epirubicin, and cyclophosphamide, called FEC) on day 1 of the first 3 cycles and with another drug (docetaxel, called D) on day 1 of the 3 last cycles (FEC-D). The residual post contrast enhancement on DCE-MRI was reported as 10 cm without significant internal response based on the RECIST criteria where (D) and (E) show, respectively, MIP and T1-weighted with fat suppression 3D-gradient echo in the 2nd minute and (F) shows the residual cancer demonstrated by DTI-MRI measuring 6 mm in the lambda-1 overlaid over T1-weighted image without fat suppression, indicating partial response. Same slice location corresponding to (F). The pathology results show overall cellularity of 5% within a 29 × 25 mm tumor bed of a residual invasive ductal carcinoma of no special type (NST) measuring 6 × 3 × 3 mm, Residual Cancer Burden Class II.
Diagnostic Breast MRI for Postoperative Assessment
In patients where there is a close or positive surgical margin, MRI can be used to detect unsuspected multicentric or multifocal disease at the current threshold of 5 mm or higher. However, MRI may have difficulties in differentiating post-surgical change from residual cancer with a very low negative predictive value (45%) when there is associated enhancement of the surgical cavity.58-60
Diagnostic Problem-Solving Breast MRI
Breast MRI is extremely useful if there is no enhancement as the negative predictive value (NPV) is close to 100%.61-63 Used when mammography or sonography is not sufficient, e.g. questionable architectural distortions; nipple discharge with inconclusive findings; suspicious lesions seen in a single view digital mammography/digital breast tomosynthesis or even when equivocal changes at site of previous surgery (Figure 5) are noted. If inconclusive sonographic findings with a clinical suspicion of BIA-ALCL, DCE (with or without DWI/DTI)-MRI is indicated to assess for peri-implant effusion or mass.36,64 Organizing an MRI in cases where there is a palpable concern and an imaging correlate in order to substitute the need of the breast tissue histological diagnosis is not appropriate. If there is a clinical focal palpable concern, the appropriate choice imaging test is sonography plus or minus diagnostic mammography and based on the imaging descriptors, it could be likely benign (BI-RADS 3) or suspicious/highly suspicious (BI-RADS 4/ 5) and biopsied to establish the histological diagnosis and appropriate management. MRI should not replace the need for tissue biopsy.
Screening mammography shows a developing focal asymmetry with associated distortion. (A) shows the left craniocaudal view and (B) shows the left mediolateral oblique view mammography. Digital breast tomosynthesis slices (not shown here) demonstrated persistent findings and a diagnostic focused sonography (C) of the left breast did not show presence of a true mass lesion associated to a focal region of decreased echogenicity correlation. Figure D is the 3D-MIP images of the 2nd minute. The diagnostic problem-solving breast MRI does not show evidence of recurrent cancer, showing scar tissues without evidence of suspicious post gadolinium enhancement.
Breast MRI Screening for Early Breast Cancer(s) Detection.
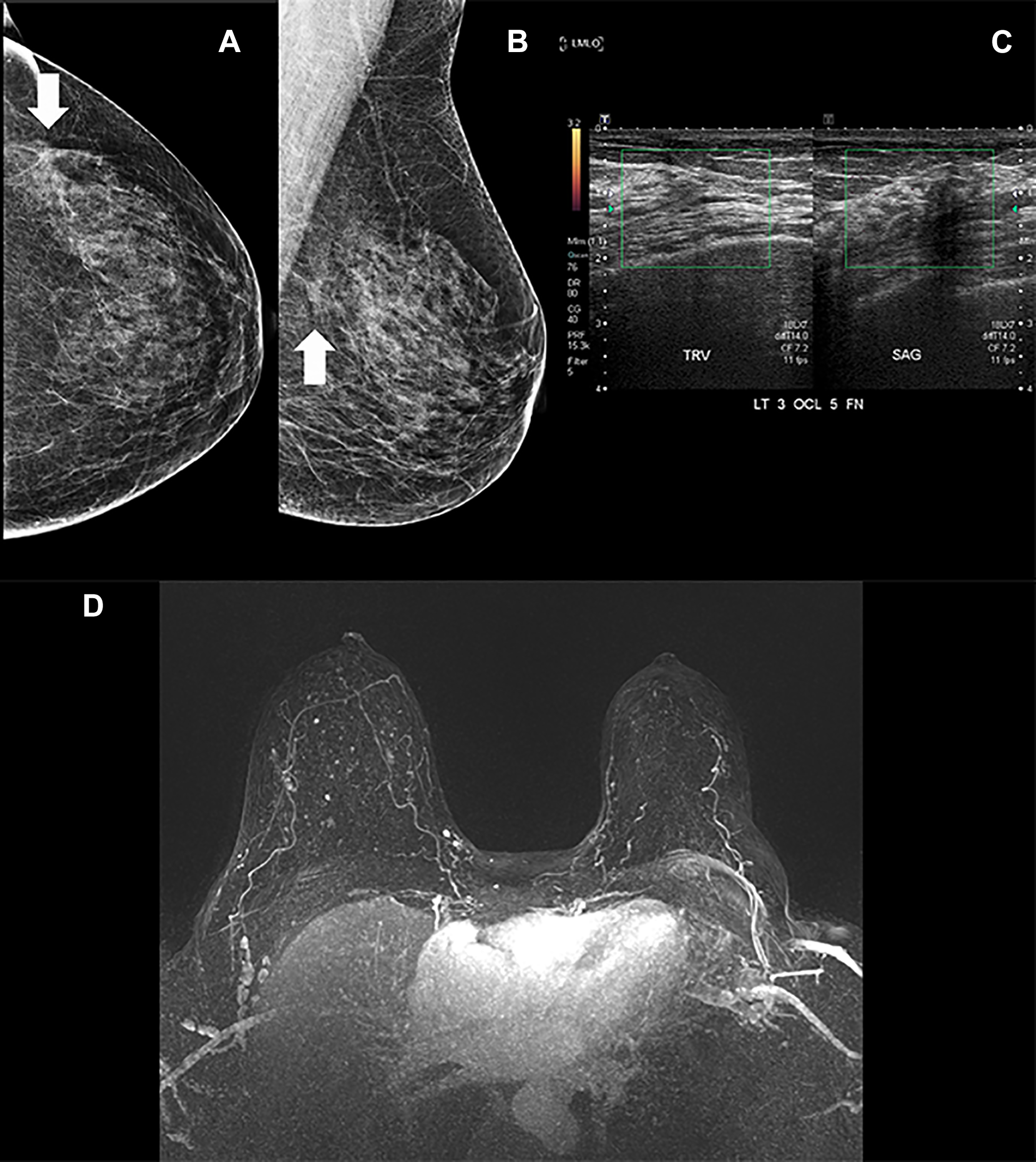
Screening breast MRI may apply to the evaluation of the contralateral breast in preoperative or pre-NAT evaluation in patients with breast cancer, and may be identified in 3-10%, dependeing on tumor histology and other risk factors. Breast MRI has become an essential screening modality for women at high-risk for breast cancer, particularly those with genetic mutations that markedly increase their risk of developing a breast cancer, such as BRCA 1/ 2 gene carriers 65 or women with a very strong family history of breast and or ovarian cancer particularly in their first degree’s relatives, women who have received chest irradiation for diseases during childhood, Hodgkin’s lymphoma, and women with a lifetime risk of breast cancer >20-25%, as estimated by multiple risk assessment models. More recently, MRI has been recommended for higher than average risk women, including women with dense breasts (Figure 6), which is an independent risk factor for breast cancer. 66 In the high-risk male population screening with breast MRI is not frequently used. 67
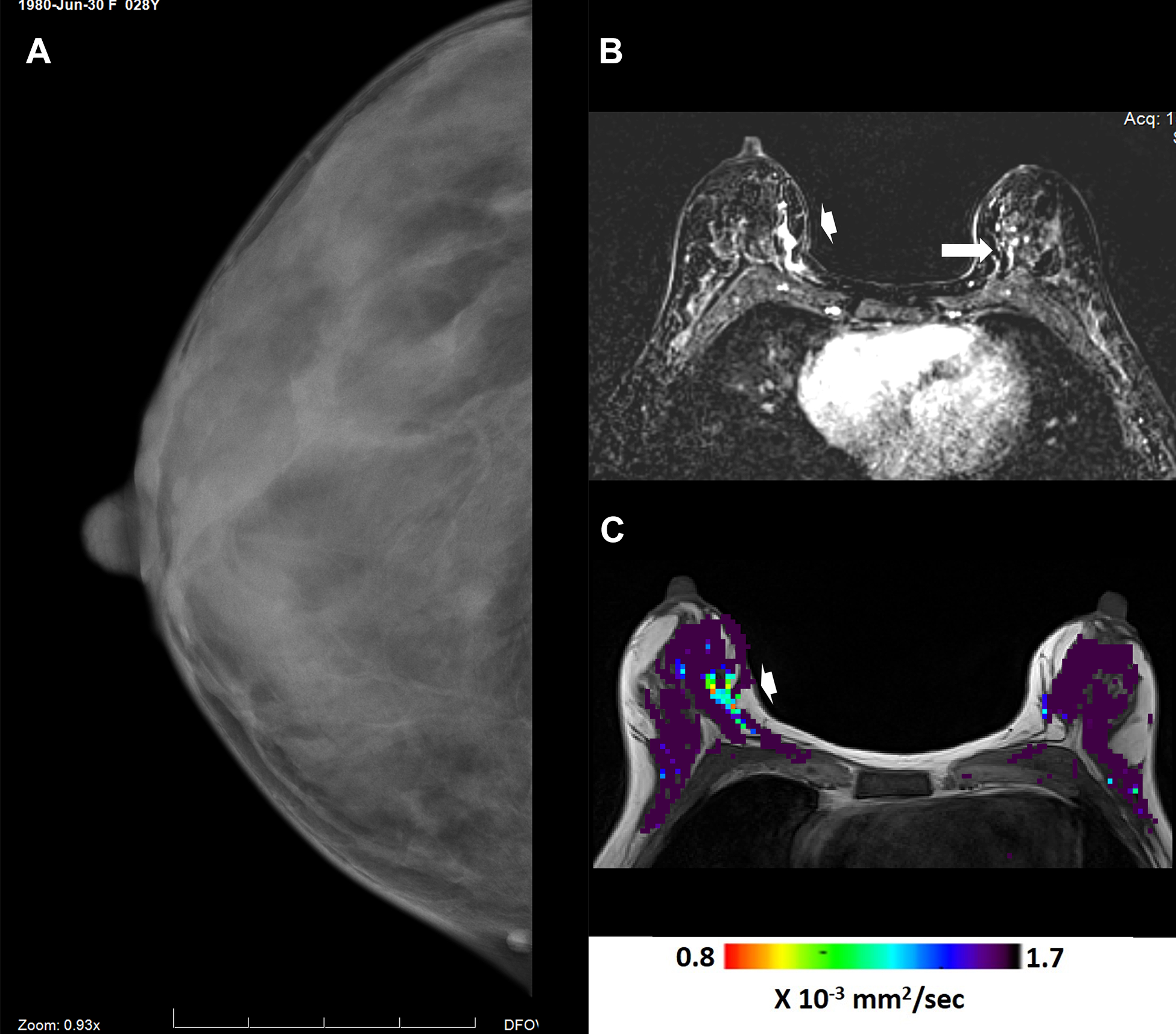
MRI detected right breast cancer. Female below age 40 with a personal history of allogeneic hematopoietic cell transplantation undergoing opportunistic screening with mammography and multiparametric breast MRI. Mammography shows extreme dense breast tissue without findings in the medial aspect of the right breast in cranio-caudal (A) view projection. Breast MRI shows the cancer (arrowhead) on both the DCE-MRI (B) and the DTI-MRI (C) images obtained in the same table position. The arrow (B) shows focal region of non-mass enhancement in the contralateral breast on DCE-MRI that is negative on DTI-MRI. No cancer found in the left breast.
No RCT studies are available to prove the efficacy of screening breast MRI in the general population, however based on that 1 in 8 Canadian women will develop breast cancer during their lifetime and 1 in 33 will die from it, 68 together with the scientific knowledge that of 9 cancers in 9 asymptomatic women will be all detected by MRI and 3 of the 9 cancers will be detected by mammography or sonography, it makes sense to develop strategies to make available for breast MRI to the general population.
The performance indicators of breast cancer screening using DCE-MRI69-73 show a wide range in published results, e.g. average sensitivity 91-98%; specificity 78-99%; NPV over 95%; positive predictive value (PPV) 53-75% that are mainly due to the radiologists’ learning curve and various MRI protocols and scanners. The ACR benchmarks for screening MRI are sensitivity >80% and specificity >85-90% with PPV3 (for biopsy performed) of 20-50% and cancer detection rates of 20-30 per 1000. 74 It is essential to audit outcomes and the false positives on DCE-MRI, given the overlap of benign and malignant enhancing lesions that must undergo proper diagnostic work up. 75 A RCT of 40,373 women with dense breasts undergoing supplemental breast cancer screening with MRI found a false positive rate of 7.98%. 76 There are also false negative DCE-MRI cases e.g. invasive lobular carcinoma and low-grade DCIS which may not be detected because of the lack of post-GBCA enhancement. One study 77 demonstrated that almost one-third of the false negative DCE-MRI were missed cancers while another third was misinterpreted. Double reading for screening breast MRI, radiologists’ audits and understanding the cognitive processes and unconscious biases are required to avoid human errors and decrease the number of unintentional missed breast cancers. 78
What Makes MRI Detect Breast Cancer?
The MRI key principle in detecting breast cancer relies upon the evaluation of the tumor angiogenesis 8 or the development of new blood vessels/capillaries in tumors demonstrating that a primary cancer cannot grow above 1-2 mm without new vessels. The secondary cancer (metastasis) genesis involves not only angiogenesis but angiogenesis and lymphangiogenesis. 79 The analysis of the physiological model of GBCA distribution in the breast 80 considers the body extracellular volume, the plasma concentration of GBCA and the breast tissue contrast uptake that is regulated by the influx transcapillary transfer constant (flow and capillary permeability times capillary surface-area) and the efflux transcapillary transfer constant (which a ratio of the extracellular volume fraction. The breast MRI curve of enhancement with wash-in and wash-out measurements is evaluated in a timeline and must have at least 3 points (preselected based on the model parameters of DCE and the MRI protocol) in order to be created. The curve of enhancement is a visual tool for the breast radiologist that represents the physio pathological features of cancer (showing wash-in phase-type 3 curve and fast rate of enhancement) versus benign (having mostly persistent wash-in phase-type 1 curve and slow rate of enhancement), when the transcapillary transfer constant is calibrated adequately in the many commercially available breast MRI software.81-83
There has been a growing interest in the investigation of modified breast MRI protocols for cancer detection, from techniques that are completely GBCA-free like DTI, 84 to the distinct sequences that focus mainly in the transcapillary GBCA transfer in the breast lesions requiring re-engineering of older MRI sequences in breast MR imaging 85 in order to achieve high temporal resolution that became feasible in light of technical MR hardware improvements such as “Ultrafast MRI.” There are smart ways of simply not acquiring some sequences of the usual clinical protocol, named “Abbreviated” MRI, 86 that led to reduced time of interpretation and scan time by acquiring only a pre-contrast and single post contrast T1-weighted sequence, using the maximum intensity projections (MIP) to obtain just one single high-contrast image.
Speeding Up Breast MRI
Does Breast MRI take less time to be reported or to be performed? In the last few years different names for breast DCE-MRI started popping up in the media for layperson and the scientific literature: abbreviated MRI also called “fast MRI” or AB-MR, abridged MRI, Ultrafast MRI, etc. They are not synonyms and do not have the same technique or carry the same MRI protocol, however all have the aim to decrease time and consequently the expectations of reducing costs associated with the standard-of-care breast DCE-MRI.
The term “Ultrafast MRI” has been used in Abdominal, Cardio and Neuro Imaging since the early 90’s87-89 to describe process of decreasing sequence’s time and increasing from a fraction of a second 90 to a fraction of a millisecond temporal resolution.91,92 The “Ultrafast” breast DCE-MRI93-95 sequences currently take over 5 seconds and are time-resolved magnetic resonance angiography (MRA) sequences with under sampling of the k-space periphery and using view-sharing that allows rapid acquisition of multiple images during the passage of the contrast bolus.
The concept is simple as a wave is characterized by its wavelength (λ), the distance between 2 corresponding points or successive peaks. The wavenumber (k) is simply the number of waves or cycles per unit distance. A typical time resolved MRA study might contain more than 20 images obtained at rates as rapid as 1-2 frames per second. Inevitably there is an inherent trade-off between spatial and temporal resolution for those techniques. The center of k-space has information about basic image contrast, while the details and edges are encoded in the peripheral k-space. The temporal resolution is highly impacted if sampling more points are required. For this reason, those time-resolved MRA techniques balance these competing resolution requirements using what is called view-sharing that is a process that start with a pre-contrast, full-resolution image of the area of interest followed by changes in the sampling of the k-space, e.g. during the passing of the contrast bolus, the center of k-space is sampled much more frequently than the periphery. The data from those different k-space sampling is then combined in order to create a series of time-resolved images with satisfactory spatial-resolution and pre-contrast full resolution image can be used as a mask for subtraction improving vascular conspicuity.
The time-resolved MRA techniques currently used in Breast MR imaging acquire 3D k-space typically using radial sampling schemes in round or oval “cylinders” and they are vendor based. For example, in our institution we have Siemens’ scanners, and it is named as TWIST (“Time-resolved angiography With Stochastic Trajectories”) 96 ; Phillips uses the name of 4D-TRAK (“4D Time-Resolved Angiography using Keyhole”) 97 ; General Electric (GE) calls theirs as TRICKS (“Time-Resolved Imaging of Contrast KineticS”) 98 ; Toshiba named Freeze Frame 99 and Hitachi named TRAQ (“Time-Resolved AcQuisition”). 100
All time-resolved MRA techniques carry a 3D-spoiled gradient echo sequences with thin slices, very short TRs and TEs, low flip angles and parallel imaging acquisition, similar to the DCE-MRI sequences used in the breast. Specific k-space parameters must be selected, the size of the central k-space region, its refresh rate (in frames per second) and the total number of frames to be acquired. This process is all not currently standardized for breast MRI use and those specifications vary by vendor. The GE’s MRI sequence name DISCO (Differential Subsampling with Cartesian Ordering) shares similar features with the Siemens’ TWIST and their own sequence TRICKS which is used for dynamic perfusion imaging instead of for MRA. DISCO uses a dual-echo 3D SPGR sequence with pseudo-random variable density k-space segmentation and a view sharing reconstruction to achieve high spatio-temporal resolution. The dual, i.e. in-phase and out-of-phase echoes allows separation of fat and water signals and this sequence can also achieve a strong fat suppression using a 2-point Dixon fat-water reconstruction algorithm. DISCO is used for breast DCE-MRI and subdivides k-space into several annular elliptical regions that are incompletely sampled and randomly together with a central k-space region that is consistently sampled. 101
The first published study of the “Ultrafast Breast MRI” in 2014 using a time-resolved technique applied in breast imaging (TWIST) showed high sensitivity (90%) with very low specificity (67%). 102 Other studies followed showing lower specificity of the fast protocols or the abbreviated ones compared to the full protocol. The differences however were not significant between the traditional full diagnostic protocol and the abbreviated ones.103,104 A recent systematic review and metanalysis 103 of available studies (2,763 women with 3,251 screening rounds) comparing the standard (full) protocols with the short (abbreviated) DCE-MRI protocols shows very low level of evidence to suggest that the abridged protocols could be accurate for breast cancer screening. Systematic reviews and metanalysis focussing exclusively on what is called “Ultrafast Breast MRI” are not yet available.
Essential Breast MRI Should be Reproducible
To date there is no standardization for the DCE-MRI protocols105-111 either for traditional ones with averaged time of 3 min to 7 min post-GBCA or the abbreviated ones where a single point post contrast is obtained with the expectation that no enhancement at all is detected. There is even less standardization for the “ultrafast breast MRI.”80,88 Indeed, there isn’t full agreement about what exactly is meant by “Ultrafast” for other subspecialties.112,113 On the other hand, the International Breast Diffusion-Weighted Imaging Working group have established basic requirements about DWI-MRI protocols used in association to DCE-MRI in the Multiparametric Breast MRI22,95 in order to have the diffusion breast MRI incorporated to the BIRADS in the near future.
One Step Forward and Two Steps Fast Forward
Current clinical practice shows that adding Diffusion-MRI (either DWI-MRI or DTI-MRI) is helpful to increase specificity4,95 of DCE-MRI (Multiparametric Breast MRI). The T2-weighted imaging can be interchangeable with the b0 images (b value = 0 s/mm2) from the Diffusion-MRI decreasing time. In order to achieve this interchangeable status, the amount of noise must be minimal, and the fat suppression should be optimized which is a limitation if scanners are not optimized and calibrated for diffusion sequences.25,55,84
The Multiparametric Breast MRI (Table 2) is appropriate to be used in Diagnostic Breast MRI and the patient’s time in the scanner would range 4-8 min for the diffusion sequences and 4-7 min for the DCE-MRI (T1-weighted fat suppressed 3D gradient echo (GE) pulse sequence obtained before contrast injection and then repeated as rapidly as possible during 4-5 min after GBCA injection, with slice thickness of 3 mm or less and in-plane resolution of 1 mm or less). If GBCA-free T1-weighted images for the higher axillary regions or GBCA-free T2-weighted images without or with fat suppression (Dixon with both water and fat suppression is considered) are required because of poor quality b0 images additional time may range up to 4-12 min for both sequences. This full protocol has a total duration of 8 to 27 minutes scanning plus patient’s time in and out scanner. To overcome those long scan times in toto that may limit the widespread use of breast MRI for all patients, protocols that shorten the image acquisition are appealing and called as abbreviated shortened.
Comparison Among the Different Breast MRI Clinical Protocols.a
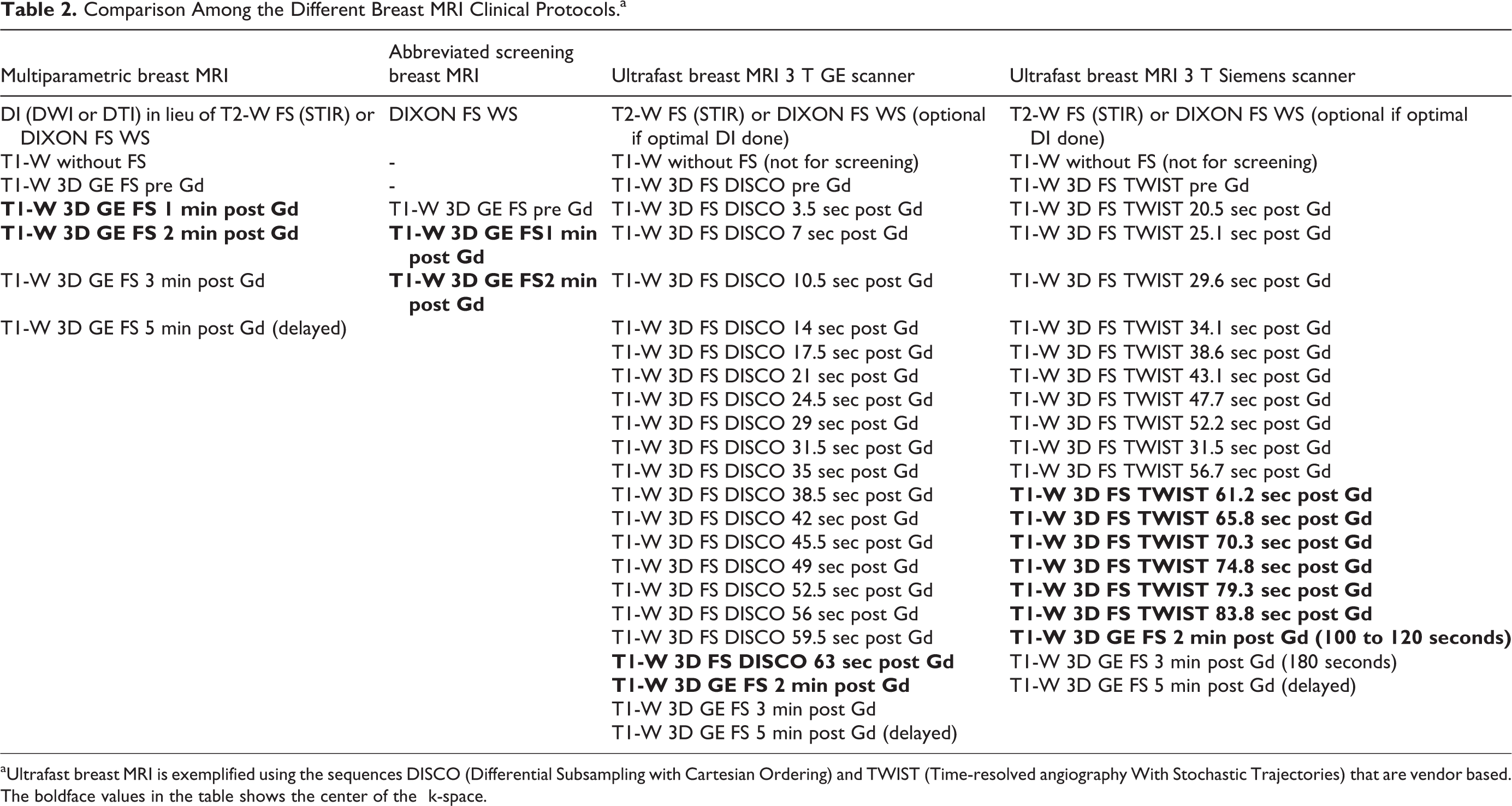
a Ultrafast breast MRI is exemplified using the sequences DISCO (Differential Subsampling with Cartesian Ordering) and TWIST (Time-resolved angiography With Stochastic Trajectories) that are vendor based.
The boldface values in the table shows the center of the k-space.
The abbreviated breast MRI (Table 2) was adapted in the ECOG/ACRIN trial (EA1141) 73 and is defined as having a total scan time of less than 10 min (including localizer) and including a T2 weighted sequence. The advantage of the abbreviated standard DCE-MRI techniques with or without adding the T2-weighted images components is to decrease the patient’s time in the scanner as well to improve the radiologist reading time with the goal of having an output of a simple binary answer to screening, yes or no presence of a potential actionable region of enhancement. Assuming absent enhancement equals negative screening, and a known high NPV with breast MRI data, a negative MIP image may lead to an interpreting time of less than 3 seconds, but caution is advised as some tumors may only be seen on the source images.
The “Ultrafast breast MRI” is composed of sequences that will shorten only the initial phase of the current DCE-MRI as the delayed phase is not usually done using this sequence (Table 2). It can be used with either diagnostic or screening Breast MRI, replacing the initial 0-60 seconds of the current DCE-MRI T1-weighted fat suppressed GE sequence that has one scan before contrast and 1 to 5 scans post contrast. The Ultrafast breast MRI generates consecutive images scanning using a fast temporal resolution after i.v contrast. There is no current determined optimal cut off value to the initial scanning and can be initiated at 3.5 seconds for 3 T magnets and 7 seconds for 1.5 T magnets keeping the slice thickness of 3 mm or less and the in-plane resolution of 1 mm or less.
Independent of using ultrafast temporal scanning, the classic Abbreviated MRI protocols, or the traditional protocols without fast temporal resolution breast DCE-MRI, it is recommended that post contrast image of maximum enhancement should have the center of the k-space (Figure 7) of ∼120 seconds post contrast administration. 69 A dynamic sequence demands at least 3 time points to be measured: (1) one before the administration of GBCA, (2) one approximately 90 to 120 seconds later to capture the peak and (3) one in the late phase 5-7 min post contrast to evaluate whether a lesion continues to enhance, will show a plateau or will show early wash-out of the contrast agent (decrease of signal intensity). One study of Rapid Abridged Multiphase Protocol (RAMP) shortened breast MRI protocol found that kinetic analysis was reliable if the late phase obtained at 3-4 min. 102 Some of the abbreviated MRI protocols that are solely based on MIP and one point after contrast do not allow determination of the enhancement curves. They are highly sensitive, but the specificity may decrease.73,114 In addition, generating MIP images leads to very fast lesion detection to the radiologist’s visual assessment, however chemical shift artifacts, suboptimal fat suppression and patient’s motion artifacts may cause poor MIP quality and obscure small lesions. For this reason, it makes sense to have an ideal breast MRI protocol that could contemplate both screening and diagnostic indications having the shortest occupation of the scanner by the patient without compromising the tests results. One group investigated the use of diffusion techniques as a substitute of the DCE-MRI delayed phase in a diagnostic study of patients with equivocal or suspicious US and/or mammography. 115 It was found that the initial enhancement rate (1 min post contrast) and ADC determined from DWI yield a higher AUC than a dynamic analysis with the late phase enhancement. Similar results were reported for a retrospective diagnostic study. 95 This approach could be explored more broadly and systematically in prospective studies although the shortening of DCE scan still requires patient time and facilities for the injection procedure.
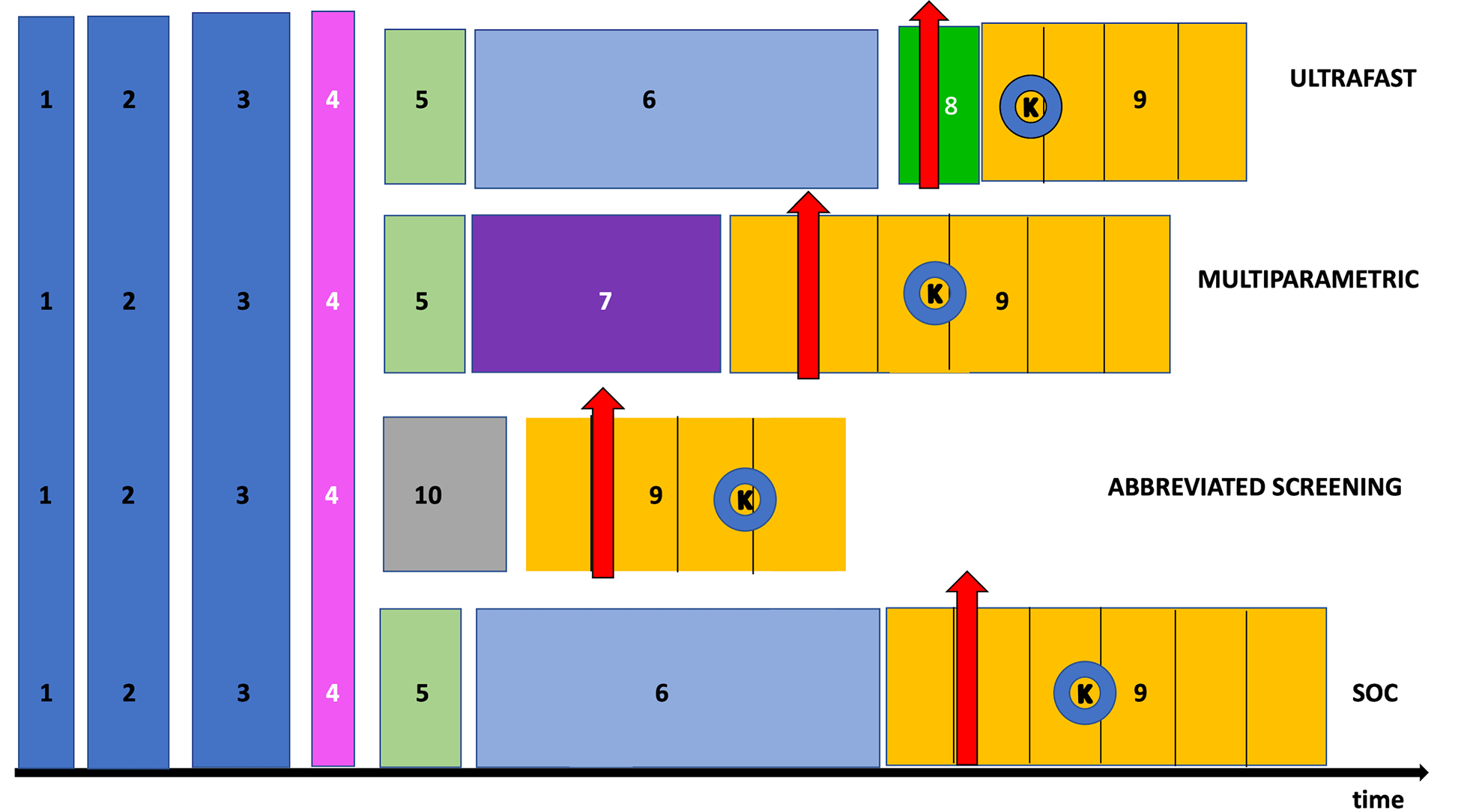
The acquisition breast MRI protocols with their sequences are compared based on the timeline. (1) Patient’s registration time. (2) MRI preparation 1: safety screening. (3) MRI preparation 2: venipuncture first, and then positioning on the table. (4) Scout. (5) T1-weighted without fat suppression (covering axillary regions). (6) T2-weighted with fat suppression (e.g. STIR, “FAT-SAT,” SSFP). (7) DWI or DTI. (8) Ultrafast. (9) DCE. (10) Fat suppressed and water suppressed sequences (e.g. DIXON). The red arrows represent the moment when the GBCA intravascular injection occurred. The letter K with circle in blue present in all DCE sequences (9) indicates the center of k-space.
Whether combining what is suggested as a feasible state of the art “shortened” breast MRI protocol would be a simple equation: “Ultrafast” + Diffusion is an open question. All MRI techniques continue to evolve, Ultrafast Breast MRI is meant to replace the initial phase DCE-MRI; Diffusion should be done instead of T2-weighted images and possibly the delayed phase of DCE-MRI. Images to be reported available as postprocessed subtracted and non-subtracted images with MIP reconstruction, DWI and ADC maps or DTI color coded maps (λ-1, λ-2, λ-3, MA, FA, MD) and DTI-MIP. Further research is required to determine the effects of this shortened protocol on clinical outcomes.
What Is Coming Up in Breast MRI
To conclude this review, I would like to point out that other areas that are not MRI exclusive are the fields of artificial intelligence, machine learning tools and radiomics that will almost certainly facilitate the rapid improvement of clinical practice and consequently better patient care.116-119 Breast radiologists shall expect that the use of DeepContrast AI algorithms in gadolinium-free breast MR images may be a reliable to tool to predict the contrast-enhancement completing the breast MRI history circle-of-life when i.v. contrast was not used.
Footnotes
Acknowledgments
This paper is dedicated to the memory of my friend, Dr Pavel, who died, tragically, in 2016. It is gratifying that I have been able to honor him with a review which substantially overlaps with his research interest in concise breast MRI techniques. I would like to thank Professor Hadassa Degani from her solid scientific knowledge of gadolinium distribution in the breast, her mentorship and collaboration with the advances of sophisticated non-invasive, safe, gadolinium-free MRI techniques and for bringing to my attention gaps in my knowledge. A special word of gratitude to Dr Edna Furman-Haran, who has read and commented about this manuscript. I am also grateful to Ms Lina Tham and Mrs Eva Markus to their assistance with the data acquisition. The completion of this undertaking could not have been possible without the participation and assistance of so many people whose names may not all be enumerated. Their contributions are sincerely appreciated and gratefully acknowledge. Finally, I would like to thank all the women that provided their anonymous images to illustrate the article and the medical imaging technologists that acquired them. This work was not supported by a specific grant, but I would like to thank to JDMI for priors dedicated academic time support received.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
