Abstract
Introduction
Contrast-enhanced breast MRI (CE-MRI, referred hereafter as MRI) is often used after breast cancer diagnosis but before surgery to detect additional breast lesions or provide additional information on disease distribution or extent to guide surgery or systemic therapy. Sensitivity and specificity are generally >90% in recent studies and sometimes reported >97%.1,2 The BI-RADS benchmark for sensitivity in MRI screening is 85 to 90%. 3 While it is generally acknowledged that preoperative breast MRI will detect additional lesions, there is controversy as to whether detecting these lesions improves patient outcomes.
The objective of this systematic review is to summarize the evidence on whether additional information on disease extent obtained by use of preoperative breast MRI after mammography and/or ultrasound in patients with newly diagnosed breast cancer changes the relative use of breast conserving surgery (BCS) vs mastectomy or improves rates of recurrence, survival, re-excision, and early detection of bilateral cancer. The systematic review protocol was registered on PROSPERO. 4 This systematic review and accompanying clinical practice guideline5,6 were sponsored by Ontario Health (Cancer Care Ontario) to provide guidance on the use of preoperative breast MRI.
Methods
Ethics approval
Informed consent and ethics approval were not required as this systematic review is based on published studies.
Literature search
Embase, MEDLINE, Cochrane Central Register of Controlled Trials, and Cochrane Database of Systematic Reviews were searched until July 3, 2019 and updated until January 18, 2021 (see Supplementary Data). A targeted search was conducted in July 2022 to identify any additional publications related to the included RCTs and studies identified as ongoing. Articles with terms for both breast cancer and MRI were evaluated against the following inclusion/exclusion criteria. Relevant studies not found in the database search but mentioned in other trial reports or systematic reviews were also examined. A review of the titles and abstracts was done by 1 reviewer (GGF). For studies that warranted full-text review, the same author reviewed each study. In cases of uncertainty, co-authors were consulted.
Inclusion criteria
Studies that met all the following criteria were included: 1. Included patients with newly diagnosed breast cancer evaluating use of breast MRI prior to surgery, or patients referred for biopsy due to suspicion of breast cancer (but not yet diagnosed with cancer). 2. Were either (a) a randomized controlled trial (RCT) of MRI vs no MRI (≥30 patients per group) or (b) a comparative study with ≥100 patients comparing use of MRI vs no MRI in 2 or more groups with equivalent disease and patient characteristics or using methods such as multivariable analysis or propensity score matching to control potential confounders. As a minimum, studies should have considered stage and patient age and/or menopausal status. Cancer subtype/histology was considered important for outcomes of margin status, reoperations, recurrence, and survival. Systemic therapy, radiotherapy, and HER2 and ER/PR status were also considered important for recurrence/survival outcomes. 3. Primary or secondary outcomes included at least 1 of the following: recurrence; survival outcomes such as disease-free survival (DFS), event-free survival (EFS), distant metastasis-free survival (DMFS), or overall survival (OS); rates of mastectomy, re-excision, or re-operation; adverse effects/morbidity due to surgery; or quality of life.
Exclusion Criteria
Studies of the following were excluded: 1. MRI as the initial screening or diagnostic test, or when no index cancer was previously identified (occult cancer). 2. MRI as a tool to monitor response to neoadjuvant treatment. 3. Studies reporting on performance characteristics of MRI (eg, sensitivity, specificity) or detection rates of multicentric, multifocal, or contralateral breast cancer (CBC), but without outcomes listed in the inclusion criteria.
Data Extraction, Assessment of Risk of Bias, and Trial Quality
Studies underwent data extraction by 1 author (GGF), with independent verification by a student assistant. Study name and location, number of patients, patient demographics, study design, disease stage/subtype or other inclusion criteria, MRI details, and outcome data were extracted for all included studies. Odds ratios (OR) or hazard ratios (HR) were expressed with a ratio of <1.0 indicating that the experimental group (MRI use) had more favourable outcome than the control group, a ratio of >1.0 indicating worse outcome with MRI, and a ratio of 1.0 indicating no difference between groups. The exception to this was the case of synchronous CBC detection (identified at the same time or sometimes defined as occurring within 6 months of the index cancer). Higher detection is considered a favourable outcome, but the convention is to report increased detection with HR >1.0. The risk of bias for randomized studies was assessed per outcome and per study using the Cochrane risk-of-bias (RoB) tool (revised version RoB2) for RCTs and ROBINS-I for non-RCTs as outlined in the Cochrane Handbook for Systematic Reviews of Interventions. 7 For RCTs, factors considered in the assessment of bias were randomization process, deviations from planned interventions, missing outcome data, measurement of the outcomes, and selective reporting of results. For non-randomized studies, risk of bias assessment included evaluation of confounding, selection bias, classification bias, departure from planned interventions, missing data, measurement of outcomes, and selection of results to report. The RoB2 tool is available as an MS Excel-based form at https://www.riskofbias.info/.
Synthesizing the Evidence
When clinically homogeneous results from 2 or more studies were available, a meta-analysis was conducted using Review Manager 5.4 software 8 using a generic inverse variance model with random effects. Analysis was conducted by a Health Research Methodologist at the Program in Evidence-Based Care (PEBC) familiar with meta-analysis in consultation with others at PEBC with statistical expertise. Multiple logistic regression calculates adjusted ORs and these were used for short-term outcomes. 9 For time to event data, HRs were used. For RCTs or studies with matched/propensity-score matched groups, if ORs or HRs and CIs were not reported they were derived from event rates or P-value. For retrospective studies with multivariate analysis to adjust for confounding, only outcomes with adjusted ORs were reported. Additional sensitivity analysis was conducted excluding cancer registry data, or excluding 2 RCTs with high risk of bias for non-mastectomy outcomes. Subgroup analysis was conducted for categories of in situ disease, invasive disease, and ILC; RCTs and non-RCTs; and subtypes of recurrence (local, locoregional, ipsilateral, or combination of these 3 subtypes, and distant recurrence). The GRADE approach (see https://www.gradeworkinggroup.org/) was used to rate the overall quality of evidence (also referred to as strength of evidence or certainty of evidence). The GRADEPro guideline development tool 10 was used to determine the GRADE rating and present the results. Assessment of certainty of evidence takes into account the risk of bias along with inconsistency, indirectness, imprecision, and other factors such as a strong association (large/very large effect).
Results
A PRISMA diagram showing the search results is provided as Figure 1. Study characteristics of 51 trials meeting the inclusion criteria11-65 are reported in Table 1; Table 2 contains a summary of the ORs or HRs calculated for each outcome. Selected forest plots are included as well. The Supplementary Data contains the full evidence tables, additional forest plots, risk of bias assessment, and GRADE profiles. Included are 8 RCTs, 1 prospective cohort study, and 42 retrospective studies. The patient population was limited to those with an initial treatment plan of BCS in 17 trials (6 RCTs). The retrospective studies included 8 with propensity-matched controls, 4 with historical or equivalent controls, 15 with multivariable/multivariate analysis of data from a single or small number of institutions, and 15 using cancer registry data and multivariable/multivariate analysis. There is also a table of excluded studies that compared outcomes with vs without MRI but were judged to have insufficient matching/adjustment for confounders. A series of forest plots created using RevMan
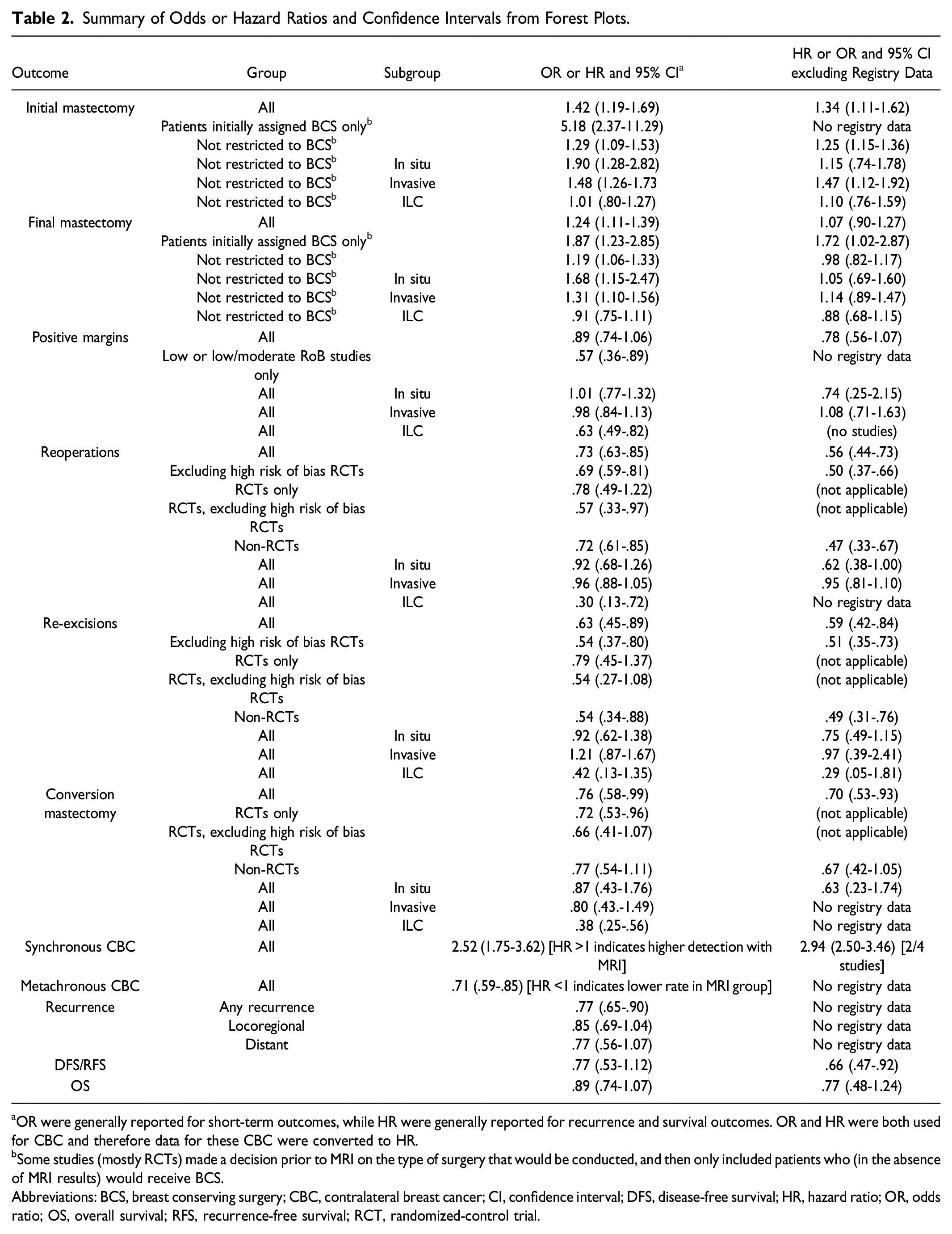
8
provide graphical summaries to aid in the interpretation of the tabulated results. PRISMA Flow Diagram. Characteristics of Included Studies. Abbreviations: ACRIN, American College of Radiology Imaging Network; BCS, breast-conserving surgery; BCT, breast-conserving therapy (BCS + RT); BI-RADS, Breast Imaging and Reporting and Data System; CPM, contralateral prophylactic mastectomy; DCIS, ductal carcinoma in situ; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; IBTR, ipsilateral breast tumour recurrence; IBC, invasive breast cancer; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; MRI, magnetic resonance imaging; pts, patients; PR, progesterone receptor; PSM, propensity score matching; RT, radiotherapy; SEER, Surveillance, Epidemiology, and End Results database. Summary of Odds or Hazard Ratios and Confidence Intervals from Forest Plots. aOR were generally reported for short-term outcomes, while HR were generally reported for recurrence and survival outcomes. OR and HR were both used for CBC and therefore data for these CBC were converted to HR. bSome studies (mostly RCTs) made a decision prior to MRI on the type of surgery that would be conducted, and then only included patients who (in the absence of MRI results) would receive BCS. Abbreviations: BCS, breast conserving surgery; CBC, contralateral breast cancer; CI, confidence interval; DFS, disease-free survival; HR, hazard ratio; OR, odds ratio; OS, overall survival; RFS, recurrence-free survival; RCT, randomized-control trial.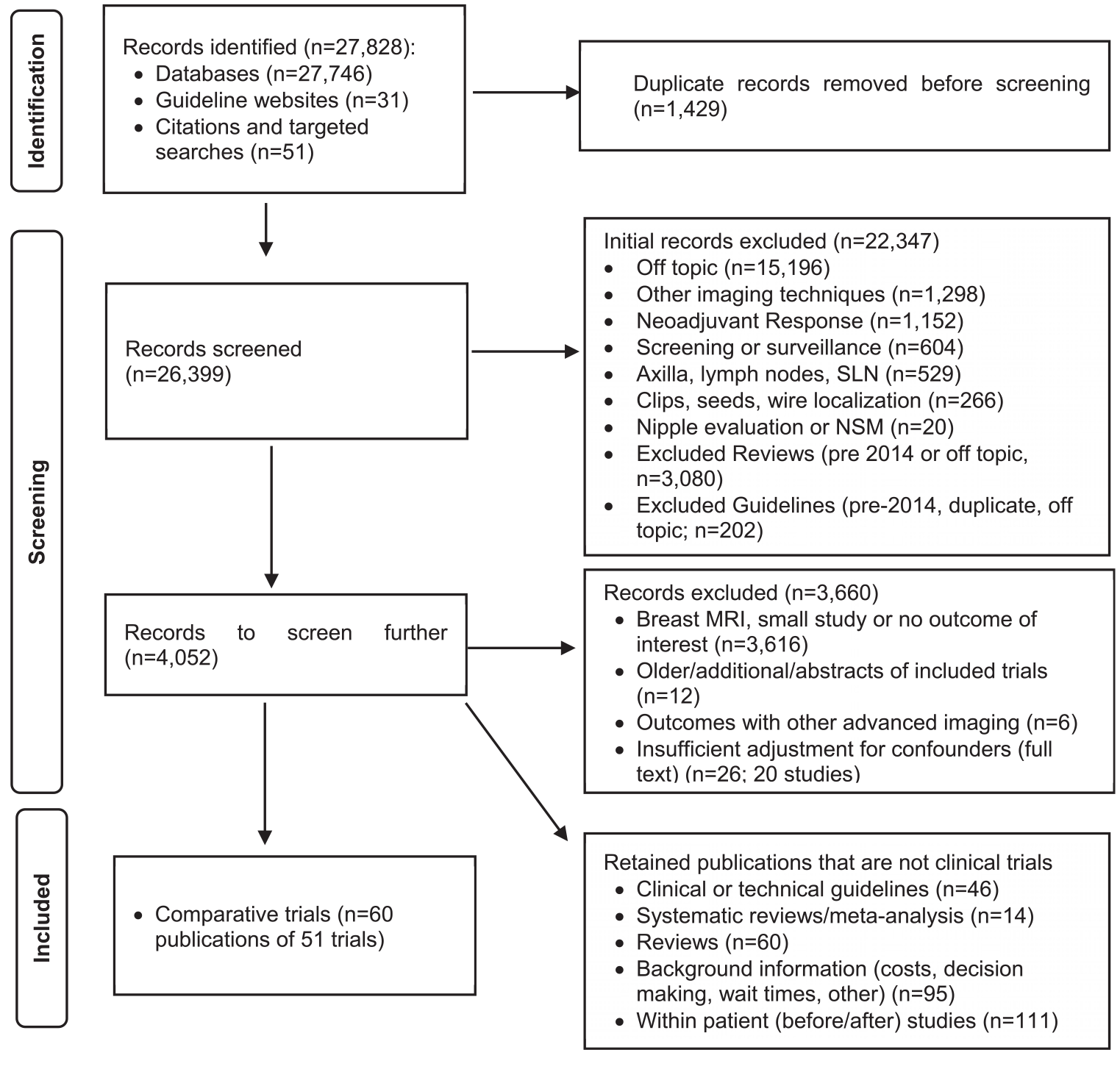

Risk of bias and quality of evidence
Randomized controlled trials
While well-conducted RCTs generally comprise the highest level of trial evidence, the RCTs in this review have limitations that affect their generalizability. Of the 6 RCTs that reported mastectomy rates, all had high risk of bias and certainty of evidence was very low for this outcome (Supplementary Data Tables S-D1 and S-E1) Studies that restricted the patient population to only patients with a treatment plan of BCS have a high risk of bias for mastectomy rate outcomes, leading to an overestimation of the effect of MRI on increasing mastectomy rates. IRCIS, 11 Turku University, 12 BREAST-MRI, 15 COMICE 18 and the ongoing Alliance AO11104/ACRIN 6694 20 were conducted in patients preselected for BCS.
Other sources of bias apply to all outcomes. IRCIS 11 had more patients with dense breasts and premenopausal status in the MRI arm, the BREAST-MRI trial 15 was only reported as an abstract and had more premenopausal patients in the MRI arm, and COMICE 18 had more patients with multifocal and multicentric tumours in the MRI arm. COMICE also had 53 patients in the MRI group who did not receive MRI compared to 9 in the non-MRI group who received MRI; results were not adjusted to account for this protocol violation. COMICE categorized initial mastectomy due to patient choice alone as reoperation, and those lost to follow-up as not having a primary endpoint event. The other 2 RCTs with mastectomy outcomes also had high risk of bias. POMB13,14 randomized some patients only after multidisciplinary team discussion, groups had unequal baseline characteristics (prior to MRI, the suggested treatment was BCS in 153 vs 132 patients), 10 patients without MRI were analyzed together with the MRI group, 1 out of 3 study sites did not perform MRI, and 1 site did not follow conventional MRI procedures. MONET 16 randomized 463 patients with suspicious lesions (and conducted power calculations based on this) but only 149 had surgery and therefore the study was greatly underpowered; there are also concerns about the MRI technique as sensitivity was only 51% and prior to the start of the trial there were substantial problems that the investigators thought were resolved. B-SMART 17 was terminated early, reported interim results only in an abstract, and gave no MRI details and is therefore also at high risk of bias.
Non-Randomized studies
Some non-randomized studies may provide similar or greater levels of evidence than RCTs that have high risk of bias. Retrospective studies that were restricted to BCS candidates have the same bias as RCTs with this restriction and were judged as having serious or critical risk of bias for mastectomy outcomes. For other outcomes, these studies may be grouped with the other non-randomized trials.
Retrospective studies are of variable risk of bias, depending primarily on how well the patient and disease factors were matched in the MRI and non-MRI groups, or how adequate the correction for confounders was made in the multivariable analyses. The most rigorous studies controlled for many disease characteristics (size or stage, subtype or histology) and physical characteristics of patients (age, menopausal status, breast density); studies with obvious imbalance and failure to control for key factors (see inclusion criteria) have been excluded. However, the number of factors measured, reported, and corrected for varied widely; in general, those with more factors in the matching or multivariate analysis have less risk of bias. Provided they included the key factors, such studies have low risk of bias for confounding. Some studies followed a controversial practice of only including factors with significant correlation (e.g., P < .05) in the multivariate analysis. Such studies were excluded if they did not adjust for key factors and are listed in Supplementary Data Table S-B5; otherwise, they are noted in the data tables and have high risk of bias. Evaluation of individual included studies is provided in the Supplementary Data Table S-D2.
Most studies did not have data available as to why patients elected mastectomy. Only in those studies with historic controls26,30-32 from the time period immediately before MRI was implemented are patient decision factors expected to be more similar in the MRI and non-MRI groups, although change in practice might still be an issue. One other study compared results for 2 surgeons in the same institution and with similar surgical practices. 31 In this trial patient and disease characteristics appeared equivalent and it is expected that decision factors were also similar. These studies are assessed as having low to moderate risk of bias. The remaining non-randomized trials have moderate to critical risk of bias for mastectomy outcomes.
Studies in which data was extracted from patient records in single or a small group of institutions tended to have better reporting of MRI and subsequent biopsy procedures and therefore generally had low risk of bias related to MRI techniques or reporting (with a few concerns noted in the data tables). Studies using registry data had inadequate documentation of MRI methodology and are considered to have serious risk of bias for all outcomes.
Mastectomy rates
Trials reporting initial mastectomy or BCS rates in patients with or without preoperative MRI are presented in Figure 2; further information is provided in Supplementary Data. Figure 2(a) reflects the fact that for patients selected for BCS prior to MRI, MRI cannot reduce mastectomy rates. The OR for initial mastectomy with vs without MRI is 5.18 (95% CI = 2.37 to 11.29) in the studies of BCS patients only (as determined prior to MRI), and 1.29 (95% CI = 1.09 to 1.53) in studies that did not restrict the study to BCS. Figure 2(b) groups the results according to subtype of cancer. MRI appears to have no overall effect on mastectomy rates in ILC (OR = 1.01, 95% CI = .80 to 1.27) but increases rates in other subgroups (in situ OR = 1.90, 95% CI = 1.28 to 2.82; invasive OR = 1.48, 95% CI = 1.26-1.73). MRI does not influence mastectomy rates in studies with equivalent or historic controls. Forest plots for initial mastectomy rate. (A) By type of surgery determined prior to MRI. (B) By subtype (excluding studies assigning all patients to BCS prior to MRI). Abbreviations: BCS, breast conserving surgery; DCIS, ductal carcinoma in situ; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma MRI, magnetic resonance imaging; RCT, randomized controlled trial.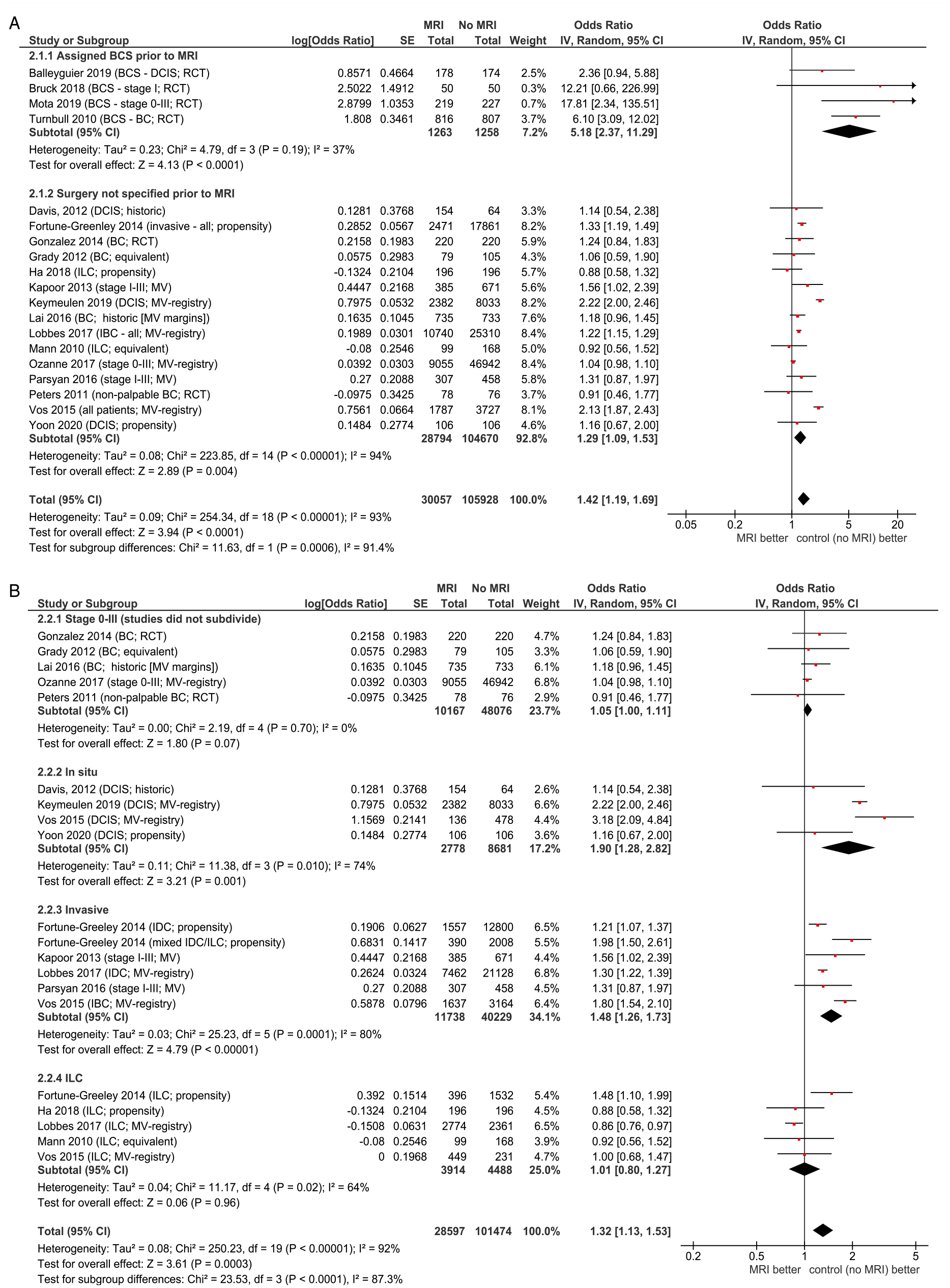
While trends are similar, the ORs are lower for final mastectomy than for initial mastectomy for most of the subgroups analyzed, indicating that in patients without MRI there is more conversion from initial BCS to final mastectomy than when MRI is initially performed. This effect is most evident in RCTs limited to patients whose treatment was determined to be BCS prior to MRI; the odds ratio was 5.18 (95% CI = 2.37 to 11.29) for initial mastectomy and 1.72 (95% CI = 1.02 to 2.87) for final mastectomy. Similar trends were found when dividing by cancer subtype. In trials not limited to predetermined BCS, the effect of MRI on both initial and final mastectomy rates was similar (OR = 1.29, 95% CI = 1.09 to 1.53 and OR = 1.19, 95% CI = 1.06 to 1.33, respectively).
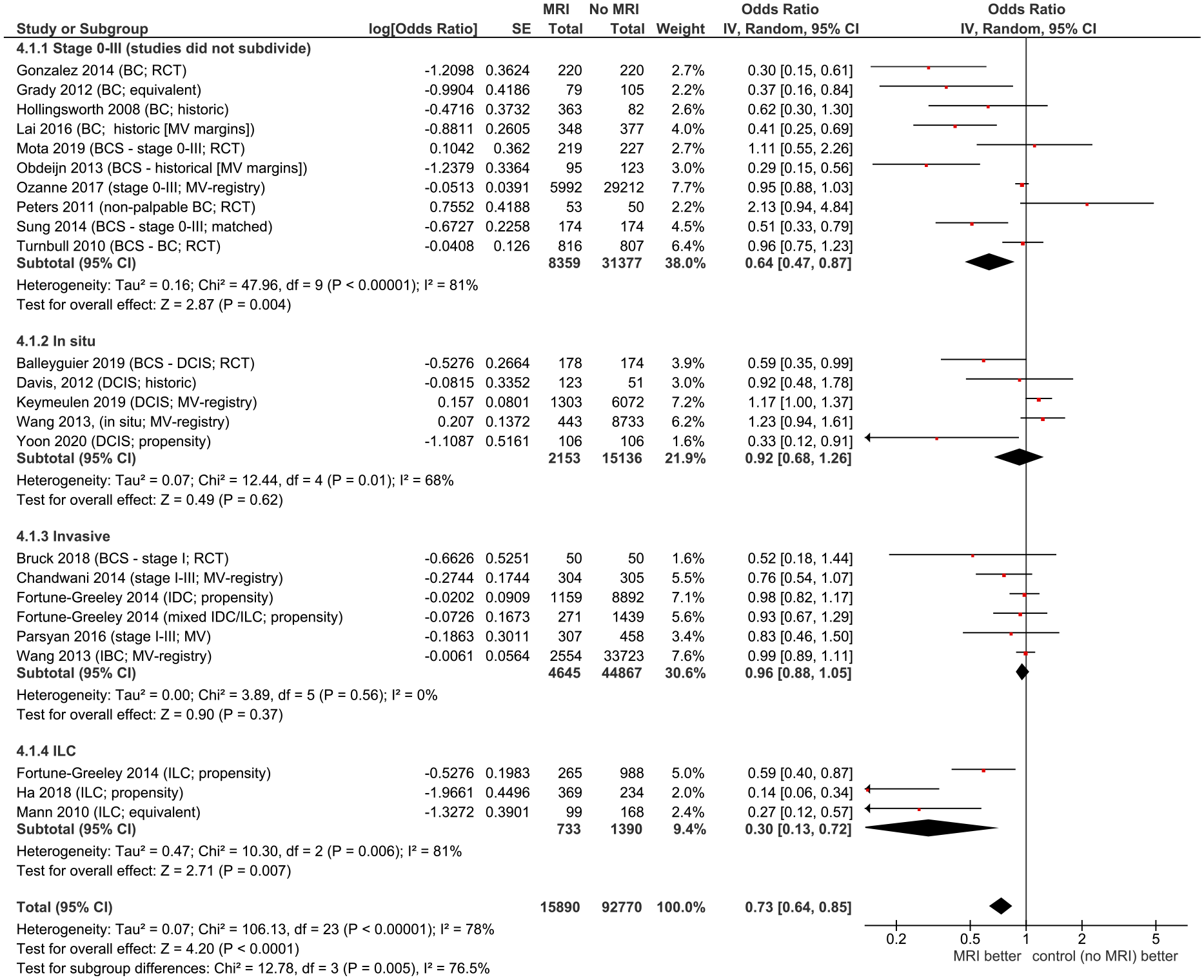
Positive margin and reoperation rates
Positive margins often result in reoperation (re-excision or conversion to mastectomy). Figure 3 summarizes positive margin rates and Figure 4 summarizes reoperations. MRI appears to decrease rates of positive margins (overall OR = .89, 95% CI = .74 to 1.06), reoperations (OR = .73, 95% CI = .63 to .85), and re-excisions (OR = .63, 95% CI = .45 to .89). Definitions of close or positive margins and when these should result in reoperation varied among studies. Forest plots for rates of positive margins. Abbreviations: BCS, breast conserving surgery; DCIS, ductal carcinoma in situ; IBC, inflammatory breast cancer; MRI, magnetic resonance imaging; RCT, randomized controlled trial. Forest plot for reoperations by stage/subtype. Abbreviations: BCS, breast conserving surgery; DCIS, ductal carcinoma in situ; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; MRI, magnetic resonance imaging; RCT, randomized controlled trial.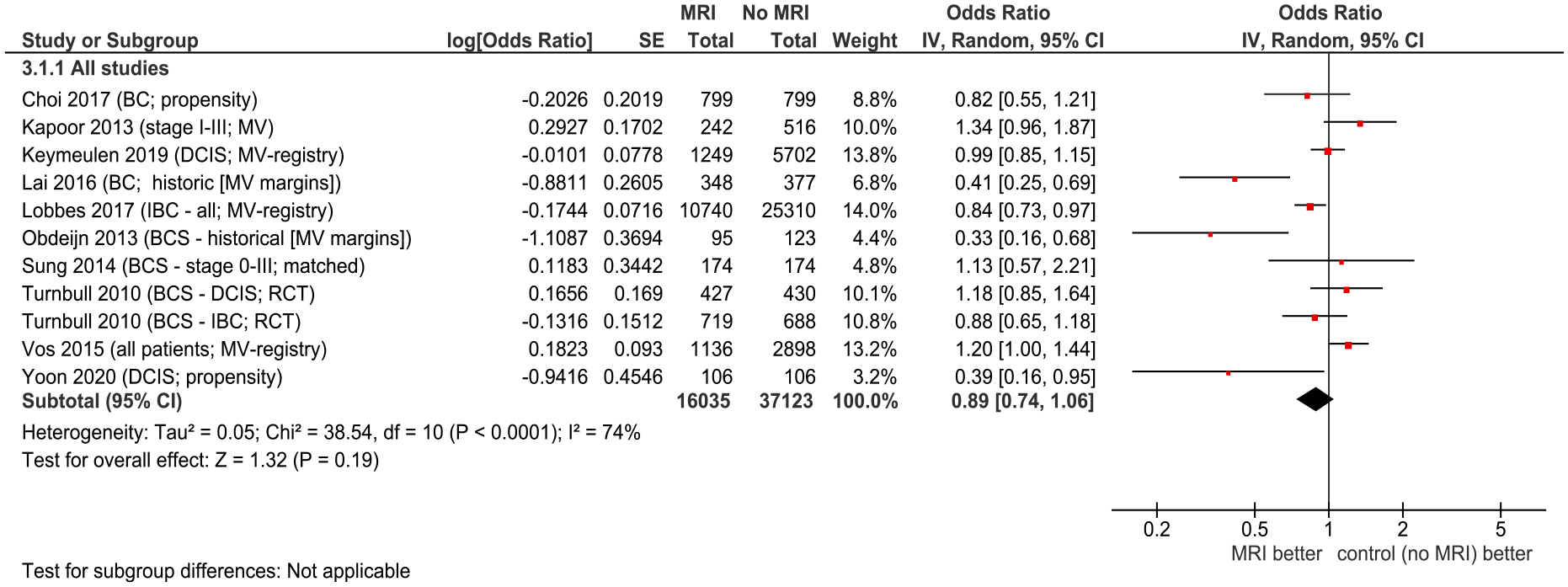
Contralateral breast cancer, recurrence, and survival outcomes
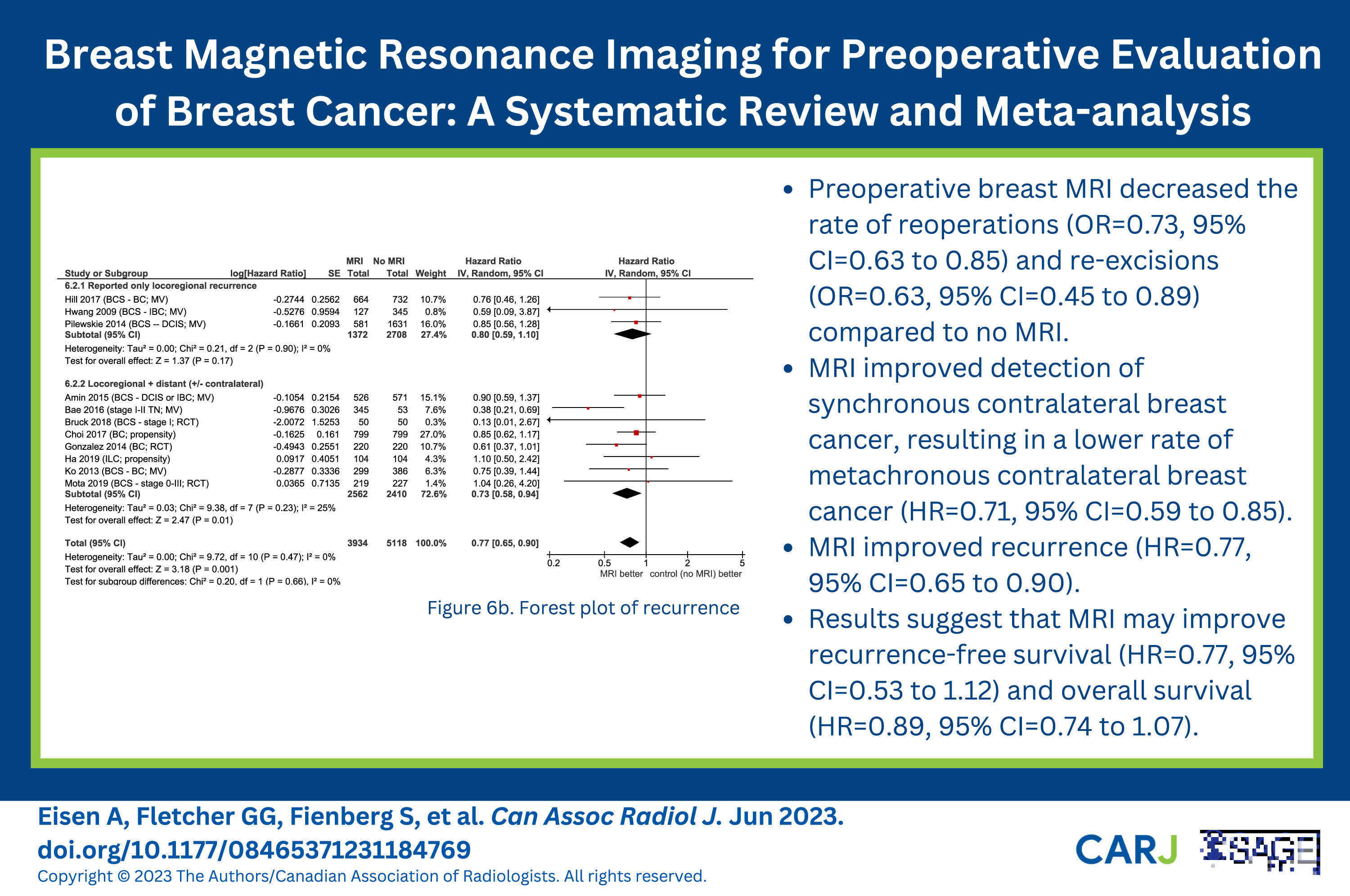
Table S4 includes 22 trials with outcomes of synchronous or metachronous CBC, recurrence, or survival. Figure 5 illustrates that MRI improved detection of synchronous CBC (HR = 2.52, 95% CI = 1.75 to 3.62). Rates of metachronous breast cancer were lower with preoperative MRI overall (HR = .71, 95% CI = .59 to .85). Forest plots for contralateral breast cancer. (A) Synchronous. (B) Metachronous. Abbreviations: BCS, breast conserving surgery; DCIS, ductal carcinoma in situ; ILC, invasive lobular carcinoma; MRI, magnetic resonance imaging; RCT, randomized controlled trial.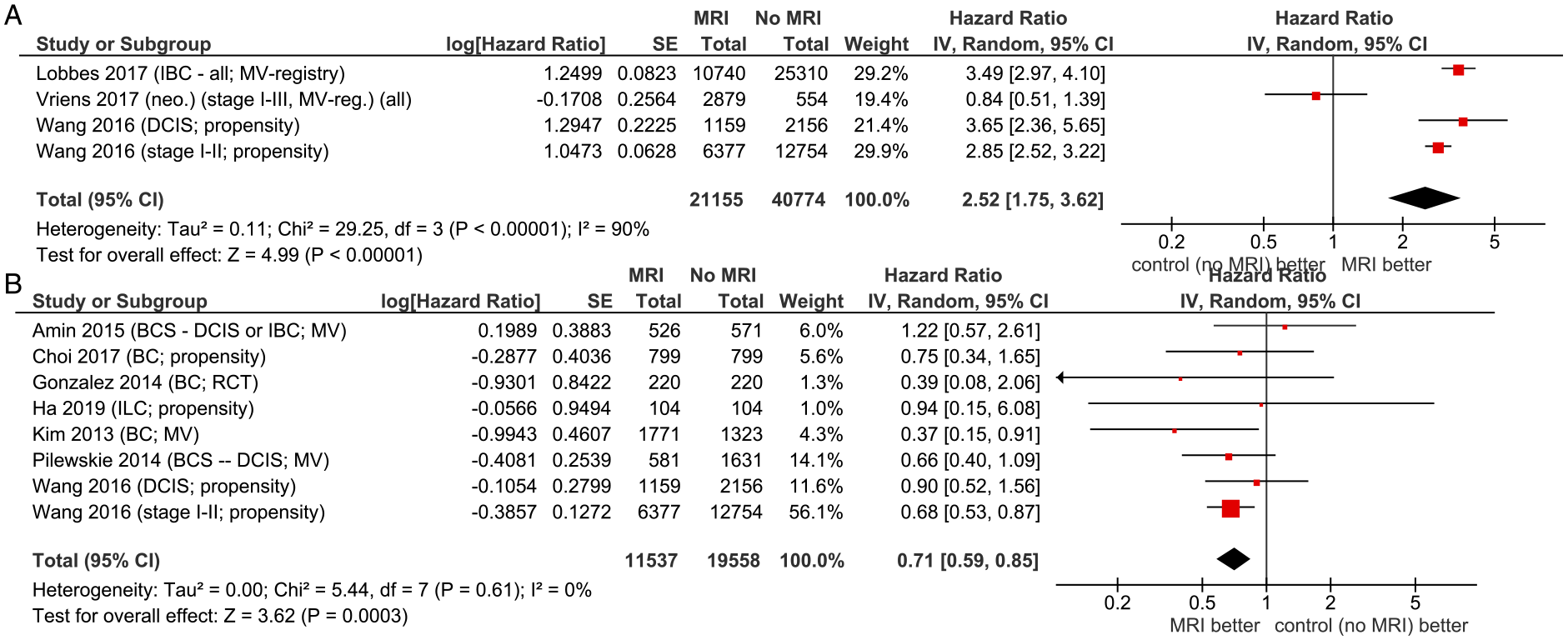
Measures of recurrence and survival used varied among studies (see Table S4 and Figure 6). MRI improved recurrence (HR = 0.77, 95% CI = 0.65 to 0.90). When data was subdivided, HR = .85 (95% CI = .69 to 1.04) for locoregional recurrence and HR = .77 (95% CI = .56-1.07) for distant recurrence. Results suggest that MRI improves RFS (OR = .77, 95% CI = .53 to 1.12) and overall survival (OR = .89, 95% CI = .74 to 1.07), although likely due to low numbers of events these were not statistically significant. Forest plots for survival or recurrence. (A) Recurrence by recurrence type reported. (B) Recurrence (any type). (C) Recurrence-free survival/disease-free survival. (D) Overall survival. Abbreviations: BC, breast cancer (not otherwise specified); BCS, breast conserving surgery; DCIS, ductal carcinoma in situ; IBC, inflammatory breast carcinoma; ILC, invasive lobular carcinoma; MRI, magnetic resonance imaging; MV, multivariate analysis; RCT, randomized controlled trial; TN, triple-negative breast cancer.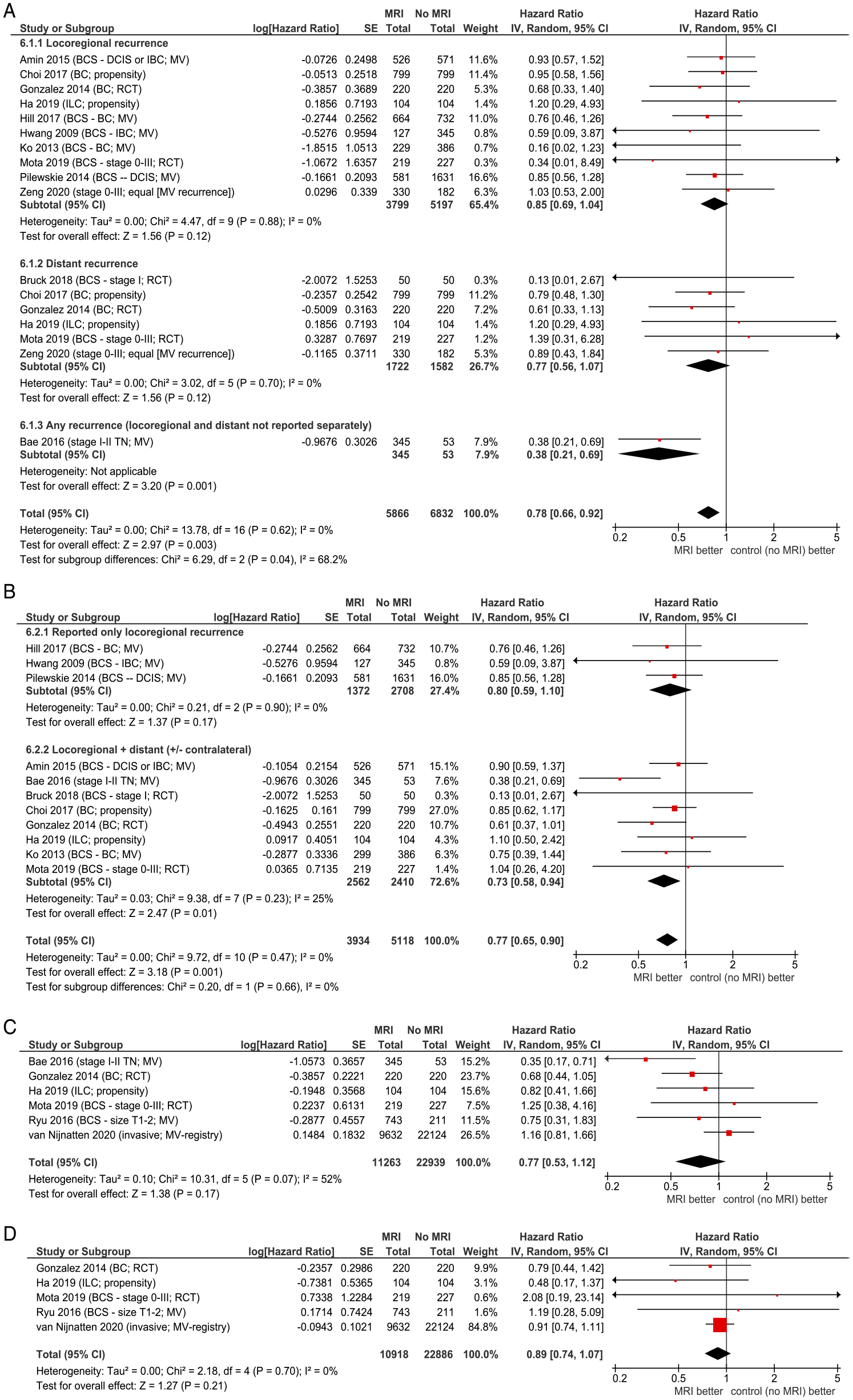
Ongoing, unpublished, or incomplete studies
Ongoing RCTs and prospective studies have been included in the data tables. RCTs include the ACRIN 6664/Alliance A011104 20 and Breast-MRI 15 trials, and the B-SMART trial that was terminated but reported interim data. 17
Discussion and Conclusions
This systematic review compiled a comprehensive set of data from trials comparing patient outcomes with and without preoperative MRI. Data from RCT trials was of limited usefulness as all had some deficiencies with a high risk of bias and evidence was considered moderate to low quality. Evidence from non-randomized trials was determined to be of similar quality and therefore evidence from all trial types was combined. A strength of this approach is that a much larger number of trials informs the observations and conclusions, and consistency among various designs leads to stronger overall evidence.
The outcome of mastectomy rates is commonly reported but of limited use in determining whether MRI should be used. There were serious limitations to the data for this outcome: several trials preselected only patients who were scheduled for BCS, patients may have received MRI for specific but non-recorded reasons related to tumour characteristics or patient factors, and mastectomy decisions are often not due to medical necessity as exemplified by differences in mastectomy rates according to surgeon, institution, ethnic, and socioeconomic factors. Some of the trials that reported increased rates of mastectomy with MRI did not confirm whether the additional lesions were benign or cancerous; analysis of mastectomy specimens suggested that a change in some surgical plans (extent of surgery) was not indicated clinically. Best practice is that additional suspicious lesions be biopsied or otherwise confirmed if they could alter surgical procedures. Sites performing MRI should have the capacity for biopsy to both reduce surgical delays and improve quality. Sites should also monitor their quality indicators for performance of MRI. Familiarity with the complete process may result in better expertise in reading and interpreting MRI images as well as dedication to advances in the field. 33
Other outcomes such as positive surgical margins, reoperations, recurrence, and survival, are less influenced by non-clinical patient factors. With OS >95% in several studies, an extremely large number of patients would be needed to measure a difference in survival due to MRI. Advances in systemic therapy and radiotherapy, and the ability to re-treat recurrence have made survival an outcome not expected to be altered with preoperative MRI, and as indicated in Figure 6 no difference in OS was found. Recurrence outcomes are more sensitive, and a decrease in recurrence in patients with MRI was found.
The remaining outcomes, namely positive surgical margins and reoperations (including re-excisions and conversion to mastectomy), are those for which additional information obtained from imaging prior to surgery is likely to make the most difference. The literature review found that MRI use resulted in a reduction in reoperations. Reoperation may delay adjuvant treatment, result in poorer cosmetic outcome, cause emotional distress, increase recovery times, and be a financial burden to the health care system and patients. 66 The American Society of Breast Surgeons indicates that “a goal of breast cancer care is to minimize the number of operations a patient requires in order to optimize their oncologic outcomes and minimize their local recurrence”. 67 It has been proposed that the goal should be a single surgery32,33 and more than 1 re-excision should not be necessary for most patients. The United Kingdom National Health Service Breast Screening Programme target is that the reoperation rate for incomplete excision should not be more than 10%.18,19 EUSOMA set a minimum standard (quality indicator) of 80% and target of 90% for proportion of patients with invasive cancer that should receive a single breast operation (excluding reconstruction); for DCIS standards were 70% and 90%. 68 While this was achieved in some studies in the current review, such as the 1 by Hollingsworth32,33 in which rates dropped from 12% to 15% prior to MRI implementation to 9% afterwards, most did not. Better information upfront could allow more BCS including oncoplastic surgery and multiple lumpectomies without conversion mastectomy, or preplanned mastectomy with a wider range of reconstruction options including skin and nipple-sparing procedures.
A common theme in many of the publications was the high rate of CBC detected by MRI but not mammography. Another meta-analysis of 22 studies found the incremental CBC detection rate over conventional imaging to be 4.1%. 69 Some studies suggest most CBC are second primary cancers. 70 The mammographically occult CBC is sometimes larger or has a worse prognosis than the initial cancer detected. While chemotherapy or other systemic therapy may help with the CBC, not all patients receive systemic therapy, and none would receive the standard treatment of radiation therapy. Some have suggested that in cases where the contralateral tumour is larger or more advanced than the index tumour, failing to detect and treat the contralateral tumour could be considered inappropriate operation.
Mammographically occult ipsilateral lesions are larger than the index lesion in about 20% of cases 71 and unless detected coincidentally during operation of the index tumour would be untreated surgically. While whole breast irradiation would provide some treatment, partial breast irradiation would be inadequate.
While the degree of benefit appears small for most outcomes and not always statistically significant, for all outcomes (except mastectomy rates) the trend is towards MRI being beneficial for each outcome, and therefore this consistency strengthens the conclusion that preoperative MRI has a positive impact in general. It is recognized that this may be higher for patients with certain characteristics such as high breast density or risk factors of multifocal or multicentric cancer and may be lower for patients with a single small well-defined lesion and no other risk factors. While these factors are of interest, data were not available for them in the comparative studies meeting the review inclusion criteria. As seen in the results, the evidence for benefit of preoperative MRI is stronger for ILC than for the overall data. This is consistent with results of studies that reported that, compared to invasive ductal carcinoma, ILC has been found more difficult to detect by mammography, is more likely multifocal, more likely to have synchronous CBC, and has a higher rate of involved margins after initial resection.72-74 There was insufficient information to report on other subtypes of cancer separately.
These outcomes were measured in a large number of studies and with data available for most patients. Included studies also indicate an increase in synchronous CBC and corresponding decrease in metachronous CBC; this is consistent with more rigorous studies designed specifically to investigate CBC. MRI resulted in a decrease in rates of recurrence of any type. Subtype of recurrence was not reported consistently among the studies and therefore subgroup information was limited and therefore less likely to be statistically significant due to low number of participants and events for each outcome. Due to the long follow-up required, potential to retreat patients upon recurrence, and generally high survival in early breast cancer, survival outcomes are less sensitive to interventions than the other outcomes. Overall survival results were only available from 5 studies, of which 4 suggest there may be a small (not statistically significant) OS benefit of MRI; the combined data for OS were similar with and without MRI (OR = .90, 95% CI = .75 to 1.09). RFS or DFS was also reported in a small number of studies and with lower number of events than for short-term outcomes that could be measured in all patients. As RFS or DFS includes a component of recurrence, these results are similar to recurrence results. Lastly, real-world issues with MRI availability were not assessed, and it is recognized that many Canadian centres lack timely MRI access.
In conclusion, this systematic review indicates a benefit of MRI in reducing positive margins and reoperations, decreasing rates of recurrence, and improving synchronous CBC detection.
Supplemental Material
Supplemental Material - Breast Magnetic Resonance Imaging for Preoperative Evaluation of Breast Cancer: A Systematic Review and Meta-Analysis
Supplemental Material for Breast Magnetic Resonance Imaging for Preoperative Evaluation of Breast Cancer: A Systematic Review and Meta-Analysis by Andrea Eisen, Glenn G. Fletcher, Samantha Fienberg, Ralph George, Claire Holloway, Supriya Kulkarni, Jean Seely, and Derek Muradali in Canadian Association of Radiologists Journal
Footnotes
Acknowledgements
The authors thank Emily Vella, Sarah Kellett, Jonathan Sussman, Cindy Walker-Dilks, and Caroline Zwaal for providing feedback on draft versions and Megan Smyth for conducting a data audit.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In accordance with the PEBC Conflict of Interest Policy, the authors were asked to disclose potential conflicts of interest. JS declared >$500 as Consultant to Hoffman Roche in 2018 in an advisory capacity and was site principal investigator for the TMIST (Tomosynthesis Mammography Intervention Screening Trial) in Ottawa, funded by National Cancer Institute, to the Canadian Clinical Trials Group. RG declared consultant fees, grants for topics unrelated to this review.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ontario Health (Cancer Care Ontario ongoing operating funds, no specific grant/award). This work is editorially independant of the funder.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
