Abstract
Atopic dermatitis (AD) is symptomatically worse in the evening, but the mechanism driving nocturnal eczema remains elusive. Our objective was to determine the circadian rhythm of skin barrier function measured by transepidermal water loss (TEWL) in AD patients and explore the molecular underpinnings. A pilot study was performed on a diverse group of AD (n = 4) and control (n = 2) young patients. We used an inpatient tightly controlled, modified, constant routine protocol. TEWL was measured at least every 90 min in the antecubital fossa (lesional) and forearm, while whole blood samples were collected every 4 h. Results show a significant difference in the antecubital fossa TEWL in the AD group versus controls. TEWL in control skin decreases starting a few hours prior to bedtime, both in the antecubital fossa and in the forearm, while in the AD forearm skin, pre-bedtime TEWL increases. We identified 1576 differentially expressed genes using a time-dependent model. The top 20 upregulated gene ontology pathways included neuronal pathways, while the downregulated functional terms included innate immune signaling and viral response. Similar pathways positively correlated with forearm TEWL in controls and inversely with the AD group. Upregulation in sensory perception pathways correlated with increases in lesional (antecubital fossa) TEWL in the evening. Results show skin barrier function worsens in the evening in the AD group, at a time when barrier is normally rejuvenating in healthy skin. This timing and the detection of transcriptomic signatures of sensory perception and diminished viral response might correspond to the nocturnal itch. Larger studies are needed to evaluate these associations in the skin.
Atopic dermatitis (AD), popularly known as eczema, is the most common chronic skin disease of early childhood afflicting ~10% of US children (Fishbein et al., 2020), with a nocturnal predominance of symptoms (Fishbein et al., 2015). AD is a disease of skin barrier and systemic immune dysregulation. Previously considered to be a local skin problem, it is now clear that AD is a systemic inflammatory condition which responds well to systemic treatment (Guttman-Yassky et al., 2019). Blood biomarkers reflect skin-directed inflammation (Esaki et al., 2016), and whole blood RNA-seq has been used to explore relevant disease pathways (Suárez-Fariñas et al., 2015). In addition to systemic inflammation, skin barrier disruption is central to AD disease flares (Fishbein et al., 2020). In fact, the gold standard measure of skin barrier function, transepidermal water loss (TEWL), is one of the most notable objective biomarkers differentiating AD versus control patients (Alexander et al., 2018). In more detail, TEWL is a measurement that represents the amount of water that escapes from the stratum corneum (the outermost layer of the epidermis) per area of skin, which reflects the integrity of skin water barrier. The increase in the TEWL value is interpreted as disruption of the barrier, while decrease in TEWL value is linked to the restoration of the skin barrier. It is important to note that TEWL varies between the individuals and across the anatomical locations (Alexander et al., 2018). AD is symptomatically worse in the evening, with increased patient self-report of pruritus (itchy skin; Cheng et al., 2022) and sleep disturbance (Fishbein et al., 2020). Beyond general reports of upregulated nocturnal inflammatory cytokines measured in whole blood (such as interleukin-6; Bender et al., 2008), the mechanism driving “nocturnal eczema” remains elusive. In healthy control patients, skin barrier function has a clear circadian (~24 h) rhythm that remains stable, even after barrier disruption following tape stripping or the application of topical steroids (Yosipovitch et al., 2004). However, the circadian rhythm of TEWL in diseased skin has not been evaluated.
Given the nocturnal worsening of AD, we hypothesize that nocturnal eczema is, at least in part, driven by nocturnal worsening of TEWL. In this pilot study, we sought to explore AD versus control TEWL rhythms and potential molecular underpinnings from whole blood biomarkers.
Methods
We recruited AD patients ages 15-25 years and age-/sex-matched controls. Patients were admitted for a modified constant routine protocol (Alexander et al., 2018). After an adaptation night and intravenous line placement, lighting (<20 lux), humidity, and temperature were kept constant with isocaloric snacks provided every 2 h during wake time. There were 28 h of continuous monitoring with 9 h of sleep opportunity at habitual bedtime. Measurement of skin barrier function, TEWL, was performed using the closed chamber Aquaflux device in triplicates at each location, right non-dominant arm antecubital fossa (lesional) or forearm (non-lesional), taken hourly during the day and every 90 min overnight. Antecubital fossa refers to the elbow crease, which is typically where the AD lesions are, whereas the forearm is generally lesion-free. The existence of lesional skin in the antecubital fossa was an inclusion criterion for our study. Patients were prompted to hold their arm out for 20 min prior to measurements to acclimate. Overnight, a nurse would quietly enter the room and help hold their arm out. Multiple skin biopsies are not practical, so we collected whole blood every 4 h in PAXgene tubes to explore potential mechanisms underlying TEWL circadian rhythm (Braun et al., 2018). All subjects had samples for zeitgeber time (ZT) 9, 13, 21, and 25. ZT0 was determined to be lights-on based on 9 h after habitual bedtime. RNA-seq was performed using the Illumina platform.
Detailed methods for RNA-seq processing and TEWL rhythm detection can be found in the Supplementary Methods. Raw fastq files were aligned to hg38 assembly. To correct for batch and unwanted variation we utilized an R package RUVSeq v1.20.0, specifically the RUVr method which considers the residuals from a first-pass generalized linear model regression of the counts on the covariates of interest. Resulting weights were passed to DESeq2 as parameters of the model. Genes with false discovery rate–adjusted p-values of less than 0.05 and|log2FC| ≥ 0.584 were considered as differentially expressed unless otherwise specified. Functional analysis was carried out using R package clusterProfiler v.3.16.0.
Subject-centered TEWL rhythm analysis was performed using a non-parametric algorithm called RAIN, which is implemented as R library (for details, see Supplementary Methods) which can work with outliers and is optimized for small-size time series. The subject’s TEWL rhythm was assumed to be significant if the Benjamini-Hochberg–adjusted p-value was less than 0.05. Pearson correlation coefficients between the subject-calibrated TEWL and gene expression profiles were calculated in R and were deemed significant if correlation coefficient was greater than 0.3.
Results
Patient Characteristics and TEWL Measures
In this pilot study, we recruited AD (n = 4) and control patients (n = 2; for details, see Supplementary Table S1). TEWL measurements taken over 24 h (Figure 1) show the relative change in TEWL over time, represented as a subject-centered, log2-transformed TEWL. In control group, the group-averaged rhythm is more pronounced in the antecubital fossa rather than forearm skin barrier when comparing to AD group. Notably, phase estimation using RAIN algorithm (Thaben and Westermark, 2014) predicted differences in phase estimates of the AD versus control TEWL, showing a coordinated oscillation profile in controls, and lack of synchronization and flattening of the oscillations among AD subjects (Supplementary Table S2). Consistent with previous publications (Yosipovitch et al., 2004, 1998), TEWL in control skin, both in antecubital fossae and in forearm, decreases (improves) starting a few hours prior to bedtime (ZT15, ~22 local time). However, during this time in AD patients, TEWL does not improve and, in fact, in forearm skin appears to worsen (TEWL increases). Absolute values for TEWL by group are plotted in Supplementary Figure S1, with subject-specific profiles in Supplementary Figure S2. Patient groups were similar in terms of their hormonal melatonin and cortisol profiles (Suppl. Fig. S3) demonstrating the controlled modified constant routine protocol.
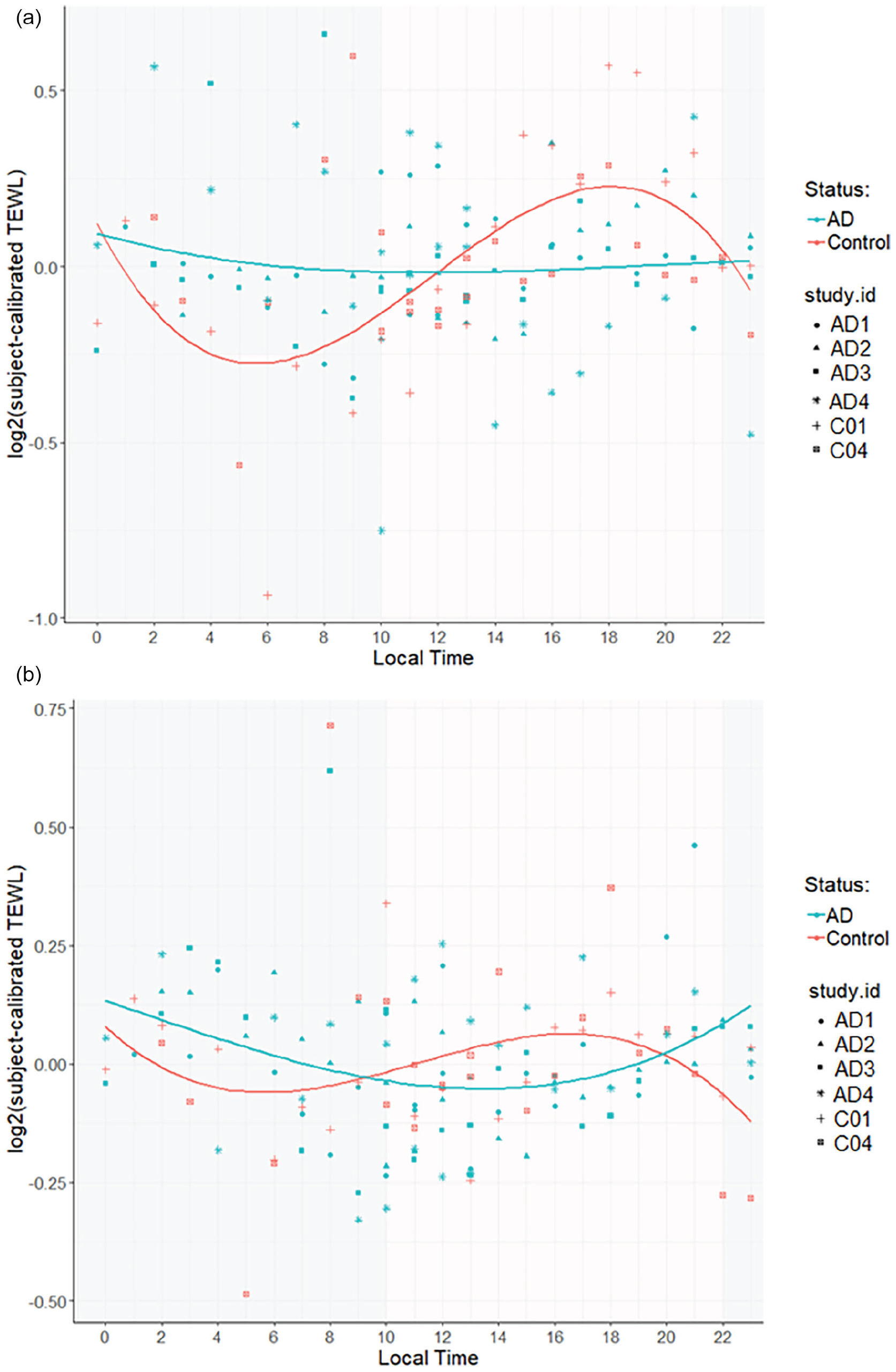
TEWL across 24 h in (a) lesional (antecubital fossa) and (b) non-lesional (forearm skin) in AD versus control patients, group-averaged profiles. Plotted with local time on the x-axis and TEWL in g/m2/h on the y-axis, expressed as a log2 (subject-calibrated TEWL) then averaged across subjects. Plot of model predicted values (smoothed lines) and individual patient values at each time point denoted. The shaded region denotes sleeping time range across all patients, which varied by patient but was set at 9 h per subject with bedtime at habitual bedtime from 7 days prior to admission. Abbreviations: TEWL = transepidermal water loss; AD = atopic dermatitis.
Differential Gene Expression and Gene Ontology (GO) Pathways
Differential gene expression in whole blood samples (principal components analysis plots in Suppl. Fig. S4) identified 1576 differentially expressed genes in AD group (Figure 2a) as compared to controls using a time-dependent model (Supplementary Methods). We evaluated these 1576 genes for significant GO pathways in AD versus control patients, including time as a factor in the model. The top 20 upregulated GO pathways in AD group versus controls demonstrated several neuronal pathways, including myelination, ensheathment of neurons, and axon ensheathment (Figure 2b).
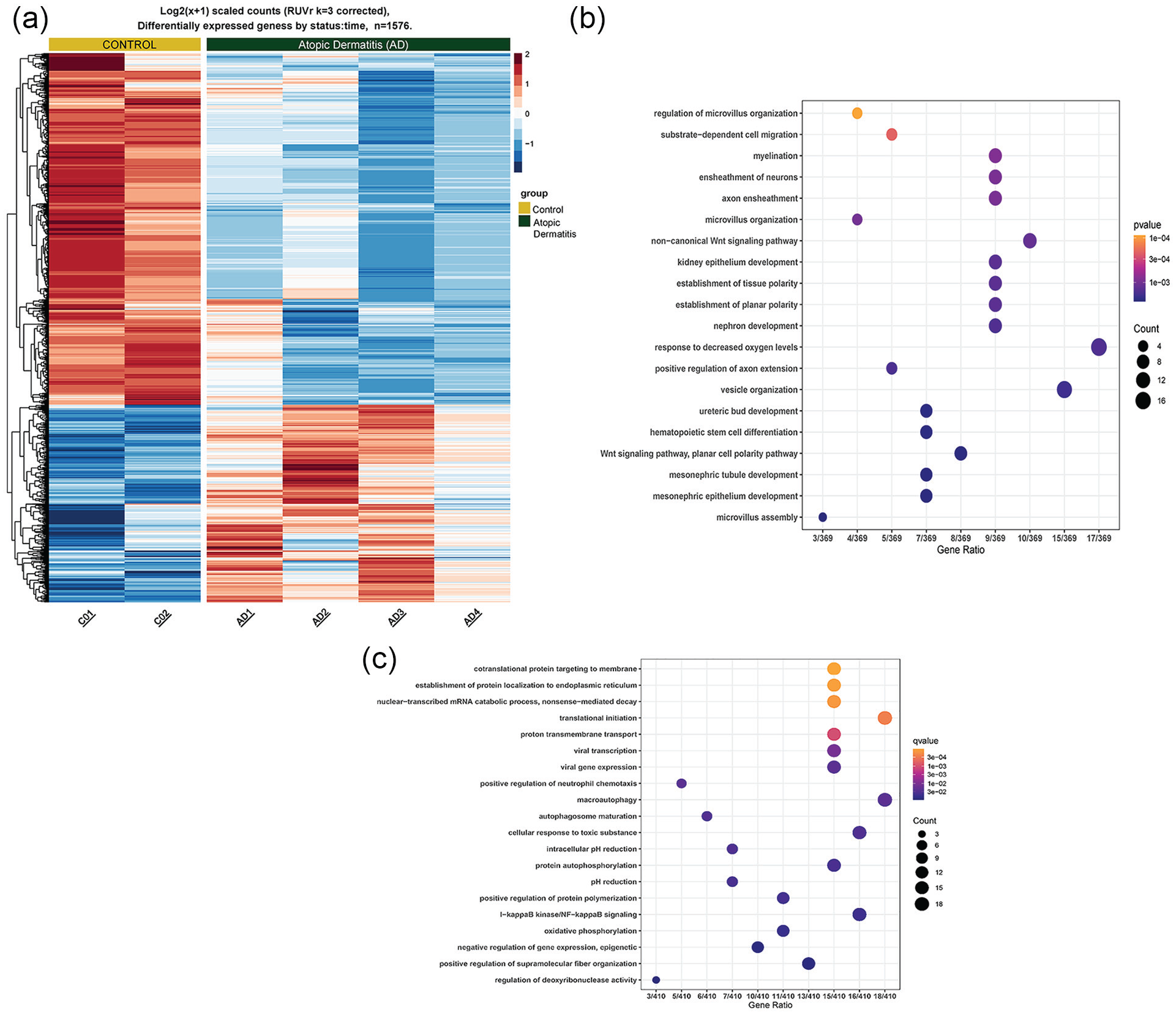
Transcriptional differences between AD and control subjects. (a) Heat map displaying differential gene expression in a time-dependent model. AD patients have a distinct transcriptome. (b) Top 20 GO terms in upregulated genes in AD patients compared to controls in a time-dependent model of differential gene expression. (c) Top 20 GO terms in downregulated genes in AD patients compared to controls in a time-dependent model of differential gene expression. Abbreviations: AD = atopic dermatitis; GO = gene ontology.
The top 20 GO terms downregulated in AD group included innate immune signaling pathways (such as positive regulation of neutrophil chemotaxis and IκB kinase/nuclear factor-κB signaling), as well as viral response pathways (such as viral transcription and viral gene expression; Figure 2c). These similar pathways also appeared in GO terms enriched in genes positively correlated with TEWL in the forearm of controls and correlated inversely with the forearm TEWL in AD group (Suppl. Fig. S5).
TEWL and Molecular Correlations
We explored correlations between lesional TEWL rhythms with the whole blood transcriptome (at ZT9, 13, 21, and 25) in AD versus control patients (Supplementary Table S2 and Supplementary Methods correlation thresholds used). We identified the top 10 GO pathways that were correlated with antecubital fossa TEWL in AD group and inversely correlated with antecubital fossa in control group, which included several pathways of sensory perception (Suppl. Fig. S6).
Discussion
In this pilot study, we found that skin barrier function (measured by TEWL rhythm) in AD skin worsens in the evening in AD group, at a time when barrier is normally rejuvenating in healthy, control skin. The lack of synchronization and flattening of the TEWL rhythm in AD skin contrasts with robust and consistent melatonin and cortisol profiles. By averaging TEWL across the AD patients, we noted a flatter curve which could be due to the phase (Suppl. Fig. S2). Regardless, the conclusion is that TEWL profile is similar among the controls but is quite different between the AD subjects.
TEWL rhythm worsening before bed in AD group corresponds with timing of patient/parent-proxy report of itch worsening before bedtime (Cheng et al., 2022). To our knowledge, this is the first paper to suggest the potential mechanistic link between nocturnal scratching with worsening TEWL rhythms. This is further supported by correlation of this timing with the transcriptomic signature of upregulation in sensory perception. Indeed, AD skin is noted to be hyperinnervated (Tominaga and Takamori, 2014), a finding that can be detected systemically with serum factors such as nerve growth factor (Toyoda et al., 2002).
This is further supported by upregulated GO pathways we identified in AD versus control group, which have been shown in mouse models of itch. This suggests the potential relevance of our barrier dysfunction findings and itch. Specifically, in TRPA1-expressing mice treated with acetone to induce chronic itch, skin biopsies demonstrate pathways of neurogenesis and axonogenesis, not present in TRPA1 knockout mice (Bender et al., 2008). In addition, similar GO pathways in neonatal mouse skin are noted when the aryl hydrocarbon receptor is constitutively expressed (Yosipovitch et al., 2004). Likely, these pathways were previously undiscovered in humans due to only daytime collection of samples.
With regard to downregulated pathways, our study is the first to discover the relevance of time as a factor in poor viral immunity in AD group. It is known that AD patients are at risk for viral infections in the skin, such as eczema herpeticum (Berdyshev et al., 2022), and even other systemic viruses, such as influenza (Jalbert et al., 2020). Although none of our patients reported a history of eczema herpeticum, a previous paper identified a poor innate immune response RNA-seq signature in peripheral blood mononuclear cells of AD patients with a history of eczema herpeticum after in vitro herpes simplex virus stimulation (Bin et al., 2014). AD patients experience significant sleep disturbance, also known to suppress viral immunity in healthy individuals (Prather et al., 2015).
Although it might be a bit far reaching, these findings suggest consideration of sleep disturbance and circadian health in AD patients as one strategy to strengthen viral immunity.
Limitations to this pilot study include the small sample size and lack of skin-derived molecular samples. Further work will focus on increasing sample size, including inflammatory circadian rhythms, and exploring whether these similar transcriptomic pathways are noted in skin and skin homing cells.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304231220695 – Supplemental material for Circadian Rhythms in Skin Barrier Function in Atopic Dermatitis: A Pilot Study
Supplemental material, sj-docx-1-jbr-10.1177_07487304231220695 for Circadian Rhythms in Skin Barrier Function in Atopic Dermatitis: A Pilot Study by Marta Iwanaszko, Nathan Waldeck, Ron Anafi, Amy S. Paller, Phyllis C. Zee and Anna B. Fishbein in Journal of Biological Rhythms
Supplemental Material
sj-docx-2-jbr-10.1177_07487304231220695 – Supplemental material for Circadian Rhythms in Skin Barrier Function in Atopic Dermatitis: A Pilot Study
Supplemental material, sj-docx-2-jbr-10.1177_07487304231220695 for Circadian Rhythms in Skin Barrier Function in Atopic Dermatitis: A Pilot Study by Marta Iwanaszko, Nathan Waldeck, Ron Anafi, Amy S. Paller, Phyllis C. Zee and Anna B. Fishbein in Journal of Biological Rhythms
Footnotes
Acknowledgements
This work was funded, in part, by National Institute of Arthritis and Musculoskeletal and Skin Diseases (grant number K23AR075108 to A.B.F.), Ann & Robert H. Lurie Children’s Hospital of Chicago, Northwestern University Illumina Pilot Grant, and the National Cancer Institute R50 (grant number CA265372 to M.I.).
Author contributions
Conflict of interest statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
RNA-seq data were deposited as Gene Expression Omnibus repository. Gene lists and TEWL values are available upon request.
Note
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
