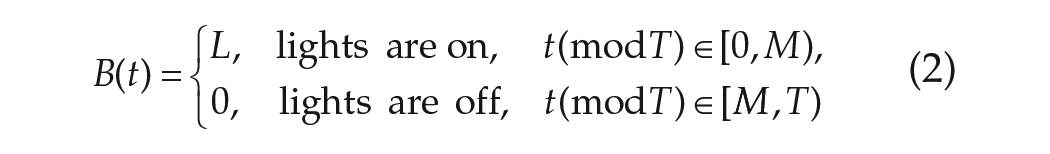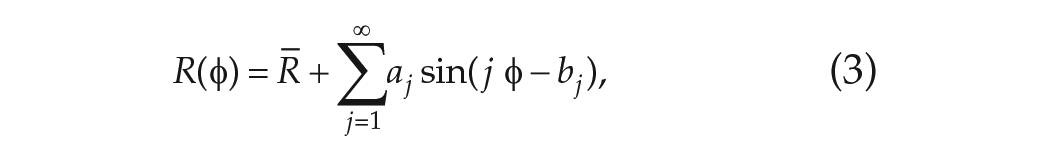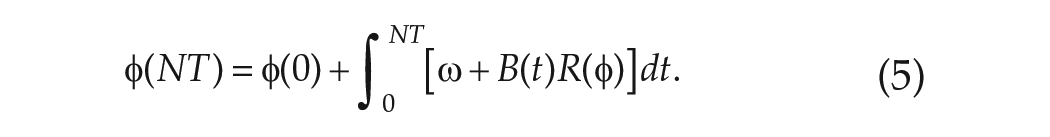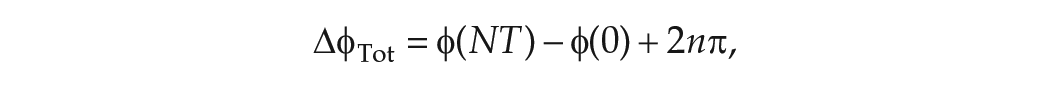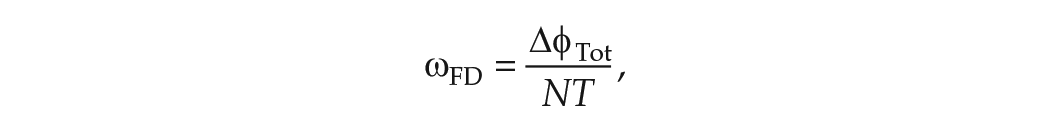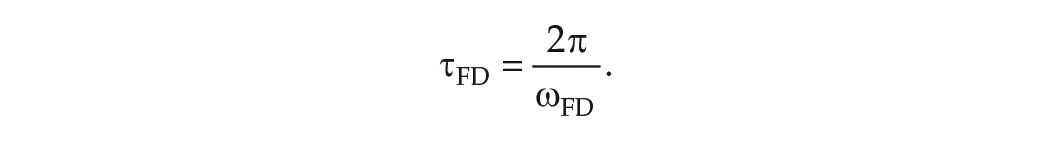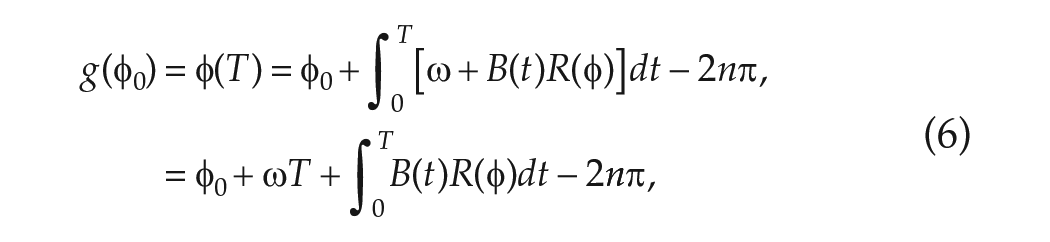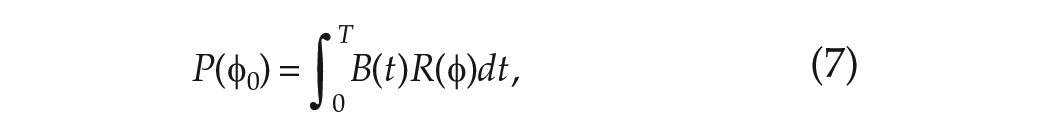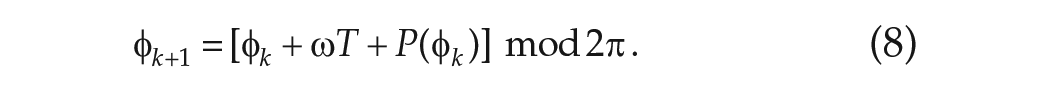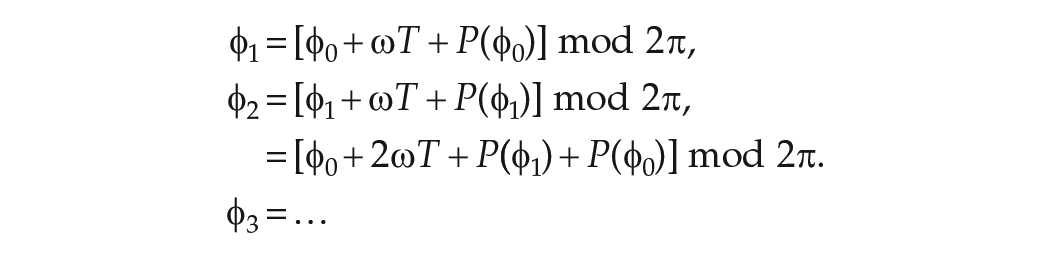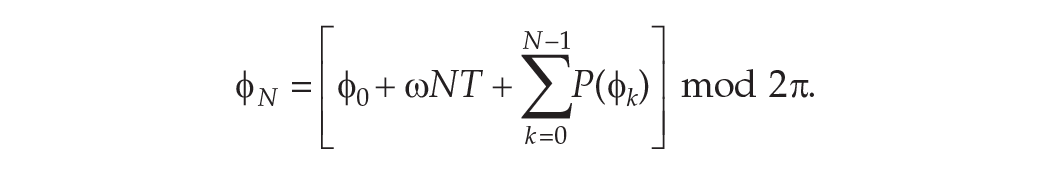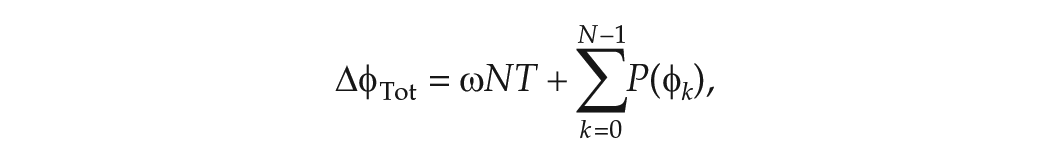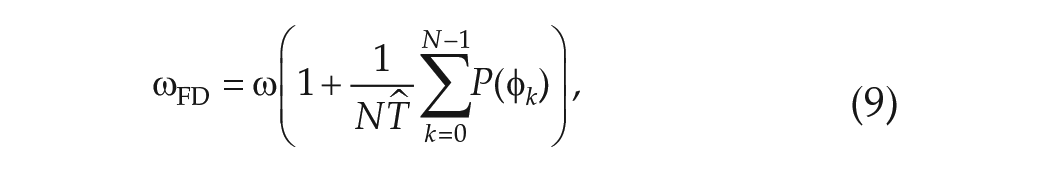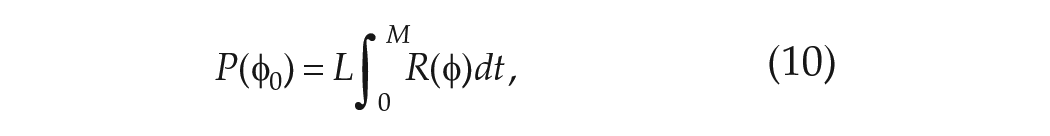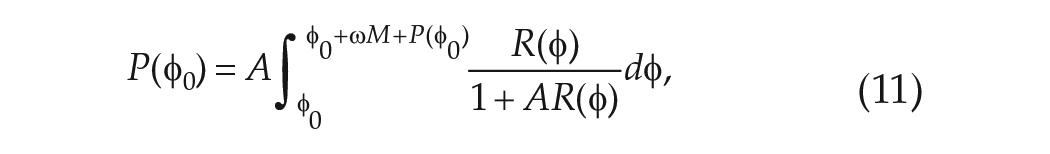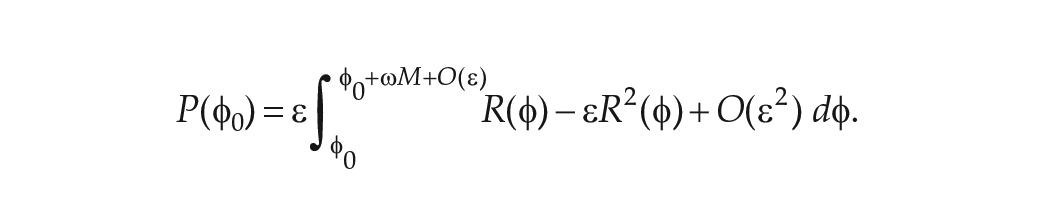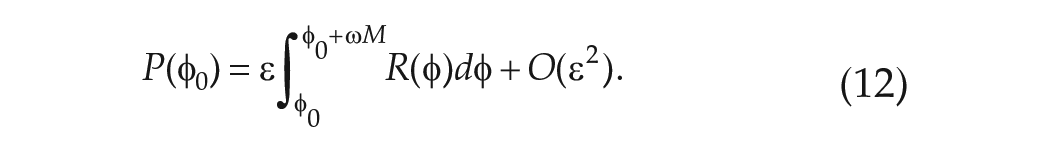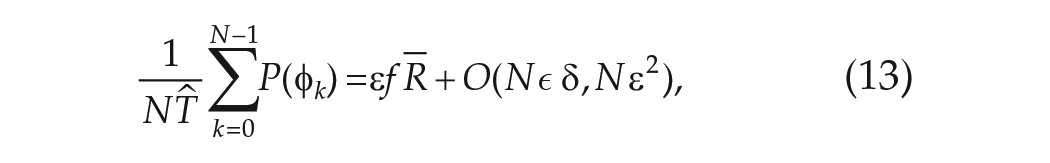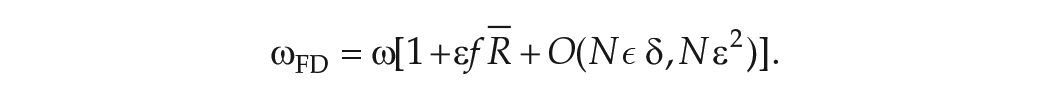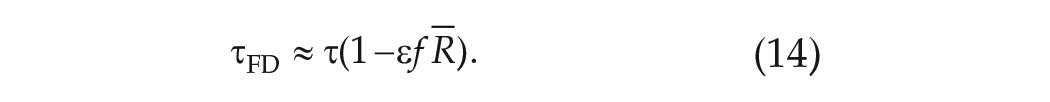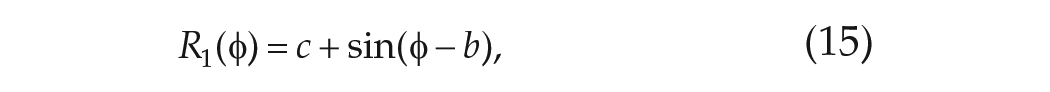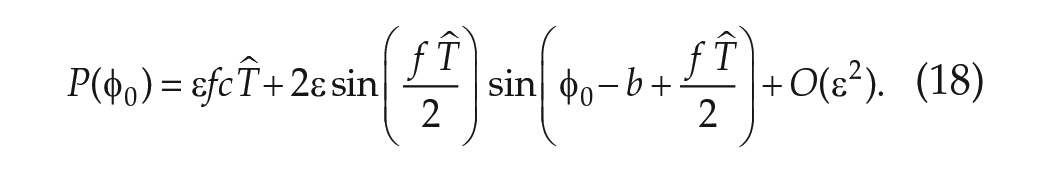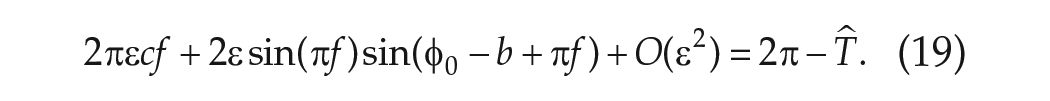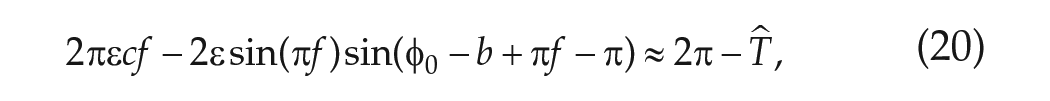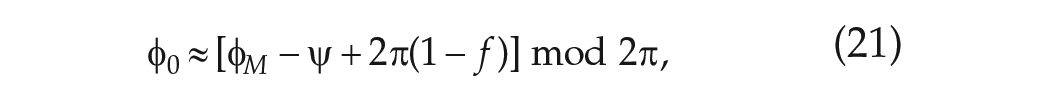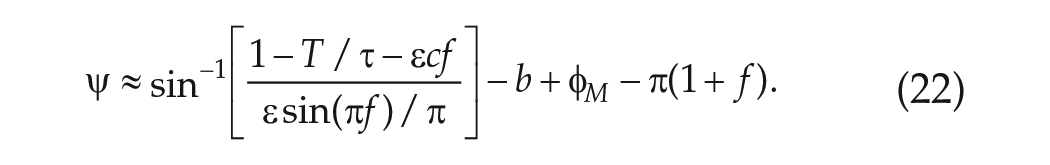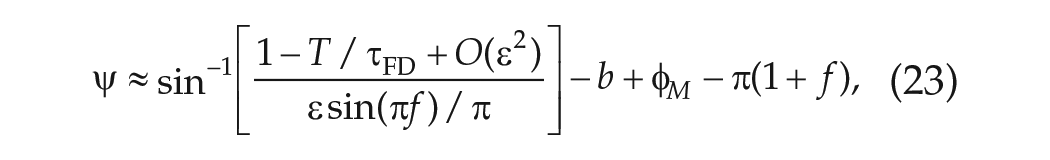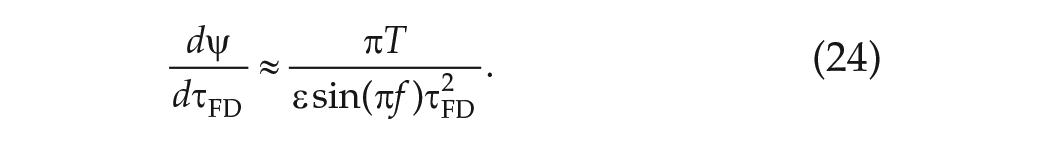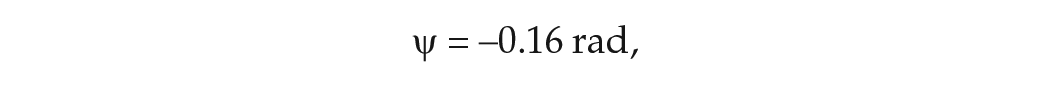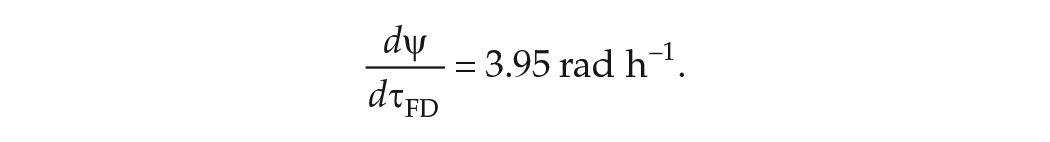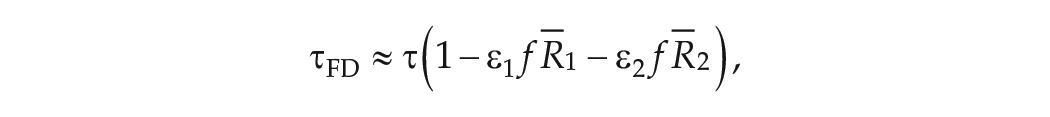Abstract
Accurate assessment of the intrinsic period of the human circadian pacemaker is essential for a quantitative understanding of how our circadian rhythms are synchronized to exposure to natural and man-made light-dark (LD) cycles. The gold standard method for assessing intrinsic period in humans is forced desynchrony (FD) which assumes that the confounding effect of lights-on assessment of intrinsic period is removed by scheduling sleep-wake and associated dim LD cycles to periods outside the range of entrainment of the circadian pacemaker. However, the observation that the mean period of free-running blind people is longer than the mean period of sighted people assessed by FD (24.50
Keywords
Appropriate timing of physiology and behavior to temporal niches associated with geophysical cycles contributes to fitness of biological systems (West and Bechtold, 2015), including the health of humans (Fishbein et al., 2021). This “appropriate timing” is reflected in 24-h rhythmic variation in gene expression, translation, physiology, and behavior and is referred to as circadian rhythmicity. A defining feature of circadian rhythms is that they are self-sustaining (Pittendrigh, 1960). The rhythms are generated by oscillators, whose activity persists in the absence of cyclical changes, known as zeitgebers, in the external environment (Aschoff, 1960). In the study of the central circadian pacemaker in mammals, the intrinsic period refers to the period of the pacemaker in the absence of zeitgebers. The intrinsic period is close to, but rarely equal to, 24 h (Czeisler et al., 1999) and entrainment to 24 h is achieved by an adjustment to the intrinsic rhythm of the pacemaker through exposure to 24-h zeitgebers (Daan, 1977, 2000).
Accurate estimation of the intrinsic period is important for two main reasons. First, the intrinsic period is a key factor in determining whether the pacemaker can entrain to 24-h light-dark (LD) cycles, since the magnitude of the adjustment required for entrainment depends on the difference between the intrinsic period and the period of the LD cycle (Pittendrigh and Daan, 1976b). Second, when the pacemaker entrains to LD cycles, the intrinsic period determines the phase of entrainment, that is, the timing of endogenous rhythmicity relative to the zeitgeber, with longer intrinsic period associated with later sleep timing (Duffy et al., 2001). Thus the intrinsic period of the circadian pacemaker and its variation between individuals informs the interpretation of circadian rhythm sleep-wake disorders (Meyer et al., 2022; Micic et al., 2016) as well as the variation in the timing of rhythmicity in the general population. For example, mathematical models suggest that those with a longer intrinsic period are more sensitive to the delaying effects of access to evening light (Skeldon et al., 2017). Furthermore, guidelines on healthy light exposure requirements critically depend on an assessment of the average and between-individual variation of this key parameter along with the sensitivity of the pacemaker to light.
The most reliable way to assess the intrinsic period of the pacemaker is to place a person or animal in constant darkness (DD). This is because in DD, the principal zeitgeber to the pacemaker, namely light (Czeisler et al., 1981; Dijk et al., 1995), is removed. The rest-activity cycle and behaviors associated with the rest-activity cycle, such as feeding, persist in DD, but the non-uniform distribution of these behaviors across the circadian cycle is assumed not to affect the period of the pacemaker to a significant extent (but see Kas and Edgar, 2001 for a counterexample). In nocturnal animals, the period in DD is measured readily (Pittendrigh and Daan, 1976a), but there are practical and ethical barriers to studying sighted humans in DD. Consequently, sighted people are rarely studied in DD, although in the 1970s Wever (1979) did assess the intrinsic period of 5 sighted humans who lived in DD for approximately 2 weeks. The intrinsic period of humans has traditionally been assessed in classical free-run (Wever, 1979) and forced desynchrony (FD) protocols (Czeisler et al., 1999; Wang et al., 2023). In FD, participants are exposed to LD cycles with a period very different from 24 h, usually 28 h or 20 h. In standard protocols, lights are on and participants are required to be awake for two-thirds of the time. Lights are off and participants are in bed and encouraged to sleep for the remaining one-third. With 28 (20) h cycles, wake is therefore scheduled to occur 4 h later (earlier) each day. Since 28 (20) h is outside the limits of entrainment, over the course of (an integer multiple of) 6 LD cycles the circadian clock is exposed to light at (approximately) all different phases. In this design, the aim is to minimize the effects of light, which allows the human circadian clock to progress at its natural period.
More recently, the intrinsic period has been assessed by measuring the in vitro period of fibroblasts taken from individual participants (Pagani et al., 2010). The fibroblast period is measured by introducing firefly luciferase genes into the fibroblast cells via a lentivirus. Since the expression of firefly luciferase is then driven by the circadian gene Bmal1, the fibroblasts exhibit periodic patterns of bioluminescence which are measured via luminometry.
Figure 1 summarizes estimates of the intrinsic period in blind and sighted humans using these various methods. Here, only blind participants with nonentrained rhythms are included, where blind means having no subjective perception of light. For studies where melatonin suppression was measured, we have further restricted those who had no melatonin suppression by light. For example, Flynn-Evans et al. (2014) studied 127 blind people of whom 41 had no light perception and 16 of these were nonentrained. In Figure 1, only the 16 nonentrained participants are included.
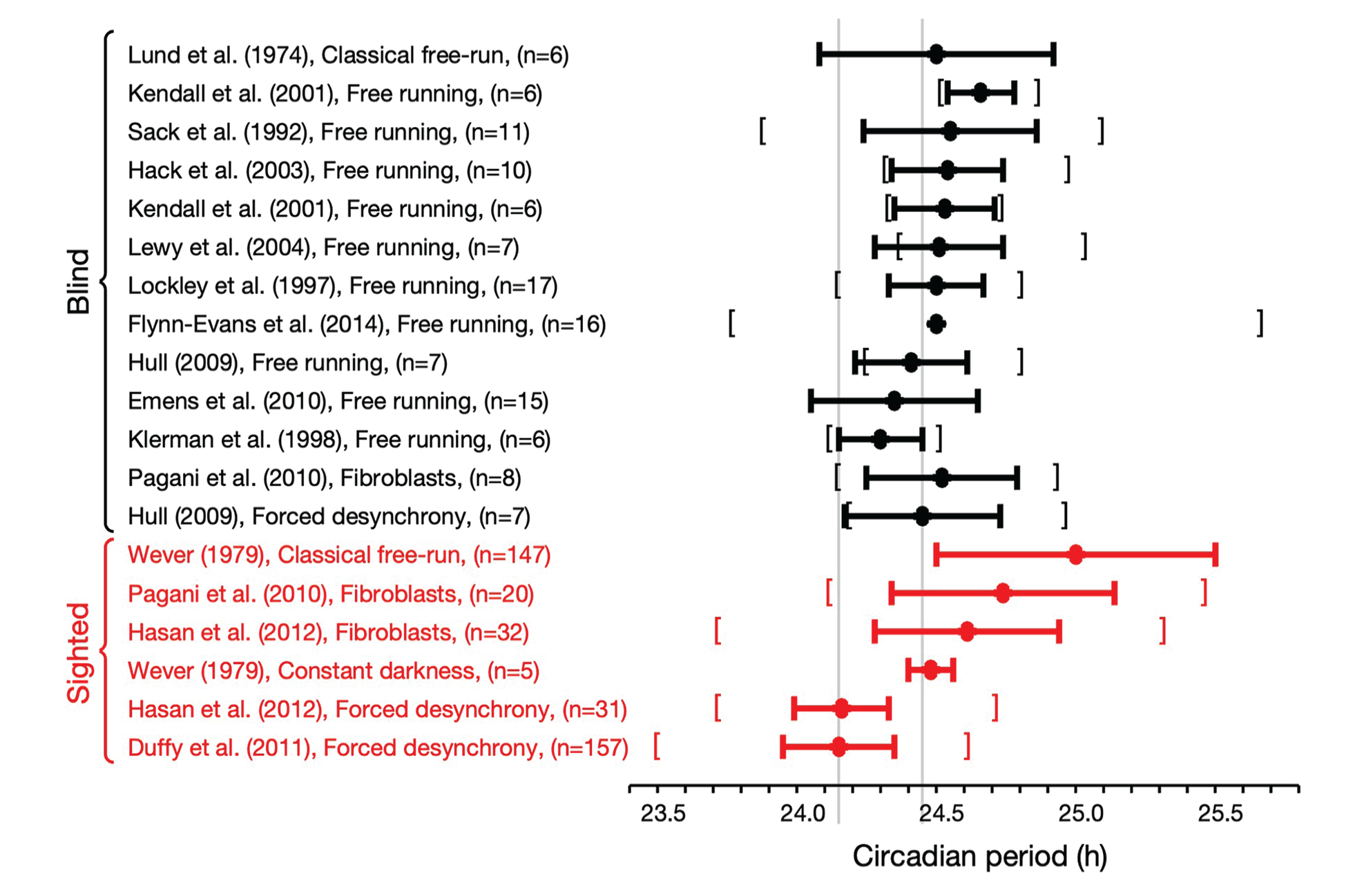
Estimates of the intrinsic period of the circadian pacemaker in blind and sighted humans using various protocols. Circles indicate the mean with the horizontal bars indicating the mean ± standard deviation. Where available, the square brackets indicate the range of the measurements. In some cases (e.g. Lewy et al., 2004) where the studies only include a small number of participants, the distribution of periods is skewed so that the smallest value recorded is greater than the mean – standard deviation. The gray vertical lines indicate accepted values for the circadian period of sighted (as measured in forced desynchrony) and blind individuals, respectively.
In Figure 1, it can be seen that the mean period of blind people is consistent across different protocols (Emens et al., 2010; Flynn-Evans et al., 2014; Hack et al., 2003; Hull, 2009; Kendall et al., 2001; Klerman et al., 1998; Lewy et al., 2004; Lockley et al., 1997; Lund, 1974, Sack et al., 1992), and consistent within individuals assessed in both free-running field conditions and in an FD protocol in the laboratory (Hull, 2009). In contrast, the mean period of sighted people is variable depending on the protocol (Duffy et al., 2011; Hasan et al., 2012; Wever, 1979). The period of sighted people in DD is consistent with intrinsic period estimates in the blind. In addition, there is no significant difference between the mean period of fibroblasts from sighted people and the period of fibroblasts from blind people (p = 0.17). The reported standard deviations of estimates of periods appear larger in classical free run and fibroblasts than observed in FD protocols but are similar between sighted people in FD and assessments in the blind.
Motivated by concerns over the impact of room lighting in classical free-run protocols, FD has emerged as a widely accepted gold standard method for assessing the intrinsic period of the circadian pacemaker in sighted humans (Dijk and Duffy, 2020; Wang et al., 2023). It has been proposed that the shorter period of sighted people in FD compared to blind people is due to aftereffects of prior entrainment in sighted people (Duffy and Wright, 2005). The presence of aftereffects implies that the period of sighted people in FD should be variable depending on prior period of entrainment. However, in humans, the period of the zeitgeber during prior entrainment appears to have only a modest effect on the subsequent period of the pacemaker in FD (mean difference 0.1 h; Scheer et al., 2007). It is also interesting, and maybe surprising, to note that the average periods of fibroblasts in sighted and blind people in vitro are comparable with each other and similar to the intrinsic period of blind people (see Figure 1), although in sighted participants the fibroblast period does not correlate with the period of plasma melatonin as assessed in FD (Hasan et al., 2012).
In classical free run, the self-selected light exposure of participants is likely to modulate the period of the pacemaker in sighted people (Klerman et al., 1996). To minimize the effect of light, FD protocols aim to distribute light evenly over the circadian cycle and use dim light. For example, in Wang et al. (2023) it is recommended that light levels in FD should be less than 15 lux, and it is reported that in many FD experiments light of intensity less than 4 lux has been used. Even at these low intensities, there is evidence that dim LD cycles may modulate the period of sighted people. For example, Wright et al. (2001) demonstrated that sighted people can entrain to dim (~~1.5 lux) 24-h LD cycles in a carefully controlled experiment with an imposed 8:16 rest-activity cycle.
In view of these discrepancies and unresolved issues relating to the intrinsic period of the human circadian pacemaker, and an absence of a formal mathematical analysis of how light may affect the human circadian pacemaker, a further analysis seems warranted. Here, we use a simple mathematical model of the circadian pacemaker to describe the effect of dim LD cycles on the circadian pacemaker in sighted humans. Using this model, we derive an expression relating the period in FD to the intrinsic period, which highlights the dependence of assessed period on the symmetry of the velocity response curve (VRC) in the model. We estimate parameters of our model using Wright’s data on the entrainment of humans to dim LD cycles (Wright et al., 2001). Then, we present a hypothesis for the observed shorter period of sighted people in FD compared to blind people. We describe experimental protocols to test this hypothesis. Our hypothesis offers one solution to a long-standing discrepancy and has implications for quantitative models that predict the effect of the light environment as mandated by policies about light exposure requirements and work schedules.
Materials and methods
Simple Clock Model of the Human Circadian Pacemaker for Dim Light Conditions
It is well established that the human circadian pacemaker behaves as a phase-amplitude oscillator, perturbations of which can lead to changes in phase and amplitude (Czeisler et al., 1989; Khalsa et al., 1997; Strogatz, 1990). Kronauer’s model of the human circadian pacemaker (Jewett and Kronauer, 1998) and its later versions (Forger et al., 1999; Jewett et al., 1999; St Hilaire et al., 2007) are the most widely used in human circadian research. These models were designed to replicate phase resetting studies, including amplitude reduction, Type-1 phase resetting, and Type-0 phase resetting (Khalsa et al., 1997). These models are currently being used to predict human circadian phase from ambulatory light data (Huang et al., 2021; Rea et al., 2022; Woelders et al., 2017) across a range of populations including students (Phillips et al., 2017) and shiftworkers (Stone et al., 2019) and are competitive with traditional phase assessment methods in terms of accuracy (Dijk and Duffy, 2020). Kronauer-type models have been used to suggest interventions to minimize the disruptive effects of jet lag (Serkh and Forger, 2014), non-24-h sleep-wake disorder, shiftwork, and social jet lag (Diekman and Bose, 2022). Kronauer-type models have also been combined with models of sleep regulation to investigate changes in sleep timing preferences (Phillips et al., 2010; Skeldon et al., 2016), sleepiness and cognitive performance due to shift-working (Postnova et al., 2014; Postnova et al., 2018), the impact of light and social constraints on sleep timing preferences and social jet lag (Skeldon et al., 2017), and the effects of daylight saving (Skeldon and Dijk, 2019), and used to propose quantitative light “availability” interventions (Skeldon et al., 2022) to normalize sleep timing.
All the Kronauer-type models use a two-dimensional oscillator with a strongly attracting limit cycle to reproduce the self-sustaining activity of the clock coupled with a model for the effect of light on the clock. Earlier versions of the model were developed from experiments in which the light intensity varied from 10 to 9500 lux. The most recent version (St Hilaire et al., 2007) was adapted to more accurately reflect light sensitivity for intensities below 150 lux and additionally includes a non-photic zeitgeber. Here, the focus is on the effect of dim light, which constitutes a weak zeitgeber. It has been established that models with a strongly attracting limit cycle when exposed to a weak zeitgeber are well approximated by phase-only models (Guckenheimer and Holmes, 1983; see the Supplementary Material for further details). Therefore, for the purpose of analyzing the effects of dim light, here we use a simple phase-only model. In addition, we assume that the effect of light is to continuously modulate the velocity of the clock, which means that the model is parametric. Parametric models are generally considered to be good models of the circadian system in diurnal animals (Daan, 2000).
In phase-only models, the state of the clock at any time is described only by its phase
The velocity
where
In parametric models, LD cycles of period
where
In parametric models, the VRC typically contains an advance region, in which the effect of a stimulus is to speed up the clock, and a delay region, in which the effect of a stimulus is to slow down the clock. The VRC is a periodic function with period
where
In phase-only parametric models, the effect of light on the velocity of the clock is assumed to be smaller than the intrinsic velocity, that is
so that the phase of the clock advances monotonically. A schematic of the CBT rhythm and example VRCs are shown in Figure 2.
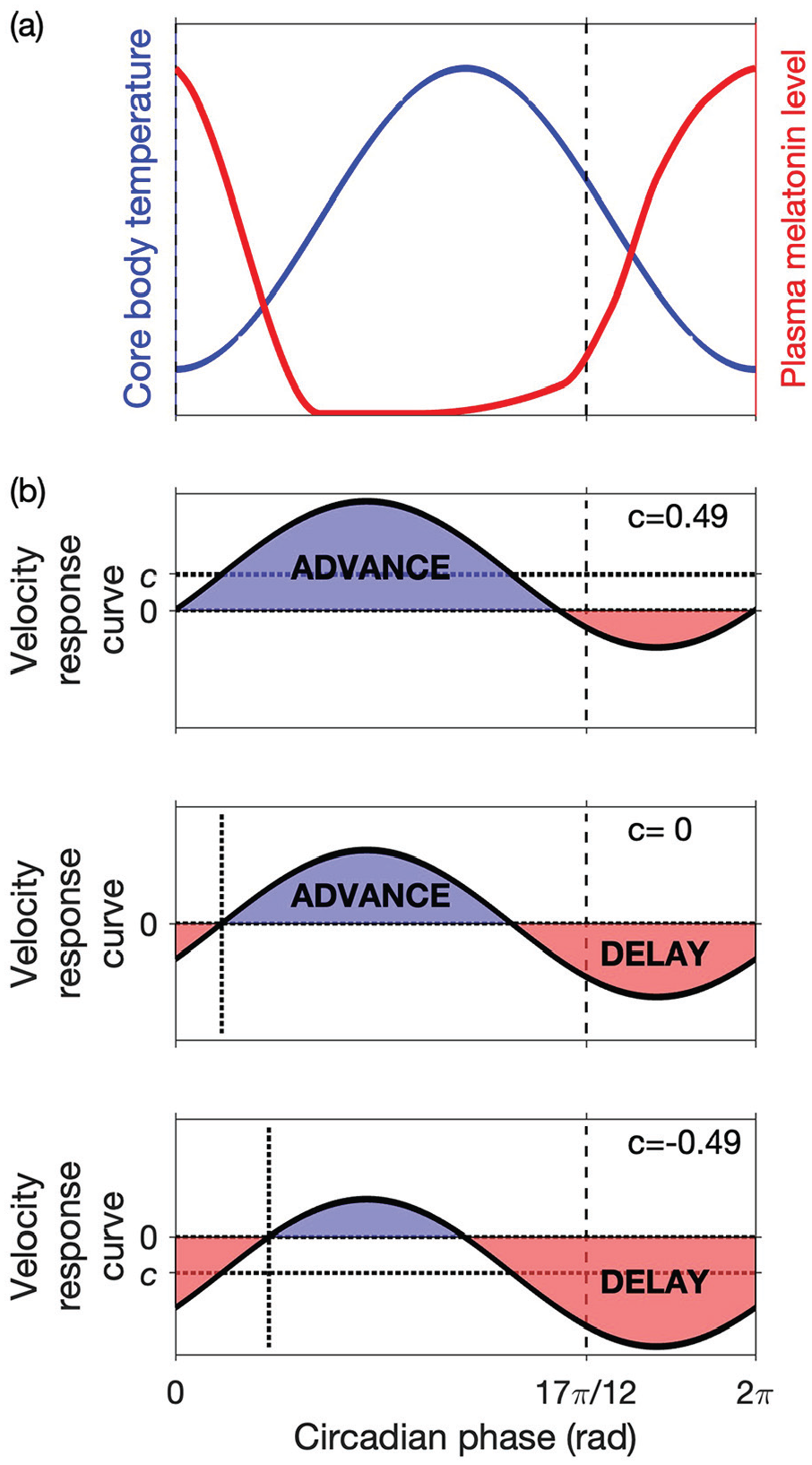
Schematic diagrams of the CBT, plasma melatonin rhythms, and the VRC in humans. (a) CBT and plasma melatonin rhythms are shown. The minimum of the CBT occurs at circadian phase
Results
Analytical Expression for the Period in FD Protocols as Found Using the Simple Clock Model
In experiments, the mean period in FD is evaluated in one of two ways. When data of a phase marker such as CBT are collected throughout the protocol, the non-orthogonal spectral analysis (NOSA) algorithm is used. The NOSA algorithm fits a mathematical function consisting of Fourier components for the mean period in FD,
Using the simple clock model, the mean period may be calculated as follows. If the FD protocol consists of
The total change in phase
where
and the mean period of the clock evaluated in FD is
Since the simple clock model does not include noise or evoked effects (e.g. the effect of the sleep-wake cycle on the circadian-regulated CBT) and there is no experimental error in calculating the phase, calculating the change in phase from the beginning to end of the protocol should lead to an accurate determination of
In general, it is difficult to carry out the integral in equation (5), and hence find
Analytical Expressions for the Phase Transition Curve and the Mean Angular Velocity
We first construct an expression for the mean angular velocity,
where
where
More generally, defining
In other words, starting at
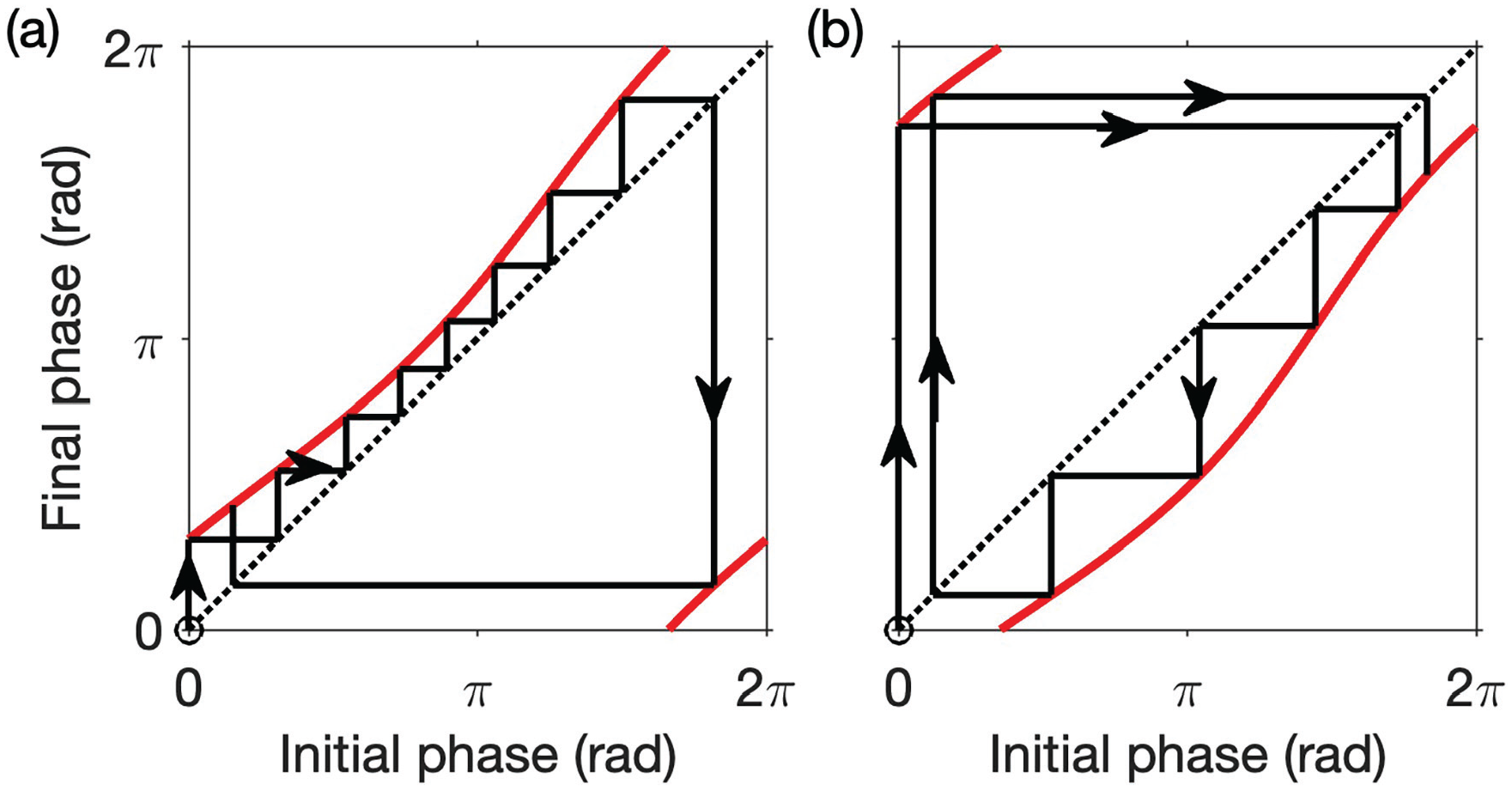
The phase transition curve as a one-dimensional map. Cobweb diagrams illustrating the map in equation (8) in simulations of FD are shown for LD cycles with different periods: (a)
Having calculated the phase after one LD cycle, we can now calculate the phase after
So after
Hence the total change in phase
where the first term on the right-hand side describes the total change in phase due to the natural angular velocity of the clock and the second term describes the cumulative phase response to successive LD cycles. The mean angular velocity is then:
where
Approximate Expression for the Cumulative Phase Response Across a FD Protocol
To calculate the mean angular velocity
We first consider the phase response to a single cycle
where
where
Thus,
Using the further assumption that the intrinsic period
where
Approximate Expression for the Period Measured in FD Protocols
Finally, combining equation (13) with equation (9) leads to an expression for the angular velocity in FD,
If the
Implications and Accuracy of the Approximate Expression for
Implications of the Approximate Expression for
Equation (14) states that, to lowest order, the observed circadian period in FD,
So, if the advance region is larger than the delay region
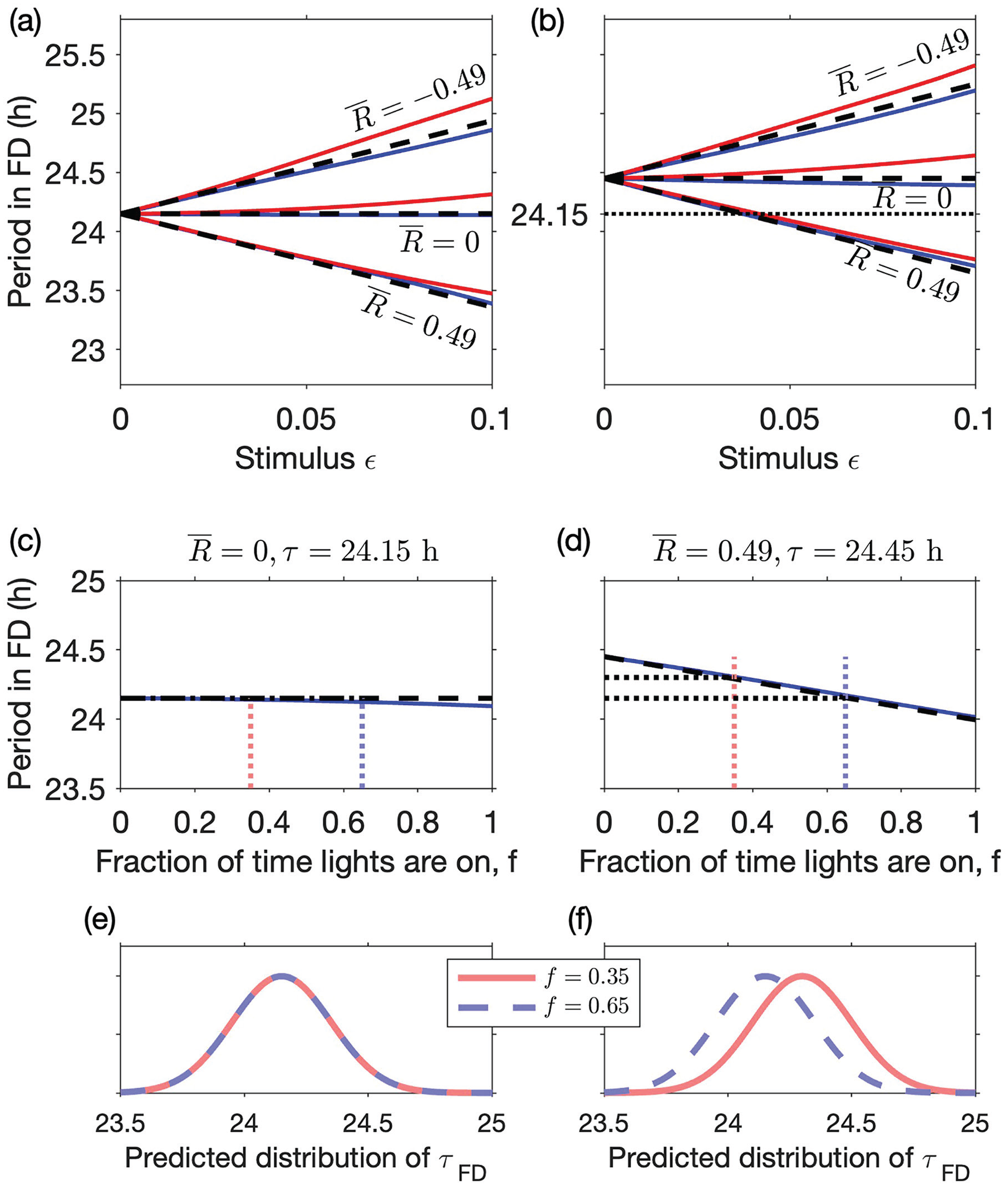
Effect of zeitgeber strength, VRC asymmetry, and duration of light period on the observed circadian period in FD predictions using a phase-only model. Upper panels show the period in FD predicted by equation (14) (dashed lines) for a protocol in which the lights are on for two-thirds of the time
Furthermore, equation (14) predicts that the distribution of circadian periods measured in FD will be similar to the distribution of intrinsic circadian periods. For example, if
Equation (14) also suggests possible approaches for assessing if there is asymmetry in the VRC. Specifically, since the size of the deviation from the intrinsic period is dependent on the fraction of time in which lights are on,
Accuracy of the Analytical Expression for
To illustrate the accuracy of our analytical expression, we compare it to simulations of FD using a simple clock model (see Figure 4). Equation (14) holds for a general VRC with mean value
where the VRC to light has been scaled in such a way that the coefficient of the sine term is unity (see Figure 2). For this specific form for the VRC,
Equation (14) assumes that
The deviations of the simulated results from the approximate solution can be explained qualitatively. There are two distinct effects on the observed period as
Second, as
When the period of the LD cycles in FD is
Parameter Estimation
Having shown that our analytical formula accurately describes how asymmetry in the VRC and dim LD cycles affects the estimate of intrinsic circadian period in FD in a phase-only model of the circadian pacemaker, the expected next question is whether it is possible to estimate the model parameters from available data. The simple clock model with the sinusoidal VRC contains a total of 4 parameters, namely the intrinsic period
In Wright et al., 4 participants were shown to entrain to dim LD cycles with period
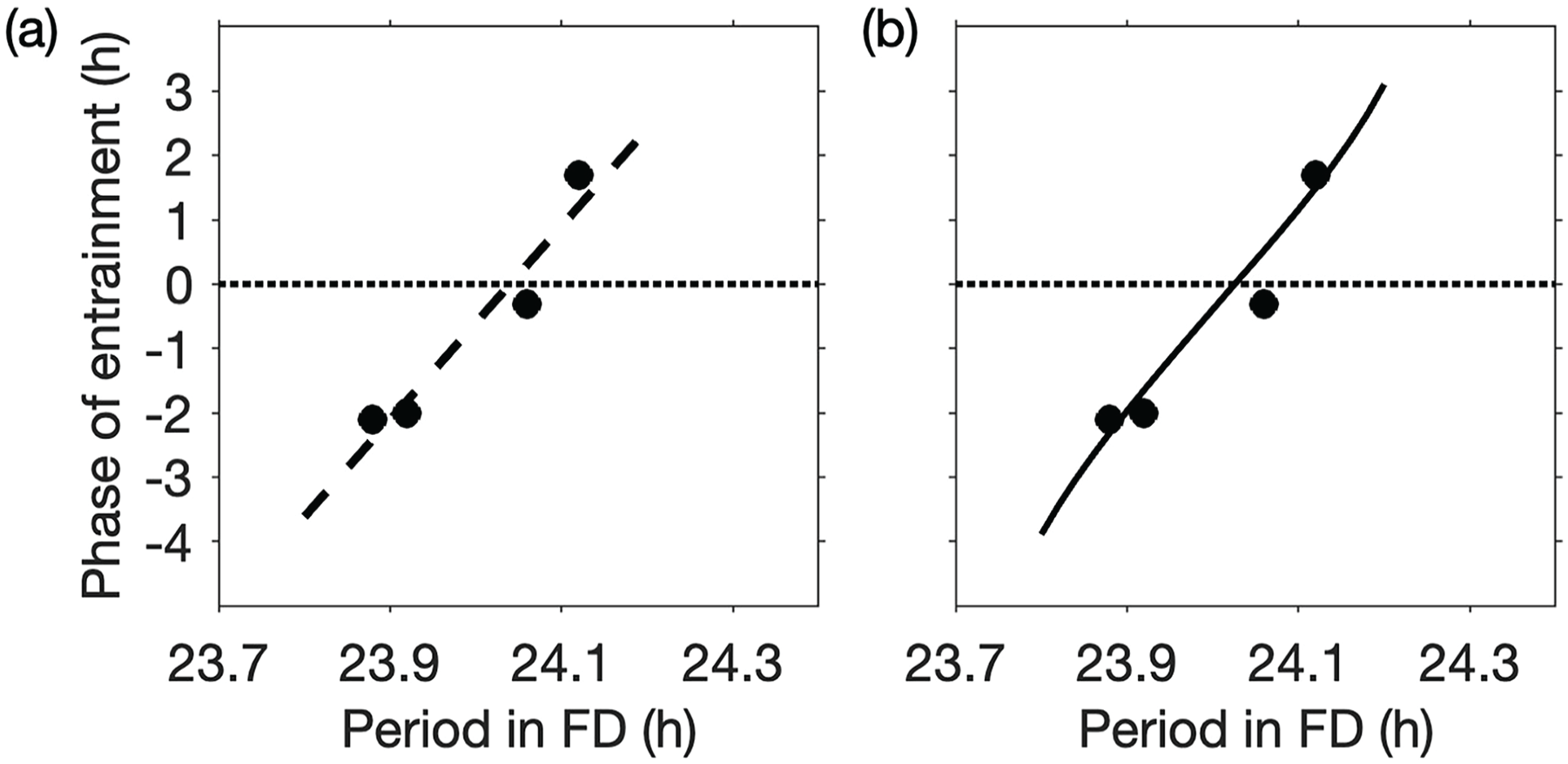
Phase of entrainment to 1.5 lux LD cycles with period
The map in equation (8) applies both in FD and during entrainment to LD cycles. Moreover, during entrainment, the clock completes exactly one cycle in each LD cycle, that is
where
where
In dim LD cycles and making the assumption that the VRC is sinusoidal, equation (12) gives:
Since
In deriving an explicit expression for the phase of entrainment
where the argument of the sine function involving
The phase of entrainment,
where
Substituting equation (21) into equation (20) and rearranging gives an explicit expression for the (stable) phase of entrainment
Since
and to the lowest order approximation, the parameter
Using measurements of
The linear regression line has been superimposed on the data in Figure 5a. Using these data and equations (23) and (24), we obtain
In another part of Wright’s study, 5 participants were exposed to dim LD cycles with period
Possible Explanation for the Difference Between the Period in FD and the Period in DD
Equation (14) relates the period in FD to the intrinsic period. Equation (14) contains the parameter
However, if
We expect that light of intensity approximately 15 lux, as frequently used in FD protocols, produces a stimulus of greater magnitude, but without further data we cannot describe how
Discussion
The intrinsic period of the central circadian pacemaker is an important parameter that determines the ability of the pacemaker to entrain, and the phase of entrainment, to LD cycles. FD carried out in dim light conditions is considered the gold standard method for measuring circadian period in humans (Dijk and Duffy, 2020; Wang et al., 2023).
We have used a phase-only parametric model to describe the effect of dim LD cycles on the assessment of period in FD. The key result is that the model-predicted period measured in FD,
where
Equation (25) suggests that FD gives a very accurate assessment of intrinsic circadian period provided the VRC to dim light has equal sized advance and delay regions
Our formal estimation of the error between intrinsic circadian period and period as measured in FD also confirms that the design of the protocol means that confounds due to dim light are small. Indeed, we emphasize that this confound is much smaller than that found for sighted individuals in classical free run (see Figure 1), and we therefore still expect FD to give a much more accurate estimate of intrinsic circadian period than classical free run.
Period Evaluation and Aftereffects
Several explanations have been offered for differences between the observed circadian period in blind and sighted individuals assessed in FD protocols (e.g. see Czeisler et al., 1999; Lockley et al., 2007; Lewy, 2007). The current dominant view is that observed differences are due to the presence of aftereffects, that is, that the period measured in FD in sighted people is a consequence of their prior entrainment to 24 h. In this scenario, after a sufficiently long time in DD, the period of sighted people would converge to the period observed in the blind. Such long-term transients have been observed in nocturnal rodents (Pittendrigh and Daan, 1976a).
The simple clock model cannot model aftereffects—it responds instantaneously to changes in the light environment, so the mechanism suggested here is fundamentally different. To capture aftereffects, amplitude-phase models (such as the van der Pol oscillator/Kronauer-type models) are required. Indeed, an amplitude-phase model that captures the dependence of measured circadian period on prior light exposure for a diurnal rodent (Bano-Otalora et al., 2021) has been constructed (see Usmani, 2022, Chapter 6).
To capture aftereffects, using an amplitude-phase model requires the parameter m to be small (e.g. in Usmani, 2022, to fit to the diurnal rodent data,
Period Evaluation and Physiological Changes Caused by Blindness
The only study we have found of sighted people in DD (Wever, 1979, n = 5) reported an average intrinsic period consistent with that in the blind. Nevertheless, given the small size of the study, a further explanation is that there are physiological changes in the blind that result in a fundamental change to the circadian system. For example, Yamazaki et al. (2002) show that in hamsters intrinsic circadian period is shorter and more variable following enucleation. Yamazaki et al. suggest a possible explanation is that coupling of retinal clocks with the suprachiasmatic nucleus is an important determinant of the intrinsic period. Hull (2009) also highlights a role for retinal clocks. If there are physiological differences to the circadian system that occur as a result of the lack of light perception, then it may be unrealistic to expect the same mathematical model to describe both blind and sighted people.
Period Evaluation and Data Selection
A further argument that has been used for the observation of a longer mean intrinsic period in the blind versus sighted people has been that data from studies in the blind have been biased toward those that do not entrain. The accepted value from FD for sighted people is 24.15 h with a standard deviation of 0.20 h (Duffy et al., 2011). In Hull (2009), it was argued that deviations of less than 0.10 h from 24 h could not be detected. If data are normally distributed, then 11% have an intrinsic period less than 23.90 h and 60% have an intrinsic period greater than 24.10 h. The mean of the 60% with a period greater than 24.10 h is 24.28 h. To find a mean value of 24.50 h requires selection of the 11% of people with an intrinsic period greater than 24.40 h. For context, in Flynn-Evans et al.’s (2014) study, 41 people with no light perception were studied. Of these, 16 (39%) did not entrain and had a mean period of 24.50 h, including 1 participant with an intrinsic period less than 24 h. Together, these results suggest that bias in data collection cannot explain the magnitude of the difference between sighted and blind individuals.
Period Evaluation and Stimulus
We estimated that light of intensity 1.5 lux produces a stimulus
where
Implications and Limitations
Mathematical models, combined with longitudinal light data collected from people in their natural every day environment, have been suggested as a non-invasive low-cost method to estimate circadian phase (Woelders et al., 2017). For day-to-day living, the estimates from models are comparable with DLMO. However, models have so far proved less accurate for irregular LD schedules as occur during shiftwork (Stone et al., 2019) and when the natural day length is short (Cheng et al., 2021). Current mathematical models are largely variants of those developed by Kronauer (e.g. Jewett and Kronauer, 1998).
One reason for reduced accuracy in shiftworkers may be that models do not currently adequately capture the response to light levels below 50 lux (e.g. see Figure 9 in St Hilaire et al., 2007) typical of night-working (Price et al., 2022). Inaccuracy in the modeling of the response to dim light could also explain the reduced accuracy for short natural photoperiods when observed light levels are typically lower (Shochat et al., 2019). One approach to the further development of models is to return to data collected in highly constrained laboratory environments and re-consider whether models adequately capture previous and newly available data, including data on spectral sensitivity of the human circadian pacemaker (e.g. St Hilaire et al., 2022). For example, Usmani (2022) highlights that current models cannot capture circadian phase alignment in both dim and bright light laboratory studies.
Here, we have focused on period assessment in dim light FD protocols. In dim light, the original Kronauer-type models (e.g. Jewett and Kronauer, 1998) describe a velocity response that is symmetric
A limitation of our hypothesis is that it depends on the value of the asymmetry parameter
We note that our results are consistent with previous simulations using Kronauer-type models in the relevant limits, that is, dim light, so that a phase-only model is reasonable, and close to symmetric VRC as occurs in Kronauer-type models. Specifically, the simulations of Lok et al. (2022) indicate that an FD protocol with a LD cycle length of 18 h gives a more accurate estimate of the intrinsic circadian period than a 28-h protocol. Stack et al. (2017) simulated an ultradian protocol of 4 h and FD protocols of 5 and 7 h and systematically varying light intensity, number of days in the protocol, and initial circadian phase. They found that more accurate estimates occurred when light intensity was low and the number of days and length of protocol further facilitated an even distribution of light across circadian phases. In dim light with
Finally, validated mathematical models describing the effects of light on the human circadian pacemaker are a prerequisite for understanding the effects of light exposure, which in our society is increasingly dominated by biologically effective human-made light. Novel technologies for monitoring this light exposure longitudinally in people going about their daily lives, combined with validated mathematical models, will enable a better prediction of the circadian health consequences of changes in policies related to light exposure and novel light sources.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304231215844 – Supplemental material for Mathematical Analysis of Light-sensitivity Related Challenges in Assessment of the Intrinsic Period of the Human Circadian Pacemaker
Supplemental material, sj-pdf-1-jbr-10.1177_07487304231215844 for Mathematical Analysis of Light-sensitivity Related Challenges in Assessment of the Intrinsic Period of the Human Circadian Pacemaker by Imran M. Usmani, Derk-Jan Dijk and Anne C. Skeldon in Journal of Biological Rhythms
Footnotes
Acknowledgements
IMU was supported by the Engineering and Physical Sciences Research Council (grant number: EP/R513350/1).
Author contributions
Conceptualization (IMU); mathematical analysis (IMU, ACS); supervision (ACS, D-JD); writing the manuscript (ACS, D-JD, IMU).
Conflict of interest statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
NOTE
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.