Abstract
Sex hormones are well known to modulate circadian timekeeping as well as the behavioral and physiological responses to circadian disruption. Gonadectomy, reducing the amount of circulating gonadal hormones, in males and females produces alterations to the free-running rhythm and the responses to light exposure by the central oscillator of the suprachiasmatic nucleus (SCN). In this study, we tested whether estradiol plays a role in regulating the circadian responses to acute (light pulses) and chronic light exposure (constant light [LL] vs standard light:dark [LD] cycle) in female C57BL6/NJ mice. Mice were either ovariectomized or given sham surgery and given a placebo (P) or estradiol (E) pellet for hormone replacement so that there were 6 groups: (1) LD/Sham, (2) LL/Sham, (3) LD/OVX + P, (4) LL/OVX + P, (5) LD/OVX + E, and (6) LL/OVX + E. After 65 days of light cycle exposure, blood and SCNs were removed and serum estradiol plus SCN estradiol receptor alpha (ERα) and estradiol receptor beta (ERβ) were measured via ELISA. The OVX + P mice exhibited shorter circadian periods and were more likely to become arrhythmic in LL compared with mice with intact estradiol (sham or E replacement mice). The OVX + P mice exhibited reduced circadian robustness (power) and reduced circadian locomotor activity in both LD and LL compared with sham controls or OVX + E mice. The OVX + P mice also exhibited later activity onsets in LD and attenuated phase delays, but not advances, when given a 15-min light pulse compared with estradiol intact mice. LL led to reductions in ERβ, but not ERα, regardless of the surgery type. These results indicate that estradiol can modulate the effects of light on the circadian timing system and that estradiol can enhance responses to light exposure and provide protection against a loss of circadian robustness.
Introduction
One increasingly common form of circadian disruption is exposure to light-at-night or light pollution. Not only can aberrant light exposure affect our sleep-wake cycle, but it can also desynchronize the central circadian oscillator, the suprachiasmatic nucleus (SCN), from all of the peripheral clocks in our bodies, which include brain areas downstream of the SCN and endocrine glands. Some of the behavioral responses to circadian misalignments are sex-dependent and are associated with sex-specific hormones, including differences in temperature regulation and metabolism (Qian et al., 2019). In addition, both human and animal research report that sex differences also exist regarding the circadian regulation of sleep (Santhi et al., 2016) and the prevalence of circadian disorders (e.g., sleep and phase syndromes—Reis and Paiva, 2019).
Previous research highlights links between a proper circadian functioning and gonadal hormone signaling, including estrogens and estrogen receptors (ERs). Estrogen is known to affect the transcription and phase of core clock genes within the SCN and peripheral tissues (Nakamura et al., 2001, 2005). Estrogen levels are also correlated with prokineticin 2 expression, an important output of the SCN (Xiao et al., 2014). In addition, ERs are located within the SCN in mice and humans (Kruijver and Swaab, 2002; Zuloaga et al., 2014). Activation of ERα (ERS1) and ERβ (ERS2) receptors can affect the transcription and translation of core clock genes, suggesting links between estrogen signaling and rhythmicity (Gery et al., 2007; Cai et al., 2008). At the behavioral level, mice with depleted estrogen levels via ovariectomy (OVX), compared with gonadally intact controls, display abnormal light-induced phase response curves and exhibit impaired rhythms (Mukai et al., 2008; Brockman et al., 2011). These studies illustrate that estrogen and ERs directly affect the circadian rhythm of behavioral outputs and the molecular clock.
Recent work from our lab has also shown that female mice which underwent OVX experience reduced circadian robustness and have an increased likelihood of becoming arrhythmic, independent of the reductions in overall activity levels (Michaud et al., 2022). However, as estradiol was not replaced after OVX in that study, it is not definitive that the estradiol itself mediated the effects of light exposure on circadian timing in those animals. This study aims to determine the relationship between ovarian estrogen and circadian light responses using OVX, with either estradiol or placebo supplementation, and non-OVX female C57BL/6NJ (B6NJ) mice. In the first experiment, we determined the effects of OVX on chronic or constant light exposure, and in the second experiment, we investigated how OVX modulates circadian phase shifting responses to acute photic stimuli.
Methods
All experiments listed below had the approval of Bridgewater State University’s Institutional Animal Care and Use Committee. All of the surgeries were done at Jackson Laboratories.
Experiment 1
Animals
Fifty-two female C57BL/6NJ (B6NJ—Strain #5304; Jackson Laboratories, Bar Harbor, ME, USA) mice were purchased at 9 weeks of age. Thirty-four mice underwent an OVX, while 18 mice experienced a Sham surgery. Afterward, 17 OVX mice received a 1.5 mg/90 day 17β-estradiol pellet (E), while 17 received a 1.5 mg/90 day placebo pellet (P); all sham mice received a 1.5 mg/90 day placebo pellet (Innovative Research of America, Sarasota, FL, USA).
Circadian Rhythm Monitoring
Upon arrival, the mice were acclimated to a 12:12 h light:dark (LD) cycle for 10 days, consuming regular chow (LabDiet5001, St. Louis, MO, USA) and water ad libitum. The mice were individually housed in infrared beam home-cage monitoring cages (StarrLife Sciences, Oakmount, PA, USA). After acclimation, half of each surgery type were placed into either ~175 lux constant light (LL) or the standard LD cycle. Thus, there were 6 groups: (1) SHAM/LD (n = 9), (2) SHAM/LL (n = 9), (3) OVX + P/LD (n = 8), (4) OVX + P/LL (n = 9), (5) OVX + E/LD (n = 8), and (6) OVX + E/LL (n = 9). All mice were exposed to their experimental lighting cycle for 64 days.
Estradiol and ERs
All the tissue collections were conducted during the middle of each animal’s inactive time (approximately ZT or CT 6) and in the light. Animals that were considered arrhythmic at the time of the tissue collection were tested during the middle of the experimental light cycle (real time), corresponding to ZT 6 for the animals held in the standard LD cycle. On the 65th day, the mice were euthanized via CO2 narcosis, their blood was collected, allowed to clot, and then centrifuged at 2000g at 4 °C for 20 min to obtain serum. The serum was used to measure 17β-estradiol via ELISAs (General Estradiol—MBS2700152; MyBioSource, San Diego, CA, USA). In addition, whole mouse brains were removed, rinsed in ice-cold phosphate-buffered saline (pH = 7.4), immediately frozen on dry ice, and stored in −80 °C. Afterward, the SCN was dissected manually into approximately 1 mm3 cubes and weighed. SCNs were removed by making 4 cuts immediately around the optic chiasm with very fine razor blade, followed by a single cut underneath. The SCN was visually identified using a dissecting microscope, by its location at the base of third ventricle and by locating the medial optic chiasm as a point of reference. SCN homogenates were created in a cocktail containing Pierce Lysis buffer and Halt protease inhibitor (Thermo Scientific, Rockford IL, USA), as previously described (Michaud et al., 2022). The supernatant was used in ERα (ESR1) (Mouse Estrogen Receptor Alpha—MBS776118) and ERβ (ESR2) (Mouse Estrogen Receptor Beta—MBS776119) ELISAs (MyBioSource). For each protein tested using ELISA, assay standards were run in triplicate, samples run in duplicate, using at least 2 plates per protein type and an intra-assay coefficient of variation (CV) of 20% or less.
Statistical Analyses
Chi-square periodograms were calculated using Clocklab’s (Actimetrics, Wilmette, IL, USA) batch analysis function (significance p = 0.001). The chi-square periodogram method was chosen due to having 64 continuous days of recording and with further analysis of intervals longer than 2 weeks in length. Two-way analyses of variance (ANOVAs) with Tukey post hoc pairwise comparisons were used to determine mean differences among the groups. In addition, 2-way repeated-measures ANOVAs with Scheffe-corrected post hoc analyses were conducted to ascertain any changes in free-running period, circadian power, and activity levels over the course of the 64-day photoperiod exposure (four 16-day increments) to determine whether these variables changed over time during the course of the experiment. A chi-square contingency table (with continuity correction) was conducted to determine whether the probability of a specific group becoming arrhythmic was more likely compared with others.
Experiment 2
Animals and Surgery
Twenty female B6NJ mice were purchased from Jackson Laboratories at approximately 9 weeks old. OVX was performed on 14 mice, while 6 received a sham surgery. Seven OVX mice received a 1.5 mg/90 day 17β-estradiol pellet (E), while the other 7 OVX mice received a 1.5 mg/90 day placebo pellet (P); the 6 mice that underwent the sham surgery also received a 1.5 mg/90 day placebo pellet (Innovative Research of America). There were 3 groups: (1) Sham (n = 6), (2) OVX + P (n = 7), and (3) OVX + E (n = 7). After recovery from the surgeries, the mice were shipped to Bridgewater State University and given standard chow and water ad libitum. The mice were placed into a standard 12:12 LD cycle and in circadian home-cage monitoring cages, as described in Experiment 1.
Assessment of Light-Induced Phase Shifts
After a 3-week entrainment period, all animals were tested sequentially for responses to phase-delaying and phase-advancing light pulses (15 min, ~175 lux) using the Aschoff type II protocol. In this protocol, animals are kept under an entraining LD cycle until the day of light pulse presentation and then subsequently placed in constant darkness (DD) for 1 week to assess the phase of the free-running rhythm. Light pulses were administered at phases expected to yield phase delays (ZT 15) and phase advances (ZT 21). Successive phase-response tests were separated at 3 weeks of re-exposure to the LD cycle, ensuring stable entrainment prior to the delivery of all light pulses.
Estradiol Measurements
Following the ZT 21 light pulse, mice were re-entrained to the 12:12 LD cycle for 3 weeks. Mice were euthanized and their blood was collected and processed as described in Experiment 1. The serum was tested for 17β-estradiol using ELISAs described in Experiment 1.
Statistical Analyses
Activity onsets were calculated for animals during the initial LD phase, using ClockLab’s automated activity-onset detection algorithm with manual correction of inaccurate onset markers performed by an experimenter blind to experimental group. Activity onsets were defined as a minimum of 3 consecutive 10-min activity bouts above the daily activity average after 3 consecutive hours of 10-min activity bouts below the daily activity average. The magnitude and direction of circadian phase responses were determined using the aforementioned method for activity-onset detection. Prestimulus phase was estimated as the mean time of activity onset over the last 7 days of LD entrainment, and poststimulus phase was estimated by a regression line fit to activity onsets over 7 DD free-running circadian cycles following the test stimulus, excluding the first two activity onsets due to the possible occurrence of “transients” prior to the establishment of a steady-state free-running phase; phase responses were then determined as the difference between these 2 phase estimates’ phase responses (Seggio et al., 2009). One-way ANOVAs were conducted to determine mean differences among the groups for all dependent variables.
Results
Experiment 1
Circadian Activity
Representative actograms are presented in Figure 1a-1d. All animals regardless of the surgery type were able to entrain to the standard LD cycle. For the overall 64-day interval, an interaction (Light Cycle × Surgery Type) was found for circadian period (F2,46 = 15.24, p < 0.001). OVX + P/LL mice had shorter circadian periods compared with OVX + E/LL (p < 0.001) and Sham/LL (p < 0.001) (Figure 2a). An interaction was also found for rhythm power (F2,46 = 15.38, p < 0.001), where OVX + P/LL mice exhibited reduced power compared with OVX + E/LL (p < 0.001) and Sham/LL (p < 0.001) (Figure 2c). While no interaction was found for circadian activity (F2,46 = 0.78, p = 0.47), both LL (F1,46 = 7.30, p = 0.010) and OVX without estrogen replacement (F2,46 = 45.69, p < 0.001) reduced circadian home-cage activity levels (Figure 2d).
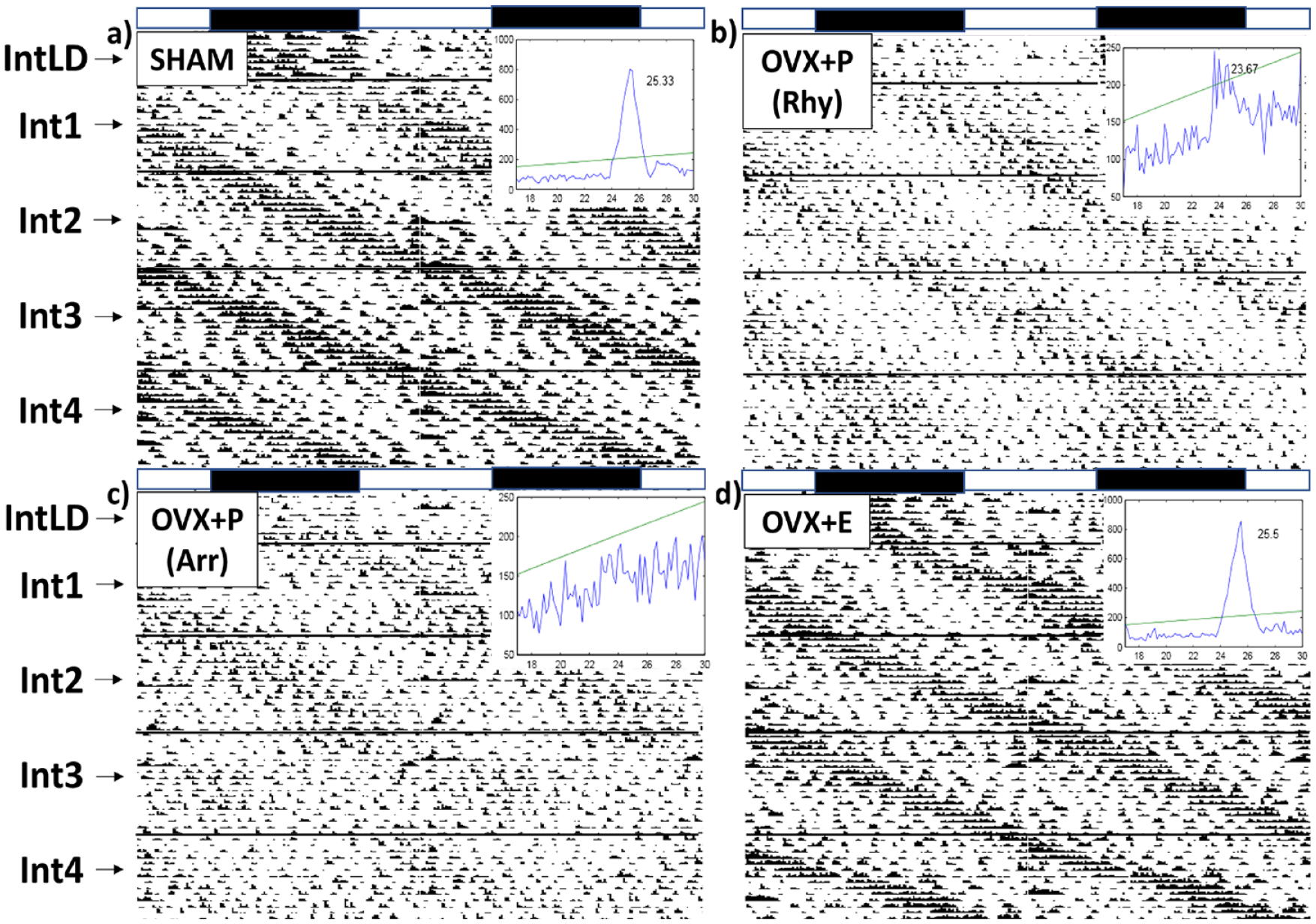
Representative actograms: Experiment 1. Actograms are divided into 5 intervals (Int): IntLD: LD entrainment before placement into LL, Int1: first 16-day LL interval, Int2: second 16-day LL interval, Int3: third 16-day LL interval, and Int4: fourth 16-day LL interval. (a) Sham representative actogram and Qp during Int4, (b) OVX + P rhythmic (Rhy) representative actogram and Qp during Int4, (c) OVX + P arrhythmic (Arr) representative actogram and Qp during Int4, and (d) OVX + E representative actogram and Qp during Int4. Inserted graph into each actogram: Qp, which is the power of the chi-square periodogram, is used to measure the robustness of circadian rhythms. LL = constant light; OVX = ovariectomy.
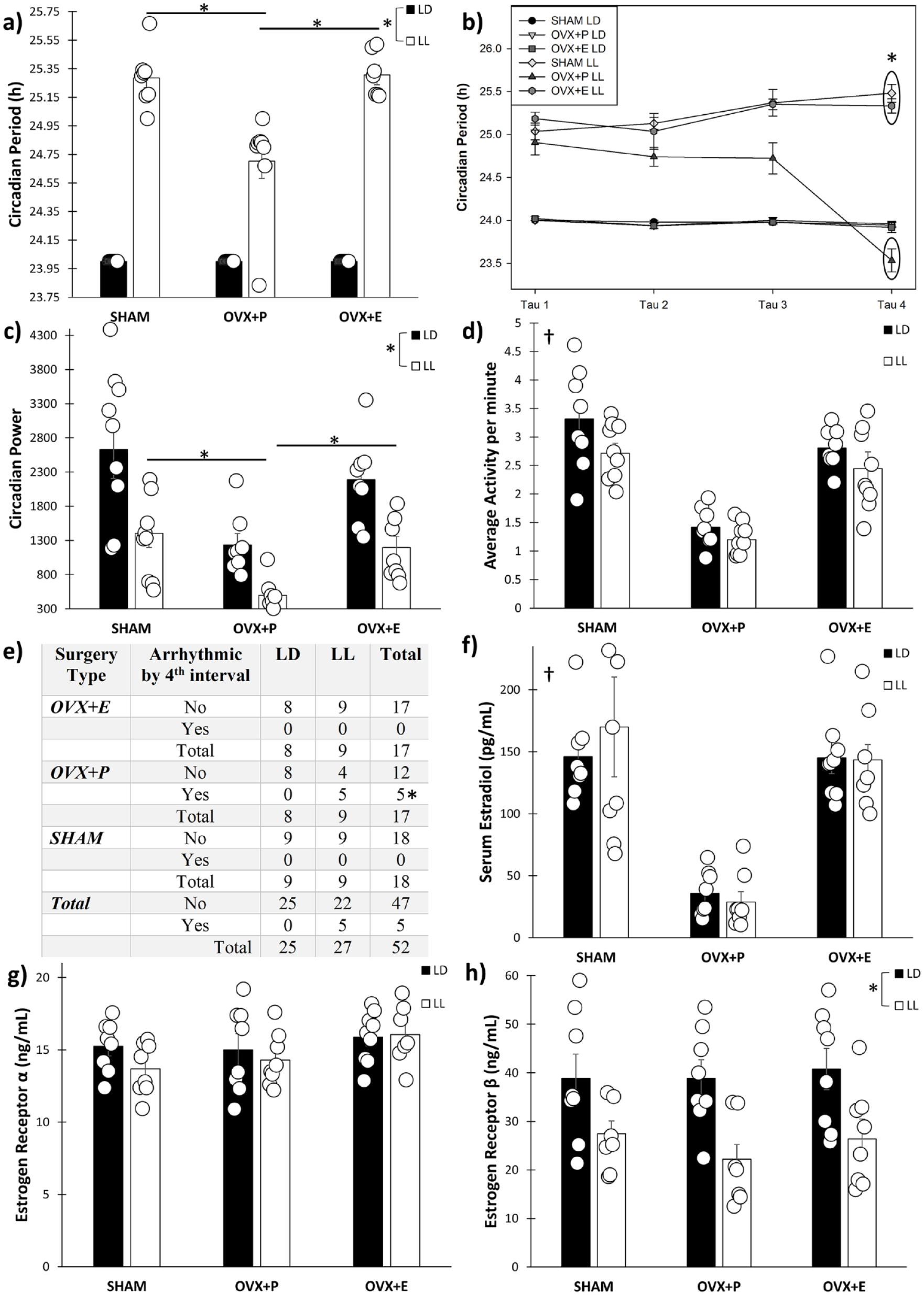
Experiment 1. (a) Circadian period during the entire 64-day light cycle exposure, (b) circadian period over time divided by interval, separated into 16-day increments, (c) circadian Power during the entire 64-day light cycle exposure, (d) home-cage circadian locomotor activity during the entire 64-day light cycle exposure, (e) number of animals that became arrhythmic during the fourth 16-day interval, (f) serum 17β-estradiol, (g) SCN ERα, and (h) SCN Erβ. SCN = suprachiasmatic nucleus; ERα = estradiol receptor alpha; Erβ = estradiol receptor beta; LD = light:dark; OVX = ovariectomy; LL = constant light.
Regarding changes in circadian rhythms over the course of the 64 days, a within-subjects interaction was found for Light Cycle × Surgery Type × Tau (F6,120 = 12.57, p < 0.001). OVX + P/LL mice that were rhythmic exhibited shorter free-running periods during the fourth 16-day interval compared with the other 3 (all p < 0.001), while circadian periods for both Sham and OVX + E mice did not change over time (all p > 0.85) (Figure 2b). Circadian power (F6,120 = 1.21, p = 0.31) and activity levels (F6,135 = 1.87, p = 0.091) did not change over the course of the experiment for any of the groups. Five of the 9 OVX + P/LL mice became arrhythmic before the end of the experiment (the fourth 16-day interval), while none of the OVX + E or Sham mice became arrhythmic in LL. As such, OVX + P/LL had a statistically significant increased probability of being arrhythmic compared with OVX + E and Sham mice in LL (χ = 3.91, p = 0.048) (Figure 2e).
Estradiol and ER Levels
As expected, OVX + P mice exhibited reduced serum 17β-estradiol (F2,42 = 27.93, p < 0.001) compared with OVX + E (p = 0.001) and Sham mice (p < 0.001), but LL had no effects (F1,42 = 0.12, p = 0.73) (Figure 2f). While no differences among any of the groups were found for ERα (ESR1) within the SCN (F2,42 = 0.70, p = 0.51) (Figure 2g), LL produced reductions in ERβ (ESR2) levels compared with animals in LD, in all surgery conditions (F1,40 = 16.09, p < 0.001) (Figure 2h).
Experiment 2
Representative actograms illustrating phase-delay responses are presented in Figures 3a-3c. OVX + P mice exhibited significantly reduced phase delays in response to a light pulse at ZT 15 (F2,16 = 4.71, p = 0.023), compared with OVX + E (p = 0.034) and Sham mice (p = 0.047) (Figure 3d); no differences were found among the groups for phase advances (F2,17 = 0.31, p = 0.74) (Figure 3e). The onset of daily activity (i.e., the start of the active period using activity onsets as defined in the methods) in a standard LD cycle was significantly later for OVX + P mice (F2,17 = 7.06, p = 0.004) compared with OVX + E (p = 0.005, approximately 0.5 h) and Sham mice (p = 0.050, approximately 0.33 h). Serum 17β-estradiol was significantly lower in OVX + P mice (F2,17 = 4.97, p = 0.020) compared with OVX + E mice (p = 0.033) and Sham controls (p = 0.043) (Figure 3f).
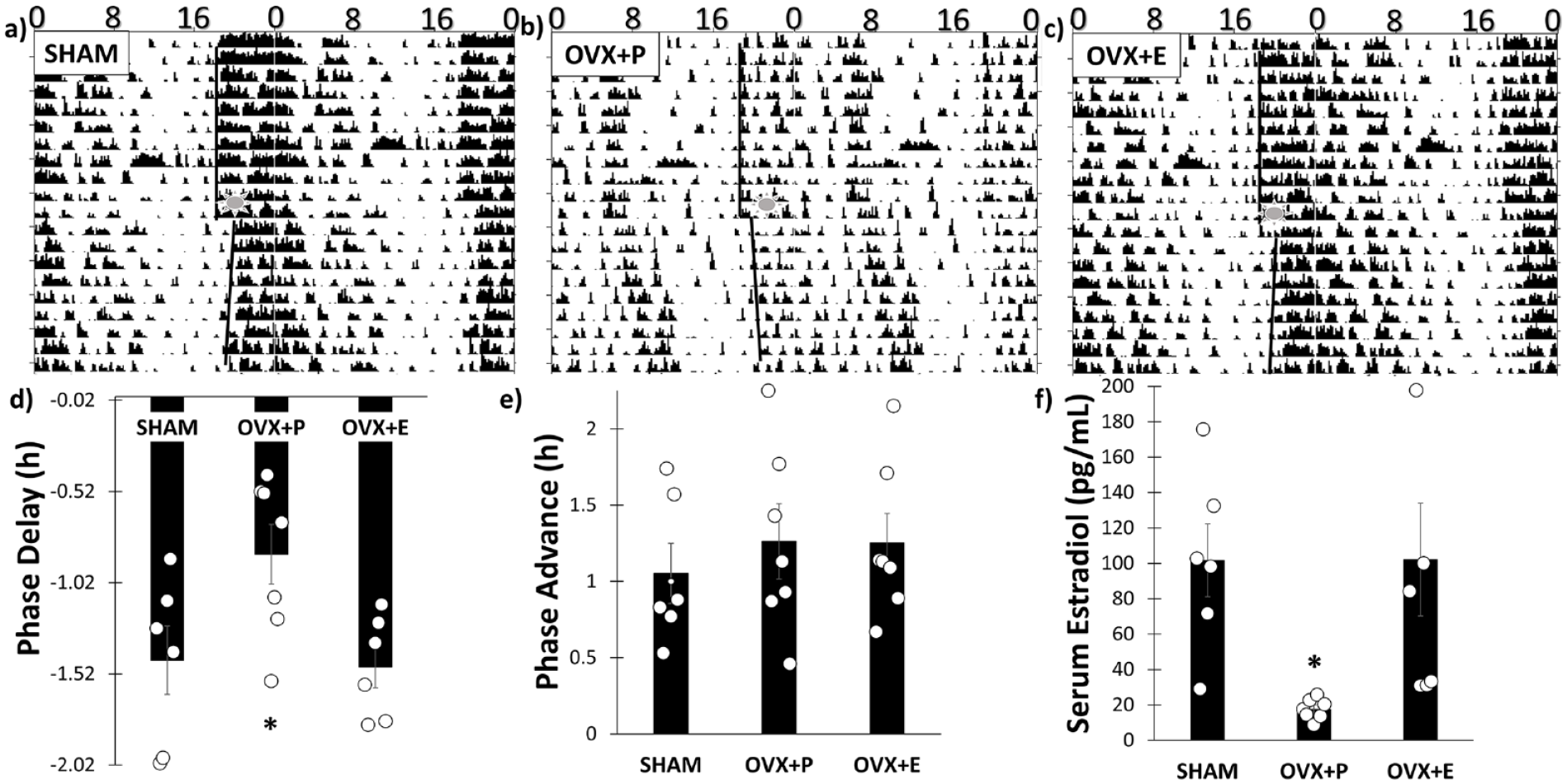
Experiment 2. (a) Sham Representative actogram for light pulse at ZT 15, (b) OVX + P representative actogram for light pulse at ZT 15, (c) OVX + E representative actogram for light pulse at ZT 15, (d) phase delays were significantly blunted in OVX + P mice compared with SHAM and OVX + E mice, (e) phase advances were not significantly different among the groups, and (f) 17β-estradiol was reduced in OVX + P compared with OVX + E and SHAM. OVX = ovariectomy.
Discussion
In this study, we show that mice with depleted estrogen due to OVX exhibit impairments in circadian responses to both chronic and acute light exposure. Mice with depleted estrogen through OVX exhibited significantly different circadian periods in LL compared with mice with intact estrogen signaling. Specifically, the circadian rhythm in LL exhibited period shortening over time in the OVX mice, but not the Sham operated or the OVX mice with estradiol replacement. The period shortening seen here in OVX mice in LL is in contrast to DD, where OVX by itself produces either period lengthening in hamsters (Morin et al., 1977) or no change to the free-running period in female mice (Iwahana et al., 2008). Meanwhile, gonadectomy in male mice leads to lengthened free-running periods (Karatsoreos et al., 2007; Iwahana et al., 2008; Brockman et al., 2011), so there seems to be sex and species differences in how gonadal hormones regulate the behavioral rhythm. Nevertheless, both androgen and estradiol treatment tend to shorten periods and cause an advance in activity onset in DD (Morin et al., 1977; Iwahana et al., 2008). which may also explain why gonadectomized animals (male or female) in LD or DD exhibit later activity onsets compared with estrogen and ER intact controls (Brockman et al., 2011; Blattner and Mahoney, 2014; Michaud et al., 2022, current study).
While sex hormones have clear roles in regulating the timing of the circadian clock, the results from this study and others also indicate that estrogens and ERs also modulate how photic inputs to the circadian timing systems are regulated. The period shortening seen in the OVX + P mice placed in LL in this study may indicate reduced circadian photosensitivity, rather than estradiol modulating the periodicity of the clock. The length of the free-running period in LL is directly proportional to the light intensity and circadian photosensitivity of the organism. Both melanopsin knock-out (KO) mice (with reduced light cues to the SCN) and human and animal models experiencing blindness also exhibit shorter periods in lengthened light exposure (Ruby et al., 2002; Lockley et al., 2007; Deane et al., 2021). Another study also reported that the greater the degree of light intensity, the longer the circadian period (Steinlechner et al., 2002). These effects may be mediated by ERs. For example, both male and female ERα (ESR1) KO mice exhibit slightly shorter circadian periods compared with wild-type animals when placed into LL (Blattner and Mahoney, 2012). In particular, ERβ (ESR2) is expressed in the SCN in female mice and humans (but not rats or hamsters) (Kruijver and Swaab, 2002; Zuloaga et al., 2014). In this study, LL led to decreases in ERβ levels, which may be due how photic inputs regulate circadian gene expression within the SCN, subsequently modulating ERβ levels. ERβ expression is cyclically regulated by the circadian genes period1, period2, clock, and bmal1, where clock and bmal1 lead to increased levels, while period expression leads to decreases in ERβ (Cai et al., 2008). As LL leads to increased period expression, since period is photic-transcribed (Shigeyoshi et al., 1997), this may be a reason why ERβ levels within the SCN were reduced in the female mice in this study. While all groups experienced a reduction in ERβ, the reduction in ERβ still may be the cause for why OVX animals exhibited period shortening and increased propensity for arrhythmicity in LL compared with animals with intact estrogen signaling (i.e., less estrogen binding to fewer receptors). However, estrogen binding levels or efficacy nor period transcripts were not directly measured in this study, so future work is still needed to determine the precise mechanism of how intra-SCN ERβ affects circadian responses to light exposure.
It is also worth noting that all estradiol and ERs were measured only for single time point during the middle of the animal’s inactive period (ZT 6 or CT 6) and in the light. While the circadian locomotor behavior of the animals was in phase, other physiological rhythms may be out of alignment between the LD and LL groups and among the 3 groups in LL. In addition, if the ER levels were tested at different time points, perhaps different results would have been obtained. ERs are known to move among the nucleus, cytosol, and membrane depending upon time of day in many different tissue types (Levin, 2009). So although no differences were found in ERα levels, that does not necessarily preclude a role for ERα or membrane-bound ERs in response to continuous light exposure or acute photic stimuli, given that our assessment measures for ERs do not account for cellular localization, and in light that removal of the ER genes leads to altered rhythmicity in both sexes. Systemically, estradiol is known to produce anxiolytic effects by directly modulating the hypothalamic-pituitary adrenal (HPA) axis (Liu et al., 2012), although stress and anxiety-like behaviors were not measured in this study. LL can be considered stressful and activate the HPA axis, leading to glucocorticoid secretion, which in turn can negatively affect estradiol and ER signaling (Isgor et al., 2003; Romano-Torres et al., 2010).
Not only does OVX alter responses to chronic light, OVX also produces blunted responses to acute light exposure. Mice that underwent OVX without estrogen replacement exhibited reduced circadian phase shifting in response to light pulses during the early night, but not the late night. This result corroborates with a previous study that also found blunted phase delays, but not advances, in OVX mice in the B6 background (Brockman et al., 2011). These results also indicate that OVX leads to irregular reactions to photic inputs to the circadian clock and that estrogen supplementation is able to restore normal light responses. Female ERα KO mice also exhibit reduced phase delays in response to acute light pulses, indicating a role of ERs and estrogen signaling in the regulation of photic responsiveness (Blattner and Mahoney, 2013). Both estrogen-cycling SHAM animals and non-estrogen-cycling OVX + E mice exhibited similar phase responses to acute light exposure during the night. A previous study investigating responses to 15-min light pulses during the different time points in the time of the estrus cycle (i.e., proestrus, estrus, metestrus, and diestrus) found that neither the steady-state phase shifts nor immediate phase shifts were significantly different from each other during the subjective night (Mizuta et al., 2018). Interestingly, female mice in proestrus (higher levels of estradiol) shift and resynchronize to a 6-hour phase advance behaviorally and in peripheral tissues more quickly compared with mice in metestrus (lower levels of estradiol) independent of the SCN (Pilorz et al., 2020). Taken together, these results imply that the estrous cycle may not directly regulate photic entrainment, but rather estradiol may exert its effect downstream of the SCN through peripheral oscillators or modulate photic responsiveness through brain areas responsive to estrogen signaling, which may or may not include the SCN.
While both male and female mice exhibit reduced phase delays when gonadectomized, this effect seems to be much stronger in females than in males (Brockman et al., 2011), indicating a sex and hormonal difference. The mechanism behind these reductions in light-induced phase responses may be different in males and females. For example, castration in males reduces the number of photic-induced c-fos neurons within the SCN (Karatsoreos et al., 2007), but OVX in females (Brockman et al., 2011) or ER KO (Blattner and Mahoney, 2013) does not affect photic-induced c-fos expression (c-fos being a marker for photic inputs to the SCN). In addition, OVX does not significantly change vasoactive intestinal polypeptide (VIP) levels within the SCN of female rodents nor does estradiol application modify vasoactive intestinal polypeptide (VIP) signaling in LD (Krajnak et al., 1998). Future studies connecting estrogen signaling and photic inputs to the circadian timing system would be of great interest to determine why depleted estrogen signaling affects responses to light exposure.
In addition to abnormal responses to both chronic and acute light exposure, mice which underwent OVX without estradiol implantation exhibited significantly reduced rhythm cohesiveness and robustness (circadian power) and an increased likelihood of becoming arrhythmic by the end of the experiment compared with Sham controls and OVX animals with an estradiol pellet. These results are consistent with previously published work also showing that OVX leads to irregular behavioral rhythms and that estradiol replacement can rescue these abnormalities (Thomas and Armstrong, 1989; Michaud et al., 2022). Interestingly, these effects are once again relegated to LL—if the OVX animals are placed back into DD, they resume exhibiting a normal free-running rhythm with no differences in period length (Thomas and Armstrong, 1989). Other studies also report abnormal behavioral and physiological rhythms in OVX animals even in LD (Wollnik and Döhler, 1986; Yin et al., 2019). These results implicate the role of estrogens in modulating photic signaling to the circadian clock as reductions in estradiol lead to significant variations in rhythmic outputs in both LD and LL.
In conclusion, OVX mice without estradiol treatment exhibited poorer circadian responses to both chronic and acute light exposure. Estradiol implantation was able to prevent abnormal responses to light exposure in LL and light-at-night (phase shifts) in the OVX animals. The results presented in this study implicate estradiol and its circulation in regulating photic signaling to the circadian timing system and engendering a synchronous, cohesive circadian rhythm in female animals. This study supports the notion of the importance of estrogen signaling on biological timing, which may improve our understanding of how sex hormones regulate nonreproductive biological processes and provide a basis for future research and clinical work.
