Abstract
Astrocytes are densely present in the suprachiasmatic nucleus (SCN), which is the master circadian oscillator in mammals, and are presumed to play a key role in circadian oscillation. However, specific astrocytic molecules that regulate the circadian clock are not yet well understood. In our study, we found that the water channel aquaporin-4 (AQP4) was abundantly expressed in SCN astrocytes, and we further examined its circadian role using AQP4-knockout mice. There was no prominent difference in circadian behavioral rhythms between Aqp4-/- and Aqp4+/+ mice subjected to light-dark cycles and constant dark conditions. However, exposure to constant light induced a greater decrease in the Aqp4-/- mice rhythmicity. Although the damped rhythm in long-term constant light recovered after transfer to constant dark conditions in both genotypes, the period until the reappearance of original rhythmicity was severely prolonged in Aqp4-/- mice. In conclusion, AQP4 absence exacerbates the prolonged light-induced impairment of circadian oscillations and delays their recovery to normal rhythmicity.
Keywords
The hypothalamic suprachiasmatic nucleus (SCN) is the master circadian clock that dictates the daily behavior and physiology cycles (Herzog et al., 2017). Cellular rhythms generated by the cell-autonomous transcription-translation feedback loop (TTFL) of clock genes (Takahashi, 2017) synchronize to produce a robust and coherent daily oscillation through circuit-level interactions among SCN neurons (Yamaguchi et al., 2003; Hastings et al., 2020). In addition to neurons, astrocytes are densely distributed in the SCN and encircle synaptic elements, neuronal somata, and capillaries (Tamada et al., 1998; Ibata et al., 1999). Now it is established that astrocytes enriched in the SCN play a critical role in generating circadian rhythms through their metabolic interaction with neurons inside the SCN (Tso et al., 2017; Brancaccio et al., 2017, 2019).
Since the SCN is the key site governing all circadian physiology of the body, we have been progressing to identify key SCN signaling molecules that are specific for circadian generation and synchronization (Okamura, 2007). Through this project, we identified the circadian roles of Rgs16 (Doi et al., 2011), vasopressin (Yamaguchi et al., 2013), and Gem (Matsuo et al., 2022). In the present study, we focused on the substances expressed in SCN astrocytes. In search of targets by Allen Mouse Brain Atlas website, we found that aquaporin 4 (AQP4) mRNA was abundantly expressed in the SCN (Suppl. Fig. S1).
Water channel aquaporins are approximately 30-kDa proteins that form 6 transmembrane water channels as homotetramers in the cell membrane and are involved in the uptake and release of water in cells (Nagelhus and Ottersen, 2013). In mammals, aquaporins have 14 homologs that are distributed in most cells of the body (Papadopoulos and Verkman, 2013). Aquaporin 4 (AQP4) is most abundantly expressed in the brain and is ubiquitous in ependymal cells and astrocytes, especially in specialized membrane domains, including astroglial end-feet ensheathing blood vessels and glutamatergic synapses (Nielsen et al., 1997; Nagelhus et al., 2004). AQP4 plays a crucial role in water movement in the brain (Mestre et al., 2018; Hablitz et al., 2020), and is speculated to be pathophysiologically involved in cerebral edema, which causes abnormal water retention in the brain parenchyma (Manley et al., 2000), and demyelination (neuromyelitis), which causes severe visual loss due to myelin sheath destruction in the optic nerve and spinal cord (neuromyelitis optica [NMO]) (Abe and Yasui, 2022).
In our study, we examined the circadian role of AQP4 abundantly expressed in the SCN by using AQP4-knockout (Aqp4-/-) mice (Ikeshima-Kataoka et al., 2013) (see Supplemental methods). We used male mice to avoid the effect of hormonal cycle on circadian rhythms. Before examining the physiology, we first conducted morphological analysis of AQP4 expression in the SCN which revealed dense AQP4-immunoreactive structures in the SCN in Aqp4+/+ but not in Aqp4-/- mice (Suppl. Fig. S2A and S2B). Strong astroglial AQP4 immunoreactivity was observed in Aqp4+/+ mice in the central and ventrolateral portions of the nucleus (Suppl. Fig. S2A arrows). Outside the SCN, AQP4 immunoreactivity was observed in the basal part of ependymal cells and optic chiasma astrocytic cover attached to the pia mater, as reported previously (Frigeri et al., 1995; Nagelhus and Ottersen, 2013). Using double immunofluorescence (Alexa488-labeled GFAP and Alexa594-labeled AQP4) in Aqp4+/+ mice (Suppl. Fig. S2 C) by confocal microscopy, we found that GFAP immunoreactivity was localized mostly in the cell body and proximal processes in astrocytes, but that of well-stained AQP4 was found in the distal processes encircling blood vessels (Suppl. Fig. S2 C asterisks), which were only weakly labeled by GFAP. These results are consistent with the electron microscopic findings of highly polarized AQP4 localization in the basal plasma membranes of astrocytes facing blood vessels (Nagelhus et al., 2004; Nielsen et al., 1997). Moreover, diffusely stained AQP4 dots were evident in the central and ventrolateral portions of the Aqp4+/+ mice SCN (thin arrows; Suppl. Fig. S2 C). These were completely absent in the Aqp4-/- mice (Suppl. Fig. S2D), although clear cobweb-like extensions and processes with GFAP immunoreactivity were detected in the SCN.
We examined whether circadian behavioral profiles were changed in Aqp4-/- mice. Mice housed in 12 h:12 h LD cycles for more than 2 weeks were subsequently exposed to DD conditions. In both genotypes, behavioral rhythms easily entrained to the LD cycle, and the period length in DD were virtually the same (Aqp4+/+:23.81 ± 0.02 h; Aqp4-/-:23.92 ± 0.05 h; n = 8) (Figure 1a-1c). The phase shifts at CT14 induced by a brief light pulse in DD were not different between the 2 genotypes (Aqp4+/+:2.11 ± 0.07 h; Aqp4-/-:2.14 ± 0.25 h; n = 8) (Figure 1d).
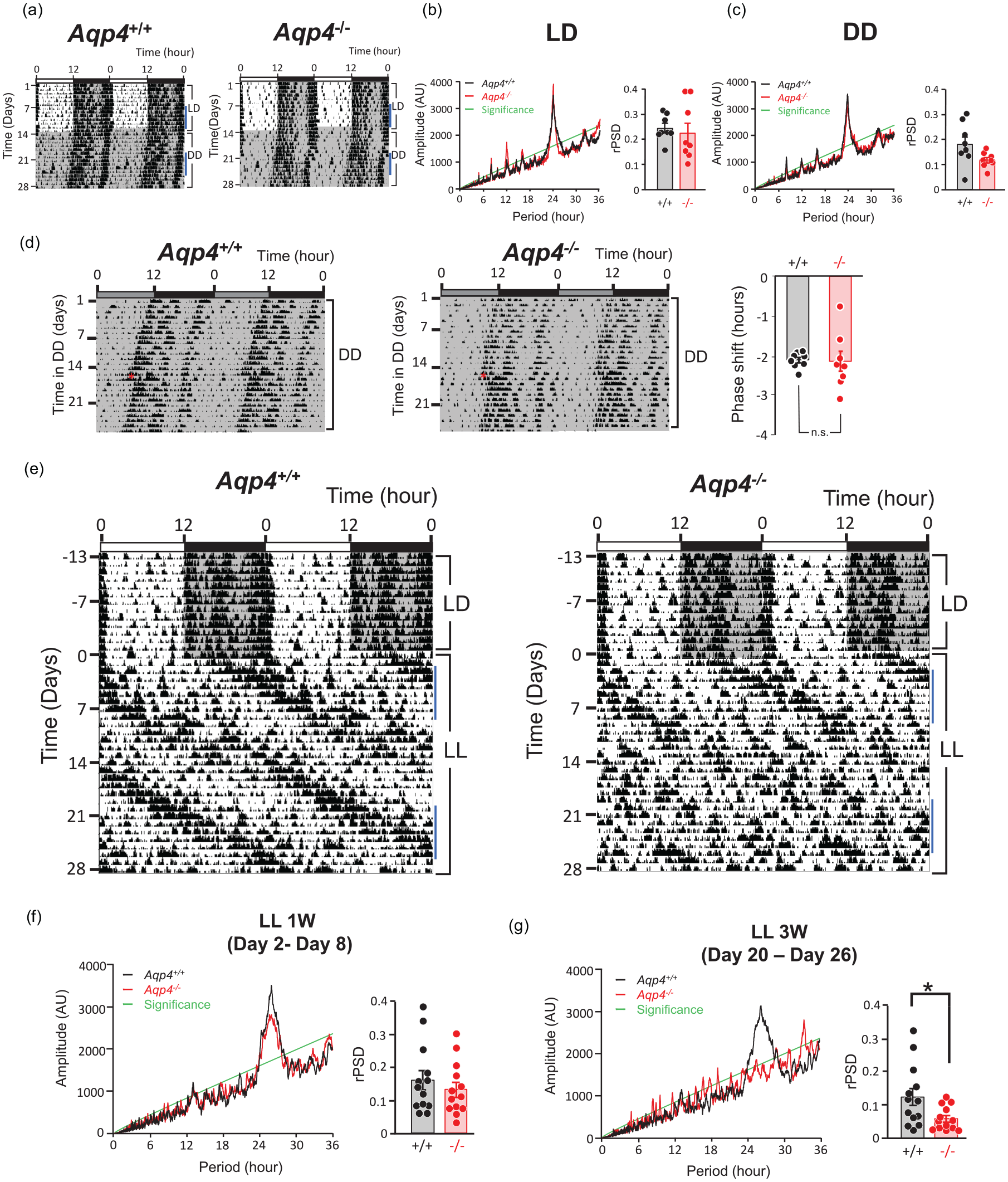
(a) Representative Aqp4+/+ and Aqp4-/- mice actograms in 12 h:12 h LD cycles, followed by DD. The horizontal axis represents the zeitgeber time (ZT), whereas the vertical axis represents the experiment days. Dark periods are depicted in gray backgrounds. Actograms are depicted in double-plotted form. (b, c) Representative χ2 periodograms and rPSD (see text or detail) for Aqp4+/+ (black) and Aqp4-/- (red) mice in LD (b) and DD (c). Data were evaluated on 7 days activity rhythms as indicated by blue line in (a). The green line indicates 0.001 confidence interval. (d) Representative actograms (left) and statistical analysis (right) of Aqp4+/+ and Aqp4-/- mice housed under DD conditions exposed to a brief light (200 lux, 30 min) at CT14. Statistical analysis of the phase shift magnitude was conducted using Welch’s t-test. Data are presented as the mean ± standard error of the mean (n = 8). (e) Representative Aqp4+/+ and Aqp4-/- mice actograms housed under 12 h: 12 h LD conditions, then transferred to prolonged LL conditions. The first day of exposure to LL is depicted as day 1. (f) Representative χ2 periodograms and rPSD for Aqp4+/+ (black) and Aqp4-/- (red) mice in early LL days (1 W: day 2- day 8). (g) Representative χ2 periodograms and rPSD for Aqp4+/+ (black) and Aqp4-/- (red) mice in prolonged LL days (3 W: day 20-26) as indicated by blue line in (e). Data were evaluated on each 7 days of activity rhythm. The green line indicates 0.001 confidence interval. p < 0.05, Welch’s t-test. Abbreviations: DD = constant darkness; rPSD = relative power spectrum distribution; LD = light-dark cycles; LL = constant light.
As depicted in representative actograms and χ2 periodograms, there were no obvious changes in the amplitude of rhythms in Aqp4-/- mice under both LD and DD conditions (Figure 1b and 1c). We further statistically evaluated the circadian rhythmicity amplitude in Aqp4+/+ and Aqp4-/- mice by measuring the rPSD of the dominant 24 h rhythm produced from a Fast Fourier transform (FFT) analysis of 7 days of animal activity (Siepka and Takahashi, 2005), but we found no difference in the rhythmicity strength in LD and DD between these genotypes (Figure 1b and 1c). These results indicate that AQP4 deletion does not alter endogenous circadian rhythmicity or entrainment ability to external light.
Since it is known that continuous (constant) light exposure (LL) prolongs the endogenous circadian period (Daan and Pittendrigh, 1976; Ohta et al., 2005; Doi et al., 2019), perturbing and damping rhythm oscillations in the SCN (Ohta et al., 2005; Chen et al., 2008), we examined the effect of prolonged LL exposure on circadian behavioral rhythms in Aqp4-/- mice (Figure 1e). Locomotor activity rhythm periods in LL were prolonged with a gradual decrease in rhythmicity in the Aqp4+/+ and Aqp4-/- genotypes. At 3 weeks after LL (between LL day 20 and 26), among 13 mice examined in each genotype, we found that the phenotypic arrhythmicity judged by actograms and χ2 periodograms were 7 in Aqp4+/+ mice and 10 in Aqp4-/- mice. We further evaluated this phenotypic impression by measuring the rPSD for the first week of LL (between days 2 and 8 after LL initiation) (Figure 1f) and the third week of LL (between day 20 and 26 after LL initiation) (Figure 1 g). Although FFT analysis for the first week in LL demonstrated a decrease in rhythmicity in both genotypes (Figure 1f), we found that Aqp4-/- mice depicted a more severe reduction than Aqp4+/+ mice at 3 weeks (Aqp4+/+:0.1209 ± 0.0262; Aqp4-/-:0.0557 ± 0.0101; p = 0.034, n = 13, Welch’s t-test) (Figure 1 g).
Previously, Chen et al. (2008) demonstrated that the damped oscillation of behavioral rhythms induced by the LL condition quickly reappeared immediately post LL cessation in wild-type C57BL6 mice. Since they also demonstrated that the reappearance accompanied the Per rhythm in the SCN, they speculated that the behavioral rhythms recovery might be derived from the SCN functional clock recovery. Here, we examined whether AQP4 deletion affects the reappearance of the rhythm in DD after severe dampening of rhythm due to prolonged LL exposure.
We transferred phenotypically arrhythmic mice of each genotype (n =5 for each genotype) after 4 weeks of LL, as judged by the representative actograms and χ2 periodograms, to DD. In Aqp4+/+ mice, the activity rhythm immediately reappeared after transfer to DD, and the original circadian period length was stabilized almost from the beginning (Figure 2a left). Conversely, Aqp4-/- mice demonstrated a pattern of behavior different from that of Aqp4+/+ mice after transfer to DD (Figure 2a right). Unstable activity onset and offset were observed for much longer, and, for activity offset time, it took approximately 9 days to restabilize at original intervals (Figure 2b; Aqp4+/+:4.40 ± 0.40 days; Aqp4-/-:9.60 ± 0.87 days; p = 0.00063, n = 5, Student’s t-test).
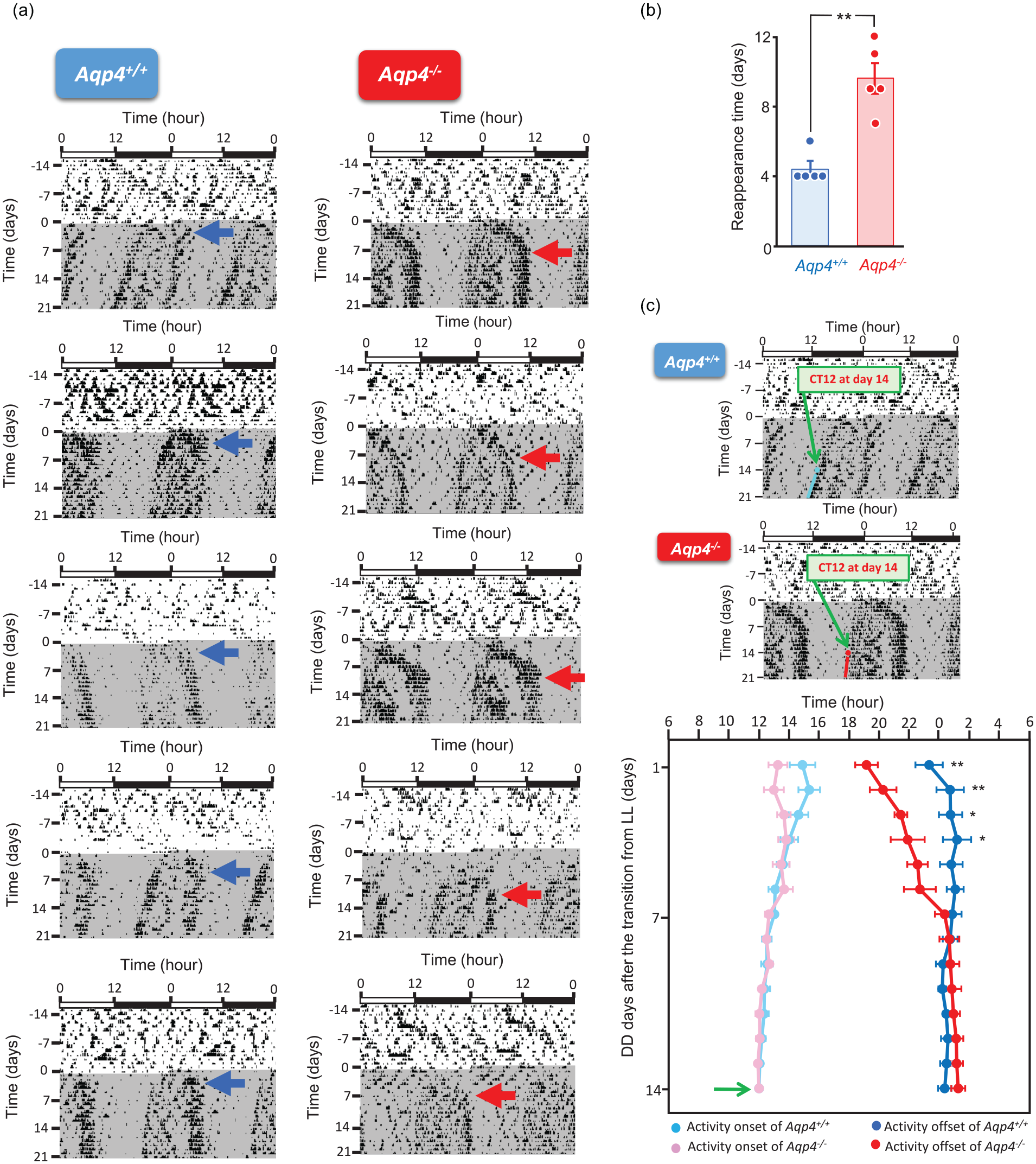
(a) Aqp4+/+ (left; n = 5) and Aqp4-/- (right; n = 5) mice actograms under LL conditions, followed by putting these mice in DD conditions. Blue and red arrows indicate the day of reappearance of stable circadian rhythms. (b) Statistical analysis of the date of reappearance of the intrinsic rhythm under DD conditions in Aqp4+/+ and Aqp4-/- mice (n =5 mice per group) was conducted using Student’s t-test. Data are presented as the mean ± SEM. (c) Change of activity onset and offset time after the transition of DD from LL. CT12 at the day 14 of DD (green arrows in upper actograms) after the transition from LL was defined as Time 12. Green arrow in lower graph indicates Time 12 at DD14. Bright blue circle (•) depicts the activity onset time of Aqp4+/+ mice, dark blue circle (•) depicts the Aqp4+/+ mice activity offset time, pink circle (•) depicts the Aqp4-/- mice activity onset time, and red circle (•) depicts the Aqp4-/- mice activity offset time. We basically examined Aqp4+/+ (n = 5) and App-/- (n = 5), but since it was difficult to judge the activity onsets and offsets of 2 App4-/- mice at the initial DD days, the number of App4-/- animals we used for this analysis was 3 (n = 3) for 1st, 2nd, 3rd and 4th day, and 4 (n = 4) for 5th day. Two-way ANOVA analysis for interaction between genotype and time (DD-days) was applied to activity onset and offset time. Abbreviations: LL = constant light; DD = constant darkness; SEM = Standard error of the mean.
Since behavioral rhythms became prominent and stable after 2-3 weeks of DD transfer in both genotypes, we examined the transition of activity onset and offset 14 days after DD transfer for each animal in either genotype (n = 5 for Aqp4+/+ and Aqp-/- mice), defining Time 12 as the activity onset on day 14 after DD transfer (Figure 2c). For activity onset, according to a 2-way analysis of variance (ANOVA), the interaction of the Aqp4+/+/Aqp-/- effects and time (days in DD) was not significant (F13, 103 = 1.486). For the activity offset, according to a 2-way ANOVA, the interaction between the Aqp4+/+/Aqp-/- effects and days in DD for offsets was significant (F13, 103 = 4.302, p < 0.0001). The offset time in the Aqp-/- genotype between DD-day 1 and -day 4 was significantly shorter than that in the Aqp4+/+ genotype.
It is known that AQP4 in astrocytes regulates water influx in the cerebrospinal fluid microcirculation, resulting in waste products clearance, such as amyloid β (Kang et al., 2009), and excess extracellular glutamate and potassium from the synaptic cleft during synaptic activity (Haj-Yasein et al., 2012; Nagelhus and Ottersen, 2013). Overexcitation of postsynaptic elements is toxic to neurons (Choi, 1988; Popoli et al., 2011). Because the SCN receives strong glutamate input from the retina (Moore, 1997; Ibata et al., 1999), long-term light stimulation increases glutamate metabolism in the SCN, and if the absence of AQP4 impairs its removal, dysfunction of rhythm generation might occur.
Our present results demonstrate that the AQP4 deletion perturbs circadian rhythmicity more severely under LL conditions and delays recovery from LL-induced perturbation, although there was no prominent difference in circadian behavioral rhythms between these genotypes subjected to LD and DD conditions. On transfer to DD, the Aqp4-/- mice exhibit a behavioral pattern different from that of wildtype, with the delayed offset recovery demonstrating a “hump” pattern, although the activity onset recovery was similar to wildtype. It is still unclear whether this difference derives from the different regulation in astrocytes on circadian activity offset and onset in SCN clock function.
Although we speculate that the abnormal circadian behavior in Aqp4-/- mice in the LL was derived from the absence of AQP4 in the SCN, retinal AQP4 could also exaggerate the LL-induced circadian change, as retinal Müller glial cells express abundant AQP4, and its deletion mildly impairs retinal function (Li et al., 2002; Ruiz-Ederra et al., 2007; Ozawa et al., 2019). Further analyses should be conducted to address the role of AQP4 in the SCN-specific conditional knockout.
The brain AQP4 system, which removes waste products, may be related to disturbances and symptomatic progression in neurodegenerative dementia (Nedergaard and Goldman, 2020). In addition, we demonstrated the significance of AQP4 in the SCN for maintaining circadian oscillations under constant lightness. It is highly plausible that AQP4 malfunction in the SCN may augment light-induced perturbation of circadian rhythmicity in our “24/7 society,” wherein artificial light at night worsens the quality of sleep in humans (Chang et al., 2015).
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304221146242 – Supplemental material for Prolonged Light Exposure Induces Circadian Impairment in Aquaporin-4-Knockout Mice
Supplemental material, sj-pdf-1-jbr-10.1177_07487304221146242 for Prolonged Light Exposure Induces Circadian Impairment in Aquaporin-4-Knockout Mice by Atsumi Murakami, Kouki Tsuji, Minako Isoda, Masahiro Matsuo, Yoichiro Abe, Masato Yasui, Hitoshi Okamura and Keiko Tominaga in Journal of Biological Rhythms
Footnotes
Acknowledgements
This work was supported by the Core Research for Evolutionary Science and Technology, Japan Science and Technology Agency (JPMJCR14 W3), Ministry of Education, Culture, Sports, Science and Technology of Japan (17K01986, 18H02606, 20K20864, 22K18384), and SRF.
Author Contributions
H.O. and K.Tominaga conceived of the project and designed the study. A.M. and K. Tsuji. contributed equally as first authors, who conducted experiments in collaboration with M.I., Y.A., M.Y., M.M., H.O., and K. Tominaga. A.M., H.O., and K. Tominagaminaga drafted the manuscript, supported by M.Y.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data supporting the study findings are available from the corresponding author upon reasonable request.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
