Abstract
Beyond visual perception, light has non-image-forming effects mediated by melanopsin-expressing, intrinsically photosensitive retinal ganglion cells (ipRGCs). The present study first used multielectrode array recordings to show that in a diurnal rodent, Nile grass rats (Arvicanthis niloticus), ipRGCs generate rod/cone-driven and melanopsin-based photoresponses that stably encode irradiance. Subsequently, two ipRGC-mediated non-image-forming effects, namely entrainment of daily rhythms and light-induced arousal, were examined. Animals were first housed under a 12:12 h light/dark cycle (lights-on at 0600 h) with the light phase generated by a low-irradiance fluorescent light (F12), a daylight spectrum (D65) stimulating all photoreceptors, or a narrowband 480 nm spectrum (480) that maximized melanopsin stimulation and minimized S-cone stimulation (λmax 360 nm) compared to D65. Daily rhythms of locomotor activities showed onset and offset closer to lights-on and lights-off, respectively, in D65 and 480 than in F12, and higher day/night activity ratio under D65 versus 480 and F12, suggesting the importance of S-cone stimulation. To assess light-induced arousal, 3-h light exposures using 4 spectra that stimulated melanopsin equally but S-cones differentially were superimposed on F12 background lighting: D65, 480, 480 + 365 (narrowband 365 nm), and D65 − 365. Compared to the F12-only condition, all four pulses increased in-cage activity and promoted wakefulness, with 480 + 365 having the greatest and longest-lasting wakefulness-promoting effects, again indicating the importance of stimulating S-cones as well as melanopsin. These findings provide insights into the temporal dynamics of photoreceptor contributions to non-image-forming photoresponses in a diurnal rodent that may help guide future studies of lighting environments and phototherapy protocols that promote human health and productivity.
In addition to permitting visual perception, light has profound “non-image-forming” effects on behavior and physiology, including entraining circadian rhythms and directly promoting sleep or wakefulness (Foster and Hankins, 2002; Fu et al., 2005). Light promotes sleep in nocturnal rodents (e.g. laboratory mice, rats) but promotes arousal and wakefulness in diurnal ones, as in humans (Redlin, 2001; Hagenauer and Lee, 2008; Cohen et al., 2010; Shuboni et al., 2012).
In both diurnal and nocturnal mammals, the non-image-forming effects of light are mediated by intrinsically photosensitive retinal ganglion cells (ipRGCs), whose endogenous photopigment melanopsin is maximally sensitive to 480 nm “blue” light (Berson et al., 2002; Hattar et al., 2002). ipRGCs also receive input from rods and cones and, therefore, can integrate light across a broad spectrum (Lucas et al., 2012). Studies using mice lacking melanopsin or rod/cone photoreceptors have demonstrated that all photoreceptors contribute to circadian photoentrainment (Hattar et al., 2003). Moreover, cones that are sensitive to short or middle wavelengths (“S-cones” or “M-cones”) have distinct roles, with a recent mouse study suggesting that S-cones contribute more to photoentrainment (van Diepen et al., 2021). In addition to mediating entrainment, stimulation of melanopsin can acutely inhibit activity in nocturnal rodents, with melanopsin-deficient mice showing impairments in bright light–induced suppression of locomotor activity (Mrosovsky and Hattar, 2003). In humans, blue light has been found to preferentially improve alertness and cognitive performance, likely by stimulating melanopsin (Cajochen et al., 2005; Lockley et al., 2006; Chellappa et al., 2011; Rahman et al., 2014; Smotek et al., 2019; Grant et al., 2021) but potentially also blue-light-sensitive S-cones.
Findings from nocturnal rodents and humans have consistently indicated that melanopsin, in addition to playing an important role in circadian entrainment, promotes sleep in mice and alertness/wakefulness in humans. However, the roles of rod/cone photoreceptors are less understood, particularly in diurnal mammals. Human studies indicate that both cones and melanopsin contribute to non-image-forming responses but with different time and irradiance dependencies (Gooley et al., 2010, 2012; St Hilaire et al., 2022). Dichromatic diurnal rodents provide a unique opportunity to investigate these dynamic contributions in more detail. Compared to the largely overlapping spectral sensitivities of photoreceptors in humans, S-cones in most rodents are maximally sensitive to ultraviolet (UV) light with λmax at 360 nm, which minimally overlaps with the sensitivity spectra of other photoreceptors and can be independently stimulated (Jacobs et al., 1991).
Nile grass rats (Arvicanthis niloticus) are diurnal in the wild and show diurnal patterns of sleep, locomotor activity, mating behavior, and body temperature in the laboratory (Yan et al., 2020). Many features of the grass rat retina are specialized for operating in the daylight hours, with 30%-40% of their photoreceptors being cones compared to ≤3% in nocturnal mice or rats. In addition, their gross retinal circuitry is typical for diurnal species, including humans (Gaillard et al., 2008), and genetic analysis suggests their S-cones are UV sensitive as in most other rodents (Gaillard et al., 2009). The distribution, morphological types, and projections of melanopsin-expressing ganglion cells have also been characterized in Nile grass rats (Langel et al., 2015), although their photoresponses have not been recorded.
The present study first examined the light-evoked responses of grass rat ipRGCs using multielectrode array (MEA) recording from ganglion cells in isolated retinas. These recordings detected non-rod/cone-dependent photoresponses, confirming the existence of ipRGCs in this species. They further showed that ipRGCs generate rod/cone-driven as well as intrinsic photoresponses, with both response components being sustained “ON,” akin to those of mouse and rat ipRGCs. Next, to explore the role of ipRGCs and other photoreceptors in sculpting daily activity rhythms under entrained condition, as well as light-driven arousal and wakefulness, grass rats were studied under lighting conditions with distinct spectra targeting different photoreceptors. The results showed spectrum-dependent patterns of daily rhythms and arousal responses, collectively suggesting dynamic time- and irradiance-dependent interactions of photoreceptors contributing to photoentrainment and light-induced arousal/wakefulness in diurnal mammals.
Methods
Animals
Grass rats were obtained through a laboratory breeding colony at Michigan State University (McElhinny et al., 1997). The colony was kept in standard animal housing under a 12:12 h light/dark cycle, with lights-on at 0600 h and lights-off at 1800 h, defined as zeitgeber time (ZT) 0 and 12, respectively. Light was supplied by a light-emitting diode (LED) ceiling light (GE current, 14BDT8/G4/840, 4000 k); illuminance was around 300 photopic lux in the center of the room and ~50 lux in the cage. A metal hut was provided in each cage for shelter and enrichment. Food (Prolab 2000 #5P06, PMI Nutrition LLC, MO, USA) and water were available ad libitum. All procedures were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 80-23) and were approved by the Institutional Animal Care and Use Committee of Michigan State University.
Retinal Preparation and MEA Recording
Under ambient room light, a male adult (6-month-old) grass rat was euthanized by intraperitoneal injection of 150 mg/kg sodium pentobarbital. Both eyes were enucleated and hemisected, and after removal of the vitreous using forceps, the eyecups were transferred to a 50-mL centrifuge tube containing Ames’ medium (Sigma, St. Louis, MO) that had been bubbled with 95% O2 and 5% CO2. After being tightly capped, the tube was placed in a lightproof box and driven from Michigan State University to the University of Michigan. Upon arrival, the eyecups were transferred to a Petri dish containing fresh Ames’ medium bubbled with 95% O2 and 5% CO2, and allowed to dark-adapt for an additional 2 h. Under dim red light, one retina was gently isolated from the pigment epithelium using a paint brush. A ~5 mm × 5 mm piece was cut from the retina and MEA recordings were obtained as previously detailed (Wong, 2012). After a series of 10-s duration, full-field 480 nm light of increasing intensities had been presented to the retina during superfusion by normal Ames’ medium, rod/cone signaling was blocked by adding to the reservoir 50 µM L-(+)-2-amino-4-phosphonobutyrate, 40 µM 6,7-dinitroquinoxaline-2,3-dione, and 25 µM D-2-amino-5-phosphonovalerate, and another intensity series of light steps presented (Figure 1a). After the drug-containing solution had been rinsed out and replaced with fresh Ames’ medium, the other retina was processed as described above for MEA recording.
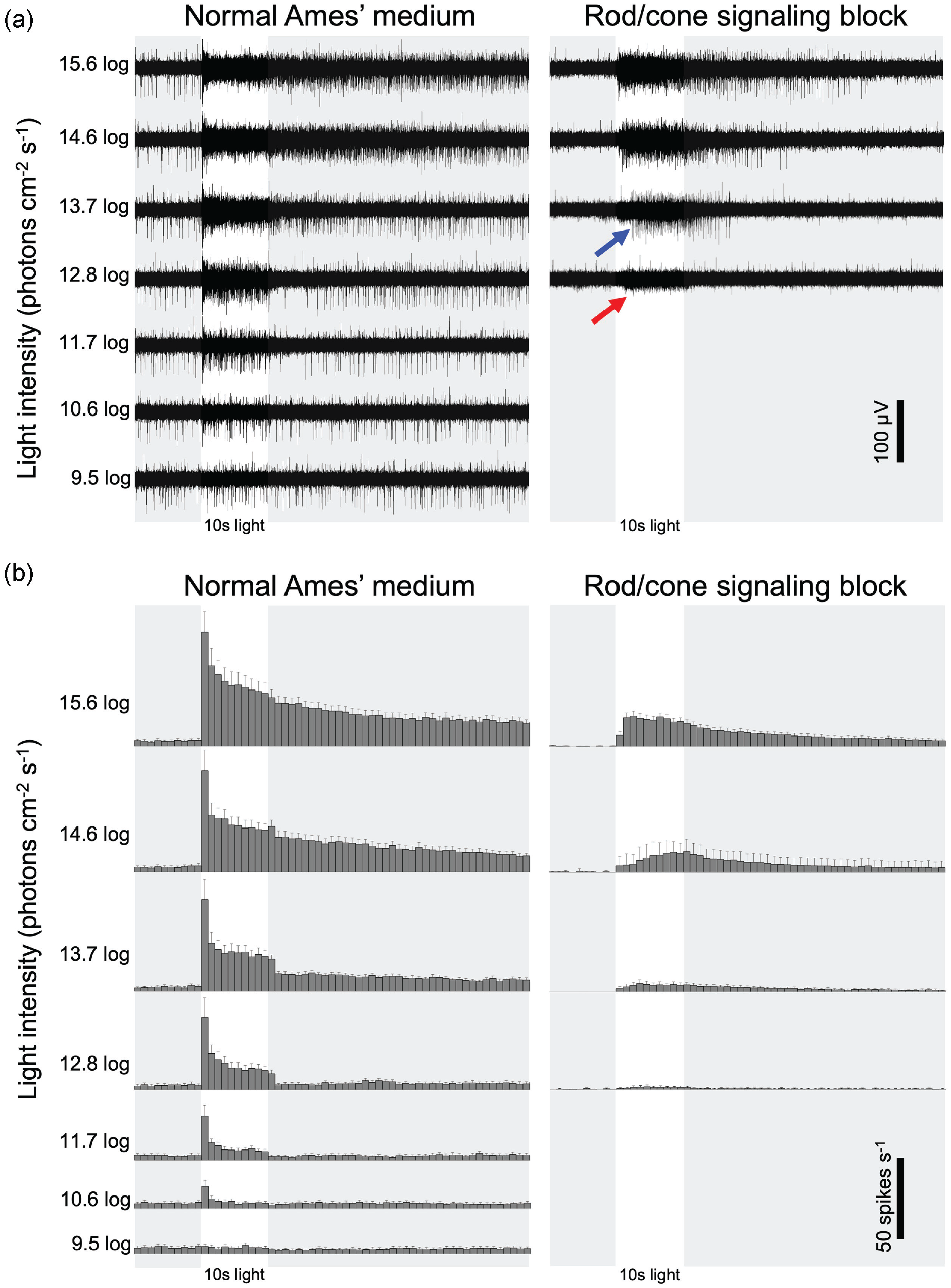
Light-evoked spiking responses of ipRGCs. (a) Raw extracellular recordings from two ipRGCs that generated spikes of two different amplitudes (blue and red arrows). The retina was superfused first in normal Ames’ medium (left) and then in the presence of rod/cone signaling blockers to isolate intrinsic photoresponses (right). (b) Population-averaged spike histograms generated from all 11 ipRGCs with well-isolated spikes, including the larger-spike ipRGC in panel a. Error bars depict SEM. Abbreviation: ipRGCs = intrinsically photosensitive retinal ganglion cells. Color version of the figure is available online.
Lighting Conditions for Behavioral Studies
During the behavioral studies, lighting conditions were provided by custom 8-channel spectral light emulators with six narrowband and two broadband LEDs whose peak emission wavelengths were 360-658 nm (Telelumen Inc., Moorpark, CA, USA). Seven channels were specified to provide appropriate spectral fidelity to stimulate all known rodent retinal photopigments and one red channel was specified to fall outside of the rodent photopigment response to aid in subjective-night experimental activities. In addition to spectral fidelity, these emulators were engineered with pulse width dimming, up to 1000:1, and frame rate control, up to 1/s, to add dimensions of fidelity for intensity and time.
There were five spectra used in this study: CIE standard illuminant fluorescent 12 (“F12,” Figure 2a), a narrowband 480 nm (“480,” Figure 2b) light, CIE standard illuminant daylight 65 (“D65,” Figure 2c), and two derivations that either added or subtracted a narrowband 365 nm (“365”) light to 480 or D65, respectively, named “480 + 365” (Figure 2d) and “D65 − 365” (Figure 2e). Except F12, all spectra used in the behavioral assays were calibrated to produce equal melanopic irradiance (Enezi et al., 2011; Lucas et al., 2014), 500.1 ± 5.9 μW/cm², roughly corresponding to 15.1 log photons cm−2 s−1 of 480 nm light, sufficient to activate melanopsin (Figure 2), although the photon flux reaching the retina in vivo would be lower. Light was measured between 360 and 830 nm using a calibrated spectroradiometer (MSC15, Gigaherz-Optik) at the top of cage level. The photic input for each rodent photoreceptor was then calculated using spectral irradiance measurements of each light stimulus inputted into the Lucas Rodent Irradiance Toolbox (https://lucasgroup.lab.manchester.ac.uk/measuringmelanopicilluminance), which was developed off a toolbox for humans (Lucas et al., 2014). It should be noted that the toolbox was developed for laboratory rodents (i.e. mice or rats), thus the calculated value of irradiance to each photoreceptor was based on the assumption that the spectral sensitivity of photoreceptors is equivalent among rodents or at least between mice and grass rats. Corresponding melanopic equivalent daylight illuminance (m-EDI) lux values were calculated using the CIE S 026 α-opic Toolbox (CIE, 2020), which are shown in Figure 2g.
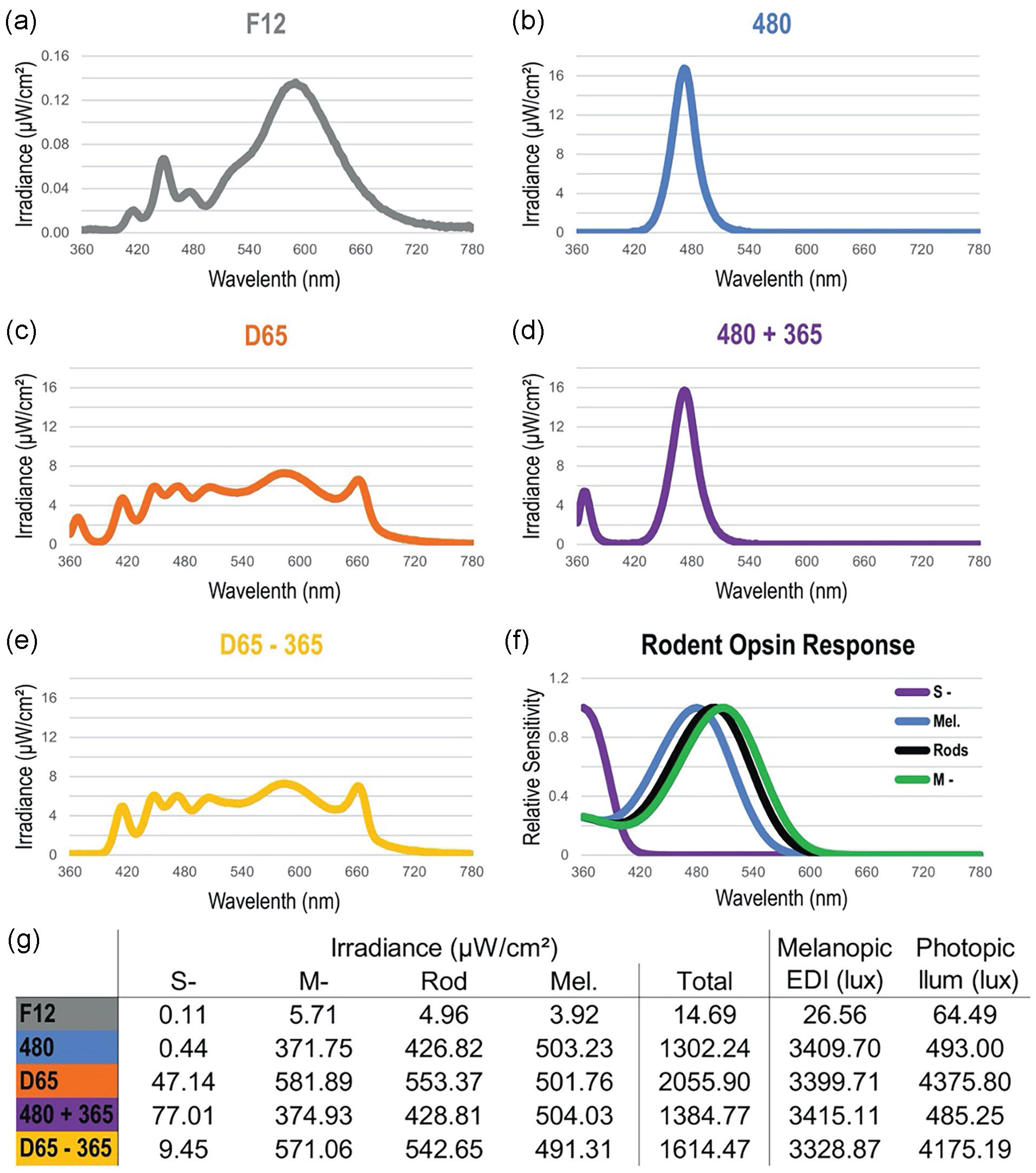
Spectral irradiance measurements for all light stimuli used in the behavioral experiments: F12 (a), 480 (b), D65 (c), 480 + 365 (d), and D65 – 365 (e). Notice the expanded y-axis in panel a. Panel f shows the rodent photoreceptor spectral response functions used to calculate the values in panel g. Irradiance, weighed by the response functions in panel f, was calculated in total and by individual photoreceptor type (g). Melanopic equivalent daylight illuminance (EDI) and photopic illuminance in panel g are human-centric measures and were included for reference purposes only.
Experimental Design for Behavioral Studies
Male grass rats (4- to 6-month old, n = 12) were singly housed under the same 12:12 h light/dark schedule as in the colony room. Animals were first housed under a F12/dark cycle, a baseline control condition for 2 weeks, and then randomly assigned into two groups. Group 1 (n = 6) was housed in 480/dark for 2 weeks, followed by a D65/dark for 2 weeks (Figure 3a). Group 2 (n = 6) received the same exposures but in inverse order to control for any order effect (Figure 3b). Both groups were then placed back under a F12/dark cycle for 2 weeks of reconditioning prior to the next experiment to measure light-induced arousal and wakefulness. Over the following week, a 3-h light exposure (“pulse”) was administered every other day from 1000 to 1300 h (ZT4 to ZT7) using four different pulses: 480, 480 + 365, D65, and D65 − 365 superimposed on the background F12 light. The order of pulses was counterbalanced in group 1 and group 2 as shown in Figure 4a and 4b, respectively.
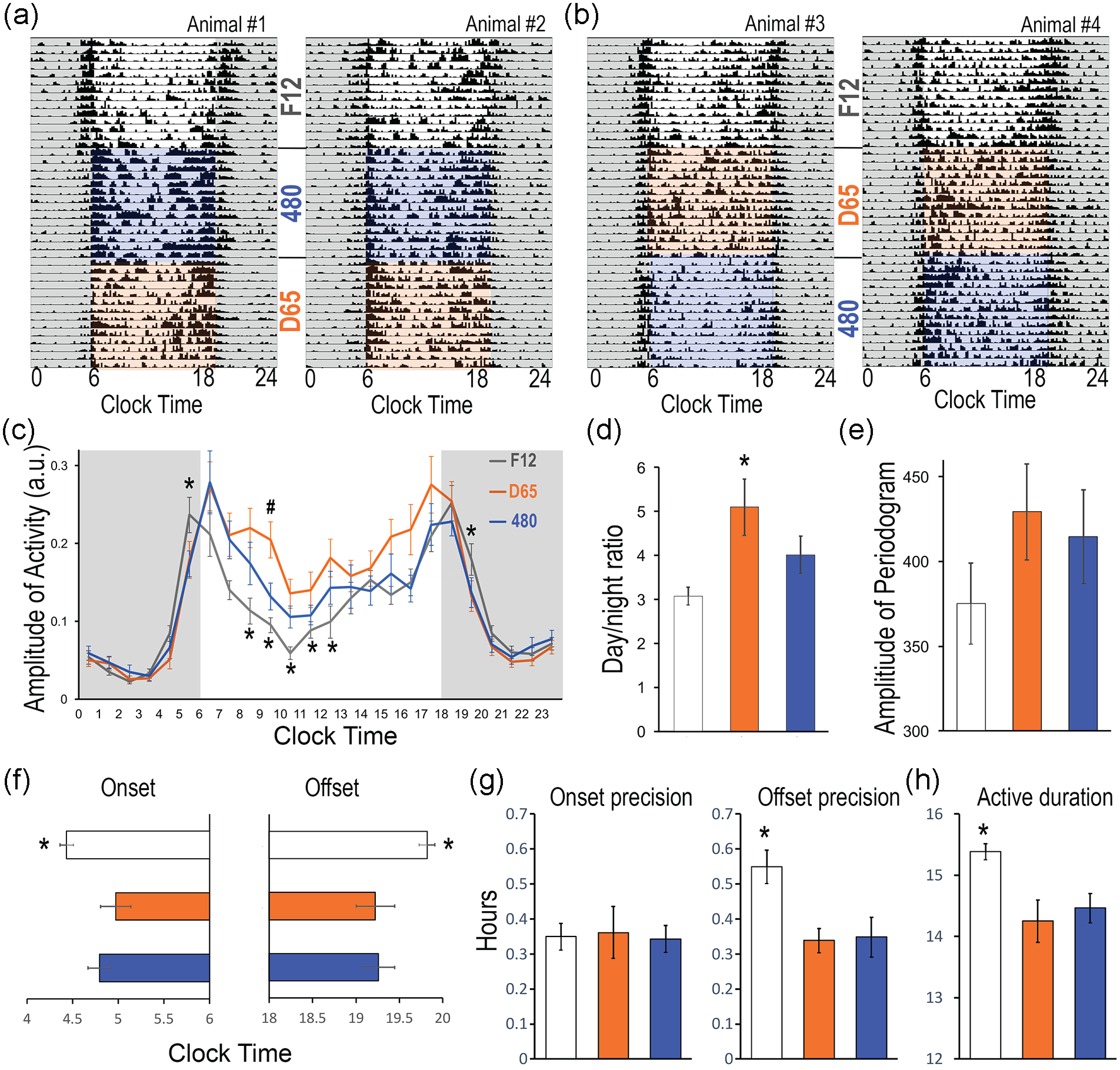
Entrainment of daily activity rhythms under the F12, 480, and D65 spectra during the light phase. (a and b) Representative actograms of four animals showing daily rhythms of locomotor activities under each light condition. Animals were first entrained under F12, half of them were then switched to 480 followed by D65 (a, two animals), and the other half was in reversed order (b, two animals). Quantitative analyses were performed for daily activity in hourly bins (c), day/night activity ratio (d), amplitude of periodogram (e), activity onset and offset (f) and their precision (g), and the daily active duration (h). Results are shown as mean ± SEM. * or # p < 0.05. In panel c, “*” indicates significant difference between F12 and the other two conditions, while “#” indicates significant difference between 480 and D65.
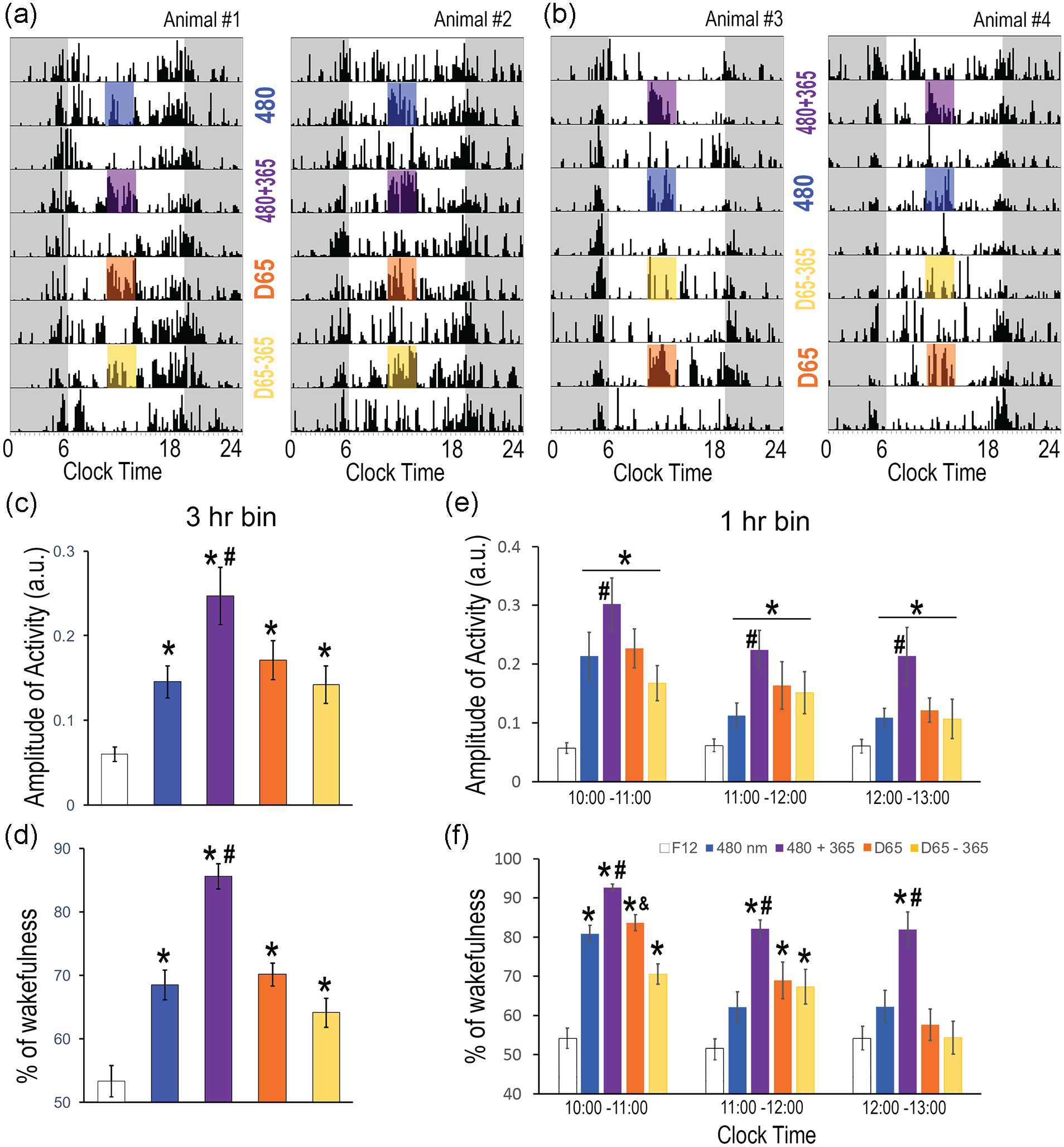
Light-induced locomotor activity and wakefulness. (a and b) Representative actograms of four animals receiving light pulses in different order, the top row corresponding to the last day of the 2-week reconditioning period under F12. (c and d) Locomotor activity and wakefulness during the 3 h of light exposure and the baseline F12 condition. (e and f) Activity and wakefulness analyzed in 1-h bin during the time of exposure (1000-1300 h). Results are shown as mean ± SEM, with “*” indicating significant difference from F12, the baseline condition; “#” indicating significant difference between 480 and 480 + 360; “&” indicating significant differences between D65 and D65 – 365; p < 0.05.
In-cage locomotor activity was monitored continuously using an Actimeter system and an Internet of Things sensor platform (Geocene Inc., Vallejo, CA). The platform consisted of battery-powered wireless sensor nodes, a cloud-based backend for data stacking, and a graphic user interface for semi real-time, ~20-s latency, data visualization/analytics. The hardware used a passive infrared sensor positioned right above the cage lid and angled to provide 100% cage area detection for movement. The system was set for 20-s sample intervals with data outputted as fractional, that is, 0.00 to 1.00. Thus, a data point of 0.69 would translate to 69% cage floor grid coverage (activity in arbitrary unit) for that sample interval.
Sleep/wakefulness was monitored during the light pulse study using a piezoelectric system (Signal Solutions, Lexington, KY). Piezoelectric sensor plates were placed underneath each cage to detect pressure changes associated with movement and breathing. The piezo signals were acquired as described in previous studies (Donohue et al., 2008; Mang et al., 2014; Yaghouby et al., 2016). This method is noninvasive and has attained >90% accuracy for sleep/wake classification based on validation with simultaneous recording through electroencephalogram/electromyography (EEG/EMG) and piezoelectric sensor (Mang et al., 2014).
Data Analysis
Locomotor activity data collected by motion sensors were processed using ClockLab (Actimetrics, Inc. IL, MSU) to generate actograms and for quantitative analysis. To analyze daily rhythms under entrained conditions, the last 7 days during each condition were used to analyze daily activity profiles, day/night activity ratios, amplitude of periodograms, activity onset/offset and their precisions (one standard deviation of the 7-day mean of onsets or offsets), and active duration as described in previous studies (Leach et al., 2013a, 2013b). For the light pulse experiment, the average of five no-pulse days under F12, including the day right before, 3 days in between, and the day right after the last pulse, was used as the baseline. Outlying motion data from one animal was excluded from the analysis due to sensor node misposition. Piezoelectric signals collected during the light pulse study were converted into FeatVec files (FeatVecMaker, Signal Solutions) before analysis using SleepStats (version 2.28, Signal Solutions). Locomotor activity and % of wakefulness were analyzed during the light pulse (1000-1300 h) at 3-h or 1-h interval. Data from the two groups with reversed housing or pulse light order were pooled by light condition since no significant order effect was observed. A one-way or two-way analysis of variance (ANOVA) with repeated measures was used to evaluate the effects of lighting condition using SPSS (IBM). Post hoc pairwise comparisons were used to further evaluate group differences. Statistical significance was indicated when p < 0.05.
Results
Light-Evoked Responses of Individual ipRGCs
Well-isolated spikes were recorded from 11 ganglion cells that exhibited light-evoked spike rate changes during rod/cone signaling block, indicating these cells were ipRGCs. Figure 1a shows recordings from an electrode that detected two ipRGCs, whose spikes had two different amplitudes (blue and red arrows) presumably because one cell was closer to the electrode than the other. Figure 1b shows population-averaged spike histograms generated from all ipRGCs with well-isolated spikes, including the larger-spike ipRGC in Figure 1a, but excluding the smaller-spike cell, as the latter’s spikes could not be analyzed reliably. During normal Ames superfusion, the 10.6 log photons cm−2 s−1 stimulus induced small, transient spiking increases in 8 cells while the remaining 3 cells were nonresponsive, and all 11 cells were responsive at higher stimulus intensities. As stimulus intensity increased, light-evoked spiking became more sustained, and the steady-state spike rate near the end of the 10-s illumination increased. At and above the 13.7 log intensity, a poststimulus response persistence was observed. Following the addition of rod/cone signaling blockers, the lowest stimulus intensity evoking a clear response was 12.8 log for one of the 11 ipRGCs, 13.7 log for 8 cells, and 14.6 log for 2 cells. Notice that the smaller-spike ipRGC in Figure 1a also responded to the 12.8 log stimulus during rod/cone blockade. These rod/cone-independent responses were very sustained, had an onset delay of up to ~2.5 s (Figure 1a, blue arrow), and persisted for many seconds after stimulus offset.
Effects of Daytime Spectra on Daily Rhythms Under Entrained Conditions
Under the baseline F12 condition (Figure 2a), a clear diurnal activity pattern was observed with more activity in light than in the dark phase (Figure 3a-3c). A crepuscular pattern was observed in 3 out of 11 animals (Figure 3a, animal #1), showing prominent active bouts around lights-on and lights-off. Under either 480 or D65 light, daytime activity was higher than in F12. A repeated-measures ANOVA showed a significant effect of lighting condition in the morning, in the hour starting at 0500, 0800, 0900, 1000, 1100, 1200 h, and in early evening at 1900-2000 h (after lights-off at 1800 h) (Figure 3c, p < 0.05 in all cases). Post hoc pairwise comparison revealed higher activity in D65 than in 480 at 0900-1000 h (p = 0.047). Lighting condition had a significant effect on day/night activity ratio (Figure 3d, F2,20 = 9.431, p = 0.006), with higher ratios under D65 light than in 480 or F12 conditions (pairwise comparison: p < 0.01 in all cases). The effect of light on periodogram amplitude was not statistically significant (Figure 3e, F2,20 = 3.455, p = 0.051). The activity onset time was significantly later and offset time significantly earlier under D65 and 480 compared to F12 (Figure 3f, onset: F2,20 = 6.095, p = 0.009; offset: F2,20 = 7.106, p = 0.005). There was no significant difference in onset precision (Figure 3g, F2,20 = 0.031, p = 0.97), but offset precision was significantly less variable in D65 and 480 (F2,20 = 6.241, p = 0.008). The active duration was also significantly more consolidated in light phase under D65 and 480 than in F12 (Figure 3h, F2,20 = 10.762, p = 0.004).
Effects of Spectra on Light-Induced Arousal and Wakefulness
A 3-h light exposure was given during the day from 1000 to 1300 h (ZT4 to ZT7), during which the animals were typically least active under the background F12 condition as shown in Figure 3c. Among the first two pulses, regardless of whether 480 was administered first (Figure 4a) or second (Figure 4b), the animals were more active when exposed to 480 + 365 (Figure 4a and 4b). Quantitative analysis of activity data over the 3-h illumination confirmed that the animals were more active during the exposure to pulses (Figure 4c, repeated-measures ANOVA, F4,40 = 13.429, p < 0.001), compared to the F12-alone condition (pairwise comparison, p < 0.01 in all cases), and they were most active during the 480 + 365 pulse (p < 0.05 in all cases). A similar pattern was also observed with wakefulness data (Figure 4d, F4,44 = 28.935, p < 0.001), with the largest wakefulness-promoting effect produced by 480 + 365 (pairwise comparison, p ≤ 0.001 in all cases). The activity (Figure 4e) and wakefulness data (Figure 4f) were further analyzed across 1-h bins, revealing distinct temporal profiles of each spectrum. For light-induced activity (Figure 4e), there was a significant effect of light condition (F4,80 = 13.437, p < 0.001) and time (F2,80 = 6.851, p = 0.005), but no significant interaction between the two (F8,80 = 1.442, p = 0.192). Pairwise comparison revealed that the activity was higher during the first hour compared to the second (p = 0.029) or third hour (p = 0.015). Between light conditions, all four spectra induced significantly higher activity compared to F12, while 480 + 365 had greater effect than 480 alone (p < 0.05 in all cases). For light-induced wakefulness (Figure 4f), there was a significant interaction between light and time (F8,88 = 3.07, p = 0.004). The 480 + 360 pulse induced higher wakefulness than 480 alone or in F12 throughout the 3-h period (p < 0.05 in all cases). The 480 pulse induced higher wakefulness than in F12 only during the first hour (p < 0.05). The wake-promoting effects of D65 and D65 − 365 pulses lasted 2 h, but the effect of D65 − 365 was lower than D65 during the first hour (p < 0.05 in all cases).
Discussion
We have presented the first recordings of both rod/cone-mediated and intrinsic ipRGC photoresponses in A. niloticus. One previous report recorded from ipRGCs in a related species, Arvicanthis ansorgei, although only intrinsic photoresponses were recorded in neonatal retinas that lacked rod/cone photosensitivity (Karnas et al., 2013). While we have not proved that the intrinsic photoresponses of grass rat ipRGCs are melanopsin-mediated, they probably are given their remarkable similarities to the melanopsin-based responses of ipRGCs in other species such as rat, mouse, and primates (Dacey et al., 2005; Wong et al., 2007; Zhao et al., 2014). Specifically, in those species, melanopsin induces sluggish, sustained “ON” responses (i.e. a prolonged increase in spiking during constant illumination) comparable to the intrinsic responses we detected in grass rat ipRGCs. Such sluggishness is not an artifact of the rod/cone signaling blockers, because mechanically isolated ipRGCs exhibit similarly sluggish intrinsic photoresponses in the absence of rod/cone blockers (Berson et al., 2002; Hartwick et al., 2007; Graham et al., 2008). We have observed additional similarities between the ipRGC photoresponses of grass rats and of other species: (1) the rod/cone input evokes sustained “ON” responses; (2) the rod/cone-driven response component has faster kinetics and a lower intensity threshold than the melanopsin component; (3) the rod/cone-driven response adapts rapidly during the first several seconds, before stabilizing at a steady state; (4) the steady-state spike rate increases with light intensity, providing a retinal output suited for driving irradiance-dependent non-image-forming responses to light; and (5) at sufficiently high light intensities, both response components persist for many seconds after stimulus offset. While we found the rod/cone input’s irradiance threshold to be 3-4 log units higher than that for mouse and primate ipRGCs (Dacey et al., 2005; Zhao et al., 2014), this finding likely reflects the suboptimal health of the grass rat retinas caused by inadequate oxygenation during the intercity transport. The grass rat ipRGCs exhibited diverse intensity thresholds for the intrinsic photoresponse, suggesting the existence of multiple physiologic types as previously documented in MEA studies of other species including humans (Tu et al., 2005; Karnas et al., 2013; Walch et al., 2015; Mure et al., 2019). In terms of rod/cone signaling, electroretinographic analysis of grass rat retinas using different photic stimuli showed similar features to the responses seen in humans, further supporting grass rats as a valuable model for understanding human photic responses (Gilmour et al., 2008).
Daily rhythm was examined under either 480 light, which stimulated mainly M-cones, rods, and melanopsin, or D65 light which also stimulated S-cones (Figures 2 and 3). Overall, the animals were more active under 480 or D65 compared to the F12 control (Figure 3c). The results are consistent with a recent study on another diurnal rodent Rhabdomys pumilio, commonly referred to as four-striped grass mice or four-striped grass rats, which found that a higher melanopic irradiance at 1940 mel EDI (~60% of the 480 or D65 in the present study) during the day resulted in higher amplitude daily rhythms in locomotor activity and suprachiasmatic nucleus (SCN) electrical activity (Bano-Otalora et al., 2021). Light condition also had significant effect on a few other parameters reflecting the entrainment of daily rhythms including the activity onset or offset (Figure 3f), the offset precision (Figure 3g, right plot), and the active duration (Figure 3h). Most studies on rhythms under entrained condition have been conducted in laboratory settings where illumination is supplied by electrical lighting with much lower irradiance and different spectral composition than natural daylight. The daylight-emulating D65 and narrowband 480 used in the present study allowed us to explore the question of how quality of light impacts photic entrainment in a diurnal mammalian species. Compared to 480, D65 induced higher daytime activity (Figure 3c) and a higher day/night activity ratio (Figure 3d), due to D65’s higher irradiance targeting S-cones and/or M-cones (Figure 2g). The rods were maximally hyperpolarized under both spectra because 480’s and D65’s irradiances of 426.82 and 553.37 μW/cm2, equivalent to 15.03 log and 15.14 log photons cm-2 s-1 at λmax, respectively, are well within the rod-saturating photopic range (Dacey et al., 2005). Given that D65 was 107-fold more effective than 480 in stimulating S-cones, but stimulated M-cones only 56.5% more strongly than did 480, the two conditions’ different effects on daily rhythms are likely due mainly to differences in S-cone stimulation.
The direct arousal (assessed by monitoring activity) and wakefulness-promoting effects of light were assessed using 3-h light exposures from 1000 to 1300 h when the animals were generally least active under F12 (Figure 4a and 4b). In non-human primates and human subjects, S-cones contribute to the non-image-forming effects of light by acting on ipRGCs (Brown et al., 2021; Patterson et al., 2022). In grass rats, S-cones’ distinct opsin sensitivity enables these cells to be specifically targeted by either adding or removing narrowband 365 nm. Therefore, we focused on two sets of comparisons: 480 versus 480 + 360, and D65 versus D65 − 360. Because 480 and 480 + 365 stimulated M-cones and melanopsin similarly, as did D65 and D65 − 365 (Figure 2g), and all four spectra saturated rods (see the previous paragraph), differences in S-cone stimulation were the only variable. Accordingly, the higher arousal and wakefulness under 480 + 365 than under 480 (Figure 4c-4f, blue vs purple bars) were due to the recruitment of S-cones. On the other hand, removing the 365 nm band from D65 only modestly reduced waking effects (Figure 4f, orange vs yellow bars); in fact, statistically significant reductions were only detected during the first hour of the pulse (Figure 4f “1000-1100 h”). This outcome likely resulted because the difference in S-cone stimulation was smaller between D65 and D65 − 365 compared to that between 480 and 480 + 365 (Figure 2g).
Analyzing the arousal and wakefulness-promoting effects in hourly intervals showed unique temporal dynamics of the four light spectra. Light-induced activity declined over time under all four spectra, likely in part due to physical fatigue (Figure 4e). However, a significant light by time interaction was found for light-induced wakefulness (Figure 4f). 480 + 365 produced more sustained wakefulness over 3 h, while 480 significantly increased wakefulness for just the first hour (Figure 4f). D65 and D65 − 365 both promoted wakefulness over 2 h, with a stronger effect produced by D65 during the first hour. These results indicate that potent stimulation of S-cones in addition to the other photoreceptors is crucial to continuously promote wakefulness. It is possible that the behavioral outcomes were regulated by brain sites downstream of the retina, for example, hypothalamic or thalamic regions that have been implicated in direct behavioral responses to light (Gall et al., 2013, 2014, 2017). Alternatively, or additionally, part of this time-dependent activity reduction could be explained by light adaptation of melanopsin, which causes a gradual decline in the melanopsin-based photoresponse during prolonged photostimulation (Wong et al., 2005). To compensate for such a decline, ipRGCs in Nile grass rats could rely on S-cone input, which has been shown to be remarkably sustained in mice (van Diepen et al., 2021). The rod input to rat ipRGCs is likewise very sustained (Wong, 2012), but the 3-h pulses superimposed on the F12 background were unlikely to have stimulated rods as the rods were probably already maximally hyperpolarized under F12 with a rod-stimulating irradiance of 4.96 μW/cm2, equivalent to 13.1 log photons cm−2 s−1 at λmax which is within the photopic range (Dacey et al., 2005). It is noteworthy that, under our experimental conditions, the temporal patterns of melanopsin- and S-cone-mediated wakefulness during the day appear opposite from those seen in light-induced melatonin suppression at night, in which S-cones mediate the initial response while melanopsin evokes a more sustained response (Brown et al., 2021; St Hilaire et al., 2022). It is conceivable that the kinetics and/or sensitivity of photoreceptors are under circadian modulation (i.e. varies between day and night), as has been reported in other sensory systems (Frederickson et al., 1977; Pickard, 1987; Aviram et al., 2015; Daguet et al., 2022), although differences in testing conditions (e.g. different stimulus intensities, and the use of Ganzfeld domes in the human studies) probably also contributed.
Although the broad effects of light on brain and behavior have been documented on a variety of human health and performance outcomes, the underlying mechanisms are not well understood. The present study stands as one of the initial steps for understanding how the lighting environment is sensed by specific types of photoreceptors in diurnal mammals, ultimately influencing behaviors including daily rhythms, arousal, and wakefulness. Such information will be valuable for the design of human-based lighting systems that promote health whether it be inside buildings or out, on this planet or off, and/or for targeted clinical applications such as bright light therapy for the treatment and prevention of depression and dementia.
Footnotes
Acknowledgements
This work was supported by NIH grants EY018863 to K.Y.W. and MH111276 to L.Y. The content is solely the responsibility of the authors and does not necessarily represent the official views of funding agencies.
Conflict of Interest Statement
S.W.L reports commercial interests from the last 3 years (2020-2023). His interests are reviewed and managed by Brigham and Women’s Hospital and Partners HealthCare in accordance with their conflict-of-interest policies. S.W.L has received consulting fees from Hintsa Performance AG, Stantec and View Inc, and has consulting contracts with Absolute Rest, Akili Interactive, Apex 2100 Ltd, Ashurst Risk Advisory, Consumer Sleep Solutions, KBR Wyle Services, Light Cognitive; Lighting Science Group Corporation/HealthE; Mental Workout/Timeshifter. He has received honoraria and travel or accommodation expenses from Bloxhub, Clifton College, Danish Centre for Lighting, and University of Toronto; and travel or accommodation expenses (no honoraria) from Wiley; and royalties from Oxford University Press and Monash University. He holds equity in iSleep pty. He has received an unrestricted equipment gift and investigator-initiated grant from F. Lux Software LLC, and a Clinical Research Support Agreement and Clinical Trial Agreement with Vanda Pharmaceuticals Inc. He is an unpaid Board Member of the Midwest Lighting Institute (non-profit). He is part-time adjunct professor at the University of Surrey, UK. He holds several pending (US20190366032; US20210162164 US20220151552) and one awarded (USD943612) patents. He has served as a paid expert in legal proceedings related to light, sleep and health.
