Abstract
In mammals, 24-h rhythms are controlled by a hierarchical system of endogenous clocks, with a circadian pacemaker located in the suprachiasmatic nuclei (SCN) of the hypothalamus that synchronizes peripheral oscillators throughout the body. The molecular clock machinery is regulated by interlocked transcriptional translational feedback loops (TTLs). The core TTL includes the transcriptional modulators PER (1-3) and CRY (1/2) that feed back on their own expression by interaction with CLOCK/BMAL1. An accessory loop involving the transcription factors DEC1 and DEC2 has been described that also impinges on CLOCK/BMAL1-mediated transactivation. In Drosophila, the DEC ortholog CWO shows synergistic activity to PER. This prompted the authors to analyze PER1-DEC interaction in the mammalian SCN. They generated Per1/Dec double and triple mutant mice to monitor activity rhythms under entrained and free-running conditions. Furthermore, they analyzed expression of the clock genes Per2, Rev-Erbα, and Bmal1 in wild-type and Per1/Dec mutant SCN by in situ hybridization. The experiments reveal a critical role for Per1-Dec interaction in regulating activity phase under entrained conditions. In constant darkness, a synergistic function for Per1 and Dec1/2 in period regulation was found, correlating with disrupted clock gene mRNA levels in the SCN. Luciferase reporter gene assays indicate an activatory function of DECs on Bmal1 expression. Together, the results suggest a partially redundant and bidirectional regulatory function for the 2 Dec genes in the TTL and a conservation of Per-Dec (Cwo) synergism between vertebrate and invertebrate clocks.
In most species, 24-h rhythms of physiology and activity are regulated by endogenous circadian clocks. In mammals, the circadian pacemaker located in the suprachiasmatic nuclei (SCN) of the hypothalamus (Moore and Eichler, 1972; Schibler and Sassone-Corsi, 2002; Stephan and Zucker, 1972) shows electrical activity rhythms of approximately 24 h under free-running conditions (Inouye and Kawamura, 1979). Therefore, to keep in synchrony with external time, the SCN have to be reset each day by environmental zeitgebers in a process termed entrainment. The predominant zeitgeber of the mammalian system is light that reaches the SCN via the retino-hypothalamic tract (Moore and Lenn, 1972). From the SCN, time information is transferred by various means to peripheral circadian oscillators found in all tissues and controlling local physiological rhythms (Buijs and Kalsbeek, 2001; Oster, 2006; Reppert and Weaver, 2002).
At the molecular level, circadian clocks are based on interlocked transcriptional/translational feedback loops (TTLs) comprised from a set of clock genes that are highly preserved between vertebrate and invertebrate species (Zhang and Kay, 2010). In the mammalian core TTL of the SCN, the transcription factors CLOCK and BMAL1 (ARNTL) activate the transcription of cis-regulatory E-box containing target genes, including Period (Per1-3) and Cryptochrome (Cry1-2). PER/CRY protein complexes accumulate during the course of the day and in the night relocate back into the nucleus, where they inhibit their own transcription by interaction with CLOCK/BMAL1 (Griffin et al., 1999; Kume et al., 1999; Reppert and Weaver, 2002). Recently, two other transcription factors, DEC1 (BHLHE40) and DEC2 (BHLHE41), have been described to interact with the components of the SCN clock (Honma et al., 2002; Noshiro et al., 2005). Dec mutant mice show defects in light entrainment and free-running periodicity (Rossner et al., 2008). Dec transcription is activated by CLOCK/BMAL1, and nocturnal light exposure acutely stimulates Dec1 expression in the SCN (Butler et al., 2004; Honma et al., 2002). On the other hand, the DECs impinge on circadian transcription by competing with CLOCK/BMAL1 for E-box occupation and by physical interaction with BMAL1 (Honma et al., 2002; Sato et al., 2004). The interactivity of the DECs with the other CLOCK/BMAL1 regulators—PERs and CRYs—however, remains unclear. Recent results suggest that DEC function can be either activatory or inhibitory, depending on tissue type as well as on promoter of target genes (Azmi et al., 2003; Dardente et al., 2009; Hamaguchi et al., 2004; Honma et al., 2002; Kawamoto et al., 2004; Li et al., 2004; Li et al., 2003; Rossner et al., 2008). Studies in flies propose that clockwork orange (cwo), the Drosophila ortholog of the Decs, has a synergistic function to the Per1/2 ortholog period (per) (Kadener et al., 2007). To test if this mode of interaction is preserved in mammals, we analyzed Per1-Dec interaction in the regulation of circadian activity rhythms and clock gene expression in the SCN in the corresponding Per1/Dec double and triple mutant mice. Our results show a synergistic function of Per1 and Dec1 together with Dec1/Dec2 redundancy in the regulation of photic activity entrainment and of period length under free-running conditions. Thus, it appears that per-cwo interaction in the Drosophila TTL is largely conserved in the mammalian pacemaker.
Materials and Methods
Animals
Per1–/– mutant mice (Per1Brdm1; Zheng et al., 2001) on a C57BL/6J background (N10) were mated with congenic Dec1–/– and Dec2–/– mice (Rossner et al., 2008) to produce Per1/Dec double and triple mutant animals. The genotype of the offspring was determined by PCR as described (Rossner et al., 2008; Zheng et al., 2001). All experiments were done on male adult animals (2-10 months). Congenic age-matched C57Bl/6J mice were used as controls. All animal experiments were performed with permission from the Office of Consumer Protection and Food Safety of the State of Lower Saxony and in accordance with the German Animal Welfare Act.
Behavioral Analysis
Mouse housing and handling were performed as described (Albrecht and Oster, 2001). Wheel-running activity was analyzed using ClockLab software (Actimetrics, Evanston, IL). Prior to the experiments, animals were entrained to a 12-h light/dark (LD) cycle for at least 10 days (light intensity 250 lux). For analyzing free-running locomotor activity under constant darkness conditions (DD), lights were turned off at the end of the light phase (zeitgeber time [ZT] 12) and not turned on again the next day. Activity profiles in LD, DD, and constant light (LL; light intensity 50 lux) were assessed over 4 to 7 consecutive days. The onset phase angle was determined by fitting a straight line to 7 to 10 consecutive activity onsets. Onset variation depicts the mean deviation of real activity onsets from a regression line through 7 to 10 consecutive onsets under stably entrained or free-running conditions. Rhythmicity and period length (τ) in DD and LL were determined by χ2 periodogram analysis over an interval of 7 to 10 consecutive days. Period differences were calculated by comparison of period lengths between two different DD intervals separated by at least 10 days in LD.
In Situ Hybridization
Animals were entrained to LD for 10 days, released into DD, and sacrificed 14, 20, 26, and 32 h after the end of the last light phase. Brains were dissected under dim red light, fixed by immersion in 4% paraformaldehyde (PFA) in phosphate-buffered saline (PBS), dehydrated, and paraffin embedded. Then, 8-µm sections were hybridized with 35S-UTP-labeled antisense RNA probes for clock gene transcripts as described (Oster et al., 2003; Oster et al., 2002). The Rev-Erbα template was generated by PCR (forward primer: CCCTCTACAGTGACAGCTCCA; reverse primer: TCAGACACCGTTTGTACTGGA) from murine adipose tissue cDNA. Relative quantifi-cation of expression levels was performed by densitometric analysis of autoradio-graph films using Quantity One software (Bio-Rad, Munich, Germany). Background correction was performed by subtracting the optical density measured in the lateral hypothalamus. For each experiment, 3 animals per genotype were used and 3 adjacent SCN sections per animal were analyzed.
Reporter Gene Assays
Dual reporter gene assays in HEK293 cells were performed as previously described (Rossner et al., 2008; Wehr et al., 2006) using a firefly luciferase Bmal1 reporter gene (Bmal1::luc; Nagoshi et al., 2004) and a Renilla luciferase reporter as normalizing transfection control. Cytomegalovirus (CMV)–driven expression plasmids coding for HA-tagged rat DEC1, Flag-tagged mouse DEC2 (Wehr et al., 2006), and V5-tagged mouse PER1 were used (Rossner et al., 2008). The pSCT1HARev-Erbα plasmid was a kind gift by Jürgen Ripperger and Urs Albrecht, University of Fribourg, CH. The PER1 expression plasmids were kindly provided by Dr. Pablo Szendro, Max Planck Institute for Biophysical Chemistry, Göttingen, Germany.
Statistical Analysis
Statistical evaluation was performed using GraphPad Prism software (GraphPad Software, La Jolla, CA). Groups were compared by one-way analysis of variance (ANOVA) followed by Tukey posttest for multiple comparisons. A p value <0.05 was considered significant. The rhythmicity of clock gene expression was evaluated using sine curve fitting with CircWave v1.4 (Oster et al., 2006).
Results
Synergistic Interaction of Per1 and Dec1/2 under Entrained Conditions
Per1/Dec double and triple mutant mice were born at the expected Mendelian ratios—taking into account that both Dec genes are located relatively close together on the same chromosome (Fujimoto et al., 2001; Sun et al., 1999). Mutant mice of all genotypes were viable and fertile and did not show any gross anatomical or behavioral abnormalities (data not shown). Under a 12-h LD cycle, all animals entrained readily to the light/dark cycle (Fig. 1). However, significantly advanced activity onsets were observed in Per1 single, Dec1/2 double, and Per1/Dec double and triple mutant mice (Fig. 2A and statistics in Suppl. Table S1). Per1–/–Dec1–/– as well as Per1–/–Dec1/2–/– mice showed significantly earlier activity onsets in comparison to wild-type (WT) and to single Per1 mutant animals, indicative of an additive Per1-Dec1 interaction during photic entrainment. In addition, the activity onsets of Per1/Dec1/2 triple mutant mice were significantly advanced compared to those of Dec1/2 and both Per1/Dec double mutant strains, indicating a synergistic interaction of Per1 and Dec1/2 and a certain redundancy of Dec1 and Dec2 in photic entrainment. These observations were supported when we measured entrainment stability by quantifying the day-to-day variation of activity onsets under LD conditions. Per1 single, Dec1/2 double, and Per1/Dec double and triple mutant mice displayed decreased onset stability when compared to WT animals (Fig. 2B). Single mutants showed onset variations between 0.11 and 0.19 h in the wild-type range. In double and triple mutants, this variability increased in an additive fashion (from 0.26-0.48) (Fig. 2B and statistics in Suppl. Table S1), suggesting that a fully functional Per1-Dec1/2 interaction is necessary for efficient synchronization of activity phase with the external LD cycle.

Wheel-running behavior under entrained and free-running conditions. (A-G) Representative locomotor activities of (A) WT, (B) Per1–/–, (C) Dec1–/–, (D) Dec2–/–, (E) Per1–/–Dec1–/–, (F) Per1–/–Dec2–/–, (G) Dec1/2–/–, and (H) Per1–/–Dec1/2–/– mice under LD and DD conditions. Actograms are double-plotted with the activity of the following day plotted to the right and below the previous one. Gray shadings indicate dark phases.
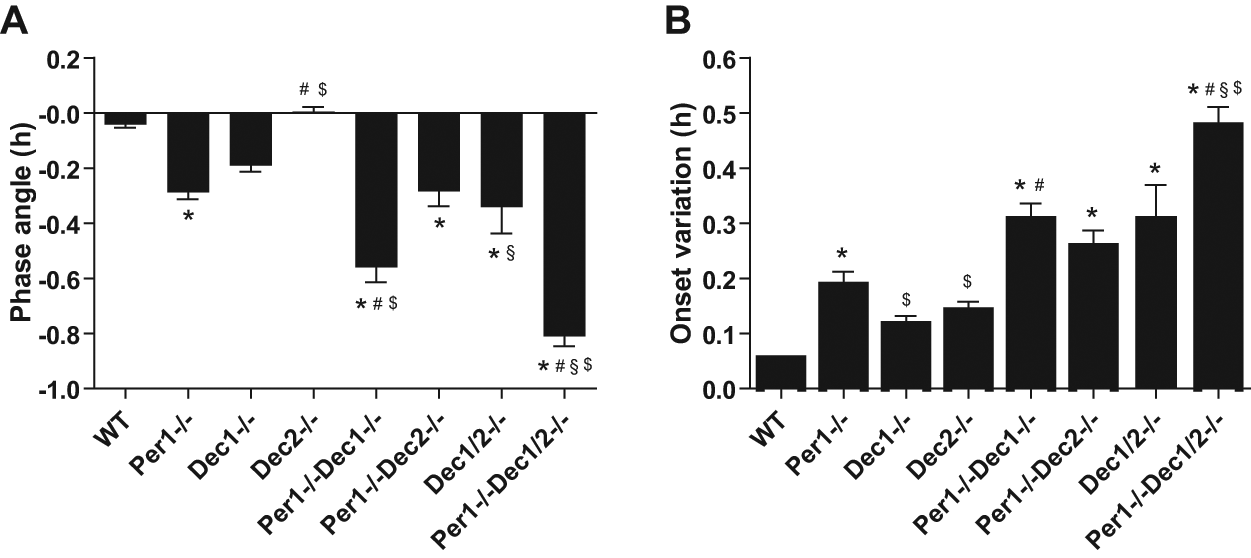
Analysis of locomotor behavior under entrained conditions. (A) Activity onset phase angles relative to “lights off” under LD conditions: WT −0.04 ± 0.01 h (n = 20), Per1–/– −0.29 ± 0.02 h (n = 20), Dec1–/– −0.19 ± 0.02 h (n = 20), Dec2–/– 0.01 ± 0.02 h (n = 12), Per1–/–Dec1–/– −0.56 ± 0.05 h (n = 20), Per1–/–Dec2–/– −0.28 ± 0.05 h (n = 17), Dec1/2–/– −0.34 ± 0.10 (n = 8), and Per1–/–Dec1/2–/– −0.81 ± 0.04 h (n = 11). (B) Activity onset variations under LD conditions: WT 0.05 ± 0.004 h (n = 20), Per1–/– 0.19 ± 0.03 h (n = 20), Dec1–/– 0.11 ± 0.02 h (n = 20), Dec2–/– 0.14 ± 0.02 h (n = 12), Per1–/–Dec1–/– 0.30 ± 0.03 h (n = 19), Per1–/–Dec2–/– 0.26 ± 0.03 h (n = 17), Dec1/2–/– 0.31 ± 0.06 (n = 10), and Per1–/–Dec1/2–/– 0.48 ± 0.04 h (n = 12). Data are represented as mean ± SEM. *p < 0.05 compared to WT; #p < 0.05 compared to Per1–/–; §p < 0.05 compared to Per1–/–Dec1–/– or Per1–/–Dec2–/–; $p < 0.05 compared to Dec1/2–/–.
Functional Redundancy of Dec1 and Dec2 in Period Regulation and Per1 Interaction under Free-Running Conditions
We next asked whether the Per1-Dec1/2 interaction in LD is conserved in the absence of external zeitgeber signaling. To test this, we released the animals into constant darkness (DD). In DD, all mutants retained rhythmic circadian activity patterns (Fig. 1), but marked changes in the period of these rhythms were observed. With the exception of Dec2–/– and Dec1/2–/– mice, all single, Per1/Dec double, and triple mutant genotypes displayed a significantly shortened DD period length when compared to WTs (Fig. 3A and statistics in Suppl. Table S1). While in the Per1/Dec double mutant mice, period length appeared mainly to be determined by the absence of Per1, Per1/Dec triple mutants showed a significant reduction in period length when compared to all single and double mutant strains. Similar, though statistically less powerful, effects were observed under constant light (LL) conditions. In LL, a loss of Per1 led to an increase in period length by about 1 h (Fig. 3C and statistics in Suppl. Table S1). While no period change was observed in either Dec single or double mutants, a combination of Per1 and Dec1/2 deficiency further additively increased LL free-running period (Fig. 3C and statistics in Suppl. Table S1). Together, the DD/LL data suggest a synergistic interaction between Per1 and Dec1/2 in the regulation of free-running period together with a functional Dec redundancy.
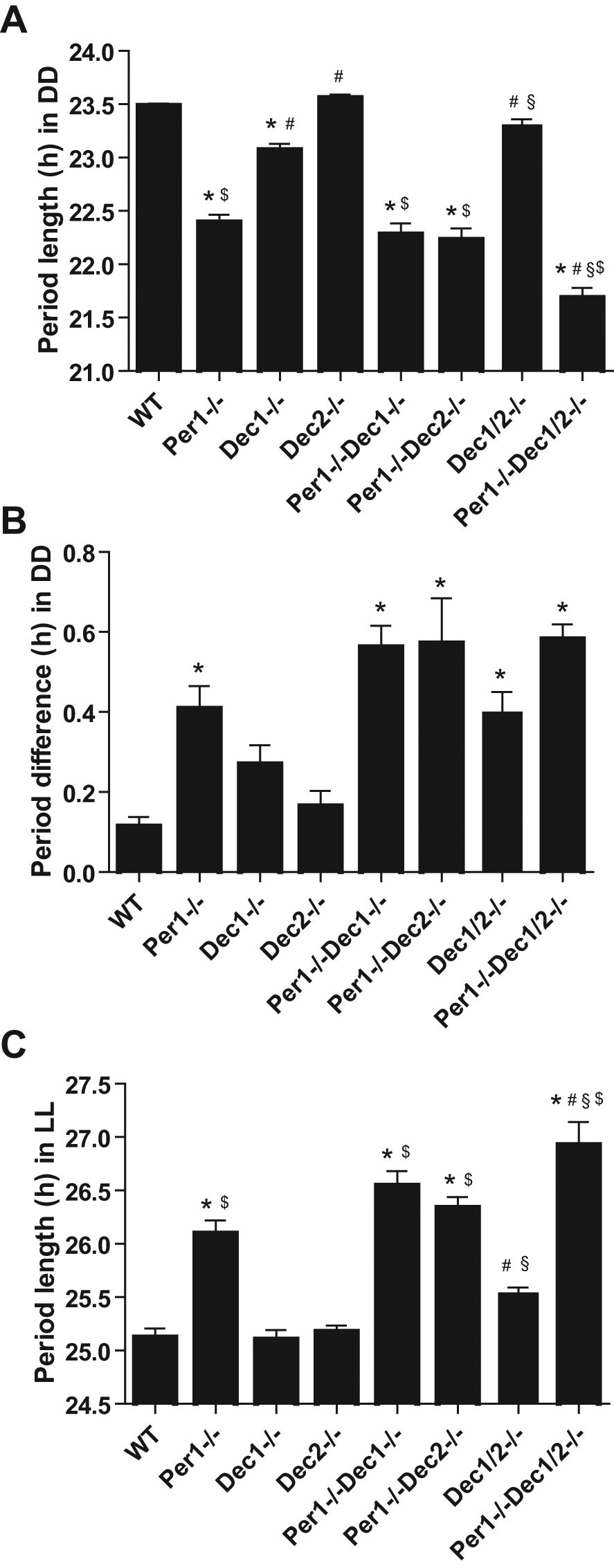
Analysis of locomotor behavior under free-running conditions. (A) Average free-running period length in DD: wild-type (WT) 23.48 ± 0.02 h (n = 20), Per1–/– 22.39 ± 0.07 h (n = 20), Dec1–/– 23.07 ± 0.06 h (n = 20), Dec2–/– 23.56 ± 0.03 h (n = 12), Per1–/–Dec1–/– 22.28 ± 0.10 h (n = 14), Per1–/–Dec2–/– 22.23 ± 0.11 h (n = 17), Dec1/2–/– 23.31 ± 0.05 (n = 10), and Per1–/–Dec1/2–/– 21.68 ± 0.10 h (n = 12). (B) Period variability between 2 DD intervals: WT 0.11 ± 0.02 h (n = 10), Per1–/– 0.41 ± 0.06 h (n = 10), Dec1–/– 0.27 ± 0.05 h (n = 10), Dec2–/– 0.16 ± 0.04 h (n = 10), Per1–/–Dec1–/– 0.56 ± 0.05 h (n = 10), Per1–/–Dec2–/– 0.57 ± 0.11 h (n = 10), Dec1/2–/– 0.40 ± 0.05 (n = 10), and Per1–/–Dec1/2–/– 0.58 ± 0.04 h (n = 12). (C) Average free-running period length in constant light: WT 25.12 ± 0.08 h (n = 12), Per1–/– 26.09 ± 0.12 h (n = 15), Dec1–/– 25.10 ± 0.09 h (n = 11), Dec2–/– 25.18 ± 0.06 h (n = 12), Per1–/–Dec1–/– 26.54 ± 0.14 h (n = 11), Per1–/–Dec2–/– 26.34 ± 0.10 h (n = 11), Dec1/2–/– 25.54 ± 0.05 (n = 10), and Per1–/–Dec1/2–/– 26.92 ± 0.22 h (n = 11). Data are represented as mean ± SEM. *p < 0.05 compared to WT; #p < 0.05 compared to Per1–/–; §p < 0.05 compared to Per1–/–Dec1–/– or Per1–/–Dec2–/–; $p < 0.05 compared to Dec1/2–/–.
A previous study had shown that Per1 deficiency results in a marked instability of free-running period length (Zheng et al., 2001). To test whether the observed Per1-Dec interaction in LD entrainment extends to this phenotype, we re-entrained all mice to LD and then re-released them into DD to compare activity period lengths of individual mice between both DD intervals. In WT animals, period length was very stable with an average period difference between both DD episodes of less than 0.2 h (Fig. 3B). Increased period variations were observed in Per1 mutant mice (Fig. 3B, statistics in Suppl. Table S1), while no significant changes in period variability were seen in Dec1 and Dec2 single mutant animals (Fig. 3B, statistics in Suppl. Table S1). Although a trend toward a further increase in period destabilization was seen in Dec1/2 and Per1/Dec double and triple mutants (Fig. 3B, statistics in Suppl. Table S1), none of the data reached significance when compared to Per1 single mutant mice, indicating that long-term period stability is predominantly dependent on Per1. In summary, our free-running activity data suggest a dominant role for Per1 in the regulation of period length and stability while, in contrast to LD, functional interaction between Per1 and Dec1/2 was only detectable under triple mutant conditions.
Effects of Per1 and Dec1/2 on Bmal1 Expression in the SCN
To extend our investigations to the molecular level, clock gene expression profiles were determined by in situ hybridization of coronal SCN sections of animals sacrificed at 14, 20, 26, and 32 h after release into DD. For Per2, no significant changes in expression levels were found on a single Per1 or Dec deficient background (Suppl. Fig. S1A). All genotypes showed rhythmic Per2 mRNA levels with peak expression at 20 h after release into DD and comparable amplitudes (Fig. 4A). This situation was preserved in Per1/Dec double mutants. In Per1/Dec1/2 animals, Per2 expression was significantly reduced 26 h after release into DD, indicative of a synergistic activatory function of Per1 and Dec1/2 on Per2 expression during the early subjective night.
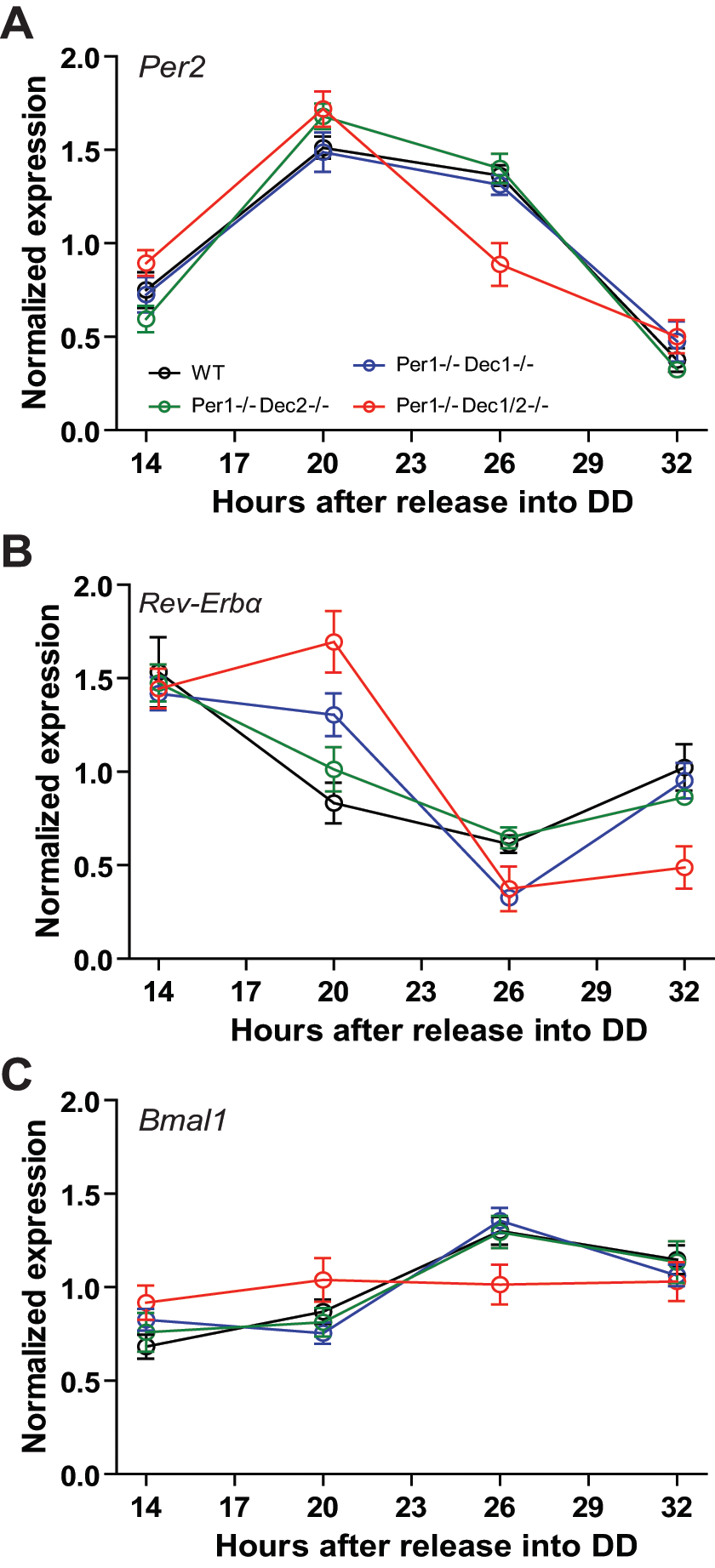
SCN clock gene expression. mRNA levels of (A) Per2, (B) Rev-Erbα, and (C) Bmal1 in the SCN 14, 20, 26, and 32 h after release into DD of WT (black), Per1–/–Dec1–/– (blue), Per1–/–Dec2–/– (green), and Per1–/–Dec1/2–/– (red) mice determined by X-ray autoradiography of 35S-labeled in situ hybridizations (ISH). Data are normalized to the average expression in WTs. All values are means ± SEM (n = 3).
The effect on Bmal1 expression was even stronger in these animals. While only minor changes in Bmal1 transcript levels were observed in Per1/Dec single and double mutants (Fig. 4C and Suppl. Fig. S1B; no changes in amplitude), Bmal1 mRNA rhythms were completely abolished in the SCN of Per1/Dec triple mutant animals. In WT, Per1/Dec single, and double mutant mice, Bmal1 showed rhythmic expression peaking between 26 h and 32 h in DD. In contrast, triple mutant Per1–/–Dec1/2–/– mice displayed medium-level Bmal1 transcript concentrations throughout the whole day. Thus, deficiency of Per1, Dec1/2, or a combination of Per1 and one Dec gene is dispensable for rhythmic Bmal1 expression, but at least one set of Dec alleles seems necessary for rhythmic Bmal1 transcriptional regulation in the SCN.
It is thought that PER and DEC proteins affect transcriptional regulation mainly by interfering with CLOCK/BMAL1-activated transcription via E-box enhancer elements (Honma et al., 2002; Reppert and Weaver, 2002; Sangoram et al., 1998; Sato et al., 2004; Shearman et al., 2000). To test if the observed strong interactive effect of Per1 and Dec1/2 on Bmal1 transcription is mediated via an intermediate E-box-controlled gene/protein other than Per2/PER2, we examined the circadian expression of Rev-Erbα (Nr1d1), a potent negative regulator of circadian Bmal1 transcription (Preitner et al., 2002). In wild-type animals, Rev-Erbα expression in the SCN showed robust rhythms with peak levels at 14 h, corresponding to the early subjective day (Fig. 4B and Suppl. Fig. S1). Interestingly, and unlike what had been observed for Per2 and Bmal1, Rev-Erbα levels were markedly changed in Per1, Dec1, and Dec2 single mutant mice with elevated expression at 20 h in DD (Suppl. Fig. S1C). This effect was similarly seen in Per1/Dec double and triple mutant animals. In Per1–/–Dec1/2–/– mice, this increase resulted in a shift of expression peak phase by 4 h (Fig. 4B). In addition, Rev-Erbα expression at 32 h in DD was reduced. Together, this resulted in an increase in amplitude of Rev-Erbα expression in the triple mutants, indicating an altered phase relationship between Per2 and Rev-Erbα expression rhythms in these mice.
To further analyze the interaction of Per1 and the Decs in the regulation of Bmal1 expression, we performed Bmal1 luciferase reporter gene assays (Nagoshi et al., 2004). In reporter cells transfected with Per1 plasmid alone, no changes in Bmal1-driven luciferase activity were observed. Transfection of one Dec plasmid increased luciferase activity by a factor of 2 to 4.5, with a higher induction capacity of Dec2 (Fig. 5A and statistics in Suppl. Table S2; Dardente et al., 2009). This induction was unchanged after co-transfection with Per1. However, co-transfection of both Decs together with Per1 resulted in a further increase of luciferase activity (Fig. 5A and statistics in Suppl. Table S2). Thus, both DECs have an activatory role on Bmal1 expression in vitro that is further potentiated by addition of Per1. We tested if this DEC/PER1-mediated activatory function on Bmal1 expression can overcome REV-ERBα-mediated repression of Bmal1 transcription. Therefore, we also transfected Rev-Erbα in our assay. Transfection of Rev-Erbα plasmid alone decreased luciferase activity (Fig. 5B, second column) (Guillaumond et al., 2005). By additional transfection of Per1 plasmid, no further changes were observed (Fig. 5B, third column). However, by co-transfection of Rev-Erbα plasmid and one Dec plasmid, the luciferase activity was elevated when compared to the effect of Rev-Erbα alone, with a higher induction capacity of Dec2 (Fig. 5B and statistics in Suppl. Table S3). Additional co-transfection of Per1 in the presence of one or both Decs and Rev-Erbα plasmids did not further affect luciferase activity (Fig. 5B, columns 6-8). Thus, DECs, but not PER1, exhibit the capacity to rescue the REV-ERBα-mediated repression of Bmal1 transcription.
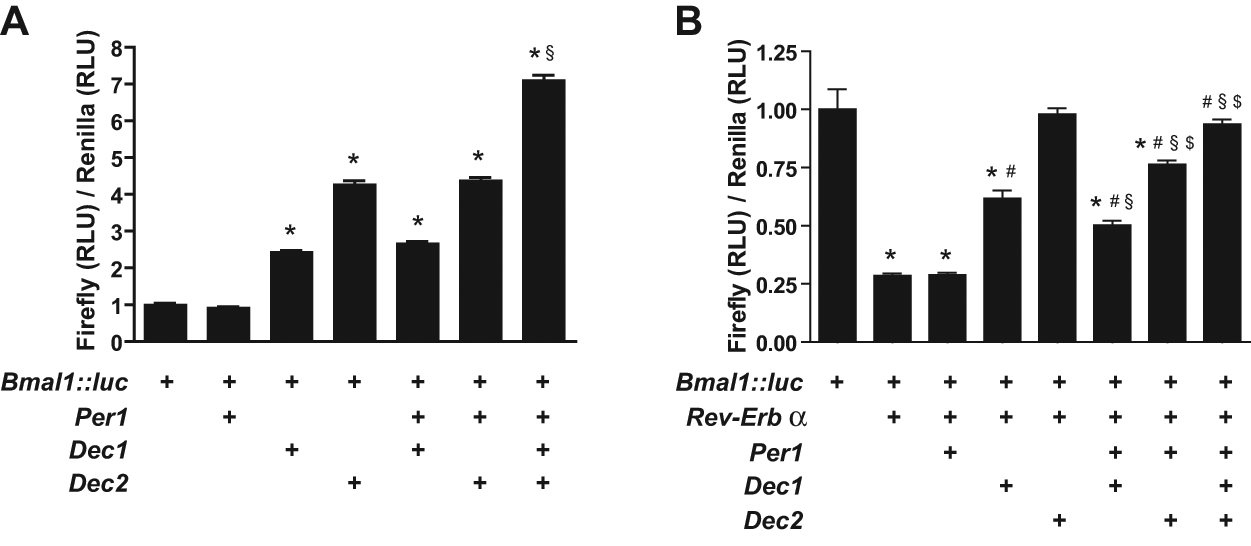
PER1/DEC influence on Bmal1 transcription. Bmal1::luc reporter gene assays using Rev-Erbα, Dec1, Dec2, and/or Per1 expression plasmids in HEK293 cells. Data are normalized to Bmal1::luc control and represented as mean ± SEM (n = 12 replicates for A and n = 6 replicates for B). (A) *p < 0.05 compared to Baml1::luc reporter (first column) or Bmal1::luc reporter and Per1 expression plasmid (second column); §p < 0.05 compared to Bmal1::luc reporter, Per1, and one Dec expression plasmid (fifth and sixth columns). (B) *p < 0.05 compared to Baml1::luc reporter (first column); #p < 0.05 compared to Baml1::luc reporter and Rev-Erbα expression plasmid (second column); §p < 0.05 compared to Bmal1::luc reporter, Rev-Erbα, and Per1 expression plasmid (third column); $p < 0.05 compared to Bmal1::luc reporter, Rev-Erbα, and one Dec expression plasmid (fourth or fifth column). RLU = relative luminescence units compared to renilla luciferase levels.
Taken together, DD expression profiling of core clock genes in the SCN and reporter gene assays revealed synergistic effects of Per1 and Dec1/2 on Per2, Rev-Erbα, and, most pronounced, Bmal1 expression together with full functional redundancy of Dec1 and Dec2, correlating with behavioral changes observed under the same conditions.
Discussion
In this study, we show a functional specificity in the interaction of Dec1 and Dec2 with Per1 in photic entrainment. Synergistic interactions of Dec1/2 genes with Per1 are critical for the normal synchronization of activity phase to the external LD cycle. Under free-running conditions, Per1-Dec1/2 synergy is preserved, but Dec1 and Dec2 show full functional redundancy in the modulation of rhythm period. In contrast, the stability of rhythm sustainment in DD appears to be exclusively dependent on Per1 function.
It has previously been shown that Per1 and Dec single as well as Dec1/2 double mutant mice entrain to LD cycles without any significant alterations in wheel-running activity when compared to wild-type littermates (Nakashima et al., 2008; Rossner et al., 2008; Spoelstra et al., 2004; Zheng et al., 2001). However, Per1 mutant mice show a tendency toward advanced activity onsets under these conditions (Spoelstra et al., 2004). In our experiments, Per1/Dec mutant mice entrained readily to an LD cycle, but Per1 single, Dec1/2 double, and Per1/Dec double and triple mutants exhibited advanced activity onsets or predark activity (Mrosovsky, 2001). The mechanism behind this phenomenon is still largely unknown but seems to involve a disruption of the negative masking capacity of light toward the end of the day (Mrosovsky, 2001). Photic masking was also suggested to underlie the normal entrainment behavior of the short period Per1 mutants in bright LD conditions (Zheng et al., 2001). The cumulative advance of activity onsets from Per1/Dec1 to Per1/Dec1/2 mutants indicates a complementary, but specific, role of both Dec genes in this process. Given the lack of significant changes in activity onsets in Dec single mutants (Rossner et al., 2008), the functional interaction with Per1 seems essential for a normal phase relationship between activity and the external LD cycle. Furthermore, the advanced onset activity in these mutants might also be explainable by an impaired sleep behavior. Per2 as well as Per1/2 mutant mice show a disruption of sleep consolidation (Kopp et al., 2002; Shiromani et al., 2004), while Per1–/– mice exhibit no abnormal sleep phenotype (Kopp et al., 2002). Mutations in the human DEC2 gene are associated with a short sleep phenotype (He et al., 2009). However, Dec2-deficient mice exhibit only minor changes in sleep architecture (He et al., 2009). Consistent with this, none of the Dec single mutants showed changes in activity onsets, unlike the Dec1/2 double mutants, suggesting a redundant function of the Dec genes in sleep regulation. In humans, equivalents of advanced activity onsets are observed in advanced sleep phase syndrome (ASPS) (Wulff et al., 2009). In ASPS patients, Per1 gene polymorphisms are associated with extreme morning preference and abnormal sleep patterns (Carpen et al., 2006). Furthermore, the ASPS phenotype is also associated with PER2 poly-morphisms, while alterations in PER3 are correlated to late chronotypes (Archer et al., 2003; Ebisawa et al., 2001; Viola et al., 2007), suggesting a high level of interaction of the 3 PERs in this process (Jones et al., 1999; Toh et al., 2001).
In constant conditions, all Per1/Dec mutant strains retained rhythmic activity patterns. As was previously shown (Zheng et al., 2001), DD period length was shortened in Per1 single mutant mice. In contrast, and contrary to what had previously been published (Rossner et al., 2008), in our setup, the DD period length of Dec1 single mutants was slightly shortened while the period length of Dec1/2 double mutant mice was unchanged compared to wild-type animals. The absolute differences, however, were small, and we believe that these effects might therefore be explainable by slightly different experimental conditions such as different lighting during the preceding LD entrainment. We also cannot exclude the possibility that these period length differences are in part based on aging effects, but the original study (Rossner et al., 2008) was performed on mice of roughly the same age as those used in our experiments. In Per1/Dec double mutants, period length was comparable to that of Per1–/– mice, while in mice deficient for Per1 and both Dec genes, dramatically shortened circadian period lengths were observed, suggesting a functional redundancy for both Decs in interacting with Per1 under free-run conditions. This is supported by the LL data. Following Aschoff’s rule (Aschoff, 1960), period lengths were increased in all investigated genotypes. Of all single mutants, this effect was most pronounced in Per1–/– animals (Steinlechner et al., 2002). While functional Dec1/2 redundancy seems to mask Per1-Dec interaction in double mutant animals, Per1/Dec1/2 triple mutants showed an increase in period length.
Our expression data suggest that Per1, Dec1, or Dec2 is dispensable for rhythmic Per2 expression in the SCN in DD (Rossner et al., 2008; Zheng et al., 2001). A moderate reduction in Per2 mRNA was seen in Per1/Dec1/2 triple mutants 26 h after release into DD, supporting a certain level of redundancy of Dec1 and Dec2 in Per2 transcriptional regulation. Studies on hepatic clock function indicate that Per2 rhythms are stabilized by systemic factors independent of local TTL function (Kornmann et al., 2007). In line with this, changes in gene activity were more pronounced for Rev-Erbα and Bmal1 in the triple mutant animals. Our in situ hybridization and reporter gene assay data postulate a synergistic activatory transcriptional role of PER1 and DEC1/2 on Bmal1 expression. This activatory PER1/DEC1/2 effect does not suffice to overcome the REV-ERBα-mediated repression of Bmal1 expression. Instead, the DECs, independently of PER1, can partially rescue the REV-ERBα-mediated repression of Bmal1 expression. It remains to be shown if this activatory function of the DECs on Bmal1 is direct or involves transcriptional regulation of intermediate factors. For example, it might depend on interactivity with retinoic acid–related orphan receptors. Of note, the mouse Bmal1 promoter contains, in addition to a retinoic acid–related orphan receptor response element (RORE), 3 noncanonical E-boxes (nucleotides 7082-7087 bp, 7674-7679 bp, and 7711-7716 bp of GenBank AB064982.1 nucleotides 6995-8103 bp), located 1033 bp, 443 bp, and 405 bp upstream of the ATG, respectively. Together, our data postulate a critical role for Per1-Dec1/2 interaction for Bmal1 rhythmicity. In contrast to Per2 and Bmal1, Rev-Erbα mRNA levels at 20 h in DD in Per1/Dec1/2 triple mutants were increased, indicating a primarily inhibitory function for Per1 and Dec on Rev-Erbα transcription in the second half of the subjective day. A similar mode of action has been postulated for Per1 (Preitner et al., 2002). In contrast, during the second half of the subjective night, Rev-Erbα expression was suppressed in Per1/Dec1/2 mutants, indicating an activating Per1/Dec function at this time. Such a time of day-dependent transactivation functionality of Per1-Dec is supported by a number of in vitro studies showing that DECs have a bidirectional effect on the transcription of E-box-containing genes (Azmi et al., 2003; Dardente et al., 2009; Hamaguchi et al., 2004; Honma et al., 2002; Kawamoto et al., 2004; Li et al., 2003; Rossner et al., 2008).
Of note, the increased amplitude of Rev-Erbα expression in the triple mutants does not result in improved rhythmicity of Bmal1 expression. Rather, the rhythmic Bmal1 expression is lost in triple mutants. A possible explanation for this effect would be that the altered phase relationship between the positive Bmal1 regulator Per2 (Schmutz et al., 2009; Shearman et al., 2000) and the transcriptional repressor Rev-Erbα (Preitner et al., 2002) results in a blunting of Bmal1 transcription. However, other uncharacterized proteins that act as transcriptional modulators as well as posttranslational processes could also be involved. Interestingly, Per1/Dec1/2 triple mutant mice did not lose activity rhythmicity, even though Bmal1 expression was disrupted in the SCN. This finding is in line with previous studies showing that the presence of arrhythmic BMAL1 is sufficient for the maintenance of behavioral rhythmicity (McDearmon et al., 2006). Nevertheless, disrupted Bmal1 transcription together with an altered phase relationship of Per2 and Rev-Erbα expression in Per1/Dec triple mutants might result in a weakened oscillator, which might be reflected in changes in period length of these animals in DD. A similar cumulative effect on free-running period length was observed in other double mutant lines such as Per1/Rev-Erbα and Per2/Rev-Erbα (Jud et al., 2010; Schmutz et al., 2009).
It was recently postulated that in Drosophila, CWO functionally synergizes with PER and has both activatory and inhibitory functions on CLK/CYC-activated transcription (Kadener et al., 2007; Richier et al., 2008). In this study, we show an analog Per(1)-Dec interaction in the mammalian TTL. The molecular mechanism behind the bi-directionality of PER1 and DEC action on E-box-mediated and Bmal1 transcription in the SCN remains to be solved. Our data suggest that it seems to depend on daytime and tissue type (Rossner et al., 2008), as well as on lighting conditions and the absence or presence of other TTL proteins.
Footnotes
Acknowledgements
We would like to thank Dr. Reshma Taneja for the generous gift of Dec1 mutant mice and Jürgen Ripperger and Urs Albrecht for the Rev-Erbα plasmid. Furthermore, we are grateful to Drs. Johanna Barclay and Lars Geffers for critical reading of the manuscript. This project was funded by a project grant (OS 353/2-1) and an Emmy Noether Fellowship (H.O.) of the German Research Foundation (DFG). M.J.R. acknowledges funding by the DFG (KFO241).
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
