Abstract
Animals adapt to a changing environment by synchronizing their circadian rhythms to different stimuli, the strongest and most reliable being the daily light-dark cycle. Photic information reaches the central circadian pacemaker, the suprachiasmatic nucleus (SCN), which drives rhythms in physiology and behavior throughout the brain and body. The endocannabinoid system (ECS) is a neuromodulatory system that is present within the SCN, including the primary receptor, cannabinoid receptor 1 (CB1). Exogenous cannabinoids that target CB1 inhibit the phase-shifting effects of light in hamsters, mice, and rats. Furthermore, there is evidence in cultured microglial cells that cannabidiol (CBD), a constituent of Cannabis sativa, alters core circadian clock genes, while the CB1 agonist delta-9-tetrahydrocannabinol (THC) does not. The CB1 agonist studies were conducted using male animals only, but cannabinoids exhibit sex-dependent effects in various aspects of physiology and behavior. In addition, the effects of CBD on circadian behavioral rhythms have yet to be investigated. Therefore, we decided to test the effects of acute injections of CBD or the CB1 agonist CP 55,940 on light-induced phase delays in male and female C57BL/6J mice. Animals received a single injection at circadian time (CT) 15.5, followed by a 10-min light or dark (sham) pulse at CT 16. Running-wheel activity was monitored to determine activity levels and the behavioral phase shifts from different treatments. We observed a sex difference in the magnitude of phase delay size in response to CP 55,940 administration. Males had attenuated phase delays with increasing doses of CP 55,940, while females did not differ from control. Various doses of CBD had no effect on the phase-delaying effects of light in either sex. Our results show a sex difference in the gating of photic phase shifts by CB1 activation.
Keywords
Circadian rhythms are approximately 24-h oscillations in physiology and behavior, and nearly every cell in the body has an endogenous, autoregulatory, transcription-translation negative feedback loop that makes up the molecular clock. The suprachiasmatic nucleus of the hypothalamus (SCN) serves as the central circadian pacemaker in mammals, synchronizing rhythms in physiology and behavior to environmental signals, the most dependable being the daily light-dark (LD) cycle (Liu et al., 2007; Mohawk et al., 2012). Photic information is transmitted to the SCN from specialized retinal ganglion cells in the retinohypothalamic tract (Hendrickson et al., 1972; Moore and Lenn, 1972; Provencio et al., 2000; Gooley et al., 2001). SCN cells generate robust, efficient, and coordinated rhythms through their interconnected neuronal and glial network (Welsh et al., 2010), which orchestrate the timing of other central and peripheral tissue clocks (Mohawk et al., 2012).
The endocannabinoid system (ECS) is widely spread throughout the brain and body and acts as a neuromodulatory signaling mechanism within the central nervous system (Lu and Mackie, 2016). Endogenous cannabinoids (eCBs) are lipid-soluble signaling molecules that activate their G-protein-coupled receptors, cannabinoid receptors 1 and 2 (CB1 and CB2); CB1 is widely spread throughout the central nervous system (Howlett et al., 2002). eCBs primarily act as retrograde synaptic messengers targeting CB1 on excitatory and inhibitory neurons (reviewed in Kano et al., 2009) but can also target CB1 on astrocytes (Navarrete and Araque, 2008; Stella, 2010; Han et al., 2012). Retrograde eCB signaling mediates both short-term (Kreitzer and Regehr, 2001; Ohno-Shosaku et al., 2001; Wilson and Nicoll, 2001) and long-term synaptic plasticity throughout the brain (Chevaleyre and Castillo, 2003; Gerdeman et al., 2002; Marsicano et al., 2002; Robbe et al., 2002). Furthermore, CB1 has influence as a temporal window component for synaptic plasticity in the amygdala (Fonseca, 2013; Madeira et al., 2020).
The major psychoactive constituent of Cannabis sativa, delta-9-tetrahydrocannabinol (THC), mainly elicits its effects through CB1 (Herkenham et al., 1990; Matsuda and Young, 1990; Felder et al., 1992). On the contrary, there is minimal evidence that the non-psychoactive constituent of C. sativa, cannabidiol (CBD), binds CB1 (Laprairie et al., 2015); however, CBD has a diverse repertoire of pharmacological targets (reviewed in Ibeas Bih et al., 2015). Interestingly, the application of CBD, but not THC, in cultured BV-2 microglial cells affected the expression of core clock genes both with and without lipopolysaccharide stimulation (Lafaye et al., 2019), indicating multiple routes for marijuana or its individual compounds to interact with circadian rhythms.
CB1 has been localized to the SCN in mice (Wittmann et al., 2007; Acuna-Goycolea et al., 2010; Hrabovszky et al., 2012), hamsters (Sanford et al., 2008), and rats (Sládek et al., 2021) and exhibits rhythmicity (Lafaye et al., 2019; Sládek et al., 2021). Exogenous cannabinoids modulate neuronal (Acuna-Goycolea et al., 2010) and astrocyte (Hablitz et al., 2020) signaling in the SCN. Furthermore, activation of CB1 with exogenous cannabinoid administration inhibits the phase-delaying effects of light on wheel-running behavior in each of these species (Sanford et al., 2008; Acuna-Goycolea et al., 2010; Filipovská et al., 2019). SCN explants from PER2::LUC mice that received exogenous cannabinoid application had time-of-day specific effects on PER2 phase (Sládek and Sumová, 2019; Hablitz et al., 2020), and inhibition of the synthesis enzymes for endocannabinoid 2-arachidonoylglycerol also had a time-of-day specific PER2 phase response when paired with the light-mimicking effect of N-methyl-
Most of the previous studies were conducted using male animals only or did not differentiate the results between males and females, but cannabinoids exhibit sex-dependent effects in various aspects of physiology and behavior (reviewed in Craft et al., 2013; Wagner, 2016). In addition, there is a sex difference in the phase response to light in mouse wheel-running behavior early in the subjective night (Blattner and Mahoney, 2013). With respect to this, we further investigated what effects acute administration of varying doses of the CB1 agonist, CP 55,940, or the exogenous cannabinoid, CBD, had on phase delay responses in male and female C57BL/6J mice.
Materials and Methods
Animals
Seven-week-old C57BL/6J female and male mice were acquired from Jackson Labs (The Jackson Laboratory, Bar Harbor, ME, USA). Animals had ad libitum access to food (Prolab RMH 3000, 5P00) (LabDiet, St. Louis, MO, USA) and water. All experiments and procedures were approved by the Institutional Animal Care and Use Committee (IACUC) at Kent State University and followed the National Institutes of Health guidelines regarding the care and use of laboratory animals for experimental procedures.
Experimental Setup
Mice were individually housed in plexiglass cages (12″ × 5″ × 5.25″) with free access to a running wheel (4.5″ × 2.25″) to monitor locomotor activity. Animals were entrained to a 12:12 LD cycle for 10-14 days to acclimate from travel. Overhead lighting was provided in the chamber (Actimetrics Circadian Cabinets; Actimetrics, Wilmette, IL, USA) and was on a programmed LD cycle (the average light intensity was approximately 100 lux), and the ambient temperature was 21 ± 2 °C. Following entrainment to the 12:12 LD cycle, animals were released into constant darkness (DD) for 10-14 days before treatments began. Animal handling during DD was conducted in dim red light (<1 lux). Cage changes occurred at random times throughout the day and were conducted several days before each treatment. During cage changes, body weight (g) was collected with a top-pan balance to an accuracy of 0.1 g. The mean body weight of each sex was used to determine the approximate dose for females and males before each treatment.
Drugs
Cannabidiol and (±)-CP 55,940 were purchased from Cayman Chemical Company (Ann Arbor, MI, USA). They were dissolved in dimethyl sulfoxide (DMSO), followed by 1:1 dilution with 0.9% saline for working solutions. Vehicle (DMSO:saline), cannabidiol (10, 25, and 50 mg/kg), and CP 55,940 (0.125, 0.25, and 1 mg/kg) were administered intraperitoneally.
Phase Delay Experiments
Mice were housed in DD for 10-14 days before the first treatment. On the day of each treatment, animals received an acute injection of vehicle or drug at CT 15.5, followed by a 10-min light pulse (or dark/sham pulse in the CBD experiment) at CT 16 of approximately 50 lux. Animals were then allowed to free-run in DD for 13-16 days before the next treatment.
For the CP55,940 experiment, the order of treatment was not completely randomized. In 1 set of mice, the initial 2 trials were counterbalanced between vehicle and 0.125 mg/kg, followed by a third trial at 0.25 mg/kg. In a second group of mice, the first 2 injections were randomized among all 4 doses (vehicle, 0.125, 0.25, and 1 mg/kg), with a third injection of 1 mg/kg being given to all mice to equalize out the sample sizes. We tested the data for order effects, but no significant differences were detected.
For the CBD experiment, each mouse was used for 4 treatments, with 2 injections before a light pulse and 2 before a dark pulse. The order of the light or dark pulses was randomized. Each mouse received all 4 CBD doses across the 4 treatments in a randomized order.
Activity Monitoring and Measurement
ClockLab data collection software (Actimetrics) was used to monitor wheel-running activity. Wheel revolutions were stored in 1-min data bins by collection with an infrared sensor. ClockLab Analysis software (Actimetrics) was used to quantify wheel-running activity into 24-h activity profiles, total activity counts per day, determine circadian time, and measure phase shifts. Phase shifts were calculated by measuring the difference in linear regression line fits on actograms for 7 days prior to the treatment day and for 10 days after, skipping the first 3 days after the treatment. The difference in the 2 regression lines on the day following the injection is the calculated phase shift.
Statistics
CP 55,940 group comparison data were assessed by 2-way analysis of variance (ANOVA) (Sex × Drug Dose) with partial repeated-measures and Dunnett’s 2-sided multiple comparison test with control. CBD group comparison data were assessed by 3-way ANOVA (Sex × Drug Dose × Pulse). Repeated-measures analysis was not used for these data as multiple factors were not repeated across individual mice. However, for both experiments, we tested the analysis using both repeated-measures and non-repeated-measures procedures, and the conclusions of the study did not vary depending on the statistical model used. p < 0.05 was set for statistical significance.
Results
CP 55,940 Phase Delay
To ascertain whether exogenous cannabinoids elicit the same circadian behavioral response to light in females as they do in male rodents, free-running male and female mice received acute intraperitoneal injections of vehicle (DMSO:saline) or varying doses of CP 55,940 (0.125, 0.25, and 1 mg/kg) at CT 15.5 followed by a 10-min light pulse at CT16 (Figure 1). There were significant main effects of sex (F1,45 = 41.64, p < 0.001) and drug dose (F3,74 = 5.76, p = 0.001), and a significant sex-drug dose interaction (F3,74 = 3.29, p = 0.025). Interestingly, none of the doses of CP 55,940 given to female mice altered the magnitude of light-induced phase shift compared with vehicle (Dunnett’s test, p > 0.05), with all treatment groups having a greater than 2-h phase delay (Figure 1). Conversely, male mice exhibit a dose-dependent attenuation of light-induced phase shifts, with increasing dosages reducing the magnitude of the phase delay (Dunnett’s test, p < 0.05), which is in agreement with previous studies (Sanford et al., 2008; Acuna-Goycolea et al., 2010; Filipovská et al., 2019).
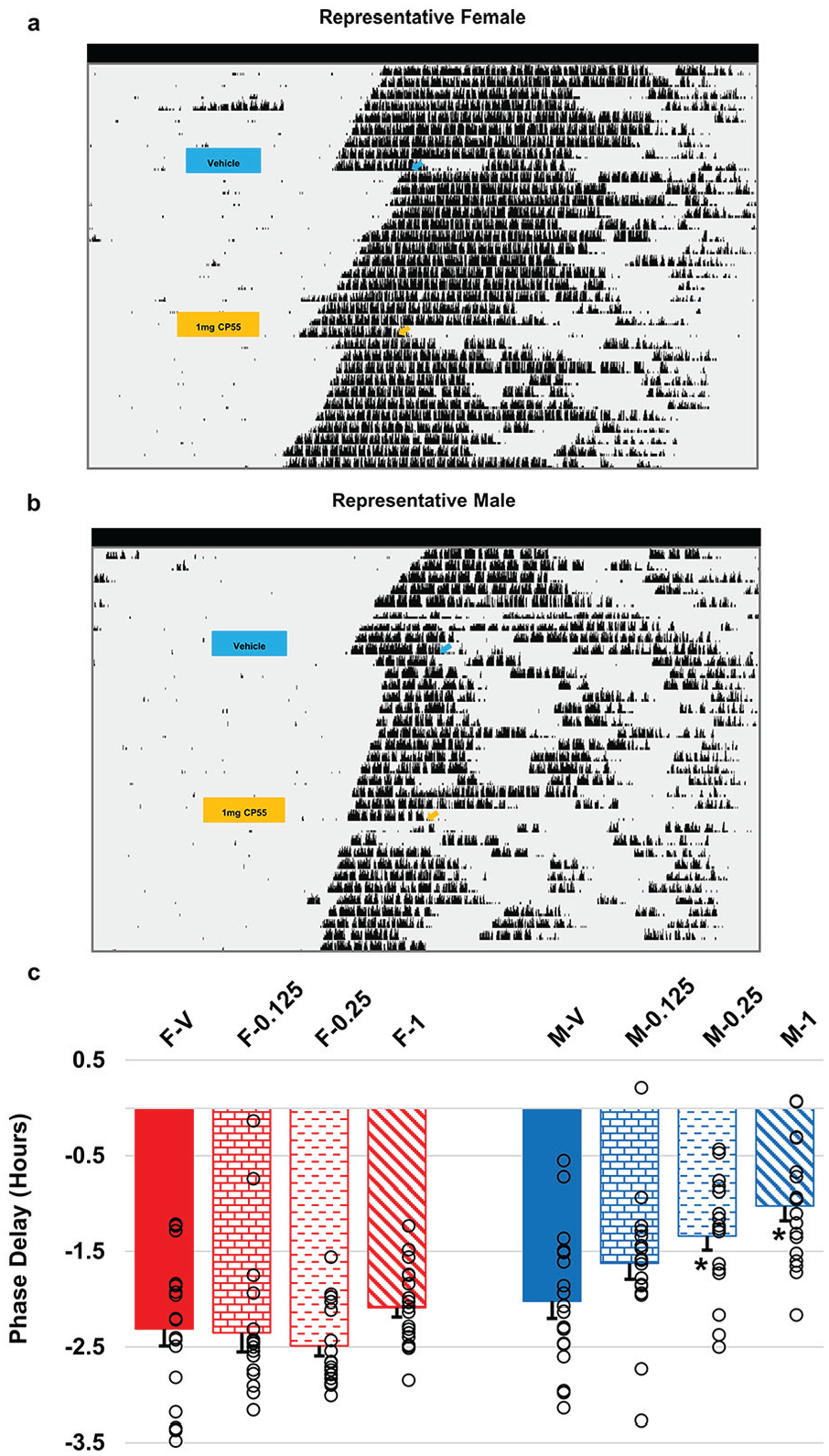
Phase delay response of female and male mice receiving cannabinoid receptor 1 (CB1) agonist injection paired with light pulse. Representative actograms of (a) female and (b) male mice on a running wheel in constant darkness (DD) that received an acute intraperitoneal injection of vehicle (DMSO:saline) or the CB1 agonist CP 55,940 (1 mg/kg) at circadian time (CT) 15.5, followed by a 10-min light pulse (~50 lux) at CT16 (represented by box/arrow) before being released back into DD. (c) Phase delay shifts in female (left) and male (right) mice that received vehicle (solid) or various doses (0.125 [brick], 0.25 [dashed], and 1 [slanted] mg/kg) of CP 55,940. Open black circles represent individual trials. (n = 16-18 per group). There was a significant interaction between sex and dose, with the 0.25 and 1 mg/kg CP55,940 doses significantly reducing the phase-shifting effects of light in males only (Dunnett’s 2-sided multiple comparison test, * denotes p < 0.05). Error bars represent standard error of the mean (SEM).
CP 55,940 Locomotor Activity
We noticed animals had an acute alteration of behavior after CP 55,940 injections during the 10-min light pulse period. In general, animals exhibited varying degrees of a sedation-like phenotype, albeit with their eyes remaining open during the light pulse. During our observations, it appeared that female mice were more active than male mice, so we decided to quantify locomotor activity on the day of injection (Figure 2). Raw wheel count data were extracted from the day of injection and quantified into 1-h segments for sex and treatment (Figure 2a and 2b). Wheel counts were high at the start of subjective night (CT 12) and began decreasing with the injection/light pulse at CT 15.5/CT 16. Animals that received vehicle and 0.125 mg/kg CP 55,940 had a rebound in wheel-running activity shortly after the light pulse, while animals that received 0.25 or 1 mg/kg CP 55,940 had minimal running afterward. To better summarize the data, we pooled wheel counts from CT 17-24 (1 h after reception of light pulse to end of subjective night). Two-way ANOVA revealed a main effect of dose (F3,3 = 15.29, p < 0.001) but not sex (F1,3 = 3.11, p = 0.080), and there was no significant interaction (F3,3 = 0.98, p = 0.406), with wheel-running significantly reduced in the 0.25 and 1 mg/kg CP 55,940 groups compared with vehicle control (Figure 3).
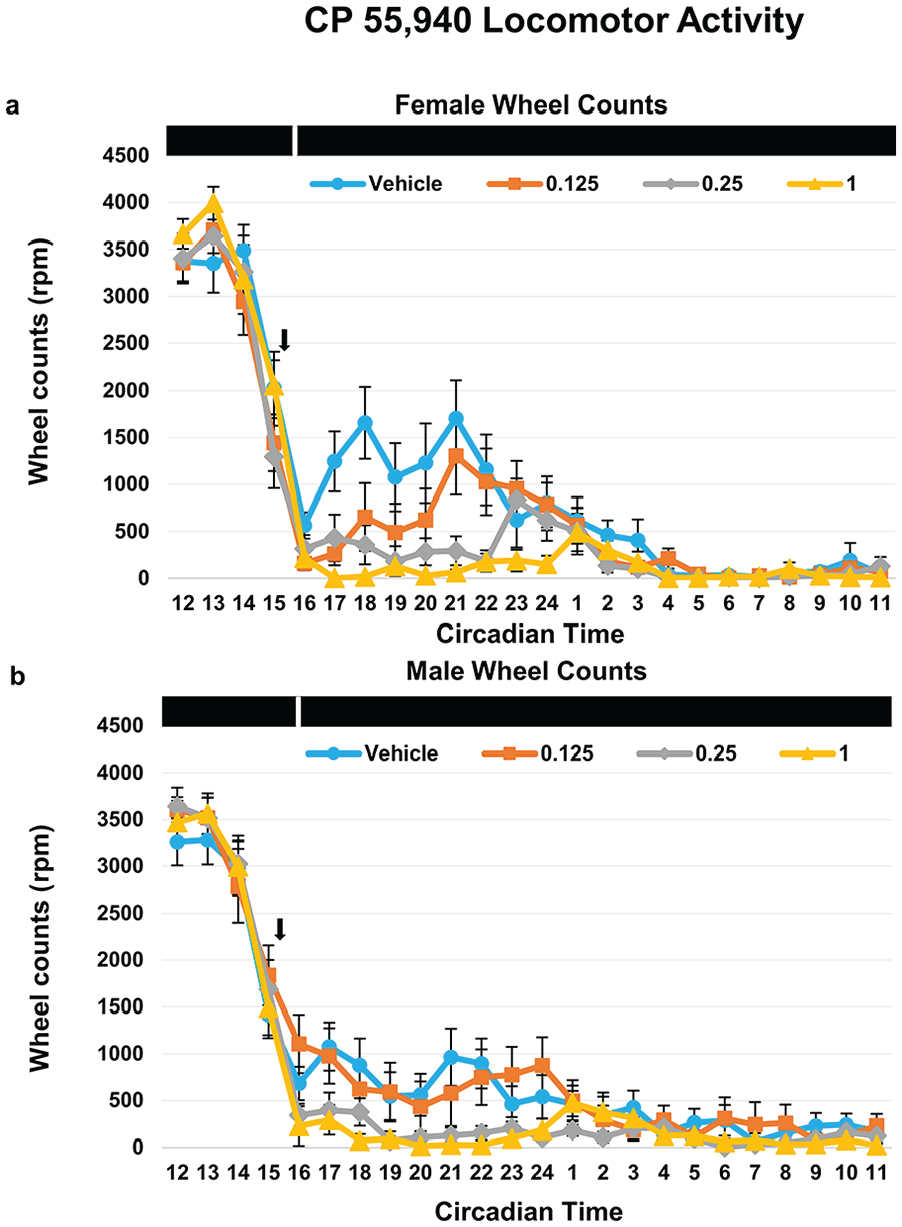
Raw wheel activity counts on the day of injection for (a) female and (b) male mice that received vehicle (circle) or varying doses of CP 55,940 (0.125 [square], 0.25 [diamond], and 1 [triangle] mg/kg). Wheel activity counts were exported in 1-min bins and summed into hourly bins to show the profile of locomotor activity following injections and the light pulse (n = 15-17 per group). Black arrow denotes time of injection (CT15.5), while black bar with white stripe on the top of graphs represents DD with the 10-min light pulse at CT 16 following the injection. Error bars represent standard error of the mean (SEM). Abbreviations: CT = circadian time; DD = constant darkness.
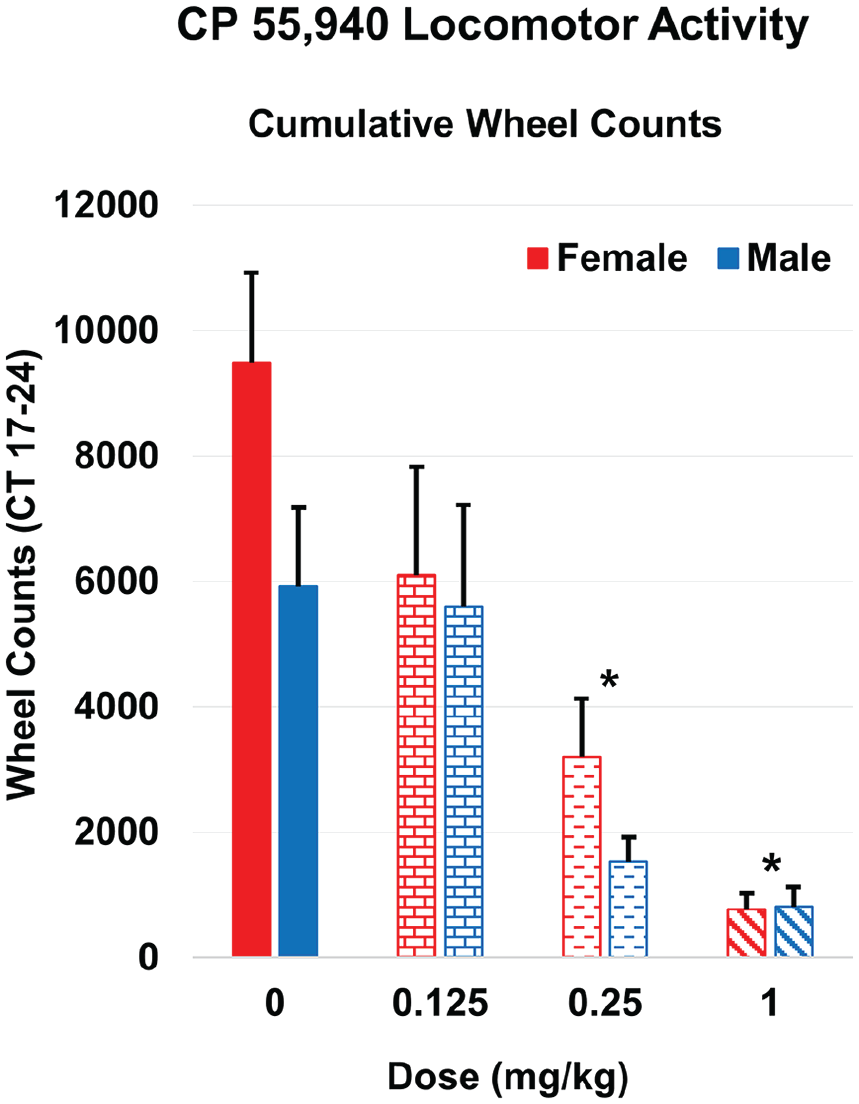
Cumulative wheel activity counts from CT 17-24 following injection of vehicle or CP 55,940. Two-way analysis of variance reveals a main effect of dose (p < 0.001) but not sex (p = 0.080), and there was no significant interaction (p = 0.406). Wheel-running was significantly reduced in the 0.25 and 1 mg/kg CP 55,940 groups (*p < 0.05) compared with vehicle control (n = 15-17 per group). Error bars represent standard error of the mean (SEM). Abbreviation: CT = circadian time.
CBD Phase Delay
Given that CBD altered core clock gene expression in BV-2 microglial cells (Lafaye et al., 2019), we sought to determine how acute administration of various doses of CBD (10, 25, and 50 mg/kg) affected the phase delay response of animals, given a 10-min light or dark (sham) pulse at CT16. Three-way ANOVA revealed no effects from any dose of CBD (F3,1 = 0.55, p = 0.646) in either sex compared with controls and no interactions with the pulse (F1,3 = 0.52, p = 0.666) or sex (F1,3 = 0.21, p = 0.891) (Figure 4). As expected, animals that received a light pulse had significantly larger phase shifts (F1,3 = 87.82, p < 0.001) compared with animals that received a dark (sham) pulse (Figure 4). We did observe a general sex difference in the magnitude of phase shift response to light at CT 16 (F1,1 = 12.77, p < 0.001), with female mice exhibiting larger shifts than male mice (Figure 4b), which is in accordance with a previous study (Blattner and Mahoney, 2013). There was no interaction between pulse and sex (F1,1 = 2.83, p = 0.095) or a 3-way interaction with drug, pulse, and sex (F3,1,1 = 0.37, p = 0.776).
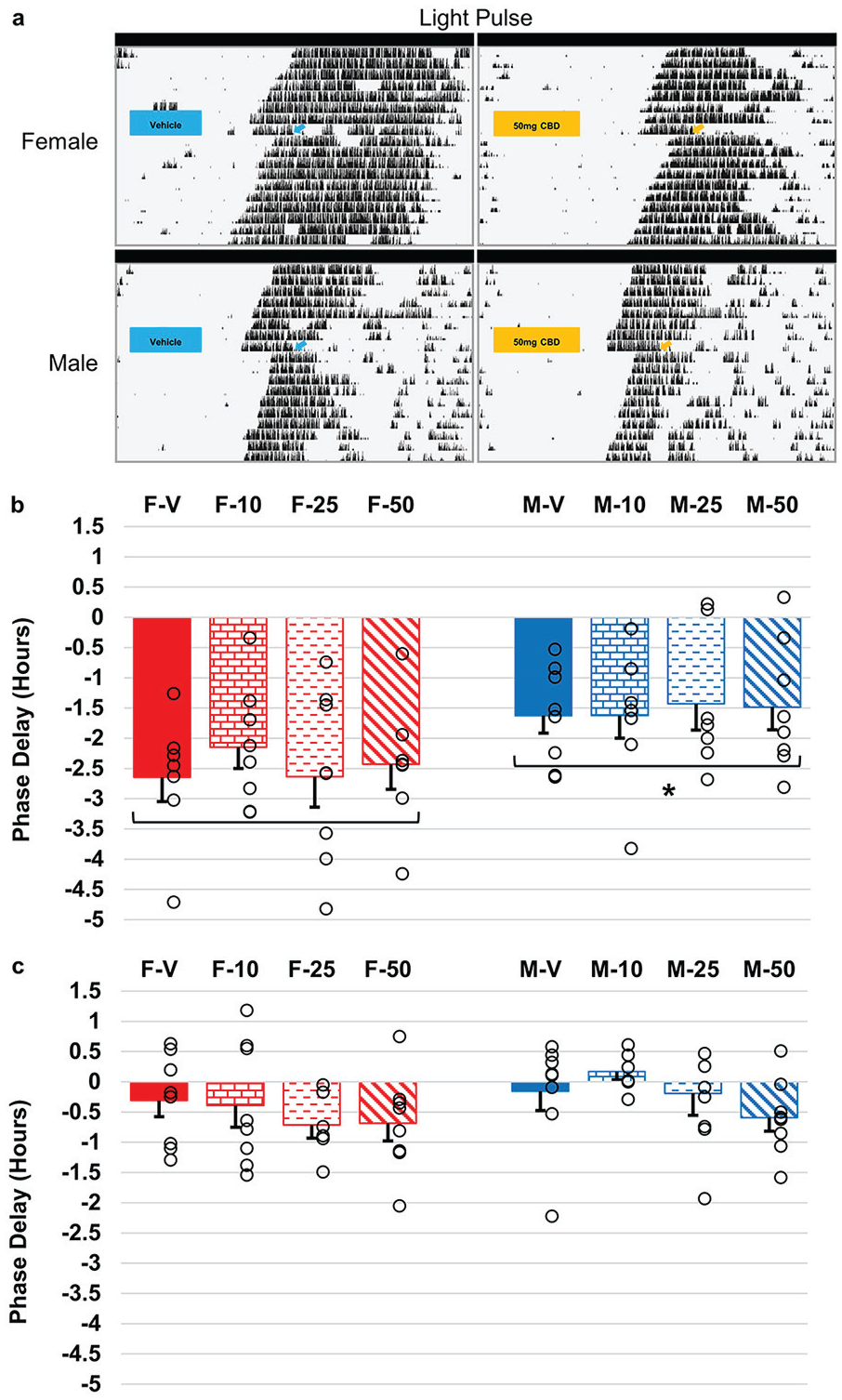
Phase delay response of female and male mice receiving cannabidiol (CBD) injections paired with a light or dark (sham) pulse. (a) Representative actograms of female and male mice on a running wheel in constant darkness (DD) that received an acute intraperitoneal injection of vehicle (DMSO:saline) or CBD (50 mg/kg) at circadian time (CT) 15.5, followed by a 10-min light pulse (~50 lux) at CT 16 (represented by box/arrow) before being released back into DD. (b) and (c) Phase delay shifts in female (left) and male (right) mice that received vehicle (solid) (DMSO:saline) or various doses (10 [brick], 25 [dashed], and 50 [slanted] mg/kg) of CBD. Injections were given at CT 15.5, followed by (b) a 10-min light pulse (~50 lux) at CT16 (c) or 10-min dark (sham) pulse at CT16. Open black circles represent individual trials. (n = 6-8 per group). Analysis by 3-way ANOVA showed significant main effects of the light pulse and sex, but no significant interaction terms (*p < 0.05). Error bars represent standard error of the mean (SEM).
Discussion
Our results revealed a sex difference in the gating of photic phase delays by CB1 activation. In line with previous studies, male mice that received acute injections of the CB1 agonist, CP 55,940, prior to a light pulse at CT16 had attenuated phase shifts (Sanford et al., 2008; Acuna-Goycolea et al., 2010; Filipovská et al., 2019). However, female mice at all doses tested did not differ from control (Figure 1). Both sexes had a reduction in wheel-running activity following CP 55,940 administration, with larger doses eliciting longer suppression of wheel activity (Figures 2 and 3). This suggests that the sex difference in the response of the circadian clock is specific to the clock mechanism and not representative of an overall difference in responsiveness to the drug. In contrast, acute delivery of various doses of CBD during the early subjective night did not shift the timing of rhythms and did not alter the phase-shifting effects of light (Figure 4).
Male and female mice had different reactions to light reception in the presence of the CB1 agonist CP 55,940, leading to differences in the photic sensitivity of the clock. The available studies looking directly at the interactions of cannabinoids and rhythms were mostly conducted in male animals (Sanford et al., 2008; Acuna-Goycolea et al., 2010; Filipovská et al., 2019; Sládek et al., 2021), with some exceptions where the data were not parsed out separately to compare males and females (Hablitz et al., 2020) or combined due to no significant effect of sex (Sládek and Sumová, 2019). Gonadal hormones interact with the SCN, its inputs, and outputs, providing several potential routes whereby they can influence the timing of the central circadian system (reviewed in Bailey and Silver, 2014). In addition, gonadal hormones can influence the effects of cannabinoids, and sex differences exist in energy homeostasis, antinociception, cannabinoid abuse, and ECS enzymatic activity (reviewed in Craft et al., 2013; Wagner, 2016). Further investigations are needed to determine whether gonadal hormones play a role in the differential photic response to CB1 activation observed between males and females.
Many groups have reported on the cataleptic behavior that follows CB1 activation with large doses of agonist administration (Fan et al., 1994; Varvel et al., 2005; Hodges et al., 2020). Important to our experiment during this cataleptic state is that all the animals’ eyes remained open for the duration of the 10-min light pulse, which was also the case in hamsters (Sanford et al., 2008). Qualitative inspection of the actograms suggested that female mice might be more active than males during the light pulse, sparking our interest to further investigate the effects of CB1 agonism on wheel-running behavior. There is indication that female rats exhibit a decline in locomotor activity and increased propensity to develop catalepsy compared with males (Tseng and Craft, 2001; Craft et al., 2012). However, our data did not reveal a sex difference in activity following CP 55,940 administration, consistent with a recent report (Hodges et al., 2020). There was a suppression of wheel-running activity with larger doses of CP 55,940 (0.25 and 1 mg/kg) for the remainder of the subjective night (Figures 2 and 3). Others report biphasic responses in locomotor activity, with low doses (0.001 mg/kg) of CP 55,940 increasing locomotion and higher doses (0.4 mg/kg) suppressing locomotor activity in the open field test (Hodges et al., 2020). We did not investigate locomotor activity after CBD administration because no behavioral differences were observed between treatments or sexes, and an overview of actograms did not show any unexpected differences in activity after injections.
The CBD doses acutely administered in this study did not affect photic or non-photic shifts in the behavioral output of the clock. Our results are somewhat surprising from the basis that CBD, but not THC, altered core clock genes in cultured microglial cells (Lafaye et al., 2019), yet we show that a THC-like synthetic agonist targeting CB1 alters behavioral rhythms while CBD did not. This could be due to the experimental design differences of working with cultured cells versus a living animal. Irrespective of the lack of effect with CBD, we confirmed a known sex difference in the magnitude of phase shifts induced by the light pulse (Figure 4b), with females displaying larger delays than males (Blattner and Mahoney, 2013). As expected, animals that received a dark (sham) pulse after CBD administration did not have a significant phase shift compared with all groups that received photic stimulation (Figure 4).
The sex difference in the gating of photic phase shifts by CB1 activation highlights the importance of evaluating both males and females in studies on basic circadian clock functions. This sex difference was not mirrored by changes in the acute suppression of locomotor activity by the CB1 agonist, indicating a circadian-clock-specific mechanism. The lack of effect of CBD in these data supports the idea that cannabinoids primarily act on the circadian clock via the CB1 receptor.
Footnotes
Acknowledgements
This research was supported by Kent State University and NIH award R15GM134528 to E.M.M. We would like to thank Hunter Frey-Burkart for his help with animal husbandry.
Author Contributions
T.D.N. and E.M.M. designed the research, analyzed the data, and wrote the manuscript. T.D.N. performed the research.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
