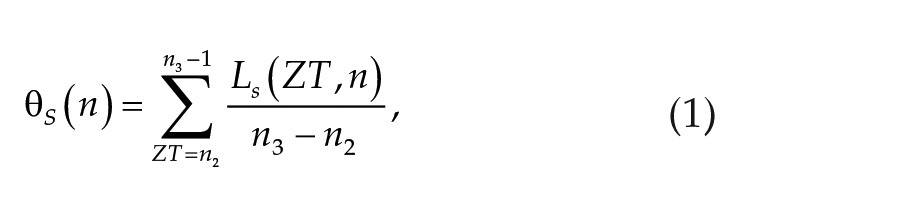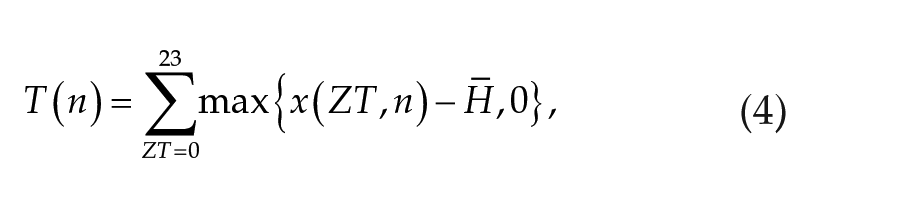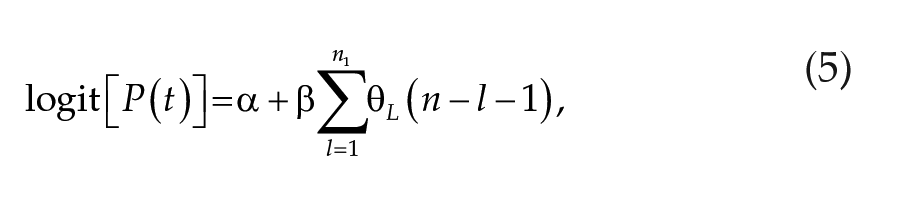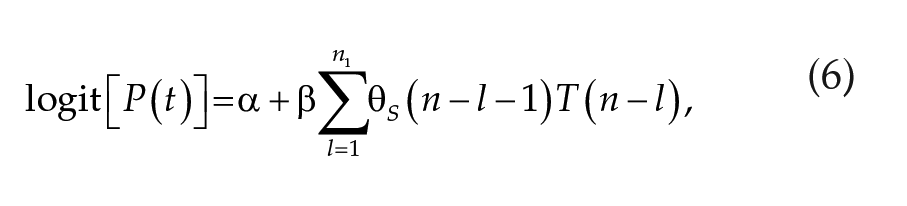Abstract
Many marine organisms synchronously spawn at specific times to ensure the success of external fertilization in the ocean. Corals are famous examples of synchronized spawning at specific lunar phases, and two distinct spawning patterns have been observed in two dominant taxa: merulinid corals spawn at regular lunar phases, several days after the full moon, whereas Acropora corals spawn at more irregular lunar phases around the full moon. Although it has been suggested that the two coral taxa have different responses to moonlight and seawater temperature, their spawning times have never been analyzed by integrating the two environmental factors, resulting in an incomplete understanding of the regulatory mechanisms of spawning. In this study, we developed a new predictive model of coral spawning days by integrating moonlight and temperature effects based on the external coincidence model for the lunar cycle. We performed model fitting using a 10-year monitoring record of coral spawning time in Taiwan. Our model successfully demonstrated the synergistic effects of moonlight and temperature on coral spawning time (days) and provided two testable hypotheses to explain the different spawning patterns regarding the preparation (maturation) process for spawning and the sensitivity to moonlight at different circadian phases: (1) Acropora corals may have an earlier onset and longer period of preparation for spawning than merulinid corals; and (2) merulinid corals may use moonlight signals near sunset, while Acropora corals may have a similar onset at approximately midnight. This is the first study to indicate the difference in circadian phase-dependent moonlight sensitivities between coral taxa, providing a basis for underlying coral spawning mechanisms for rhythmic studies.
Keywords
Lunar-controlled rhythms are widespread and important in marine organisms (Tessmar-Raible et al., 2011). A famous example is synchronous spawning of reef-building (scleractinian) corals. Most scleractinian corals are broadcast spawners (Baird et al., 2009), and many of them spawn synchronously after the full moon (Babcock et al., 1986, 1994; Harrison et al., 1984; Lin and Nozawa, 2017; Mendes and Woodley, 2002) to ensure the success of external fertilization (Harrison et al., 1984). It has also been reported that the lunar phase at which spawning occurs depends on coral species: Acropora corals, belonging to the genus Acropora, spawn in a wide range of lunar phases from the full moon to the last quarter moon (Fogarty et al., 2012; Lin and Nozawa, 2017). In contrast, merulinid corals, which belong to the family Merulinidae, including multiple genera such as Favites and Dipsastraea, spawn within a narrow range of lunar phases between 5 and 8 days after the full moon (DAFM) in Taiwan (Lin and Nozawa, 2017).
The difference in spawning time among coral species could be caused by different responses to environmental cues for spawning. Spawning time can be regulated by seawater temperature (Fujiwara et al., 2015; Nozawa, 2012; Paxton et al., 2016; Sakai et al., 2020; Shimoike, 1999), moonlight (Harrison et al., 1984; Hunter, 1988; Kaniewska et al., 2015; Lin, 2019; Lin et al., 2021), wind speeds (Sakai et al., 2020; Van Woesik, 2010), and solar flux (Sakai et al., 2020; Van Woesik et al., 2006). Among these, it has been suggested that spawning responses to moonlight and seawater temperature may differ between Acropora and merulinid corals (Lin et al., 2021; Lin and Nozawa, 2017). In Acropora corals, the presence of moonlight may be essential for spawning (Lin et al., 2021) because spawning was suppressed by blocking moonlight in Acropora millepora (Kaniewska et al., 2015) and Acropora hyacinthus (Lin, 2019). In contrast, in the merulinid coral Dipsastraea speciosa, spawning was suppressed by the presence of moonlight (Lin et al., 2021). Warmer seawater temperatures advanced the spawning time of Acropora corals (Fujiwara et al., 2015; Nozawa, 2012; Paxton et al., 2016; Sakai et al., 2020; Shimoike, 1999), while the effect of temperature on spawning time remains unclear in merulinid corals.
To unravel the different mechanisms regulating spawning time between Acropora and merulinid corals, it is necessary to understand how moonlight and temperature signals are integrated as a spawning cue. Recent manipulation experiments demonstrated that the merulinid coral D. speciosa spawned in response to the darkness between sunset and moonrise (Lin et al., 2021). This suggests that corals may respond to the presence or absence of moonlight at a certain time of day to trigger spawning. A similar example has been reported in the appearance time of marine midges Clunio tsushimensis that is regulated by lunar phase responding to moonlight at midnight (Neumann, 1995). Kaiser and Neumann (2021) demonstrated that such a mechanism is related to the external coincidence model for photoperiodism (Pittendrigh and Minis, 1964).
The external coincidence model has been proposed to explain the effect of sunlight at a certain time of day, which induces physiological responses (Pittendrigh and Minis, 1964). The model considers that the circadian oscillator entrained by sunlight generates the subjective time of day and the sensitivity to light signals over the course of the subjective time of day. We here extended the external coincidence model by integrating the role of moonlight as an induction signal of physiological response and the role of sunlight as an entrainment signal of the circadian oscillator. The physiological process of marine organisms can be inducted by the presence or absence of moonlight in the light sensitivity window regulated by a circadian oscillator entrained to sunlight. In corals, clock genes are conserved (Hoadley et al., 2011; Levy et al., 2007, 2011): the CRY1 and CRY2 genes, marker genes of the circadian oscillator, have been reported to show clear diurnal expression (Hoadley et al., 2011; Levy et al., 2007, 2011), which could be the basis of the circadian phase-dependent sensitivity. However, the role of circadian phase-dependent sensitivity to moonlight signals in the regulation of coral spawning remains elusive.
In this study, we developed a new predictive model of coral spawning days by integrating moonlight and temperature effects based on an external coincidence model for the lunar cycle. We performed model fitting using a 10-year monitoring record of coral spawning and examined how the different spawning patterns between Acropora and merulinid corals can be explained by different sensitivities to moonlight and temperature. This analysis enabled us to not only confirm the previously reported effects of those environmental factors on spawning time but also provide new testable hypotheses on the mechanisms of synchronous spawning in corals.
Materials and Methods
Data Collection
Data for spawning dates and seawater temperature were collected over a 10-year period from 2010 to 2020 (except 2011) at the two study sites, Gonggan and Dabaisha in Lyudao (Green Island), Taiwan (Suppl. Fig. S1). Monitoring methods of spawning dates and data from 2010 to 2016 have been reported previously (Lin and Nozawa, 2017). The survey was conducted during the spawning season from April to June in Lyudao. Because spawning occurs around the full moon, monitoring was performed for 1 to 2 weeks around the full moon in each month during the spawning season.
The present study focused on four dominant coral species: two Acropora species (Acropora hyacinthus and Acropora gemmifera) and two merulinid species (Favites pentagona and Dipsastraea speciosa). Because spawning patterns were distinctly different between Acropora and merulinid corals but were similar between the species within each taxon (Lin and Nozawa, 2017), we pooled spawning data for the two species within each taxon. The spawning data include a binary variable of 1 (spawning) or 0 (no spawning) in each species for each monitoring date. The variable was classified as 1 when spawning was observed regardless of the magnitude of spawning and was classified as n.a. (data not available) when the survey was canceled due to bad weather or other reasons.
For each species, spawning over multiple days at the same monitoring locations may not be independent data. Therefore, we used the first spawning date of each species observed in each monitoring month. The spawning date was recorded using the number of DAFM. We compared the first spawning dates between the two study locations or the two taxonomic groups with the exact Wilcoxon test using the package of exactRankTests in the free statistical computing software R version 3.4.1.
For the prediction of spawning day, we used the spawning data from −3 DAFM (= 3 days before the full moon) to the first spawning date in each month. When the data had a missing value at 1 day before the first spawning date, the data set was not used in merulinid corals but was used in Acropora corals. This treatment was based on the report that merulinid corals continuously spawned over a couple of nights, whereas Acropora corals often spawned over a single night in each spawning month (Lin and Nozawa, 2017).
Seawater temperature was recorded every hour at a depth of 5 m at the two study locations using HOBO temperature data loggers (Onset Computer Corp., Bourne, MA, USA). Gonggan tended to have lower seawater temperatures than Dabaisha. The presence or absence of moonlight was calculated every minute based on the lunar cycle at the two study locations using Calendar Calculation—National Astronomical Observatory of Japan (https://eco.mtk.nao.ac.jp/cgi-bin/koyomi/koyomix.cgi). The altitude was set to 0 m at both study locations for this calculation.
To examine the accuracy of our prediction models using separate independent data, we obtained spawning data of D. speciosa from a published experimental study where moonlight conditions were manipulated around the full moon in field and laboratory experiments (Lin et al., 2021). In the experiments, D. speciosa colonies were exposed to either the presence or absence of moonlight conditions for continuous nights, and the occurrence of spawning was recorded in each condition. A similar experiment was repeated at different lunar phases. From this study, we obtained seven datasets, each with four biological replicates per experimental condition. Seawater temperature data for the field experiments were recorded using the same method described above. The seawater temperature in the laboratory experiments was kept at 27.5 °C using an air conditioner. Moonlight conditions in the models were set to match the experimental conditions of moonlight.
Models for Predicting the Synchronous Spawning Day
To predict the coral spawning day, we constructed a model that integrates signals from moonlight and seawater temperature (Figure 1). Based on the external coincidence mechanism for the lunar cycle, we defined a lunar cue on date n,
where
In contrast, when corals are sensitive to dark signals for spawning,
The lunar cue on date n defined as
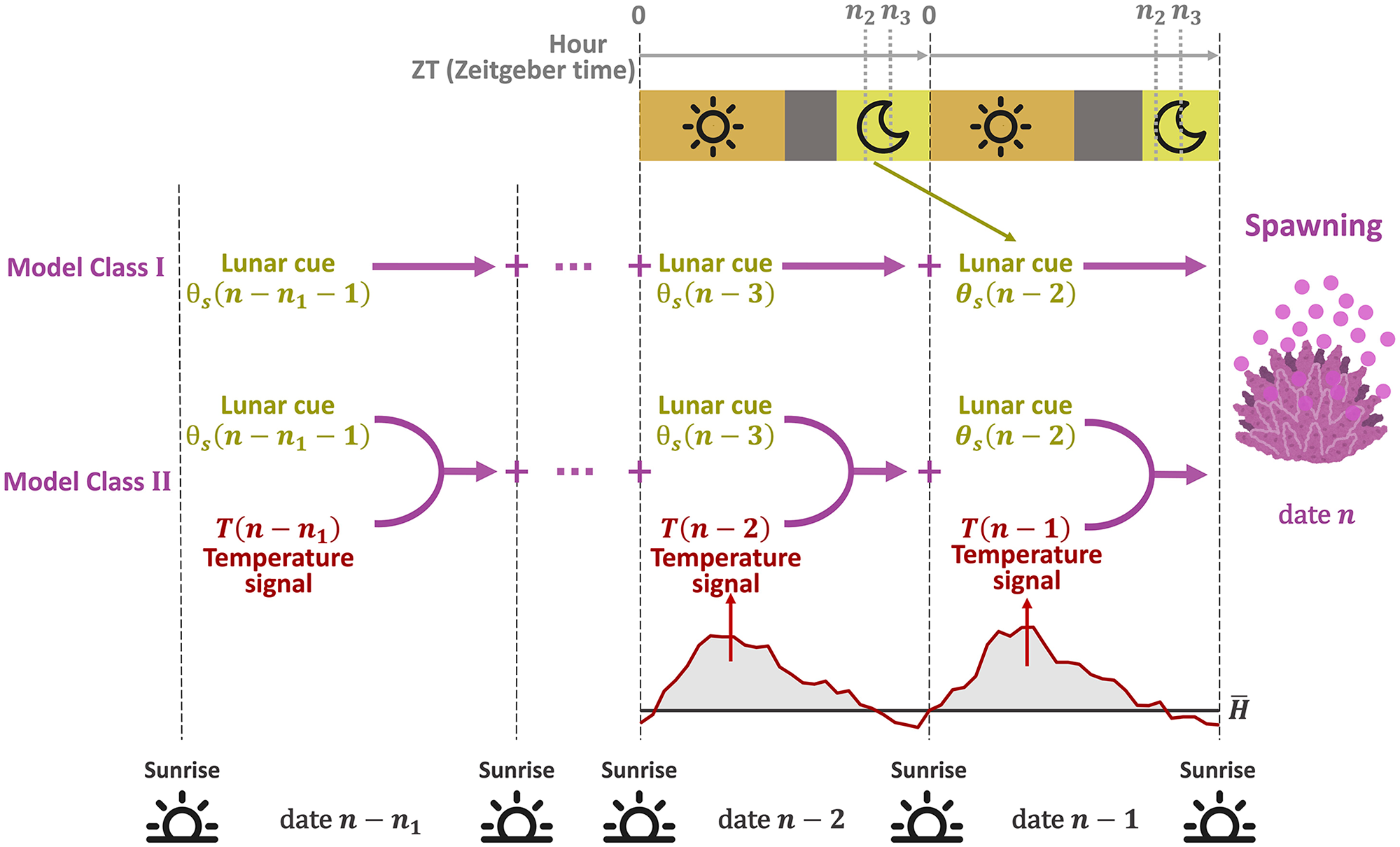
Model structure. For each day, lunar cues accumulate during the gating period (from
The temperature cue on date
where
Using lunar and temperature signals defined in equations (1) and (4), respectively, we developed two models that explain the relationship between environmental signals and spawning (Figure 1). In the first model (model class I), the spawning probability on date n, denoted as
Model class I:
where
The second model (model class II) considers that the lunar signal and the temperature signal are integrated to induce spawning:
Model class II:
where
We examined all combinations of potential values for
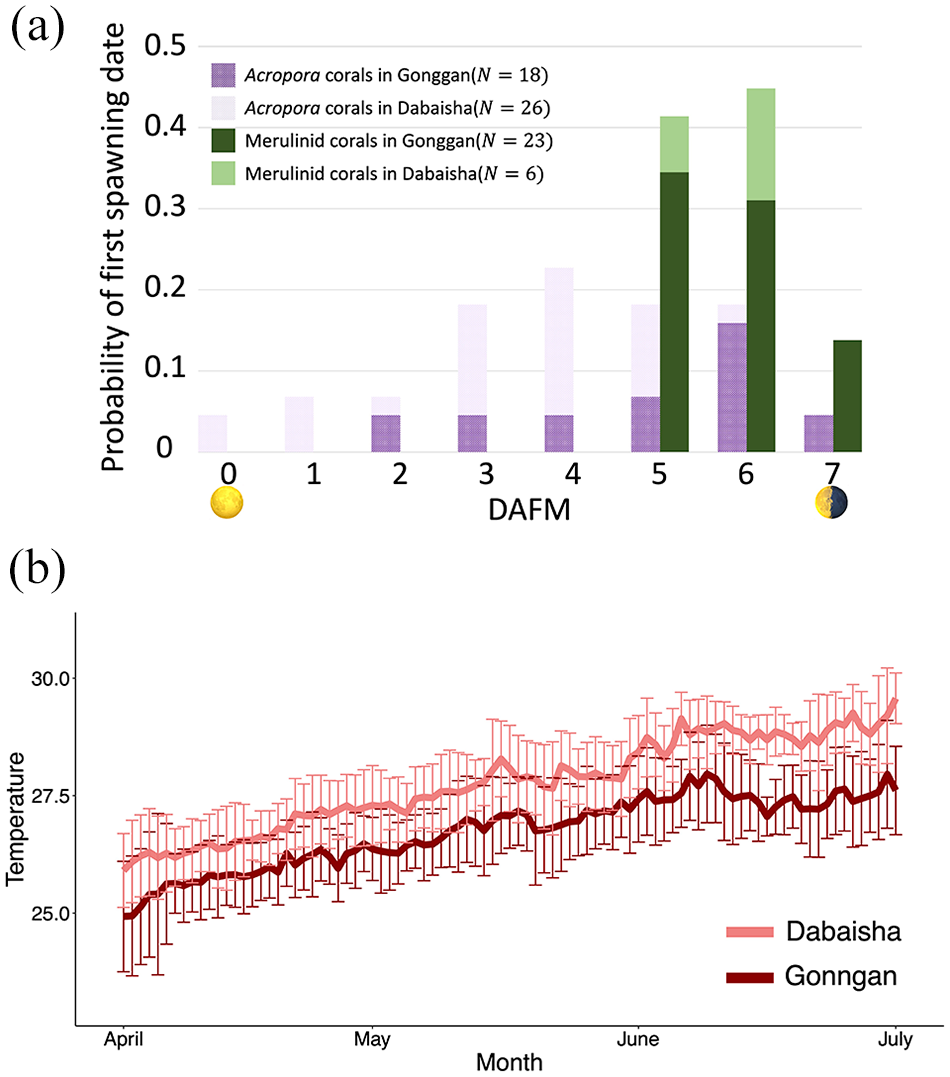
Spawning dates and seawater temperature in the spawning season. (a) Frequency distribution of the first spawning dates. The horizontal axis indicates the number of days after the full moon (DAFM). The vertical axis indicates the probability of the first spawning date. N indicates the number of observations. (b) Comparison of seawater temperature (daily mean ± SD) between the two monitoring locations, Gonggan and Dabaisha, during the spawning season in 2010-2020.
Results
The 10-year spawning records of two Acropora species (A. gemmifera and A. hyacinthus) showed a wide range of first spawning dates between the full moon and 7 DAFM (Figure 2a). The mean (SD) of the first spawning dates for the Acropora species was 3.98 (3.19) DAFM. In contrast, the first spawning days of the two merulinid species (D. speciosa and F. pentagona) were concentrated at approximately 6 DAFM, with a mean (SD) of 5.72 (0.49) DAFM on the first spawning day (Figure 2a). These distributions of the first spawning days between the Acropora and merulinid corals were significantly different (exact Wilcoxon test,
Different model classes were selected as the best models for the Acropora and merulinid corals (Table 1; Suppl. Fig. S2). Model class II, considering the combination of the moonlight signal and seawater temperature, best explained the spawning time dynamics for the Acropora corals. In contrast, in the merulinid corals, model class I, which considered only the moonlight signal, best explained the observed spawning pattern. The regression coefficients in the best models (model class I) were positive in the Acropora corals but negative in the merulinid corals (Table 1), indicating the opposite effect of the moonlight signal between the two coral taxa.
Variables and AIC values for the best models in each taxonomic group.
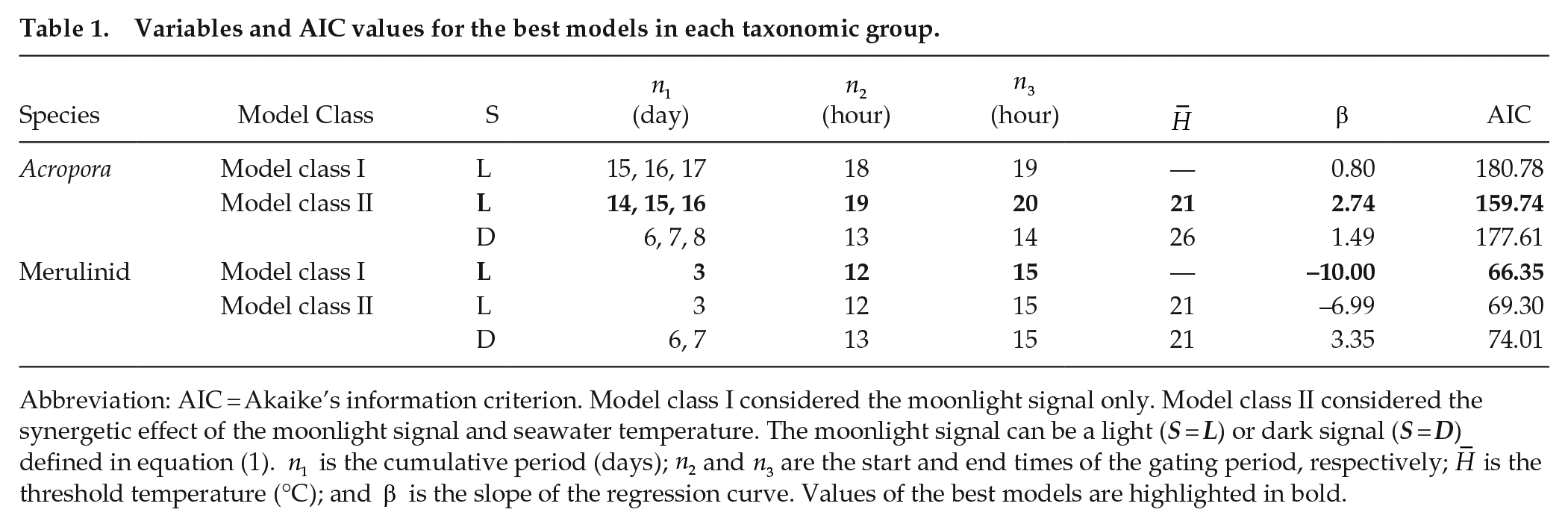
Abbreviation: AIC = Akaike’s information criterion. Model class I considered the moonlight signal only. Model class II considered the synergetic effect of the moonlight signal and seawater temperature. The moonlight signal can be a light (S = L) or dark signal (S = D) defined in equation (1).
Interestingly, the gating periods in which corals are sensitive to the moonlight signal were different between the Acropora and merulinid corals (Figure 3). The gating period for the Acropora corals was at approximately midnight (0000-0100 h [ZT = 19-20]), whereas that for the merulinid corals was at approximately sunset (1700-1900 h [ZT = 12-15]). Moreover, the difference in the cumulative period of environmental signals (n1) was approximately 2 weeks for the Acropora corals, which was approximately five times longer than that in the merulinid corals (Table 1). Plots of the log-likelihood for various values of
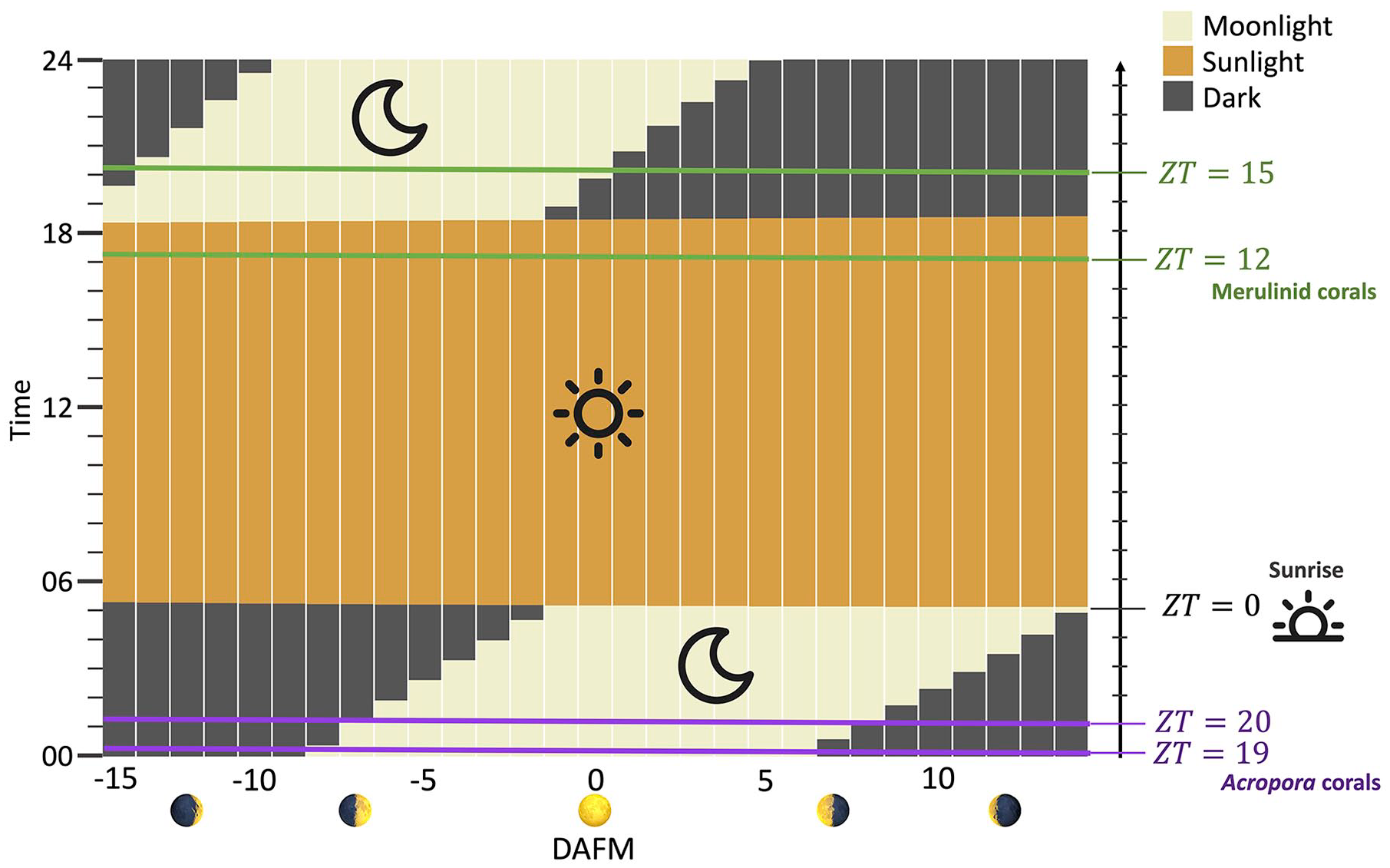
The gating periods for the Acropora and merulinid corals sensing the moonlight signal. The moonlight and sunlight times are based on the data from Dabaisha in May 2019. The horizontal axis indicates days before (−) or after (+) the full moon (= 0). The right vertical axis indicates the zeitgeber time ZT (
We next tested whether the best model estimated from the spawning data for the merulinid corals can explain the spawning patterns of D. speciosa under the light manipulation experiments in a previous study (Lin et al., 2021). Our model successfully predicted the spawning dates that were observed in both the field and laboratory conditions (Figure 4).
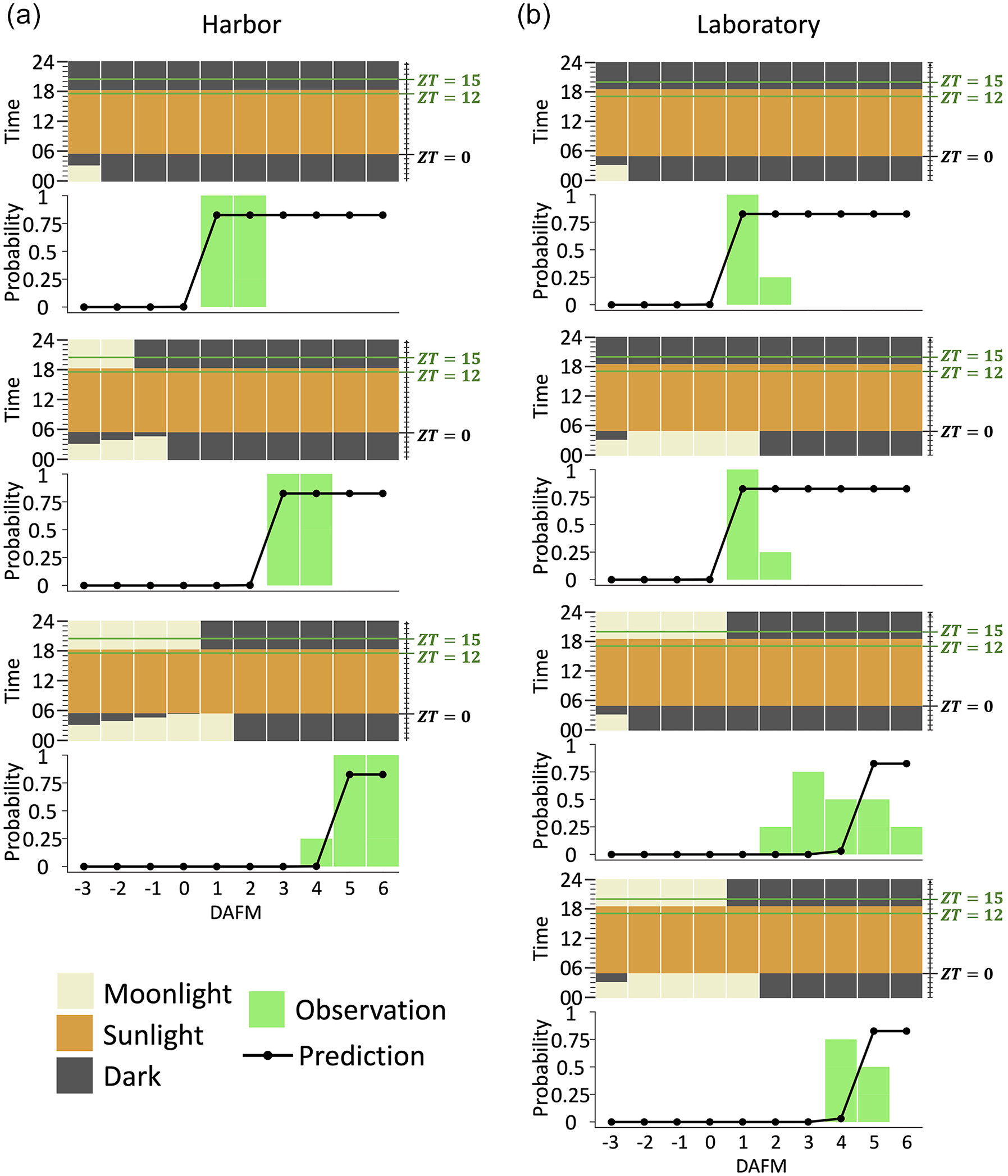
Model predictions for spawning days of Dipsastraea speciosa in the moonlight manipulation experiments. Results of model prediction for spawning days of D. speciosa under different moonlight conditions in the seven datasets from the three harbor (a) and four laboratory (b) experiments (Lin et al., 2021). Natural moonlight was used in the field, and dim white LED light (0.3 lux) was used in the laboratory. The horizontal axis indicates days before (–) or after (+) the full moon (= 0). The lower vertical axis indicates spawning probability. The prediction of spawning probability is indicated by line with dots, and the observation of spawning rate in 4 replicates is indicated by bars.
Discussion
Based on the external coincidence model, we developed the first predictive models of coral spawning, integrating the two key environmental factors of coral spawning, moonlight and seawater temperature (Lin and Nozawa, 2017). By fitting to the 10-year record of coral spawning, our models successfully reproduced previously suggested effects of moonlight and seawater temperature on coral spawning days and newly proposed two testable hypotheses to explain the different spawning patterns between the Acropora and merulinid corals.
Our models indicated the different moonlight effects on spawning between the Acropora and merulinid corals. In model class I, we found that the regression coefficient of the moonlight effect was positive for the Acropora corals but was negative for the merulinid corals (Table 1; Figure 3). This result is consistent with previous empirical findings that spawning of Acropora corals was activated by the presence of moonlight, whereas it was suppressed in merulinid corals (Lin et al., 2021).
Our models also indicated the different sensitivities to seawater temperature in spawning between the Acropora and merulinid corals. In the merulinid corals, model class I, which only considered the moonlight effect, was selected as the best model, whereas in the Acropora corals, model class II, which considered both the moonlight and temperature effects, was selected as the best model (Table 1). Previous studies indicated that higher seawater temperatures advance the spawning date in Acropora corals (Fujiwara et al., 2015; Nozawa, 2012; Paxton et al., 2016; Sakai et al., 2020; Shimoike, 1999). Lin and Nozawa (2017) observed high annual variation in spawning days for Acropora corals in contrast to the constant annual spawning days for merulinid corals, irrespective of the annual variation in seawater temperature. They hypothesized that the different spawning patterns could be caused by different sensitivities in gamete maturation between the two coral groups.
Our models provided two testable hypotheses that may explain the different spawning patterns in the Acropora and merulinid corals. The first hypothesis is that the Acropora corals may have an earlier onset and longer period of preparation for spawning than the merulinid corals. This was suggested by the longer cumulative period for environmental signals for spawning (n1) in the Acropora corals than in the merulinid corals (Table 1). The n1 value of 14 to 16 days in the best model for the Acropora corals indicated that the preparation of spawning starts around the first quarter moon and takes approximately 2 weeks (Figure 3). In contrast, the n1 value of 3 days in the best model for the merulinid corals suggests that the preparation of spawning starts after the full moon in the merulinid corals. Given that spawning of the Acropora corals sometimes occurred near the full moon (Figure 2a), it is reasonable that spawning preparation starts earlier in the Acropora corals than in the merulinid corals. The longer period for spawning preparation in the Acropora corals may result in increased sensitivity to temperature and variability in spawning time, in contrast to the short preparation period for spawning in the merulinid corals.
The second testable hypothesis from our models is the different time windows of the moonlight signal for spawning between the Acropora and merulinid corals. Our results indicated that the gating period during which corals are sensitive to moonlight signals is approximately midnight (0000-0100 h [ZT = 19-20]) for the moonlight-sensing Acropora corals, whereas in the darkness-sensing merulinid corals, the gating period was predicted to be approximately sunset (1700-1900 h [ZT = 12-15]). Although the prediction for the Acropora corals needs to be empirically examined in future studies, our prediction for the merulinid corals was consistent with the empirical finding that the merulinid coral D. speciosa responds to the darkness between sunset and moon rise after a full moon (Lin et al., 2021). The role of the circadian clock and its entrainment by environmental signals will need to be investigated to examine the mechanism underlying the possible difference in the gating periods. Although CRY1, CRY2, CLOCK, and TIMELESS genes have been reported to be affected by lunar phases (Brady et al., 2016; Kaniewska et al., 2015; Levy et al., 2007), these genes respond differently to changes in lunar phases, and it is still not clear how lunar phases influence the circadian oscillator that regulates spawning time. With new knowledge about clock genes in corals, the model presented here will be improved to consider the regulation of the circadian clock by lunar phases.
Our model suggested that the different responses to the two environmental cues (moonlight and temperature) cause the different spawning patterns between Acropora and merulinid corals. The different responses could be attributable to differences in the habitat depth range of the coral species. The Acropora corals used in the present study inhabit shallow reefs (~3-5 m depth), whereas the merulinid corals mainly inhabit deeper (~10-15 m depth). In shallow waters, moonlight intensity is high and temperature is variable. The high intensity of the moonlight signal in the shallow waters might allow the Acropora corals to utilize more detailed moonlight information, whereas the presence or absence of moonlight would be more reliable information for the merulinid corals in deep waters. The variable temperature in the shallow waters might influence the final gametogenesis process more in the Acropora corals, which determines the spawning timing (Lai et al., 2022). It will be an interesting research topic to investigate the co-evolutionary history of habitat preference and spawning behavior based on the phylogenetic relationship of diverse coral species.
We summarize the external coincidence model for the lunar cycle in Figure 5 to illustrate how moonlight and seawater temperature are integrated to synchronize spawning timing. First, the seasonal increase in seawater temperature in spring induces gametes maturation determining spawning months (Keith et al., 2016). Once gametes have matured, corals become sensitive to moonlight signals to synchronize their spawning days. The model considers that the circadian oscillator determines the sensitivity window to moonlight signals over the course of the subjective time of day, and the presence or absence of moonlight in the light sensitivity window act as a cue that initiates the final stage of reproduction, that is, germinal vesicle migration and breakdown (Suwa and Nakamura, 2018; Lai et al., 2022) and gamete bundle formation (Padilla-Gamiño et al., 2011). These could be tested via experimental studies in the future.
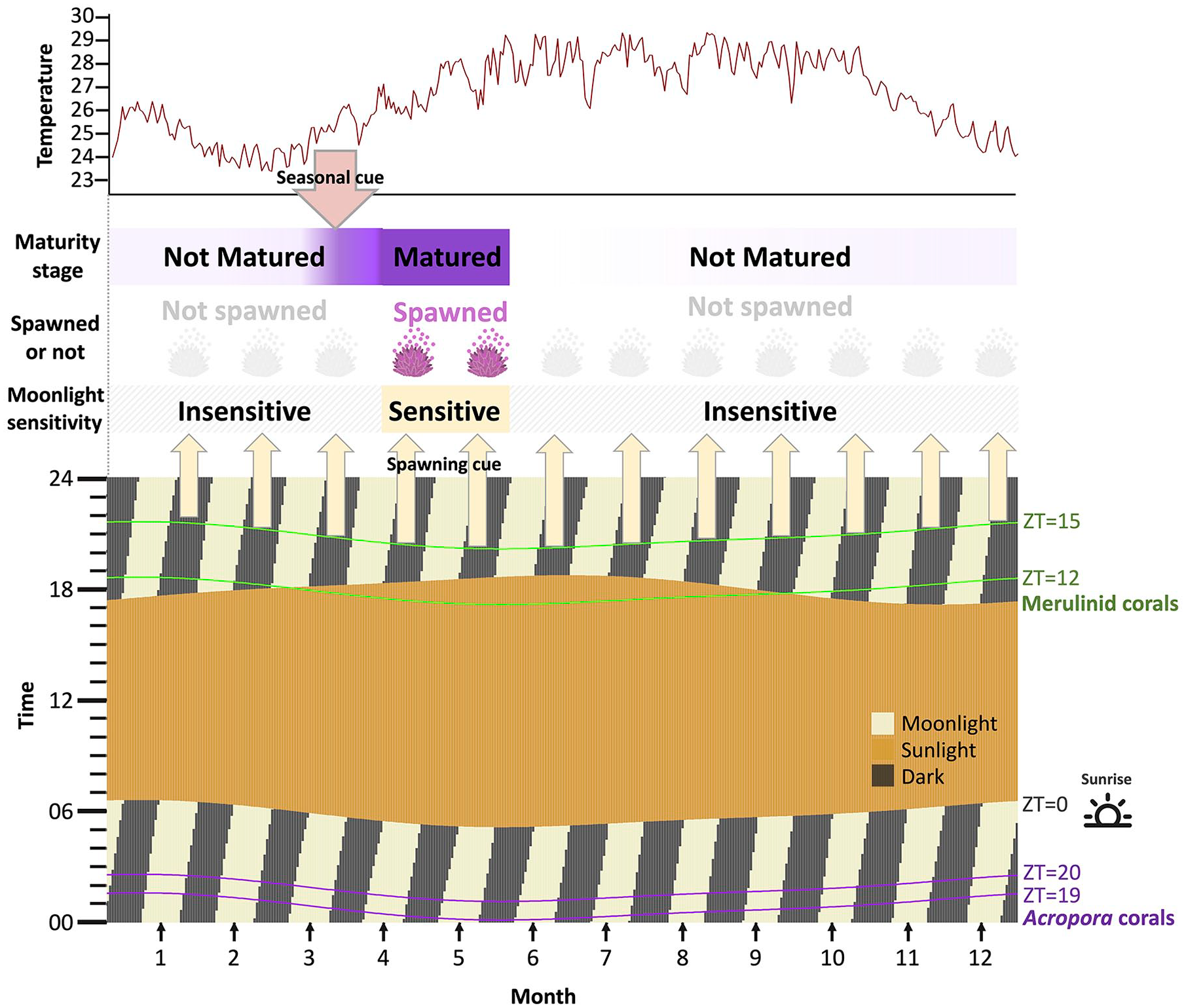
Schematic relationship of seawater temperature and moonlight that regulate the timing of synchronized spawning in Acropora and merulinid corals. The moonlight and sunlight times and temperature are based on the data from Gonggan in 2016, as an example. The horizontal axis indicates the month, and the arrows show the full moon of each month. In the vertical axis, time zones enclosed by the two lines indicate the moonlight-sensitive gating periods indicated by the best model for the merulinid corals (green) and the Acropora corals (purple). Once gametes have matured by rapidly increasing seawater temperature in April and May, corals become sensitive to moonlight signals and spawn by responding to the appropriate moonlight cue.
Coral spawning has been suggested to be deleteriously affected by artificial light at night (Ayalon et al., 2021; Kaniewska et al., 2015) and ocean warming due to climate change (Shlesinger and Loya, 2019). Therefore, understanding species-specific environmental factors for coral spawning is essential to predict the potential responses of coral reef ecosystems to anthropogenic disturbances and to propose effective conservation plans. Integration of modeling approaches and genomic resources, such as high-throughput transcriptome profiling in corals under different climatic conditions (Chiu et al., 2020; Kaniewska et al., 2015; Meyer et al., 2009; Oldach et al., 2017; Rosenberg et al., 2022), will be promising to identify proximate mechanisms of coral spawning at the genetic level and accelerate progress in understanding coral ecology and evolution in the future.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304221135916 – Supplemental material for An External Coincidence Model for the Lunar Cycle Reveals Circadian Phase-Dependent Moonlight Effects on Coral Spawning
Supplemental material, sj-pdf-1-jbr-10.1177_07487304221135916 for An External Coincidence Model for the Lunar Cycle Reveals Circadian Phase-Dependent Moonlight Effects on Coral Spawning by Hideyuki Komoto, Che-Hung Lin, Yoko Nozawa and Akiko Satake in Journal of Biological Rhythms
Footnotes
Acknowledgements
We thank S. Arakaki for his valuable comments on this study. The field survey was supported by the internal research grant of Academia Sinica to YN.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.