Abstract
Temperature influences animal physiology and behavior. Animals must set an appropriate body temperature to maintain homeostasis and maximize survival. Mammals set their body temperatures using metabolic and behavioral strategies. The daily fluctuation in body temperature is called the body temperature rhythm (BTR). For example, human body temperature increases during wakefulness and decreases during sleep. BTR is controlled by the circadian clock, is closely linked with metabolism and sleep, and entrains peripheral clocks located in the liver and lungs. However, the underlying mechanisms of BTR are largely unclear. In contrast to mammals, small ectotherms, such as Drosophila, control their body temperatures by choosing appropriate environmental temperatures. The preferred temperature of Drosophila increases during the day and decreases at night; this pattern is referred to as the temperature preference rhythm (TPR). As flies are small ectotherms, their body temperature is close to that of the surrounding environment. Thus, Drosophila TPR produces BTR, which exhibits a pattern similar to that of human BTR. In this review, we summarize the regulatory mechanisms of TPR, including recent studies that describe neuronal circuits relaying ambient temperature information to dorsal neurons (DNs). The neuropeptide diuretic hormone 31 (DH31) and its receptor (DH31R) regulate TPR, and a mammalian homolog of DH31R, the calcitonin receptor (CALCR), also plays an important role in mouse BTR regulation. In addition, both fly TPR and mammalian BTR are separately regulated from another clock output, locomotor activity rhythms. These findings suggest that the fundamental mechanisms of BTR regulation may be conserved between mammals and flies. Furthermore, we discuss the relationships between TPR and other physiological functions, such as sleep. The dissection of the regulatory mechanisms of Drosophila TPR could facilitate an understanding of mammalian BTR and the interaction between BTR and sleep regulation.
Keywords
Mammalian body temperatures fluctuate over 24 h. The body temperature rhythm (BTR) is associated with sleep and metabolism and plays an important role in the entrainment of the peripheral clock (Refinetti and Menaker, 1992; Gilbert et al., 2004; Krauchi, 2007a, 2007b; Buhr et al., 2010; Weinert, 2010). For instance, human body temperature is lowest during sleep, increases during the day, and reaches its highest level before sleep, suggesting an association between BTR and sleep (Figure 1a and 1c) (Duffy et al., 1998; Lack et al., 2008; Harding et al., 2019).
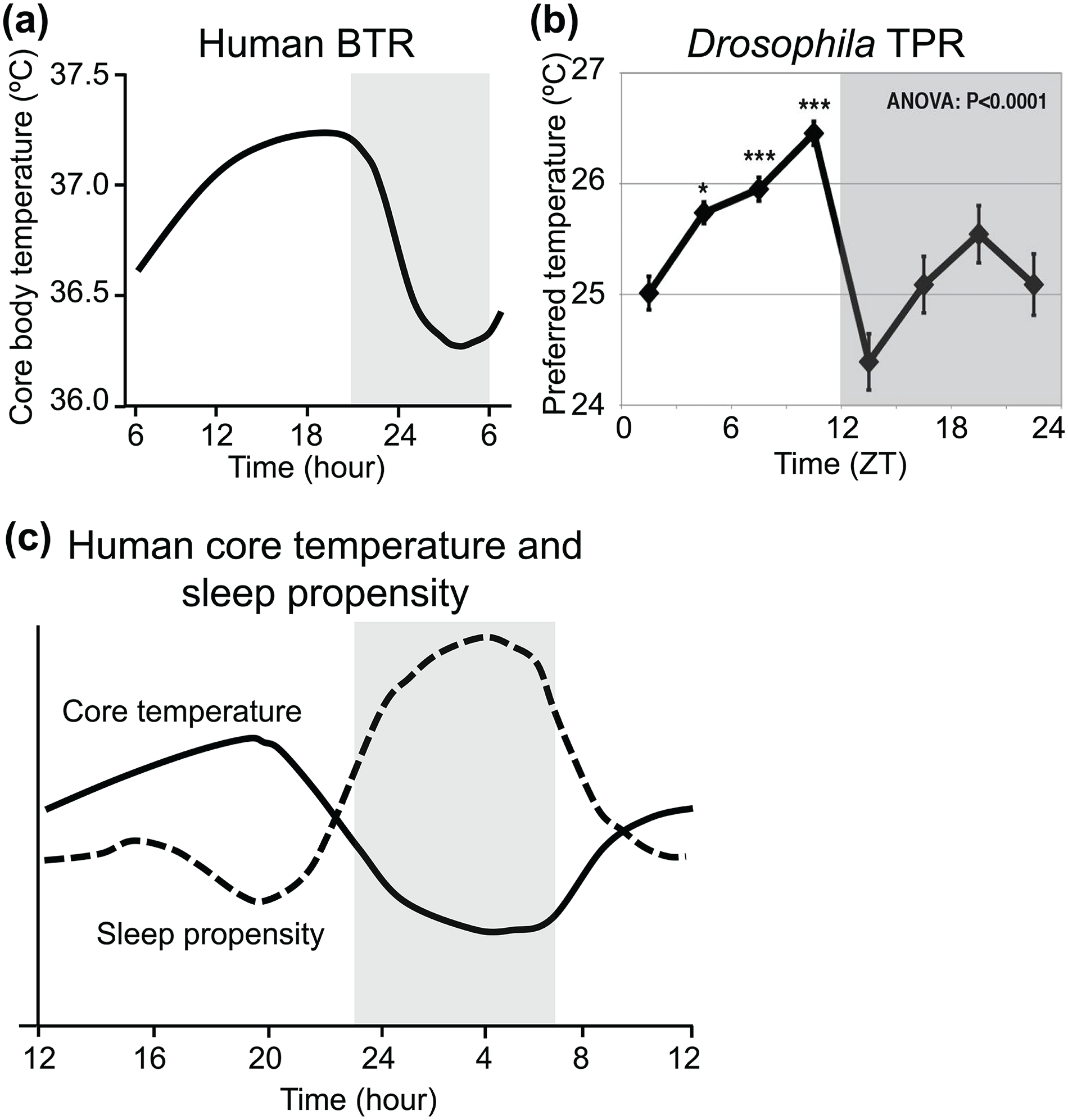
Human BTR and fly TPR. (a) Daily human core body temperature fluctuation (figures replotted using an example from Duffy et al., 1998, Figure 3). (b) Drosophila TPR (figures replotted using an example from Kaneko et al., 2012, Figure 1). (c) The inverse proportionality between daily fluctuations in core body temperature and sleep propensity in humans (figure replotted using an example from Lack et al., 2008, Figure 1). The black line and dashed line in the graph represent human core body temperature and sleep propensity, respectively. The shadowed areas in the graphs in (a) and (c) or in (b) indicate a period of sleep or nighttime, respectively. Abbreviations: BTR = body temperature rhythm; TPR = temperature preference rhythm; ZT = zeitgeber time.
The circadian clock controls daily changes in physiological activities. In nocturnal mammals, body temperatures are higher during the night and lower during the day in light-dark (LD) cycles and constant dark conditions (DD). Lesions of the suprachiasmatic nucleus (SCN) in the hypothalamus cause BTR disruption in DD (Zhang et al., 2004). These data suggest that the circadian clocks in the SCN, which is the central pacemaker, control BTR. Although BTR and locomotor activity are both robust circadian outputs, BTR and locomotor activity rhythms are separately controlled (Smith, 1969; Gander et al., 1986; Lavie, 2001; Saper et al., 2005; Cambras et al., 2007). For example, in humans, body temperature still fluctuates under conditions in which locomotor activity is restricted (Smith, 1969; Gander et al., 1986). BTR and locomotor activity rhythms can be experimentally dissociated, a phenomenon known as spontaneous internal desynchronization (Lavie, 2001; Cambras et al., 2007). BTR and locomotor activity rhythms are controlled by different output pathways originating from the SCN (Saper et al., 2005). Therefore, the mechanisms controlling BTR must differ from those controlling locomotor activity rhythms. However, the regulatory mechanisms of BTR are not fully understood.
Not only mammals but also ectotherms generate BTRs. Ectotherms control their body temperatures by behavioral strategies. For example, reptiles move to warmer and colder places throughout the day and use the ambient temperature to alter their body temperatures. A similar phenomenon can be seen in slugs, pill bugs, crayfish, and goldfish (Ellis et al., 2007). Fruit flies, Drosophila melanogaster, exhibit robust temperature preference behavior (Sayeed and Benzer, 1996; Hamada et al., 2008; Dillon et al., 2009) in which they select a comfortable temperature by avoiding warm and cold temperatures. Their preferred temperature (Tp) increases during the day, reaches its peak in the evening, and decreases during the night; this pattern is named the temperature preference rhythm (TPR) (Figure 1b) (Kaneko et al., 2012). Because Drosophila are small ectotherms, their body temperatures are almost equal to the surrounding temperatures. Therefore, Drosophila TPR generates BTR (Kaneko et al., 2012; Goda and Hamada, 2019).
Flies still exhibit TPR in DD, and flies with mutations of the circadian clock show abnormal TPR. These data suggest that TPR is a circadian clock output (Kaneko et al., 2012). The main clock neurons controlling TPR, dorsal neurons 2 (DN2s), differ from those controlling locomotor activity rhythms, indicating that TPR is regulated separately from locomotor activity rhythms (Kaneko et al., 2012). Furthermore, mutations in the neuropeptide receptor diuretic hormone 31 receptor (Dh31r) disrupt TPR during the day, showing that DH31R regulates TPR (Goda et al., 2018). Interestingly, a mammalian homolog of DH31R, the calcitonin receptor (CALCR), is also crucial for BTR in mice, suggesting that the CALCR is a common molecule in flies and mammals controlling fly TPR and mammalian BTR, respectively. Thus, although flies and mammals have different strategies of heat generation, their mechanisms of regulating TPR and BTR are equivalent. Drosophila represent a relatively simple genetic model that allows us to study the fundamental regulatory mechanisms of animal physiology and behavior (Allada et al., 2017; Dubowy and Sehgal, 2017; Shafer and Keene, 2021). Thus, studies of Drosophila TPR could shed new light on our understanding of BTR and the circadian system.
TPR in Drosophila
Drosophila exhibit TPR in a temperature gradient apparatus (Figure 2a) (Hamada et al., 2008; Kaneko et al., 2012; Goda et al., 2014). A temperature gradient from 18 to 32 °C is generated in the chambers between the metal plate and the plexiglass cover. Flies are placed in the chambers through the holes on the cover and are left for 30 min to select their Tp (Figure 2b). The flies prefer lower temperatures in the early morning. Then, their Tp increases gradually during the daytime, reaches a peak in the late afternoon, and drops dramatically at night to its lowest value in 12 h/12 h LD cycles (Figure 2c) (Kaneko et al., 2012). Tp in one time point is compared with the other three time points for daytime and seven time points for 24 h using analysis of variance (ANOVA) with a post hoc test. Wild-type flies show a statistically significant difference in Tp between the morning (zeitgeber time 1-3 [ZT 1-3]) and evening (ZT 10-12) for the daytime and between evening (ZT 10-12) and early night (ZT 13-15). In contrast to mammals, Drosophila rely on their surrounding temperatures to control their body temperatures. Due to their TPR, the body temperature of fruit flies also fluctuates over the course of a day, showing BTR. Importantly, human BTR and Drosophila TPR show similar patterns of temperature fluctuations, with higher body temperature during the day and lower body temperature during the night (Figure 1a and 1b).
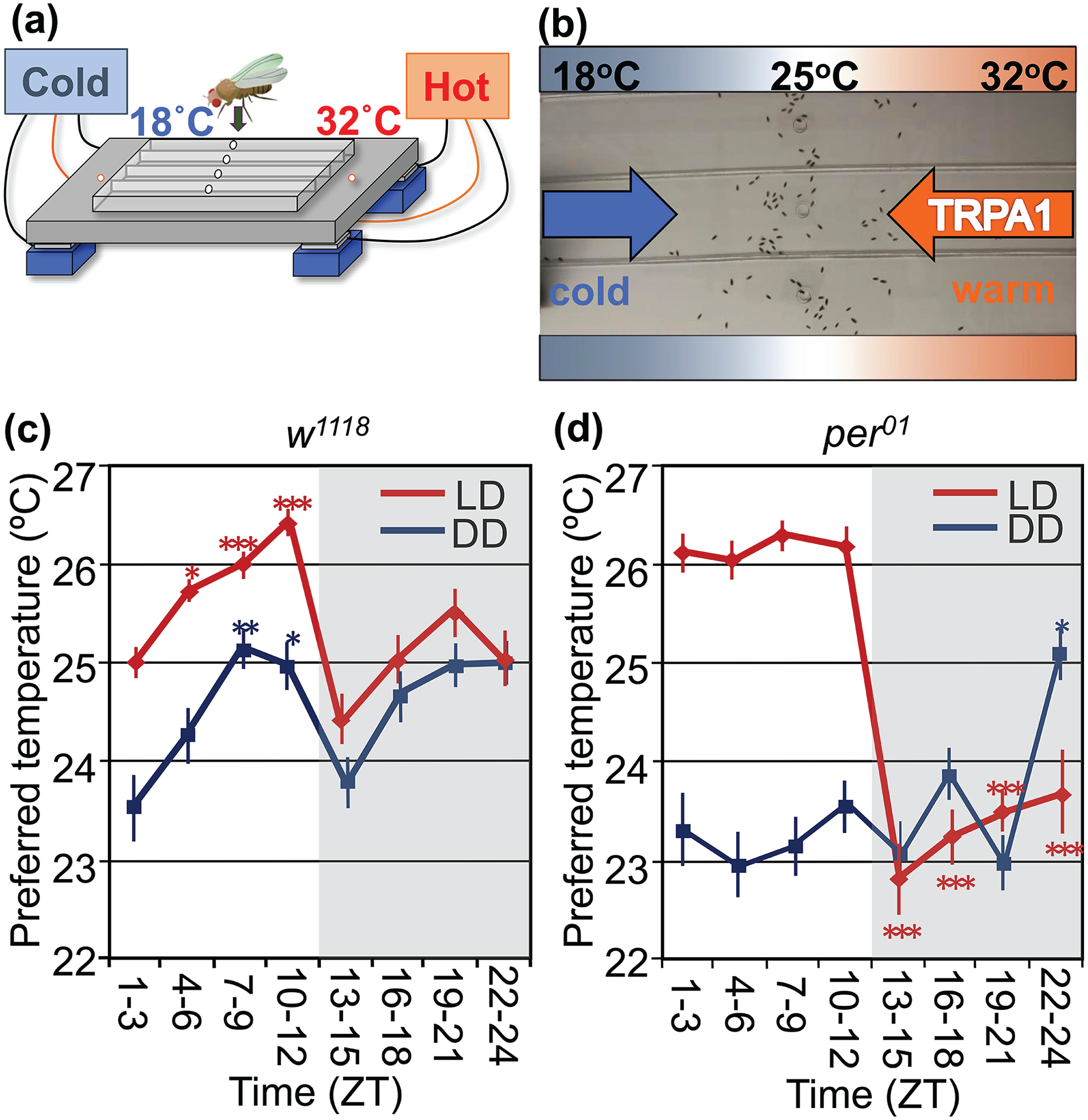
Drosophila TPR. (a) The apparatus for the Drosophila temperature preference behavior assay (modified from Goda and Hamada, 2019, Figure 2). Peltier devices are set on the bottom of the apparatus to heat and cool the metal plate. A plexiglass cover is placed on the plate, and air chambers are made between the plate and the cover with a temperature gradient from 18 to 32 °C. Flies enter the chambers through the holes on the cover. (b) Drosophila temperature preference behavior assay. Thirty minutes after the flies entered the chambers, they settled at the preferred temperature locations in the chambers. Flies avoid noxious cold and warm temperatures, and then they determine their Tp by the balance between cold and warm avoidance behaviors. TRPA1 is a warm-sensing molecule that is used for warm avoidance behavior (Hamada et al., 2008). (c, d) TPR in w1118 (c) and per01 (d) flies (figure replotted using an example from Kaneko et al., 2012, Figure 2). The red and blue lines show TPR under LD and DD, respectively. The temperature preference assays were not performed continuously for 24 h; instead, they were independently performed for 30 min at different times of the day. The flies used for the behavioral assay were never reused. More than five trials were performed in each time interval, and the preferred Tp and SEM were calculated (Goda et al., 2014). The shadowed areas in the graphs represent nighttime. Abbreviations: TPR = temperature preference rhythm; TRPA1 = transient receptor potential cation channel A1; LD = light-dark; DD = dark-dark. Color version of the figure is available online.
Wild-type flies exhibit a statistically significant increase and decrease in Tp during the day and at night, respectively, under both LD and DD (Kaneko et al., 2012) (Figures 1b and 2c). This suggests that TPR is not a mere response to light or dark but is regulated by the circadian clock, as is the mammalian BTR. Flies with null mutants of the clock genes, period (per) and timeless (tim), which are denoted as per01 and tim01 flies, respectively, show no increase in Tp during the day under both LD and DD (Figure 2d) and no Tp drop at night under DD. These data also support that daytime TPR is a clock output. Notably, clock mutant flies respond to acute lights-on and lights-off, with light causing a higher Tp. They exhibit a square-shaped TPR, which is a masking effect (Figure 2d). Therefore, the Tp drop at night is controlled by light and circadian clocks (Kaneko et al., 2012).
Approximately 150 neurons in the fly brain are considered circadian pacemakers for locomotor activity rhythms (Kaneko and Hall, 2000; Helfrich-Förster, 2003). There are nine different clusters that vary in their cell sizes and locations in the brain: four clusters are located in the dorsal area (anterior dorsal neuron 1 [DN1a], posterior dorsal neuron 1 [DN1p], dorsal neuron 2 [DN2], and dorsal neuron 3 [DN3]) and five clusters are located in the lateral area (small ventrolateral neuron (sLNv), large ventrolateral neuron (lLNv), fifth small ventrolateral neuron (5th-sLNv), dorsolateral neuron (LNd), and lateral posterior neuron (LPN)) (Figure 3a). DNs play critical roles in TPR (Kaneko et al., 2012; Chen et al., 2022). Furthermore, recent studies have shown that environmental cold- and warm-temperature inputs are conveyed into the different clusters of DNs (Yadlapalli et al., 2018; Alpert et al., 2020; Marin et al., 2020; Jin et al., 2021). These findings imply roles of DNs in the regulation of temperature-related behaviors.
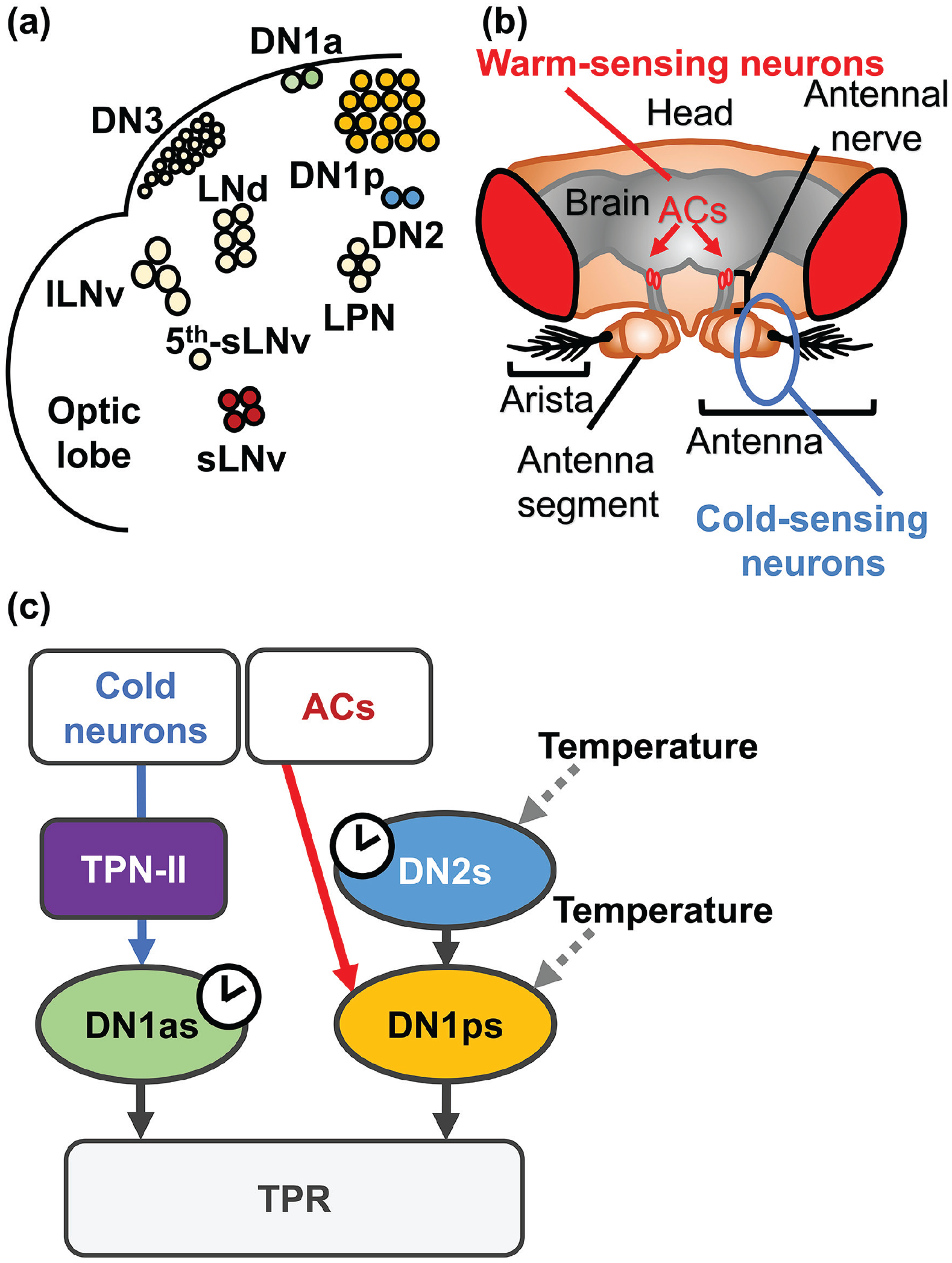
Core clock cells receive temperature inputs. (a) The core clock cells in the fly brain hemisphere (b) The warm- and cold-sensing neurons responsible for temperature preference behavior in the brain. ACs are warm-sensing neurons located at the antennal nerve between the antenna and the brain, and cold-sensing neurons are located in the antenna (modified from Umezaki 2019, Biomedical gerontology). (c) A schematic diagram of the current model for thermosensation and TPR regulation in Drosophila. In this model, the DN2-DN1p microcircuit and DN1as are responsible for TPR, and the DN2-DN1p microcircuit also regulates the Tp setpoint. In DN2-DN1p microcircuits, DN1ps receive clock information from DN2s and warm-temperature information from ACs (red arrowhead). DN1ps also receive warm- and cold-temperature information from peripheral thermosensors. In addition, cold-temperature information from the peripheral thermosensors is transferred to DN1as through TPNIIs (blue arrowhead). Warm and cold stimuli change the neuronal activities in DN1ps and DN2s (gray dotted arrowheads). The red and blue arrows represent warm- and cold-temperature information pathways, respectively. Abbreviations: AC = anterior cell; TPR = temperature preference rhythm; DN1a = anterior dorsal neuron 1; DN1p = posterior dorsal neuron 1; DN2 = dorsal neuron 2; DN3 = dorsal neuron 3; sLNv = small ventrolateral neuron; lLNv = large ventrolateral neuron; 5th-sLNv = fifth small ventrolateral neuron; LNd = dorsolateral neuron; LPN = lateral posterior neuron; TPN = thermosensory projection neuron; TPNII = type II TPN. Color version of the figure is available online.
The subsequent sections describe the roles of DNs and the thermoregulatory networks of TPR in each of the following three zones: (1) the time-dependent Tp increase during the day (ZT 1-12) (daytime increase in Tp) (Kaneko et al., 2012; Goda et al., 2018), (2) the Tp decrease at the transition from day to night (ZT 9-12 to 13-15) (night-onset TPR) (Goda et al., 2016), and (3) the Tp right before the morning starts (ZT 22-24) (predawn Tp) (Tang et al., 2017) (Figure 4d). The 24-h TPR is separated into three time zones, daytime (ZT 1-12), night-onset (ZT 9-12 to 13-15), and predawn (ZT 22-24), from the perspective of the regulatory machinery (Goda and Hamada, 2019).
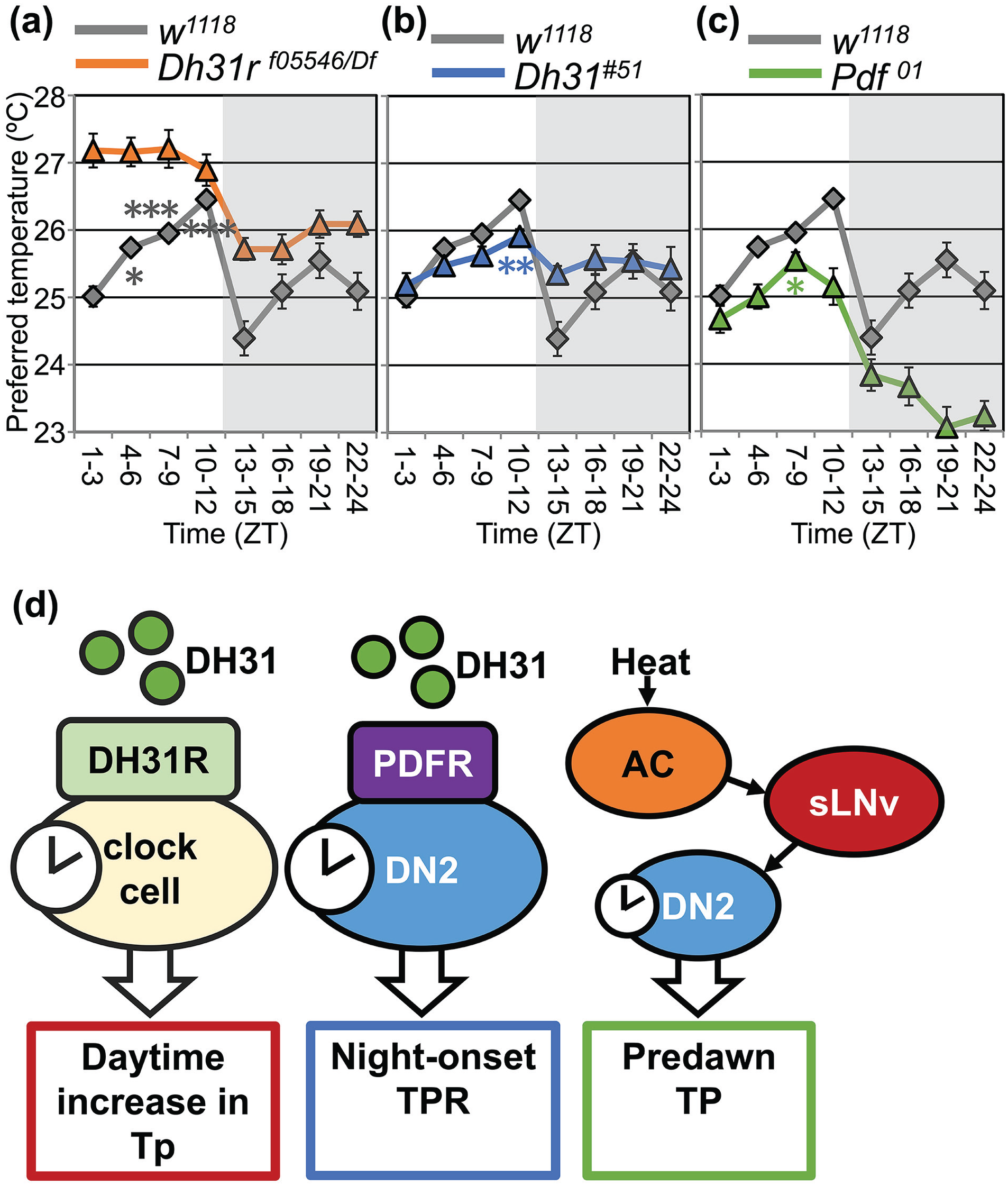
TPR phenotypes of Dh31r, Dh31, and Pdf mutant flies and models for molecular mechanisms of TPR regulation. TPR in Dh31r mutant (a: orange line), Dh31 mutant (b: blue line), and Pdf mutant (c: green line) flies compared with w1118 flies (a-c: gray line) (replotted using an example from Goda et al., 2018, Figure 1 (a) and Goda et al., 2016, Figure 3 (b, c)). The shadowed areas in the graphs represent nighttime. *p < 0.05. **p < 0.01. ***p < 0.001 (compared with ZT 1-3, only daytime)(d) Schematic diagrams of the daytime (ZT 0-12), night-onset (ZT9-12 to ZT13-15), and predawn Tp (ZT 21-24) regulations (modified from Goda and Hamada, 2019, Figure 7). Abbreviations: TPR = temperature preference rhythm; Tp = preferred temperature; ZT = zeitgeber time; DH31R = diuretic hormone 31 receptor; PDF = pigment dispersing factor. Color version of the figure is available online.
The Regulatory Mechanisms of the Daytime Increase in Tp
DN2: The Main Oscillator for the Daytime Increase in Tp
Flies exhibit daytime increasing Tp (Kaneko et al., 2012). DN2s are composed of two clock cells per brain hemisphere and are the main clock for the daytime increase in Tp (Kaneko et al., 2012; Hermann-Luibl and Helfrich-Forster, 2015). The Tp increase during the day is lost in per01 mutant flies and is restored by per expression in DN2s (Kaneko et al., 2012), suggesting that PER in DN2s is sufficient for the daytime increasing Tp. DN2-specific tim depletion using CRISPR-mediated gene disruption led to no Tp increase during the day (Chen et al., 2022). These data indicate that the circadian clocks in DN2s are required and sufficient for the regulation of daytime increasing Tp. Thus, DN2s are the main oscillators for the daytime increase in Tp. Given that DN2s are not involved in regulating locomotor activity rhythms, the data suggest that the TPR and locomotor activity rhythms are separately regulated by the circadian clock circuits (Kaneko et al., 2012; Goda and Hamada, 2019; Chen et al., 2022).
Interestingly, flies still maintain TPR in constant light conditions (LL) for 4 and 8 days (Kaneko et al., 2012). Under these conditions, locomotor activity rhythms are arrhythmic because molecular clock oscillations are dampened (Konopka et al., 1989; Qiu and Hardin, 1996). This is additional evidence that TPR is controlled separately from locomotor activity rhythms. Although the circadian molecular clock oscillations in DN2s were still maintained in larvae under LL (Klarsfeld et al., 2011), their status in adult DN2s is unclear. It will be interesting to elaborate the molecular mechanisms of TPR in LL.
DN1p Functions Downstream From DN2 to Regulate the Daytime Increase in Tp
DN1ps consist of ~15 clock cells per brain hemisphere (Hermann-Luibl and Helfrich-Forster, 2015). Although clock disruption in DN1ps does not prevent the normal daytime increase in Tp, DN1p silencing eliminates the daytime increase in Tp. These data suggest that the neural activities but not the circadian clocks in DN1ps are essential for the daytime increase in Tp (Chen et al., 2022). Therefore, the data suggest that DN1ps likely regulate the daytime increase in Tp downstream from other clock cells. Interestingly, DN2s contact DN1ps, and the number of these contacts fluctuates over the course of a day, with a minimum in the early morning and a peak in the early night (Chen et al., 2022). Calcium imaging data using GCaMP indicate that DN2s can activate DN1ps but not vice versa. DN2 inhibition causes a lower Tp, which is a similar phenotype to DN1p inhibition. These data suggest that the microcircuits from DN2s to DN1ps (DN2-DN1ps microcircuits) regulate the daytime increase in Tp (Figure 3c). Nonetheless, these findings do not exclude the possibility of microcircuits from DN1ps-DN2s, which may also play an important role in TPR (Reinhard et al., 2022b). Taken together, these findings show that DN1ps regulate TPR downstream of DN2s and that the DN2-DN1ps microcircuits play an essential role in regulating the daytime increase in Tp.
DN1a: Another Oscillator for the Daytime Increase in Tp
Another possible oscillator for the daytime increase in Tp was recently identified (Chen et al., 2022). DN1as are composed of two clock neurons per brain hemisphere and are located more anteriorly than DN1ps (Hermann-Luibl and Helfrich-Forster, 2015). DN1as-specific tim depletion using CRISPR-mediated clock gene disruption led to no Tp increase during the day (Chen et al., 2022), suggesting that the clocks in DN1as are required for the daytime increase in Tp. Therefore, DN1a is another possible main oscillator for the daytime increase in Tp. Notably, the roles of DN2s and DN1as in TPR are likely different since flies exhibit different phenotypes in the neuronal activity manipulation of DN2s or DN1as (Chen et al., 2022). DN2 silencing does not disrupt a robust daytime increase in Tp but results in overall low Tp during the day. This suggests that the temperature values the flies choose (Tp setpoint) are separately regulated from the time-dependent increase in Tp during the day. On the contrary, DN1a silencing does not affect the Tp setpoint, suggesting a functional difference between DN2s and DN1as in the daytime increase in Tp. Further elucidation is required to fully understand the regulatory mechanisms of DN2s and DN1as in the daytime increase in Tp.
The Neuropeptide Signals for Regulating the Daytime Increase in Tp
Neuropeptide signals play important roles in the regulation of animal behaviors. Pigment-dispersing factor (PDF) and its receptor (PDFR) are a well-known neuropeptide and corresponding neuropeptide receptor for Drosophila circadian rhythms, and they are essential for locomotor activity rhythms (Taghert and Nitabach, 2012). The clock cells, sLNvs, drive robust locomotor activity rhythms by releasing PDF, which activates PDFR in a subset of clock cells (Helfrich-Forster, 1995; Grima et al., 2004; Stoleru et al., 2004; Lear et al., 2009; Im and Taghert, 2010; Zhang et al., 2010; Yao and Shafer, 2014; Liang et al., 2016). PDF-PDFR signaling plays a critical role in the synchronization of molecular clocks among the core clock cells in the brain under DD conditions (Renn et al., 1999; Lin et al., 2004; Hyun et al., 2005; Lear et al., 2005; Mertens et al., 2005). However, Pdf and Pdfr mutant flies still exhibit a normal Tp increase during the day under LD (Goda et al., 2016; Tang et al., 2017), suggesting that PDF and PDFR are not the main regulators of the daytime increase in Tp.
Diuretic hormone 31 receptor (DH31R) is a member of the class B family of G-protein coupled receptors (GPCRs) to which PDFR also belongs. Immunostaining with an anti-DH31R antibody revealed that DH31R is expressed in a subset of DN1s, DN3s, and lLNvs, as well as several nonclock cells in the dorsal area of the brain (Goda et al., 2018). Dh31r loss-of-function mutant flies exhibit a constant Tp at approximately 27 °C and lose the increase in Tp during the day under LD and DD (Figure 4a). These data suggest that DH31R regulates the daytime increase in Tp under the control of endogenous circadian clocks. Dh31r expression in all clock cells restores the increase in Tp during the day, suggesting that DH31R in clock cells regulates the daytime increase in Tp. Although DH31R is expressed in clock neurons, the molecular rhythms in clock cells are still normal in Dh31r mutant flies. These data suggest that DH31R function is downstream from circadian clocks.
It is important to note that Dh31r mutants show normal locomotor activity rhythms with minor abnormal phenotypes, such as less locomotor activity at night and dampened morning anticipation (Goda et al., 2018). This finding is additional evidence that TPR and locomotor activity rhythms are separately regulated since, as we mentioned in the “DN2: The Main Oscillator for the Daytime Increase in Tp” section, DN2s are the main clock for TPR but are not involved in locomotor activity rhythms (Kaneko et al., 2012).
Although it was easily predicted that the ligand of DH31R, DH31, was also required for the daytime increase in Tp, unexpectedly, Dh31 mutant flies still exhibited a normal Tp increase during the day (Goda et al., 2018). In addition, as we mentioned above, Pdf mutant flies exhibited a normal daytime increase in Tp (Goda et al., 2016). However, the Dh31 and Pdf double mutant showed no Tp increase during the day under LD and DD, which is similar to the phenotype of Dh31r mutants (Goda et al., 2018). Furthermore, a rescue experiment was performed with the membrane-tethered neuropeptides DH31 and PDF (t-DH31 and t-PDF, respectively), which combine with membrane-localized signal peptides and do not disperse but are anchored on peptide-releasing cells (Choi et al., 2009). The expression of either tethered-Dh31 (t-DH31) or Pdf (t-Pdf) in all clock cells rescued the abnormal phenotype in Dh31 and Pdf double mutant flies (Goda et al., 2018). The data suggest that both DH31 and PDF are required for the increase in Tp during the day and that DH31 and PDF compensate for each other to control the daytime increase in Tp.
Neuronal Circuits From Thermosensory Neurons to Clock Cells
Several recent studies have identified multiple temperature-sensing pathways targeting DNs (Lamaze et al., 2017; Yadlapalli et al., 2018; Alpert et al., 2020; Marin et al., 2020; Jin et al., 2021), which could play significant roles in TPR regulation (Figure 3c). Temporal heating and cooling of adult flies induce the inhibition and excitation of both DN1ps and DN2s, suggesting that DN1ps and DN2s receive environmental temperature inputs (Yadlapalli et al., 2018). DN1ps receive inputs from anterior cells (ACs) (Lamaze et al., 2017; Jin et al., 2021). ACs are warm-sensing neurons located at the antennal nerve between the antenna and the brain and are the key neurons for temperature preference behavior (Figure 3b) (Hamada et al., 2008; Tang et al., 2013). ACs express transient receptor potential cation channel A1 (TRPA1), which is activated by innocuous warmth above ~25 to 27 °C (Hamada et al., 2008). TrpA1 mutant flies or flies with TrpA1 knockdown in ACs prefer a warmer temperature, suggesting that the flies avoid innocuous warm temperatures due to the presence of TRPA1-expressing ACs. ACs release acetylcholine and activate a subset of DN1ps (Jin et al., 2021). Thus, the microcircuits from ACs to DN1ps (AC-DN1p microcircuit) may play an important role in regulating TPR. In addition, the two microcircuits from ACs to sLNvs and from sLNvs to DN2s were determined, which suggests that ACs are associated with DN2s through sLNvs (Tang et al., 2017). These neurocircuits are responsible for the regulation of the predawn Tp (see the section “Regulation of the Predawn Tp”).
Furthermore, the antenna contains thermoreceptors for hot or cold stimuli (Figure 3b) (Gallio et al., 2011; Barbagallo and Garrity, 2015). These thermosensory neurons project to the posterior antennal lobe (PAL), where thermosensory projection neurons (TPNs) are located (Frank et al., 2015; Liu et al., 2015). TPNs innervate the brain regions in the lateral horn and posterior-lateral protocerebrum (PLP) area (Frank et al., 2015; Liu et al., 2015). Recently, subsets of type II TPNs (TPNIIs) were shown to project to DN1as (Alpert et al., 2020), but whether these TPNIIs are involved in the TPR is unclear. Chordotonal organs are another peripheral tissue responsible for temperature entrainment (Glaser and Stanewsky, 2005; Sehadova et al., 2009; Chen et al., 2015). In addition, the connectomes of temperature processing pathways in the brain were revealed (Marin et al., 2020). Thus, further investigations are necessary to fully understand the roles of the thermosensing pathways in TPR regulation.
The Regulation of the Night-Onset TPR
Human body temperature falls before sleep (Refinetti and Menaker, 1992; Krauchi, 2002, 2007a; Lack et al., 2008; Buhr et al., 2010; Kräuchi and Deboer, 2011; Morf and Schibler, 2013). The disruption of BTR causes the suppression of sleep induction (Krauchi and Wirz-Justice, 2001), and the dissociation of sleep and BTR causes insomnia (Lack et al., 2008). Therefore, there is an obvious correlation between sleep induction and body cooling (Harding et al., 2019; Harding et al., 2020). However, the neural interplay between sleep onset and body cooling is largely unclear. In Drosophila, Tp is highest in the evening and dramatically decreases at the transition from day to night (ZT 9-12 to ZT 13-15) by approximately 2 °C (night-onset TPR) (Figure 1a), suggesting that fly body temperature is decreasing at this time point. Therefore, there might be a correlation between sleep induction and body cooling in Drosophila.
DH31 was identified as a key player in the night-onset TPR. Dh31 mutant flies show an abnormal night-onset TPR, as their Tp decreases by only 0.6 to 0.8 °C (Figure 4b) (Goda et al., 2016). This phenotype of Dh31 mutant flies is rescued by the overexpression of t-Dh31 specifically in DN2s but not in other clock cells, suggesting that DH31 acts on DN2s to regulate the decrease in Tp at night-onset. What receptor is involved in this night-onset regulation? DH31 is a ligand of DH31R and PDFR in vitro (Mertens et al., 2005). Whereas Dh31r mutant flies still show a normal night-onset TPR (Figure 4a), the Pdfr mutant flies exhibit an abnormal night-onset TPR similar to that observed in Dh31 mutant flies (Goda et al., 2016). These data suggest that DH31 regulates the decrease in Tp at night-onset via PDFR. In addition, the overexpression of Pdfr in DN2s restores a robust decrease in Tp at night-onset in Pdfr mutant flies, suggesting that PDFR in DN2s receives DH31 signals and regulates the decrease in Tp at night-onset. Notably, Pdf01 flies with a null mutant of PDF show a normal night-onset TPR (Figure 4c) (Goda et al., 2016; Tang et al., 2017), suggesting that PDF is dispensable for the night-onset TPR. Taken together, these results show that DH31 acts on PDFR in DN2s to regulate the decrease in Tp at night-onset.
Although DH31 regulates the night-onset TPR via PDFR in DN2s and the daytime increase in Tp via DH31R in clock cells, the neuronal circuits controlling these Tp changes through DH31 are unclear. One of the possible scenarios is DN1ps. DH31 is expressed in glutamatergic DN1 (gDN1) cells, which are spl-DN1-Gal4-positive cells (Guo et al., 2017; Reinhard et al., 2022b). Neuronal suppression of gDN1 cells results in severe disruption of the daytime increase in Tp and night-onset TPR (Chen et al., 2022). Thus, gDN1 might be the regulator of the daytime increase in Tp and night-onset TPR through the DH31 signal. Moreover, a recent study demonstrated that LPNs rhythmically express DH31 and play an important role in locomotor activity rhythms and sleep (Reinhard et al., 2022a). Whether LPNs are involved in temperature preference behavior is an interesting question.
Regulation of the Predawn Tp
In wild-type flies, Tp increases during the daytime and decreases at night. Before dawn (ZT 22-24), the flies choose the Tp that is almost the same as the one in the early morning (ZT 1-3) in LD cycles, which is termed the “predawn Tp” (Tang et al., 2017) (Figure 1b). Although Pdf01 flies show a normal daytime increase in Tp and night-onset TPR, they show an abnormal predawn Tp. In Pdf01 flies, Tp continues to decrease during the night, and as a result, the predawn Tp is much lower than that of wild-type flies (Figure 4c). PDF is expressed in LNvs, and the suppression of neural activity or circadian molecular clock disruption in LNvs causes a phenotype similar to that of Pdf01 flies (Tang et al., 2017). These results suggest that PDF-expressing neurons play an important role in regulating the predawn Tp. Furthermore, sLNvs are shown to contact and activate DN2s using GFP Reconstitution Across Synaptic Partners (GRASP) and GCaMP calcium imaging (Tang et al., 2017). The number of temporal contacts between sLNvs and DN2s fluctuates over the course of a day, with the trough at early night and a peak before dawn. These data suggest that the interaction between sLNvs and DN2s is maximized before dawn, which might contribute to the proper setting of the Tp before dawn.
The silencing of ACs also causes an abnormal predawn Tp, which is a phenotype similar to that of Pdf01 flies (Tang et al., 2017). GRASP and calcium imaging data suggest neural contacts between ACs and sLNvs, indicating microcircuits from ACs to sLNvs (AC-sLNv microcircuits). ACs are serotonergic (Shih and Chiang, 2011), and sLNvs express the serotonin receptor 5-hydroxytryptamine receptor 1B (5HT1B) (Yuan et al., 2005). 5HT1B knockdown in sLNvs led to an abnormal predawn Tp, suggesting that sLNvs might receive temperature information from ACs through serotonin signals and regulate the predawn Tp (Tang et al., 2017). Thus, two microcircuits, AC-sLNv and sLNv-DN2s, are likely responsible for the predawn Tp.
The Fundamental Mechanisms of BTR are Likely Conserved between Flies and Mammals
As we described above, Drosophila DH31R was identified as a critical regulator of the daytime increase in Tp (Goda et al., 2018). What are the regulatory mechanisms of BTR in mammals? There are two mammalian homologs of DH31R: CALCR and calcitonin receptor-like receptor (CALCRL) (Kunst et al., 2015). Because CALCR but not CALCRL is highly expressed in the SCN, the BTR of Calcr knockout mice was examined (Nakamoto et al., 2000; Becskei et al., 2004; Doi et al., 2016; Goda et al., 2018). Mice are nocturnal animals, and their body temperatures are the lowest at midday and show bimodal peaks during the night, with a trough at midnight. Calcr knockout mice lack the trough in body temperature at midnight and exhibit higher body temperatures at night under both LD and DD conditions (Figure 5a) (Goda et al., 2018). These data suggest that CALCR regulates BTR in mice. Notably, the BTR phenotypes of Calcr knockout mice are similar to those of Dh31r mutant flies; both exhibit relatively flat and higher body temperatures than the controls during the active phase.
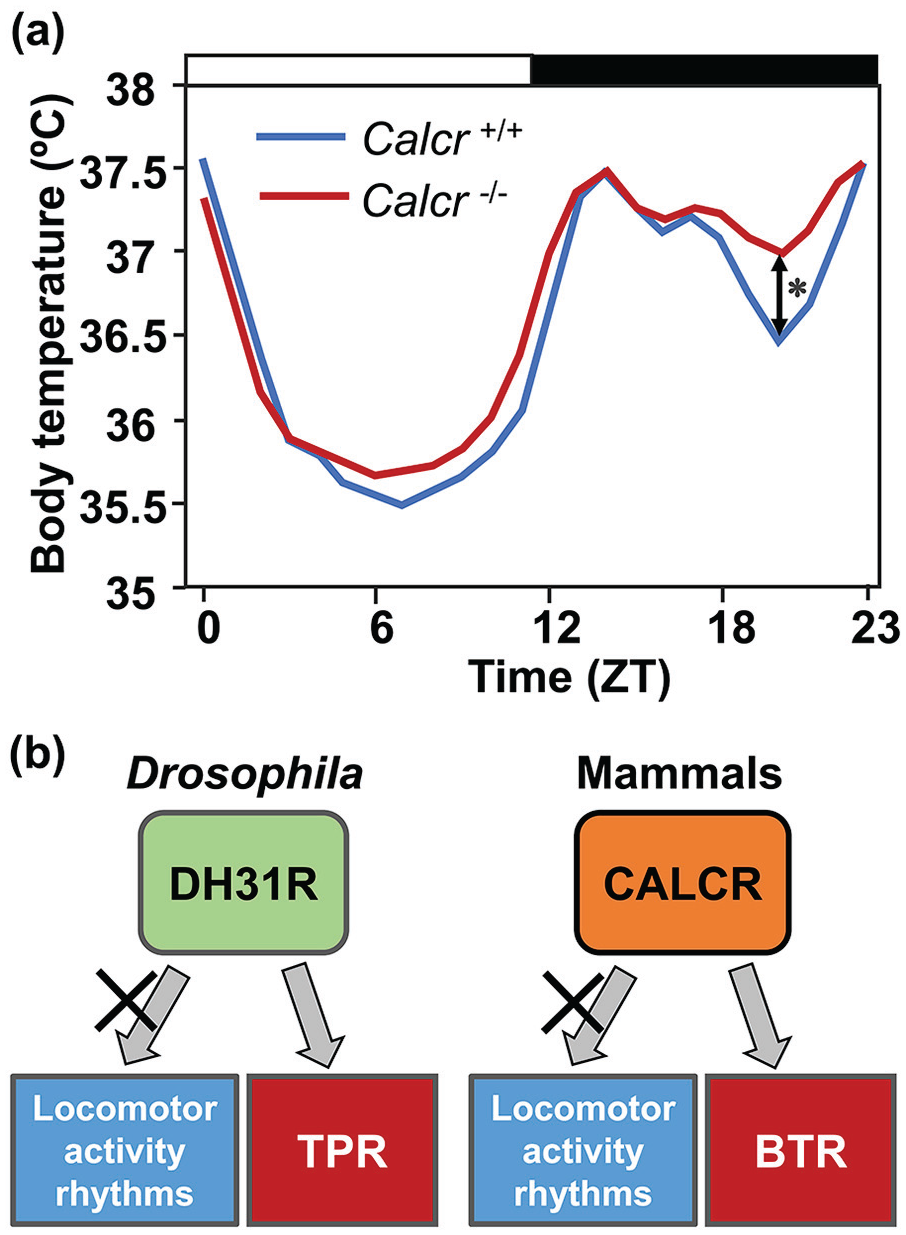
Mammal CALCR and Drosophila DH31R regulate BTR. (a) The BTR in LD cycles in control (blue line) and Calcr knockout (red line) mice (modified from Goda et al., 2018, Figure 6). The bars at the top of the graph represent light (white bar) and dark (black bar) cycles. The body temperatures in the middle of the night are significantly different between control and Calcr knockout mice. *p < 0.05 (comparison between Calcr knockout and control mice at ZT 20). (b) A schematic diagram showing the evolutionally conserved function of the calcitonin receptor for BTR in Drosophila and mammals. Calcitonin receptors in both Drosophila and mammals are responsible for BTR regulation but not for locomotor activity rhythms. Abbreviations: CALCR = calcitonin receptor; DH31R = diuretic hormone 31 receptor; BTR = body temperature rhythm; LD = light-dark; TPR = temperature preference rhythm. Color version of the figure is available online.
On the contrary, the locomotor activity rhythms in Calcr knockout mice are almost identical to those of wild-type mice (Goda et al., 2018). This suggests that mammalian BTR and locomotor activity rhythms are regulated by different pathways, similar to Drosophila TPR (Figure 5b). These findings on CALCRs in flies (DH31R) and mice (CALCR) are the first molecular evidence that BTR and locomotor activity rhythms are separately controlled. Thus, although flies and mammals employ different strategies to generate heat, the regulatory mechanisms of BTR may be conserved between flies and mammals (Goda et al., 2018; Goda and Hamada, 2019).
TPR Might be Associated with Sleep
Human body temperature starts to rise before awaking and falls before sleep (Refinetti and Menaker, 1992; Krauchi, 2002, 2007a; Lack et al., 2008; Buhr et al., 2010; Morf and Schibler, 2013). BTR disruption causes a suppression of sleep induction and is associated with insomnia (Lack et al., 2008). These data suggest that BTR is associated with sleep. Flies exhibit a similar correlation between BTR and sleep induction; they decrease their Tp dramatically before starting nighttime sleep (the night-onset TPR). Given that the neural activity of DN1ps is critical for both TPR and sleep, DN1ps might be important hubs between TPR and sleep (Guo et al., 2016; Dubowy and Sehgal, 2017; Lamaze and Stanewsky, 2019; Mazzotta et al., 2020; Shafer and Keene, 2021; Chen et al., 2022). DN1ps express DH31, which regulates the night-onset TPR (Goda et al., 2016, 2018) and arousal before dawn (Kunst et al., 2014; Goda et al., 2019; Goda and Hamada, 2019). Therefore, DH31 in DN1ps might function as a possible mediator of the link between the TPR and sleep.
As discussed previously, the pattern of Drosophila TPR is similar to that of human BTR (Figure 1a and 1b), and the fundamental regulatory mechanisms of BTR are likely shared between mammals and flies (Goda et al., 2018; Goda and Hamada, 2019). The mechanisms controlling sleep in flies are conserved in mammals (Allada et al., 2017; Ly et al., 2018). Thus, understanding the mechanistic interplay between TPR and sleep in Drosophila might facilitate the elucidation of sleep mechanisms in mammals.
Interestingly, the application of warm temperature to flies induces delays in the onset and offset of siesta. While either TrpA1 mutation or AC inhibition suppresses both delays, the inhibition of DN1ps delays only the onset and not the end of siesta (Lamaze et al., 2017). These data suggest that temperature affects sleep plasticity via distinct pathways at different times, supporting the model in which TPR is regulated by multiple networks working at different times of the day (Figure 4d).
Drosophila Body Temperature Regulation is Associated with Feeding Status
In mammals, body temperatures are strongly associated with metabolism (Cannon and Nedergaard, 2004; Brychta and Chen, 2017; Chechi et al., 2018). The body temperatures of mammals and birds change according to fasting/feeding conditions (Ben-Hamo et al., 2010; Refinetti, 2020). For instance, the basal body temperature of goats is approximately 39.0°C. Under starved conditions, goats exhibit gradual decreases in body temperature to approximately 38.0°C, which is called fasting (starvation)-induced hypothermia and dampens BTR. As soon as the goats are refed, their body temperatures and BTR recover to their original states (Piccione et al., 2002). Therefore, the feeding status of goats directly affects their internal metabolic states, which leads to body temperature changes.
What about Drosophila? Is their body temperature regulation also associated with their metabolic state? Starvation was demonstrated to result in a lower Tp in Drosophila (Umezaki et al., 2018). As a fly’s body temperature is almost equal to the ambient temperature, the lower Tp in the starved condition reduces Drosophila body temperature. Thus, starvation induces lower body temperatures in both mammals and flies (Piccione et al., 2002; Umezaki et al., 2018). Interestingly, as soon as the hungry flies are refed with fly food, they restore their normal Tp. This physiological phenomenon is also observed in mammals.
Ectotherms lower their metabolic rates at cold temperatures (Nespolo et al., 2011; Staples, 2016). Recent findings suggest that at cold temperatures, the metabolic rate and food intake of Drosophila are lower, and their lifespan is extended (Klepsatel et al., 2019). Furthermore, malnutrition or starvation causes a lower metabolic rate in Drosophila (Stahl et al., 2017; Fiorino et al., 2018). These data suggest that starved flies prefer lower temperatures to save their metabolism for survival. Interestingly, while starved flies prefer cold areas to save their energy, they also increase their locomotor activity until they die (Yang et al., 2015). Perhaps, under starved conditions, flies want to balance the cold-temperature preference for energy saving with foraging food for extended survival.
Although the association between metabolism and TPR is unclear, feeding timing is associated with circadian clocks (Xu et al., 2008; Ro et al., 2014; Panda, 2016), and feeding state strongly affects body temperatures in both flies and mammals (Zammit et al., 1992; Challet et al., 1996; Kaur et al., 2008). Therefore, TPR likely interacts with and influences metabolic regulation, and vice versa.
Drosophila have powerful genetic tools, a relatively simple nervous system and a long history of use in circadian research. Drosophila TPR is one of the robust outputs of the circadian clock. In contrast to the other clock outputs, locomotor activity rhythms, the research history of TPR was just started in 2009. Although it has been revealed that TPR is associated with and influenced by various physiological activities and environmental cues, the regulatory mechanisms of TPR are still largely unclear. Because of the functional parallels between BTR in mammals and TPR in flies, studying fly TPR could markedly contribute to unraveling the mechanisms underlying BTR. Thus, studies in Drosophila could contribute to a better understanding of the complex regulatory system of body temperature in mammals and provide a new breakthrough in circadian clock research.
Footnotes
Acknowledgements
This research was supported by NIH R01 grant GM107582 and R21 grant NS112890 to F.N.H. We thank Dr. Shyh-Chi Chen, Benjamin Falkenstein, and Madison Aikawa for comments on this article.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
