Abstract
Data from human and animal studies are highly suggestive of an influence of time of day of vaccine administration on host immune responses. In this population-based study, we aimed to investigate the effect of time of day of administration of a COVID-19 vector vaccine, ChAdOx1 nCoV-19 (AstraZeneca), on SARS-CoV-2 anti-spike S1 immunoglobulin (IgG) levels. Participants were 803 university employees who received their first vaccine dose in March 2021, had serology data at baseline and at 3 weeks, and were seronegative at baseline. Antibody levels were determined in binding antibody units (BAU/mL) using enzyme-linked immunosorbent assay (ELISA). Generalized additive models (GAM) and linear regression were used to evaluate the association of time of day of vaccination continuously and in hourly bins with antibody levels at 3 weeks. Participants had a mean age of 42 years (SD: 12; range: 21-74) and 60% were female. Time of day of vaccination was associated non-linearly (“reverse J-shape”) with antibody levels. Morning vaccination was associated with the highest (9:00-10:00 h: mean 292.1 BAU/mL; SD: 262.1), early afternoon vaccination with the lowest (12:00-13:00 h: mean 217.3 BAU/mL; SD: 153.6), and late afternoon vaccination with intermediate (14:00-15:00 h: mean 280.7 BAU/mL; SD: 262.4) antibody levels. Antibody levels induced by 12:00-13:00 h vaccination (but not other time intervals) were significantly lower compared to 9:00-10:00 h vaccination after adjusting for potential confounders (beta coefficient = −75.8, 95% confidence interval [CI] = −131.3, −20.4). Our findings show that time of day of vaccination against SARS-CoV-2 has an impact on the magnitude of IgG antibody levels at 3 weeks. Whether this difference persists after booster vaccine doses and whether it influences the level of protection against COVID-19 needs further evaluation.
The coronavirus disease 2019 (COVID-19) pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is still ongoing. A key element in controlling the pandemic is the development and application of safe and effective vaccines. Therefore, identifying factors that could potentially enhance the immune response to vaccination (antibody levels) could be very important. Parameters related to vaccine administration, such as the time of day of vaccination, would be comparatively simple to implement.
Circadian rhythms regulate the immune system by a temporal modulation of the immune function (Giri et al., 2021; Sengupta et al., 2021), with human immune cell counts and inflammatory markers showing daytime variation (Wyse et al., 2021). Circadian parameters such as the time of day of vaccine administration may influence vaccine-induced immune response. As shown in a few previous studies on vaccines against seasonal influenza, Hepatitis A, and antituberculosis vaccine bacillus Calmette-Guérin (BCG), morning vaccination generally enhanced production of antibodies compared to afternoon vaccination (de Bree et al., 2020; Long et al., 2016a, 2016b; Phillips et al., 2008). However, findings on optimal time are not entirely consistent across studies, and Langlois et al. (1995) found a significantly higher antibody response if an influenza vaccine (strain A/Philippines) was administered at 1 pm, as compared to morning or afternoon, but no significant differences for the other two vaccine strains tested (A/Chile or B/USSR). This was in line with Kurupati et al. (2017), as well as Long et al. (2016a, 2016b) who also reported no significant effect of timing of seasonal influenza vaccination for two of their three subgroups, the H3N2 A and the B-influenza strain vaccines. Evidence on whether time of day of vaccination affects the responsiveness of vaccinated persons to the recently developed COVID-19 vaccines is sparse. In a small prospective cohort study conducted among 63 health care workers in Guangzhou, China, vaccination with an inactivated COVID-19 vaccine, BBIBP-CorV (Sinopharm, Beijing), in the morning led to significantly higher levels of neutralizing antibodies (NAbs) against the receptor-binding domain of SARS-CoV-2 spike protein and higher B-cell counts 56 days after vaccination (Zhang et al., 2021). A recent UK-based study of 2784 health care workers vaccinated with either an mRNA-based (BNT162b2, Pfizer) or vector-based (ChAdOx1 nCoV-19, Astra Zeneca) vaccine against COVID-19 corroborated an influence of time of day of vaccination on anti-spike immunoglobulin (IgG) levels measured over 2-10 weeks after vaccination, but results were not consistent across subgroups of exposure (age, sex, type of vaccine) and follow-up time points (Wang et al., 2021). Additional studies among non-shift workers, with more detailed information on time of day of vaccination and on other vaccine types, are warranted.
We sought to investigate the potential association of time of day of administration of a vector vaccine against SARS-CoV-2, ChAdOx1 nCoV-19 (AstraZeneca), on the SARS-CoV-2 anti-spike S1 subunit (S1) IgG levels 3 weeks after vaccination in a retrospective analysis of a large sample of university employees vaccinated continuously throughout the day. We hypothesized that time of day of vaccination would be associated with antibody levels.
Materials and Methods
Study Population and Data Collection
Study participants were employees of the Medical University of Vienna (MUV) from five centers/departments with regular daytime working hours, who received their first dose of ChAdOx1 nCoV-19 (AstraZeneca) vaccine in March 2021, as part of the MUV COVID-19 vaccination program. On the day of their vaccination, employees could opt for having their antibody levels measured at baseline and at 3 weeks after vaccination. Among those who agreed to antibody measurements, blood was drawn and anti-S1 IgG levels were determined at baseline (same day as the first vaccine dose) and at 3 weeks (21 or 22 days) after vaccination. Information on age and sex was also available. The study received approval by the Ethics Committee of the Medical University of Vienna (ECS 1588/2021).
Information on time of day of vaccination was available for a total of 1160 employees. Of them, 853 (74%) had serological information on antibody levels at 3 weeks. Employees with missing antibody levels at 3 weeks (non-participants) comprised of those that missed their appointment or received the vaccine but opted out of the antibody measurement. Non-participation was not associated with time of day of vaccination, but females and slightly younger employees were more likely to participate (Suppl. Table S1). Employees with missing baseline antibody levels (N = 22) and those who were seropositive or gray zone (antibody levels > 25.6 binding antibody units/mL [BAU/mL]; N = 28, 3.4%) at baseline were excluded from the analysis (Figure 1).

Flow chart of study participants included in the analysis.
Time of Day of Vaccination and Blood Sampling
All vaccinations were administered in 2021 between 2 and 29 March. Information on the exact time of day (hours and minutes) of vaccination was collected based on fixed predefined vaccination appointments, scheduled in 2-min intervals between 9:00 and 16:00 h by the administrative personnel of each sub-unit. There was no possibility for appointment re-arrangements according to personal preferences. Blood samples for antibody measurements were taken just prior to vaccination at baseline and between 9:00 and 12:00 h at 3 weeks after vaccination.
Serology
Blood samples were stored at 4 °C overnight, centrifuged the next day, and stored again at 4° until their analysis, which took place on the same day or the following. Serum samples were prepared to determine anti-S1 IgG antibody levels (in BAU/mL) using the Anti-SARS-CoV-2 QuantiVac (IgG) ELISA (Euroimmun). Antibody levels at 3 weeks which were above the assay’s upper sensitivity limit (limit of detection [LoD]) of 384 BAU/mL (N = 124 samples; 15.4%) were not further titrated; these values were replaced by two times the upper LoD (768 BAU/mL). Values of baseline antibodies below the lower LoD of 3.2 BAU/mL (N = 621 samples; 53.5%) were replaced by half the lower LoD (1.6 BAU/mL). We used manufacturer’s instructions (Euroimmun, 2021) to classify antibody level test result as negative (<25.6 BAU/mL), gray zone (25.6-35.2 BAU/mL), positive (35.2-384 BAU/mL), and above LoD (>384 BAU/mL).
Statistical Analysis
The analytic dataset consisted of participants with complete information on vaccination time, antibody levels at baseline and 3 weeks, and covariates (age and sex). Descriptive analyses of the exposure (time of day of vaccination), outcome (antibody levels at 3 weeks), and covariates (age, sex, and baseline antibody levels) were conducted. Time of day of vaccination was analyzed as a continuous variable and in hourly bins (9:00-10:00, 10:01-11:00, 11:01-12:00, 12:01-13:00, 13:01-14:00, 14:01-15:00, 15:01-16:00 h). Summary statistics of the continuous time variable and frequencies of the hourly bins were described. Mean and median antibody levels 3 weeks after vaccination were determined for each hourly bin. Linear regression analysis (natural and log-transformed antibody levels) was used for calculation of beta coefficients (β coef) and 95% confidence intervals (CIs) for each hourly bin of time of day of vaccination, in order to estimate the difference in antibody levels associated with later vaccination time points; the first morning hourly bin (9:00-10:00) was used as the reference group. Linear and quadratic terms of vaccination time were added in linear regression models to inspect the linearity of associations. We used generalized additive models (GAM) to explore the shape of the association (smooth function) of time of day of vaccination with antibody levels at 3 weeks after vaccination. We used natural splines (normal distribution) with 3 degrees of freedom and the identity link and report the p > Gain which is a measure of goodness of fit of the GAM compared to the linear model (StataCorp, 2021). We used crude and multivariable adjusted models for age, sex, and baseline antibody levels. In the main analysis, antibody levels were analyzed in the original scale and in secondary analysis they were log-transformed. In order to assess effect modification by sex and age, interactions were tested in linear regression models using the linear and quadratic term of time of day of vaccination. Statistical interactions between vaccination time (linear and quadratic term) and (1) sex (female/male) and (2) age (continuous) were tested by adding both interaction terms (e.g., age × time and age × time2) in regression models and by using log-likelihood ratio tests to compare models with and without the interaction terms. Analyses were stratified by sex and 10-year age categories (<30, 30-40, 40-50, >50 years). p-values were not adjusted for multiple testing and should be interpreted exploratorily only.
Several sensitivity analyses were conducted to account for the uncertainty related to the non-titration of antibody levels >384 BAU/mL and imputation of these values. First, Cox proportional hazards models (Dinse et al., 2014; Wood et al., 2011) using antibody levels as the “time” variable and censoring participants with levels >384 BAU/mL were used and hazard ratios (HR) and 95% CI were calculated. Hazard in these models can be interpreted as lower antibody levels. Second, values > 384 BAU/mL were replaced with one and three times the assay’s upper LoD (384 and 1152 BAU/mL, respectively), and results were compared to the main analysis that used two times the upper LoD (768 BAU/mL). Third, a categorical outcome was created (seronegative/gray zone: <35.2 BAU/mL, seropositive: 35.2-384 BAU/mL, highly seropositive: >384 BAU/mL), and the association of time of day of vaccination with odds of being seronegative and highly seropositive compared to seropositive was assessed. Odds ratios (ORs) and 95% CIs were estimated in multinomial logistic regression models adjusted for age, sex, and baseline antibody levels. Finally, values >384 BAU/mL were excluded in sensitivity analysis. All statistical analyses were performed using Stata/IC 16.1. All tests were two-sided and significance level was set to 5%.
Results
The analytical dataset consisted of 803 SARS-CoV-2 naïve participants, 485 (60.4%) women, and 318 (39.6%) men (Table 1). Mean age of the participants was 42 years (SD = 12). Time of day of vaccination ranged between 9:00 and 16:00 h, with a mean of 12:06 h (Suppl. Fig. S1). Age and sex were evenly distributed across hourly bins of time of day of vaccination (Suppl. Table S2). Three weeks after vaccination, anti-S1 IgG levels were negative in 4.7%, borderline in 3.1%, positive in 76.2%, and above LoD in 15.4% of participants (Table 1).
Age and anti-S1 IgG antibody levels and test results at baseline and at 3 weeks after vaccination overall and by gender (N = 803).
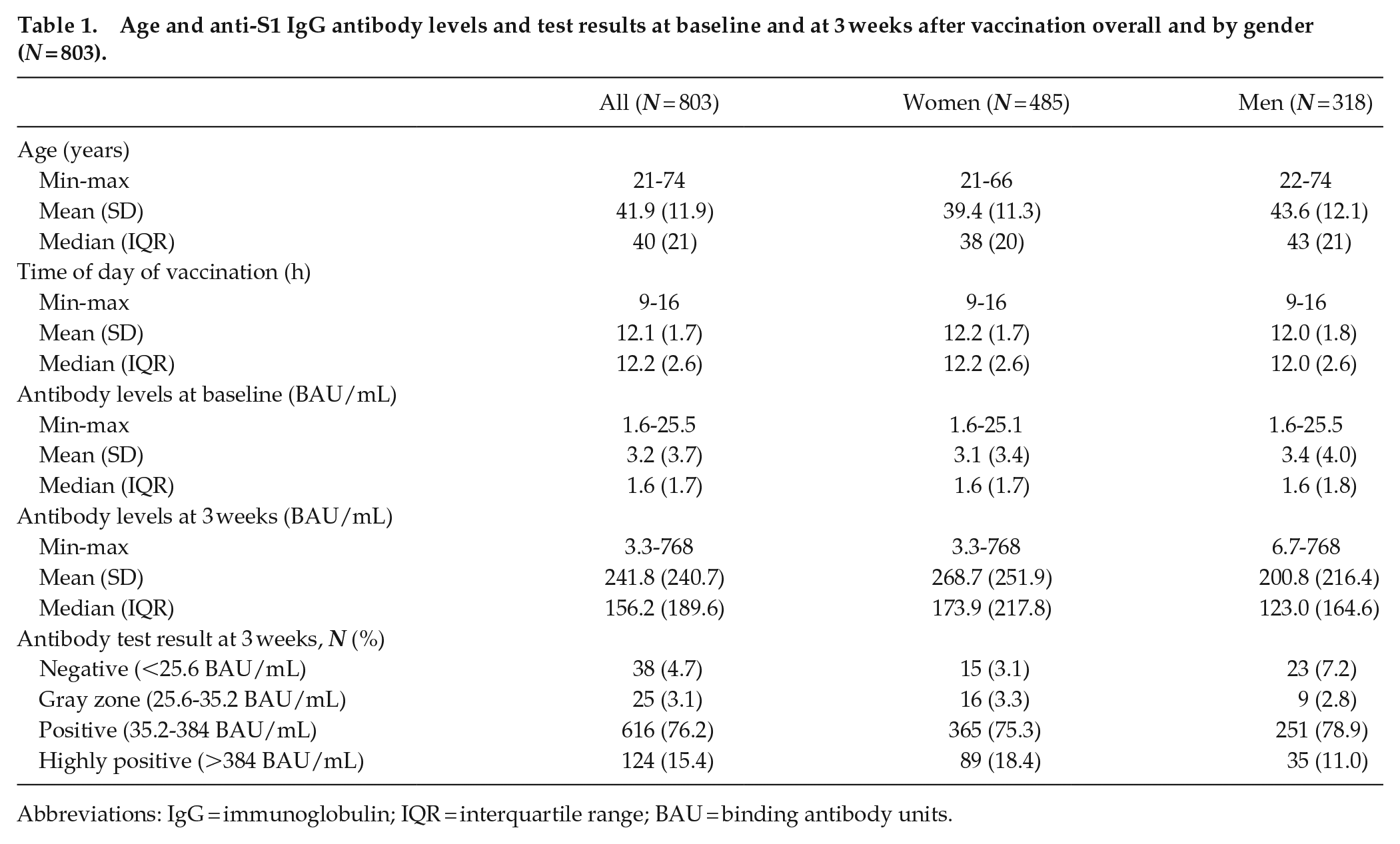
Abbreviations: IgG = immunoglobulin; IQR = interquartile range; BAU = binding antibody units.
Time of day of vaccination was associated with anti-S1 IgG levels at 3 weeks according to the multivariable-adjusted GAM spline (Figure 2, Suppl. Fig. S2), and the association was U-shaped or reverse J-shaped (p > Gain = 0.036). The quadratic term of time of vaccination was statistically significant (p = 0.01) in the regression model, consistent with departure from linearity (results not shown).
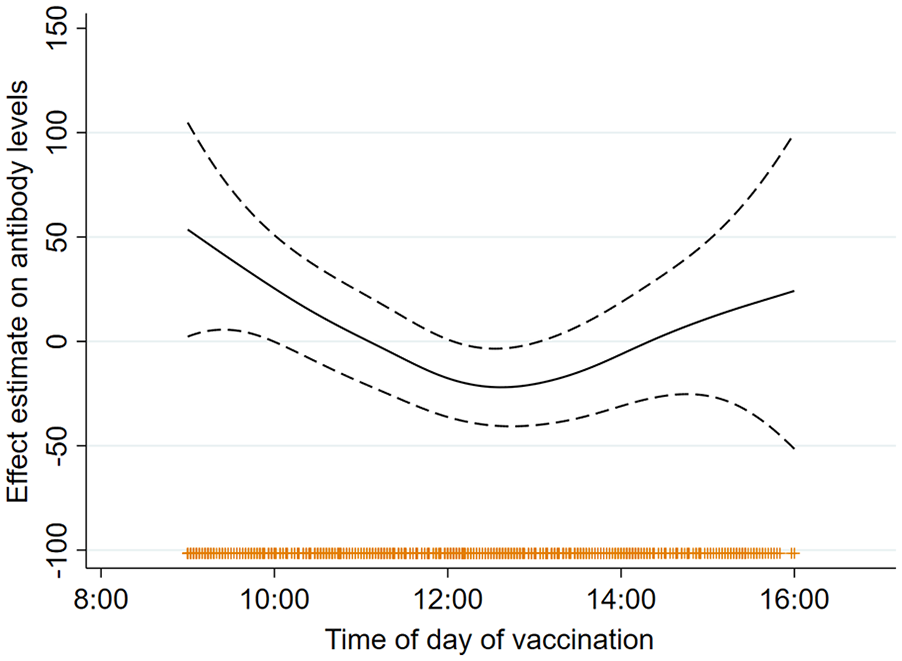
Multivariable adjusted general additive model (GAM) splines (smooth function) for the association of time of day of vaccination and anti-S1 IgG antibody levels at 3 weeks after vaccination in the full study population (N = 803). Abbreviation: IgG = immunoglobulin.
Mean antibody levels were highest after morning (9:00-10:00 h) vaccination (292.1 BAU/mL), lowest after midday or early afternoon (12:00-13:00 h) vaccination (217.3 BAU/mL), and intermediate after late afternoon (14:00-16:00 h) vaccination (264.4 BAU/mL) (Table 2). In the linear regression models, vaccine administration between 12:00 and 13:00 h was associated with significantly lower antibody levels compared to 9:00-10:00 h administration and differences remained significant after adjusting for age, sex, and baseline antibody levels (beta coefficient: −75.8; 95% CI: −131.3, −20.4).
Association of hourly bins of time of day of vaccination with anti-S1 IgG antibody levels (N = 803) in linear regression models.
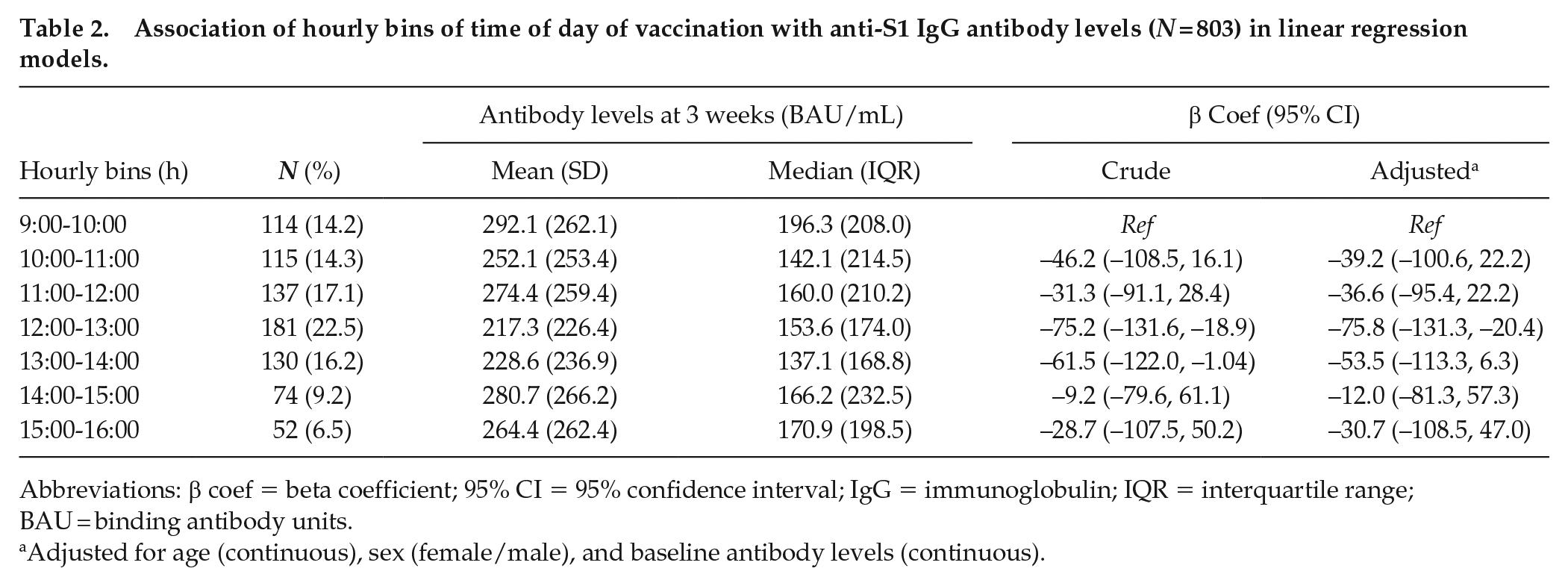
Abbreviations: β coef = beta coefficient; 95% CI = 95% confidence interval; IgG = immunoglobulin; IQR = interquartile range; BAU = binding antibody units.
Adjusted for age (continuous), sex (female/male), and baseline antibody levels (continuous).
The difference in antibody levels between morning and afternoon vaccination was larger among men (Table 3, Figure 3); however, the interaction with sex was not statistically significant (LR-test p for interaction = 0.43). Evidence of statistically significant variation in the effect of time of vaccination (size and shape) was found across age groups (LR-test p for interaction = 0.04). A U-shaped association was observed among younger participants (≤40 years) suggesting that morning or late afternoon vaccination may both predict higher antibody levels. The association was linear in participants older than 40 years, suggesting that in this age group morning vaccination led to a higher antibody response, compared to later in the day vaccination.
Time of day of vaccination with anti-S1 IgG antibody levels 3 weeks after vaccination grouped by sex (N = 803) in linear regression models.

Abbreviations: β coef = beta coefficient; 95% CI = 95% confidence interval; IgG = immunoglobulin.
Adjusted for age (continuous) and baseline antibody levels (continuous).
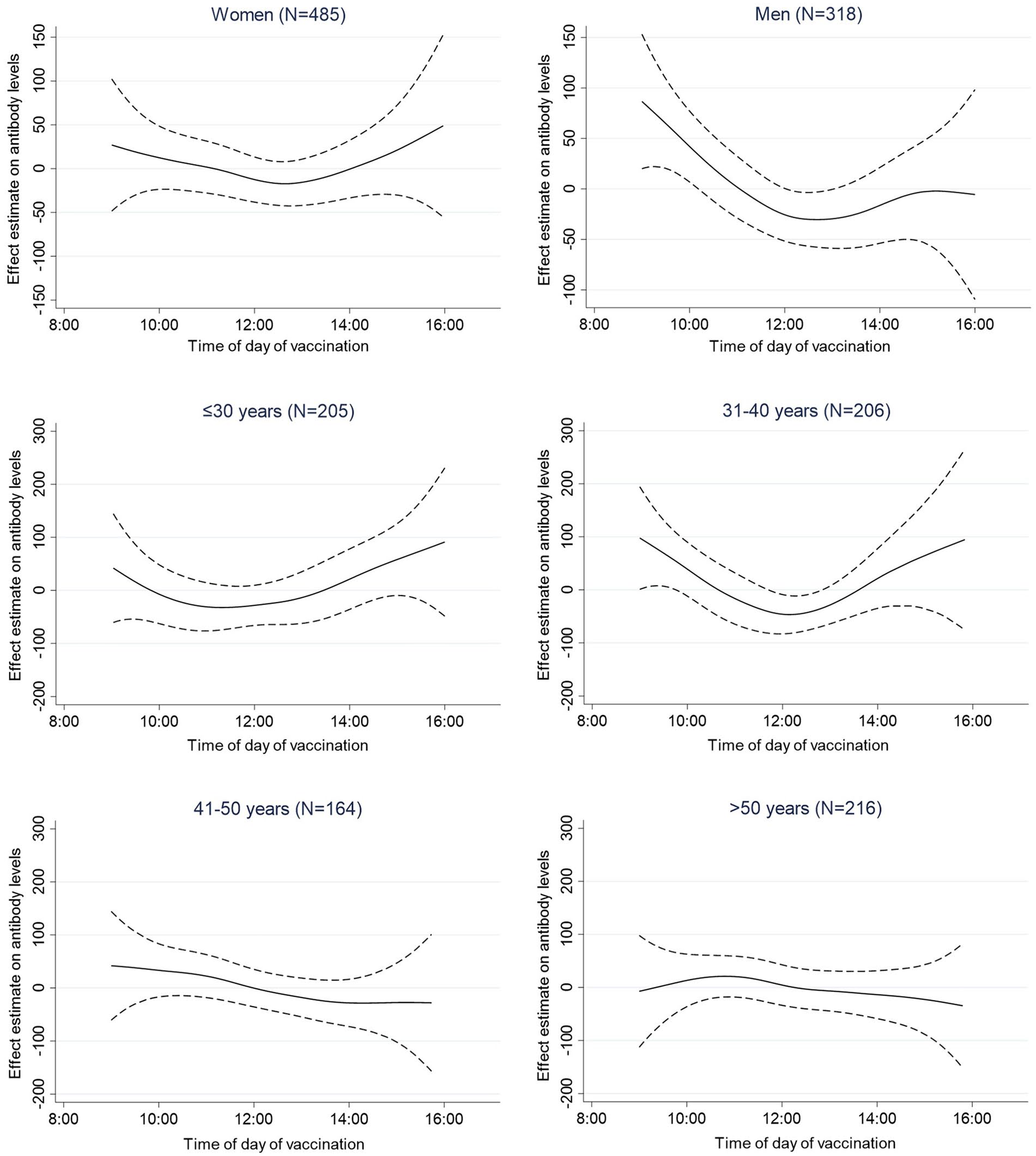
Multivariable adjusted general additive model (GAM) splines (smooth function) for the association of time of day of vaccination and anti-S1 IgG antibody levels at 3 weeks after vaccination grouped by sex and age. Abbreviation: IgG = immunoglobulin.
Results of the several sensitivity analyses were in line with the main analysis. Cox-proportional hazard regression of the hourly time bins censoring participants with antibody levels of >384 BAU/mL yielded hazard ratios similar in direction and significance to the main results (Suppl. Table S3). Results imputing 1 times upper LoD and 3 times upper LoD mirrored the main results that used the 2 times factor but the magnitude was smaller in the first and greater in the latter (Suppl. Table S4). In logistic regression analysis, we found that compared to 0900-1000 vaccination, 1200-1400 vaccination significantly increased the odds of being seronegative, which supports the results of the main analysis (Suppl. Table S5). The proportion of samples below the limit for seronegativity/gray zone (<35.2 BAU/mL) and above the upper LoD (>384 BAU/mL) were evenly distributed with small variation across hourly bins of time of day of vaccination (Suppl. Table S5). Finally, the inclusion of positive or gray zone antibody levels at baseline to the analysis (Suppl. Table S6) and the exclusion of non-titrated high antibody levels from the analysis (Suppl. Table S7) did not change the results. Analysis of log-transformed antibody levels was consistent with the analysis results based on non-transformed levels (Suppl. Table S8).
Discussion
In summary, our results from a study population of 803 SARS-CoV-2 infection naïve adults demonstrate an association of time of day of the first vaccination dose with the vector-based vaccine ChAdOx1 nCoV-19, Vaxzevria (AstraZeneca) on the magnitude of anti-S1 IgG levels after 3 weeks. Antibody levels were highest after morning vaccination (0900-1100), lowest after early afternoon vaccination (1200 and 1400), and intermediate after late afternoon vaccination (1400-1500).
We found a reverse-J association between time of day of vaccination and antibody levels, with strongest antibody responses after morning vaccination. Our results, although exploratory, are in line with a recent comparable study (Zhang et al., 2021) of an inactivated COVID-19 virus vaccine, BBIBP-CorV (Sinopharm, Beijing), among 63 health care workers. Participants vaccinated with both doses in the morning (0800-1000) showed 1.8-fold higher levels of NAbs against the receptor-binding domain of SARS-CoV-2 spike protein, compared to those vaccinated in the afternoon (1500-1700). Participants vaccinated in the morning also yielded stronger B cell and T follicular helper cells responses, and higher frequencies of monocytes and dendritic cells. However, this study compared predefined groups of exposure (morning vs afternoon), whereas we analyzed vaccination times continuously and described a daily pattern of variation in vaccine response. A significant influence of time of day of vaccination on SARS-CoV-2 anti-spike IgG antibody levels after vaccination was also supported in a recent study of 2784 health care workers vaccinated with an mRNA-based (BNT162b2, Pfizer) or vector-based (ChAdOx1 nCoV-19, Astra Zeneca) vaccine against SARS-CoV-2 (Wang et al., 2021). This study was indicative of slightly higher antibody levels (measured at 2-10 weeks) among those vaccinated later in the day, though not consistently across sex, age, and vaccine type subgroups. This finding is partly in agreement with the non-linear association pattern we observed. Furthermore, our study included persons working on day schedules with presumably more intact circadian rhythms, whereas both previous studies (Wang et al., 2021; Zhang et al., 2021) were conducted among health care workers. As such, they were likely exposed to night shift work, circadian misalignment, and sleep disruption (and thus presumably vaccinated at different internal times), which may interfere with vaccine-induced antibody responses (Fatima et al., 2021; Maidstone et al., 2021; Prather et al., 2021; Ruiz et al., 2020). We also found that morning vaccination increased the odds of being seropositive, compared to afternoon vaccination, which supports and strengthens our main analysis findings and if confirmed might be relevant for optimizing antibody responses among individuals with weak or no vaccination responses. In our study, the shape of the association changed (from reverse J-shape to linear) with age, but morning vaccination was the most potent in mounting antibody titers across all age groups. The age-specific results might be explained by a cross-talk of aging of the circadian system and sleep homeostasis (Giri et al., 2021) with age-related declines in immunity such as impaired immune cell function, diminished chemokine production, that may lead to reduced antibody response following vaccination or infection (Bartleson et al., 2021; Blomberg and Frasca, 2013). We also found suggestive evidence of effect modification by sex, with morning vaccination (vs early afternoon) stimulating a stronger vaccine response among male participants, although our stratified analyses were of limited power. Similarly, one previous study (Phillips et al., 2008) also reported that men—but not women—mounted a better peak antibody response after morning administration of hepatitis A and seasonal influenza compared to afternoon. Age and sex-specific differences in daily variation of vaccine response, underlying mechanisms, and their consequences for immunity need further study.
The impact of time of day of vaccination on vaccine responsiveness is biologically plausible (Giri et al., 2021; Sengupta et al., 2021). Circadian rhythms regulate many aspects of immune function, including the innate but also the adaptive immunity (Abele et al., 2019; Scheiermann et al., 2018; Wang et al., 2022). A number of animal and human studies support the occurrence of circadian oscillations in immunity including cytokine-mediated chemotaxis, T-cell differentiation, and leukocyte mobilization and activation (Haspel et al., 2020; Wyse et al., 2021). In animal models, immune response to antigenic challenge varies with time of day, with mice injected with lipopolysaccharides (LPS) in the evening exhibiting increased mortality compared with those challenged in the morning (Silver et al., 2012). Similarly, experimental animal studies have demonstrated time-of-day-dependent immune responses after vaccination (Fortier et al., 2011; Nobis et al., 2019; Silver et al., 2012). In one experiment, mice vaccination with ovalbumin (OVA) at night resulted in increased antigen-induced lymphocyte proliferation and increased interferon (IFN)-γ production compared with day vaccination. This phenomenon was correlated with rhythmic expression of toll-like receptor 9 (TLR9) on macrophages and B cells (Silver et al., 2012). In another experiment, Nobis et al. (2019) also showed circadian vaccination responses after injection of mice with dendritic cells loaded with the OVA peptide antigen (DC-OVA) vaccine. These effects were mediated by the intrinsic clock of CD8 T-cells that led to differential T-cell activation and proliferation according to time of day of vaccination. On the contrary, when circadian clock function in lymphocytes is lost, the rhythmic lymphocyte trafficking through lymph nodes and lymph—which is an important immune surveillance mechanism—is ablated (Druzd et al., 2017). Finally, there is significant amount of evidence connecting circadian rhythms to the function of the digestive system, gut microbiota, and immunity (Wang et al., 2022). The observed reverse J-association pattern in our data could be explained by behavioral aspects such as meal timing that exerts a strong influence in peripheral clocks and thus may impact the gut microbial diversity, gut cell motility, and nutrient absorption which might in turn influence immune responses to infection or vaccination (Brooks et al., 2021; Zheng et al., 2020). Despite the longer time frame compared to innate immunity, interestingly and despite earlier skepticism, adaptive immunity exhibits circadian rhythmicity even weeks after the initial challenge (Wang et al., 2022). However, it is still unclear how circadian rhythms in immune responses observed weeks or months after the initial stimulus are defined and maintained, and this is a high-priority research area for future studies (Haspel et al., 2020).
Another relevant question is whether reported differences in antibody levels correlate with higher levels of protection. Antibody-mediated immunity in protection against SARS-CoV-2 infection is supported by results from a large natural experiment in health care workers from the United Kingdom (N = 12,541) (Lumley et al., 2021) showing that the presence of anti-S or anti-nucleocapsid IgG antibodies due to infection was associated with a substantially reduced risk of SARS-CoV-2 reinfection after 6 months. A large meta-analysis pooling data from three single-blind randomized controlled trials of ChAdOx1 nCoV-19 (Voysey et al., 2021) reported a relationship between antibody levels and vaccine efficacy against primary symptomatic COVID-19 for both binding and neutralizing antibody levels. Data from an efficacy trial of the ChAdOx1 nCoV-19 vaccine in the United Kingdom (Feng et al., 2021) showed that a vaccine efficacy of 80% against symptomatic infection with SARS-CoV-2 was achieved with an average of 264 (95% CI: 108, 806) BAU/mL of anti-S antibodies. Results from a large UK-based cohort of 92,584 individuals without evidence of prior infection after having received two doses of ChAdOx1 found that 67% of individuals were protected against infection at estimated levels of 107 BAU/mL anti-spike IgG (Wei et al., 2021). Therefore, the observed variation in antibody levels in our study could indicate a clinically relevant effect of time of day of vaccination.
Strengths of our study include (1) the large sample size, (2) the collection of detailed information on time of day of vaccination in a large time window covering from early morning to late afternoon, (3) the inclusion of an infection naïve population of regular (day) working hours, and (4) the synchronous assessment of antibodies at 3 weeks (day 21 or 22) after vaccination for all participants that increased precision. Our study also has a number of limitations. First, it included only limited information on potential confounders, for example, chronotype and thus confounding might have occurred, although vaccination appointments were predefined and not influenced by personal preferences. Second, antibody levels greater than the upper LoD of the assay (17.2% of samples) were imputed, which might have led to an under- or overestimation of the true effects. However, several sensitivity analyses showed that this did not substantially impact the results in terms of effects direction and significance. Third, the time of day of blood collection for antibody measurements at 3 weeks was not known, but there is no conclusive evidence of daily variation in antibody titers (Kurupati et al., 2017; Reinberg et al., 1977; Wyse et al., 2021), and in a recent relevant study, SARS-CoV-2 anti-spike IgG antibody levels post-vaccination were not influenced by time of day of blood collection (Wang et al., 2021). Fourth, p-values were not adjusted for multiple testing and results should be interpreted explanatorily only. In terms of generalizability, participants were university employees and therefore of working age and presumptive above-average socioeconomic and educational status. Our analysis focused on the short-term (3 weeks) effects of only the first dose of a vaccination against SARS-CoV-2 in a naïve population without considering the second and third doses that are recommended for optimal protection (European Centre for Disease Prevention and Control, 2021). However, our results are in line with studies that have evaluated first and second doses of a COVID-19 vaccine (Zhang et al., 2021). Finally, although vaccine effectiveness is reflected in both cellular and humoral immune responses (He et al., 2021), we evaluated only one type of antibody response (anti S1 IgG), but binding and neutralizing antibodies have been shown to correlate in healthy populations (Dolscheid-Pommerich et al., 2022; Mendrone-Junior et al., 2021).
Overall, our data indicate that circadian parameters such as time of day of vaccination may play a role in human COVID-19 vaccination immune responses. When feasible circadian rhythms could be harnessed to help elicit a stronger immune response and optimize vaccination strategies against SARS-CoV-2. Future studies need to evaluate if time of day of vaccination has an impact on the responsiveness to the full vaccination scheme against SARS-CoV-2 and whether this might influence the protection rate. Our study included healthy participants, but a “timed” vaccination approach might be in particular useful among people showing weaker vaccine-induced antibody responses. Further studies should aim to include vulnerable populations such as immunocompromised patients and older adults.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304221132355 – Supplemental material for The Association of Time of Day of ChAdOx1 nCoV-19 Vaccine Administration With SARS-CoV-2 Anti-Spike IgG Antibody Levels: An Exploratory Observational Study
Supplemental material, sj-pdf-1-jbr-10.1177_07487304221132355 for The Association of Time of Day of ChAdOx1 nCoV-19 Vaccine Administration With SARS-CoV-2 Anti-Spike IgG Antibody Levels: An Exploratory Observational Study by Astrid C. Erber, Angelika Wagner, Marianna Karachaliou, Maren Jeleff, Polyxeni Kalafatis, Manolis Kogevinas, Beata Pepłońska, Isabel Santonja, Eva Schernhammer, Hannes Stockinger, Kurt Straif, Ursula Wiedermann, Thomas Waldhör and Kyriaki Papantoniou in Journal of Biological Rhythms
Footnotes
Acknowledgements
We would like to thank Christina Hössel, Beate Syrch, and Melitta Poturica for the coordination of the vaccination campaign. Furthermore, we would like to thank the medical team Dooa Al-Mamoori, Lisa Dohr-Loufouma, Romana Klasinc, Mateusz Markowicz, Peter Pichler, Peter Tauber, Karin Schreitmüller, Claudia Seidl-Friedrich, Brigitte Stuckart, Andrea Wessely, and Maja Zabel for their effort in the COVID-19 vaccine campaign for the staff members and people affiliated with the pre-clinical institutes of the Medical University of Vienna. We are grateful for the commitment of the serology team Tatjana Matschi, Vanessa Maurer, Barbara Schaar, Karin Schoiswohl, and Andrea Wendl to perform all the antibody measurements. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization and methodology: ACE, KP, AW, TW; Formal analysis: KP; Writing—original draft: ACE, KP; Writing—review and editing: ACE, KP, MK, KS, AW, TW, ES, MJ, PK; Supervision: KP. All authors have read and agreed to the published version of the manuscript.
Conflict of Interest Statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Grants from NIH/NHCHD (ES), Pfizer Inc. (IS), and Synovo GmbH (HS) in the previous 3 years unrelated to this manuscript, participation on advisory boards (ES) and leadership/fiduciary roles in societies (HS) unrelated to this manuscript, stock ownership (AE, HS) unrelated to this manuscript; the remaining co-authors have no conflicts of interest to disclose.
Note
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
