Abstract
The COVID-19 pandemic has necessitated novel approaches and collaborative efforts across multiple disciplines. It is known that various aspects of our physiology and response to pathogens are under tight clock control. However, the assimilation of circadian biology into our clinical and research practices is still evolving. Using a focused review of the literature and original analyses of the UK Biobank, we discuss how circadian biology may inform our diagnostic and therapeutic strategies in this pandemic.
The coronavirus disease 2019 (COVID-19) epidemic, which as caused by severe acute respiratory coronavirus-2 (SARS-CoV2), has prompted a scramble for safe and effective treatments. Our focus remains on vaccine development and repurposing approved drugs. However, reviewing our current understanding of chronobiology as applicable to other viral pathogens may help us realize opportunities to harness the power of circadian biology for SARS-CoV-2.
Circadian rhythms are evolutionarily conserved pathways that serve as a means for living organisms to adapt to the environment. It is an anticipatory system, preparing us to deal with challenges to our resilience, including infections. Molecular clockworks are highly conserved, tightly regulated, and exhibit a high degree of redundancy (Takahashi, 2017). Directed by a master clock in the suprachiasmatic nucleus, peripheral tissue clocks are among the most prominent biological networks that integrate cross-tissue functions, such as are intrinsic to cardiovascular homeostasis, metabolism, and the immune response (Yang et al., 2013).
The clock may be relevant to the pathophysiology and treatment of COVID-19 in the following ways:
As we reopen institutions and design schedules and workspaces, we could test whether time of day influences the susceptibility to or severity of COVID-19 infection. Historically, shift workers have been known to have increased susceptibility to many diseases (Smith and Eastman, 2012) and demonstrate differences in their immunological profile modified by their chronotype and the time from last night shift (Loef et al., 2019). However, in the context of the pandemic, the central question is whether the risk of infection among shift workers is influenced by the time of their shift. This is applicable to all shift workers, including health care workers, but it has particular relevance to the clustered outbreaks in workplaces where social distancing has been a challenge, such as the meat-processing industry (Waltenberg et al., 2020). In many ways, the COVID-19 pandemic presents us with a natural experiment. Thus, an intriguing possibility would be to leverage the many well-designed contact-tracing initiatives to address the time-of-day–specific susceptibility of various populations.
Further, these differences in the immune system based on time of day should be considered when working on animal models of COVID-19. That there are differences between the mouse and man, in terms of immune architecture and response, is well documented (Mestas and Hughes, 2004; Shay et al., 2013). However, specifically relevant here is the fact that mice are nocturnal and humans are not. Promising results from mice containing a humanized ACE2 (Hassan et al., 2020) and other models need to be interpreted in the light of the nocturnal habitus of mice. Appreciation of the differences in their circadian rhythms may improve the reproducibility of data and directly affect the translation of findings from animal to human studies.
In conclusion, our molecular clocks may modulate viral cellular entry and the consequent immunological response to infection. Timing of administration may influence the response to administration of a vaccine or to the drugs used to attenuate the severity of COVID-19. The various intersections of the circadian clocks and immunobiology as well as the epidemiology of COVID-19 (Fig. 2) constitute biologically plausible hypotheses that are readily testable.
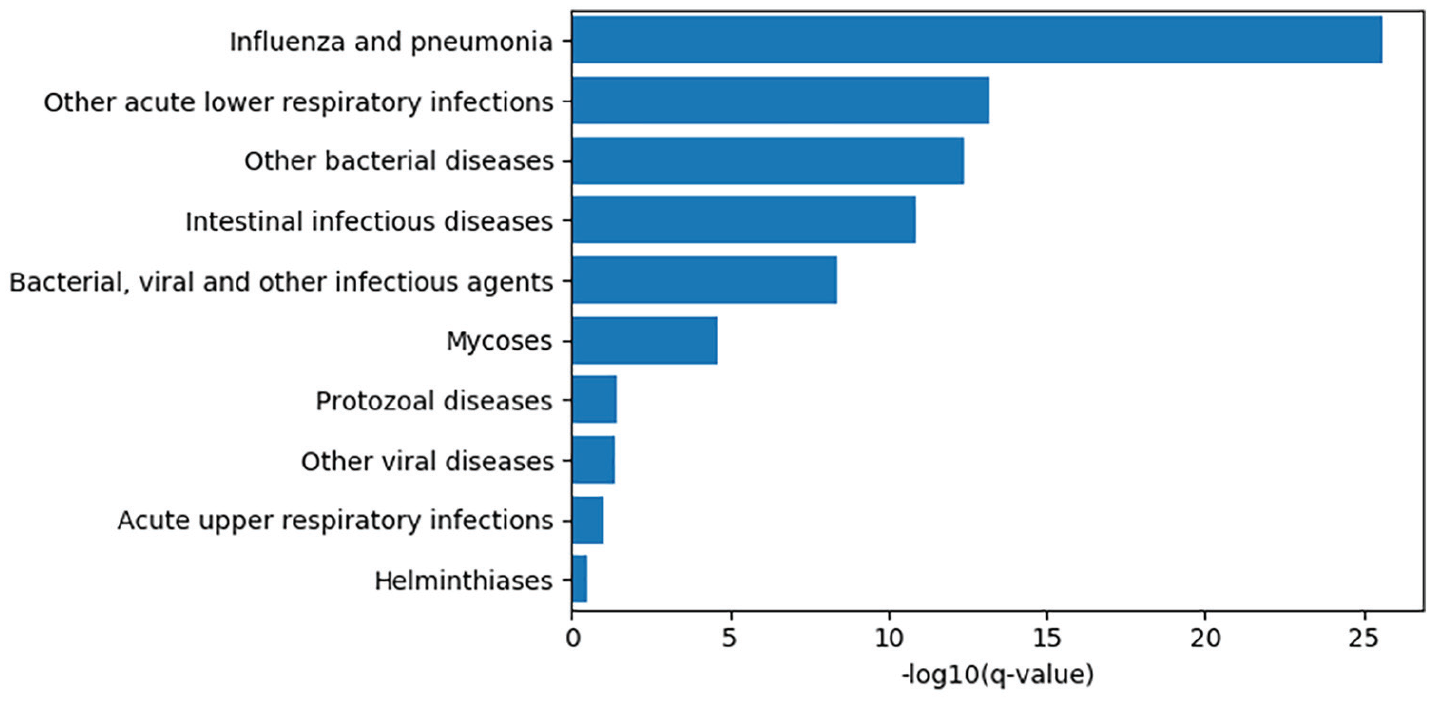
The top 10 infection phenotypes associated with relative circadian amplitude among participants of the UK Biobank, by −log10 of the Benjamini-Hochberg q-values. Cases were extracted by lifetime occurrence of ICD-10 codes and associations computed by a linear model controlling for age, sex, socioeconomic status, body mass index, smoking status, and self-reported overall health. Relative amplitude was computed as a ratio (M10-L5/M10+L5) where M10 and L5 are are the participant’s average activity levels of the ten hours of highest activity and five hours of the lowest activity, respectively ( Witting et al., 1990).
Summarized representation of the ways circadian rhythms interface and thus may be harnessed against COVID-19.
Footnotes
Acknowledgements
This work was supported by the NHLBI-K08HL132053 (to S.S.), NIH/NCRR RR023567 (to G.A.F.), a Merit Award from the American Heart Association (to G.A.F.), a grant from the Volkswagen Foundation (to G.A.F.), and 5T32MH106442-04 and NCATS-5UL1TR000003 (to T.G.B. and G.R.G). Dr. FitzGerald is the McNeil Professor of Translational Medicine and Therapeutics and a senior advisor to Calico Laboratories. This research has been conducted using the UK Biobank Resource under project ID 50398. We thank Kaitlyn Forrest for her help with the graphics.
