Abstract
Foraging and feeding are indispensable for survival and their timing depends not only on the metabolic state of the animal but also on the availability of food resources in their environment. Since both these aspects are subject to change over time, these behaviors exhibit rhythmicity in occurrence. As the locomotor activity of an organism is related to its disposition to acquire food, and peak feeding in fruit flies has been shown to occur at a particular time of the day, we asked if cyclic food availability can entrain their rhythmic activity. By subjecting flies to cyclic food availability, that is, feeding-starvation (FS) cycles, we provided food cues contrasting to the preferred activity times and observed if this imposed cycling in food availability could entrain the activity-rest rhythm. We found that phase control, which is a property integral to entrainment, was not achieved despite increasing starvation duration of FS cycles (FS 12:12, FS 10:14, and FS 8:16). We also found that flies subjected to T21 and T26 FS cycles were unable to match period of the activity rhythm to short or long T-cycles. Taken together, these results show that external food availability cycles do not entrain the activity-rest rhythm of fruit flies. However, we find that starvation-induced hyperactivity causes masking which results in phase changes. In addition, T-cycle experiments resulted in minor period changes during FS treatment. These findings highlight that food cyclicity by itself may not be a potent zeitgeber but may act in unison with other abiotic factors like light and temperature to help flies time their activity appropriately.
Keywords
Circadian clocks integrate cues from the environment and temporally regulate physiological and behavioral programs to aid animals in fulfilling their daily needs by anticipating cyclic changes in their day-to-day environment and timing their physiology accordingly (Dunlap et al., 2003). This is achieved by the process of entrainment which is the ability of the clock to synchronize to cyclic cues in the environment. Abiotic factors that cycle with time of day such as light and temperature serve as time cues or “zeitgebers” to the clock (Pittendrigh, 1960; Johnson et al., 2003). Similarly, biotic factors such as food resources may also serve as time cues to the clock of many animals (Frisch and Aschoff, 1987; Hau and Gwinner, 1996; Sharma et al., 2000).
Food resources frequently undergo changes in quality and quantity. While these changes are more apparent over seasons, daily food availability may also change as is documented in the case of various plant-pollinator and prey-predator interactions (Bloch et al., 2017; Kronfeld-Schor et al., 2017). The cyclicity of these interactions is made possible by the circadian clocks that regulate diverse aspects of pollination such as those involved in flower advertisements in plants (Overland, 1960; Matile, 2006; Fenske et al., 2015; Yon et al., 2016) and foraging activities in pollinator species (Fenske et al., 2018). Activity of several insect species such as bees (Bloch et al., 2017), mosquitoes (reviewed in Sougoufara et al., 2017), and bedbugs (reviewed in Barrozo et al., 2004); (Romero et al., 2010) have been shown to be influenced by food availability.
Restricted food access in rodent models under laboratory conditions invokes an anticipatory response in the form of an activity bout before food availability called the food anticipatory activity (FAA; Richter, 1922). FAA occurs for as long as food is restricted and even at times when animals are not usually active. For example, when food is restricted to daytime, FAA is observed during daytime which is otherwise a period of low activity in nocturnal animals (Mistlberger, 2011; Carneiro and Araujo, 2012). FAA is not dependent on the canonical light-entrainable oscillator (LEO) located in the suprachiasmatic nucleus (SCN; Stephan et al., 1979), and is thought to be controlled by another clock which is termed food-entrainable oscillator (FEO) which has not yet been localized (Pendergast and Yamazaki, 2018). Furthermore, it has been shown that feeding entrains several peripheral clocks, such as those in the liver, adipose tissue, and skeletal muscle (Damiola et al., 2000; Yang et al., 2006; Mohawk et al., 2012). Therefore, food provided at unusual times of the day can disrupt the phase relationship between the LEO and the liver peripheral clock. Other than the occurrence of FAA, the overall activity-rest pattern of these animals remains largely unaffected by change in food availability when SCN is intact. However, in SCN-lesioned animals, FEO can completely entrain activity rhythms to food availability cycles (Pendergast and Yamazaki, 2018), suggesting that food can act as a secondary zeitgeber.
In the fruit fly, Drosophila melanogaster, a peripheral clock in olfactory receptor neurons of the antenna regulate a circadian rhythm in olfactory responses with a peak in the middle of the night (Krishnan et al., 1999; Tanoue et al., 2004). Similarly, a diurnal rhythm in electrophysiological responses of the labellar gustatory receptor neurons (GRNs) has been reported with a peak in the morning hours. The GRN clock also regulates a behavioral gustatory rhythm in proboscis extension reflex; an appetitive behavior with a peak in the morning (Chatterjee et al., 2010). Fruit flies also feed rhythmically with a peak in the morning in light-dark cycles and early subjective day in constant conditions (Xu et al., 2008). While most of these rhythms are controlled by either peripheral clocks (Xu et al., 2008, Krishnan et al., 1999; Chatterjee et al., 2010) or central clocks (Helfrich-Forster, 1998), or both (Fulgham et al., 2021; Barber et al., 2021), whether and how food availability affects any of these rhythms is unknown.
An organism’s active phase is the time when most of the resource gathering and energy requirements are likely to be fulfilled. Hence locomotion of most animals is a function of various drives such as foraging, feeding, mating, oviposition, and so on. It is imperative to bring congruence between the internal drive to feed and the availability of food resources in the environment. Circadian clocks could facilitate this by adjusting the active phase of the organism such that the animal performs foraging and feeding behaviors when food is likely to be available in the environment. Since being in an active state is closely tied to an organism’s disposition to acquire food resources, we asked if changing the time of food availability can bring about changes in the activity patterns and affect the underlying clock in D. melanogaster. In this study, we test the hypothesis that food availability cycles can act as a zeitgeber in entraining the activity-rest rhythm of D. melanogaster by imposing various types of feeding cycles.
Materials and Methods
Locomotor Activity Assay
Locomotor activity rhythm of flies was recorded using the Drosophila Activity Monitor (DAM, Trikinetics, USA). Four- to five-day-old virgin male flies, unless mentioned otherwise, were recorded in LD 12:12 with ad libitum food (standard cornmeal medium) at 25 °C for 2 to 3 days following which feeding-starvation (FS) cycles were imposed in constant dark (DD). The period of starvation lasted for either 12, 14, or 16 h depending upon the regime. Experimental flies received standard cornmeal food during the “feeding” phase of the cycle and were transferred into 2% agar during the “starvation” phase. FS cycles were imposed for 7 to 8 days following which flies were shifted to DD ad libitum food (DD ad lib) conditions for the next 7 days. Age-matched flies that were transferred into fresh food tubes at the same time as experimental flies served as disturbance controls. All transfers were conducted under far-red light illumination (> 630 nm) in DD.
For the phase-shifted FS cycle experiment, 5- to 6-day-old flies were subjected to the first FS (FS1) for 5 days after which the second FS (FS2) was imposed by either advancing or delaying the food transfers by 6 h with respect to FS1 for a period of 7 days. Offset estimated from ClockLab (Actimetrics, Wilmette, IL, USA) and acrophase (calculated using Actogram J; Schmid et al., 2011) were used as phase markers to obtain the phase of the rhythm during and after FS cycles in all the regimes.
DD1-FS-DD2 experiment was performed in the same way, with one major exception. Flies were subjected to LD 12:12 only during rearing, that is, from the embryo stage to 2 days post-eclosion. Two-day-old flies were transferred to DD ad lib for 8 days (DD1), following which FS 8:16 was imposed for 11 days, where period of starvation was phase shifted by 6 h with reference to lights-on of the rearing LD regime. This was followed by DD ad lib food (DD2) for 8 days.
For T26 and T21 cycles, 5- to 6-day-old flies were recorded in LD 12:12 with ad libitum food for 5 days following which FS cycles were imposed in DD. A T26 FS cycle was imposed such that the flies experienced 13 h of food availability and 13 h of starvation. Similarly, a T21 FS cycle was imposed where flies experienced 10.5 h of food availability and 10.5 h of starvation. Age-matched flies served as disturbance controls as previously described. Seven cycles were imposed following which flies were shifted to DD ad lib food for the next 7 days. To determine periodicity during T26 and T21 feeding regimes and DD ad lib phase, χ2 periodogram analysis was carried out using ClockLab.
All DAM recordings were made at 1-min intervals, and activity data were binned at 15-min intervals for activity profiles and actograms. Fly transfers per monitor took about 10 to 12 min, during which time, activity of flies was continuously recorded from DAM monitors. Data loss due to monitor reconnections did not exceed 4 to 5 min per transfer, and such readings were recorded as 0 thereby maintaining the time stamp of activity over the course of the experiment.
Immunocytochemistry
Fly brains were sampled at 4-h intervals on the first and seventh cycle of the FS 12:12 treatment. Brains were dissected in ice cold phosphate buffered saline (PBS) and fixed with 4% paraformaldehyde (PFA) for 30 min. Fixed brains were blocked with 10% horse serum for 1 h at room temperature and 6 h at 4 °C. Samples were then stained with antibodies against PERIOD (anti-PER rabbit, 1:20,000, gift from Jeffrey C Hall, Brandeis University, previously pre-adsorbed in per0 embryos) and Pigment Dispersing Factor (anti-PDF, mouse, 1:5000 DSHB, PDF C7, RRID: AB_760350) and incubated for 48 h at 4 °C. After eight to nine washes with 0.5% PBT (0.5% TRITON-X 100 in PBS), brain samples were incubated for 24 h at 4 °C with secondary antibodies anti-rabbit-Alexa Fluor 488 (Invitrogen, #A11094, RRID: AB_221544, 1:3000) and anti-mouse-Alexa Fluor 546 (Invitrogen, #A11003, RRID: AB_141370, 1:3000). After eight to nine washes with 0.5% PBT, brains were mounted on glass slides using 7:3 glycerol: PBS as the mounting medium. Immmunocytochemistry for the brains sampled on the first cycle and those sampled on the seventh cycle was performed separately. Confocal images were taken in Zeiss LSM 880 (airyscan) microscope under 20× or 40× (oil immersion) lens.
Statistical Analyses
Daily offsets and acrophases were analyzed using the circular statistics toolbox for MATLAB R2018a ver. 9.4.0 (Mathworks, Inc.; Berens, 2009). A small number of flies that became arrhythmic during DD ad lib were not considered in these analyses. Rayleigh’s tests were performed on each circular dataset to test if circular means belonged to a von Mises distribution. Since none of the datasets followed von Mises distribution, we compared angular phases using non-parametric multiple pair-wise common median tests, followed by Bonferroni corrections. Inter-individual phase synchrony between controls and experimental flies was tested by measuring the degree of dispersion of mean phases averaged across last 3 days of the FS cycles. Wallraff rank sum test for angular dispersion was performed on phase values (radians) using R core team (version 3.6.1, 2019, R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/). Δ phase was defined as change in phase on the first day of DD ad lib from the mean phase (last 3 days) during FS regime. Two-sample t test was used to compare Δ phases between controls and experimental flies. For T21 and T26 experiments, a χ2 test for proportions was performed using GraphPad Prism (version 9.2.0 for Windows, GraphPad software, San Diego, California, USA, www.graphpad.com) to compare proportions of flies exhibiting different periodicities. Change in period was tested using Mann-Whitney U test. Activity levels during starvation for all FS regimes were analyzed using repeated-measures analysis of variance (ANOVA) with day as the repeated measure and treatment as the between-group factor. Mauchly’s test for sphericity was performed on all the data sets and Greenhouse-Geisser corrections were applied when the assumption for sphericity was not met. The above tests were performed using IBM, SPSS Statistics for windows (version 26, 2019, IBM corp., Armonk, NY, USA). Multiple post hoc pair-wise comparisons were performed using t tests with Bonferroni corrections. Kruskal-Wallis ANOVA was performed on number of PER+ LNv at different time points for each treatment and cycle followed by a post hoc Dunn’s multiple comparisons test using GraphPad Prism. All other analyses were performed on STATISTICA (version 7, 2004, StatSoft Inc, Tulsa, OK, USA).
Results
To address if external food availability cycles, that is, the FS cycles, entrain the activity-rest rhythm of D. melanogaster, we subjected flies to three different FS cycles of increasing starvation duration—namely, FS 12:12 (12 h of feeding followed by 12 h of starvation, Figure 1a), FS 10:14 (10 h of feeding followed by 14 h of starvation, Figure 1b), and FS 8:16 (8 h of feeding followed by 16 h of starvation, Figure 1c). We also subjected flies to 18 h of starvation, although feeding duration as small as 6 h resulted in high rates of mortality within the first few FS cycles. Therefore, we did not use feeding durations smaller than 8 h in our study. Activity-rest rhythms were recorded in LD 12:12 at 25 °C on ad libitum food for 2 to 3 days before shifting to one of the aforementioned FS regimes in DD. Previously, it has been reported that starvation causes hyperactivity (Connolly, 1966; Keene et al., 2010). We reasoned that starvation during night would result in nocturnal hyperactivity, which would clearly show phase changes in activity due to FS cycles, if any. Thus, in all three FS regimes, the feeding duration overlapped, partly or completely with daytime of the previous LD cycles. Following 7 to 8 days of FS regime, flies were subjected to DD ad lib.
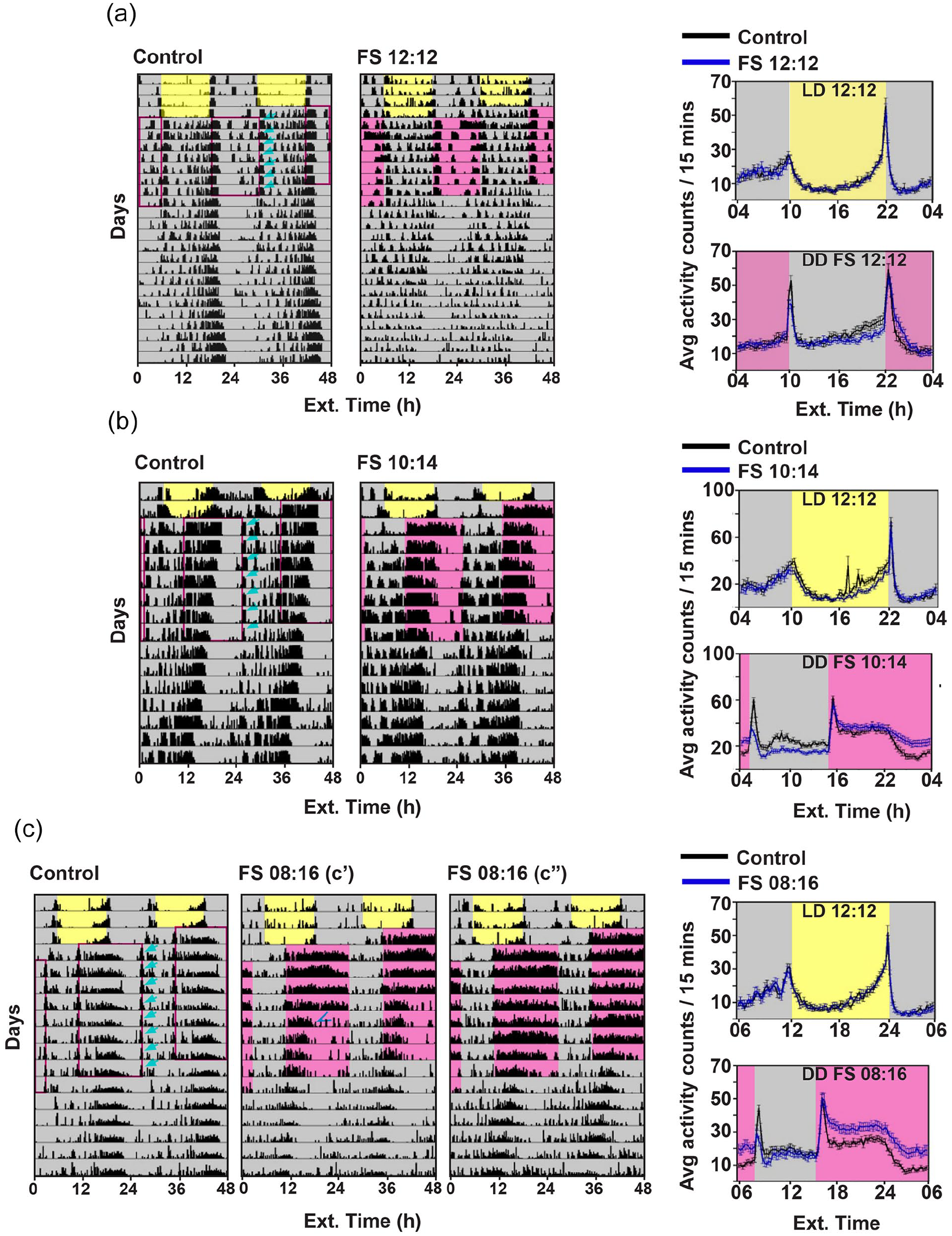
Flies subjected to FS cycles display a combination of free-running and masking behavior. Representative double plotted actograms of (left) age-matched disturbance controls and flies subjected to FS cycles; (a) FS 12:12, (b) FS 10:14, (c) FS 8:16 and (right) average profiles of activity binned in 15 min intervals (top) before and (bottom) during FS cycles, (a) FS 12:12, (b) FS 10:14, (c) FS 8:16. Error bars for activity profiles are ± standard error of the mean (SEM). Yellow-shaded region represents daytime and gray-shaded region represents nighttime with ad libitum food. Pink-shaded region represents starvation. In every regime, disturbance caused due to transfer of flies in fresh tubes results in small bursts in activity, arrows indicate the startle bouts. In FS 8:16, (c′) a small fraction of flies (7/23) shows gradual reduction in activity (arrowhead) during the starvation window across days of the treatment; (c″) another fraction of flies (14/23) displays elevated activity levels across all days of the treatment. x-axis indicates external time.
Disturbance control and experimental flies show startle bouts of activity when they were moved to new tubes (Figure 1a-c, left, arrows). While these startle bouts can be attributed to disturbance due to change of tubes, we expected to observe changes in the activity-rest rhythm because of FS cycles over and above the disturbance caused during the assay (Figure 1). For example, as expected from previous studies (Connolly, 1966), experimental flies show increased activity level during starvation compared with control flies (Figure 1a-c, right). To determine whether FS cycles are indeed entraining the activity-rest rhythm, we examined classical criteria of entrainment, namely, day-to-day phase stability, inter-individual phase synchrony, phase control, and period matching with zeitgeber cycle (τ = T; Moore-Ede et al., 1982). We used offsets and acrophases which is equivalent to the radial center of mass of activity (Diez-Noguera, 2013), as the phase markers in all experiments.
Phase Changes During FS Cycles Are Transient in Nature
To determine whether flies are entraining to FS cycles, we compared offsets and acrophases between control and experimental flies in the three regimes within a run—LD 12:12 ad lib, FS cycles, and DD ad lib. In addition, we divided the FS cycles into two parts—first four cycles (FS cycles 1-4) and last three cycles (FS cycles 5-7), to separate the effect of transients observed in the first few cycles (Figure 1). As expected, offsets during LD ad lib were highly consolidated and mean phase angles (theta) of control and FS 12:12 experimental groups were comparable with each other (Figure 2a top row, Common median test, p = 0.78). Initially (FS cycles 1-4, second row), both disturbance control and experimental groups show change in phase angle as compared with their respective LD phase angle (Figure 2a, top and second rows, Common median test, p < 0.0005 for both groups). However, subjecting flies to FS 12:12 did not induce greater phase change than the control group (Figure 2a second row, p = 0.15, FS cycles 1-4). Indeed, day-to-day mean offset phases of control and experimental flies resembled each other during the FS regime (Suppl. Fig. 2A). Offset phases in FS cycles 5 to 7 are also different from LD in both control and experimental groups (Figure 2a, top and third rows, control: p < 0.0003, experimental: p < 0.003). Nevertheless, in FS cycles 5 to 7, offsets of both control and experimental groups appear to return to previous LD phase (Suppl. Fig. 2A). Finally, in DD ad lib, offsets of both control and experimental groups are comparable with LD offset phase (Figure 2a, first and fourth rows, control: p = 0.4, experimental: p = 0.14). These results show that subjecting flies to disturbance or FS 12:12 induces a transient change in phase, which returns to previous LD phase. Importantly, FS 12:12 does not induce changes greater than disturbance (Control vs. Experimental, FS 5-7: p = 0.08; DD ad lib: p = 0.77).
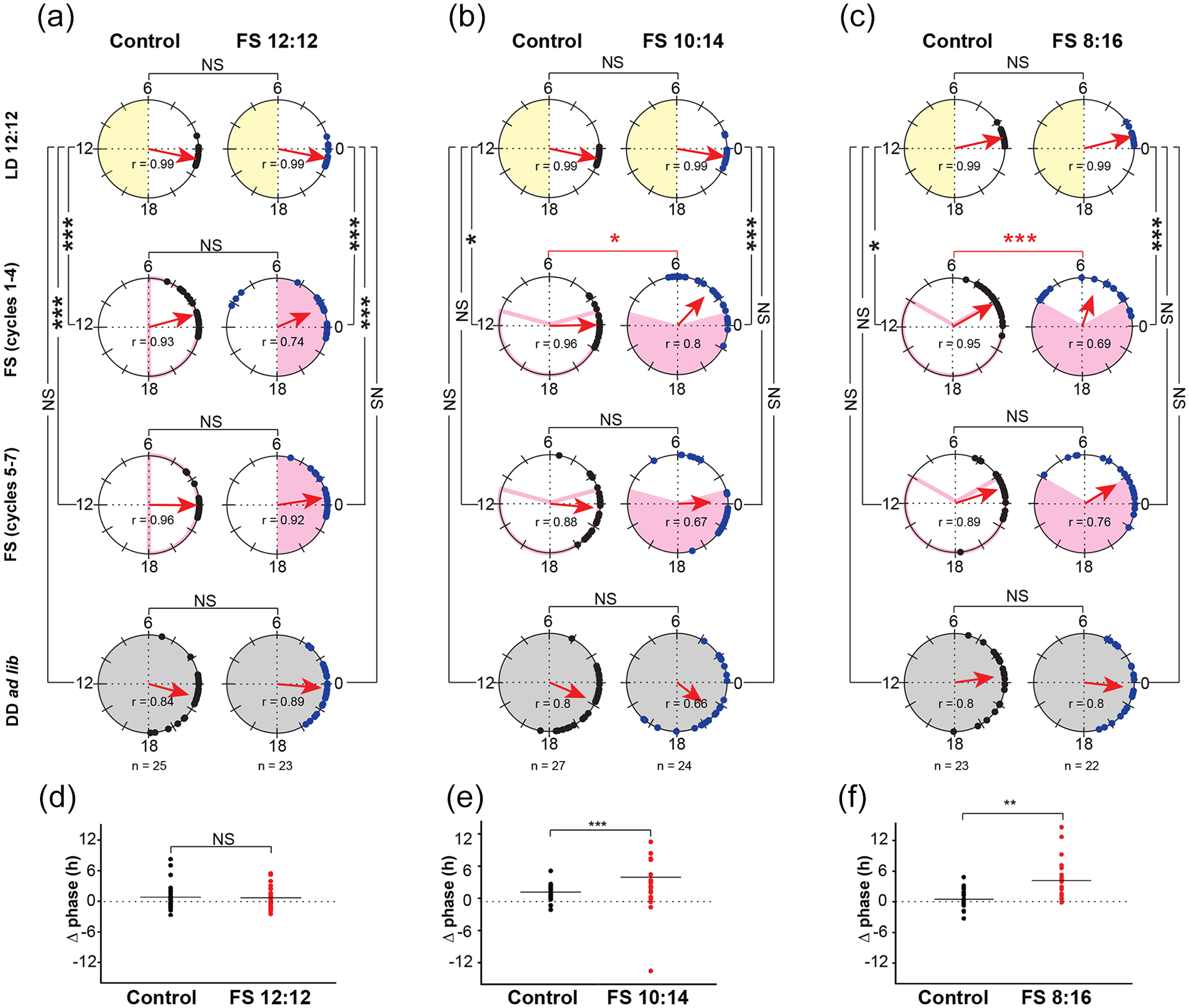
FS cycles result in transient changes in offsets, do not result in higher inter-individual phase synchrony and phase control. Polar plots representing activity offsets of disturbance control flies (black circles) and experimental flies (blue circles) subjected to (a) FS 12:12, (b) FS 10:14, and (c) FS 8:16. Each pair of polar plots show mean phase during different regimes within an experiment (LD 12:12, FS 1-4 cycles, FS 5-7 cycles and DD ad lib). Yellow-shaded region indicates lights-on time during LD 12:12, pink-shaded region indicates period of starvation during FS cycles in DD, gray-shaded region indicates constant dark (DD ad lib). Red arrows show consolidation (r) of phases during each regime. Length of the arrow indicates strength of consolidation, while angle represents mean phase. External time 0 is set to ZT 18 as per LD cycle. Pair-wise Common median tests were performed to compare phases across different regime and treatment groups. NS = not significant, *p < 0.003, **p < 0.0006, ***p < 0.0003 after Bonferroni corrections. Scatter plots depicting differences in offsets of FS cycles 5 to 7 from DD D1 (Δ phase) of individual control (black) and experimental (red) flies; horizontal line represents the mean Δ phase. (d) FS 12:12, (e) FS 10:14, (f) FS 8:16. *, **, *** indicate p < 0.05, 0.01, 0.001, respectively (Student’s t test).
We reasoned that lack of change in phase due to FS 12:12 could be because of starvation coinciding with lights-off of previous LD cycles, at a time when flies are expected to feed less (Xu et al., 2008; Seay and Thummel, 2011; Ro et al., 2014). Therefore, in our next experiments, we shifted the FS cycles with respect to previous LD by 4 to 5 h, such that feeding was restricted to periods partially overlapping with the previous lights-on phase, thereby allowing for visualization of phase changes. In addition, we increased the starvation duration to 14 or 16 h, to ensure maximum impact on activity-rest rhythm. Subjecting flies to FS 10:14 resulted in a transient change in phase during the first 4 cycles which was significantly different from controls (Figure 2b, FS cycles 1-4, p < 0.003). These phase changes were most prominent during the first 2 cycles (Suppl. Fig. 2B). Similar to FS 12:12, these transient changes appear to return to previous LD phase (Figure 2b, control: p = 0.06, experimental: p = 0.77). Similarly, in DD ad lib, both control and experimental groups show phases comparable with LD phases (Figure 2b, control: p = 0.78, experimental: p = 0.06 after Bonferroni corrections). Flies subjected to FS 8:16 showed similar trend as in FS 10:14, with transient phases significantly different from control initially (Figure 2c, FS cycles 1-4, p < 0.003), with maximum changes occurring on cycles 2 to 3 (Suppl. Fig. 2C). Phases in FS cycles 5 to 8 returned to previous LD phases in both control and experimental groups (Figure 2c, control: p = 0.64, experimental: p = 0.07 after Bonferroni corrections). Same trend was observed for comparison of DD ad lib with LD phase (Figure 2c, control: p = 0.77, experimental: p = 0.05, after Bonferroni corrections). These results suggest that subjecting flies to FS cycles causes transient change in phase, which eventually tracks to previous entrained phase under LD.
FS Cycles of Different Starvation Durations Do not Exert Phase Control on Activity-rest Rhythm
To test whether the phase in constant conditions follows from the previously entrained phase (i.e., to establish phase control), the change in offset (Δ phase) was quantified by subtracting offset on day 1 (D1) DD ad lib from the mean offset during FS cycles 5 to 7 (to avoid initial transient cycles). We expected maximum phase change (if any) to be manifested on D1 of DD ad lib condition. Δ phase is expected to be small if the flies are free-running, since their τ is ~ 24 h (Suppl. Table 1), and daily drift in phase is minimal. Under entrained conditions, the expectation is that phase on D1 would not be very different from the previously entrained phase, also resulting in a small Δ phase value. However, if masking was determining the offset phase, the expectation is that a detectable change in phase would occur as soon as constant conditions ensue.
While disturbance in controls caused some phase shift in the initial few cycles, in the later cycles, phases seem to drift apart suggesting that control flies begin to free-run (Suppl. Fig. 2A-C). We compared Δ phase of flies subjected to FS 12:12, FS 10:14, and FS 8:16 with their respective controls, assuming that controls are free-running. We found that Δ phase of control and experimental flies experiencing FS 12:12 (Figure 2d, two-sample t test, p = 0.84) are not different from each other. This indicates that FS 12:12 has no effect on activity-rest rhythm of flies. However, flies experiencing FS 10:14 (Figure 2e, two-sample t test, p < 0.001) and FS 8:16 (Figure 2f, two-sample t test, p < 0.001) showed significantly higher Δ phase compared with controls. This suggests that the phase attained during the last three FS cycles changed quickly on D1 of DD ad lib, which is characteristic of masking. These trends observed with offsets are replicated when acrophase is used as a phase marker, although, it should be noted that acrophase is a reliable phase marker when rhythms are unimodal as in DD (FS or ad lib) but performs poorly in LD where rhythms are bimodal (Suppl. Fig. 1). These results show that flies subjected to FS cycles do not exhibit phase control.
FS Cycles Do Not Increase Inter-individual Phase Synchrony
An entrained rhythm is phase-locked to the zeitgeber cycle, and this phase relationship is stable across multiple cycles and reproducible across individuals. This implies that in an entrained condition, flies are expected to exhibit similar phases resulting in higher inter-individual synchrony (for instance, Figure 2a top row, LD 12:12). Here, we examined the extent of phase dispersion within control and experimental fly groups during FS regimes. If FS cycles were entraining the activity-rest rhythm, we would expect a smaller dispersion with greater consolidation of phases. However, we find that the r-value, which is the length of the mean resultant vector of angular phases, an indicator of phase synchrony, is lower in experimental flies during FS cycles as compared with those of controls experiencing disturbance (Figure 2a-c, middle two rows). We find that the degree of dispersion of offsets in each of the experimental regimes of FS 12:12 (Wallraff rank sum test for angular dispersion, Kruskal-Wallis χ2 = 4.23, df = 1, p = 0.04), FS 10:14 (Kruskal-Wallis χ2 = 6.395, df = 1, p = 0.011), and FS 8:16 (Kruskal-Wallis χ2 = 4.18, df = 1, p = 0.041) was significantly different from their respective disturbance controls. This implies that phases during FS cycles are more dispersed than their respective controls suggesting lack of inter-individual synchrony during FS cycles.
In addition, we asked whether abrupt shifts in food availability schedules may reveal any features that were not previously detectable. Two jetlag FS 12:12 experiments were conducted, where the second set of FS cycles (FS2) were either phase delayed (Suppl. Fig. 3A and C) or advanced (Suppl. Fig. 3B and D) with respect to previous FS cycle (FS1). Like the previous FS 12:12 (Figure 1), the activity rhythm of the experimental flies during FS1 and FS2 was similarly phased as their disturbance controls (Figure 3a and b). Moreover, the phase of the activity-rest rhythm continued to follow from the previous entrained phase in LD 12:12 irrespective of FS2, suggesting lack of phase control (Figure 3a and b, Suppl. Fig. 3). Furthermore, Δ phase of control and flies experiencing FS 12:12 (Figure 3c, phase delay: p = 0.12 and Figure 3d, phase advance: p = 0.34) are comparable with each other. The degree of phase dispersion during the last 3 days of FS2 was not different in experimental flies as compared with their controls in both phase-delayed (FS2: Kruskal-Wallis χ2 = 3.072, df = 1, p = 0.08) and phase-advanced conditions (FS2: Kruskal-Wallis χ2 = 0.252, df = 1, p = 0.61) suggesting that FS cycles are not more consolidated than controls. These results suggest that FS cycles fail to increase inter-individual synchrony of activity rhythms. Taken together, lack of phase synchrony during FS cycles and presence of a masking component as detected by Δ phase (for both offsets and acrophases), we conclude that FS cycles do not entrain activity-rest rhythms but cause transient phase changes resulting in incomplete masking. Finally, our results suggest that phases during the FS cycles were possibly intermediary between the internal clock and masking components.
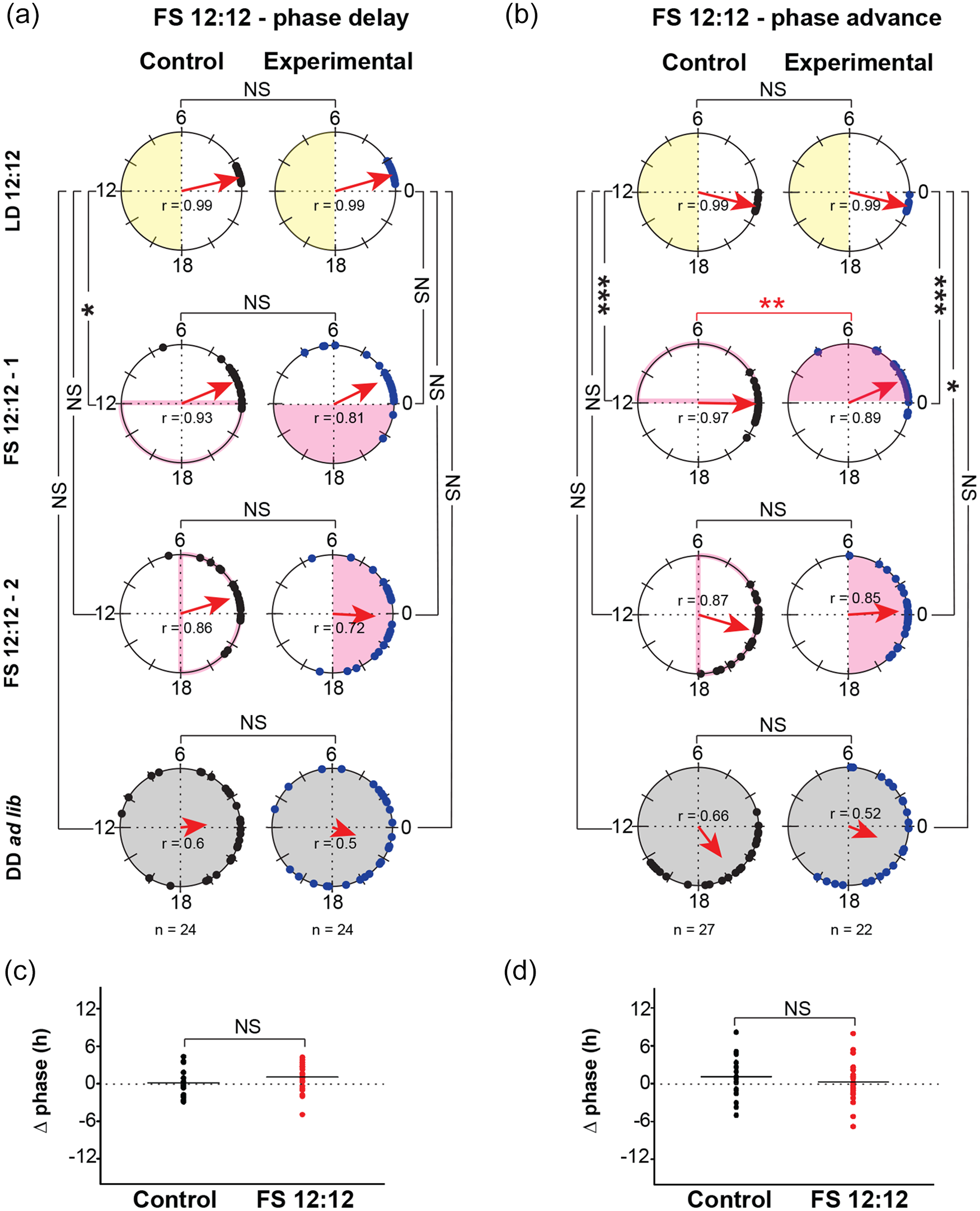
Phase-shifted FS cycles do not reveal any difference in inter-individual phase synchrony. Polar plots representing activity offsets of disturbance control flies (black circles) and experimental flies (blue circles) subjected to (a) phase-delayed FS 12:12, (b) phase-advanced FS 12:12. Each pair of polar plots show mean phase during different regimes within an experiment (LD 12:12, FS 1, FS 2, and DD ad lib). All other details as in Figure 2. Pair-wise Common median tests were performed to compare phases across different regime and treatment groups. NS = not significant, *p < 0.003, **p < 0.0006, ***p < 0.0003 after Bonferroni corrections. Scatter plots depicting differences in offsets of FS 2 (last 3 days) from DD D1 (Δ phase) of individual control (black) and experimental (red) flies; horizontal line represents the mean Δ phase. (c) Phase-delayed FS 12:12, (d) Phase-advanced FS 12:12. *, **, *** indicate p < 0.05, 0.01, 0.001, respectively (Student’s t test).
FS 8:16 Cycles Do Not Entrain the Activity-rest Rhythm in Female Drosophila Flies
Since female flies feed more compared with males (Wong et al., 2009), we asked if FS cycles can entrain activity-rest rhythm in female flies. We found similar results when female flies were subjected to FS 8:16 wherein most flies showed excessive activity in the starvation window (Suppl. Fig. 4a). Average phase was significantly higher from the controls during FS 8:16 which immediately reverted to pre-FS phase after the FS treatment (DD ad lib; Suppl. Fig. 4B, Common median test for control vs. experimental FS cycles 1-4 and FS cycles 5-8, p < 0.003 after Bonferroni corrections). In addition, we found that there was no phase control—as Δ phase was significantly higher from controls (Suppl. Fig. 4C, two-sample t test, p < 0.001) nor was there inter-individual synchrony (Wallraff rank sum test for angular dispersion Kruskal-Wallis χ2 = 0.38, df = 1, p = 0.53). Altogether, these results indicate that activity rhythms of both males and females do not entrain to FS cycles.
Starvation-induced Hyperactivity (SIH) Is Not Sustained Across FS Cycles
Starvation has been shown to result in excess activity levels which fall to the baseline levels after food has been provided (Connolly, 1966; Yang et al., 2015). Interestingly, we observed that cyclic food availability for several cycles does not result in consistent hyperactivity across all cycles. Flies that experienced 12 h of starvation per day for 7 consecutive days showed activity levels comparable with the controls in the 12-h starvation window each day. However, activity levels on the first 2 days were higher as compared with other days, (Supplemental Figure 5A, repeated-measures ANOVA, Greenhouse-Geisser ε = 0.44, F2.67, 149.3 = 28.39, main effect of day, p < 0.001, followed by pair-wise t tests with Bonferroni corrections for 21 comparisons). When flies were subjected to FS 10:14, they showed an immediate increase in activity in response to lack of food (Figure 1b right, Suppl. Fig. 5B). The activity levels were higher than the controls in the first three cycles of the treatment, after which they were comparable with the controls, (Supplemental Figure 5B, Greenhouse-Geisser ε = 0.52, F3.13, 175.32 = 3.72, day × treatment, p = 0.012, followed by pair-wise t tests with Bonferroni corrections for 49 comparisons). Interestingly, we found two types of behaviors among individuals when subjected to 16 h of starvation, 7 out of 23 flies showed excessive activity during starvation hours throughout the 8 days of FS 8:16 (type a flies, Figure 1c; center), and 14 out of 23 flies appeared to show excessive activity during starvation only for the first few days after which the activity seemed to decrease (type b flies, Figure 1c; right). Day-to-day activity levels of FS 8:16 flies, (Supplemental Figure 5C, Greenhouse-Geisser ε = 0.29, F2.05, 108.54 = 6.99, day × treatment, p = 0.001, followed by pair-wise t tests with Bonferroni corrections for 64 comparisons), showed reduction in activity levels after 6 days (Figure 1c right, Suppl. Fig. 5C). This shows that acrophases during FS are masked as a result of hyperactivity occurring in response to starvation.
Under Constant Conditions, Flies Continue to Free-run With Slight Changes in Phase During FS Cycles
Classical experiments testing for entrainment follow a schedule wherein animals are housed in constant conditions prior to and after exposure to zeitgeber cycles. In our studies so far, we subjected flies to a strong LD 12:12 cycle before imposing FS cycles in DD, and we observe no discernible entrainment. To avoid the possible lingering effect of LD cycles, we housed flies in DD, 2 days after adult emergence, followed by FS cycles, such that they are the only cycling environment experienced by flies. Since FS 8:16 cycles showed maximum effect on phase, we subjected flies to the following regime: DD1 → FS 8:16 → DD2. Here, we included two types of controls—a set which remained undisturbed throughout the experiment (apart from two instances when flies were given fresh food, Figure 4a, right, arrows) and a disturbance control where flies were flipped into fresh tubes as described previously. We observed that activity-rest pattern (Figure 4a) and mean phase of flies experiencing FS cycles were comparable with those of disturbance control and undisturbed control flies during FS 8:16 (Figure 4b). Furthermore, phase of rhythm changed with respect to DD1 when FS cycles were imposed (Figure 4b, DD1 vs. FS experimental: p < 0.0003); however, these changes did not continue into DD2 (Figure 4b, FS vs. DD2, p = 0.54 after Bonferroni corrections). Moreover, this phase change was also observed in control flies subjected to disturbance (DD1 vs. FS disturbance control: p < 0.0003), but not in undisturbed controls (DD1 vs. FS undisturbed control: p = 0.36 after Bonferroni corrections). Furthermore, DD2 phase was different from phase during disturbance (Figure 4b, FS vs. DD2 disturbance control: p < 0.0003; FS vs. DD2 undisturbed control: p = 0.8), although it is difficult to ascertain if this change is due to disturbance, daily drift in phase, or both. Importantly, subjecting flies to FS cycles did not change phase more than disturbance alone (Figure 4b, disturbance control vs. experimental: p = 0.18 after Bonferroni corrections). These results clearly show that FS cycles do not entrain activity-rest rhythms in flies lacking all time cues.
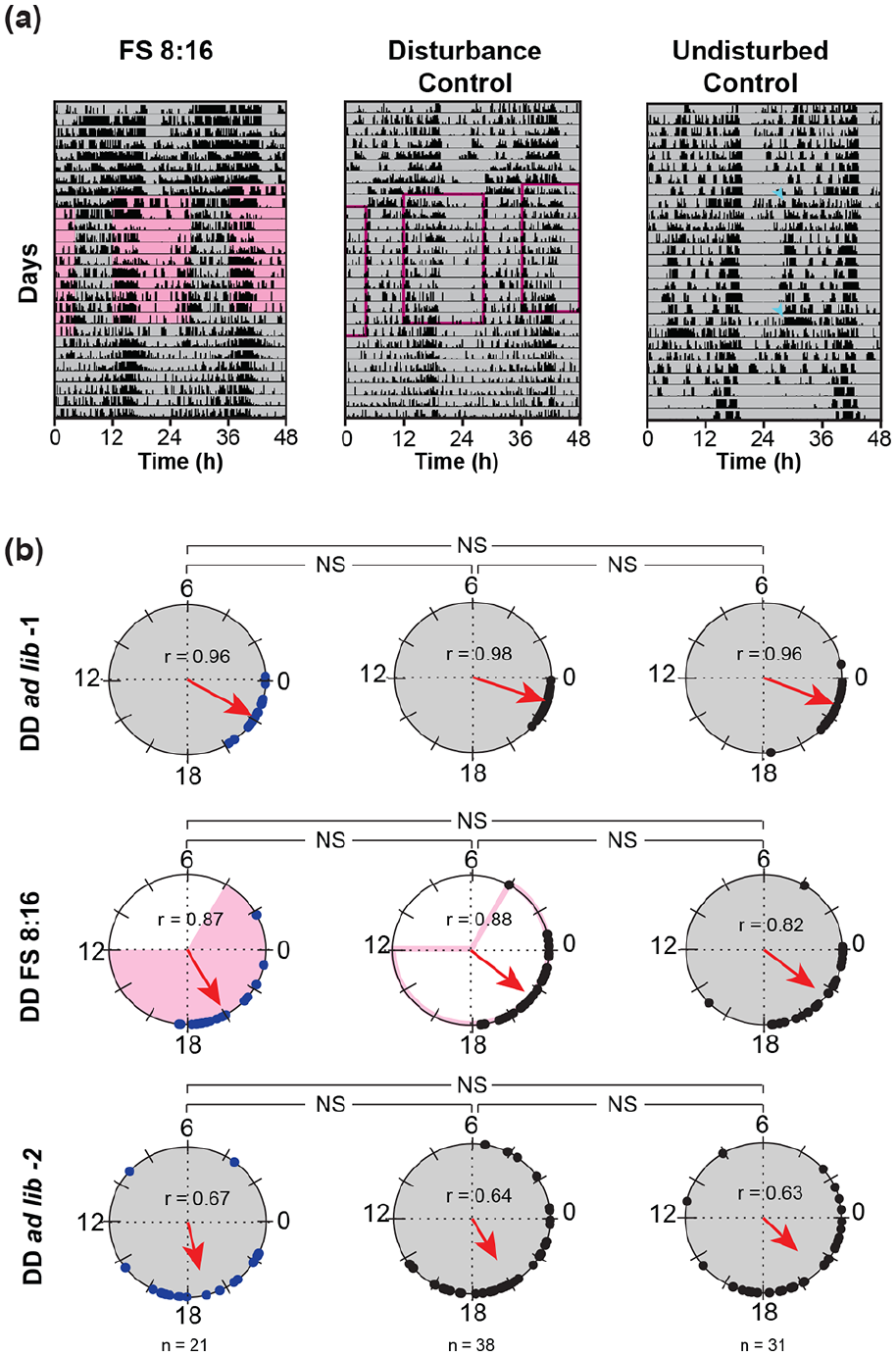
Flies exhibit minimal changes in phase during FS cycles under DD1-FS 8:16-DD2 regime. (a) Representative double-plotted actograms of (left) FS 8:16, (middle) age-matched disturbance controls, and (right) age-matched undisturbed controls. Blue arrows indicate two instances when undisturbed control flies were changed into fresh food tubes. (b) Polar plots representing offsets of (left) experimental flies subjected to FS 8:16 cycles (blue circles), (middle) disturbance control flies (black circles), and (right) undisturbed control flies (black circles). Each polar plot shows mean phase during different regimes within an experiment (DD1, FS 8:16, and DD2). All other details as in Figure 2. Pair-wise Common median tests were performed to compare phases across different regime and treatment groups. NS = not significant, *p < 0.003, **p < 0.0006, ***p < 0.0003 after Bonferroni corrections.
T26 and T21 FS Cycles Do Not Synchronize the Activity-rest Rhythm of D. melanogaster
We next assessed another criterion for entrainment, that is, period matching between activity-rest rhythm (τ) and external FS cycles (T). In the previous experiments, T was 24 h, and τ was also close to 24 h (Suppl. Table 1); hence it was not possible to test this criterion using data from FS 12:12, 10:14, and 8:16 experiments. We therefore imposed either a T26 or a T21 FS regime and asked if this could result in period matching of the activity-rest rhythm to external FS cycles. We subjected the flies to T26 FS cycles wherein the flies were provided food for 13 h and were starved for 13 h (Figure 5a and b) in an otherwise aperiodic environment (DD, 25 °C). A large majority (Figure 5c) of the experimental flies appear to free run with a phase similar to the previous entrained phase in LD 12:12 despite being subjected to T26 FS regime. However, they also exhibited a masked component to the disturbance caused due to change of tubes (Figure 5a, dashed line). A χ2 periodogram analysis during T26 FS revealed two significant periodicities; one that was close to 24 h (henceforth referred to as free-running component) and another that was close to 26 h (Suppl. Table 2, Figure 5a and b). Since both control and experimental flies exhibit this long period (~26 h) component, the same can be attributed to the physical disturbance experienced by both (henceforth referred to as the masking component). Presence of two significant periodicities in a large proportion of experimental flies suggests that T26 FS regime is unable to synchronize activity (Figure 5c, χ2 test for proportions shows no difference in the two groups; p = 0.18, χ2 = 3.22, df = 2). We estimated the difference in free-running component (~24-h period) during FS (24τ1, T26 FS) and after FS (24τ2, DD ad lib) for each fly. Interestingly, we found that experimental flies exhibited approximately 23 min longer period during T26 FS regime. This difference was significantly greater than the controls suggesting that T26 FS indeed significantly lengthens the internal period of the flies, albeit to a small degree (Figure 5d, U = 2388, p < 0.0001, Mann-Whitney U test, Suppl. Table 2).
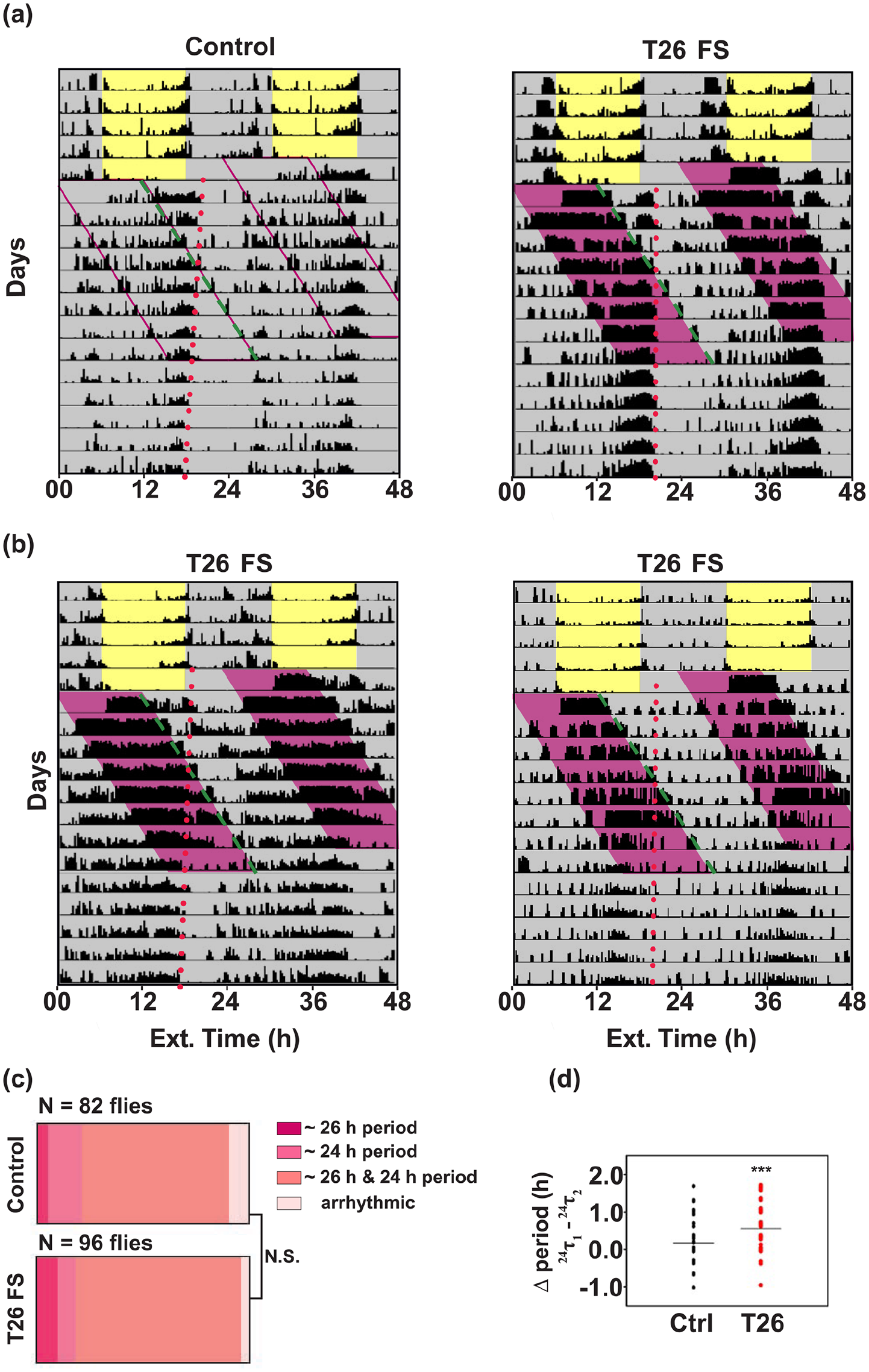
FS T26 cycles do not entrain the locomotor activity-rest rhythm. (a) Representative actograms of (left) control and (right) experimental flies recorded under LD 12:12 conditions for the first 4 days before being subjected to T26 FS cycles in DD conditions. Pink-shaded region represents 13 h of starvation on each day. T26 FS cycles continued for 7 days followed by DD ad libitum conditions. Both the control and the experimental flies show masked responses to the T26 regime along with a free-running component of activity (24τ1) that seems to follow the phase from the LD 12:12 cycle. Dashed line tracks the startle activity due to food changes (green) and dotted line tracks activity offset phase after LD 12:12 (red). (b) Two example actograms of flies exhibiting a single period (26 h, τ = T) during FS treatment. (c) Proportion of flies showing two different periodicities (26 and 24 h), only 26-h or only 24-h periodicity, or arrhythmicity during T26 FS are indicated. A major proportion of both control and experimental flies show dual periodicities. (d) Distribution of changes in the ~24-h periodicity during the T26 regime for controls and experimental flies. Differences were calculated by subtracting periods after T26 FS (24τ2) from the periods during T26 FS (24τ1). T26 FS flies have a longer free-running component (~24 h) during T26. NS denotes no difference between controls and T26 FS flies. *** denotes p < 0.001.
We next subjected the flies to a T21 regime with 10.5 h of food availability followed by 10.5 h of starvation in DD. We found similar results wherein the experimental flies continued to free run from the previously entrained LD phase along with a masked response to the external food availability cycles (Figure 6a and b, dashed line). Akin to T26, we found flies exhibiting two periodicities, a near 24 h component (free-running component) and a near 21 h component (masked component) as estimated by χ2 periodogram (Figure 6c). A higher proportion of T21 FS flies show only 21-h periodicity as compared with controls, (χ2 test for proportions shows a significant difference p < 0.0005, χ2 = 15.37, df = 2) yet majority of the experimental flies still show both 21- and 24-h periodicities. This further provided evidence for inability of the external T21 FS cycle to synchronize the activity-rest rhythm because of incomplete period matching. Moreover, T21 regime also affects the intrinsic period during the treatment. We estimated the difference in free-running period during FS (24τ1, T21 FS) and after FS (24τ2, DD ad lib) for each fly, and we found that experimental flies exhibited approximately 24 min shorter period during T21 FS regime. This difference was significantly lower than the controls suggesting that T21 FS significantly shortens the intrinsic period of the experimental flies (Figure 6d, U = 2389, p = 0.00096, Mann Whitney U test, Suppl. Table 3). Overall, inability to match the intrinsic period to T cycles suggests that FS cycles are inefficient in synchronizing the activity-rest rhythm. Thus, another criterion for entrainment (period matching: τ = T) is not met under conditions where food availability is cyclic. However, it is possible that T-cycles closer to 24 h (such as T23.5 or T24.5) may yet be able to entrain activity-rest rhythms. However, given that our flies exhibit free-running periods within this range, testing this hypothesis is challenging. Taken together, our results suggest that the FS cycles that we imposed do not entrain the activity-rest rhythms controlled by the central clock.
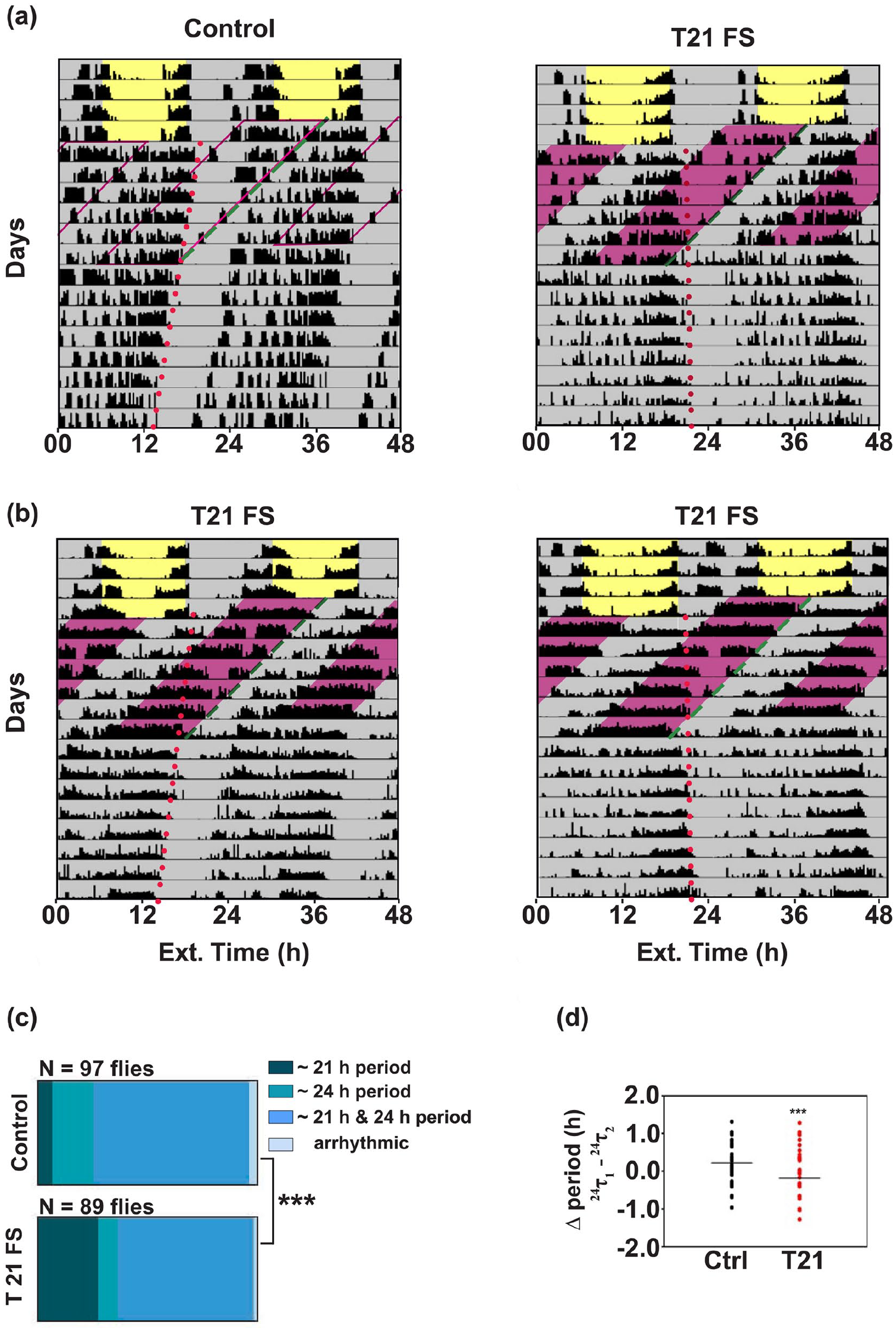
T21 cycles do not entrain the locomotor activity-rest rhythm in D. melanogaster. (a) Representative actograms of (left) control and (right) experimental flies recorded in LD 12:12 conditions for the first 4 days before being subjected to T21 FS cycles in DD. Pink-shaded region represents 10.5 h of starvation on each day. T21 FS cycles continued for 7 days followed by DD ad libitum conditions. Both the control and the experimental flies show masked responses to the T21 regime along with a free-running component of activity (24τ1) that seems to follow the phase from the LD 12:12 cycle. (b) Two example actograms of flies exhibiting a single period (21 h, τ = T) during FS treatment. (c) Proportion of flies showing two different periodicities (21 and 24 h), only 21-h or only 24-h periodicity, or arrhythmicity during T21 FS are indicated. A major proportion of both control and experimental flies show dual periodicities. A higher proportion of T21 FS flies show only 21-h periodicity. (d) Distribution of changes in the ~24-h period during the T21 regime for (left) controls and (right) experimental flies. Differences were calculated by subtracting periods after T21 FS (24τ2) from the periods during T21 FS (24τ1). T21 FS flies have a shorter free-running (~24 h) component during T21. *** denotes p < 0.001. All other details as in Figure 5.
FS Cycles Neither Improve Synchrony nor Affect the Phase of Molecular Clock in the Central Pacemaker Neurons
Finally, to examine whether we may uncover any molecular signatures of circadian clock phase under FS cycles, we conducted a preliminary study examining anti-PERIOD staining in the LNv of control and FS treated flies at two stages, first after 1 day of FS 12:12 when we expect no change between controls and experimental flies and again after seven cycles of FS 12:12. We counted the number of PER+ LNv at 4-h intervals as a readout of the PER accumulation rhythm. On the first day, we found that there was a clear peak and trough in the number of LNv cells marked by the PER antibody in both control, (Supplemental Figure 6, top left, Kruskal-Wallis ANOVA by ranks, H5 = 34.13, p < 0.0001 followed by Dunn’s multiple comparisons test, and FS 12:12 groups, Supplemental Figure 6, top right, Kruskal-Wallis ANOVA by ranks, H5 = 38.14, p < 0.0001 followed by Dunn’s multiple comparison test). After seven FS 12:12 cycles, we observed that both control, (Supplemental Figure 6, bottom left, Kruskal-Wallis ANOVA by ranks, H5 = 21.12, p = 0.0008 followed by Dunn’s multiple comparison test), and experimental, (Supplemental Figure 6, bottom right, Kruskal-Wallis ANOVA by ranks, H5 = 16.98, p = 0.0045 followed by Dunn’s multiple comparison test), flies show a damped PER accumulation rhythm most probably because of divergence of individual peak phases in DD. This suggests that FS 12:12 cycles do not bring about a significant enhancement in synchrony of the molecular clock in the central pacemaker cells.
Discussion
Flies exhibit a rhythmic feeding pattern which is under circadian control (Xu et al., 2008; Seay and Thummel, 2011; Xu et al., 2011; Ro et al., 2014; Barber et al., 2016; Murphy et al., 2017). Maximum food intake is shown to occur during the first half of the day, although a few studies also show a prominent evening bout of feeding (Seay and Thummel, 2011; Ro et al., 2014). Nevertheless, it is clear that feeding is rhythmic and is under the control of a circadian clock. We examined whether cyclic food availability can act as an entraining cue for the activity clock in fruit fly D. melanogaster by subjecting flies to FS cycles, with the feeding time restricted to reported peak hours of feeding (feeding time spanning 4-6 h after lights-on from previous LD). Under these regimes, we assessed basic criteria for entrainment—day-to-day phase stability, inter-individual phase synchrony, phase control, and period matching. We found that subjecting flies to FS cycles of different durations of starvation does not result in inter-individual synchrony and phase control. These results build on the findings of previous studies in which subjecting flies to FS 7:17 and FS 12:12 cycle with feeding restricted to evening or night time did not affect activity-rest pattern (Xu et al., 2011; Oishi et al., 2004, respectively). However, in each of the T24 cycles, we found that first four cycles, especially in FS 10:14 and FS 8:16, had a dramatic effect on phase which was transient in nature. Furthermore, T26 and T21 FS cycles could bring about a significant lengthening and shortening of period while only partially synchronizing the activity-rest rhythm to the external periodicity. These results suggest that while food cannot act as an entraining agent, it affects the clock properties such as phase and period. This indicates that the clock is perceptive to food availability in the environment and can respond by making small changes to clock properties without changing the overall pattern of activity-rest.
Drosophila larvae are known to feed voraciously until they acquire critical weight before pupation; as adults however, feeding is meager. Nevertheless, when deprived of food, flies are known to respond by increasing their locomotor activity (Knoppien et al., 2000; Keene et al., 2010) which has been attributed to foraging behavior in flies (Yang et al., 2015). Indeed, in most of our experiments, we observed Starvation-induced Hyperactivity (SIH) in both male and female flies. Since hyperactivity is a direct and immediate response to starvation, it can be considered as masking. In FS 10:14 and FS 8:16 regimes, we found that SIH decreased over subsequent cycles. This masking to lack of food is also reflected in the phase changes observed during FS cycles. We observed that day-to-day phase changes indeed mirror the activity levels that were also higher than baseline during the first few cycles and tapered to baseline in the subsequent cycles. Since acrophase is a phase marker that depicts the radial center of mass of activity, SIH may influence the day-to-day phases. This influence of cyclic starvation disappeared after release into constant conditions with ad lib food. This was evident from the fact that the Δ phase values were found to be significantly different from respective controls in all three T24 FS paradigms tested (Suppl. Fig. 2D-F). Therefore, while food availability is unable to entrain the activity-rest rhythm, masking to starvation may bring about some changes which indirectly affect the phase of the clock. This is of significance to organisms that often encounter unpredictability in food availability in their environment. Masking to changes in food availability becomes more relevant for female flies who in addition to their own survival, also need food patches for laying eggs. In fact, most female flies show excessive activity throughout the period of starvation when subjected to FS 8:16 (Suppl. Fig. 4A) in contrast with male flies that displayed reduction in SIH midway into the treatment (Figure 1c and c′). Mean phase during the FS treatment changes in female flies but they immediately revert to a value similar to pre-FS cycle when shifted to constant conditions (Suppl. Fig. 4B and C). However, despite SIH and its influence on the phases, the activity-rest rhythm continues to free run during the FS cycles. This suggests that in females like in males, masking and free-running components together regulate day-to-day phases.
If flies were consistently masking to lack of food by increasing their activity levels, we would expect a higher synchrony in inter-individual phases. However, we found that the inter-individual synchrony was lacking both in male and female flies irrespective of short or prolonged hyperactivity response. Moreover, this increased activity was not only variable across days but also variable across individuals. Therefore, unlike typically masked responses which consists of consistent all-or-none responses, in our paradigm, we observed a graded response to lack of food. This graded response may in part explain why inter-individual synchrony is absent despite occurrence of masking.
Food availability has been shown to affect activity patterns of many insects. Solitary bee species such as carpenter bees and orchid bees show foraging behavior, which is partially regulated by circadian clocks (Bloch et al., 2017). Social honey bees Apis mellifera entrain to food availability in their environment (Frisch and Aschoff, 1987) underlining the importance of circadian clocks in anticipating availability of food. Blood mealtimes are carefully phased to host availability in various hematophagous species such as bed bugs, kissing bugs, and mosquitoes. For example, bed bug Cimex lectularius are active during dawn, a time when the humidity is relatively high and hosts are resting (Barrozo et al., 2004). Similarly, kissing bug Triatoma infestans, the vector for Chagas disease shows peak locomotion, feeding, and carbon-dioxide sensitivity in the early night presumably when hosts are inactive (Barrozo et al., 2004). Flight activity and feeding/biting patterns in mosquitoes are influenced by many environmental factors apart from the internal clock mechanisms (Barrozo et al., 2004). However, these feeding patterns have been shown to change in nocturnal Anopheles spp from peak feeding occurring during the nighttime to a relatively earlier phase due to change in host availability as a result of interventions to prevent spread of malaria in some African countries (Reddy et al., 2011; Gatton et al., 2013; Sougoufara et al., 2017). Such flexible patterns are also observed in Culex pipiens when blood meals were restricted to a daytime window (Fritz et al., 2014). Ground beetle Feronia madida change their nocturnal activity patterns to diurnal activity patterns under starved conditions (Williams, 1959; Beck, 1980). Cockroach Periplaneta americana that have nocturnal activity and feeding show masking response to food availability when food is restricted to daytime; however, the activity is still higher during the night suggesting little change to overall activity-rest pattern (Beck, 1980). Therefore, non-alignment of food availability to an active state of the organism can influence the activity patterns and possibly the underlying clock. In addition, mice gauge the food reward-to-predation risk ratio and accordingly change their temporal niche depending upon the environmental conditions (van der Vinne et al., 2019). Thus, activity patterns may change from nocturnal to diurnal in response to change in food availability and predation pressure. We sought to ask if similar pressure in the form of restricted food access to a certain time of day can bring about a change in activity-rest of D. melanogaster. We found that while the clock does not entrain to the food availability cycle, it perceives these changes in food availability and accommodates them by adjusting its clock properties. Different extent of clock responses to food could be because of difference in amount of food required for sustenance and/or difference in natural history and ecology of these species.
It has been previously shown that starved flies feed more than fed controls upon re-supply of food. However, this increase is observed only in the first 30 min of food presentation post 24 h of starvation (Regalado et al., 2017). Starved flies linger on the food end of activity tubes significantly longer when presented with food after starvation as opposed to the controls that were fed throughout (Regalado et al., 2017). In our study, while we have not directly measured food intake, we observe a decrease in activity immediately after flies are presented with food tubes after at least 14 or 16 h of starvation (Figure 1b-c, right), suggesting that flies are near food thereby resulting in lower activity counts. Further studies that can simultaneously measure activity-rest rhythm and food intake will address how FS cycles can affect peripheral oscillators.
In mammals, anticipatory response to food availability suggests that circadian clocks are directly responsive to food availability. While the identity of such a clock (FEO) remains elusive to date, it is clear that FEO controls food anticipatory activity (Mistlberger, 1994; Stephan, 2002). In Drosophila, response to restricted food access is masked, which suggests a homeostatic control. Our study demonstrates that this masked response in the form of SIH indirectly affects the activity-controlling central clock. The physiological basis for this interaction between the homeostatic and clock components has not been studied so far. Future studies directed at understanding this interaction between clocks and a food homeostat will help to understand how food availability in the environment can shape the activity-rest patterns in animals.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304221111287 – Supplemental material for Effects of Food Availability Cycles on Phase and Period of Activity-rest Rhythm in Drosophila melanogaster
Supplemental material, sj-pdf-1-jbr-10.1177_07487304221111287 for Effects of Food Availability Cycles on Phase and Period of Activity-rest Rhythm in Drosophila melanogaster by Viveka Jagdish Singh, Sheetal Potdar and Vasu Sheeba in Journal of Biological Rhythms
Footnotes
Acknowledgements
We thank two anonymous reviewers for their valuable inputs on a previous version of the manuscript. We are grateful to Todd C Holmes for sharing anti-PER antibody originally generated by Jeff C Hall. We thank Srikant Venkitachalam for help with statistics, Chitrang Dani for comments on the manuscript, and other lab members for discussions. Sunil Kumar S for help with imaging. We also thank Rajanna Narasimhaiah and Muniraju Muniappa for technical assistance. This work was supported by intramural funds of Jawaharlal Nehru Centre for Advanced Scientific Research and a consumable grant from the Department of Biotechnology (DBT), Government of India, to V.S. (BT/INF/22/SP27679/2018).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Any underlying research materials related to this publication will be provided upon request by email to the corresponding author.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
