Abstract
Circadian rhythms, which respond to the day-night cycle on the earth, arise from the endogenous timekeeping system within organisms, called the “biological clock.” For accurate circadian rhythms, daily fluctuations in light and temperature are considered one of the important time cues. In social insects, both abiotic and biotic factors (i.e., social interactions) play a significant role in activity-rest rhythm regulation. However, it is challenging to monitor individual activity-rest rhythms in a colony because of the large group size and small body size. Therefore, it is unclear whether individuals in a colony exhibit activity-rest rhythms and how social interactions regulate their activity-rest rhythms in the colony. This study developed an image-based tracking system using 2D barcodes for Diacamma cf. indicum from Japan (a monomorphic ant) and measured the locomotor activities of all colony members under laboratory colony conditions. We also investigated the effect of broods on activity-rest rhythms by removing all broods under colony conditions. Activity-rest rhythms appeared only in isolated ants, not under colony conditions. In addition, workers showed arrhythmic activities after brood removal. These results suggested that a mixture of social interactions, and not light and temperature, induces the loss of activity-rest rhythms. These results contribute to the knowledge of a diverse pattern of circadian activity rhythms in social insects.
Most organisms show daily changes in various behaviors (Saunders, 2002), such as locomotor activity (Cymborowski, 1973), feeding activity (Frisch and Aschoff, 1987), mating (Groot, 2014), and egg hatching (Itoh and Sumi, 2000). In addition, physiological phenomena, such as body temperature (Refinetti et al., 1992), blood pressure (Floras et al., 1978), endocrine hormone release (Ungar and Halberg, 1962), and metabolic activity (Chew et al., 1965), fluctuate throughout the day (Glass, 2001). This daily rhythm is not a simple response to alternating daily changes in the external environment; instead, it has an endogenous basis. When organisms are settled in constant photoperiodic and temperature conditions (i.e., “free-run” conditions), circadian rhythms (~24 h cycles) arise from the endogenous timekeeping system of the organism, called the “biological clock.” Circadian rhythms allow the organism to anticipate and prepare for changes in physical states, depending on the day-night cycle (Sharma, 2003). For accurate circadian rhythms, endogenous oscillations are synchronized to external cues (zeitgebers or time givers), such as light, temperature, and feeding time (Aschoff, 1960; Refinetti and Piccione, 2005). However, little is known about how biological factors, such as ecological interactions within and among species, affect behavioral and/or circadian rhythms.
Studies have reported on social synchronization by which animals adjust their circadian rhythms in response to social signals (Eban-Rothschild and Bloch, 2012a; Favreau et al., 2009; Regal and Connolly, 1980). To increase mating opportunities and cooperative behaviors, especially in the absence of time cues, animals require synchronization of individual activity-rest cycles (Bloch et al., 2013; Davidson and Menaker, 2003; Eban-Rothschild and Bloch, 2012b; Regal and Connolly, 1980). Interactions among mature individuals are an important time giver for cavity-dwelling and social animals (Eban-Rothschild and Bloch, 2012b; Fuchikawa et al., 2016; Mistlberger and Skene, 2004). In addition, in humans, other mammals, and birds, parents lose their daily rhythms during the reproductive phase because of interactions with offspring (Bulla et al., 2016; Lyamin et al., 2005; Nishihara et al., 2002). Similarly, bee and ant nurses lose their daily rhythms (Bloch et al., 2013; Fujioka et al., 2017; Moore et al., 1998). Therefore, parent-offspring interactions are one of the strongest and most important social interactions in human and nonhuman animals for changing daily rhythms.
Social insects have a subterranean habitat and highly sophisticated social life. Therefore, they are an excellent model for investigating the effects of environmental and social factors on daily rhythms. The subterranean habitat is an “arrhythmic” habitat with few variations in light and temperature (Beale et al., 2016). In addition, individuals within a colony engage in several tasks, such as foraging and brood care, which depend on various social contexts and interactions. The daily rhythms of insects highly depend on social contexts. For example, foragers have strong circadian activity rhythms, whereas queens and nurse bees exhibit around-the-clock activity for egg-laying and brood care (Mildner and Roces, 2017; Moore et al., 1998; Sharma, Lone, Goel, and Chandrashekaran, 2004; Shemesh et al., 2007, 2010).
In ants, in addition to the brood effect on the activity-rest rhythm of workers, the behavioral rhythm seems to be highly sensitive for worker-worker interactions. When individual workers of D. cf. indicum sp. were isolated from a colony reared under a light-dark cycle and individual activity-rest rhythms were evaluated under constant dark (DD) or light (LL) conditions, workers exhibited clear activity-rest rhythms regardless of age (Fujioka et al., 2017, 2019). Experimentally manipulating worker-worker interactions led to decreased activity-rest rhythms in small groups (up to 5) of old (forager-age) workers (Fujioka et al., 2019). In contrast, young (nurse-age) workers exhibited clear activity-rest rhythms when 5 ants were grouped together, but grouping with brood or old workers decreased activity-rest rhythms. The results indicate that individual ants harbor circadian rhythms, but the expression of behavioral rhythms varies depending on castes and stages of interacting opponents. Thus far, the variability of rhythmic states in solitary and small ant groups with experimentally controlled interactions has been determined in D. cf. indicum. However, evaluating individual activity-rest rhythms under natural colony conditions and investigating how activity-rest rhythms are integrated into colony-level activities are important.
In social insects with a large colony size, monitoring individual activity-rest rhythms under colony conditions is challenging. Colony-level observations have focused on foraging activity, oxygen consumption, and temperature; however, a few studies have addressed individual activity separately (Crailsheim et al., 1996; Jürgen Stelzer et al., 2010; McCluskey and Brown, 1972; Moritz and Kryger, 1994). A classic measurement of individual locomotor activity includes sensing devices, using transducer of vibration or infrared (IR) light beam barriers, but the system is applied only under isolated conditions (Beer et al., 2016; Fuchikawa et al., 2014; Giannoni-Guzmán et al., 2014; Ingram et al., 2009; Sharma, Lone, and Goel, 2004; Spangler, 1972). Moreover, a radio-frequency identification system is commonly used, particularly for monitoring the foraging activity of a colony (Jürgen Stelzer et al., 2010; Nunes-Silva et al., 2019; Stelzer and Chittka, 2010). However, this method has some limitations, such as group size, detectable distance, and accuracy of individual identification. Recently developed digital tools, including image-based tracking, can help monitor ant behavior at a higher resolution and under more natural conditions (i.e., all members of a colony; Greenwald et al., 2015; Mersch et al., 2013).
In this study, we developed a tracking system using 2D barcodes for D. cf. indicum from Japan and measured locomotor activities of all colony members under colony conditions. The monomorphic ant D. cf. indicum from Japan is distributed in subtropical Okinawa, Japan. The focal species is monogynous, that is, colonies contain 30 to 200 workers and a single functional queen, called the gamergate, is present in each colony (Billen and Peeters, 1991). Monodomous nest habitats are open lands, such as grasslands at the forest edge (Fukumoto and Abe, 1983). This ant has been studied as a model system in various fields of study, including circadian rhythms (Fuchikawa et al., 2014; Miyazaki et al., 2020; Okada et al., 2017; Shimoji et al., 2020; Win et al., 2018). We also examined the effect of social interactions on activity-rest rhythms. From our previous observations of small group interactions (Fujioka et al., 2019), we hypothesized that individuals lose activity-rest rhythms under colony conditions because of a combination of interactions with the brood, between young and old workers, and within old workers. To test this hypothesis, we compared individual activity rhythms under colony conditions with those under isolated conditions and then analyzed the effect of broods on activity rhythms by removing all broods.
Materials and Methods
Insect and Colony Setup
We excavated 16 colonies of the ponerine ant Diacamma cf. indicum (the only Japanese Diacamma species that had been described as D. sp from Japan previously) in Sueyoshi Park (Naha), the Tamagusuku Youth and Children’s Center (Tamagusuku), and Kenmin-no mori (Onna), Okinawa, Japan, and maintained them in the laboratory in artificial plastic nests filled with moistened plaster. To minimize environmental effects (i.e., variability of receiving light intensity), we kept the experimental colonies under a 16/8-h day-night cycle from 7:00 a.m. to 11:00 p.m. at 25 °C and then transferred them to measurement conditions with constant dim-red light (i.e., imitating the all-dark [DD] free-run condition) inside the nests by covering the top of the nests with a red film. Reared colonies were fed chopped mealworms and crickets thrice a week. Each colony comprised a single queen and workers. Workers 0- to 30-d old stayed inside the nest and cared for the brood (i.e., eggs, larvae, and pupae), while workers older than 30 d started to forage outside the nest for various terrestrial invertebrates (Nakata, 1995).
The life span of individuals is approximately 1 year (Tsuji et al., 1996). To investigate the relationship between rhythmicity and age, we marked newly enclosed workers using enamel paint (TAMIYA, Shizuoka, Japan) at least once a month to record the age of each individual (Figure 1a). At this point, we examined all individuals to check if their marks had peeled or not.

Experimental outline and 2D barcode tag-based tracking system: (a) 2D barcode tags (2.0 × 2.0 mm2) were attached to each individual. (b) We studied 2 colony conditions: colony with the brood (days 1-3) and without the brood (days 5-7). All broods (eggs, larvae, and pupae) were removed on day 4 of the experiment from 2 colonies. (c) Experimental setup. The tag-based tracking system detected coordinates (x, y) and a direction of each individual every second. The square plastic box at the center is an artificial nest that was filled with moistened plaster. (d) The artificial nest was a square box that was filled with a moistened plaster. Ants were prohibited from climbing the walls of nest and foraging arena by coating the lateral sides of the containers with fluoropolymer resin (Insect-a-Slip Insect Barrier [Fluon], BioQuip, CA, USA).
Experimental Conditions
We studied 3 conditions: (1) solitary (without the brood), (2) colony with the brood, and (3) colony without the brood (brood removal). Workers less than 30 d old were defined as young workers or nurse-age workers, while those older than 30 d were defined as old workers. Under solitary conditions, we randomly selected 1 individual from each colony and measured its locomotor activity. Individuals of isolated condition were obtained from the colonies different from those of colony conditions (young workers: n = 26 from 5 colonies; old workers: n = 21 from 4 colonies). Since the removal of a portion of workers from a colony might cause behavioral changes in the rest of the workers, we avoided this potential confound effect by using different colonies.
We used naive colonies under colony with the brood (n = 8 colonies [A-H]), and some of them (n = 2 colonies) were subjected to brood-removal treatment. Under colony with the brood condition, we investigated individuals’ activity-rest rhythms under laboratory conditions. All 8 colonies had a queen, workers, and the brood (eggs, larvae, and pupae) each. We conducted 3 d of recording (Figure 1b). During the observation period, 1 or 2 crickets were replenished at the same time every day (colonies A and B: 1800; colonies C-H: 1100), and water was provided at all times. Each colony comprised 88 to 194 individuals, including the queen, but we could not obtain data of some individuals because of the loss of tags, death, or tag detection insufficiency (see the “Data Processing and Checking Errors” section). Table 1 shows other detailed information about colonies.
Colony information: The colony size is the number of individuals in the colony.

The brood includes eggs, larvae, and pupae. We fed the ants at the same time every day.
For brood removal, we used 2 (A and B in Table 1) of 8 colonies analyzed under the with-brood condition. After recording of the 2 colonies with brood condition for 3 d, we removed all broods (eggs, larvae, and pupae) from the nests of colonies A and B on day 4. To avoid the disturbing effect of the treatment, we performed brood removal under dim-red light and opened the nest shortly thereafter. The colonies were kept under constant dim-red light throughout the experiment. From day 5 (at least 8 h after treatment), we started 3 d of recording of brood-removal treatment (Figure 1b). To investigate the effect of broods, we compared rhythmicity and total activity between colonies under with-brood and brood-removal conditions.
Recording and Analyses of Activity Rhythms in Colonies
Isolated condition was identical to the experimental setup of Fujioka et al. (2017). Briefly, we introduced 1 individual into an 8 × 8 cm2 square plastic container and continuously recorded its locomotor activity for 3 d (60 s × 60 min × 24 h × 3 d) at 25 ± 1 °C under dim-red light (DD). We recorded the experimental area using a Logicool HD Pro Webcam C920t web camera (Hitachi, Ibaraki, Japan) and used a homemade code based on the OpenCV computer vision library (Intel Corporation, mountain View, CA, USA), to track each individual’s position. We also obtained the trajectory of movement (x(t), y(t)) every second.
For colonies under with-brood and brood-removal conditions, we applied the image-based tracking system with tags to identify individuals. To do so, we attached a 2.0 × 2.0 mm2 BugTag 2D barcode tag (Robiotech, Hollywood, MD, USA) to each individual’s dorsal thorax using multipurpose, instant liquid glue (Product No. 7004; 3M, Saint Paul, MN, USA; Figure 1a and Suppl. Fig. S1); the tags and glue had no effect on mortality (Suppl. Table S1; Abe and Fujioka, 2017). We photographed the nest and foraging area every second using a Grasshopper3 charge-coupled device camera (GS3-U3-123S6C-C, 3000 × 4000 pixels; FLIR Systems, Wilsonville, OR, USA) for 3 d (60 s × 60 min × 24 h × 3 d) at 25 °C ± 1 °C under constant dim-red light. Barcode-labeled individuals were identified using a BugTag commercial vision-based tracking system (Robiotech; Figure 1c). We again obtained the trajectory of movement (x(t), y(t)) every second. Experiment nests were filled with moistened plaster (20.0 × 10.0 × 5.0 cm3), and each nest had 1 entrance of 1.0 cm diameter to allow the ants to forage (37.0 × 25.0 × 11.5 cm3) (Figure 1d).
Data Processing and Checking Errors
Unfortunately, we could not track some individuals because of the detachment of tags or death; these individuals were omitted from analysis. For others, data with missing tag detection time (>50%) were also omitted from analysis. Therefore, the number of individuals analyzed was different from the actual colony size (Table 1). The reasons included image resolution (4000 × 3000) and frame rate (1 fps) for a large tracking area. In addition, focal individuals had long legs and long antennae that sometimes covered other individuals’ tags, decreasing the tag detection rate. In addition, the focal ant often shows self-grooming behavior with a sideways posture, which prevented dorsal tag detection (H. Fujioka, personal observation).
We randomly selected 1443 discontinuous data points and manually checked the maximum movement velocity (distance/time = 36.7 mm/s). On the basis of this maximum movement velocity, we decided a threshold of 40.0 mm for excluding tracking errors. If the velocity was >40.0 mm/s, we deemed the point an error and corrected the coordinate (i.e., tag location) as not applicable (NA) data; the mean of error rates of all data was 1.39%. After processing, we filled the NA data at time t with previous coordinates (t − 1) assuming that each individual stayed in the same position.
Statistical Analysis
All statistical analyses were performed in R v.3.5.0. We converted the position coordinates (xt, yt) of the worker at every second to the amount of locomotor activity at every minute. We performed χ2 periodogram analysis for the circadian activity-rest rhythm (Fujioka et al., 2017; Sokolove and Bushell, 1978). The activity-rest rhythm power was defined as the maximum difference between χ2 and the significance threshold line at p = 0.05 (Fujioka et al., 2017; Klarsfeld et al., 2003). The power was high when the activity-rest rhythms were strong; in contrast, low or negative power indicated a weak or statistically arrhythmic state, respectively. The total activity was defined as the total movement distance during the recording period (3 d). We performed the Steel-Dwass test to compare isolated and colonies with brood conditions. We also performed one-way analysis of variance (ANOVA) and a paired t test when data followed a normal distribution with homogeneous variance (Bartlett’s test). In addition, we analyzed the difference of age among colonies with ANOVA. We used linear models to evaluate the effects of age and colony size on power or total activity. The response variables were power (a continuous value representing the activity-rest rhythm strength) and total activity, while the explanatory variables were age and colony size.
Results
Under isolated conditions, the power of most young workers and old workers was >0 (M ± SD, isolated young: 202.3 ± 186; isolated old: 144.8 ± 147.8), indicating that both young workers and old workers show activity-rest rhythms (Figure 2). In contrast, under colony with brood conditions, individuals almost completely lost activity-rest rhythms (Figure 2, M ± SD, young: −64.4 ± 43.1; old: −47.8 ± 50.9, Steel-Dwass test, p < 0.01). Arrhythmic activity patterns were observed in colony-level activities (i.e., averaged activity; Figure 3). Although there was no significant difference between young workers’ and old workers’ powers under isolated conditions (Steel-Dwass test, p = 0.96), the power of old workers was greater than that of young workers under the colony conditions (Steel-Dwass test, p < 0.01). Total activity of isolated young workers was less than that of workers under the colony condition (Suppl. Fig. S2, M ± SD, isolated young: 290.5 ± 165.7; young: 450.9 ± 266.3; old: 407.4 ± 240.1, Steel-Dwass test, p < 0.01). Total activity of isolated old workers was not significantly different between other conditions (Suppl. Fig. S2, M ± SD, isolated old: 344.5 ± 276.5, Steel-Dwass test, p > 0.05).
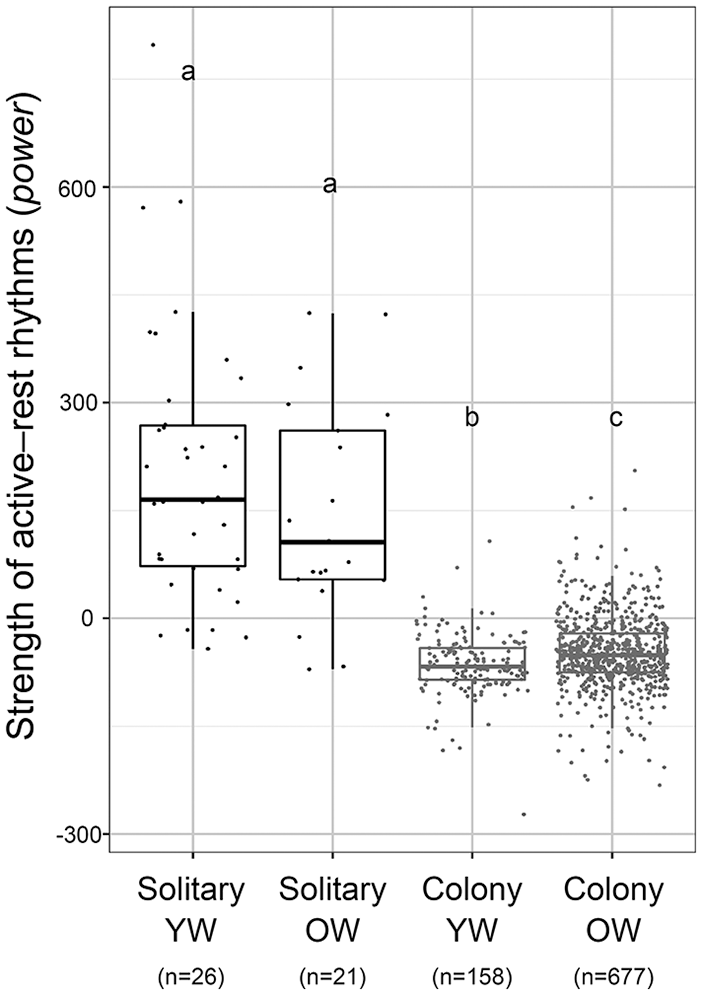
Activity rhythm under isolated conditions (solitary) and colony with the brood. Abbreviations: YW = young worker; OW = old worker. Both young workers (<30 d) and old workers (>70 d) under colony condition do not show activity-rest rhythms, whereas young workers and old workers under isolated conditions show clear activity-rest rhythms. The sample size is shown below the label of the x-axis. Different letters above the boxes indicate significant differences (Steel-Dwass test).
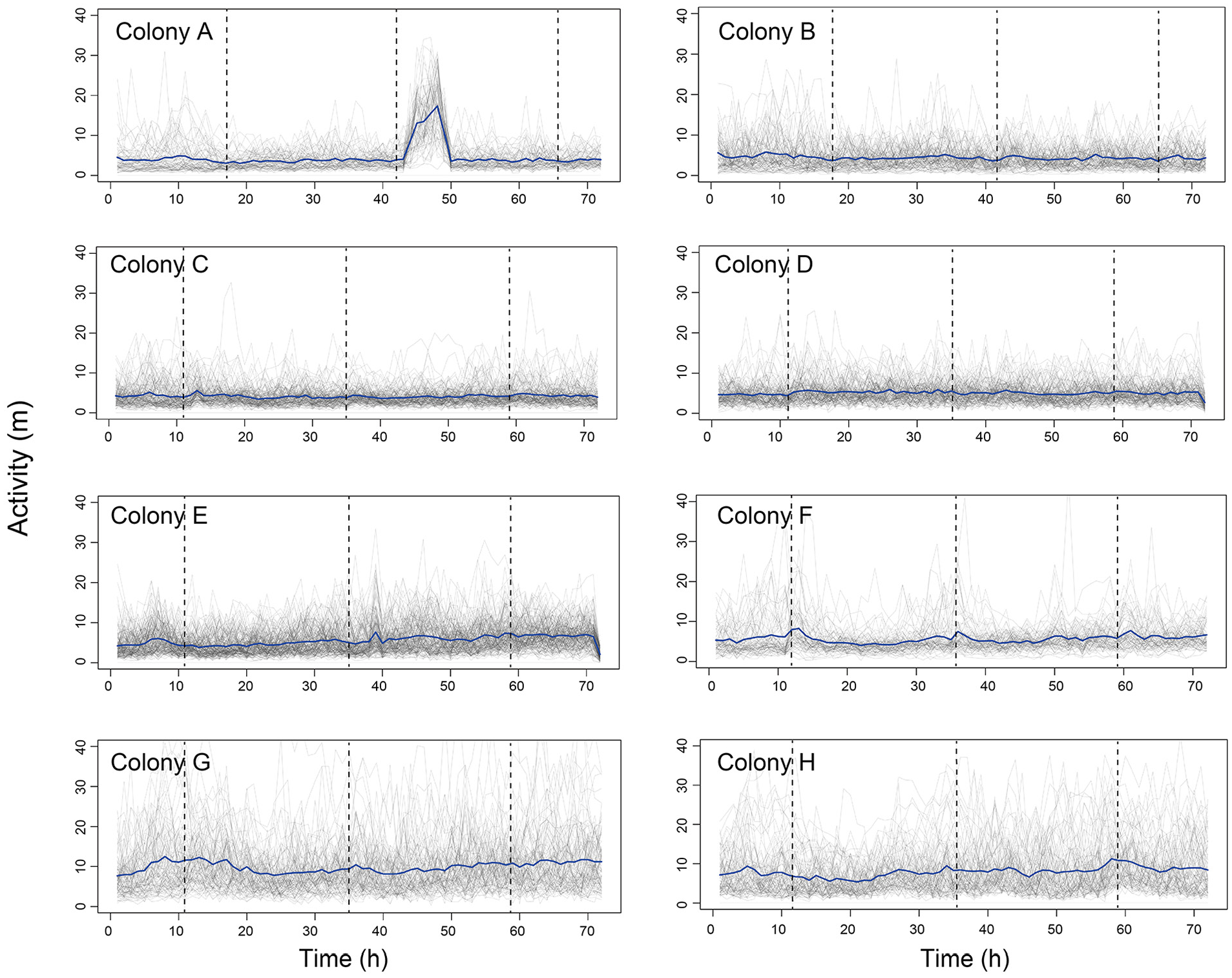
Time-series for colony- and individual-level activities for 3 d. The x-axis represents time (h), and the y-axis depicts the activity level (m/h). Thick (blue) and thin (gray) lines represent colony- (i.e., activity averaged over all individuals) and individual-level activity, respectively. Dashed vertical lines indicate feeding times (1800: colonies A and B; 1100: colonies C-H).
Under colony with brood conditions, power was significantly affected by age but not by colony size (Suppl. Fig. S3a, Suppl. Table S1, linear model, F2.824 = 12.98; age: p < 0.01; colony size: p = 0.64), while activity was strongly affected by both age and colony size (Suppl. Fig. S3b, Suppl. Table S1; linear model, F2.824 = 7.816; age: p < 0.01; colony size: p < 0.01). Midsize colonies showed high activity (Suppl. Fig. S3b). The worker age significantly differed across colonies (ANOVA, F7.189 = 27.3; p < 0.01), so we separately analyzed the colonies to detect the effect of age on behaviors. In 3 of the 8 colonies, we found a significantly positive relationship between power and age (Figure 4 and Suppl. Table S2, linear regression) and a positive relationship between total activity and age in 7 colonies (A-C, E-H) except colony D (Figure 5 and Suppl. Table S2, linear regression).
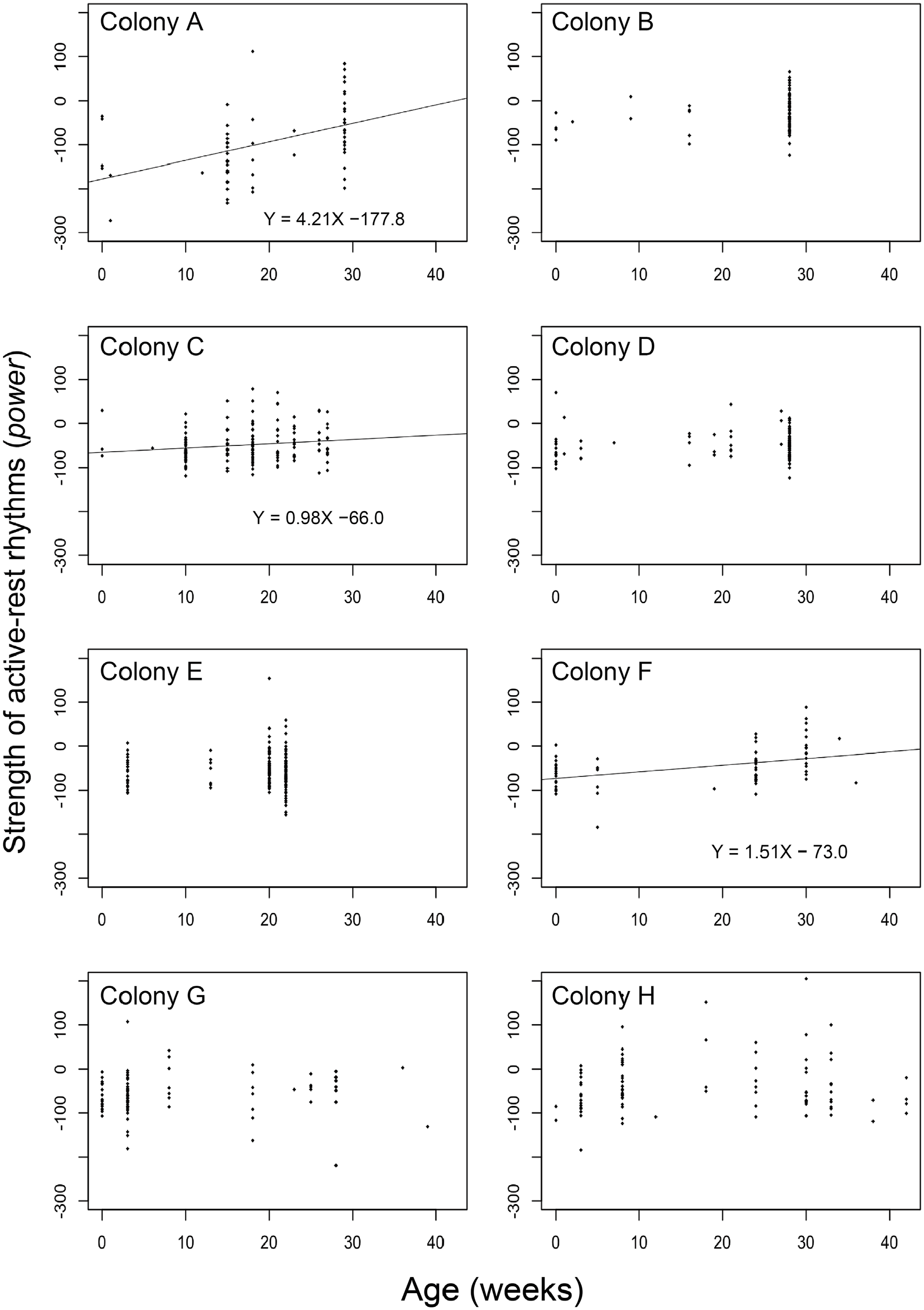
Relationship between the power (strength) of activity-rest rhythms and age (weeks) in colonies A to H. The x- and y-axes represent the circadian rhythm power and age (weeks), respectively. The regression line represents the statistically significant relationship between power and age.

Activity levels increase with age. The x- and y-axes represent the total activity throughout the experimental period (m/3 days) and age (weeks), respectively. Colony D had no significant relationship between total activity and age.
Under brood-removal conditions, some individuals showed higher power, while others showed lower power after brood removal (Suppl. Fig. S4). On average, both power and total activity after brood-removal treatment were significantly lower than those of before brood removal (Figure 6; paired t test; power: t = 2.3, df = 146, p = 0.02; activity: t = 5.2, df = 146, p < 0.01). Unexpectedly, workers in the brood-removal condition also showed arrhythmic activities.

Brood-removal treatment decreases power and total activity. Upper and lower panels represent the activity-rest rhythm power (a) and total activity per 3 d (b). There were significant differences between control- and brood-removal treatment (paired t test).
Discussion
The daily rhythm is ubiquitous in organisms; however, Diacamma workers did not show activity-rest rhythms under the laboratory colony conditions. In contrast, Diacamma workers showed activity-rest rhythms in isolated conditions. This suggests that our focal individuals inherently harbor the circadian rhythm. Given the characteristic ant ecology, arrhythmic activities might be an adaptation for “arrhythmic” subterranean habitats and the ants’ social life. Considering the difference between the isolated and the colony conditions, we conclude that social interactions induce the loss of activity-rest rhythms.
In isolated individuals, interaction with the brood decreases the rhythmicity of nursing ants (Fujioka et al., 2017). In addition, interactions with the brood or old workers decrease activity-rest rhythms of young workers in small groups (Fujioka et al., 2019). Therefore, the arrhythmic activities of young workers can be attributed to interactions with the brood and/or old workers under colony conditions. Similarly, old workers show arrhythmic activities in small groups (Fujioka et al., 2019). Arrhythmic activities of old workers can be attributed to interactions with other workers under colony conditions. Our result (Suppl. Fig. S2) and previous studies suggest that there is no relationship between the power of rhythms and total activity levels (Fujioka et al., 2017, 2019). Thus, activity level does not account for the low power of rhythmicity. Therefore, we concluded that endogenous activity rhythm emerges when they are isolated from society, but a mixture of social interactions induces them to lose activity-rest activities.
In laboratory-rearing nests under constant dark conditions, foragers are found throughout the day. Both colony- and individual-level foraging activity was arrhythmic under colony conditions. Our results support the previous evidence of changing behavioral rhythms, depending on the social context (Fuchikawa et al., 2014; Fujioka et al., 2019). However, the result that foragers lose their circadian rhythms under colony conditions is inconsistent among studies. In social insects, such as bees and ants, foragers have precise daily rhythms (Bloch et al., 2001; Bochynek et al., 2017; Crailsheim et al., 1996; Hoenle et al., 2019; Jürgen Stelzer et al., 2010; Lei et al., 2019; McCluskey, 1987; McCluskey and Neal, 1990; Medeiros et al., 2014; Mildner and Roces, 2017; Moore et al., 1998; Orivel and Dejean, 2002; Passera et al., 1994; Raimundo et al., 2009; Retana et al., 1992). Several possible reasons exist for this contradiction. First, previous studies did not investigate the activities of foragers inside a nest (Hoenle et al., 2019; Jürgen Stelzer et al., 2010; Lei et al., 2019; McCluskey, 1987; Medeiros et al., 2014; Moore et al., 1998; Orivel and Dejean, 2002; Passera et al., 1994; Raimundo et al., 2009; Retana et al., 1992). Foragers may be active during the nonforaging time inside the nest. Second, some ant species employ cooperative transport (Feinerman et al., 2018; McCreery and Breed, 2014), whereas others do not. Cooperative foraging requires that workers are active at the same time. By contrast, our focal ant is a solitary hunter (Win et al., 2018); workers can hunt prey and bring it into the nest alone (Uematsu et al., 2019). Such a difference in foraging strategies may change the foraging schedule. In addition, our focal ant is distributed in the subtropical island of Okinawa, Japan. There is a small temperature difference between day and night (approximately 5 °C; Win et al., 2018). The relatively warm night would allow all-day foraging. Last, their feeding habits can affect their activities. Most bees are specialized in floral nectar and pollen, and therefore need to forage during flowering. Therefore, for bees, foraging is reasonable during daytime. In contrast, temporal changes of resource availability are not straightforward for ants, who can be generalist or specialist predators, scavengers, or omnivores (feeding on nectars and preying on other arthropods; Cerdá and Dejean, 2011; Hoenle et al., 2019). In fact, some ants, such as Anoplolepis gracilipe (Chong and Lee, 2009), Crematogaster matsumurai (Harada, 2005), Diacamma (Win et al., 2018), Formica (Finnegan, 1973; Rosengren, 1977), Linepithema humile (Abril et al., 2007), and Solenopsis invicta (Lei et al., 2019), do not have clear circadian foraging activities. Unlike the foraging time of bees, there might be no specific foraging time for generalist arthropod predators. Our focal species feeds on various arthropods (Win et al., 2018). This generalist predator biology could be associated with the all-day-long activities of foragers.
Interactions with the brood (i.e., care-taking) can induce arrhythmic activities in bee and ant workers (Bloch and Robinson, 2001; Fujioka et al., 2017; Nagari et al., 2019). In our focal species, isolated and grouped nurses (up to 5 individuals) show circadian rhythms when freed from the brood, indicating that activity-rest rhythm is the default state (Fujioka et al., 2019). Therefore, nurses may exhibit activity-rest rhythms under the brood-removal environment. Unexpectedly, the rhythmicity for most workers was arrhythmic after brood removal under colony conditions. Although the power of several young workers increased after brood-removal treatment, this pattern is inconsistent among individuals and colonies. The variation between colonies could be due to differences in age structure among colonies. Moreover, young-old worker interactions may induce arrhythmic activities after brood removal, that is, when lacking worker-brood interactions. The detailed mechanism underlying decreased activity-rest rhythms requires further investigation of interactions among workers. In addition to activity-rest rhythms, total activity levels decrease in both colonies before and after brood-removal treatment. It is less likely that decreased activity is affected by hunger. Brood removal decreases the amount of work, and workers may become inactive in response to the decreased workload.
Here, we discuss why many workers exhibit arrhythmic activity. First, the lack of rhythmicity might be linked to the “arrhythmic” subterranean habitat. In such habitats, individuals do not need to keep daily rhythms inside colonies. That could be enough if foragers respond to outside conditions, such as temperature and rain, only when necessary. Second, if an arrhythmic activity is related to continuous task performance, it leads to an increase in colony productivity. For instance, when workers are engaged in brood care all day, brood survival increases. Similarly, when foragers collect food all day, food intake is maximized. However, sleep-loss or foraging task itself has several costs (Vaccaro et al., 2020). To understand the significance of arrhythmic activity, we need to investigate the benefit and cost of the arrhythmic activity.
In insect societies, social synchronization of activity is believed to be important for temporal coordination and efficient colony-level performance (Bloch et al., 2013). Social synchronization occurs when individuals have rhythms of self-sustained periodic oscillators. Thus, we cannot conclude that the result of arrhythmic activities indicates a lack of social synchronization. Moreover, we do not believe that ant colonies lost the ability to cooperate even if individuals showed arrhythmic activities. Cooperative behavior can be achieved by reacting to a stimulus, such as pheromones and physical contacts. Further studies that investigate the reaction to the stimulus and evaluate performance in arrhythmic ants at the colony level are needed.
Ants have diverse ecological traits, such as colony size, habitat types, nest types, caste, number of queens, diurnal/nocturnal, feeding type, and foraging strategy (Crall et al., 2018; Hölldobler and Wilson, 1990). We speculate some ecological features that may affect social rhythms of ants. First, our focal ant is monomorphic with the smaller degree of behavioral specialization. The pattern of activity-rest rhythms could differ in physically polymorphic species. In Camponotus compressus, the media worker (middle size; 11-16 mm long) displayed both nocturnal and diurnal patterns under the light-dark cycle (Sharma, Lone, Mathew, et al., 2004). The caste may accomplish all-day-long working by having different activity time. Although insufficient evidence exists, a specialization to activity patterns may be coupled with morphological specialization in other species. Second, Diacamma have a relatively small colony size (30-300 ants; Billen and Peeters, 1991; Burchill and Moreau, 2016). In larger colonies, the activity may be more synchronized between individuals. We hypothesized that interaction rates would be larger if the colony size is larger. If the interaction becomes strong, synchronization between individuals is likely to occur (Pikovsky et al., 2002). Investigating the relationship between ecological traits and activity-rest rhythms will bring a fundamental understanding of how social regulation on activity-rest rhythms has evolved in response to changes in ecological traits.
We succeeded in automatically tracking the locomotor activities of most colony members. However, it should be noted that current experimental condition (i.e., the constant temperature and dim-red light) differs from the natural nest condition. Several abiotic cycles and a fluctuation of resource availability or predators exist in the wild. It is unclear how the ants integrate both abiotic and biotic information and coordinate their activity-rest rhythms within the nest. Our future research endeavor would be to understand the regulation of activity-rest rhythms using manipulation experiments by controlling light and temperature. In addition, we can develop our experimental system to identify several interactions, such as physical contact and trophallaxis, and investigate how the actual change of interaction regulates activity-rest rhythms in ants. This will be the subject of further studies.
Conclusion
The activity-rest rhythms of ants are regulated by a mixture of worker-brood and worker-worker interactions in the colony. For an in-depth understanding of social chronological organization, future studies should incorporate the dynamics of social interactions. Given that ants have diverse ecological features (e.g., nesting habitats, geographical variations, diets, and foraging strategies), many more ant species might have arrhythmic colonial lives, and comparisons of different ant species should be an interesting topic. By integrating laboratory and field studies (Denlinger et al., 2017; Helm et al., 2017), the chronobiology of ants will provide new insight into the temporal organization of animals living in groups.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304211002934 – Supplemental material for Individual Ants Do Not Show Activity-Rest Rhythms in Nest Conditions
Supplemental material, sj-pdf-1-jbr-10.1177_07487304211002934 for Individual Ants Do Not Show Activity-Rest Rhythms in Nest Conditions by Haruna Fujioka, Masato S. Abe and Yasukazu Okada in Journal of Biological Rhythms
Footnotes
Acknowledgements
Many thanks to S. Hakataya and K. Sakiyama for helping with the ant-keeping work. We also thank J. Uematsu for kindly supporting us with ant excavation. We are grateful to anonymous referees for very helpful comments on the manuscript. This study was funded by JSPS KAKENHI Grant number JP18J13369, JP20J01766 to HF, JP17K19381, JP18H04815 to YO, and JP15H06830 to MSA and Grant in Scientific Research on Innovation Areas “Integrative Research Toward Elucidation of Generative Brain Systems for Individuality” JP17H05938 and JP19H04913 from MEXT to YO.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Note
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
