Abstract
Internal circadian clocks organize animal behavior and physiology and are entrained by ecologically relevant external time-givers such as light and temperature cycles. In the highly social honey bee, social time-givers are potent and can override photic entrainment, but the cues mediating social entrainment are unknown. Here, we tested whether substrate-borne vibrations and hive volatiles can mediate social synchronization in honey bees. We first placed newly emerged worker bees on the same or on a different substrate on which we placed cages with foragers entrained to ambient day-night cycles, while minimizing the spread of volatiles between cages. In the second experiment, we exposed young bees to constant airflow drawn from either a free-foraging colony or a similar-size control hive containing only heated empty honeycombs, while minimizing transfer of substrate-borne vibrations between cages. After 6 days, we isolated each focal bee in an individual cage in an environmental chamber and monitored her locomotor activity. We repeated each experiment 5 times, each trial with bees from a different source colony, monitoring a total of more than 1000 bees representing diverse genotypes. We found that bees placed on the same substrate as foragers showed a stronger phase coherence and a phase more similar to that of foragers compared with bees placed on a different substrate. In the second experiment, bees exposed to air drawn from a colony showed a stronger phase coherence and a phase more similar to that of foragers compared with bees exposed to air from an empty hive. These findings lend credence to the hypothesis that surrogates of activity entrain circadian rhythms and suggest that multiple social cues can act in concert to entrain social insect colonies to a common phase.
Keywords
Most studies on the entrainment of endogenous circadian clocks have focused on photic entrainment, which is considered evolutionarily ancient and the most important time-giver to circadian clocks. In addition to light, the sun also produces daily fluctuations in ambient temperature that can effectively entrain circadian clocks of many organisms (Sharma and Chandrashekaran, 2005; Albrecht, 2012). Nonphotic, nonthermal time-givers have received relatively little attention. Recent studies have advanced the importance of entrainment by feeding time that may act on clocks in tissues other than the central light-entrained clock (e.g., the SCN in mammals; Mendoza, 2007).
A somewhat more controversial line of research relates to the importance of social entrainment. Although observations consistent with entrainment by social interactions have been reported for diverse animal species, there is no clear relationship between the level of sociality and the efficacy of social entrainment (reviewed in Favreau et al., 2009; Castillo-Ruiz et al., 2012; Eban-Rothschild and Bloch, 2012b; Bloch et al., 2013). Some social animals such as Mongolian gerbils, sugar gliders, and common marmosets could not be effectively entrained by even intensive social interactions such as contacts with receptive females or aggressive opponents. On the other hand, social influences on activity rhythms have been reported for species that are not considered social, such as the fruit fly Drosophila melanogaster (Levine et al., 2002; Lone and Sharma, 2011; Bloch et al., 2013; Castillo-Ruiz et al., 2012). An additional difficulty is that no neuronal or molecular pathway transmitting social stimuli to the core clock system has been described to date. Rather, it has been suggested that social influences on the mammalian clock are mediated by changes in arousal state, by learning processes associating circadian time with specific actions, or by social gating of the time or pattern of exposure to photic time-givers (Reebs, 1989; Amir and Stewart, 1996; Mistlberger and Skene, 2004). The best evidence for social entrainment is found in cavity-dwelling social animals such as bats, ants, and bees, in which at least some individuals do not experience ambient conditions directly but rather rely on information received from group mates that experience the outside environment (Regal and Connolly, 1980; Favreau et al., 2009; Eban-Rothschild and Bloch, 2012b; Bloch et al., 2013). In insect societies, it is thought that social synchronization of activity time is essential for temporal coordination and efficient colony-level performance. For example, honey bee nectar receivers and foragers need to act in a concerted manner, and this can be achieved by means of social synchronization (reviewed in Sharma, 2003; Bloch et al., 2013; Eban-Rothschild and Bloch, 2012b).
We study the highly social (“eusocial”) western (“European”) honey bee, Apis mellifera, which has been relatively well studied in terms of both social behavior and circadian rhythms. The organization of work in honey bee colonies relates to the age of worker bees. Young workers typically stay in the constantly dark and tightly thermoregulated cavity of the hive, in which they typically care for (“nurse”) the brood. Older workers typically forage outside the hive for food and other resources (reviewed in Robinson, 1992). Foragers are diurnal and show a sleeplike behavior at night, whereas nurses typically tend the brood around the clock and show an intermitted sleep pattern (Eban-Rothschild and Bloch, 2012a; Bloch, 2010). Although around-the-clock active nurses typically do not show overt circadian rhythms in locomotor activity or whole-brain clock-gene transcript abundance, there is evidence that they do have functional and entrainable circadian clocks. First, nurses that are removed from the hive and isolated individually in a cage rapidly switch to activity and brain gene expression with robust circadian rhythms that are in phase with ambient day-night cycles (Shemesh et al., 2007, 2010). Second, the expression levels of more than 150 brain transcripts vary with a 24-h cycle (Rodriguez-Zas et al., 2012). Third, recent immunocytochemical studies show that the levels of the clock protein amPeriod and the circadian neuropeptide Pigment Dispersal Factor (PDF) cycle with a similar phase and amplitude in brain circadian neurons of foragers that show, and nurses that do not show, circadian rhythms in locomotor activity (Fuchikawa et al., 2017; Beer et al., 2018).
Nest bees as young as 2 days of age can be socially entrained to the colony cycle. Entrainment is effective for caged bees deprived of access to the hive entrance and direct contact with other individuals in the colony (Bloch et al., 2013; Fuchikawa et al., 2016). Thus, social entrainment cannot be explained by gating the time of exposure to light. Remarkably, social entrainment could override photic entrainment; nest bees confined to the inner dark nest cavity and experiencing conflicting photic and social environmental cycles were entrained to the social cycle (Fuchikawa et al., 2016).
Here we test the hypothesis that surrogates of forager activity act to synchronize circadian rhythms of individual bees and entrain the colony to a common phase (Bloch et al., 2013). We focused on 2 important surrogates of bee activity: substrate-borne vibrations and hive volatiles. Honey bees, as well as other social insects, use vibrations for intracolony communication (reviewed by Hunt and Richard, 2013). Volatile pheromones and other hive odors are also well established for their pivotal and diverse communication functions in insect colonies (Wilson, 1971; Conte and Hefetz, 2008; Leonhardt et al., 2016). The hypothesis that chemical cues affect social synchronization was supported by evidence that allowing airflow between small groups of bees facilitates their synchronization to a common circadian phase (Moritz and Kryger, 1994). There is also evidence that hive CO2 levels vary over the day (Ohashi et al., 2009; Murphy et al., 2015; Edwards-Murphy et al., 2016), and in other insects, there is evidence consistent with the premise that CO2 can entrain circadian rhythms (Nicolas and Sillans,1989).
To test the hypothesis that substrate-borne vibrations generated by forager activity can entrain circadian rhythms in locomotor activity, we analyzed the circadian phase of newly emerged bees placed on the same or on different substrate as foragers. To test the hypothesis that volatile cues can mediate social synchronization, we exposed newly emerged bees to a constant air flow drawn from either a hive housing a free-foraging colony or a similar hive but with no bees. Our findings show that both vibrations generated by forager activity and volatile cues from a free-foraging colony can entrain circadian rhythms in locomotor activity in young worker bees. These findings lend credence to the hypothesis that surrogates of forager activity mediate social synchronization in honey bees.
Materials and Methods
Bees
Honey bees were maintained according to standard beekeeping techniques at the Bee Research Facility at the Edmond J. Safra campus of the Hebrew University of Jerusalem, Givat-Ram, Jerusalem, Israel. The honey bees in our apiary were derived from a mixture of subspecies typical to Israel. To obtain newly emerged bees, we removed honeycomb frames with emerging worker pupae, brushed off all adult bees, and immediately transferred each frame into a separate lightproof container. We placed the frames in an incubator (33°C ± 1 °C, 60% relative humidity [RH] ±5%) for the bees to emerge. The emerging bees were collected within 2 h after emergence under dim red light (DD; using Edison Federal EFEE 1AE1 Deep Red LED; mean wavelength = 660 nm, maximum and minimum wavelengths = 670 and 650, respectively) to avoid influences of light on the circadian system (e.g., photic entrainment).
General Procedure
Figure 1A summarizes the general experimental outline. On day 1, we collected newly emerged worker bees and transferred them to Libfield wooden cages (12 × 8 × 4.5 cm) with glass covers in which they were exposed to the tested environmental cues. On day 6, we collected samples of bees experiencing the various treatments as well as foragers that served as a control group. We isolated each focal bee in a monitoring cage and monitored locomotor activity for at least 6 successive days under dim red light and constant lab conditions (Fig. 1A). We used the locomotor activity data to determine the circadian phase of each bee and assess social synchronization among bees subjected to the same treatment (e.g., colony volatiles or vibrations).
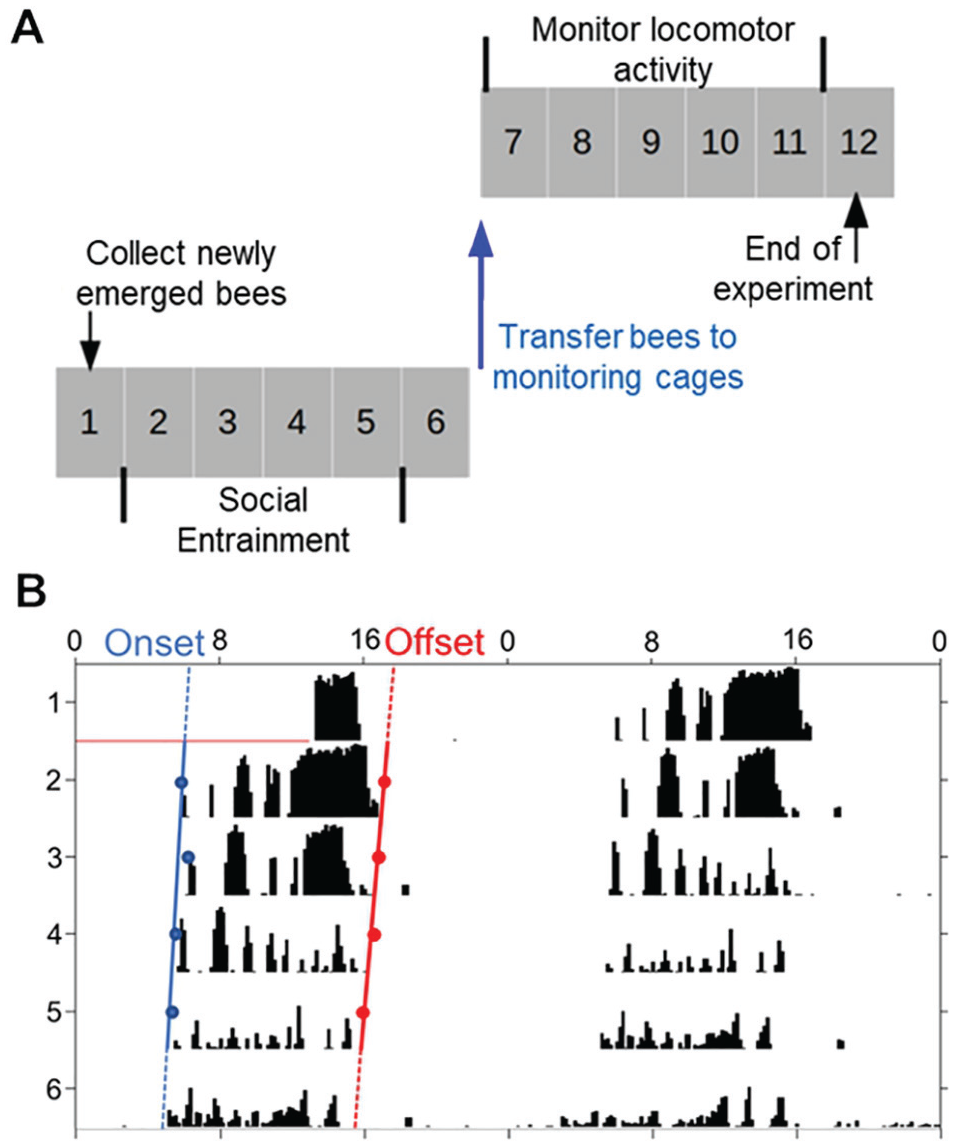
Experimental procedures. (A) General experimental outline. The numbers depict the days of the experiment, and the gray shading indicates that the bees were kept under dim red light. On day 1, we collected the bees and transferred them to cages in the lab, in which they were exposed to the tested signals. On day 6, each focal bee was collected and transferred to an individual monitoring cage in which her locomotor activity was automatically monitored under constant conditions for 6 successive days or more. (B) Representative double-plotted actogram of an individually isolated bee. The y-axis shows days in the monitoring cage, and the x-axis the time of day, double plotted for easier visual detection of rhythms. The height of the black bars within each day corresponds to the level of locomotor activity in a 10-min bin, determined by distance moved in pixels. The blue and red dots show the estimated times for the onset and offset of activity for each day, respectively. Linear regression models are fitted to these points, and the phase is determined based on the extrapolation of the regression lines on the first monitoring day (day 7 in the experiment; designated by the terms “onset” and “offset”). Color versions are available online.
Monitoring and Analyzing Locomotor Activity
We placed each bee individually in a monitoring cage made from a modified Petri dish (diameter = 90 mm) provisioned with ad libitum sugar syrup (50% w/w) and pollen. The monitoring cages with the bees were placed in a tightly regulated environmental chamber (29 °C ± 1 °C, 55% ± 5% RH). The chamber was illuminated with dim red light (Edison Federal EFEF 1AE1 Far [Cherry] Red LED; mean wavelength = 740 nm, maximum and minimum wavelengths = 750 and 730, respectively). Locomotor activity (measured as the number of pixels traveled over a time unit on the camera field of view) was recorded automatically at a frequency of 1 Hz with the ClockLab data acquisition system (Actimetrics Inc., Evanston, IL, USA). Our monitoring system was composed of 4 infrared light-sensitive black-and-white cameras (Panasonic WV-BP334, 0.08 lux CCD video cameras, or Sentech STC-MB33USB mini USB video cameras with computer TZ32910CS-IR lenses) and a high-quality monochrome image acquisition board (IMAQ 1409, National Instruments, Austin, TX, USA) or a National Instruments USB-6501 interface. Each camera recorded activity in 30 arenas (i.e., cages) on a single tray. Four cages, 1 on each tray, were left empty as controls, recording background noise.
For the analyses of circadian rhythms, we used the ClockLab circadian analyses software package (Actimetrics). We used the χ2 periodogram analysis with 10-min bins to generate actograms for the analyses of circadian rhythms. As a proxy for the strength of circadian rhythms, we used the Power, which was calculated as the height of the periodogram peak at a significance threshold greater than p = 0.01 (for more details, see Yerushalmi et al., 2006). For each day, we calculated the onset and offset of the daily bout of activity. The precise timing of the onset or offset was defined as at least 3 consecutive 10-min bins each with activity reaching at least 10% of the maximal activity per bin during this day. An additional requirement was a period of at least 5 h of reduced activity between the offset and the following onset (as described in Fuchikawa et al., 2016). We used the ClockLab software to fit the linear regression models passing through the onset or the offset points of at least 4 consecutive days. The extrapolations of these regression lines on the first day in which the bees were transferred to the monitoring system were used for estimating the timing of the onset and offset of activity (Fig. 1B). We included in our analyses only bees with statistically significant circadian rhythms (χ2 periodogram analysis; p < 0.01, period between 20 and 28 h) for which we could unambiguously determine the onset of activity.
We used the Oriana circular statistics software package (KCS, Pentraeth, Isle of Anglesey, UK) to determine the degree of synchronization and the phase coherence among bees within each treatment group. We used the time of onset as an index for the phase. The Rayleigh test was used to determine whether the phase synchronization was significantly different from random distribution, and the mean length of the Rayleigh vector was used as an index for phase coherence. We used the Watson-Williams F test to compare phases between the 2 groups.
Influence of Substrate-borne Vibrations on the Synchronization of Circadian Rhythms in Locomotor Activity
This experiment was designed to examine the hypothesis that substrate-borne vibrations entrain circadian rhythms of honey bee workers. The experimental design is summarized in Figure 2. On the first day, we caged groups of ~30 callow (i.e., newly emerged) bees or foragers in Libfield wooden cages (12 × 8 × 4.5 cm) with glass covers and provisioned each cage with ad libitum sugar syrup (50% w/w), and pollen, (collected by honey bees). Foragers were (collected at the hive entrance and identified as bees returning to the hive with pollen loads conspicuously attached to their hind legs. They were collected on the same day and from the same source colony as the callow bees. The foragers were then entrained to a light-dark cycle that was 5 h advanced relative to the phase of the colony in the first trial and to the natural day-night cycle in all other trials. We placed the cages containing groups of callow bees either on the same substrate (a canvas frame placed on a table) as the foragers (“same substrate”) or on a different table and substrate (“different substrate”). In each trial, we used 2 cages for each treatment (see Fig. 2). We placed all the cages in the same environmental chamber (30 °C ± 1 °C, 55% RH ± 5%, illuminated with dim red Edison Federal EFEE 1AE1 Deep Red LED lights). The distance between the cages of the foragers and those with callows on the same substrate and different substrate was identical (Fig. 2). We sucked out volatiles (which may mediate social entrainment) using an exhaust air unit with its opening (10-cm diameter) placed above the foragers’ cages. We repeated this experiment 5 times between May and August 2017, with each trial consisting of bees from a different source colony (i.e., a different family of bees). The survival rate was 45% (range, 30%-70%) for foragers and 87% (range, 61%-100%) for young bees. Such differences in survival are common because foragers are older and foraging activity is physically demanding.
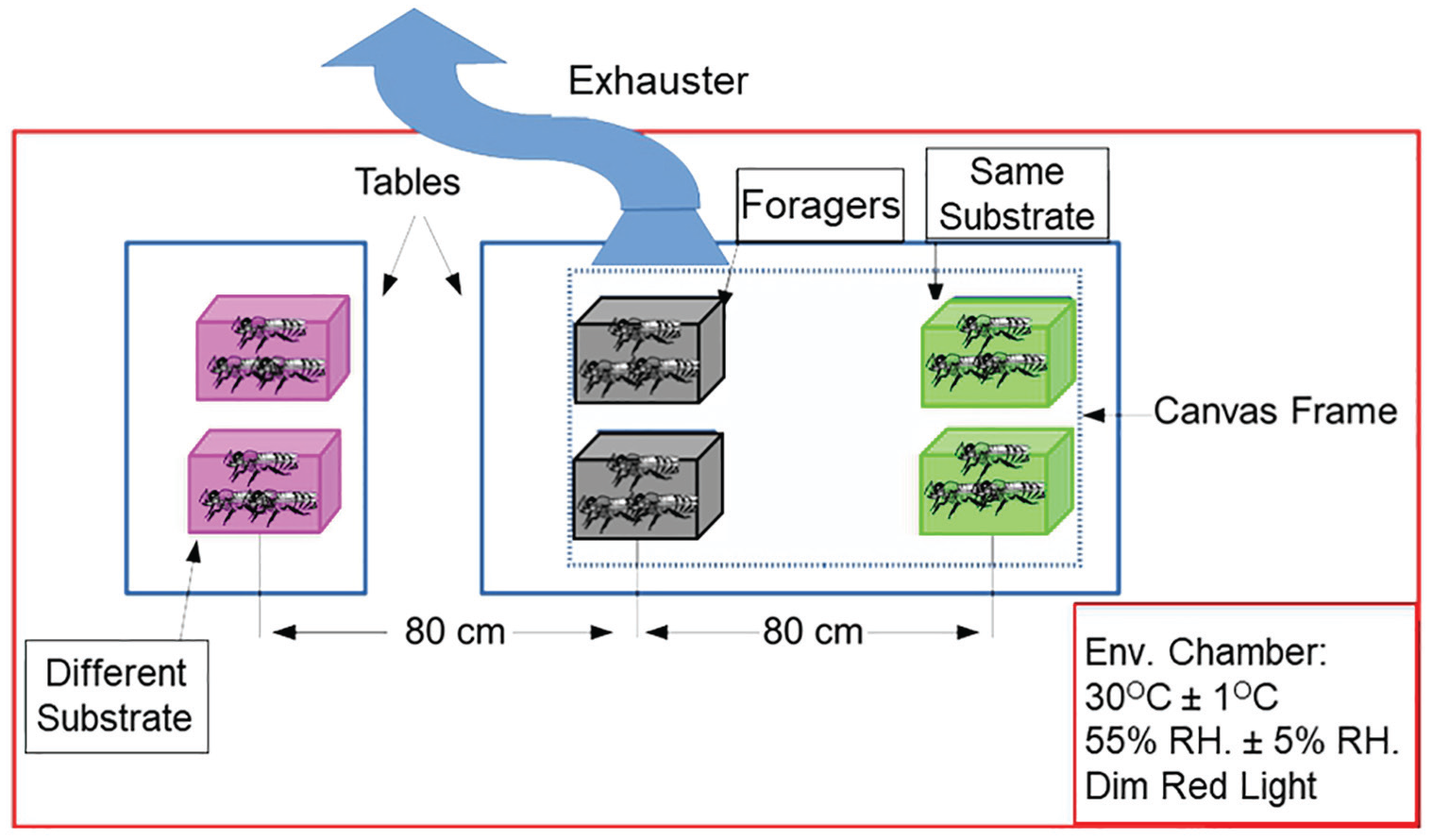
Setup for the experiment that tested whether substrate-borne vibrations entrain circadian rhythms in locomotor activity. We placed newly emerged bees in 2 small cages (30 bees/cage) on the same (“same substrate”) or on a different substrate (“different substrate”), on which we placed 2 cages with foragers. We sucked out the air above the foragers to prevent the spread of forager-emitted odorants that could influence the circadian rhythms of young bees. Color versions are available online.
Influence of Colony Volatiles on Synchronization of Circadian Rhythms in Locomotor Activity
To test the hypothesis that volatile chemicals entrain circadian rhythms of honey bee workers, we exposed callow bees to volatiles drawn out from a small hive housing a free-foraging colony (“colony”) or from a control empty hive (“empty hive”). The empty hive was of a similar size and contained the same number of empty wax combs. To keep its temperature constant and at a similar level as in the populated hive, the empty hive was heated with a reptile-heating mat (20 W, 16.5 × 11 inches). Both hives were prepared with a similar hole in the backboard through which an exhauster tube (25-mm diameter) was installed (Fig. 3).
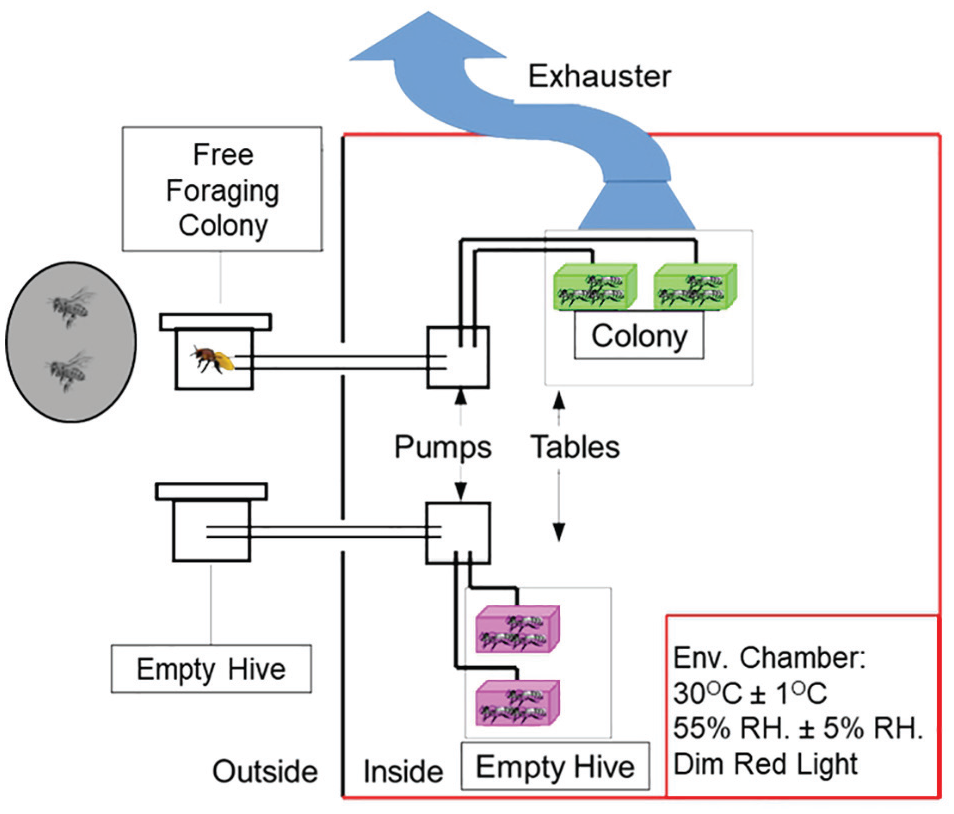
Setup for the experiment that tested whether hive volatiles entrain circadian rhythms in locomotor activity. Cages, each with 30 callow bees, were exposed to a constant air flow sucked out from either a hive housing a free-foraging colony (“colony”) or a similar control empty hive containing the same number of empty combs (“empty hive”). We placed all the cages on vibration-absorbing bases to minimize the possible transfer of vibrations. We sucked the air above the colony group to prevent the unintended release of volatiles that could affect the behavior of other bees. Color versions are available online.
We pumped the air out of the hive boxes using 2 aquarium pumps (Aquarium Air Pump AS-1063, Atman). The air from each hive flowed into 2 Libfield cages, each containing 30 callow bees (Fig. 3). The cages were provisioned with ad libitum sugar syrup (50% w/w) and pollen. We placed the cages on vibration-absorbing bases (Palziv polyethylene foam, Palziv Inc, Israel; https://www.palziv.com]) to minimize the possible transfer of vibrations. We placed a tube (10-cm diameter) from the exhaust air unit above the cages of the colony treatment cages to prevent the unintended transfer of volatiles to the control cages. The pump and the cages were housed inside a tightly regulated environmental chamber (30 °C ± 1 °C, 55% ± 5% RH) constantly illuminated with dim red light (as in the first experiment above). The populated and empty hives were placed outside the building. The colony in the populated hive was self-sustained (Fig. 3). We repeated this experiment 5 times between September and November 2017, each trial with bees from a different source colony. The average survival rate was 79% for foragers (range, 70%-100%) and 67% for the young bees (51%-84%).
Results
Influence of Substrate-Borne Vibrations on the Synchronization of Circadian Rhythms in Locomotor Activity
We repeated this experiment 5 times, each trial with bees from a different source colony, and monitored a total of 454 bees. The experimental groups differed in the strength of circadian rhythms. The foragers had stronger rhythms (Power = 222.76 ± 160.09 SD) compared with the 2 groups of young bees, which were overall similar (“same substrate”: 128.89 ± 119.81 and “different substrate”: 122.52 ± 115.04; Fig. 4A). The proportion of bees showing statistically significant circadian rhythms was 91% (range, 70%-100%) for foragers, 71% (range, 58%-85%) for young bees on the same substrate, and 68% (range, 50%-70%) for the different substrate group (data not shown).
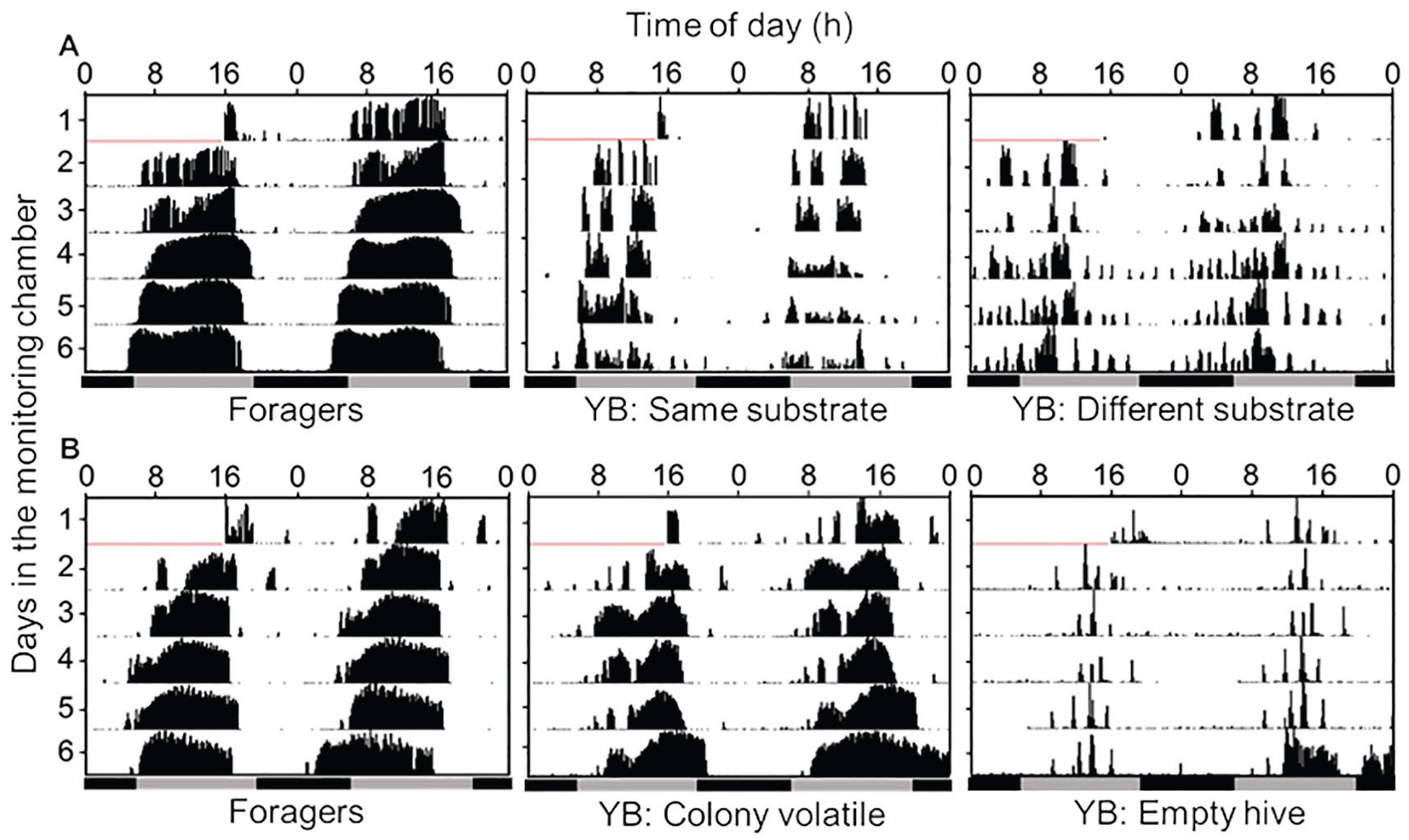
Representative double-plotted actograms. (A) Experiment 1 tested whether substrate-borne vibrations entrain circadian rhythms in locomotor activity. (B) Experiment 2 tested whether hive volatiles entrain circadian rhythms in locomotor activity. The bars at the bottom of the plots show the ambient illumination regime: Black = night; gray = day. The bees were monitored under dim red light. YB, young bees. Color versions are available online.
The phase of Same Substrate bees was overall more similar to that of the foragers compared with that of the Different Substrate treatment. In 3 of 5 trials, the phase of the Different Substrate group was significantly different from that of the foragers (Watson-Williams F test <0.05; Fig. 5A). There was a similar trend in 2 additional trials in which the different substrate bees did not show a statistically significant phase coherence; therefore, we could not compare the vectors with the Watson-Williams F test. The young bees on the same substrate also showed stronger phase coherence (longer Rayleigh vectors) compared with those placed on a substrate separated from that of the foragers (Fig. 5B; Kruskal-Wallis test, p = 0.007; Steel-Dwass-Critchlow-Fligner post hoc test: forager, same substrate: p = 0.417; forager, different substrate: p = 0.024; same substrate – different substrate: p = 0.116). In all 5 trials, the phase difference (in minutes) between that of the foragers and the Different Substrate group was larger than that compared with the Same Substrate group (binomial test, n = 5, p = 0.031; Fig. 5C).
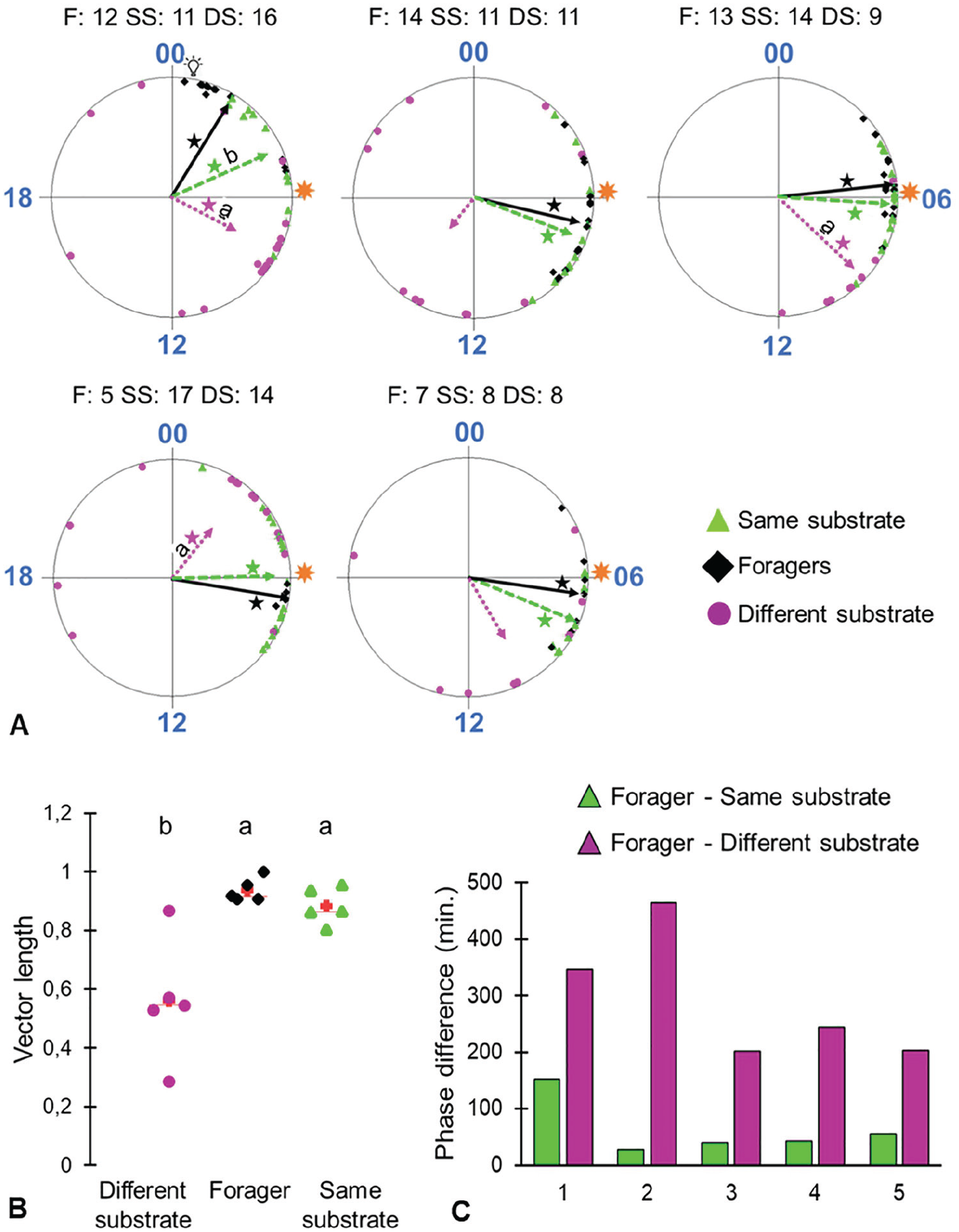
Substrate-borne vibrations entrain circadian rhythms in honey bee workers. (A) Circular plots comparing the phases of bees from the different treatment groups. Each plot summarizes a different trial. Sample sizes (number of bees with a clear onset) are shown above each plot. F = foragers; SS = callows on the same substrate; DS = callows on a different substrate. The time of day is depicted on the circular plot perimeter. Each Symbol depicts the onset of an individual bee. The vectors point to the average onset time, and their length corresponds to the degree of phase coherence. Foragers = black solid vectors; callows on the same substrate = green dashed vectors; callows on a different substrate = magenta dotted vector. Asterisks indicate that the p-value obtained from the Rayleigh test is statistically significant (*α < 0.05). Vectors marked with different letters differ in phase from that of foragers in a Watson-Williams F test. The foragers were entrained to a phase 5 h advanced (illustrated by the light bulb symbol) relative to the phase of the colony (illustrated by the sun symbol) in the first trial and to the natural sunrise in all other trials (sun symbol). (B) Scatter plot summary of the degree of synchronization (length of the Rayleigh vector) across the different treatments. The red crosses correspond to the means and the central horizontal bars to the medians. Treatments marked with different letters are significantly different in the Kruskal-Wallis test (p < 0.05) followed by Steel-Dwass-Critchlow-Fligner post hoc test. (C) The phase difference between forgers and each of the 2 groups of young bees.
Influence of Hive Volatiles on the Synchronization of Circadian Rhythms in Locomotor Activity
We performed 5 repetitions of this experiment, each with bees from a different source colony. In total, we monitored locomotor activity for 580 bees. As in the first experiment, the foragers had stronger circadian rhythms (mean Power = 234.43 ± 119.48 SD) than the 2 groups of young bees, which were similar overall (Colony, 166.93 ± 115.44; Empty Hive group, 158.94 ± 127.31; Fig. 4B). Almost all of the foragers (98%; range, 95%-100%) showed statistically significant circadian rhythms, whereas only 75% (range, 62%-89%) in the Colony and 70% (range, 58%-84%) in the Empty Hive group showed significant circadian rhythms (data not shown).
The phase of the young bees that were exposed to air from a hive housing a colony (“Colony”) was more similar to that of the foragers (collected from the same colony from which we sucked the air) compared with that of the callow bees exposed to air sucked from a similar hive with no bees. In 1 of 5 trials, the phase of the empty hive bees was significantly different from that of the foragers (Watson-Williams F test, p < 0.05; Fig. 6A). There was a similar trend in 2 additional trials in which the empty hive bees did not show a statistically significant phase coherence, and therefore, we could not use the Watson-Williams F test to compare the vectors. Bees from the colony treatment showed stronger phase coherence (longer Rayleigh vector) compared with the empty hive treatment (Kruskal-Wallis test, p < 0.0001; Steel-Dwass-Critchlow-Fligner post hoc test: forager-colony, p = 0.043; forager-empty hive, p = 0.024; colony-empty hive, p = 0.043; Fig. 6B). In a pooled analysis across all 5 trials, the phase difference relative to the foragers did not differ between the 2 groups of young bees (binomial test, p = 0.31; Fig. 6C). It is notable, however, that in the first 2 trials, the effect size for phase difference was much larger for the bees exposed to the empty hive.
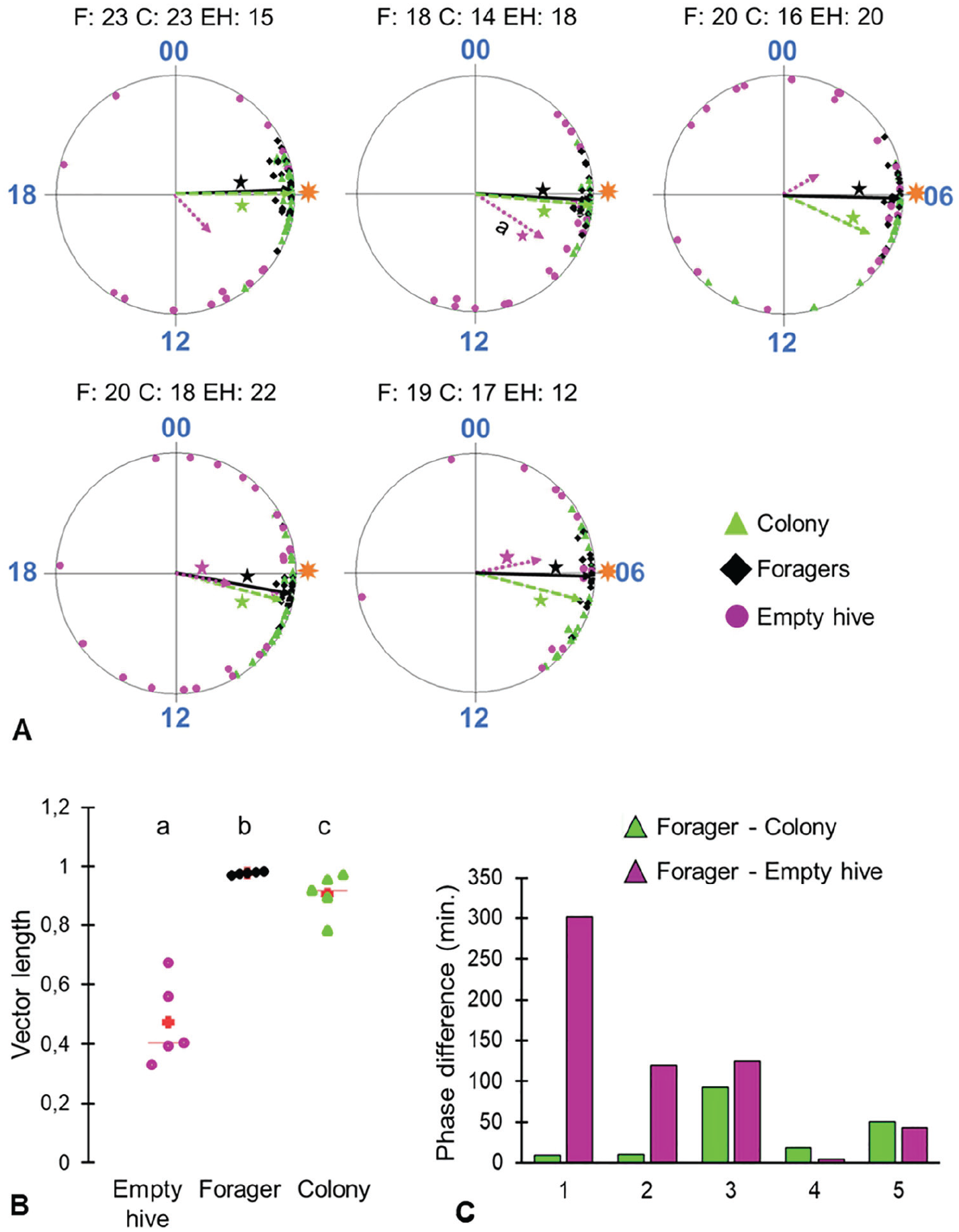
Hive volatiles mediate social synchronization. (A) Circular plots comparing the phases of bees from the different treatment groups. Sample sizes are shown above each plot (number of bees with a clear onset). F = foragers (black solid vectors); C = callows exposed to air from a colony (green dashed vectors); EH = callows exposed to air from an empty hive (magenta dotted vectors). The foragers were entrained by the natural sunrise (illustrated by the sun symbol) in all trials. (B) Scatter plot summary of the degree of synchronization (length of the Rayleigh vector) across different treatments. (C) The phase difference between each of the 2 groups of young bees relative to the foragers. Other details as in Figure 5.
Discussion
Our experiments show that both substrate-borne vibrations generated by forager activity and volatile cues emitted by a free-foraging colony can entrain circadian rhythms in locomotor activity of young honey bees. These results were obtained using a robust data set of more than 1000 bees that were each monitored individually. We repeated each experiment 5 times, each trial with bees from a different source colony. Given that bees in each colony are the offspring of a different queen and drones, our findings are not limited to certain genotypes or laboratory lines. It is also important to note that because we determined the circadian parameters for bees after removing them from the environment in which they were entrained, our measurements reflect properties of the internal clock rather than environmental (e.g., social) factors that could mask the clock effect on locomotor activity. Our experimental approach allowed us to uncouple the effects of substrate-borne vibrations and volatile colony cues. In the experiment testing the influence of substrate vibrations, we minimized the transfer of volatiles by sucking out the air above the forager cages. In the experiment testing the influence of hive volatiles, we minimized possible effects of vibrations by placing the cages on vibration-absorbing bases and on separated bases. Taken together, these findings lend credence to the hypothesis that substrate-borne vibrations and volatile olfactory cues, which are generated by active bees, mediate social synchronization among workers in a honey bee colony.
Our findings are consistent with the hypothesis that hive volatiles mediate social synchronization in honey bee colonies (Moritz and Kryger, 1994; Bloch et al., 2013; Fuchikawa et al., 2016). We show that young bees exposed to air from a hive housing a free-foraging colony are better synchronized with each other and overall have a phase more similar to that of the free-foraging colony, compared with similar bees that were exposed to air coming from a similar hive with no bees (Fig. 6). These findings are consistent with earlier studies showing that young bees that were caged inside double-mesh enclosures were similarly synchronized with the ambient day-night cycles as bees freely moving in the hive, indicating that direct contact is not necessary for social synchronization in a honey bee colony (Fuchikawa et al., 2016). Similarly, Moritz and Kryger (1994) showed that phase synchronization between 2 groups of bees was improved when the divider separating their chambers enabled air flow. What are the colony volatiles mediating this effect? We propose that the 2 most likely factors are volatile pheromones or fluctuations in the concentration of gases, such as CO2 or O2, that is influenced by bee activity.
The CO2 concentration within the hive shows a clear diurnal cycle (Murphy et al., 2015; Edwards-Murphy et al., 2016; Ohashi et al., 2009). Typically, lower levels are recorded during daytime when the foragers are outside the hive, with high levels in the morning and the late afternoon. These 2 CO2 peaks apparently correspond to the time when a large cohort of foragers leave the nest for their first morning foraging trips, and then the evening peak is when the last cohort of foragers return to the hive (Ohashi et al., 2009). Honey bees regulate their nest microenvironment homeostatically. They regulate CO2 levels by standing near the hive entrance and fanning with their wings, ventilating the nest cavity. There is a positive correlation between CO2 levels inside the hive and the number of fanning bees (Seeley, 1974; reviewed in Guerenstein and Hildebrand, 2008). Thus, we speculate that CO2 levels can entrain the circadian clock of bees by 2 nonmutually exclusive mechanisms: First, an elevation in CO2 levels leads to an increase in the number of bees fanning, and this activity entrains their circadian clocks. Second, fluctuation in CO2 levels act as a zeitgeber setting the phase of circadian clocks, which affects locomotor activity. There is some evidence that CO2 can entrain circadian rhythms in insects (Nicolas and Sillans, 1989), and in mammals, changes in CO2 concentration act at the cellular level and can phase shift oscillations in clock gene expression in a cell culture (Adamovich et al., 2019).
Honey bees use a wide variety of vibrational signals to communicate and coordinate colony-level activities. For example, both the “waggle dance” with which scouts and foragers recruit followers to a rewarding flowering patch and the “tremble dance” with which they inhibit further recruitment are at least partially mediated by comb vibrations (reviewed in Hrncir et al., 2005; Hunt and Richards, 2013). The hypothesis that substrate-borne vibrations can mediate social synchronization is also consistent with findings that in the fruit fly Drosophila melanogaster, circadian rhythms in locomotor activity can be entrained by oscillations in vibration intensity (Simoni et al., 2014). We do not know yet of any study recording comb vibrations over several days in honey bee colonies. It is reasonable, however, to assume that the daily fluctuations in forager activity generate comb vibrations that are sensed by nestmates and can mediate clock entrainment.
The evidence that substrate vibrations and colony volatiles entrain circadian rhythms lends credence to the hypothesis that surrogates of worker activity mediate social synchronization in honey bee colonies. The link between worker activity and comb vibrations is quite straightforward, although their repertoire in the hive is broad (Hrncir et al., 2005; Hrncir et al., 2019; Hunt and Richards, 2013), and additional work is needed for identifying the specific vibrations that entrain the clock. Changes in CO2 and O2 concentration inside the hive are influenced by worker activity and metabolism. Given that active bees typically have an increased body temperature, their level of activity may also affect the release (e.g., evaporation) of additional chemicals such as cuticular pheromones from their body surfaces, which may also contribute to social synchronization. However, the hive environment is very rich in volatiles, and much work is needed to identify the specific volatile chemicals that mediate social entrainment. We suggest that in natural colonies, vibrations and volatiles, and perhaps additional surrogates of worker activity, act together to create oscillations in the nest microenvironment. This idea is consistent with self-organization models in which the sum activity of individual workers produces oscillations in the microenvironment of the hive, which in turn entrain the circadian clocks of other individuals in the colony (Moritz and Fuchs, 1998; Bloch et al., 2013).
Footnotes
Acknowledgements
We thank Rafi Nir for professional beekeeping assistance. We also thank Muki Nagari, Jacob Holland, and Igor de Medici for assisting with the experiments. This study was supported by grant 1274/15 from the Israel Science Foundation.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
