Abstract
The importance of the circadian clock for the control of behavior and physiology is well established but how and when it develops is not fully understood. Here the initial expression pattern of the key clock gene period was recorded in Drosophila from embryos in vivo, using transgenic luciferase reporters. PERIOD expression in the presumptive central-clock dorsal neurons started to oscillate in the embryo in constant darkness. In behavioral experiments, a single 12-h light pulse given during the embryonic stage synchronized adult activity rhythms, implying the early development of entrainment mechanisms. These findings suggest that the central clock is functional already during embryogenesis. In contrast to central brain expression, PERIOD in the peripheral cells or their precursors increased during the embryonic stage and peaked during the pupal stage without showing circadian oscillations. Its rhythmic expression only initiated in the adult. We conclude that cyclic expression of PERIOD in the central-clock neurons starts in the embryo, presumably in the dorsal neurons or their precursors. It is not until shortly after eclosion when cyclic and synchronized expression of PERIOD in peripheral tissues commences throughout the animal.
The circadian clock is the endogenous pacemaker, which generates the daily output rhythms in behavior, physiology, metabolism and cellular processes, and persists in the absence of environmental cues (Chang, 2006; Peschel and Helfrich-Förster, 2011). Entrainment is the synchronization of the endogenous clock to fluctuating environmental factors, which are called zeitgebers, and light is the most important one (Peschel and Helfrich-Förster, 2011). A functional circadian system requires 3 basic components: 1) the endogenous circadian oscillator, which is temperature compensated, comprising key clock genes that regulate their own expression; 2) input pathways, which transmit environmental information such as light and entrain the circadian oscillator; and 3) output pathways, which manifest timed behavior and physiology (Yu and Hardin, 2006). In Drosophila, the central clock is defined as a cluster of approximately 150 clock gene-expressing neurons in the brain, which regulates circadian rhythmicity, such as locomotor activity. These central clock neurons are divided into dorsal lateral neurons (LNd), large ventral lateral neurons (l-LNv), lateral posterior neurons (LPN), small ventral lateral neurons (s-LNv), and dorsal neurons 1-3 (DN1-3) (Helfrich-Förster, 2005; Liu et al., 2015; Peschel and Helfrich-Förster, 2011). In addition to the central clock, peripheral clocks reside in various organs and tissues, characterized by the detection of clock gene or protein oscillations throughout the animal (Ito and Tomioka, 2016).
The circadian clock has profound significance in maintaining health throughout life in a range of organisms, including humans. The circadian clock machinery is largely conserved between humans and fruit flies, and Drosophila has been an effective tool for elucidating the cellular and biochemical mechanisms underlying the circadian system (Panda, 2002). The questions of when, where, and how these rhythms take place have attracted growing attention in circadian biology. A number of studies have worked on the developmental process of the circadian system in Drosophila (Glossop et al., 2014; Houl et al., 2008; James et al., 1986; Kaneko et al., 1997; Kaneko and Hall, 2000; Liu et al., 2015; Malpel et al., 2002; Malpel et al., 2004; Ruiz et al., 2010; Sehgal et al., 1992), yet the underlying mechanisms have not been fully described. Of particular interest is determining the timing of when the central molecular clock starts to oscillate and when the peripheral clocks are synchronized in the whole animal.
Adult activity rhythms are the most detailed and commonly described output of the Drosophila circadian clock. Sehgal et al. reported that adult behavioral rhythms could be synchronized by a 12-h light pulse administered as early as the first-instar larval stage (but not during embryonic stages), implying that the circadian clock might function continuously from the first larval stage onward (Sehgal et al., 1992). Subsequently, it has been demonstrated that a 10-min light pulse, different light regimes, and ethanol exposure, in the larvae can affect the adult free-running period or shift the phase of adult rhythms (Kaneko et al., 2000; Seggio et al., 2012; Sheeba et al., 2002). In summary, this supports the idea that a functional circadian clock is present from the first larval stage.
Driving behavioral rhythms is the “molecular oscillator,” which is based on the transcription-translation feedback loops of several clock genes, including period and timeless (per and tim) (Hardin and Panda, 2013). The expression of core clock genes and their product proteins, including per/PER and tim/TIM, as well as their regulators, are found in the embryonic central nervous system of Drosophila, suggesting that brain oscillator neurons may begin their development during embryogenesis. However, the restricted 24-h duration of the embryonic stage at 25°C (typical environmental rearing temperature) limits our ability to conclude the existence of circadian pattern of clock gene expression (Glossop et al., 2014; Houl et al., 2008; James et al., 1986; Liu et al., 2015; Ruiz et al., 2010).
Immunohistochemistry studies by Kaneko in 1997 demonstrated that the cyclic expression of PER and TIM occurs in the Drosophila larval central nervous system (CNS). This required animals to be sacrificed every 6 h, with data pooled from different brain samples (Kaneko et al., 1997). The question remains whether this technique has sufficient resolution to represent faithfully the key parameters of circadian oscillations. Several studies (Glossop et al., 2014; Houl et al., 2008; James et al., 1986; Liu et al., 2015; Ruiz et al., 2010) that put more emphasis on the spatial expression than temporal expression of clock genes, have concentrated on where the circadian clock may start to “tick” instead of when. Using luciferase as a real-time reporter of gene expression in vivo makes it possible to monitor clock-gene levels in living individual flies (Stanewsky, 1997). The advantages of this technique when compared with other methods include short-interval and long-term measurements, as well as the ability to collect time-series data from a single live animal.
Until now, there has been no definite conclusion on whether an entrainable clock exits at the end of embryonic development. The negative correlation between temperature and developmental timing is well defined in Drosophila (Kuntz and Eisen, 2014), offering the possibility to extend the embryonic stage to potentially reveal rhythmicity. A higher temperature speeds up development, whereas a lower temperature—within the range of 16°C and 29°C—slows it down; outside of this range leads to an increase in mortality (Economos and Lints, 1986). Thus, 16°C is an optimal choice to prolong maximally the embryonic stage, and lead to an approximate doubling of the developmental time as compared with 25°C; these results are based on our previous observations (data not shown) and the findings of other studies (e.g., Ghosh et al., 2013). In contrast, circadian clocks are temperature compensated; i.e. the frequency of molecular and behavioral rhythms remains constant at different temperatures (Hall, 1997). Therefore, extending developmental time by lowering the ambient temperature may help to clarify the timing of the emergence of an entrainable molecular clock beyond the 24-h temporal constraint.
In this study, we aimed to determine when the central and peripheral components of the circadian clock start to oscillate. First, PER expression was measured continuously from the beginning of the embryonic stage in individual flies at 25°C and 16°C using luciferase clock-gene reporters. Second, we investigated the adult locomotor activity response to timed light treatment during pre-adult stages. Third, although it is known that the establishment of behavioral rhythms in individual flies does not require exposure to light during development, we investigated here if this is also true for molecular rhythms of clock gene expression.
Materials and Methods
Measurement of Clock Gene Expression
Two transgenic reporter lines 8.0-luc and XLG-luc were used. XLG-luc contains the per promoter sequences plus almost the entire per coding region, whereas 8.0-luc contains the same coding region without the 5’-UTR sequences (Veleri et al., 2003). Flies were reared on growth-media loaded in standard plastic Drosophila vials and kept on a 12-12 h LD cycle with a constant temperature of 25°C and used for real-time bioluminescence recording of per, as described previously (Stanewsky, 2007). Individual eggs were collected within 4 h of being laid in DD and transferred into a single well of a white opaque 96-well microplate (Greiner Bio-one) under 100-lux white light. Minimal light was used because eggs cannot be distinguished under DD even with the aid of infrared night vision equipment or under red light. This short-term operation falls in the first half of the embryonic stage, when neither the molecular clock nor its light-response has established, according to our and previous findings (Houl et al., 2008). Each well contained growth-media fortified with
Monitoring of Locomotor Activity
For the locomotion assay, wild-type flies were maintained as above, and eggs were collected and loaded into standard vials all under DD with the aid of infrared night vision equipment. Fruit flies from one group were kept in DD during the entire experiment, and other groups were given a 12-h 1000-lux light pulse at different developmental times within the embryonic and early larval stages. When a particular group of flies was under light treatment, the other groups were kept in darkness (as shown in Figure 3). Fly locomotor activity was monitored automatically using Trikinetics Drosophila Activity Monitors (Waltham, MA, USA), as previously described (Zhao et al., 2018). On the days before eclosion, with the aid of infrared night vision equipment in DD, individual pupae were transferred into glass tubes (8-cm long and 5-mm diameter) with growth media at one end, sealed with plastic plugs, and plugged with cotton at the other end. Locomotor activity, temperature and relative humidity were recorded every 5 min for at least 7 days in DD.
Statistical Analysis
Data are shown as mean and standard error (mean [SEM]). Periodogram with Chi-square in ClockLab (Actimetrics, USA) was used to analyze free running periods (tau), phases, and rhythmicity strength of wild-type fly locomotor activity in DD. Lomb-Scargle periodograms in ClockLab and MetaCycle (Wu et al., 2016) were used to analyze periodicity and rhythmicity of bioluminescence signals. P values < 0.05 were considered rhythmic.
As the phases of PER expression in individual flies (both 8.0-luc and XLG-luc) raised in DD do not necessarily synchronize, rhythms of PER expression were aligned post hoc at the data analysis stage. This was achieved by manually aligning the acrophase of flies across the recording period. To ensure that no larval signal was incorrectly assumed to be an embryonic signal, we chose a cut-off of embryonic expression of 24 h (this is the standard embryonic developmental time at 25°C).
Results
Period Expression in the Central Clock during Early Development
To measure PER expression in subsets of the central-clock neurons, we used 8.0-luc flies, which express a PER-luciferase fusion protein in subsets of the dorsal neuronal clusters (DN1-3) and the LNd (Veleri et al., 2003; Yoshii et al., 2009). Over the 20 days of continuous recording at 25°C, starting with 8.0-luc embryos, we found that 81.5% of 8.0-luc individuals (n = 108) commenced rhythmic PER expression from the embryonic stage onward in constant darkness (DD) (p < 0.05); oscillations in the remaining individuals did not reach significance (Figure 1A, B). The acrophases of rhythmic individuals occurred at different developmental times, with an average of 16.0 ± 0.6 h after egg collection, corresponding to embryogenic stages 16 and 17. Similarly, over the 10 days of continuous recording at 16°C, 75.6% of 8.0-luc flies (n = 41) showed rhythmic PER expression onset from the embryonic stage onward in DD (p < 0.05); oscillations in the remaining individuals did not reach significance (Figure 1C, D). The acrophases of rhythmic individuals occurred at 36.7 ± 1.3 h after egg collection, with levels falling in the second half of embryogenesis. At this lower temperature, where embryonic developmental time is doubled, clear peaks of PER expression occurred after 24 h and 48 h, demonstrating that the molecular clock starts operating during embryogenesis (Figure 1C). Because rhythmic PER expression in DD flies was not synchronized at either temperature, we manually synchronized the signals by aligning the acrophases, grouping them as an average signal to visualize rhythmicity (Figure 1). At 25°C, grouped PER expression during the embryonic (24 h) and early larval (stages 1 and 2) stages was emphasized (Figure 1A), with a free-running period of 23.6 h. At 16°C, grouped PER expression during the embryonic (48 h) and 1st larval (48 h) stages was emphasized (Figure 1C), with a free-running period of 23.6 h.
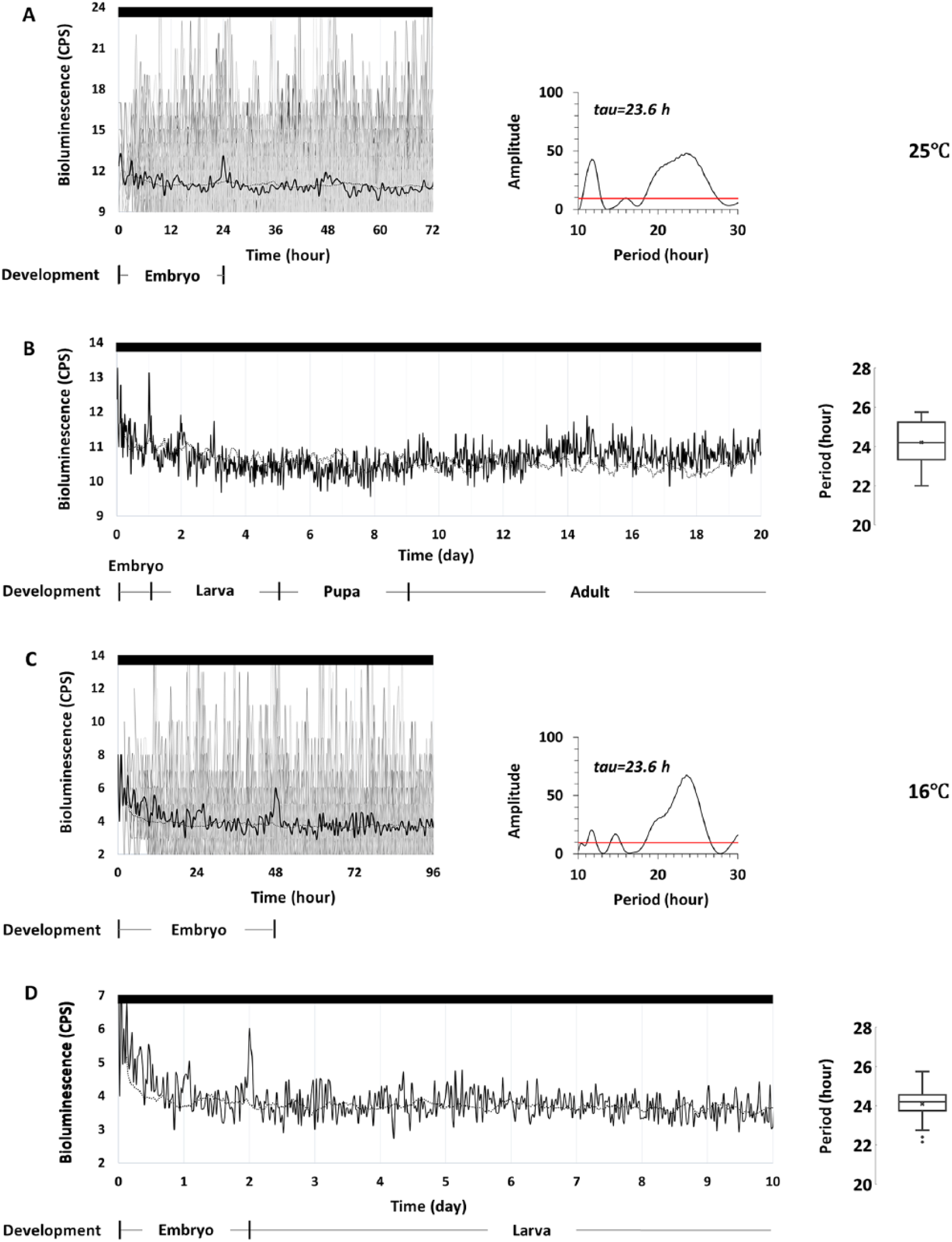
Bioluminescence time-series data of individual 8.0-luc fruit flies measured in DD at 25°C (A, B) and 16°C (C, D). Individual bioluminescence (counts per second) is plotted as grey lines and synchronized by acrophases, averaged as black lines. The control is the average signal from wells containing only luciferin-media without any flies under the same conditions, represented as black dashed lines. Corresponding developmental stages are shown at different temperatures. (A) Synchronized PER expression in 8.0-luc (n = 88) during embryonic and early larval stages of the first 72 h after fertilization. The periodogram displays significant circadian rhythm (Lomb-Scargle, p < 0.05) with a 23.6 h tau, and (B) from the embryo until adult (the first 20 days after fertilization), the periodogram shows cyclic expression, with a mean period of 24.2 ± 0.1 h, as shown in the box-and-whisker plot. (C) Synchronized PER expression in 8.0-luc (n = 31) during the embryonic and early larval stages of the first 96 h after fertilization. The periodogram displays significant circadian rhythm (Lomb-Scargle, p < 0.05) with a 23.6-hour tau, and (D) from the embryonic to the larval stages (the first 10 days after fertilization), the periodogram shows cyclic expression, with a mean period of 24.1 ± 0.2 h, as shown in the box-and-whisker plot.
Period Expression in the Peripheral Clocks during Early Development
After observing these per expression patterns at the central clock level, we decided to further study how PER oscillations develop at the peripheral-clock level. To distinguish the effect from the central clock, a second per transgenic line, XLG-luc was used, which is most likely expressed in many, if not all, per-expressing cells throughout the adult body, including the central and peripheral clock tissues (Stanewsky et al., 1997; Veleri et al., 2003). However, it mainly reports per signals in peripheral clocks because of the small number of central brain clock neurons compared with the thousands of clock cells in the rest of the body.
Again, because rhythmic PER expression in flies reared in DD was not synchronized, we manually synchronized the signals by aligning their acrophases at 25°C. During the continuous 20-day and 10-day XLG-luc recordings at 25°C or 16°C, respectively, we found that PER started to rise during the embryonic stage. All of the embryos kept in DD (n = 68) and in LD (n = 67) at 25°C showed this increase in PER signal (Figure 2A, B). The rise in PER peaked at the pupa and then decreased. Phase differences between DD and LD records most likely were caused by manual phase sorting of the DD records to aid recognition of rhythmic PER expression (see Methods for details). Importantly, no oscillations were evident during this first rise in PER. Subsequently, PER began to rise again after eclosion and, at this time, also began to cycle in DD with a period of 24 h and in LD with a period of 23.5 h. The strength of rhythmicity of the bioluminescence oscillations was more robust in LD compared with DD, as expected. At 16°C, PER expression in XLG-luc (n = 14 in DD and n = 82 in LD) begins to rise from the embryonic stage until the larval stage, without oscillations (Figure 2C, D).
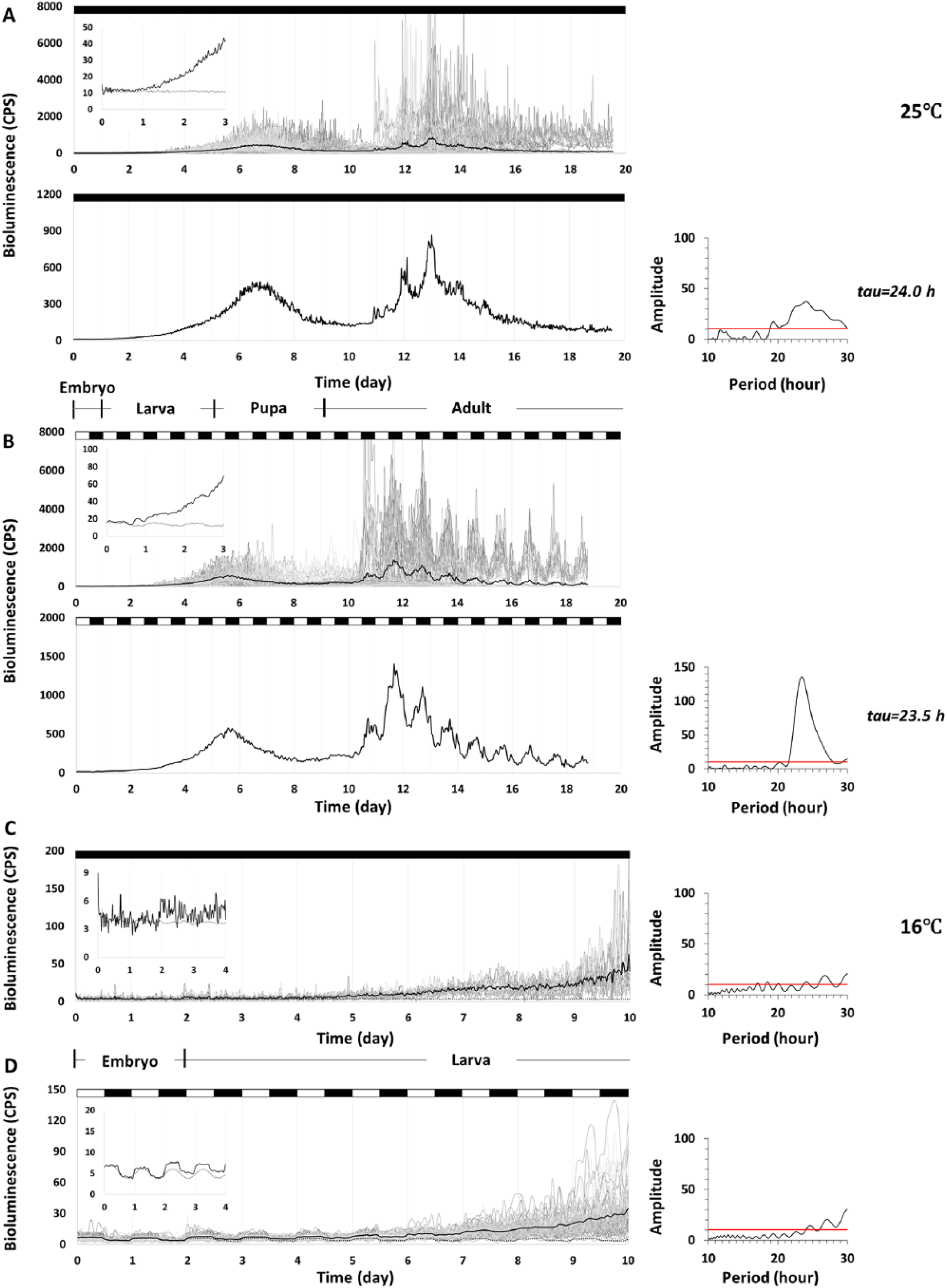
Bioluminescence time-series data of individual XLG-luc fruit flies measured at 25°C (A, B) and at 16°C (C, D). Individual bioluminescence CPS is plotted as grey lines and averaged as black lines. The control is the average signal from wells containing only luciferin media, without any flies under the same condition, noted as black dashed lines. The y-axis is rescaled to better show the first signal increase in the embryo and the pattern of rhythmicity in the adult. Corresponding developmental stages are shown at different temperatures. (A) Synchronized PER expression in XLG-luc in DD (n = 68) begins to rise from the embryo until it reaches a peak at the pupa. The signal decreases in the pupal stage but rises again and begins cycling with a period of 24.0 h (Lomb-Scargle, p < 0.05) from the adult stage. (B) Synchronized PER expression in XLG-luc in LD (n = 67) shows a similar pattern to that in DD, with oscillations beginning in the adults with a period of 23.5 h (Lomb-Scargle, p < 0.05). No oscillation is detected in the increasing signal from the embryonic and larval stages in DD or LD. PER expression at 16°C in XLG-luc in (C) DD and (D) LD (n = 14 and n = 82, respectively) begins to rise from the embryonic stage until the larval stage without oscillations (Lomb-Scargle, p > 0.05). Periodogram in LD is calculated as the average signal minus the control signal to counteract the pseudo-rhythm caused by the light-induced background (Stanewsky, 2007).
Synchronized Behavioral Rhythms in Adult Flies Exposed to Light Early in Development
The results from the above experiments indicated the existence of a functional clock in the embryo. Thus, we attempted to confirm this by testing whether light given at such early developmental stages influenced the phase of adult locomotor activity. Groups of wild-type flies kept in DD were given light treatment at different times of development. We found that the locomotor activity of individual flies raised in DD was not synchronized at either 25°C or 16°C. The probability that the distribution of individual phases within a population is random was calculated by Rayleigh’s test. Probabilities for flies reared in DD being synchronized were not significant, indicating a random distribution (p > 0.05). Flies that received light-treatment at the first half of the embryonic stage showed desynchrony at both temperatures. In contrast, those that had received light treatment at the second half of the embryonic stage showed synchronized distributions of phases (Figure 3). Even though the group of flies kept in DD was never exposed to light during their early development, they showed rhythmic locomotor activity as adults, similar to all the light-treated groups (Table 1).
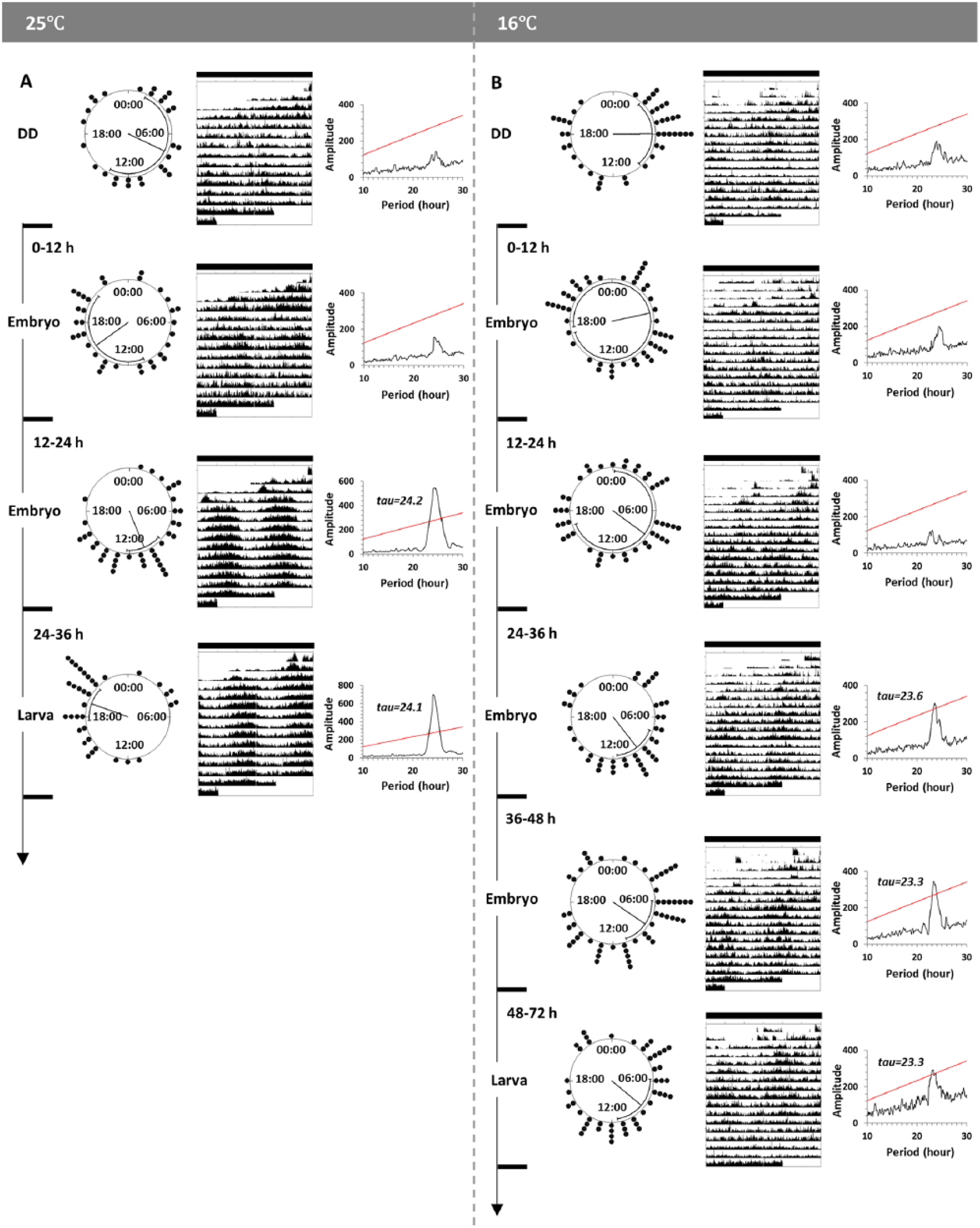
The circular distributions of the locomotor activity phases (acrophases), average actograms and periodograms at (A) 25°C (left panel) and (B) 16°C (right panel). One group per panel was under DD throughout the entire experiment (top). The12-h light pulses were administrated to other groups at different stages of early development. Each clock face shows a 24-h clock time. Black dots around clock faces indicate the timing of acrophase of every single fly in the same group. The mean phase of each population is shown by the angle of the radius with 95% confidence intervals. Double-plotted actograms were averaged from flies in each group. (A) The animals kept in DD for the entire experiment (n = 28) and given light pulses at 0 to 12 h (embryo, n = 34) show random distributions (p = 0.48 and p = 0.35, respectively), with arrhythmic average actograms supported by periodograms. Animals given light pulses at 12 to 24 h (embryo, n = 38), and at 24 to 36 h (larva, n = 35) show significantly concentrated distributions (p < 0.001), with rhythmic average actograms supported by periodograms (Chi-square, p < 0.05). (B) Animals kept in DD for the entire experiment (n = 43) show random distributions (p = 0.08). Animals given light pulses at 0 to 12 h (embryo, n = 48) and at 12 to 24 h (embryo, n = 45) show random distributions (p = 0.82 and p = 0.67, respectively), with arrhythmic average actograms supported by periodograms. Animals given light pulses at 24 to 36 h (embryo, n = 42), 36 to 48 h (embryo, n = 54), or 48 to 72 h (larva, n = 47) all show significantly concentrated distributions (p = 0.003, p = 0.01 and p = 0.03, respectively), with rhythmic average actograms supported by periodograms (Chi-square, p < 0.05).
Free-running periods and rhythmicity among DD and light-treated wild-type flies.
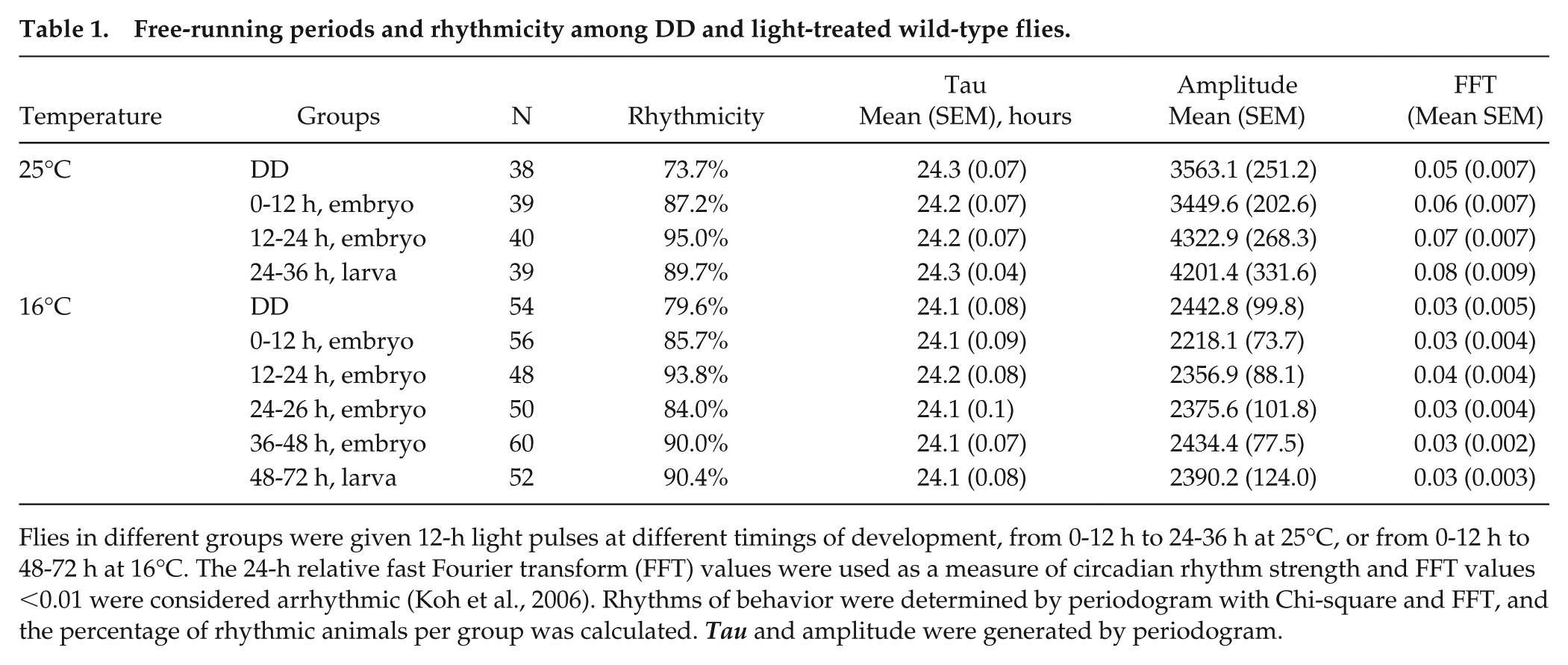
Flies in different groups were given 12-h light pulses at different timings of development, from 0-12 h to 24-36 h at 25°C, or from 0-12 h to 48-72 h at 16°C. The 24-h relative fast Fourier transform (FFT) values were used as a measure of circadian rhythm strength and FFT values <0.01 were considered arrhythmic (Koh et al., 2006). Rhythms of behavior were determined by periodogram with Chi-square and FFT, and the percentage of rhythmic animals per group was calculated. Tau and amplitude were generated by periodogram.
Discussion
In this study, we analyzed the development of the circadian clock in Drosophila by utilizing transgenic reporters that record the essential clock component PER in vivo. These tools allowed us to monitor the expression of clock genes in real time and to track the measurement throughout the life of the flies. Here, we have shown that the expression of PER starts in the embryo but does not begin to cycle until the adult stage. However, a reporter that reflects PER expression only in subsets of central clock neurons starts to show rhythmic circadian patterns during embryogenesis, coinciding with the time when light begins to affect the clock. In addition, and in agreement with previous studies, clock gene oscillations of PER as well as adult locomotor activity rhythms (Sehgal et al., 1992) initiate without the requirement of light.
Slowing down developmental speed by exposing embryos to constant 16°C allowed us to show that circadian PER expression initiates from as early as the embryonic stage in DD. PER in 8.0-luc flies is specifically expressed within subsets of the DN and LNd, but not in LNv, nor in peripheral clock tissues in adults, which explains the low bioluminescence signal detected. It implies that some of the DN and/or LNd precursor neurons may possess a functional clock already very early during development. It has been reported that LNd only differentiates in the pupal stage (Helfrich-Förster et al., 2006) when PER starts to cycle (Kaneko et al., 1997; Kaneko et al., 2000); although, CLOCK expression reveals that these cells are present in the third instar larvae (Liu et al., 2015). In addition to the finding that per is expressed in DN1 and s-LNv during embryonic stages 16 and 17 (Houl et al., 2008)—which fall within the second half of embryogenesis—we infer the signal from 8.0-luc flies potentially reflects embryonic PER expression in DN1 or their precursors. Our finding that a 12-h light pulse given at the second half of embryogenesis can synchronize adult behavioral rhythms is consistent with the molecular result, supporting the idea that at least some functional components of the central oscillator are present very early in development; i.e., in precursors of clock neurons.
Aside from the identified central clock neurons during early development, peripheral clock development has been less emphasized. It has been revealed that a significant non-cyclic expression of per mRNA initiates in the mid to late developing embryo, which is limited to the midline of the nervous system (Houl et al., 2008; James et al., 1986; Liu et al., 1988; Ruiz et al., 2010). This is consistent with our finding that PER expression begins from the embryo stage but does not oscillate in either DD or LD at the whole-animal level. However, the linearly increasing expression from embryo to larva is different from what has been reported previously, which displays low expression in larval stages and its re-emergence at the mid-pupal stage (Liu et al., 1988).
In the embryos and larvae, PER began to cycle in the 8.0-luc flies, while a non-cyclic increase was observed in XLG-luc flies, suggesting that neuronal oscillations of clock gene expression initiate before those in tissues more peripherally located. Because XLG-luc reports PER expression in both central and peripheral cells, we cannot exclude that some of the signal may come from the central clock neurons. However, of the total number of approximately 300 per-expressing cells in the central nervous system during embryogenesis, only 20 are defined as central clock neurons (Ruiz et al., 2010). This suggests that the XLG-luc signal mainly indicates the periphery. We further infer that PER is expressed within different peripheral clocks or their precursors around the body, which could be cyclic or non-cyclic, but not synchronized at the whole-animal level before eclosion. However, a limitation of our study is that, because many larval peripheral tissues are histolyzed during metamorphosis, we cannot assume that the oscillations in PER we see in the embryo are driven by the same unchanged tissues in the adult. It is widely accepted that direct light-entraining pathways exist in peripheral tissues of adult Drosophila. Because of the non-synchronized PER expression in LD, our results suggest that photo-receptors in peripheral clocks are not fully developed before the adult stage. Although the photo-transduction opsins have been detected in the larval body in Drosophila (Shen et al., 2011; Zanini et al., 2018), Rh1 rhodopsin gene expression in the compound eyes (peripheral tissues) begins in mid-to-late pupae (Kumar and Ready, 1995). This is also supported by a recent finding that although expression of Cryptochrome exists in certain larval peripheral tissues but not others, it lacks the function to mediate light-entrainment in both the central and peripheral clocks in adult Drosophila (Agrawal et al., 2017). This is different from the vertebrate system, where peripheral cells in zebrafish are synchronized by light at early embryonic stages, whereas in the adult peripheral clocks of both Drosophila and zebrafish, they are directly light-sensitive (Carr et al., 2005).
One of the important features of the circadian clock is that it is endogenous. The current developmental study shows that, in certain species, such as Drosophila, the circadian clock is able to develop and mature without the requirement of entraining stimuli, such as light (Sehgal et al., 1992). In our study light exposure did not initiate or speed up the development of the circadian clock or change the clock-gene expression level, but it synchronized the phases among individual flies. This is consistent with mammalian clocks that can start synchronized oscillations within an individual independently of zeitgebers, and independently of a functional maternal circadian clock (Jud and Albrecht, 2006). It also shares similarity with clocks in zebrafish, which can initiate but not synchronize without light (Dekens and Whitmore, 2008).
A single light pulse given during the embryonic stage in the current study, or during the larval stage in previous studies (Kaneko et al., 2000; Sehgal et al., 1992), can synchronize adult rhythmic behavior. One possible explanation is a “time-memory,” which implies the special ability of the clock to memorize such stimuli and manifest it later in life when the clock becomes more mature (Kaneko et al., 2000). The direction and magnitude of phase shifts of locomotor activity were different between wild-type and fast-clock pershort mutants, suggesting the involvement of per in the regulation of developmental “time-memory” (Kaneko et al., 2000). However, a reasonable assumption is that, at some point during early adult stages, synchronization between different clock cells is under the control of an as yet undescribed intrinsic mechanism. So in our behavioral experiment, rather than a time-memory, it is possible that the clock is “set” by the initial light and free runs in the central clock neurons (as we observe in 8.0-luc in DD) but not in most peripheral clock tissues.
An important role of the circadian clock in the regulation of development has been reported (Kumar et al., 2006; Kyriacou et al., 1990; Vallone et al., 2007). In this study, we placed emphasis on the development of the circadian clock itself. It is assumed that the development of the circadian clock should be in accordance with sequential and precise developmental processes. Consequently, the circadian clock is thought to develop as the whole body develops, in a gradual way (Vallone et al., 2007). Through the new application of established luciferase clock gene reporter techniques, we show cyclic clock-gene expression, presumably in precursors of dorsal clock neurons during the embryonic stages, which is earlier than previously thought. Light is not required to start the clock, but it can, to some extent, synchronize the clock as early as in the embryo, which is also earlier than previously thought. In total, the early cyclic expression of PER and light-entrainable behavior in this study suggests that key components of the circadian clock are present very early in life. Future studies may put emphasis on developmental details in individual pacemaker neurons, perhaps by performing simultaneous recordings of behavioral rhythms and clock-gene expression in the same animal.
Footnotes
Authors’ Contributions
J.Z., G.R.W., and J.F.C. designed research; J.Z. performed research; J.Z. and J.F.C. analyzed data; and J.Z., G.R.W., R.S., and J.F.C. wrote the paper.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
