Abstract
Sharks are an interesting group of vertebrates, as many species swim continuously to “ram” oxygen-rich seawater over their gills (ram ventilators), whereas other species “pump” seawater over their gills by manipulating buccal cavity volume while remaining motionless (buccal pumpers). This difference in respiratory physiology raises the question: What are the implications of these differences in lifestyle for circadian rhythms? We investigated the diel activity patterns of 5 species of sharks, including 3 ram ventilating species: the school shark (Galeorhinus galeus), the spotted estuary smooth-hound (Mustelus lenticulatus), and the spiny dogfish (Squalus acanthias); and 2 buccal pumping species: the Port Jackson (Heterodontus portusjacksoni) and draughtsboard (Cephaloscyllium isabellum) sharks. We measured the amount, duration, and distance traveled while swimming over multiple days under a 12:12 light:dark light regime for all species and used modified light regimes for species with a clear diel rhythm in activity. We identified a surprising diversity of activity rhythms. The school shark and smooth-hound swam continuously; however, whereas the school shark swam at the same speed and covered the same distance during the day and night, the smooth-hound swam slower at night and traversed a shorter distance. A similar pattern was observed in the spiny dogfish, although this shark swam less overall. Both the Port Jackson and draughtsboard sharks showed a marked nocturnal preference for swimming. This pattern was muted and disrupted during constant light and constant dark regimes, although circadian organization of this pattern was maintained under certain conditions. The consequences of these patterns for other biological processes, such as sleep, remain unclear. Nonetheless, these 5 species demonstrate remarkable diversity within the activity rhythms of sharks.
In many animals, biological processes are rhythmic and aligned with the naturally occurring changes in day length, allowing organisms to synchronize their physiology and behavior with predictable changes in the environment (Laposky et al., 2008). For example, the circadian clock shapes emergence times in bats (Hope and Jones, 2013), singing in songbirds (Da Silva and Kempenaers, 2017), and the phasing of sleep (Ball, 1992). Changes to day length can also influence the seasonal timing of reproduction (Robert et al., 2015). Understanding the activity rhythms of a species is an important first step in identifying the adaptive value behind those patterns. Furthermore, by measuring activity rhythms in many closely related species under the same conditions, we might find unexpected diversity in those rhythms, stimulating the creation of new questions (Bulla et al., 2016).
The basic biology and behavior of many elasmobranchs is poorly understood, especially in pelagic species. Nonetheless, the natural history surrounding fundamental behaviors, such as the timing and amount of daily swimming and resting, are paramount to understanding the role sharks play in marine ecosystems and may even assist in the mitigation of negative human-shark interactions (Gleiss et al., 2009). Sharks are typically characterized into 2 distinct groups based on their mode of respiration (Carlson et al., 2004; Kelly et al., 2019). Buccal pumping sharks facilitate respiration by lowering their mandible to draw oxygenated seawater into the buccal cavity and then raise the mandible to push the oxygen-rich water over their gills. This mechanism is also used by many teleost species and allows these animals to remain stationary for extended periods of time (Carlson and Parsons, 2001). It is still unknown, however, how these periods of sustained immobility are organized temporally in most buccal pumping species of sharks and whether they reflect sleep (Kelly et al., 2019). In contrast, ram ventilators do not possess the buccal pumping mechanism for gill ventilation and must maintain constant forward motion to push oxygen-rich seawater over their gills, rendering them obligate constant swimmers (Carlson et al., 2004). This unusual behavioral lifestyle may render the activity rhythms of these animals cryptic (if present at all), owing to the absence of conspicuous periods of restfulness. Qualitative studies have shown that many ram ventilating species of sharks appear to swim constantly (reviewed by Kelly et al., 2019), and acoustic and telemetric tracking studies have yielded powerful data on the movements and behavior on many of these species (Holland et al., 1993, 1999; Nelson et al., 1997; Weng et al., 2007; Papastamatiou et al., 2010; White et al., 2019). In recent years, advances in biologging techniques, such as the combining of accelerometry data with positioning technology, have facilitated further insight into the daily activity and behavior of elasmobranchs (Whitney et al., 2007, 2010; Gleiss et al., 2009, 2013; Shipley et al., 2018; Papastamatiou et al., 2018; Royer et al., 2020).
Assessing the reliability of accelerometry data from wild studies, however, can be difficult. Captive studies using video recordings are extremely useful for assessing the diel activity patterns of sharks as they allow for reliable, close, and continuous observations of animals. More subtle behaviors, such as respiration rates and eye movements, can also be obtained from such data. Furthermore, captive studies allow for full environmental control. In this manner, not only can we assess the presence of diel activity patterns, but importantly, we can also investigate the endogenous nature of these patterns and identify entraining cues involved in this process.
The school shark (Galeorhinus galeus), spotted estuary smooth-hound (Mustelus lenticulatus), and spiny dogfish (Squalus acanthias) are all ovoviviparous ram ventilators. They spend most of their lives in open water and on coastal shelves but are also known to inhabit shallow coastal waters during mating periods, pregnancy, or as juveniles (Simpson and Wardle, 1967; Francis, 1988; West and Stevens, 2001; Francis, 2013). Data on habitat preferences over space and time is available for the smooth-hound (Francis, 2013) as well as diel activity and movements for the closely related gray smooth-hound shark (Mustelus californicus; Espinoza et al., 2011; Freedman et al., 2017) and vertical migrations of school sharks (West and Stevens, 2001). There have been no studies, however, focusing on the activity rhythms on the spotted estuary smooth-hound or school sharks. The spiny dogfish has been reported to swim continuously and without circadian variation from behavioral observations in captivity (Karmanova et al., 1975). However, only the presence of swimming was noted, with no data collected on the intensity (speed and distance traveled) of swimming. Both the Port Jackson (Heterodontus portusjacksoni) and draughtsboard (Cephaloscyllium isabellum) sharks are oviparous buccal pumping sharks; they are also demersal and rest much of the 24-h day on cave floors and trenches in reefs and continental shelves (McLaughlin and O’Gower, 1971; Frick et al., 2010; Horn, 2016). Visual observations from captive and wild animals (O’Gower, 1995; O’Gower and Nash, 1978; Powter and Gladstone, 2008, 2009), as well as historic catch records (McLaughlin and O’Gower, 1971), have highlighted the movements and migratory patterns of Port Jackson sharks. Acoustic tracking data of wild Port Jackson sharks have shown this species to be nocturnally active and travel greater distances at night (Powter and Gladstone, 2009; Kadar et al., 2019). This is consistent with data obtained from video and accelerometry data from captive animals, which exhibited late-evening activity peaks and daytime troughs. A similar pattern has also recently been reported in the closely related horn shark (Heterodontus francisci), with wild sharks resting in shelters throughout the day and becoming active at night (Meese and Lowe, 2020). Whether these activity patterns are endogenous or regulated by external cues is unknown. Furthermore, nothing is known about activity patterns in the draughtsboard shark, although studies on closely related species, in both captivity and in the wild, have reported crepuscular and nocturnal activity (Nelson and Johnson, 1970; Awruch et al., 2012).
In this study, we first sought to investigate the amount and intensity of swimming in 3 species of ram ventilating sharks (school shark, smooth-hound, and spiny dogfish) and 2 species of buccal pumping sharks (Port Jackson and draughtsboard shark) over a 24-h day under 12:12 light:dark conditions. This was tested with a 3-day protocol to look for light regime effects on activity patterns and whether the ram ventilating animals in fact swim continuously or could show periods of immobility. Next, we explored whether any observed cyclicity in swimming was endogenously generated or influenced more by light regime conditions following a series of 9-day protocols, which included 3 days under constant light or dark conditions, and a forced desynchrony protocol, preceded and succeeded by 12:12 light:dark conditions.
Materials and Methods
Animals and Housing
Experiments were conducted between September 2017 and September 2018 at the University of Auckland, New Zealand (animal ethics permit No. 001983) and at the University of Western Australia (Indian Ocean Marine Research Centre Watermans Bay, animal ethics permit No. RA/3/100/1506). All work was carried out in strict accordance with the guidelines of the New Zealand Code of Ethical Conduct and the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes (8th edition, 2013). Animals were monitored daily before, during, and after behavioral testing to ensure their health and well-being.
Six juvenile school sharks (3 males; 510-570 mm in total length), 6 juvenile spotted estuary smooth-hound sharks (5 males; 550-610 mm in total length), and 1 mature spiny dogfish shark (female; 910 mm in total length) were collected from the Kaipara Harbour, New Zealand (−36° 24.10′ S, 174° 23.30′ E). The animals were collected via longlining (25-m longlines) and angling. All angling and longline hooks used were circular to prevent gut-hooked animals. Eight juvenile draughtsboard sharks (4 males; 320-450 mm in total length) were collected from the waters around Leigh, Little Barrier Island, and Great Barrier Island, New Zealand (−36° 04′02.3 S, 175° 19′24.2 E) using customized lobster pots. All animals were collected within the Ministry of Primary Industries’ recreational limits. Upon capture, live animals were placed into a portable, aerated seawater tank and transported to the Leigh Marine Laboratory. Eight juvenile Port Jackson sharks (6 males; 270-320 mm) were collected from waters around Perth, Australia, by an aquarium supplier and delivered to the Indian Ocean Marine Research Centre at Watermans Bay.
All species were housed indoors (in groups of no more than 6 individuals). Round, plastic housing tanks (diameter: 1800 mm, depth: 1600 mm, 4000-L capacity at Leigh Marine Laboratory; diameter: 1000 mm, depth: 600 mm, 500-L capacity at Watermans Bay) were fitted with a filtered (50 µm at Leigh; 30 µm at Watermans Bay) and temperature controlled (16-18 °C at Leigh; 18-20 °C at Watermans Bay) flow-through system (complete, hourly water changes). These setups allowed us to maintain water quality (temperature, oxygen content, salinity) and control environmental conditions (light intensity, light regime, and the timing of feeding) for the entire duration of the experiments. Animals began to feed within 24 h of collection and were fed every 72 h thereafter on a diet of pilchards and squid. Animals were kept under natural light conditions (from sunlight through windows in the housing tank rooms) for a minimum of 2 weeks before the commencement of experimental video recordings.
Experimental Setup
The experimental setup was similar to the prerecording housing conditions, except that the animals were now housed individually in smaller tanks. School sharks, smooth-hounds, and dogfish were placed in round, plastic tanks (diameter: 1900 mm, depth: 600 mm, 1500-L capacity). Port Jackson and draughtsboard sharks were placed in rectangular, glass tanks (900-mm long, 500-mm wide, 500-mm deep, 225-L capacity; 1200-mm long, 600-mm wide, 540-mm deep, 390-L capacity, respectively). Experimental tanks were situated within a windowless room to remove any uncontrolled sources of light. Each tank was isolated with a lightproof curtain to prevent unwanted light and visual stimulation from adjacent tanks and the research team. Waterproof (silicone injected), infrared (IR) light-emitting diode (LED) strip lights (12-V DC, 850-nm single chip, 120 LEDs 9.6 W/m) were fixed equidistant from one another along the length of the floor of each tank with aquarium-grade silicone (strips were spaced 110-mm apart in round tanks; 100-mm apart in rectangular tanks; Fig. 1a) and were left on at all times during behavioral tests. The wavelength (850 nm) of these IR lights fell outside the visual range of all species of sharks (Hart et al., 2011) but provided illumination for IR-sensitive cameras, enabling the researchers to track the natural movements of the animals under dark conditions. A diffuser made from opal-colored, acrylic sheeting (3-mm thick) was placed above the LED lights to provide a homogenous background for the automated tracking of animals (Fig. 1b) and was held in place 100 mm above the tank floor by evenly spaced clear, acrylic blocks (20 blocks in circular tanks; 9 blocks in rectangular tanks),which were fixed to the tank floor with aquarium-grade silicone. A 12:12 light:dark light regime was provided by a programmable, marine spectrum, aquarium LED light (Zetlight Lancia ZP4000 Marine Light, 1200 mm, 46 watt). The light was hung centrally above each tank (1950 mm above round tanks; 980 mm above rectangular tanks), to a purpose-built, overhead frame and a diffuser of 1-mm polytetrafluoroethylene sheeting was placed over each aquarium LED light to minimize the light’s reflection in the seawater below. Each aquarium light was controlled by a Zetlight I200 Lancia ZP-4000 controller, which enabled light-intensity control programming. A light logger (Hobo Pendant Temperature/Light Data Logger UA-002-xx) was also used to cross-reference light intensity over a 24-h period prior to the commencement of behavioral trials. An IR-sensitive camera (Altronics, 4.0 megapixel, weatherproof vari-focal IP PoE), fitted with a photographic red gel filter (Lee Filters, Pop Red), was placed centrally above each tank (fixed to the overhead bracket, 30 mm below the overhead aquarium light), facing downward toward the tank to capture video footage of the sharks. The red filter gel and the cameras’ sensitivity to IR light resulted in video footage that was unaffected by changes in light regime set by the overhead aquarium light, enabling the capture of clear footage in both light and dark conditions. Video footage was recorded, and a live feed was observable, via the use of personal video surveillance software (Security Monitor Pro, Deskshare, Plainview, NY), installed on a desktop computer in an adjacent room. All electrical equipment was connected to an uninterrupted power supply (Powershield Defender PSD 1200).
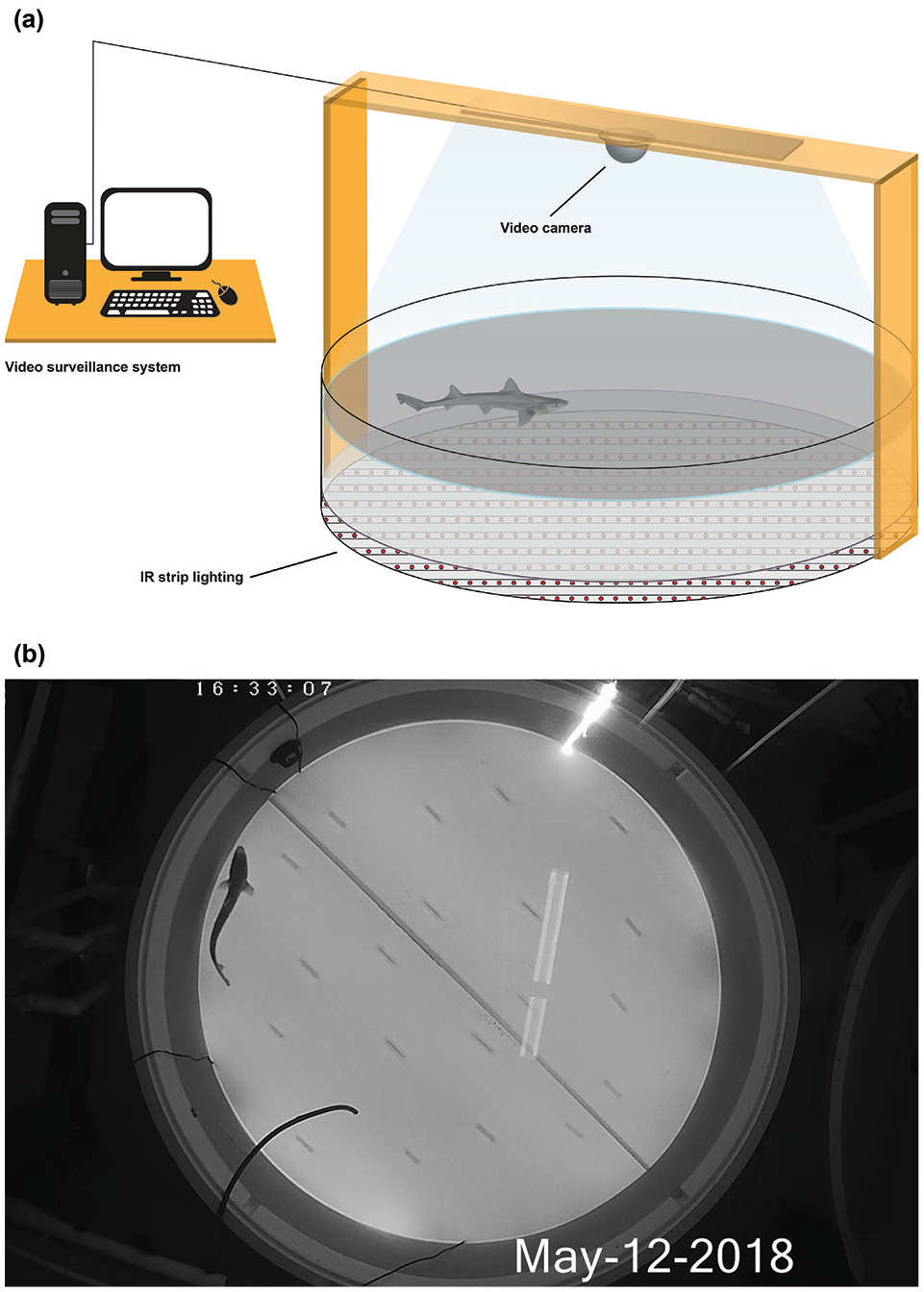
(a) Experimental apparatus used to study the activity rhythms of ram ventilating sharks. Waterproof infrared (IR) light-emitting diode (LED) light strips were secured to the bottom of a seawater-filled tank and were diffused by an acrylic sheet. A controllable, overhead aquarium LED light provided a 12:12 light:dark cycle, and an overhead IR-sensitive video camera captured animal behavior at 2 frames per second. The digital data were recorded and stored on a computer. (b) A recording in progress as seen from above via an IR-sensitive video camera. The shark swimming above the acrylic sheet provides a clear silhouette of its form, enabling the tracking of its movements and behavior.
Three-Day Experimental Protocol
Animals were placed into individual experimental tanks and given 72 h to acclimatize under 12:12 light:dark conditions. Animals experienced a 30-min, stepwise dawn and dusk at 10.8 lux (comparable to twilight) from 0700 to 0730 h and 1830 to 1900 h, respectively. Daytime light was set to 80 lux from 0730 to 1830 h. This is comparable to visible light at approximately 5 m under water on an overcast day. At night (1900-0700 h), there was no light at all, as confirmed by the aforementioned light logger. The sharks were fed 30 h before the commencement of experiments and then again at 0100 h on the second morning of the experiments (at the normal 72-h feeding interval). The animals were otherwise left undisturbed and were monitored remotely by live-feed video. The entire 3-day experiment was recorded on video for later analysis.
Nine-Day Experimental Protocols
Because we observed a marked activity rhythm for the benthic species (Port Jackson and draughtsboard sharks) in the 3-day protocol, we conducted a series of 9-day protocols, which incorporated 3 days of a modified light regime in between two 3-day periods of a standard 12:12 light:dark cycle. Each animal was exposed to 3 different light regimes in the middle 3-day period: (1) 3 days of constant dark (0:24) and (2) 3 days of constant light (24:0) to determine whether the cyclic organization of activity could be retained in the absence of light cues; and (3) 3 days of forced desynchrony (6:6:6:6) to investigate the possibility of reentrainment of activity cycles to an ultradian pattern. Animals were given a minimum of 5 days’ recovery time under 12:12 light:dark conditions between experiments. During the experiments, all animals were held in individual tanks and visually isolated from their neighbors. During the modified light regime protocols of constant light and constant dark, half of the animals (situated on one side of the room) experienced the constant dark protocol, whereas the other half (on the other side of the room) experienced the constant light protocol to control for time-correlated noise variables. The protocols were then switched to ensure all animals experienced each protocol.
Video Analysis
Video clips of behavioral trials were analyzed with the video analysis software Digi developed by J.M.H. (Smolka et al., 2011) in MATLAB (Release 2015b, The MathWorks, Inc., Natick, MA). Shark positions were automatically determined at 2 frames per second by comparing each video frame with a background image that did not contain a shark. In the resulting difference matrix, using a blob analysis in MATLAB, the largest blob closest to the position of the shark in the previous image was taken as its new position. Distance traveled per minute was calculated by filtering the data in time with a boxcar filter of 1-min length (120 frames) and then subsampled to 1 sample per minute. Animals were deemed active if they moved more than 60 mm/min; this threshold smoothed out the tracking noise introduced by image-processing noise and slight, locomotion-independent movements.
Statistical Analysis
All statistical analyses were completed in either MATLAB (MATLAB Release 2015b, The MathWorks, Inc.), R (R Core Team 2017, R, R Foundation for Statistical Computing, Vienna, Austria), or SYSTAT (SYSTAT Software, Inc., Tools for Science Version 13.2, Chicago, IL). Mixed-effects models were used to test for (1) the effects of light regime and time of day on activity patterns and distance traveled in the 3-day protocols, in all species, but the spiny dogfish, and (2) the effects of light regime, time, treatment, and interactions (treatment by day and treatment by light regime) on activity patterns in the Port Jackson and draughtsboard sharks. As we recorded only a single spiny dogfish, no statistical analysis was possible. Median activity was used for the school sharks and smooth-hounds as activity data were nonnormal. Mean activity and distance traveled were used in all other cases.
Results
Upon transition to experimental tanks, all sharks settled well, fed, and appeared to acclimatize for the duration of the protocols.
Three-Day Experimental Protocol
School Sharks
Five of the 6 school sharks exhibited 100% activity for the 3-day recordings (Fig. 2a). There was no variation in the amount of swimming or distance traveled between the day and night (activity: F1,27 = 2.97, p = 0.11; distance: F1,27 = 0.02, p = 0.88) or across the 3 days (activity: F2,27 = 0.59, p = 0.56; distance: F2,27 = 0.92, p = 0.41; Table 1a). Therefore, captive juvenile school sharks swam continuously, at an even speed, across the 24-h day. Note that the sixth shark showed intermittent reductions in swimming, mostly at night, when it remained motionless on the tank floor. These bouts of inactivity were usually brief, typically less than 30 min.
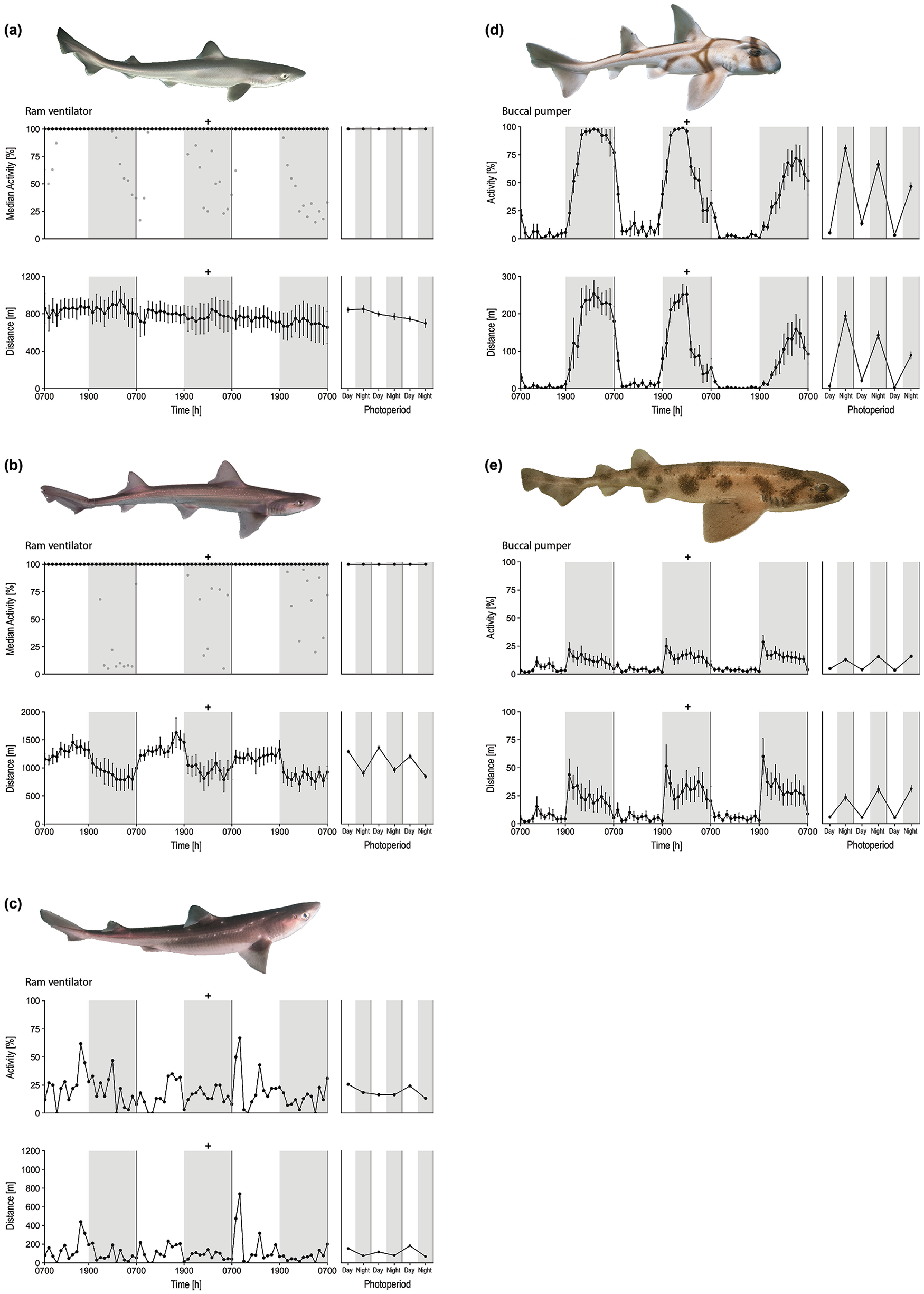
Three-day protocol showing each species and mode of respiration (labeled as ram ventilator or buccal pumper; top), percentage of each hour spent swimming (middle left), and distance traveled per hour across three 24-h days (bottom left), denoted by black dots, for the (a) school shark, (b) spotted estuary smooth-hound, (c) spiny dogfish, (d) Port Jackson shark, and (e) draughtsboard shark. Twelve hour means for each day (white background) and night (gray background) are plotted as a summary (middle and bottom right); “+” denotes the timing of feeding. For the activity plots in (a) and (b), the black dots indicate the hourly median, as data were not normally distributed. The gray dots show the unusual animals that did stop periodically (1 per species). Elsewhere, the data are shown as mean ± SE, except for the spiny dogfish (n = 1). Photos (a)–(c), (e) by Richard Taylor; photo (d) by Andy Murch/Bigfishexpeditions.com.
Results of mixed-effect models for the 3-day plots shown in Figure 2 (letters a, b, d, e in the table match those in the figure) for the amount of activity and the distance traveled over time.
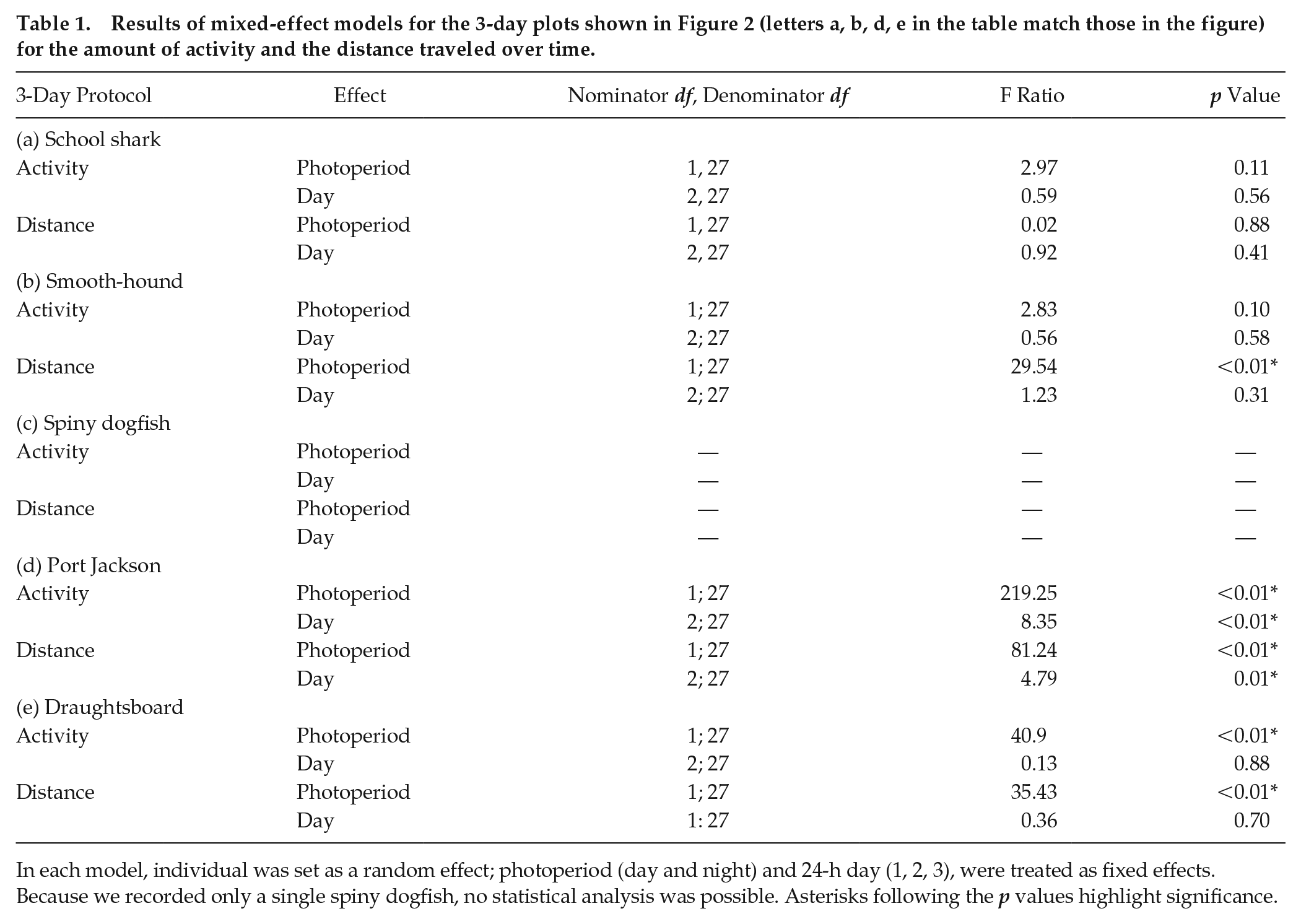
In each model, individual was set as a random effect; photoperiod (day and night) and 24-h day (1, 2, 3), were treated as fixed effects. Because we recorded only a single spiny dogfish, no statistical analysis was possible. Asterisks following the p values highlight significance.
Spotted Estuary Smooth-Hounds
As with the school shark, 5 of the spotted estuary smooth-hounds swam constantly across all days. A single animal showed a reduction in swimming at night (Fig. 2b). There was no difference in the percentage of time spent swimming between day and night (F1,27 = 2.83, p = 0.10) or across the 3 days (F2,27 = 0.56, p = 0.58; Table 1b). Unlike the school shark, however, the smooth-hound traveled significantly farther during the day than during the night (F1,27 = 29.54, p < 0.01). During the day, these sharks were the fastest of all the species studied, as they swam more than 25 km each 24-h day.
Spiny Dogfish
The single spiny dogfish studied remained motionless on the tank floor for an average of 80% of the 24-h day. The animal appeared to rest a little more during the night (Fig. 2c). This pattern was mirrored by a similar preference to swim farther during the day than during the night.
Port Jackson Sharks
The Port Jackson shark showed the most dramatic difference in day-night swimming, with all animals engaging in bouts of extended inactivity (during which the animals remained motionless on the tank floor) during the day (Fig. 2d). This was followed by a significant increase in sustained periods of swimming at night (F1,27 = 219.25, p < 0.01), often continuously swimming for hours at a time (Table 1d). The amount of swimming decreased across the 3 days (F2,27 = 8.35, p < 0.01). The distance these sharks traveled was significantly higher at night (F1,27 = 81.24, p < 0.01) but also decreased over the recording (F2,27 = 4.79, p = 0.01), notably after feeding.
Draughtsboard Sharks
The draughtsboard shark was the least active of all sharks studied, swimming just 300 m per 24-h day (Fig. 2e). However, this species showed a significant preference for nocturnal swimming (F1,27 = 40.90, p < 0.01), which was unchanged across the 3 days (F2,27 = 0.13, p = 0.88; Table 1e), and therefore led to the animals traveling more at night (F1,27 = 35.43, p < 0.01).
Nine-Day Protocol
To test whether the pronounced day-night differences of the Port Jackson and draughtsboard sharks was driven by light regime or an endogenous circadian rhythm, we conducted 3 experiments that incorporated a 3-day modified light regime (namely, 3 days of constant darkness, or constant light, or a forced desynchrony protocol of alternating light-darkness every 6 h) in the middle of the 9-day protocols. Here, we show only the amount of swimming data, as the data on the distance traveled are a near-perfect mirror of percentage activity for both species.
Port Jackson Sharks
The Port Jackson shark showed a marked increase in swimming at night, across all 6 nights under 12:12 light:dark in all 3 light treatment experiments. When switched to constant darkness (Fig. 3a), the sharks elicited an attenuated baseline rhythm, which was retained for the first 24 h (Table 2a). Any semblance to the baseline pattern was lost after 24 h of constant dark conditions. Activity levels decreased dramatically under constant light conditions, although the sharks swam more during the subjective night than during the subjective day for the first 24 h and activity decreased over each 3-day treatment (Table 2b). Two interesting patterns were present under the forced desynchrony protocol: (1) on the first and third subjective nights, the sharks swam more than during the subjective day, and (2) increased swimming could be elicited during the 6-h dark phases irrespective of when they occurred. Related to point 1, Port Jackson sharks retained the baseline (12:12 light:dark) activity rhythm during day 4 and day 6, although they displayed a reverse pattern on day 5. Related to point 2, the sharks also showed intermittent significance for an ultradian activity pattern, driven by the novel 6:6:6:6 light regime, by swimming more in the dark. In all instances, when the 12:12 light:dark light regime resumed, so too did the marked day-night difference in the amount of swimming (Table 2c). In each 9-day protocol, activity decreased in the 24 h after feeding.
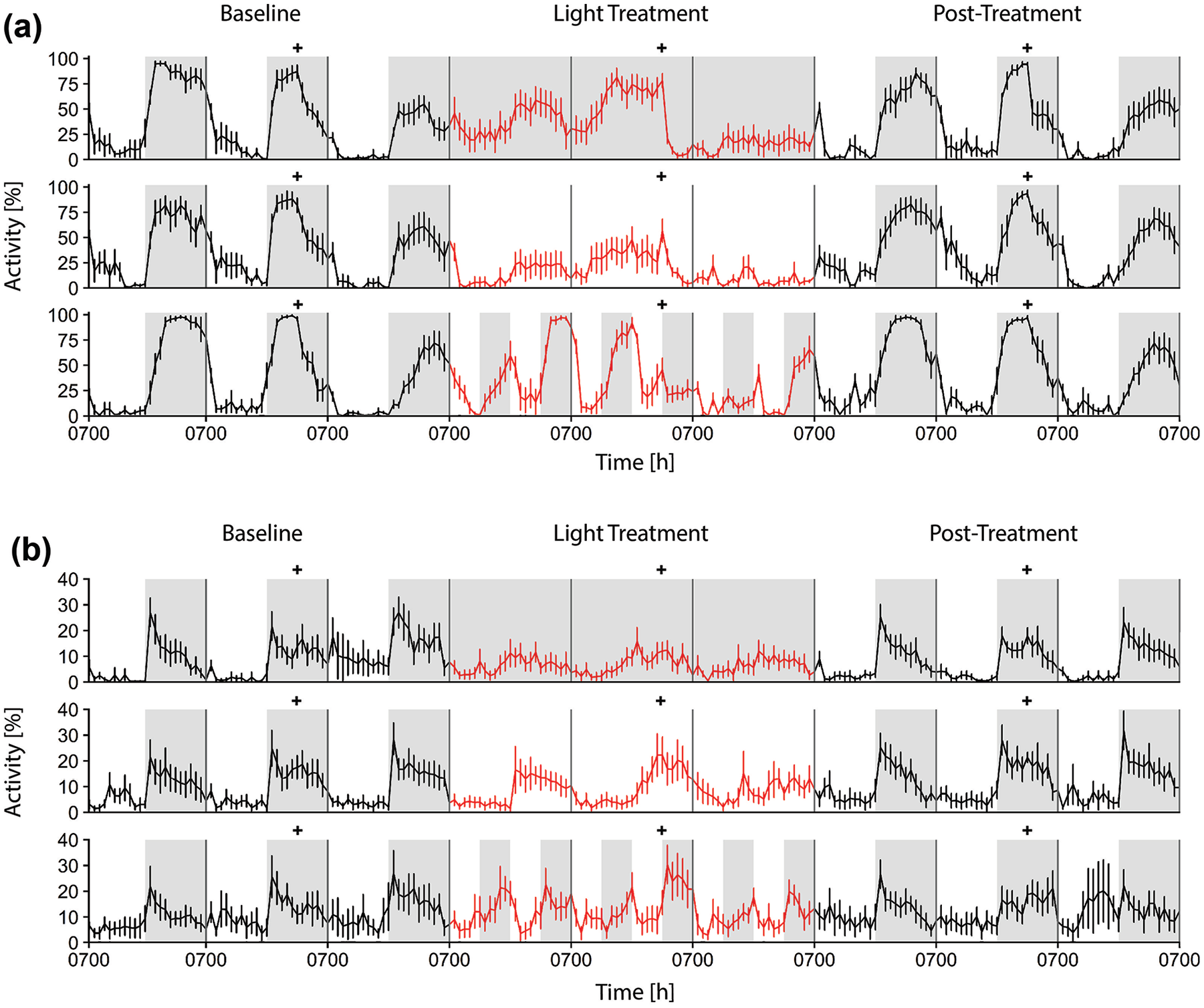
Nine-day protocols showing the percentage of each hour spent swimming under 3 days of a 12:12 light:dark light regime (baseline, in black), 3 days of a modified light treatment (constant dark 0:24, or constant light 24:0, or forced desynchrony 6:6:6:6; in red), followed by 3 days of 12:12 (posttreatment, in black) in (a) Port Jackson sharks and (b) draughtsboard sharks. Gray shading denotes the dark phase of the light regime. Data are presented as mean ± SE. + indicates time of feeding. Color version of the figure is available online.
Results of mixed-effect models for the 9-day plots shown in Figure 3 for the amount of activity.
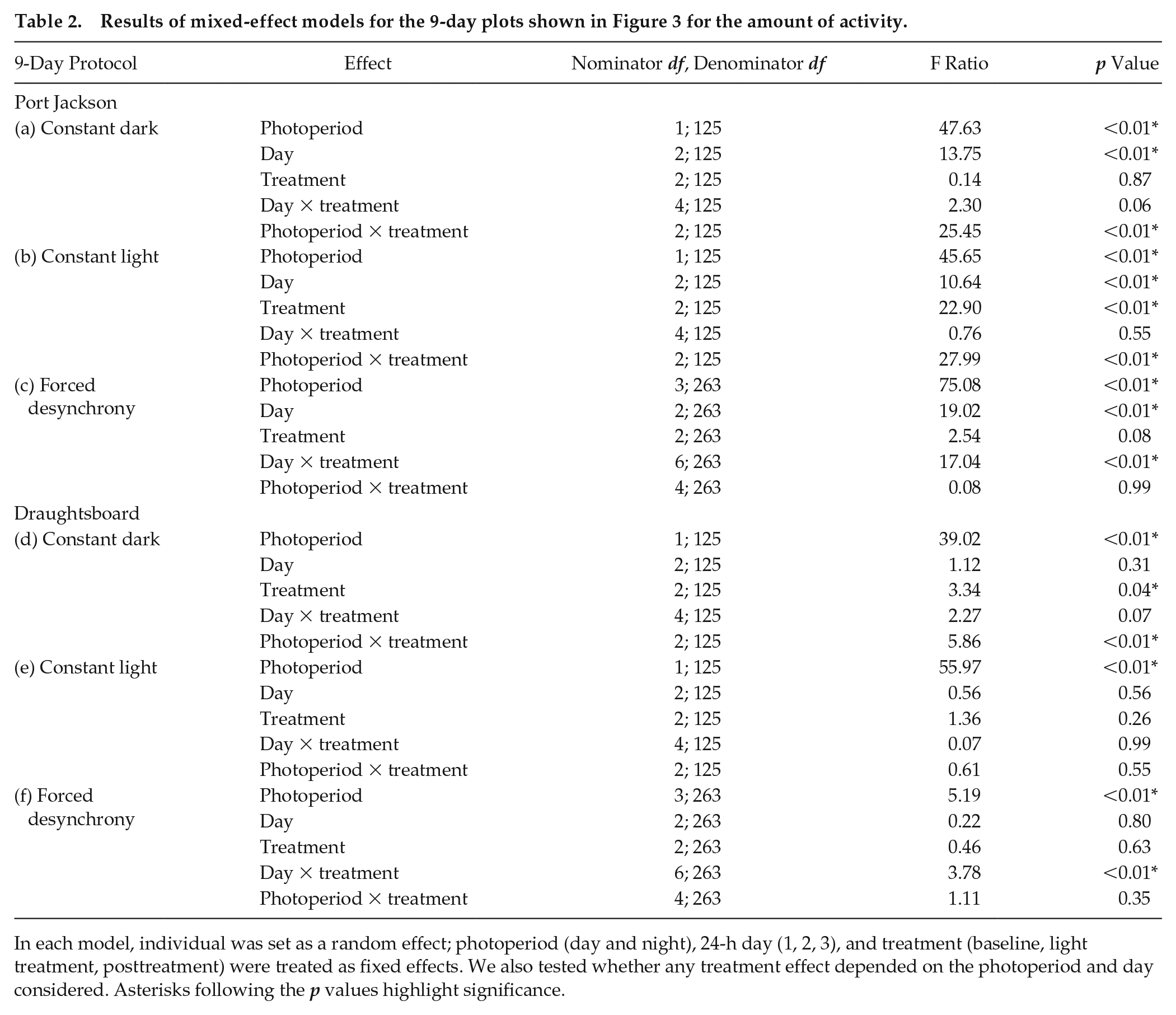
In each model, individual was set as a random effect; photoperiod (day and night), 24-h day (1, 2, 3), and treatment (baseline, light treatment, posttreatment) were treated as fixed effects. We also tested whether any treatment effect depended on the photoperiod and day considered. Asterisks following the p values highlight significance.
Draughtsboard Sharks
Data from the much less active draughtsboard shark were less clear, in part because the baseline activity pattern under 12:12 light:dark, namely, increased activity at night, was less pronounced (Fig. 3b). However, these sharks could show increased swimming during the subjective nights of constant darkness and constant light conditions (Table 2d, e). As in the Port Jackson shark, this pattern was most prominent under the preceding (baseline) and succeeding (posttreatment) 12:12 light:dark conditions. Under the forced desynchrony protocol, draughtsboard sharks showed a nonsignificant ultradian rhythm, suggesting that they also track the light regime, but further study is needed (Table 2f).
Discussion
We identified a surprising diversity of activity rhythms across 5 shark species. Even closely related species that share the same method of ventilation showed marked differences in their temporal organization and intensity of swimming. Specifically, school sharks and smooth-hounds swam nonstop during all recordings. However, while the school sharks maintained speed and distance traversed across the day and night, the smooth-hounds swam more slowly and traveled less during the night. The single spiny dogfish, thought to be a ram ventilator, was shown to rest, immobile, on the tank floor for periods of time, indicating that this shark is capable of buccal pumping. While both of the buccal pumping sharks, the Port Jackson and draughtsboard sharks, are clearly nocturnal, the magnitude of this variation differed greatly between the 2 species, with the Port Jackson sharks showing profound day/night variation in swimming behavior and a more muted rhythm in the draughtsboard sharks. Such diversity in activity rhythms is seen in other groups of animals as well. Shorebirds have shown a plethora of diverse activity patterns, from predictable activity each day, to free-running clocks, to arrhythmic patterns, which are linked to species-specific ecology and life-history strategies (Bulla et al., 2016). The causes for the interspecific variation observed in sharks require further study.
Ram Ventilators: School and Smooth-hound Sharks
School and smooth-hound sharks swam continuously for days. In other animals, sustained periods of quiescence often reflects sleep, where it is accompanied by reduced awareness of the local environment (i.e., decreased sensitivity to stimuli), rapid reversibility to an awake condition, and homeostatic regulation (Tobler, 2011). Unfortunately, there are no systematic data on sleep in elasmobranchs (Kelly et al., 2019), but this finding questions whether these sharks sleep or at least raises interesting possibilities as to the form any sleep might take. We focused our analyses on hourly time bins. It is possible that the animals experience short (seconds) periods of inactivity, during which time they sleep across the day and night, much in the same way that birds package their sleep on short time scales (Lesku et al., 2012; Lesku and Rattenborg, 2014; Rattenborg et al., 2016). Reductions in the amount of sleep have evolved many times in multiple populations of cavefish (Astyanax mexicanus), relative to their surface-living counterparts (Duboué et al., 2012), and the same may have happened in sharks. Alternatively, these sharks might sleep unihemispherically, as observed in continuously swimming marine mammals (Lyamin et al., 2008; Kelly et al., 2019; Rattenborg et al., 2019). On the other hand, ram ventilating sharks might sleep facing into currents, although evidence for this is lacking (Kelly et al., 2019). Future studies should determine whether periods of inactivity or reductions in the intensity of swimming (as in the smooth-hounds at night) represent sleep or something else.
Ram Ventilators: Dogfish Shark
In a single spiny dogfish, thought to be a ram ventilating animal, we observed surprisingly low levels of activity, suggesting that this animal can meet its oxygen requirements by buccal pumping alone. In the purported ram ventilators, sustained periods of inactivity were most obvious in the spiny dogfish, which spent ~80% of the 24-h day inactive and favored swimming more during the day. Karmanova et al. (1975) reported constant swimming in a captive specimen of spiny dogfish; however, our findings align with those of Brett and Blackburn (1978), who were unable to elicit constant swimming in this species. Our results suggest that this animal is a nonobligate ram ventilator that can use both modes of ventilation, favoring buccal pumping for most of the 24-h day in captivity, presumably when oxygen demands are relatively low. It is important to note, however, that these data were collected from only 1 individual and may not be representative of the species in the laboratory or wild environments.
We also observed periods of reduced activity in 1 school shark and 1 smooth-hound. Periods of inactivity and buccal pumping have been reported infrequently in wild species of ram ventilating sharks, except for rare observations of periods of immobility associated with anecdotes of reduced responsiveness in requiem sharks (Carcharhinus leucas, Carcharhinus perezii, Prionace glauca, Negaprion brevirostris; Clark, 1973). However, this appears to be an isolated phenomenon, likely influenced by a unique set of environmental conditions, such as increased levels of oxygen in the water. As we observed sustained restfulness in only a single individual of each ram ventilating species, it is unlikely that such immobility is a common behavior and instead might be related to an (unidentified) feature of laboratory housing.
Buccal Pumpers: Port Jackson and Draughtsboard Sharks
Both buccal pumping species were clearly nocturnal. In the Port Jackson sharks, there was a conspicuous day/night difference in activity, with swimming commencing with the onset of night. This is consistent with activity patterns observed from wild and captive animals of the same species (Kadar et al., 2019). During the 9-day protocol, a number of conclusions were drawn. Under constant conditions, the Port Jackson sharks appeared to swim more during the subjective night, which is indicative of an endogenous drive for activity at that time of the 24-h day. Nonetheless, constant light had a disruptive effect on this rhythm for several days. Furthermore, during the forced desynchrony protocol, the Port Jackson sharks swam more during some of the 6-h dark phases irrespective of when they occurred. Taken together, this indicates that light cues are a strong determinant of activity levels and can override clock-mediated regulation in this species. A drop in swimming activity and distance traveled was observed in the third, sixth, and ninth day of each protocol, which occurred shortly after feeding. This is likely due to postfeeding increases in metabolic rate due to specific dynamic action. With a larger proportion of their aerobic scope taken up by digestion, less energy can be devoted to swimming (Owen, 2001). This dramatic drop in postfeeding activity may be a factor that affects diel activity rates of this species in the wild. Many benthic sharks rely on the sensory advantages they have over prey in low-light conditions by foraging at night (Kempster et al., 2015; Hammerschlag et al., 2017). The decreased activity during the day following prolonged activity at night observed in wild Port Jackson sharks (Kadar et al., 2019) may therefore arise from the digestive processes of sharks after nocturnal feeding events.
The draughtsboard sharks showed a similar (yet smaller magnitude) preference for nocturnal activity. Unlike the Port Jackson sharks, the draughtsboard sharks were better able to maintain rhythmicity under constant conditions, suggesting that they have a stronger circadian clock and were less dependent on light cues. That said, we observed a (nonsignificant) modulation of swimming under the forced desynchrony protocol, and further study is warranted. Evidence for a circadian clock has been found in other buccal bumping sharks. Nelson and Johnson (1970) observed that although both the horn (H. francisci) and the swell (Cephaloscyllium ventriosum) shark began nighttime swimming and feeding with the onset of darkness, the swell shark maintained this rhythmicity under constant light and constant dark conditions, and the horn shark maintained rhythmicity under constant dim light in captivity (Finstad and Nelson, 1975).
Timing of Activity
For the most part, sharks elicit a nocturnal or crepuscular lifestyle in both the wild and captivity (as reviewed by Hammerschlag et al., 2017; Kelly et al., 2019). The decrease in nighttime activity levels observed in the smooth-hound and spiny dogfish, however, suggests that aspects of their behavior were day active. In the smooth-hound, this activity was more subtle, manifesting as nonstop swimming over 24 h but faster swimming over a greater distance during the day. Such diurnality has been reported in captive bonnethead sharks (Sphyrna tiburo), who engaged in patrolling behavior throughout the day and decreased both swimming speed and distance covered at night (Myrberg and Gruber, 1974). In the case of bonnetheads, a behavioral pattern may have been trained into the animals because of their consistent feeding in the afternoon by the researchers. The animals may have learned to anticipate the feeding time, reducing the pressure to forage throughout the night. In our study, we strived to avoid this scenario by consistently feeding the animals in the middle of the “dark phase,” and yet they still showed evidence of decreased activity at night.
Conclusions
This study has revealed the presence of an impressive diversity in the activity rhythms of sharks, including in a number of species whose activity patterns had gone unstudied. We have also highlighted the striking variation in the presence (and absence) of the circadian organization of these rhythms under environmentally controlled conditions, even among species that seemingly share similar lifestyles. Both the school and smooth-hound shark swim nonstop, and yet, although the school shark shows no circadian variation to its activity, the smooth-hound travels less at night. How either of these species obtains a daily amount of sleep is unknown and warrants further study. In contrast to some earlier reports, the spiny dogfish is a mostly inactive shark and swims more during the day. The Port Jackson shark shows a dramatic, light-driven, nocturnal activity pattern that is temporarily retained in the absence of cues, and the draughtsboard shark shows a similar rhythm to the Port Jackson shark, albeit less pronounced, with a stronger retention under constant conditions. Although this is the first study of its kind to show the endogenous nature of activity patterns in these species of sharks, the presence of endogenous rhythms in other closely related species (such as horn and swell sharks) suggests this may be a common evolutionary adaptation, at least in buccal pumping sharks. Given the remarkable diversity in the activity rhythms of sharks highlighted in this study, there is a clear need for more circadian rhythm studies to find the causes, consequences, and adaptive value of these patterns.
Footnotes
Acknowledgements
The authors would like to acknowledge Stefan Spreitzenbarth for his contribution to animal collection and husbandry as well as experimental construction, Peter Browne for his contribution to experimental construction, Brendan Jeffrey for his contribution to animal collection and husbandry, Martin Lefley for his contribution to animal collection, Noah Radford for his contribution to animal collection, and John Montgomery for his contribution to species identification. We would also like to express our sincere gratitude and appreciation for the continued support and generous financial assistance provided by Craig and Katrina Burton. This study was supported by the University of Western Australia, the University of Auckland, the Endeavour Leadership Program, and the Sea World Research & Rescue Foundation Inc.
Author Contributions
M.L.K., E.R.P.M., C.C.K., S.P.C., and C.A.R. collected all animals and were responsible for the husbandry and maintenance of animals and experiments; M.L.K. conducted all experiments; M.L.K., J.A.L., and J.M.H. produced all figures; M.L.K and J.A.L wrote the first draft; all authors then contributed to editing the manuscript.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
