Abstract
Investigators typically study one function of the circadian visual system at a time, be it photoreception, transmission of photic information to the suprachiasmatic nucleus (SCN), light control of rhythm phase, locomotor activity, or gene expression. There are good reasons for such a focused approach, but sometimes it is advantageous to look at the broader picture, asking how all the parts and functions complete the whole. Here, several seemingly disparate functions of the circadian visual system are examined. They share common characteristics with respect to regulation by light and, to the extent known, share a common input neuroanatomy. The argument presented is that the 3 hypothalamically mediated effects of light for which there are the most data, circadian clock phase shifts, suppression of nocturnal locomotion (“negative masking”), and suppression of nocturnal pineal function, are regulated by a common photic input pathway terminating in the SCN. For each, light triggers a relatively fixed interval response that is irradiance-dependent, the effective stimulus can be very brief light exposure, and the response continues to completion in the absence of additional light. The presence of a triggered, fixed-length response interval is of particular importance to the understanding of the circuitry and mechanisms regulating circadian rhythm phase shifts because it implies that the SCN clock response to light is not instantaneous. It also may explain why certain stimuli (neuropeptide Y or novel wheel running) administered many minutes after light exposure are able to block light-induced phase shifts. The understanding of negative masking is complicated by the fact that it can be represented as a positive change, that is, light-induced sleep, not just as a reduction in locomotion. Acute nocturnal light exposure also induces adrenal hormone secretion and a rapid drop in body temperature, physiological responses that appear to be regulated similarly to the other light effects. The likelihood of a common regulatory basis for the several responses suggests that additional light-induced responses will be forthcoming and raises questions about the relationships between light, SCN cellular anatomy, the molecular clockworks of SCN neurons, and SCN throughput mechanisms for regulating disparate downstream activities.
Circadian rhythm investigators “know” what masking is despite the absence of a rigorous definition. In his review of the topic, Mrosovsky (1999) defined negative masking in nocturnal animals as “a decrease in activity” and positive masking as “an increase in activity.” Other varieties of masking have also been defined (Mrosovsky, 1999). Most investigators are familiar with negative masking as an index of light’s suppressive effects on wheel-running behavior. Masking has also been used much more generally in reference to the extent to which virtually any intrinsic or extrinsic stimulus impairs expression of an action of the circadian clock. For example, Redlin (2001) considers light-induced suppression of wheel-running and melatonin to be negative masking responses (also see Mrosovsky, 1999, for discussion and references). Rensing prefers to consider the stimulus effects as “direct” effects rather than as “masking” effects (Rensing, 1989). Redlin has combined terms, referring to masking as resulting from direct effects of light (Redlin, 2001).
Redlin’s suggestion that light-induced activity suppression and inhibition of melatonin production are both masking phenomena illustrates some of the semantic confusion surrounding the concept. This arises because the two variables are fundamentally different. Melatonin level is a physiological measure that varies on a continuum from low to high, according to the prevailing conditions, and it is not known to covary with other measures. In contrast, measures of “negative masking” have been used as indices of behavioral change, without considering the fact that by definition, all behaviors must always sum to 100%. In other words, when light reduces activity to 0% of ongoing behavior (meaning that “negative masking” reaches 100%), at least 1 alternate behavior must have correspondingly increased. If we could identify and measure that elevated behavior, would it be an index of positive masking or negative masking? Clarification of this semantic difficulty is necessary in order to uncover organizing principles governing the effects of light on physiology and behavior.
A second problem regarding the historical use of “negative masking” is the implication that the behavioral change to which it refers is contingent upon the presence of light. A second assumption is explicit: masking effects of light occur “by a route that does not involve a pacemaker” (Mrosovsky, 1999). Despite wide acceptance of both characteristics (Aschoff, 1960, 1981; Minors and Waterhouse,1989; Mrosovsky, 1999; Redfern et al., 1994; Redlin, 2001), neither expectation has been proven. More important for the purposes of the present discussion is the recent demonstration that sustained suppression of locomotion does not require the continued presence of light (Morin and Studholme, 2009; Vidal and Morin, 2007). Moreover, research questioning the relationship between light and negative masking has revealed several unexpected results. One is the occurrence of light-induced sleep, hereafter referred to as “photosomnolence.” The fact that nocturnal light induces sleep in mice and hamsters (Morin and Studholme, 2009, 2011; Studholme et al., 2013) is a clear example of the above point that if one behavior becomes less likely (e.g., active wakefulness), at least one other behavior must become more likely (e.g., sleep).
The revelation of photosomnolence requires reconsideration of what negative masking actually represents in order to understand how light modifies physiology and behavior. Here, it is proposed that light induces circadian rhythm phase shifts and suppresses pineal function in ways that are remarkably similar to the effects of light on negative masking. Indeed, it is plausible that all 3 responses, mediated through the circadian visual system, share a single, light-activated, time-limited mechanism necessary for their expression which, once initiated, continues to completion without additional light being necessary.
The present discussion focuses on the above highlighted issues, emphasizing the need for better understanding of the nature of light-induced changes in physiology and behavior while addressing some of the ramifications that the issues may present to circadian rhythm research and the understanding of global SCN function. For the remainder of this presentation, masking refers to negative masking unless otherwise specified and, to avoid semantic confusion, will be generally replaced by the term locomotor suppression.
Locomotor Suppression and Phase Shifting Are Simultaneously Induced by Light
Mrosovsky used several novel ways of assessing light-induced locomotor suppression, but the basic procedure has been to provide nocturnal rodents with a running wheel under a standard light-dark (LD) photoperiod and expose them to a 30- or 60-min light pulse during the early activity phase (Mrosovsky, 1999; Mrosovsky et al., 1999 2000, 2001; Redlin and Mrosovsky, 1999b). In a typical study with saturating light, running declines to zero within a few minutes, where it remains during continued exposure to the stimulus (Redlin and Mrosovsky, 1999b).
Phase shift studies are conducted similarly. Depending on whether an Aschoff Type I or II procedure is used, animals are either in constant dark (DD) or LD and are exposed to a 5- to 60-min light pulse. In rhythm studies, a great deal of emphasis has been placed on effects of light on the circadian clock and the phase response curves (PRCs) that describe those effects. Masking receives little or no mention. Nevertheless, it should be abundantly clear that procedures for demonstrating masking and phase shifting are essentially identical and that every phase shift study is, by default, a masking study. The major difference between the two is the elapsed time before a result can be considered reliable. From a practical perspective, useful results about negative masking can be obtained within a few minutes of stimulus onset, whereas reliable phase shift detection requires many hours or days after the stimulus to collect the data.
More bluntly, 2 different responses are elicited via the identical stimulus (Fig. 1). One is considered to be a demonstration of the most important function of the circadian visual system, namely, the ability to maintain a stable temporal relationship with the environmental photoperiod through the phase shifting action of light on the circadian clock. Masking, in contrast, is usually considered (if it is considered at all) to be a hindrance to circadian rhythm assessment, a mere by-product resulting from a procedure by which light is used to induce a phase shift. Because identical methods are used to test phase shifting and masking, an obligatory conclusion is that in the absence of other qualifying information, experiments designed to evaluate how light alters intrinsic function of the circadian clock (i.e., the SCN) cannot distinguish between effects on clock phase and effects on masking. Mrosovsky (1999) lamented, with good reason, that so “little study has been devoted to masking compared to that lavished on entrainment.”
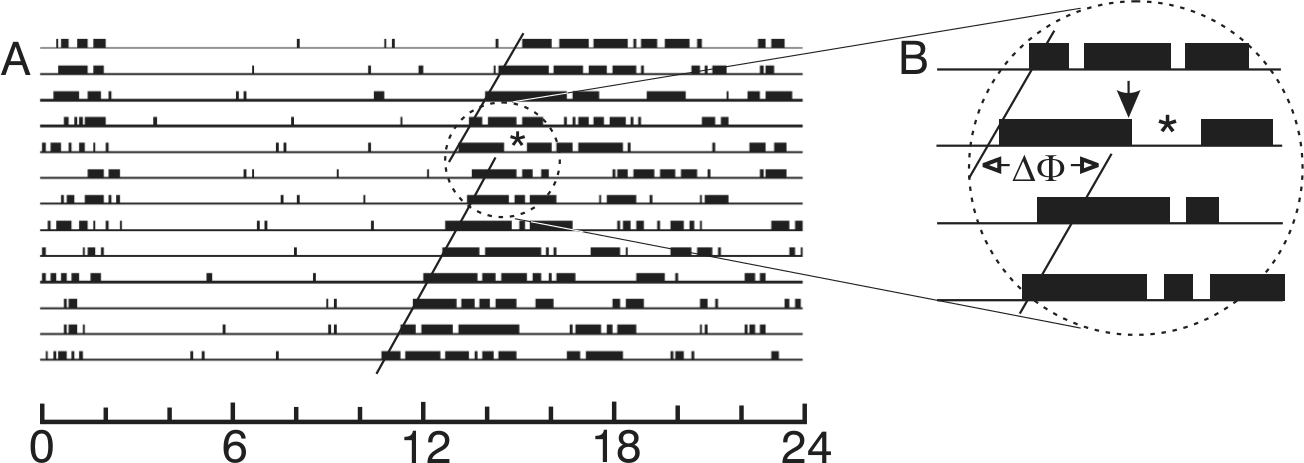
A millisecond light stimulus effective for eliciting phase shifts is also effective for eliciting a masking response. Here, several days of a running record obtained from a constant dark-housed mouse (Mus musculus) are shown. The stimulus consisted of 10 flashes of 2 msec each, distributed equally across a 5-min interval beginning at CT13. In (A), the circadian periods before and after the stimulus are indicated by the slope of each black line fitted through the daily activity onsets. The effect of the light stimulus yielded a subsequent phase shift indicated by the Greek letters ΔΦ in the enlargement (B). The exact time of onset for the flash sequence is indicated by the black arrow in (B). The asterisks in (A) and (B) indicate the interval of locomotor suppression induced by the flashes. Modified after Fig. 1 in Morin and Studholme (2009).
The Photic Input Pathways for Masking and Phase Shifting May Be Identical
Circadian rhythm phase shifts and masking respond to photic information arriving in the brain via very similar, if not identical, input pathways involving classical rod/cone and ganglion cell photoreception. As is now well known, about 1% to 2% of mammalian retinal ganglion cells are photoreceptive (pRGCs) (Berson et al., 2002; Hattar et al., 2002) and use melanopsin as a photopigment (Panda et al., 2005; Qiu et al., 2005). Both classical (rod/cone) photoreceptors and pRGCs mediate masking and circadian rhythm phase and account for all non-image-forming visual responses (Hattar et al., 2003; Mrosovsky and Hattar, 2003; Mrosovsky et al., 2001; Thompson et al., 2008). Moreover, photic information received by classical photoreceptors apparently must pass through pRGCs in order to elicit those non-image-forming visual responses (Goz et al., 2008; Guler et al., 2008; Hatori et al., 2008).
Retinal ganglion cells project to about 30 retinorecipient brain regions (Morin and Blanchard, 1999) and the pRGCs project to many of these regions (Ecker et al., 2010; Gooley and Saper, 2003; Hattar et al., 2006; Morin et al., 2003). In particular, pRGCs densely innervate the SCN, intergeniculate leaflet (IGL), and olivary pretectum and less densely innervate the dorsal lateral geniculate and superior colliculus.
It has been known since 1972 that destruction of the SCN eliminates circadian rhythmicity in rodents (Moore and Eichler, 1972; Stephan and Zucker, 1972). Lesions elsewhere in the brain generally have no effect on rhythm expression. Two exceptions are the IGL and pretectal-tectal region, both of which mediate rhythm response to light (Harrington and Rusak, 1986; Marchant and Morin, 1999; Morin and Pace, 2002; Pickard et al., 1987). This information is background for a series of studies by Mrosovsky and colleagues designed to determine which brain region is necessary for masking. Masking has been tested after lesions of the retinorecipient pretectum, tectum, dorsal lateral geniculate, ventral lateral geniculate, IGL, and visual cortex (Edelstein and Mrosovsky, 2001; Mrosovsky, 1999; Redlin et al., 2003; Redlin et al., 1999). The results are unequivocal: masking persists in the absence of these brain regions (although 1 or more may modulate “positive masking” responses to low irradiance).
Considering the foregoing results, the SCN is the most likely location for the control of masking. This, together with the observation that the photic input pathway for circadian rhythm phase control seems to be the same as that for masking, argues by analogy that the SCN is essential to both functions. Unfortunately, 2 tests of SCN contribution to masking have yielded opposite conclusions (Li et al., 2005; Redlin and Mrosovsky, 1999a). The studies were similarly conducted except that Li et al. (2005) used a powerful method (Pickard and Turek, 1982) that determined complete destruction of the SCN by absence of any SCN retinal terminal field. The idea that the SCN regulates masking is also supported by work showing that effects of light are absent from published running records generated by animals with complete transection of the retinohypothalamic tract (RHT), a procedure that blocks entrainment (Johnson et al., 1988a). Similarly, transplantation of an embryonic SCN to an arrhythmic adult restores locomotor rhythmicity without entrainment or apparent masking (Lehman et al., 1987). Thus, complete SCN lesions not only cause arrhythmicity (and loss of entrainment by light) (Rusak, 1977 but also block masking (Li et al., 2005). The existing data support the view that there is no difference between the photoentrainment pathway and the pathway for light-induced locomotor suppression. Although unlikely, it remains possible that the functional pathway for light-induced locomotor suppression terminates not in the SCN but in another retinorecipient hypothalamic region (Johnson et al., 1988b; Muscat et al., 2003).
The Photic Input Pathway for Pineal Regulation Is Similar to that for Masking and Phase Shifting
A third non-image-forming visual response initiated via rod/cone or pRGC photoreception is light suppression of pineal activity (Hattar et al., 2003; Panda et al., 2003). SCN lesions that render animals arrhythmic and abolish locomotor suppression also abolish photic regulation of the pineal (Antle et al., 2007; Klein and Moore, 1979; Li et al., 2005; Rusak and Morin, 1976; Stetson and Watson-Whitmyre, 1976). Such results support the view that photic regulation of pineal function occurs through the same input pathway influencing locomotor suppression and phase shifting.
Very Brief Light Exposure Has Similar Effects on Suppression of Pineal Activity, Induction of Masking, and Phase Shifting
Redlin’s (2001) detailed review describes numerous points of similarity between light suppression of pineal activity and masking. Of particular importance for the present discussion is the fact that melatonin suppression in rodents is initiated by light and persists well beyond the actual presence of light. The “duration” characteristic is important because it also exists for locomotor suppression, but it has received only limited investigative attention or discussion (Antle et al., 2007; Lall and Biello, 2003a; Redlin, 2001). Informal observations (Fig. 1B in Redlin, 2001) show that a light pulse lasting only 15 min is sufficient to induce locomotor suppression that endures for more than an hour. A 1-min light pulse also suppresses melatonin for about the same amount of time (Fig. 1A in Redlin, 2001). The responses of the 2 variables to identical stimuli have not been studied in sufficient depth to know the extent of their similarity.
Exploration of light-induced suppression of pineal function in nocturnal rodents has also demonstrated its persistence in response to mere seconds of light exposure, which is sufficient to elicit a rapid decline in pineal activity (Fig. 2; as indicated by the amount of melatonin or N-acetyltransferase [NAT]) (Illnerova and Vanacek, 1979, 1982; Illnerova et al., 1979; Lerchl, 1995; Reiter et al., 1986). A 1-msec flash of light is sufficient to massively suppress NAT and melatonin within 10 min (Vollrath et al., 1989) (duration of this effect was not examined). In other words, the effect of light on pineal suppression in nocturnal rodents is not contingent upon the continued presence of light.
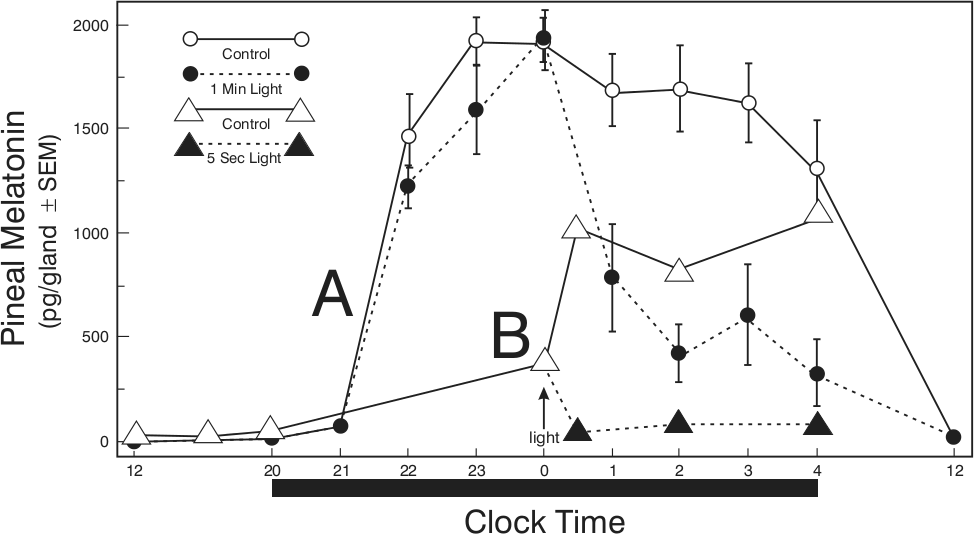
Short-duration light pulses suppress pineal function. (A) Nocturnal pineal melatonin levels in Siberian hamsters (Phodopus sungorus) in a LD 16:8 photoperiod (light off at 2000 h; black bar at bottom of figure) and exposed to 1 min of light (●) or no light (○) at midnight (arrow; data plotted from Lerchl, 1995). (B) Nocturnal pineal melatonin levels in Syrian hamsters (Mesocricetus auratus) housed in a LD 8:16 photoperiod and exposed to 5 sec of light at midnight (▲); (△) no-light controls (data plotted from Reiter et al., 1986).
The fact that brief light exposure is sufficient to elicit prolonged suppression of pineal activity raises the possibility that similar light exposure might elicit locomotion suppression or phase shifts. The answer is unequivocally affirmative. In fact, as few as 4 flashes (each 2 msec long with a 16-sec interflash interval) are sufficient to rapidly induce complete locomotor suppression (Fig. 3) that endures for 35 to 40 min without additional light (Morin and Studholme, 2009; Vidal and Morin, 2007). As indicated in Figure 1, light flash stimuli also elicit robust phase shifts (Morin and Studholme, 2009; van den Pol et al., 1998; Vidal and Morin, 2007).
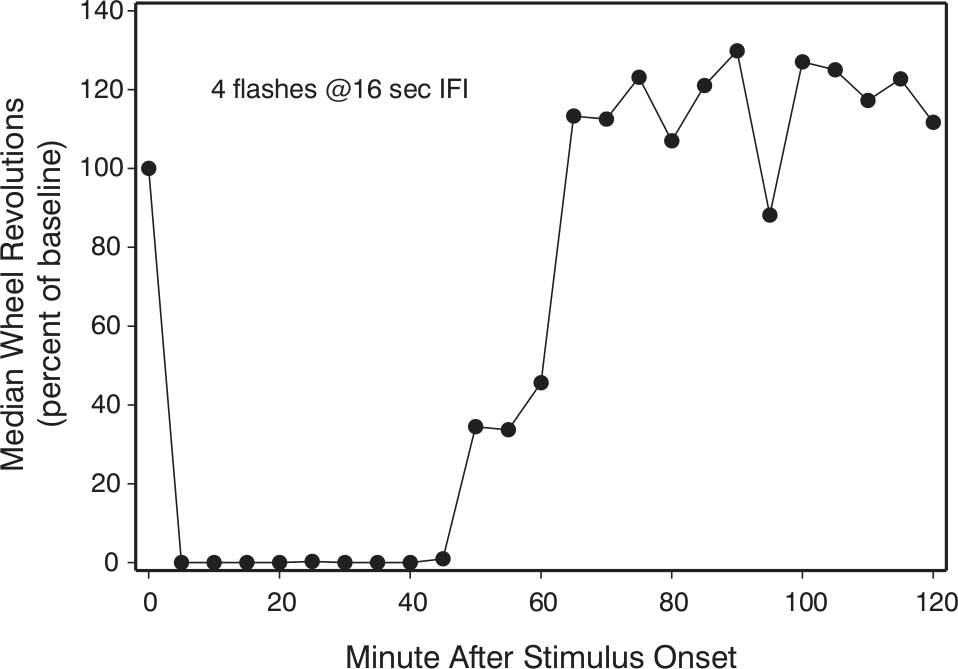
Prolonged locomotor suppression is robustly induced by 8 msec of light presented as 4 equally spaced light flashes, each 2 msec long with the final flash 48 sec after the first flash. Wheel-running declines rapidly with little or no running evident for many minutes in the dark after the last flash. IFI = interflash interval. After data from Morin and Studholme (2009).
Temporal Integration in the Circadian System
The circadian visual system is thought to have the capacity to estimate the energy in a light stimulus and generate phase shifts proportional to that energy. This process of “temporal integration” has been demonstrated by showing reciprocity between stimulus irradiance and duration (Dkhissi-Benyahya et al., 2000; Muscat and Morin, 2005, 2006; Nelson and Takahashi, 1991b). Both light-induced locomotor suppression (Mrosovsky et al., 1999; Redlin and Mrosovsky, 1999b) and light suppression of pineal function (Nelson and Takahashi, 1991a) show typical sigmoidal irradiance sensitivity curves with a shape similar to what is known for phase shifts.
Tests for temporal integration related to locomotor suppression have been absent until recently. The reason for this is almost certainly related to the prevailing assumption that the continued presence of light is required for suppression of locomotion. However, that assumption is incorrect. In fact, tests of flash or pulse stimuli of various durations reveal that light has 2 distinct effects on locomotor suppression. First, it triggers or activates a response that continues for a relatively fixed interval (K) in the absence of further light (Morin and Studholme, 2009). The magnitude of this effect is dependent on irradiance but not on stimulus duration (Morin et al., 2010). Second, if the stimulus endures longer than K, then the light maintains locomotor suppression. In general, such long stimuli will maintain locomotor suppression equal to the stimulus duration (e.g., an 11-h stimulus will induce 11 h of suppression) (Morin et al., 2010), a result consistent with Mrosovsky’s studies with bright 60- or 180-min stimuli (Mrosovsky and Hattar, 2003; Redlin and Mrosovsky, 1999b). Thus, temporal integration cannot be demonstrated for locomotor suppression because of the maintenance effect of light.
The maintenance effect may also confound interpretation of studies designed to test temporal integration with respect to phase shift responses to light pulses (Morin et al., 2010; Nelson and Takahashi, 1991b; see Morin et al., 2010, for a complete discussion).
Temporal integration of photic stimuli is absent when either phase shift or locomotor suppression responses to millisecond stimuli are tested (Morin and Studholme, 2009; Vidal and Morin, 2007). For example, a series of 100 flashes (2 msec each) does not yield either phase shifts or locomotor suppression equal to those in response to 10 flashes at 10 times the energy per flash (Morin et al., 2010; Vidal and Morin, 2007). In other words, 2 stimulus sequences having equal energy do not elicit equal responses for either phase shifting or suppression of locomotion. Similar studies concerning temporal integration and suppression of pineal activity are lacking.
Toward a Synthesis of SCN Function: Acute Photic Modulation of Multiple Systems
To this point, the similarities between photic regulation of circadian rhythm phase, locomotor suppression, and pineal suppression have been discussed, and the fact that light-induced locomotor suppression is actually an index of a positive change, namely sleep induction, has been acknowledged. These facts alter appreciation of how photic information must interact with SCN activity to regulate multiple bodily functions, not just circadian rhythm phase. One prediction is that the initial events in the light-activated sequence are likely to be same regardless of whether the response of interest is photosomnolence, phase shifting, or pineal hormone suppression. At some location, presumably in the SCN circuitry, there will be 1 or more points of pathway divergence that lead to expression of the 3 outputs. Each output response would theoretically be subject to idiosyncratic modification at or beyond the point of pathway divergence.
It is parsimonious to conclude that light triggers 1 event sequence in the SCN to initiate all 3 responses. Therefore, a search for light-dependent changes in the SCN having time constants similar to the interval, K, is likely to be a worthwhile endeavor. One class of physiological processes known to be activated by light during the subjective night is induction of SCN gene expression. The immediate early gene product FOS is induced in cells throughout the SCN by light pulses or a few millisecond flashes (Aronin et al., 1990; Kornhauser et al., 1990; Vidal and Morin, 2007). In the SCN, the region of FOS expression corresponds to that of retinal input (Hattar et al., 2006; Morin et al., 2006). It should be apparent that light will induce FOS or other gene products in the SCN irrespective of whether the behavioral or physiological response being measured is photosomnolence, phase shifting, or suppression of pineal activity. As for the photic input pathway, all retinorecipient SCN cells are likely to participate in regulation of all SCN functions, and the separation of functions occurs further downstream.
Most studies of light and SCN function focus exclusively on issues relating to circadian rhythm entrainment without consideration of the possibility that photic information affecting the SCN affects multiple functions via a common mechanism. As a result, there has been no attempt to establish the point or points at which the several SCN-mediated outputs diverge. It should be possible to obtain such information using a combination of neuroanatomical and electrophysiological methods to determine intra-SCN circuitry. One investigation has already narrowed the scope of the issue by showing that nocturnal light can induce FOS and Per1 in SCN neurons even when phase shifts are blocked by a nonphotic stimulus (Edelstein et al., 2003; but see Maywood et al., 1999; Yokota et al., 2000). Thus, the location at which the nonphotic input is acting must be downstream of the signal transduction cascade promoting expression of those genes. It remains to be seen whether the same nonphotic stimulus would block light-induced suppression of pineal activity or some other response to light.
Light-Induced Locomotor Suppression and Phase Shifts Are Linked
The recent evidence that a single stimulus induces both phase shifts and photosomnolence highlights an issue simmering in the circadian rhythm literature. It concerns whether wheel running, a standard behavior used to evaluate rhythmicity, is causally related to generation of light-induced phase shifts. A number of experiments now support the principle that nonphotic stimuli (enforced locomotion being one of several types) greatly reduce or abolish the phase shifting effects of light (Edelstein et al., 2003; Mistlberger and Antle, 1998; Mistlberger and Holmes, 1999; Ralph and Mrosovsky, 1992). In other words, the data show that locomotor activity must be suppressed (or photosomnolence induced) in order for light to maximally elicit phase shifts.
Importantly, the foregoing studies suggest a pathway by which locomotion suppresses light-induced phase shifts. Light and behavioral activity oppose each other at the SCN cellular level (Schaap and Meijer, 2001). Locomotion (or a correlate) (Mistlberger et al., 2003) is thought to modify SCN function via release of NPY from terminals of IGL neurons (see Yannielli and Harrington, 2004, for a review). Light-induced phase shifts are blocked by NPY infusion onto the SCN (Gamble et al., 2005; Lall and Biello, 2002, 2003a, 2003b; Yannielli and Harrington, 2000). Several additional results are consistent with the idea that light initiates a prolonged sequence of events, mediated by the SCN, that promotes both locomotor suppression and phase shifts. These include the fact that running in a novel wheel induces NPY release in the SCN (Glass et al., 2010) and that both NPY treatment and novel wheel activity block light-induced phase shifts even when administered an hour after the photic stimulus (Lall and Biello, 2002, 2003a, 2003b; Yannielli and Harrington, 2000, 2001a, 2001b). Lall and Biello (2003b) also noted that individual NPY treatments simultaneously block or greatly attenuate both light-induced phase shifts and the duration of locomotor suppression. This observation is consistent with the view that the 2 effects of light are mediated by the SCN. In the case of pineal melatonin synthesis, direct infusion of NPY onto the SCN mimics the inhibitory effect of light (Gamble et al., 2006). It remains to be seen whether photic suppression of pineal melatonin is or is not blocked by locomotion. The interval during which NPY is able to antagonize nocturnal light effects may be related to the “early phase resetting” interval determined from SCN cell physiology and characterized by increasing spike frequency in VIP neurons and reduction of the potassium current (see Fig. 8 in Kuhlman et al., 2003).
Conclusions from Studies of Light-Induced Locomotor Suppression
Exposure of mice or hamsters to light during the early night suppresses locomotion, whether measured as wheel running or open field activity. A variety of studies have yielded the following conclusions:
Classic and pRGC photoreceptors contribute to locomotor suppression. The SCN is necessary for light-induced locomotor suppression. Very brief light exposure is sufficient to induce locomotor suppression. Single light pulses must be more than 3 sec long to suppress locomotion. An effective light stimulus “triggers” an interval, K, of locomotor suppression. Long light exposure “maintains” the interval of locomotor suppression. Higher irradiance light pulses yield greater locomotor suppression. Locomotor suppression by flashes does not display temporal integration. Conclusions 1 through 8 are also true for light-induced phase shift responses. Light induces rhythm phase shifts simultaneously with locomotor suppression. Inactivity is necessary to obtain normal light-induced phase shifts.
The sole indication that the effect of light on rhythm phase differs from its effect on locomotor suppression comes from phase shift data which have been interpreted as suggesting that the phase shift mechanism shows temporal integration of light pulse energy. The difference between the phase shift and locomotor suppression results may lie with the confound that long light pulses have both activating and maintenance effects (see (Morin et al., 2010, for a discussion). Given the numerous similarities between photic regulation of phase shifts and locomotor suppression, it is parsimonious to infer that phase shift magnitude is modified by light according to the same rules governing the duration and magnitude of locomotor suppression.
Light Acts through the SCN to Modify the Activity of Multiple Systems
Photic regulation of circadian rhythm phase and locomotor suppression appear to be regulated by similar activating and maintaining mechanisms, but the details of such responses are not identical. For example, subjective night is divided into delay and advance portions of the light-type phase response curve; there is no equivalent for locomotor suppression (Redlin and Mrosovsky, 1999b). It is also possible that melatonin suppression by light in the early night is regulated by an entirely different mechanism than is active during the later night (Illnerova and Vanacek, 1982). At the very least, there are likely to be multiple regulatory circuits involved in the most basic light-initiated responses—a pathway intrinsic to the SCN for resetting circadian clock phase; SCN efferent pathways for suppressing locomotion and pineal function and for inducing sleep—and there should be an SCN-afferent route by which locomotor suppression/sleep enables light-induced phase shifts.
The SCN, as master circadian clock, dictates the harmony of many bodily oscillations through its efferent distribution of rhythm phase. It is more surprising that photic input to the SCN initiates additional forms of physiological response, including suppression of pineal activity and melatonin production, as well as photosomnolence. The list of direct and indirect light-induced actions is expected to lengthen with additional research, especially given the presence of multiple pathways by which the SCN can modulate peripheral organ function (Bartness et al., 2001; Gerendai et al., 2009) and their role in regulation of rhythmicity in certain tissues but not others (Guo et al., 2006). A candidate for inclusion on this list is the adrenal corticosterone surge induced by nocturnal light (Ishida et al., 2005; Mohawk et al., 2007). The level of response is dependent upon stimulus irradiance, abolished by SCN lesions and mediated through SCN efferent activity in the splanchnic nerve, which provides input to the adrenal (Ueyama et al., 1999). Elevated ACTH does not mediate the effect. In addition, light pulses as short as 1 min are known to increase splanchnic nerve activity, which is also irradiance-dependent. A 10-min light pulse induces a steady increase in splanchnic nerve activity over at least 2 h which, once initiated by light, persists in the dark (Niijima et al., 1992, 1993). It is not yet known whether adrenal hormone synthesis responds to brief light exposure in the same manner as locomotor suppression or splanchnic nerve activity.
A new physiological event on the list of changes induced by nocturnal light is a rapid drop in mouse core body temperature (Tc; Fig. 4; see Studholme et al., 2013). The large, abrupt drop in Tc occurs in parallel with locomotor suppression and recovery. The pattern of Tc change, its induction by millisecond light flashes, the fact that it endures well beyond the presence of the stimulus, and that it is prolonged by long-duration light stimuli strongly suggest that the Tc response is regulated by the same mechanistic sequence triggering the other events. The light-induced drop in Tc, along with photic control of corticosterone, must be considered central to a multifaceted, whole body response involving, at the very least, the circadian visual, sleep, neuroendocrine, thermoregulatory, and autonomic nervous systems.
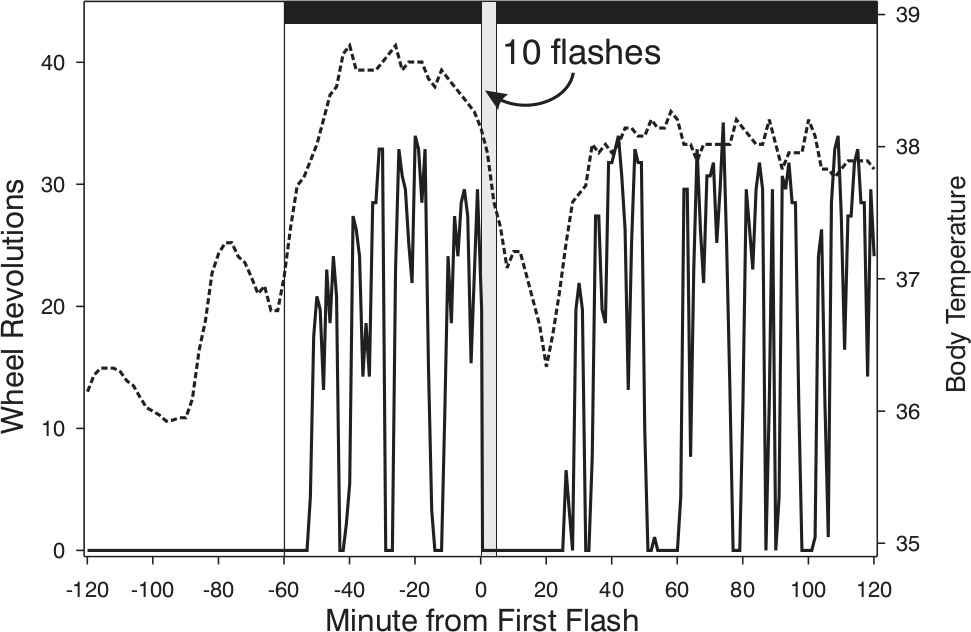
Core body temperature (Tc; broken line) drops acutely in response to brief light exposure and remains reduced well beyond the presence of light. In this example, the stimulus consisted of 10 light flashes, each 2 msec long, distributed equally across a 5-min interval (gray vertical bar beginning at Minute 0). The Tc change is temporally associated with suppression of locomotion (solid line) but is not caused by it. The behavioral and physiological changes attain full recovery at approximately the same time, although Tc rises from its nadir well in advance of locomotion. Morin and Studholme, unpublished data.
The suggestion that light initiates a time-limited event sequence common to its suppression of pineal function, induction of photosomnolence, phase shifting, augmented adrenal activity, and thermoregulatory change is interesting but says little about how the brain might be organized to enable those responses. Figure 5 is offered as a guide for thought experiments regarding how photic information might act through the SCN to elicit the patterns of physiology and behavior discussed here. The figure does not imply any causal relation between presently known intrinsic SCN anatomy and efferent functional control. In fact, recent research has revealed the SCN to be a plastic structure capable of organizing its own rhythmic internal activities to reflect the needs imposed by the prevailing photic environment (De la Iglesia et al., 2004; Karatsoreos et al., 2011; Lee et al., 2009; Liu et al., 2007; Watanabe et al., 2006). This being the case, what is considered “normal” SCN activity and efferent function must be accepted with caution. Perhaps more important is the fact that unexpected or seemingly outlandish light-induced responses (or lack thereof) are likely to provide invaluable clues as to ordinary function (Barakat et al., 2004; de Groot and Rusak, 2002; Erkert et al., 2006; Ruby et al., 2000).
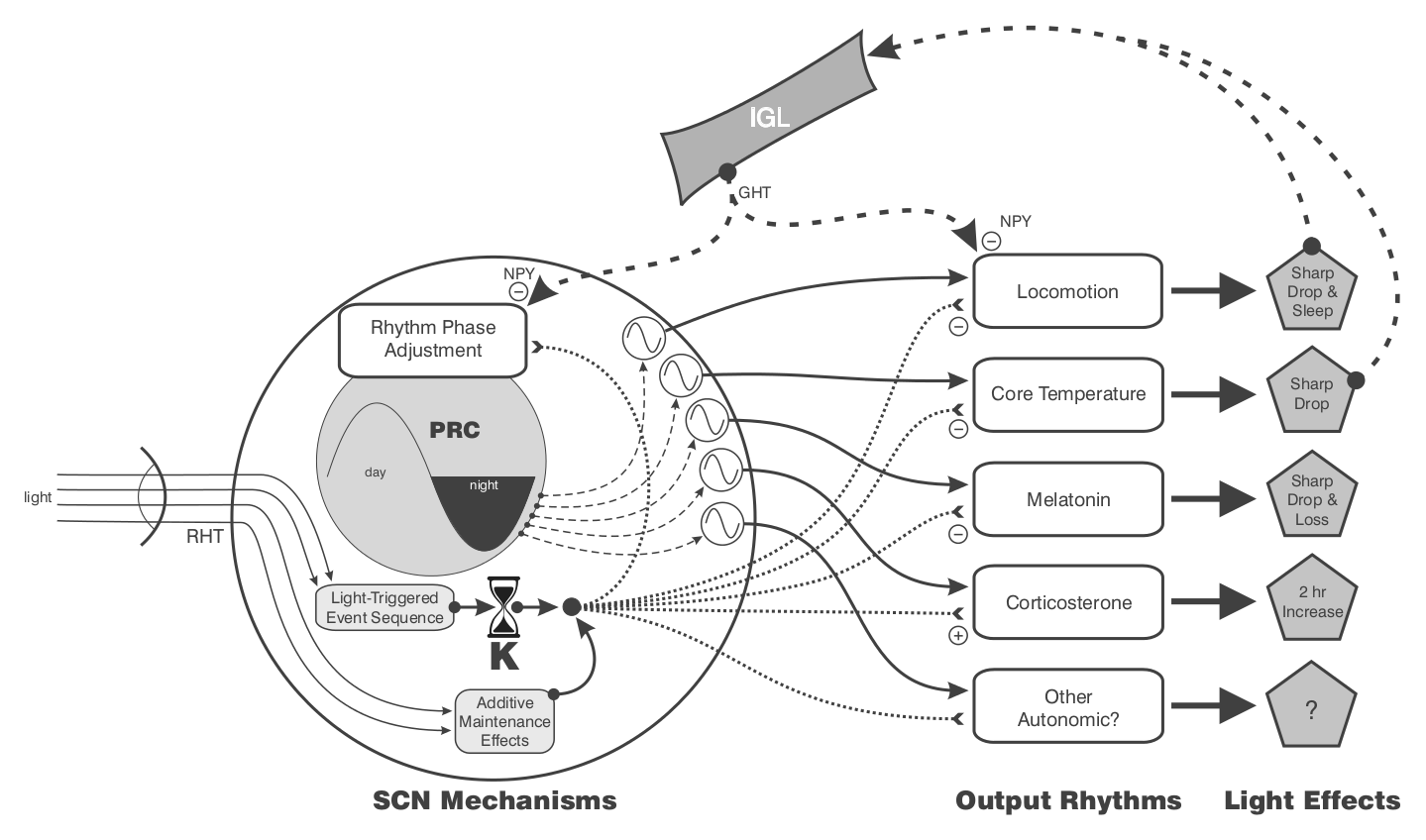
Theoretical relationships between the retinorecipient SCN, the master circadian clock in the SCN (the two not necessarily being different), and efferent circuits controlling the rhythms of locomotion/sleep, core temperature, pineal melatonin, and adrenal corticosterone. “Other autonomic” rhythmic outputs are predicted (see text). In this diagram, “rhythm phase adjustment” is also a rhythm as defined by the light-type phase response curve (PRC). The generation and photic control of the PRC are fully intrinsic to the SCN. The retinorecipient mechanism underlying the “light-triggered event sequence” (a presumed interval timer with duration = K) resides in the SCN and mediates light effects on all rhythms. Additional light can prolong K via “additive maintenance effects.” These will be seen as larger phase shifts or longer intervals of photosomnolence. The diagram presumes that the irradiance-sensitive, fixed-interval response sequence is activated for all light effects indicated. The immediate effect of nocturnal light is to inhibit locomotion, cause an abrupt drop in core temperature, induce a phase shift, cause a rapid decline in melatonin, and facilitate adrenal corticosterone synthesis. The drop in nocturnal locomotion (or corresponding sleep induction), and conceivably the drop in core temperature, may act directly or indirectly through the IGL to inhibit release of NPY in the SCN. Only in the absence of such release will light-induced phase shift and locomotor suppression responses be normal. The intra-SCN release of NPY may also alter other listed responses to light (see text).
Footnotes
Acknowledgements
Supported by NIH grant NS061804.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
