Abstract
The suprachiasmatic nucleus (SCN) is the main brain clock in mammals. The SCN synchronizes to the light-dark cycle through the retinohypothalamic tract (RHT). RHT axons release glutamate to activate AMPA-kainate and N-methyl-D-aspartate (NMDA) postsynaptic receptors in ventral SCN neurons. Stimulation of SCN NMDA receptors is necessary for the activation of the signaling cascades that govern the advances and delays of phase. To our knowledge, no research has been performed to analyze the functional synaptic modifications occurring during postnatal development that prepare the circadian system for a proper synchronization to light at adult ages. Here, we studied the pre- and postsynaptic developmental changes between the unmyelinated RHT-SCN connections. Spontaneous NMDA excitatory postsynaptic currents (EPSCs) were greater in amplitude and frequency at postnatal day 34 (P34) than at P8. Similarly, both quantal EPSCs (miniature NMDA and evoked quantal AMPA-kainate) showed a development-dependent increase at analyzed stages, P3-5, P7-9, and P13-18. Moreover, the electrically evoked NMDA and AMPA-kainate components were augmented with age, although the increment was larger for the latter, and the membrane resting potential was more depolarized at early postnatal ages. Finally, the short-term synaptic plasticity was significantly modified during postnatal development as was the estimated number of quanta released and the initial release probability. All of these synaptic modifications in the unmyelinated RHT-SCN synapses suggest that synchronization to light at adult ages requires developmental changes similar to those that occur in myelinated fast communication systems.
The suprachiasmatic nucleus (SCN) of the hypothalamus is the main brain clock that regulates circadian rhythms in mammals (Moore and Eichler, 1972). The SCN synchronizes to the light-dark cycle through the retinohypothalamic tract (RHT; Johnson et al., 1988), which is a bundle of unmyelinated axons (Gûldner, 1978) that emerge from a subpopulation of melanopsin-containing photosensitive ganglion cells in the retina (Hankins et al., 2008). RHT synaptic boutons release glutamate (de Vries et al., 1993) and the pituitary adenylate cyclase-activating polypeptide (Hannibal, 2006) to the ventral SCN neurons (Abrahamson and Moore, 2001). Glutamate stimulates ionotropic AMPA-kainate and N-methyl-D-aspartate (NMDA) postsynaptic receptors (Kim and Dudek, 1991), and thus the Ca2+ entrance through NMDA receptors plays a crucial role in the activation of the signaling cascades that control the delays and advances of phase needed for the synchronization of the circadian pacemaker to environmental light (Ding et al., 1994; Ding et al., 1997).
It should be noted that the mechanisms described above have been studied in adult animal models, and thus there is no information about the physiological modifications occurring during development of the circadian system that allow the proper synchronization to light at adult stages. Nonetheless, anatomical and behavioral evidence suggest physiological and structural changes during postnatal development. For instance, early connexions are established from the RHT to the SCN at postnatal days (P) 1 to 4 (Mason et al., 1977; Munoz Llamosas et al., 2000; Speh and Moore, 1993; Stanfield and Cowan, 1976), but Fos immunoreactivity is found beginning at P0 (Hannibal and Fahrenkrug, 2004; Sekaran et al., 2005). Innervation of RHT fibers to the ventral SCN reaches an adult appearance between P10 and P15 (Frost et al., 1979; Gûldner, 1978; Mason et al., 1977; Munoz Llamosas et al., 2000; Speh and Moore, 1993). Notably, Duncan and colleagues (1986) pointed out P6-8 as a critical period when enough maturity is reached in the RHT-ventral SCN connexions, which permits the circadian system to synchronize to light without maternal influence (Reppert and Schwartz, 1986).
On the other hand, there is fairly profound knowledge about the physiological changes occurring during development in synapses that process rapid sensorial information, as well as in hippocampal circuitry. For example, quantal or miniature excitatory postsynaptic currents (EPSCs) and quantal inhibitory postsynaptic currents (IPSCs) increase in amplitude (Taschenberger et al., 2002; Lu and Trussell, 2007; Case et al., 2011; Kim and Kandler, 2003; Awatramani et al., 2005). Also, the AMPA-kainate component augments over that of NMDA, although this latter diminishes in amplitude (Hsia et al., 1998; Chen and Regehr, 2000; Lu and Trussell, 2007; Case et al., 2011) and the decay time of EPSCs and IPSCs shortens (Hsia et al., 1998; Kotak et al., 1998; Chen and Regehr, 2000; Taschenberger and von Gersdorff, 2000; Lu and Trussell, 2007; Case et al., 2011; Alamilla and Gillespie, 2013; Kramer et al., 2014). In addition, the neurotransmitter release machinery is modified during development, including a reconfiguration of voltage-activated calcium channels (Iwasaki and Takahashi, 1998; Fedchyshyn and Wang, 2005; Alamilla and Gillespie, 2013) and a reduction in the probability of neurotransmitter release, which is reflected by a decrease in the short-term synaptic depression (STD), while augmenting the short-term synaptic facilitation (STF; Bolshakov and Siegelbaum, 1995; Taschenberger and von Gersdorff, 2000; Taschenberger et al., 2002; Iwasaki and Takahashi, 2001; Case et al., 2011; Alamilla and Gillespie, 2013).
All of the developmental changes described above correspond to fast synapses, capable of communicating high frequencies of stimulation (Taschenberger and von Gersdorff, 2000), while the unmyelinated RHT fibers are unable to conduct similar frequencies. Thus, it is unknown if the postnatal pattern in myelinated axons also occurs for the RHT-SCN pathway. Hence, the aim of this research was to study the functional pre- and postsynaptic changes during postnatal development between the RHT and ventral SCN neurons. Using electrophysiological experiments, we found evidence of comparable modifications to those described in myelinated fibers, such as an increment in miniature EPSCs, increase in the AMPA-kainate component over that of NMDA, and changes in short-term synaptic plasticity (STP), which imply modification in the vesicle pool and a decrease in the release probability. By contrast, we detected differences such as an increment in the decay time of the evoked EPSCs and an augmentation of the evoked NMDA EPSCs.
Materials and Methods
Ethical Approval
Experimental procedures were approved by the University of Colima Institutional Animal Care and Use Committee and comply with the Guide for the Care and Use of Laboratory Animals of the US National Institutes of Health (NIH Publication No. 85-23, revised 1996) and with UK regulations on animal studies (Grundy, 2015). For the experiments of this study, hypothalamic brain slices were obtained from animals aged between P3 and P34. The experimental protocols, including the use of sodium pentobarbitone as the anesthetic agent, were approved by the Institutional Animal Care and Use Committee of the University of Colima. Animals were killed by decapitation while under anesthesia, which was induced with sodium pentobarbitone (60 mg kg−1, intraperitoneally). The level of anesthesia was monitored by confirming the absence of pedal withdrawal reflexes. All efforts were made to minimize the number of animals used in this study and their suffering.
Animals and Slice Preparation
Experiments were carried out in Wistar rat pups of either sex, weighing between 10 and 85 g (N = 166) and bred in our animal facilities from parents supplied by Harlan, Mexico. They were housed in pairs in polycarbonate cages under 12:12 light-dark cycles (lights-on at 0700 h). Food and water were available ad libitum. The runt animals of the litter were excluded.
After decapitation, the brain was quickly removed from the skull (at light phase ZT 3-6), and coronal brain slices (~300 µm) containing the hypothalamus were obtained with a VT1000 microtome (Leica Microsystems, Wetzlar, Germany) in cold (4 °C) artificial cerebrospinal fluid (ACSF), which contained (in mM) 124 NaCl, 1 MgSO4, 5 KCl, 1.25 KH2PO4, 10 glucose, 26 NaHCO3, and 2 CaCl2; bubbled with 95% O2/5% CO2; pH 7.4. Coronal brain hypothalamic slices were maintained in ACSF at room temperature (22-24 °C) for at least 45 min before they were transferred to the recording chamber.
Electrophysiological Recordings
Electrophysiological experiments were performed in ventral neurons of the medial SCN. Slices were transferred to a recording chamber (1.5 mL) and bathed with extracellular solution through a gravity-driven device (~5 mL min−1). Recordings were made at room temperature (22-24 °C) in the whole-cell configuration of the patch-clamp technique using an Axopatch 200B amplifier (Molecular Devices, Sunnyvale, CA). Neurons were visualized with an upright Axioexaminer A1 microscope equipped with differential interference contrast optics with 10× (W Achroplan, NA 0.3) and 63× (W Plan-Apochromat, NA 1.0) water immersion objectives (Carl Zeiss, Oberkochen, Baden-Württemberg, Germany). Data acquisition and command potentials were carried out with a Digidata 1322A interface controlled by the pClamp 8.2 software (Molecular Devices). Recording electrodes of 2 to 4 MΩ were made in a P97 puller (Sutter Instrument Co., Novato, CA) and filled with intracellular solution containing (in mM) 64 D-gluconic acid, 64 CsOH, 11 EGTA, 56 CsCl, 1 MgCl2, 1 CaCl2, 10 HEPES, and 5 QX-314. Series resistance was compensated (~80%) in all recordings, and the seal was monitored throughout the whole experiment. Cells were discarded if the membrane resistance was <150 MΩ or the access resistance >15 MΩ or changed more than 15%. Currents were low-pass filtered at 2 kHz and sampled at 10 kHz. Current traces obtained throughout this study were not corrected for a −4.6-mV junction potential. Passive membrane properties were obtained by analyzing capacitive currents transients evoked during 10-msec depolarizations (20 mV) at −60 mV of holding potential. An agar-KCl bridge was used to ground the bath. All electrophysiological recordings were performed during the light phase (ZT ~4-8).
Recording of Spontaneous and Miniature NMDA EPSCs
Spontaneous NMDA EPSCs were recorded in ventral SCN neurons (Abrahamson and Moore, 2001) in an Mg2+-free ACSF solution (MgSO4 was replaced by K2SO4). Recordings were performed in the presence of 50 µM picrotoxin (GABA-A receptors antagonist) and 5 µM 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX; antagonist of AMPA/kainate receptors). Each neuron was recorded at a holding potential of −60 mV for at least 100 sec.
Miniature EPSCs were carried out in similar conditions to those used for spontaneous EPSCs and recorded after the perfusion of 1 µM tetrodotoxin (TTX; Na+ channel inhibitor) for 10 min in the bath solution (Mg2+-free ACSF with picrotoxin [50 µM] and CNQX [5 µM]). Recordings were done at a holding potential of −60 mV and lasted a total of 10 min, in 10-sec segment files. To validate the NMDA origin of both spontaneous and miniature EPSCs, 50 µM D-(-)-2-amino-5-phosphonopentanoic acid (D-APV; NMDA receptor antagonist) or 10 µM (5S,10R)-(+)-5-methyl-10,11-dihydro-5H-dibenzo[a,d]cyclohepten-5,10-imine maleate (MK-801, NMDA receptor antagonist) was perfused in the bath solution at the end of the experiments.
Quantal Events Coming from the RHT Terminals, Recorded in Ventral SCN Neurons
A bipolar stimulating electrode (external diameter 125 µm, inner diameter 12.5 µm; FHC, Bowdoinham, ME) was positioned on the optic chiasm. A square electrical pulse (50-300 µs of duration; 0.1-1 µA of amplitude) was applied at 0.1 Hz using a Master 8 stimulator and an Iso-Flex stimulus isolator (A.M.P.I., Jerusalem, Israel). Stimulus strength was adjusted to produce a reliable response with the lowest intensity. To evoke EPSCs at a resolution of quantal events by desynchronization of neurotransmitter released by the RHT stimulation, the extracellular recording solution (standard ACSF) included the substitution of Ca2+ by Sr2+ (3 mM; Chen and Regehr, 2000), plus picrotoxin (50 µM) and D-APV (50 µM). At the end of the experiments, CNQX (5 µM) was added to the extracellular solution to confirm the AMPA-kainate nature of the evoked quantal EPSCs.
Measurement of NMDA/AMPA-kainate EPSCs Ratio
A square electric pulse with similar conditions to those described in the previous paragraph was delivered on the optic chiasm. Evoked EPSCs elicited by RHT stimulation were recorded alternately at 2 holding potentials, −60 mV and +40 mV. Twenty pulses at each membrane potential were applied with a frequency of 0.1 Hz. Extracellular recording solution was standard ACSF with picrotoxin (50 µM). EPSC amplitude was calculated from the average of 20 responses obtained in each holding potential. NMDA EPSC amplitude (+40 mV) was calculated at the time when AMPA-kainate EPSCs (−60 mV) decayed at 95% of the amplitude (for instance, see the vertical dotted lines in Fig. 4A). The probability of response was calculated as the result of the ratio of postsynaptic responses and the electrical pulses applied on the RHT (Alamilla et al., 2015). To corroborate the glutamatergic source of the postsynaptic responses, CNQX (5 µM) and D-APV (50 µM) were added to the perfusion bath at the end of experiments.
Recording of the Membrane Resting Potential
Measurement of the resting membrane potential of SCN ventral neurons was performed by means of an accurate (±1 mV) noninvasive approach, the cell-attached current clamp mode when the seal resistance is higher than 5 GΩ (Mason et al., 2005; Perkins, 2006). The extracellular solution (standard ACSF) contained no blocking compound of any synaptic input, whereas the intracellular recording solution was the same used for all experiments, but without QX-314.
Evaluation of Short-term Plasticity in Ventral SCN Neurons
Paired electrical pulses of different frequencies of stimulation (2, 5, 10, 20, and 50 Hz) were delivered on the optic chiasm. Electrical stimulation was similar to that described above. Twenty pairs of RHT stimulations (applied at 0.1 Hz) were recorded in ventral SCN neurons for every frequency. Extracellular recording solution was standard ACSF with picrotoxin (50 µM) and D-APV (50 µM). To determine the paired pulse ratio (PPR) and the type of STP, the average EPSC amplitude (20 pulses at every frequency) generated by the second pulse was divided by the average amplitude elicited by the first pulse. If the PPR was lower than 1, the synaptic plasticity was denominated short-term synaptic depression (STD); if the PPR was larger than 1, it was named short-term synaptic facilitation (STF). The AMPA-kainate origin of elicited EPSCs by RHT stimulation was validated by the specific blocking action of CNQX (5 µM), which was perfused in the recording bath at the end of the experiments.
Trains of High-frequency Stimulation
The train was composed of 15 stimuli delivered with a frequency of 50 Hz (200-msec interval). A total of 10 trains were applied on the RHT fibers, and each was administered every 10 sec (0.1 Hz). These experiments were performed in the same neurons used for evaluation of STP in the presence of the inhibitor of AMPA receptor desensitization, cyclothiazide (CTZ). The extracellular recording solution was standard ACSF with picrotoxin (50 µM), D-APV (50 µM), and CTZ (20 µM). After the application of the trains, CNQX (5 µM) was added to the bath to confirm the glutamatergic origin of the postsynaptic responses.
Evaluation of Presynaptic AMPA Modulation
Two pulses were applied on the RHT fibers with a frequency of 5 Hz (200-msec interval). Each paired pulse was delivered every 10 sec (0.1 Hz). A total of 20 paired pulses were performed before and after CTZ (20 µM) to evaluate changes in the PPR. NMDA postsynaptic response was recorded at +40 mV. The extracellular solution was Mg2+-free ACSF with picrotoxin (50 µM). The effect of AMPA presynaptic modulation was evaluated after 5 min of adding CTZ to the bath.
Drugs and Reagents
Picrotoxin, CNQX, D-APV, MK-801, QX-314, TTX, and CTZ were obtained from Tocris Bioscience (Ellisville, MO), and all other reagents were purchased from Sigma-Aldrich (St. Louis, MO). QX-314 was dissolved directly in the internal pipette solution at the desired concentration and prepared fresh daily. Drugs were prepared as stock solutions and kept at −20 °C until use: picrotoxotin (50 mM in ethanol), CNQX (5 mM in DMSO), D-APV (50 mM in water), MK-801 (10 mM in water), TTX (1 mM in chlorhydric acid aqueous solution), and CTZ (20 mM in ethanol). Aliquots of the stock solutions were diluted to final desired concentrations in the external (bath) solution.
Analysis
Data are reported as mean ± SEM, except for evoked quantal EPSCs (for which the median and interquartile range were expressed) and for the evaluation of the STP (categorical data). Spontaneous EPSCs, miniature EPSCs (TTX-independent), and evoked quantal EPSCs were analyzed using Mini Analysis V 6.0.7 (Synaptosoft, Decatur, GA) and pClamp V 10.2 software (Molecular Devices). For measurement of the NMDA/AMPA-kainate ratio, PPRs, the average amplitude and decay time (tau), as well as the amplitude of the EPSCs generated by the trains of high-frequency stimulation, we used the pClamp 10.2 software. Statistical analysis was performed using Prism 5 (GraphPad Software, La Jolla, CA), SPSS V 19 (IBM Corporation, Armonk, NY), and Origin V 8.5 software (OriginLab, Northampton, MA). The significance of differences was evaluated by parametric statistical tests (unpaired t test, 1-way analysis of variance [ANOVA] with Dunnett’s or Tukey’s post hoc tests, and 2-way ANOVA) when the normal distribution (Shapiro-Wilk and Kolmogorov-Smirnov tests) and variance homogeneity (Levene test) of data were confirmed. Otherwise, nonparametric tests (Kruskal-Wallis test with Dunn’s post hoc test) were used. The Cochran-Armitage trend test was used for the analysis of the STP. Statistical significance was set to p < 0.05.
Results
Development-dependent Difference in Spontaneous NMDA EPSCs from Ventral SCN Neurons
We first asked if spontaneous NMDA EPSCs are different at 2 postnatal ages: P8, when the rat pup starts to synchronize to the light-dark cycle through the RHT (Duncan et al., 1986), and P34, a juvenile postnatal age when this synchronization is completed. We found a significant difference in the amplitude of spontaneous NMDA EPSCs between the 2 analyzed ages (P8: 17 ± 0.3 pA, in 1617 EPSCs; P34: 20 ± 0.2 pA, in 6193 EPSCs; t = 9.2, df = 7808, p < 0.001; unpaired t test; Fig. 1A-C). Amplitude histograms of spontaneous NMDA currents at P34 moved to the right in the x axis (Fig. 1B). Analysis of the cumulative amplitude histograms of NMDA EPSCs indicated that 75% of the events were smaller than 19 pA, whereas at P34, the same percentage of postsynaptic currents was lower than 25 pA (Fig. 1D, E). Moreover, spontaneous NMDA currents exhibited a higher frequency at P34 in comparison with that at P8 (0.90 ± 0.21 Hz and 0.38 ± 0.10 Hz, respectively; t = 1.9, df = 25, p = 0.03; unpaired t test; Fig. 1F).
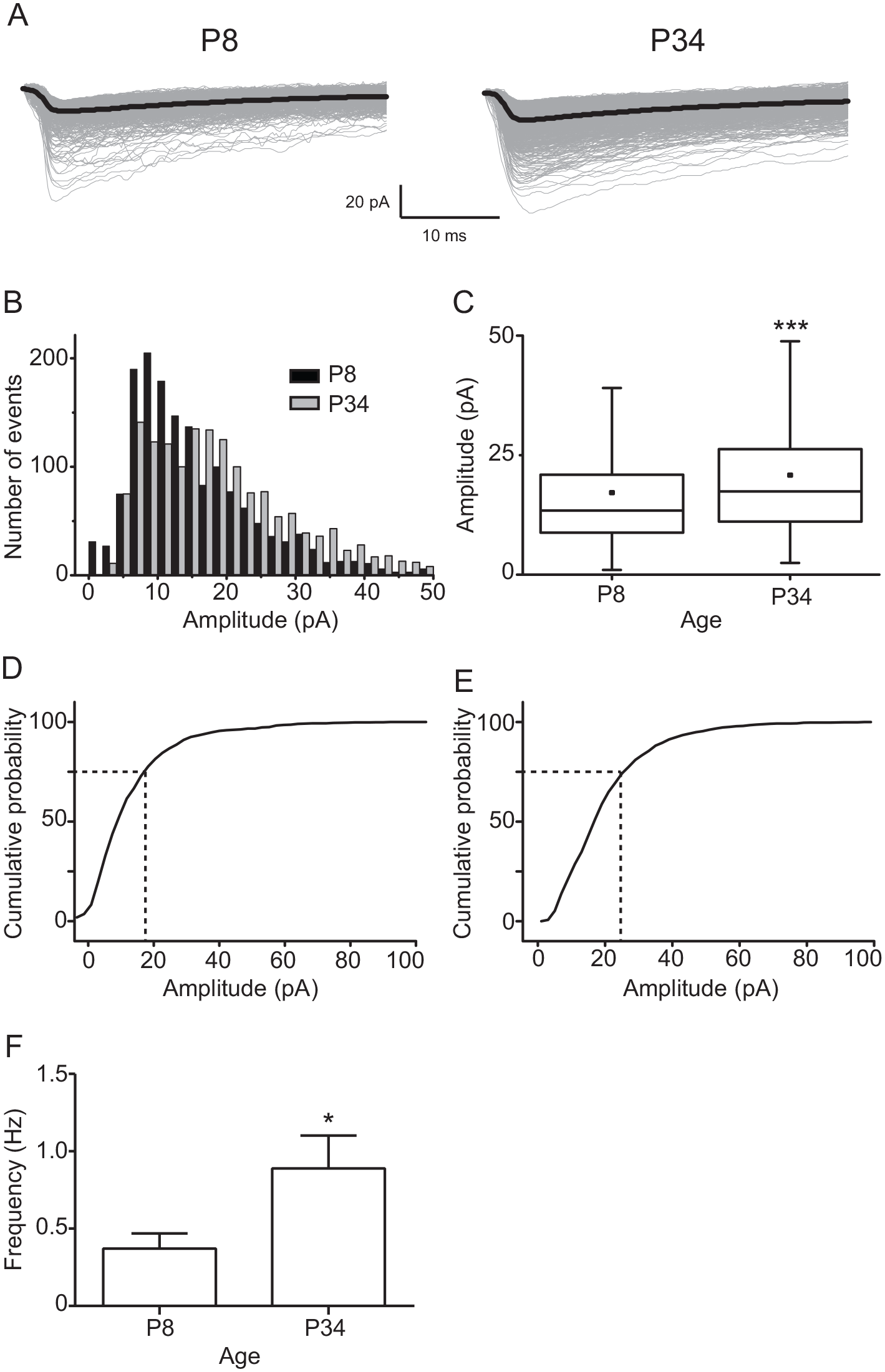
Spontaneous N-methyl-D-aspartate (NMDA) excitatory postsynaptic currents (EPSCs) from ventral suprachiasmatic nucleus neurons at postnatal day 8 (P8) versus P34. (A) Example of traces of NMDA EPSCs recorded in P8 (1197 events) and P34 (1023 events). For this and subsequent Figures 2 and 3, the black color trace represents the average of EPSCs. (B) Amplitude histograms of spontaneous NMDA currents recorded at P8 and P34. (C) Box plot graphs illustrating the amplitude of the population of spontaneous NMDA EPSCs at P8 and P34. For this and subsequent Figures 2 and 3, the small dot inside the box denotes the mean value. Cumulative amplitude histograms of spontaneous NMDA current population at P8 (D) and P34 (E). (F) Frequency of spontaneous NMDA EPSCs at both analyzed ages. n = 17 and 23 at P8 and P34, respectively. Unpaired t test: *p < 0.05 and ***p < 0.001 versus P8.
Postnatal Development of Miniature NMDA EPSCs
If there is a development-dependent change in the amplitude of spontaneous postsynaptic glutamatergic currents, it is possible that it is due to an increase of the glutamatergic miniature events in ventral SCN neurons. To test this idea, we recorded TTX-treated NMDA EPSCs along rat development, in 3 groups of postnatal ages, P3-5, P7-9, and P13-18 (Fig. 2A). We found significant differences in the amplitude of miniature NMDA EPSCs in P7-9 (5.7 ± 0.1 pA, in 408 EPSCs) and P13-18 (7.0 ± 0.1 pA, in 570 EPSCs), in comparison with that at P3-5 (4.1 ± 0.1 pA, in 333 EPSCs; F2,1308 = 170, p < 0.0001; 1-way ANOVA; Fig. 2C). At an older age, the amplitude histograms were shifted to larger values (Fig. 2B) as well as the cumulative amplitude histograms (Fig. 2E-G). In addition, miniature NMDA EPSCs had a higher frequency at the older analyzed stage (P13-18) (0.16 ± 0.05 Hz) in comparison with that at P3-5 (0.06 ± 0.01 Hz; F2,22 = 4.2, p = 0.026; 1-way ANOVA; Fig. 2D).
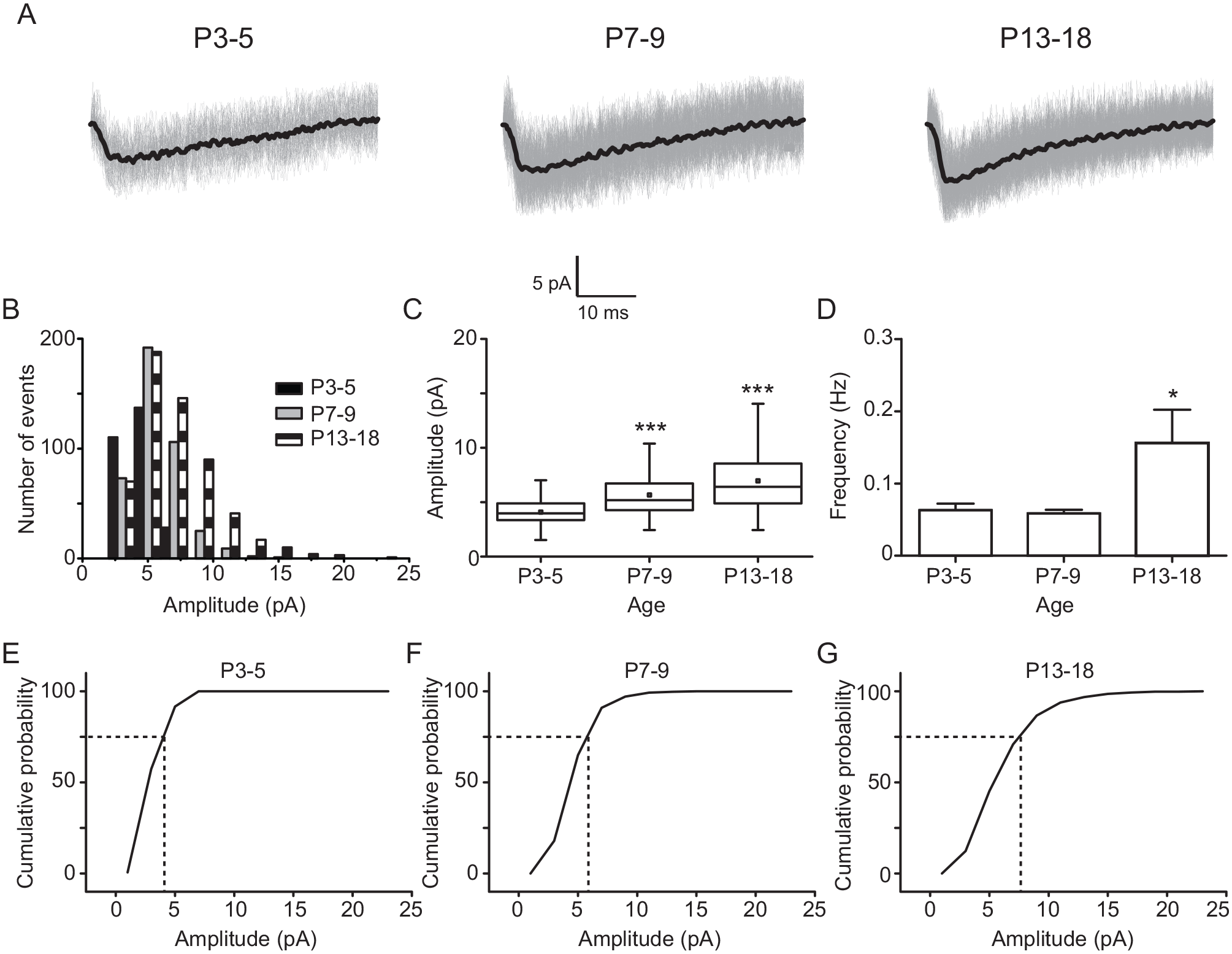
Miniature N-methyl-D-aspartate (NMDA; tetrodotoxin independent) excitatory postsynaptic currents (EPSCs) along rat development. (A) Representative traces of miniature NMDA EPSCs recorded at postnatal day 3 to 5 (P3-5; 123 events), P7-9 (266 events), and P13-18 (383 events). (B) Amplitude histograms of miniature NMDA currents belonging to the 3 postnatal ages. Note that the histograms shifted to the right in the x axis at older ages of postnatal development. (C) Box charts depict the amplitude populations of miniature NMDA currents at the examined ages. (D) Frequency of miniature NMDA postsynaptic currents at the 3 studied ages. Cumulative amplitude histograms of miniature NMDA currents at P3-5 (E), P7-9 (F), and P13-18 (G). n = 9, 10, and 9 at P3-5, P7-9, and P13-18, respectively. Dunnett’s post hoc test: *p < 0.05 and ***p < 0.001 versus P3-5.
As miniature NMDA EPSCs are difficult to identify and analyze (Chen and Regehr, 2000), we assessed if the reversal potential of the currents recorded in neurons of the ventral SCN is in agreement with that described for the glutamatergic component (Hille, 2001). Accordingly, we analyzed current recordings of 6 neurons belonging to the P13-18 group, at 3 holding potentials, −60, −40, and +40 mV. As depicted in Suppl. Fig. 1, the reversal potential of the currents was close to 0 mV, suggesting that they indeed come from synaptic glutamate release.
Evoked Quantal Non-NMDA EPSCs Increase with Postnatal Age
Next, we wondered if the evoked non-NMDA EPSCs coming from RHT fibers vary with postnatal age. Hence, by electrically stimulating the RHT, we recorded AMPA-kainate EPSCs in ventral SCN neurons in the presence of Sr2+, as previously reported by Chen and Regehr (2000; Fig. 3A). Median (and interquartile range; IQR) values of evoked AMPA-kainate EPSCs showed significant differences in amplitude at P7-9 (6.7 pA [IQR: 5.2-10.1 pA], in 1780 events) and P13-18 (8.5 pA [IQR: 6.1-12.5 pA], in 1977 events) when compared with those at P3-5 (5.2 pA [IQR: 3.1-8.2 pA], in 1414 events; K = 495, p < 0.001; Kruskal-Wallis test; Fig. 3C). Histograms and cumulative histograms show that the amplitude of evoked AMPA-kainate EPSCs increased at the older postnatal examined ages (Fig. 3B and 3E-G). In addition, these AMPA-kainate EPSCs displayed a larger frequency at P13-18 than at P3-5 (0.60 ± 0.08 and 0.44 ± 0.03, respectively; F2,58 = 4.4, p = 0.017; 1-way ANOVA; Fig. 3D). These results indicate that there is also a development-dependent increase of the amplitude and frequency of evoked quantal glutamatergic EPSCs coming from the RHT.
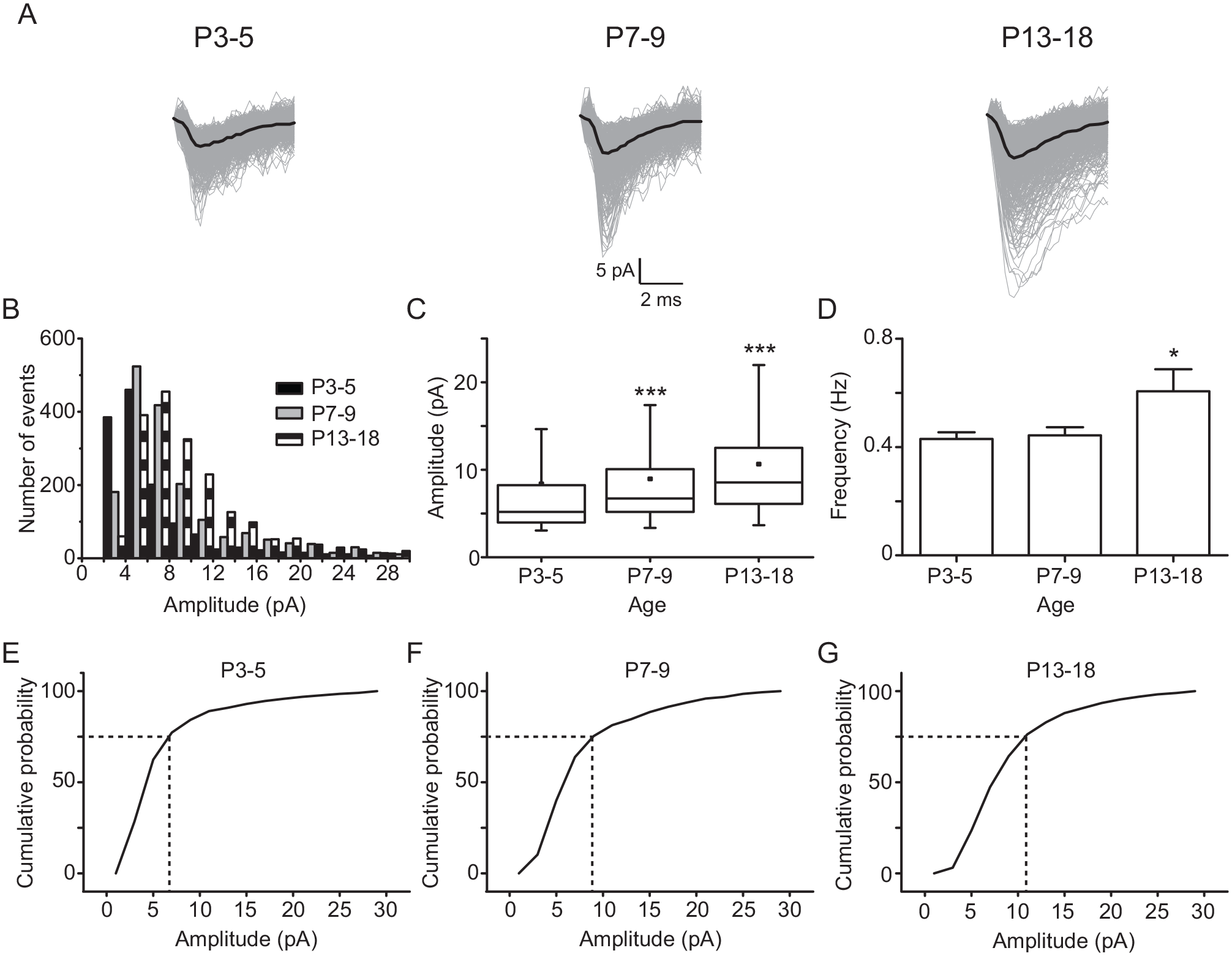
Age-dependent changes of evoked quantal non–N-methyl-D-aspartate excitatory postsynaptic currents (EPSCs). (A) Illustrative AMPA-kainate EPSCs recorded at postnatal day 3 to 5 (P3-5; 794 events), P7-9 (695 events), and P13-18 (811 events). Histograms (B) and amplitudes (C) of the populations of quantal AMPA-kainate EPSCs measured at the 3 studied ages. (D) Frequency of AMPA-kainate events at the analyzed ages. Cumulative amplitude histograms of AMPA-kainate EPSC at P3-5 (E), P7-9 (F), and P13-18 (G). n = 32, 20, and 22 at P3-5, P7-9, and P13-18, respectively. Dunnett’s post hoc test: *p < 0.05 and *** p < 0.001 versus P3-5.
To support that the changes in the amplitude of miniature and quantal EPSCs are the result of physiological modifications along postnatal development, and thus discarding the possibility that EPSCs would have been affected by variations in the passive membrane properties or the access resistance, we monitored these parameters by applying a 10-msec depolarization (20 mV), from a holding potential of −60 mV, in the same neurons used to record the miniature EPSCs. As shown in Table 1, we did not find significant differences in the passive membrane properties at the 3 ages investigated.
Passive membrane properties of ventral suprachiasmatic nucleus neurons corresponding to the 3 postnatal ages.

ANOVA = analysis of variance; P = postnatal day.
Ratio of NMDA and AMPA/Kainate Components Elicited by RHT Stimulation Is Reduced by Growth
In the above described results, the amplitude of NMDA EPSCs was lower than that for AMPA-kainate EPSCs at the same ages (Figs. 2 and 3). To assess if there is a different functional expression of NMDA receptors and AMPA-kainate postsynaptic receptors along development, we analyzed the amplitude ratio of NMDA and AMPA-kainate components induced by RHT electrical stimulation at different postnatal ages (Fig. 4A). The amplitude of the NMDA component evoked by electrical RHT stimulation was increased at older postnatal ages (4.1 ± 0.4 pA, 10.6 ± 1.9 pA, and 9.8 ± 1.6 pA at P3-5, P7-9, and P13-18, respectively; F2,29 = 3.6, p = 0.04; 1-way ANOVA; Fig. 4B) as was the AMPA-kainate component (9 ± 1 pA, 55 ± 15 pA, and 39 ± 8 pA at P3-5, P7-9, and P13-18, respectively; F2,23 = 3.7, p = 0.04; 1-way ANOVA; Fig. 4C). However, the augmentation of the latter was greater than that of the NMDA component and thereby the NMDA/AMPA-kainate ratio was reduced at P7-9 (0.29 ± 0.06) and P13-18 (0.31 ± 0.04) in comparison with that at P3-5 (0.51 ± 0.04; F2,24 = 3.4, p = 0.04; 1-way ANOVA). In addition, the decay kinetics (tau) of the NMDA component were enhanced at P13-18 (350 ± 60 msec) in comparison with that at P3-5 (142 ± 11 msec; F2,36 = 3.7, p = 0.03; 1-way ANOVA; Fig. 4E). Analyses of probability of response by a 2-way ANOVA test indicate a significant interaction by age (F2,78 = 10, p = 0.0001), but the interaction by the type of glutamatergic receptors that originated the postsynaptic response (NMDA or AMPA-kainate) was not significant (Fig. 4F).
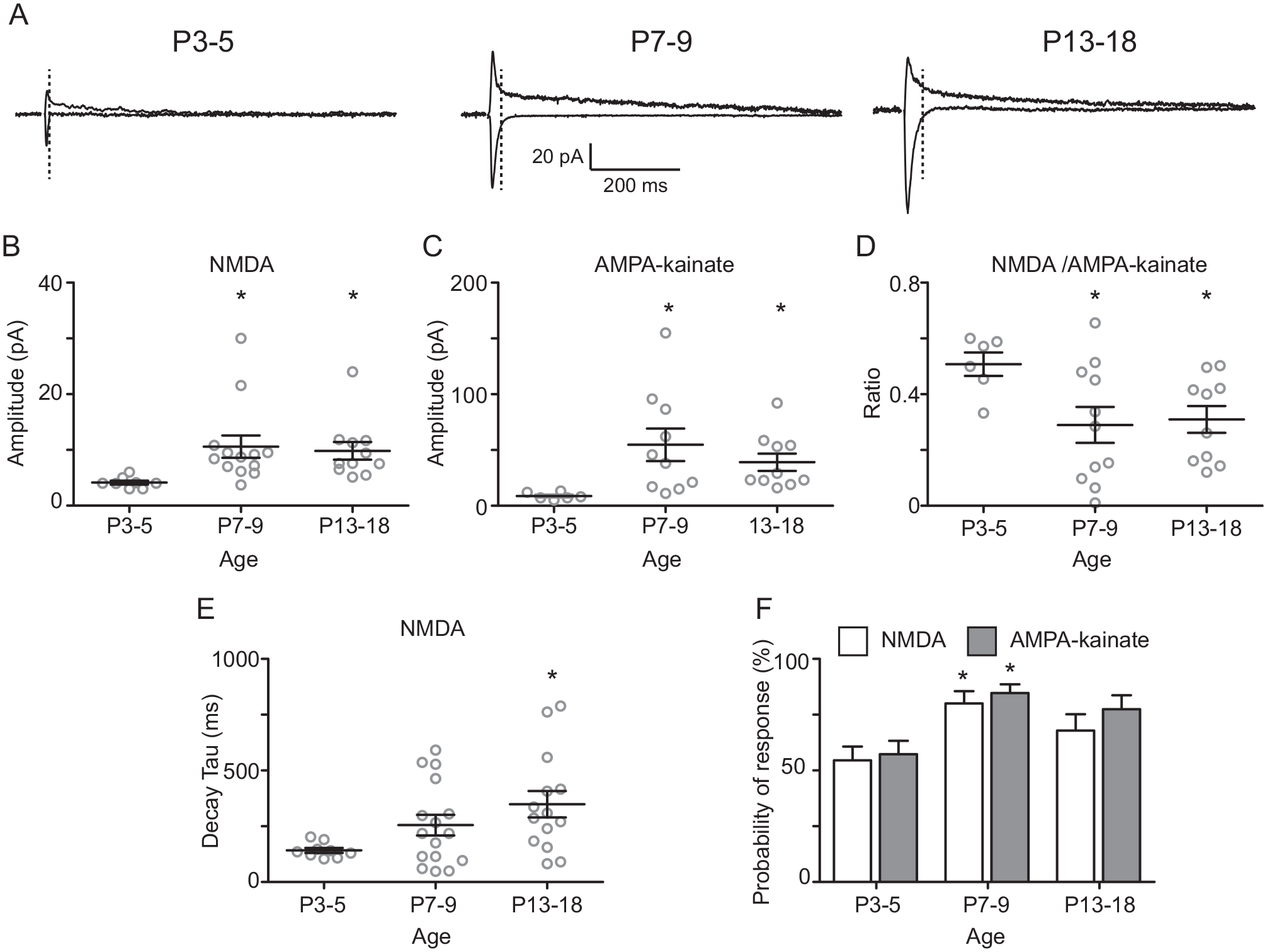
N-methyl-D-aspartate (NMDA)/AMPA-kainate ratio during postnatal development. (A) Average of 20 current traces of glutamatergic postsynaptic responses elicited by retinohypothalamic tract electric stimulation at −60 mV (AMPA-kainate) and +40 mV (AMPA-kainate and NMDA) recorded in suprachiasmatic nucleus ventral neurons of the different postnatal ages. The vertical dotted lines indicate the 95% decay time of the AMPA-kainate component, which was used as the time point for measuring the NMDA peak component. For clarity, the stimulus artifact is not shown. Amplitude of the NMDA (B) and AMPA-kainate (C) components and their respective NMDA/AMPA-kainate ratio at the studied ages (D). (E) Decay time (tau) of the NMDA component at the 3 ages. (F) Probability of response of the NMDA and AMPA-kainate transients. Dunnett’s post hoc test: *p < 0.05 versus postnatal day 3 to 5.
For the evaluation of the decay time (tau) of isolated AMPA-kainate EPSCs (i.e., in the presence of 50 µM D-APV and 20 µM CTZ), we performed additional recordings using stimuli of variable intensities (50-600 µA) to induce them to have similar amplitudes (Fig. 5A, B). We did not find differences in the decay kinetics of non-NMDA EPSCs along postnatal development (Fig. 5C). However, lower stimulation intensity was required at older developmental stages (F2,33 = 15.7, p < 0.0001; 1-way ANOVA; Fig. 5D) to obtain comparable EPSC amplitudes in ventral SCN neurons. In addition, the membrane resting potential was measured in SCN neurons because this parameter can directly affect the activation of NMDA receptors. Significant differences were found in the resting potential at P3-5 (−31.7 ± 1.1 mV) in comparison with P7-9 (−39.8 ± 2.0 mV) and also with respect to the oldest analyzed stage, P13-18 (−45.2 ± 1.7 mV; F2,41 = 18.6, p < 0.0001; 1-way ANOVA; Fig. 6).
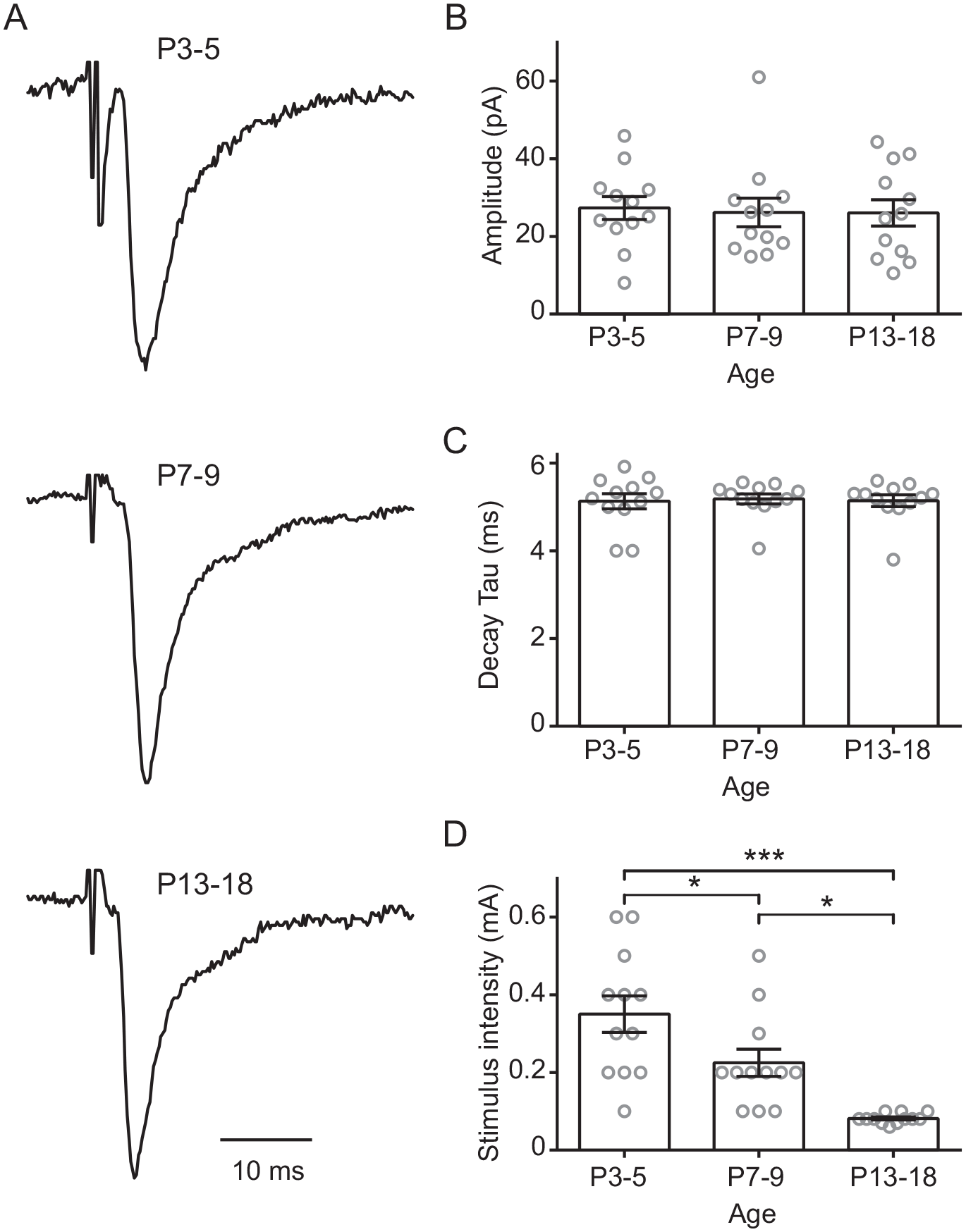
AMPA-kainate component (generated by retinohypothalamic tract stimulation) during postnatal development. (A) Normalized amplitude of non–N-methyl-D-aspartate currents recorded in the suprachiasmatic nucleus ventral neurons during different postnatal ages, applying variable stimulus intensity to reach a comparative amplitude (B). (C) Decay time constant and (D) stimulus strength applied at the 3 studied ages. Tukey’s post hoc test: *p < 0.05; ***p < 0.001.
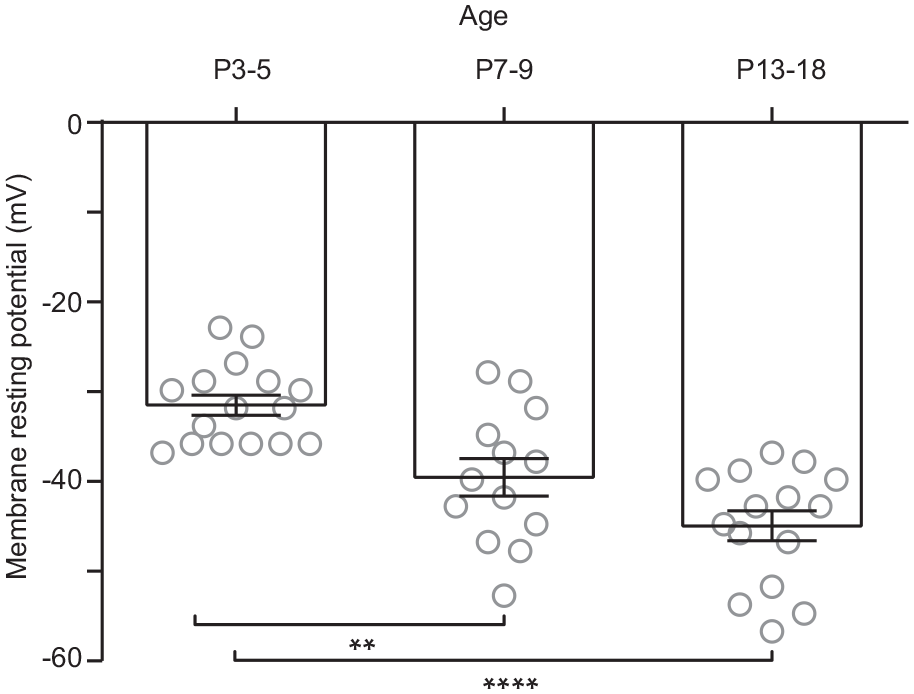
Membrane resting potentials recorded during postnatal development in the ventral suprachiasmatic nucleus neurons. Postnatal day 3 to 5 (P3-5) showed significantly more depolarized membrane potential than P7-9, and the former developmental stage was more depolarized than P13-18. Dunnett’s post hoc test: **p < 0.01; ****p < 0.0001.
These results suggest that there is a development-dependent rise in the expression of AMPA-kainate postsynaptic receptors in comparison with NMDA receptors and also a strengthening of RHT-SCN synapses as result of pre- and postsynaptic mechanisms.
Developmental Changes of STP
As previously described, the frequency of spontaneous and miniature events was augmented at older ages (Figs. 1F, 2D, and 3D), and there was a significant interaction between age and the probability of response elicited by RHT stimulation (Fig. 4F). These results could indicate presynaptic changes in the neurotransmitter release machinery along postnatal development. Thus, we analyzed the STP using the PPR of currents activated by electrical stimulation of the RHT fibers that impinge ventral SCN neurons at different frequencies (Suppl. Fig. 2). We did not find significant differences at any frequency of stimulation when data were pooled by developmental stage (data not shown). When we grouped our results by the type of STP (STF or STD), at 2 and 50 Hz we found that there was a trend in which most of the ventral SCN neurons exhibited STD at P3-5, whereas at the 2 later postnatal stages, the trend was toward STF, although these tendencies were not statistically significant (p = 0.14 and p = 0.15 for 2 Hz and 50 Hz, respectively; Cochran-Armitage test of trend; Suppl. Fig. 2A, E).
As we did not find developmental changes in the STP, we asked if desensitization of postsynaptic AMPA receptors could have complicated the results of PPR. Thus, we used an inhibitor of AMPA receptor desensitization, 20 µM CTZ (Chen and Regehr, 2000). Analyses of the PPR showed significant differences at P13-18 in comparison with P3-5 at frequencies of 10, 20, and 50 Hz. The PPR was significantly higher at older ages (~6- to 9-fold change) than that found at the earliest postnatal analyzed age (Table 2). Furthermore, the number of neurons with a specific type of STP changed during development. A significant trend to observe STF was found in ventral SCN neurons aged P13-18 at frequencies of 5 Hz (Cochran-Armitage test of trend: score = 4.24, df = 1, p = 0.039; Fig. 7B), 10 Hz (score = 4.24, df = 1, p = 0.039; Fig. 7C), and 50 Hz (score = 4.3, df = 1, p = 0.038; Fig. 7E). These results indicate that there is a change in the STP during postnatal development, which implies a modification in the neurotransmitter release machinery in the RHT terminals.
Paired pulse ratios obtained in ventral suprachiasmatic nucleus neurons, in the presence of cyclothiazide, and belonging to 3 postnatal ages.
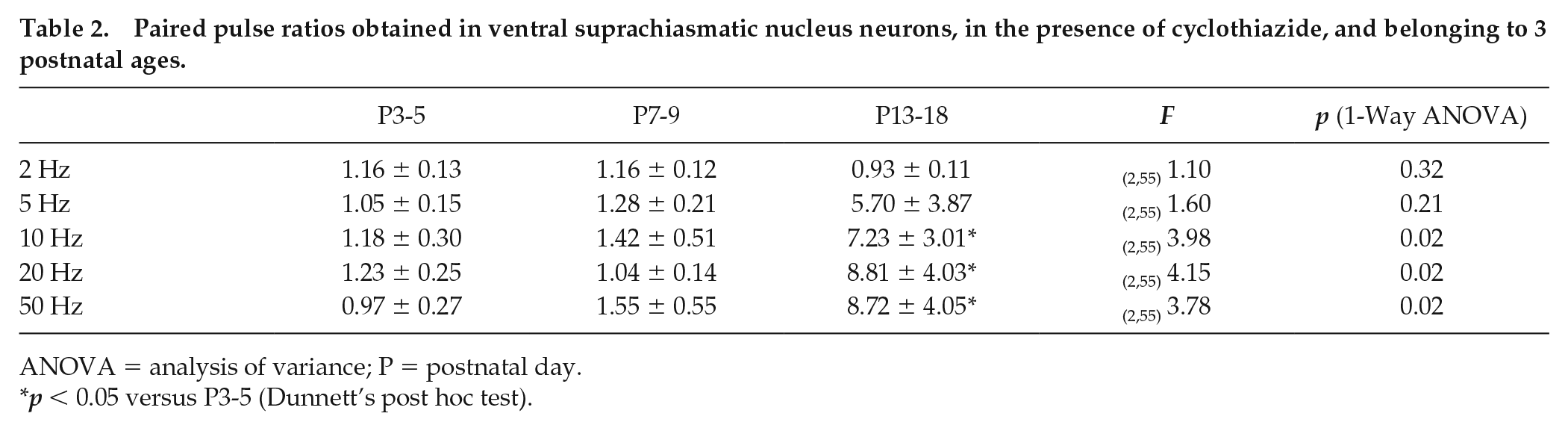
ANOVA = analysis of variance; P = postnatal day.
p < 0.05 versus P3-5 (Dunnett’s post hoc test).
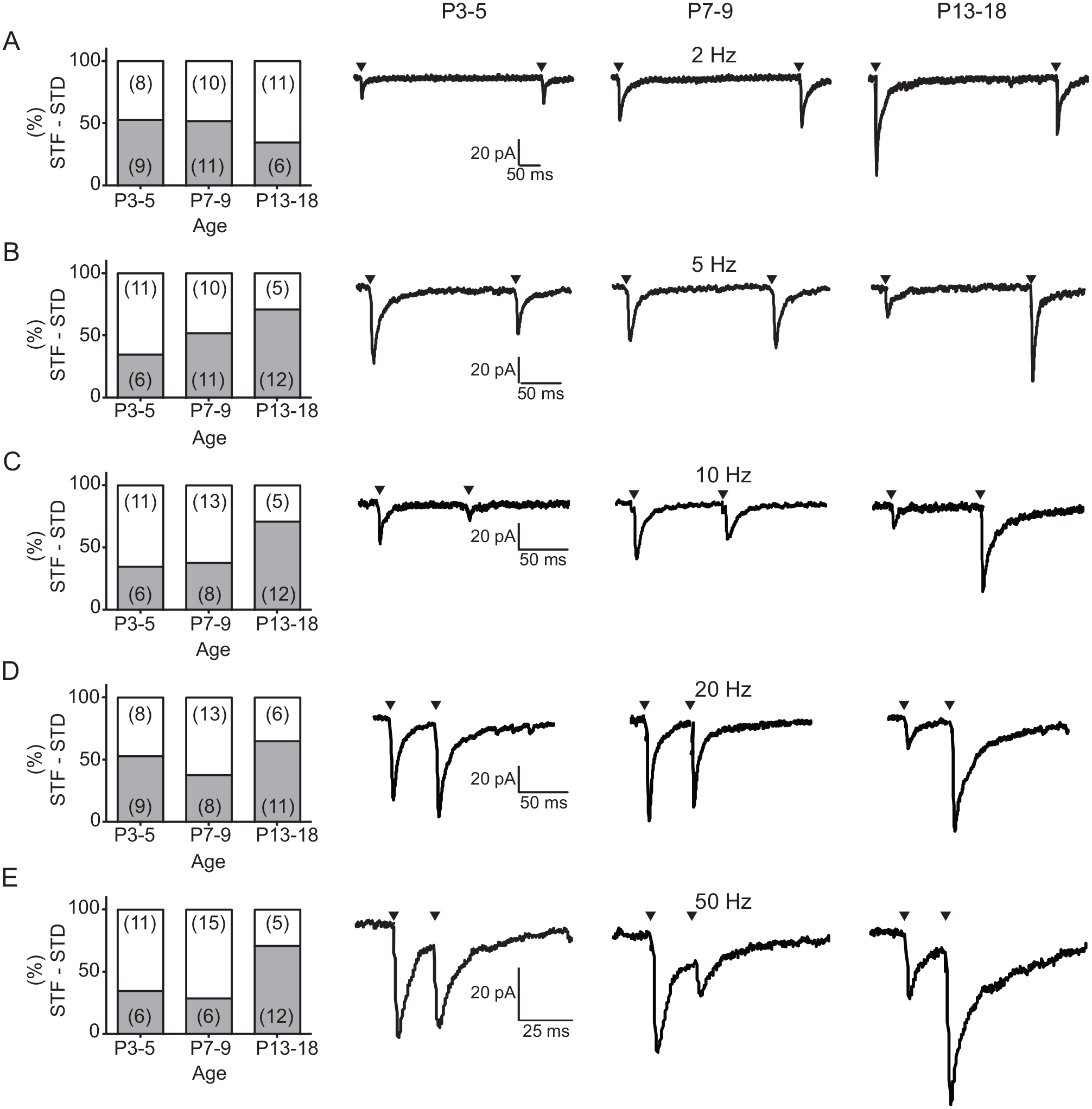
Short-term plasticity of the retinohypothalamic tract (RHT) and suprachiasmatic nucleus (SCN) ventral neurons in the presence of cyclothiazide. Percentage of SCN ventral neurons that showed short-term synaptic facilitation (gray boxes) or short-term synaptic depression (white boxes) by RHT electrical stimulation at 2 Hz (A), 5 Hz (B), 10 Hz (C), 20 Hz (D), and 50 Hz (E). The number of neurons that displayed each type of plasticity is indicated in parentheses. At the right of the graphs, representative current traces illustrating the dominant short-term plasticity for the 3 studied postnatal stages are shown. Arrowheads indicate when the electrical stimulation was applied.
Developmental Differences in the EPSCs Elicited by Trains of High-Frequency Stimulation
We next sought to investigate whether there is a relationship between the type of STP and the estimated number of quanta released. As the cumulative EPSCs can be used to compare the total number of released quanta during high-frequency trains (Taschenberger and von Gersdorff, 2000), we used 50-Hz trains of 15 stimuli to induce EPSCs at the 3 postnatal studied ages (Fig. 8A). With this approach, we found different EPSC amplitudes along postnatal development (Fig. 8B). Also, the cumulative amplitude of EPSCs at P3-5 was 27.0% ± 4.1% to that found at P13-18, whereas the cumulative amplitude of EPSC at P7-9 was 91.6% ± 9.2% of that exhibited by neurons at the oldest analyzed age (Fig. 8C). Estimation of the number of quanta released can be obtained by dividing the cumulative amplitude of EPSC by the AMPA quantum (Taschenberger and von Gersdorff et al., 2000). These analyses revealed that the stimulus of 50-Hz trains induced an increase of the estimation of quanta released at P7-9 and P13-18 in comparison with P3-5 (F2,48 = 9.2, p = 0.0004; 1-way ANOVA; Fig. 8D). Moreover, the initial release probability (i.e., the ratio between the first and the total pool size; cumulative EPSCs; Taschenberger and von Gersdorff, 2000) yielded values of 0.26 ± 0.09 for P3-5, 0.20 ± 0.04 for P7-9, and 0.14 ± 0.02 for P13-18. Furthermore, we found a positive and significant correlation between the probability of STF in the paired pulses and the estimation of vesicle released with the 50-Hz train stimulation (ρ = 0.26, p = 0.045, 54 pairs, Spearman test; Suppl. Fig. S3). All of these results support the idea that there is a relationship between the type of STP and the estimated quanta released, and thus implying an increase in the release sites but a lower release probability at older postnatal ages in RHT terminals.
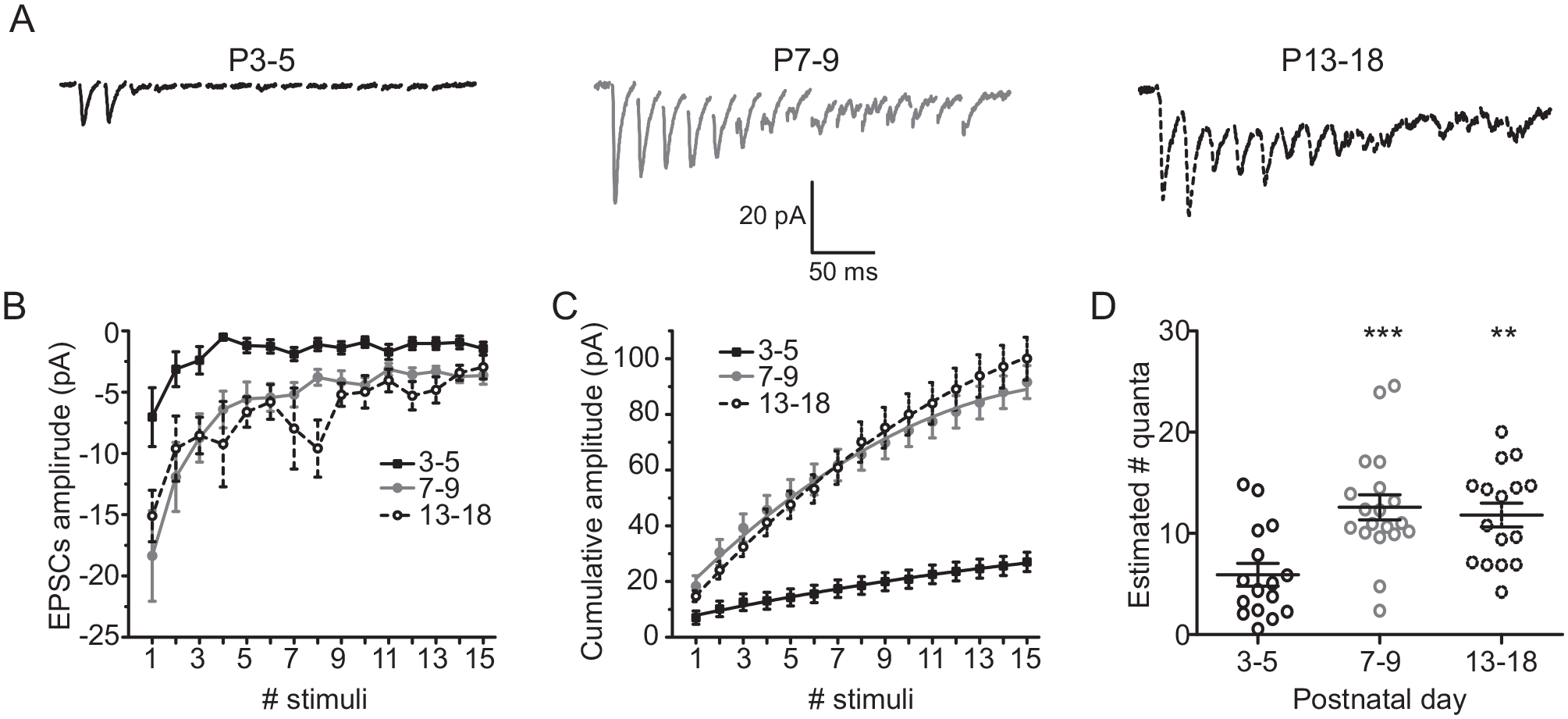
Developmental differences in the excitatory postsynaptic currents (EPSCs) elicited by 50-Hz stimulation trains delivered at the retinohypothalamic tract (RHT). (A) Current traces (average of 10 trials) induced by 15 stimuli trains of 50 Hz in neurons belonging to the 3 postnatal stages. (B) Average amplitude of the EPSCs generated by each of the 15 stimuli trains. (C) Cumulative amplitude of the EPSCs elicited by the 50-Hz trains, obtained from measurements as for panel B. (D) Estimated number of quanta released by RHT terminals as a consequence of the 50-Hz train stimulation, along postnatal development. Dunnett’s post hoc test: **p < 0.01 and ***p < 0.001 versus P3-5.
Cyclothizide Does Not Presynaptically Increase the Amount of Neurotransmitter Release
As we found changes in the STP in the presence of CTZ, a possible explanation could be an increase of neurotransmitter release by removal of the desensitization of presynaptic AMPA receptors. To test this possibility, we analyzed the PPR of NMDA EPSCs before and after CTZ administration. We did not find significant differences in the PPR between the control conditions and after the addition of 20 µM CTZ at any of the 3 ages investigated (Fig. 9). These results ruled out a presynaptic modulation of AMPA receptors during postnatal development.
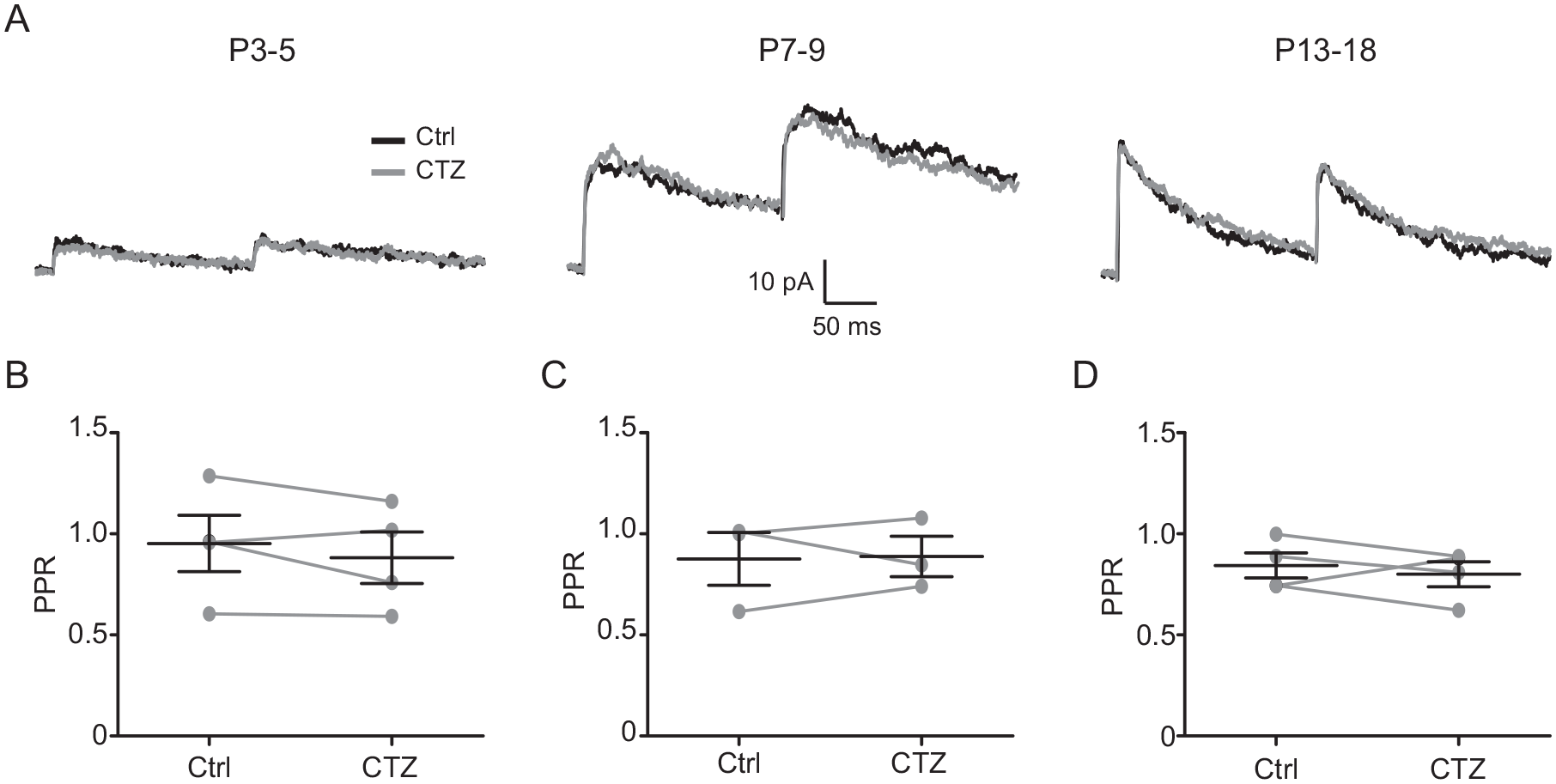
Effect of cyclothiazide (CTZ) on the activation of presynaptic AMPA receptors by paired pulse application. (A) N-methyl-D-aspartate current recordings obtained in the suprachiasmatic nucleus ventral neurons as a result of 5-Hz retinohypothalamic tract stimulation, before (Ctrl) and after the administration of 20 µM CTZ. The paired pulse ratio was not significantly modified by CTZ at postnatal day 3 to 5 (P3-5; B), P7-9 (C), or P13-18 (D).
Discussion
The main results of this work are as follows. There is a development-dependent increase of miniature and quantal EPSCs and a larger increment of the evoked AMPA-kainate EPSCs than that for the evoked NMDA at ages older than P3-5. As far as we know, this is the first research to study developmental functional changes in synapses from unmyelinated axons, whereas most investigations of this type have been performed in fast synapses, such as the auditory and visual systems, and the hippocampal circuitry.
Our findings show an increment of ~40% in both miniature NMDA and quantal AMPA
On the other hand, to our surprise we found a larger augmentation in the functional expression of AMPA-kainate receptors than that observed for NMDA receptors. This is an interesting result because several lines of evidence highlight the role of NMDA receptors in the activation of the signaling cascades that control the synchronization to light (Ding et al., 1994; Ding et al., 1997). Therefore, our results implicate the importance of AMPA-kainate receptors in the physiological fine-tuning of glutamatergic synapses in ventral SCN neurons. AMPA-kainate receptors are involved in the Mg2+ removal from the pore of the NMDA receptor and its subsequent activation; thus, it is tempting to speculate that a greater functional expression of AMPA-kainate receptors will promote the stimulation of NMDA receptors by glutamate released from RHT terminals. During development, an increment of the AMPA-kainate component accompanied by a decrease in the NMDA component has been reported (Hsia et al., 1998; Chen and Regehr, 2000; Lu and Trussell, 2007; Case at al., 2011), which contrasts with the augmentation of the latter in unmyelinated axons and supports the relevance of NMDA receptors in the RHT-SCN pathway. Also, with growth, we observed an increase in the decay time of electrically evoked NMDA but not the AMPA-kainate component, which differs from the developmental decrease of this parameter reported for several sensory information–processing synapses (Hsia et al., 1998; Chen and Regehr, 2000; Taschenberger and von Gersdorff, 2000; Lu and Trussell, 2007; Case et al., 2011). Importantly, in the experiments of the decay time of the isolated AMPA-kainate component, the fact that lower stimulation intensity was required at older postnatal ages to induce comparable amplitudes suggests that RHT-SCN synapses are strengthened more at these developmental stages, which could involve some of the above mentioned pre- and postsynaptic mechanisms, such as a higher number of release sites, increase in AMPA-kainate postsynaptic receptors, and so forth.
Electrical activity is very important for guiding the wiring of neural connections (Aamodt and Constantine-Paton, 1999). Our data of the more depolarized membrane resting potential of SCN neurons at younger ages is consistent with that reported for other brain areas (Dehorter et al., 2011; Sanchez et al., 2012). Considering the voltage-dependent Mg2+ blockade of NMDA receptors, the development-dependent resting potential of SCN neurons could be relevant for a differential activation of these receptors, especially at earlier postnatal stages, when the total expression of AMPA-kainate receptors has not yet been achieved. Moreover, a depolarized potential suggests that developmental synaptic plasticity of SCN ventral neurons is the result of spontaneous activity, as shown for the auditory system (Wang and Bergles, 2015). It is likely that the development of SCN neurons relies on mechanisms independent of action potentials at depolarizing conditions (due to inactivation of Na+ channels), whereas the more hyperpolarized potential at older ages would allow firing of action potentials. Indeed, before hearing onset in the nucleus laminaris of the auditory brainstem of the chick, high-frequency synaptic activity produces only 1 or 2 action potentials (Sanchez et al., 2012).
It is interesting that we found an increase in the decay kinetics of the evoked NMDA component (Fig. 4A, E), but this was not evident in the miniature NMDA EPSCs (Fig. 2A). It has been shown that evoked glutamate release can induce glutamate spillover and activate extrasynaptic NMDA receptors, which can be of different NR2 subunit subtypes (Alamilla and Gillespie, 2011), whereas synaptic NMDA receptors are activated by glutamate released by low-frequency evoked events, spontaneous synaptic events, and miniature synaptic events (Hardingham and Bading, 2010). Probably, our recordings of miniature NMDA EPSCs were due to synaptic NMDA receptors, whereas the evoked release of glutamate recruited the activation of extrasynaptic NMDA receptors. In addition, a modification in the current decay kinetics could imply a change of the subunits that conform the ionotropic glutamatergic receptor. In fact, we have previously reported a development-dependent expression of NR2C and NR2D subtypes of the NMDA receptors (Herrera-Zamora et al., 2019), which could explain the enhancing of the decay time of the evoked NMDA EPSCs.
At older ages, we found an increment in the number of evoked quantal events as well as a higher probability of STF (in the PPR) that could be accounted for by similar synaptic mechanisms, such as an increase in the excitability of RHT terminals and in the number of release sites (Chamberland et al., 2014), which, in turn, could be determined by a rise in the projections coming from the retina (Speh and Moore, 1993). Likewise, our results agree with the reported attenuation of synaptic depression at more developed ages (Bolshakov and Siegelbaum, 1995; Taschenberger and von Gersdorff, 2000; Iwasaki and Takahashi, 2001; Taschenberger et al., 2002; Case et al., 2011; Alamilla and Gillespie, 2013). The pre- and postsynaptic mechanisms associated with the reduction of synaptic depression during development include augmentation of the pool of synaptic vesicles (Blue and Parnavelas, 1983; Taschenberger and von Gersdorff, 2000; Iwasaki and Takahashi, 2001), increment of active zones or release sites (Taschenberger et al., 2002; Chamberland et al., 2014), decrease in release probability (Bolshakov and Siegelbaum, 1995; Taschenberger and von Gersdorff, 2000; Iwasaki and Takahashi, 2001; Taschenberger et al., 2002), and reconfiguration of calcium machinery (Iwasaki and Takahashi, 1998; Fedschyshyn and Wang, 2005; Alamilla and Gillespie, 2013). Furthermore, the estimation of the released quanta by stimulation trains can be explained by an increment in the synaptic vesicle pool and in the number of release sites, although the latter seems more plausible because our data were not obtained from a single synapse. These processes may enhance the amount of neurotransmitter released by the second stimulus of the paired pulse, particularly when the frequency is relatively high, which implies that the synapse has acquired the ability to respond to such frequencies of stimulation. Previously, it was found that RHT terminals mainly display paired-pulse depression and STD during train stimulation (Moldavan and Allen, 2010; Moldavan and Allen, 2013). In the absence of CTZ, the inhibitor of AMPA receptor desensitization, we observed no difference in the PPR nor a significant trend to exhibit a particular type of synaptic plasticity during postnatal development. However, in the presence of CTZ, we noticed a higher probability of STF in the PPR and a decrease in the synaptic depression when stimulation trains were applied. However, the compound (Aniracetam) used by Moldavan and Allen (2010) to study the effects of desensitization on the steady state of stimulus train modulates the AMPA receptor kinetics in a different manner to that produced by CTZ (Sekiguchi et al., 2002). At older postnatal ages, we deem that similar synaptic mechanisms underlie a higher tendency for STF in the PPR and a reduction in the synaptic depression by the train of stimuli, although the latter was more patent because the synapse was under a more demanding situation. Notably, at older ages, the synaptic depression by stimulation trains was not as evident as that found at earlier ages.
It is worth noting the pioneering study by Duncan and colleagues (1986), who demonstrated with behavioral experiments that the rat pup is able to synchronize to the light-dark cycle at a time point close to P7. Our results imply that the developmental stage between P7-9 is more similar to the oldest examined group (P13-18) than to the earliest analyzed stage (P3-5). It is likely that synchronization to light is not achieved before P7 because synaptic changes that enable SCN ventral neurons to depolarize (due to photic information) have not taken place yet. After P7, there are pre- and postsynaptic changes that prepare the SCN for a robust postsynaptic response induced by glutamate released from RHT terminals, which elicits a higher calcium influx necessary to differentially activate signaling pathways (depending on the time of the light stimulation) that induce the expression of clock genes, thus ending in delays and advances of phase shifts (Hirota and Fukada, 2004; Suppl. Fig. S4).
In conclusion, this investigation provides evidence of pre- and postsynaptic changes occurring during postnatal development between the RHT and ventral SCN neurons (including an increment in the glutamatergic quantal content, a larger augmentation of the evoked AMPA-kainate EPSCs than those of NMDA, and enhancement of the decay time of electrically evoked NMDA EPSCs), implying modifications in the quantal glutamatergic content and in the expression of glutamatergic postsynaptic receptors as well as changes in the STP, which support an increment in the estimated number of quanta released and a decrease in the probability of release. These changes seem to be inherent to the synaptic development rather than exclusive to fast synaptic transmission and could be important for the circadian clock to achieve a proper synchronization to the light-dark cycle at adult stages.
Supplemental Material
supp_fig_1 – Supplemental material for Functional Pre- and Postsynaptic Changes between the Retinohypothalamic Tract and Suprachiasmatic Nucleus during Rat Postnatal Development
Supplemental material, supp_fig_1 for Functional Pre- and Postsynaptic Changes between the Retinohypothalamic Tract and Suprachiasmatic Nucleus during Rat Postnatal Development by Miriam E. Reyes-Mendez, Fernando Osuna-López, J. Manuel Herrera-Zamora, Ricardo A. Navarro-Polanco, Eloy G. Moreno-Galindo and Javier Alamilla in Journal of Biological Rhythms
Supplemental Material
supp_fig_2 – Supplemental material for Functional Pre- and Postsynaptic Changes between the Retinohypothalamic Tract and Suprachiasmatic Nucleus during Rat Postnatal Development
Supplemental material, supp_fig_2 for Functional Pre- and Postsynaptic Changes between the Retinohypothalamic Tract and Suprachiasmatic Nucleus during Rat Postnatal Development by Miriam E. Reyes-Mendez, Fernando Osuna-López, J. Manuel Herrera-Zamora, Ricardo A. Navarro-Polanco, Eloy G. Moreno-Galindo and Javier Alamilla in Journal of Biological Rhythms
Supplemental Material
supp_fig_3 – Supplemental material for Functional Pre- and Postsynaptic Changes between the Retinohypothalamic Tract and Suprachiasmatic Nucleus during Rat Postnatal Development
Supplemental material, supp_fig_3 for Functional Pre- and Postsynaptic Changes between the Retinohypothalamic Tract and Suprachiasmatic Nucleus during Rat Postnatal Development by Miriam E. Reyes-Mendez, Fernando Osuna-López, J. Manuel Herrera-Zamora, Ricardo A. Navarro-Polanco, Eloy G. Moreno-Galindo and Javier Alamilla in Journal of Biological Rhythms
Supplemental Material
supp_fig_4 – Supplemental material for Functional Pre- and Postsynaptic Changes between the Retinohypothalamic Tract and Suprachiasmatic Nucleus during Rat Postnatal Development
Supplemental material, supp_fig_4 for Functional Pre- and Postsynaptic Changes between the Retinohypothalamic Tract and Suprachiasmatic Nucleus during Rat Postnatal Development by Miriam E. Reyes-Mendez, Fernando Osuna-López, J. Manuel Herrera-Zamora, Ricardo A. Navarro-Polanco, Eloy G. Moreno-Galindo and Javier Alamilla in Journal of Biological Rhythms
Footnotes
Acknowledgements
This work was supported by Consejo Nacional de Ciencia y Tecnología (CONACYT): CB-2015-01- 254887 to J.A.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
