Abstract
The suprachiasmatic nucleus (SCN) is the main brain clock that regulates circadian rhythms in mammals. The SCN synchronizes to the LD cycle through the retinohypothalamic tract (RHT), which projects to ventral SCN neurons via glutamatergic synapses. Released glutamate activates N-methyl-D-aspartate (NMDA) receptors, which play a critical role in the activation of signaling cascades to enable phase shifts. Previous evidence indicates that presynaptic changes during postnatal development consist of an increase in RHT fibers impinging on SCN neurons between postnatal day (P) 1 to 4 and P15. The aim of this study was to evaluate postsynaptic developmental changes in the NR2 subunits that determine the pharmacological and biophysical properties of the neuronal NMDA receptors in the ventral SCN. To identify the expression of NR2 subtypes, we utilized RT-PCR, immunohistochemical fluorescence, and electrophysiological recordings of synaptic activity. We identified development-dependent changes in NR2A, C, and D subtypes in mRNA and protein expression, whereas NR2B protein was equally present at all analyzed postnatal ages. The NR2A antagonist PEAQX (100 nM) reduced the frequency of NMDA excitatory postsynaptic currents (EPSCs) at P8 significantly more than at P34, but the antagonists for NR2B (3 μM Ro 25-6981) and NR2C/D (150 nM PPDA) did not influence NMDA EPSCs differently at the 2 analyzed postnatal ages. Our results point to P8 as the earliest analyzed postnatal age that shows mRNA and protein expression similar to those found at the juvenile stage P34. Taken together, our findings indicate that postsynaptic development-dependent modifications in the NR2 subtypes of the NMDA receptor could be important for the synchronization of ventral SCN neurons to the LD cycle at adult stages.
The hypothalamic suprachiasmatic nucleus (SCN) is the main clock responsible for the regulation of circadian rhythms. The SCN synchronizes with the environmental LD cycle through the retinohypothalamic tract (RHT). Ablation of the RHT abolishes the synchronization of circadian rhythms to the LD cycle, allowing its free-running expression (Johnson et al., 1988). The RHT originates from a population of melanopsin-containing photosensitive ganglion cells in the retina (Hankins et al., 2008) and projects its fibers to various hypothalamic nuclei, including the SCN. The RHT releases glutamate and the pituitary adenylate cyclase-activating polypeptide (Hannibal, 2006; You et al., 2012) to the ventrolateral part of the SCN, which receives its principal innervation from the retina (Abrahamson and Moore, 2001). Glutamate released from the RHT activates ionotropic glutamatergic α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA)/kainate and N-methyl-D-Aspartate (NMDA) receptors (Kim and Dudek, 1991). Activation of NMDA receptors is a crucial step in phase shifts induced by light (Colwell and Menaker, 1992), which subsequently activate signaling pathways to achieve phase advances or delays (Ding et al., 1994; Ding et al., 1997).
The NMDA receptor is a heteromeric protein principally composed of 2 NR1 and 2 NR2 subunits. The 4 known types of subunits are termed NR2A to NR2D. The 2 NR2 subunits of the NMDA receptor can comprise the same or different types, thus providing diversity to the biophysical and pharmacological properties of NMDA receptors (see Cull-Candy and Leszkiewicz, 2004). Several reports indicate that the NR2B subtype plays an important role in the synchronization to light. In SCN neurons, Clark and Kofuji (2010) found that the NMDA current is reduced to about 50% by the NR2B antagonist ifenprodil. It was furthermore shown in mice that ifenprodil microinjected into the SCN region significantly reduces light-induced phase shifts in wheel running activity and that the phosphorylated form of this NR2 subtype varies in the ventral SCN according to circadian rhythms (Wang et al., 2008).
When NR2A and NR2C are individually knocked out in the whole animal, they do not seem to play a role in the synchronization of SCN neurons to light, even though there is biophysical and pharmacological evidence of their presence in NMDA currents of SCN neurons. Clark and Kofuji (2010) showed that the remaining NMDA current after NR2B inhibition with ifenprodil presents pharmacological characteristics of the NR2A subtype, even though they exhibit a Mg2+ sensitivity similar to that of NR2C and NR2D subunits. Thus, these authors suggest that a subpopulation of NMDA receptors in SCN neurons consists of NR2A and NR2C/D heteromers. Moriya and colleagues (2000) showed that synchronization to light was slower in NR2A-knockout animals under low-intensity light conditions, indicating that NR2A indeed plays a role in synchronization. However, no effects were found in NR2C-knockout animals, and the role of the NR2D subunit in the synchronization of SCN neurons to light has not been reported yet.
Pre- and postsynaptic changes during development are critical for all sensory systems to prepare them for an optimal performance at adult stages, and the photic circadian system is no exception. It has been reported that RHT fibers start the SCN innervation at an early stage, i.e., postnatal days (P) 1 to 4 (Mason et al., 1977; Munoz Llamosas et al., 2000; Speh and Moore, 1993; Stanfield and Cowan, 1976); although, functional Fos immunoreactivity is already found at P0 (Hannibal and Fahrenkrug, 2004; Sekaran et al., 2005). The RHT achieves an adult appearance at P10 to P15 (Frost et al., 1979; Guldner, 1978; Mason et al., 1977; Munoz Llamosas et al., 2000; Speh and Moore, 1993). At the behavioral level, neonatal rats synchronize to LD cycles by P7 (Duncan et al., 1986), when the maternal influence wanes (Reppert and Schwartz, 1986) and the RHT synaptic terminals innervate the SCN neurons. Anatomical and behavioral evidence of the maturation in RHT - SCN connections may imply pre- and postsynaptic changes during this development that help to prepare the circadian system to correctly synchronize to light. To further understand these processes, this study was aimed to determine the postsynaptic changes in NR2 subunit expression (both at the mRNA and protein levels) in rats during development and determine their influence on synaptic activity.
Materials and Methods
All studies were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996). All procedures were approved by the Institutional Bioethics Committee of the University Center for Biomedical Research of the University of Colima. Experiments were carried out in Wistar rat pups (n = 111) bred in our animal facilities from parents supplied by Harlan (Mexico). We analyzed postnatal developmental changes in the ventral SCN of animals at the ages of P4, P8, P12, and P34. For tissue sampling, animals were euthanized (between ZT 3 and 6) with sodium pentobarbital (125 mg/kg, i.p.).
RNA Extraction and Quantitative Real-Time PCR Analysis
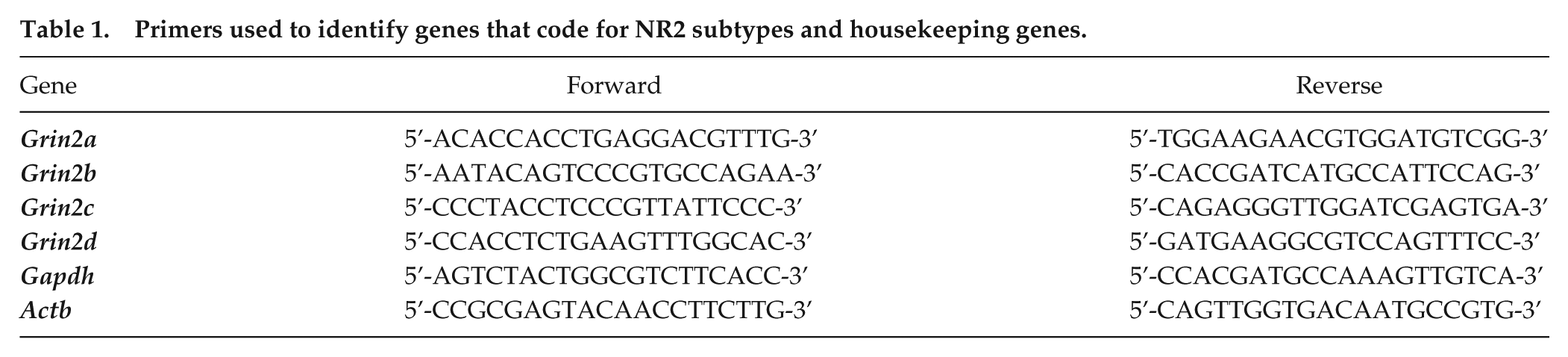
Coronal brain slices (300 µm) containing the hypothalamus were obtained with a VT1000 microtome (Leica Microsystems; Wetzlar, Germany). Brain slices were maintained in artificial cerebrospinal fluid (ACSF) containing 124 mM NaCl, 1 mM MgSO4, 5 mM KCl, 1.25 mM KH2PO4, 10 mM dextrose, 26 mM NaHCO3, 2 mM CaCl2; bubbled with 95% O2/5% CO2; pH 7.4. Tissue from the SCN was microdissected from 8 animals belonging to each of the above-mentioned postnatal ages (Fig. 1A). Total RNA was extracted from the pool (20 mg) of SCN samples using the Aurum Total RNA Mini Kit (Bio-Rad; Hercules, CA). RNA (500 ng) was reverse transcribed using an iScript cDNA Synthesis Kit (Bio-Rad). The cDNA was used as a template for real-time PCR experiments using the iQ SYBR Green Supermix (Bio-Rad) with the primers for the following genes that code for the different NR2 subtypes Grin2a (NR2A), Grin2b (NR2B), Grin2c (NR2C), and Grin2d (NR2D) in addition to the housekeeping genes Gapdh (glyceraldehyde-3-phosphate dehydrogenase) and Actb (β-actin) (Table 1). Specific primers were purchased from Invitrogen (Carlsbad, CA). Real-time PCR was performed using a CFX96 detection system (Bio-Rad). The thermal cycling parameters were 95°C for 3 min, 40 cycles of 95°C for 15 s, followed by 60°C for 1 min. The experiment for each postnatal age was run in triplicate and normalized to Gapdh and Actb mRNA levels. We used Actb as an additional housekeeping gene because it has been shown that the mRNA levels remain very steady throughout the development of the whole brain in Wistar rats (Fedoseeva et al., 2014). Three replicates of measurements from the same pool were then averaged, and the fold induction was determined in a ∆∆CT-based fold-change calculation. Furthermore, raw data (not normalized to P4) were obtained with the ∆CT method (Schmittgen and Livak, 2008). A melting curve analysis was performed to confirm the specificity of the PCR products.
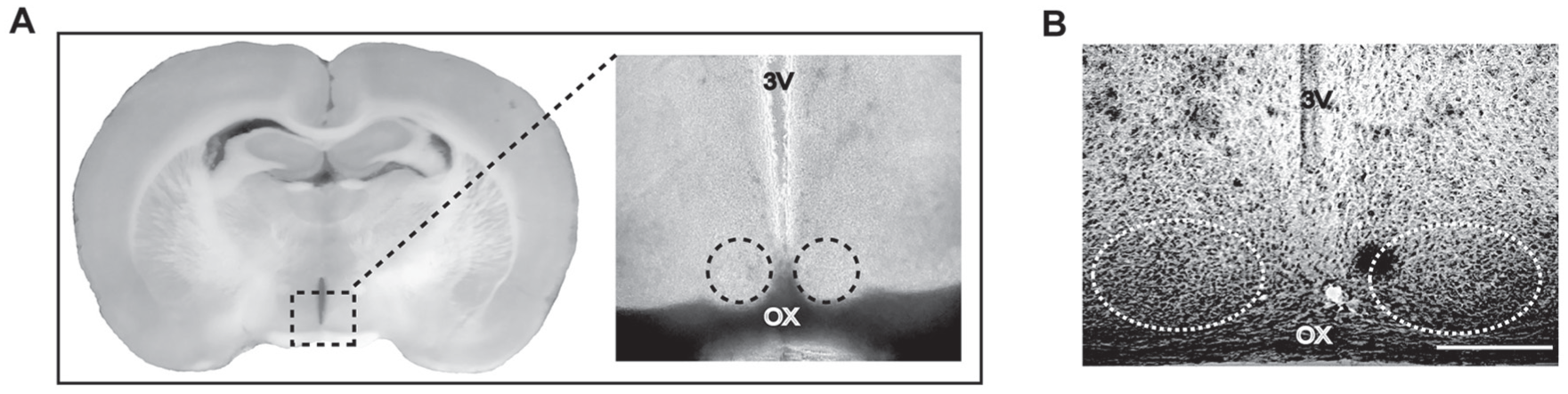
Coronal brain slices containing the SCN used in the different methodological approaches of this study. (A) Slice corresponding to a P12 animal, illustrating the sectioned area where RT-PCR assays were performed. (B) Ventral and medial SCN examined for immunofluorescence experiments. Coronal slice of a P8 animal imaged with transmitted light containing the ventral SCN and reference structures such as the third ventricle (3V) and optic chiasm (OX). Circles indicate the ventral parts of the SCN analyzed in confocal microscopy. Scale bar, 150 µm.
Primers used to identify genes that code for NR2 subtypes and housekeeping genes.
Immunohistochemistry
A total of 47 rat pups were used at different postnatal ages: 15 animals for P4, 14 animals for P8, 11 for P12, and 7 for P34. After the animals were euthanized with sodium pentobarbital (125 mg/kg), the brain was perfused transcardially with 0.1 M phosphate-buffered saline (PBS; pH 7.4), followed by cold (4°C) 4% paraformaldehyde diluted in 0.1 M phosphate buffer (PB; pH 7.4). Thereafter, the brain was extracted and post-fixed in 4% paraformaldehyde for 1 h before being transferred to 10% sucrose (overnight), 20% sucrose (24 h), and finally 30% sucrose (48 h). Consecutive frozen coronal sections (40 µm) containing the hypothalamus were obtained with an SM2010R microtome (Leica) and collected into 0.1 M PBS. Brain sections belonging to the different postnatal ages were stained for the antibodies listed below and imaged at the same time for consistency between runs. On average, 4 consecutive sections (per brain) were carefully chosen with the help of a stereomicroscope (MicroscoSB; Commack, NY). They contained the medial SCN and were immunostained for protein pairs consisting of 1 of the 4 NR2 subunit subtypes of the NMDA receptor (A to D) and the scaffolding protein PSD-95; i.e., NR2A and PSD-95, NR2B and PSD-95, NR2C and PSD-95, NR2D and PSD-95. Immunofluorescence labeling was performed with free-floating sections at 4°C for 72 h in 1% normal donkey serum and 0.03% Triton X-100 in 0.1 M PBS (PBSDT). Secondary antibodies were incubated in PBSDT for 2 h at room temperature in black tubes protected from light. Slices were then washed in PBS (3 times, 10 min each), mounted, and coverslipped with 97% 2,2’-thiodiethanol (Staudt et al., 2007).
Antibodies
All antibodies used in this study (Table 2) were tested and characterized by the supplier companies. Negative control experiments were performed by incubation with only the secondary antibody, and no unspecific immunolabeling was observed (Suppl. Fig. S1A). Dilution-fluorescence curves were carried out for primary antibodies (Table 2, Suppl. Fig. S1B). Secondary antibodies (anti-rabbit Alexa Fluor 488 and anti-mouse Alexa Fluor 647; both from Abcam, Cambridge, UK) were used at a 1:500 dilution.
Specifications of antibodies used in this research.
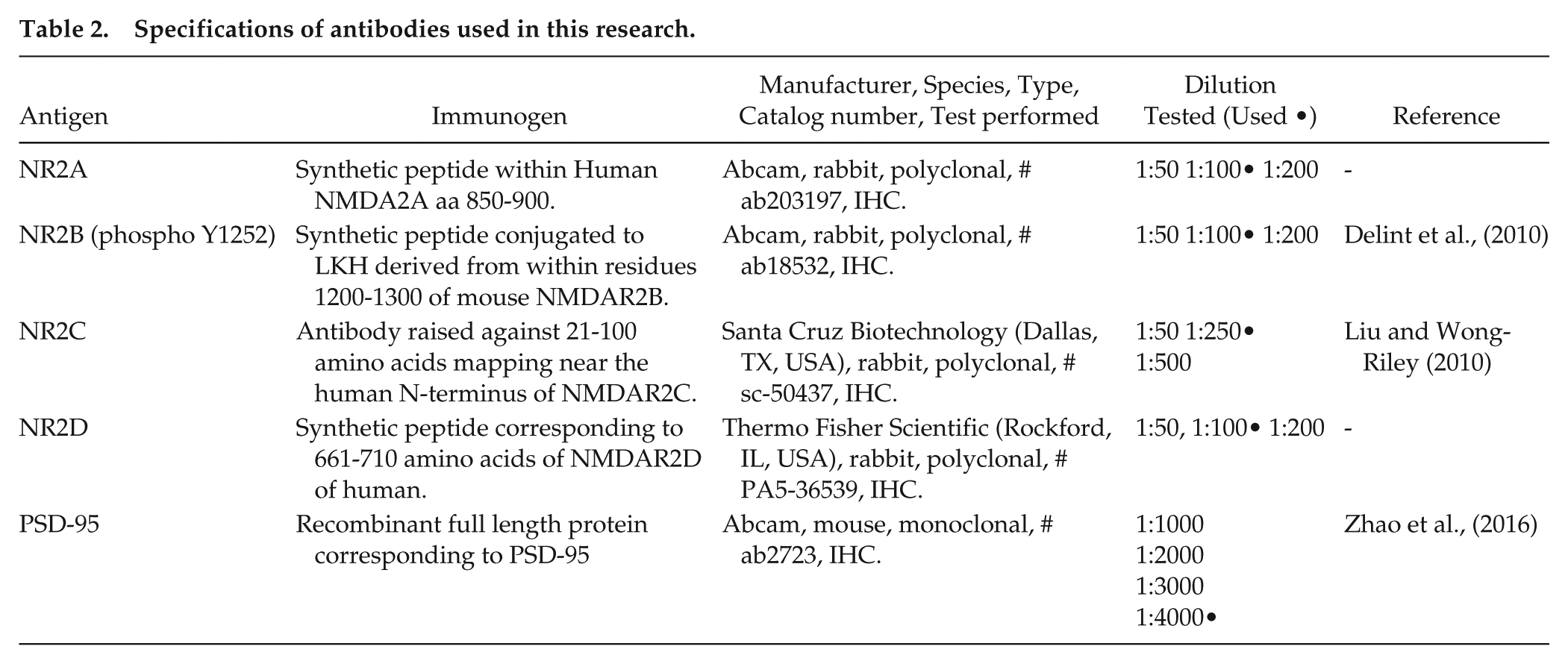
Image Acquisition and Analysis
SCNs were imaged with an LSM-700 confocal microscope (Carl Zeiss; Oberkochen, Baden-Württemberg, Germany). The best mounted and coverslipped brain slices containing the medial SCN were chosen for imaging by confocal microscopy (1 or 2 slices were selected from each animal at every stage of postnatal development) using the Zen software (Carl Zeiss) for image acquisition. A 10× objective (NA 0.3) was used to obtain a panoramic view of the SCN and adjacent structures. Then, 40× (NA 1.3) and 63× (NA 1.4) objectives were used to visualize the ventral SCN (Abrahamson and Moore, 2001) (Fig. 1B), and optical slices were obtained at 1 µm steps (on average, we obtained more than 20 optical slices per image). Two images (medial and lateral) were taken for each ventral SCN (left and right) for a total of 4 images for each slice. For intensity fluorescence measurements, PMT gain, laser intensity, and all other parameters (e.g., acquisition and exposure time) were adjusted for each age to show the highest brightness without saturation and were maintained constant for all subsequent images obtained within the same run.
Images were 3D blind deconvoluted using the AutoQuant X3 (Media Cybernetics Inc., Rockville, MD) and then analyzed using the ImageJ software (National Institutes of Health, Bethesda, MD). The signals were separated into 2 channels, and the mean pixel immunofluorescence intensity was measured in the entire image, i.e., the stack of optical slices (at least 20). A stack of optical slices was obtained with the maximum intensity projection function of the Zen software. Immunofluorescence intensity measurement obtained from medial and lateral images were pooled. Co-localization was analyzed on the entire image using the Zen software, and this co-localization analysis was performed on a pixel by pixel basis. Each pixel was plotted in a scatter diagram based on its intensity level from each channel. Thereafter, the Pearson correlation coefficient was calculated with a value of 1 indicating a perfect co-localization.
Electrophysiological Recordings
Spontaneous postsynaptic currents from ventral SCN neurons of slices obtained at P8 and P34 were recorded at room temperature (22°C to 24°C) in the whole-cell configuration of the patch-clamp technique using an Axopatch 200B amplifier (Molecular Devices; Sunnyvale, CA). Neurons were visualized with an upright Axioexaminer A1 microscope (Carl Zeiss). Data acquisition and command potentials were carried out with a Digidata 1322A interface controlled by the pClamp 8.2 software (Molecular Devices). Recording electrodes of 2 to 4 MΩ were made in a P97 puller (Sutter Instrument Co., Novato, CA) and filled with intracellular solution containing 64 mM D-gluconic acid, 64 mM CsOH, 11 mM EGTA, 56 mM CsCl, 1 mM MgCl2, 1 mM CaCl2, 10 mM HEPES, and 5 mM QX-314. Series resistance was compensated (~80%), and the seal was monitored throughout the whole experiment. ACSF Mg2+-free solution (124 mM NaCl, 1 mM K2SO4, 5 mM KCl, 1.25 mM KH2PO4, 10 mM dextrose, 26 mM NaHCO3, 2 mM CaCl2; bubbled with 95% O2/5% CO2; pH 7.4) was used as the external recording solution. Cells were discarded if the input resistance was less than 150 MΩ or the access resistance greater than 15 MΩ, or changed more than 15%. Currents were low-pass filtered at 2 kHz and sampled at 10 kHz.
Contribution of NR2 Subtypes to the Frequency and Amplitude of NMDA EPSCs
To analyze the contribution of NR2 subtypes to isolated spontaneous NMDA excitatory postsynaptic currents (EPSCs), 50 µM picrotoxin (a GABA-A receptor antagonist) and 5 µM 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX, an antagonist of AMPA/kainate receptors) were added to the external recording solution (ACSF Mg2+-free solution). After 5 min of recording basal NMDA isolated currents, each of the selective NR2 subtype antagonists were applied in a random sequence; for NR2A, 100 nM [[[(1S)-1-(4-Bromophenyl) ethyl]amino] (1,2,3,4-tetrahydro-2,3-dioxo-5-quinoxalinyl)methyl] phosphonic acid tetrasodium salt (PEAQX); for NR2B, 3 µM (αR,βS)-α-(4-Hydroxyphenyl)-β-methyl-4-(phenylmethyl)-1-piperidinepropanol maleate (Ro 25-6981); or for NR2C/D, 150 nM (2S*,3R*)-1-(Phenanthren-2-carbonyl)piperazine-2,3-dicarboxylic acid (PPDA). PEAQX has an IC50 of 8 nM on NMDA receptors containing the NR2A subunit. PEAQX is over 100-fold more selective to NR2A than NR2B subunits (Auberson et al., 2002). Thus, we chose a concentration of 100 nM, according to previous reports demonstrating the highest specificity for NR2A (Tackenberg et al., 2013). By contrast, Ro 25-6981 exhibits a higher affinity for NR2B over the NR2A subtype, since its IC50 values are 0.009 and 52 µM, respectively (Fischer et al., 1997). Hence, we used 3 µM Ro 25-6981 to detect the presence of NR2B subunits, which agrees with previous studies (Alamilla and Gillespie, 2011). Likewise, PPDA exhibits a Ki of 0.096, 0.125, 0.31, and 0.55 µM for the NR2C, D, B, and A subtypes, respectively (Morley et al., 2005). Based on this information, we assumed that 150 nM would be sufficient to distinguish between NR2C/D and NR2B/A subunits. The effects of these NR2 subtype blockers on the frequency and amplitude of NMDA EPSCs were analyzed, and the percentages of NR2A, NR2B, and NR2C/D subunit contributions were calculated for each recorded neuron.
Drugs and Reagents
Picrotoxin, CNQX, PEAQX, PPDA, and Ro 25-6981 were obtained from Tocris Bioscience (Ellisville, MO), and all other reagents were purchased from Sigma-Aldrich (St. Louis, MO).
Data Analysis
Data are reported as the mean ± SEM. GraphPad Prism 5 (GraphPad Software; La Jolla, CA) was used to perform statistical analyses. Where appropriate, the unpaired t-test or one-way ANOVA with Dunnett’s post-hoc test were used to evaluate statistical differences. The non-parametric Kruskal–Wallis test with Dunn’s post-hoc test was used to analyze the fold-change in mRNA expression (Hoang et al., 2009). A two-tailed alpha value of 0.05 (P < 0.05) was considered statistically significant.
Results
mRNA Expression of NR2 Subunits during Postnatal Development in the SCN
As mRNA expression of NR2 subtypes must be normalized to that of housekeeping genes according to the ΔΔCT method, we first sought to validate that the mRNA levels of the housekeeping genes displayed no development-dependent variation. Although there was a statistically significant Gapdh variation between P8 and P12, we did not find a significant difference in CT between the control age P4 and any other analyzed age (Suppl. Fig. S2). Likewise, Actb did not show a development-dependent variation in CT at all analyzed ages (Suppl. Fig. S2).
Next, the mRNA expression levels of NMDAR2 subtypes in the SCN were analyzed at different postnatal ages (Fig. 1A), using P4 as a reference (Fig. 2). Normalized to both housekeeping genes, Grin2a (NR2A subtype) mRNA expression levels only peaked at P8 (Fig. 2A). When normalized to Gapdh, Grin2b (NR2B) showed a decrease in mRNA expression at P12 and P34, whereas when normalized to Actb, mRNA levels significantly increased at P8 (Fig. 2B). The mRNA levels of Grin2c (NR2C) were elevated at P34 with respect to those at P4 (Fig. 2C) with a similar result for Grin2d (NR2D), which also displayed a significant increase in mRNA expression at P34 (Fig. 2D). Similar tendencies were found when data were not normalized to P4 (ΔCT) (Suppl. Fig. S3). These results indicate that mRNA levels of NR2 subtypes in SCN neurons are differentially expressed during postnatal development.
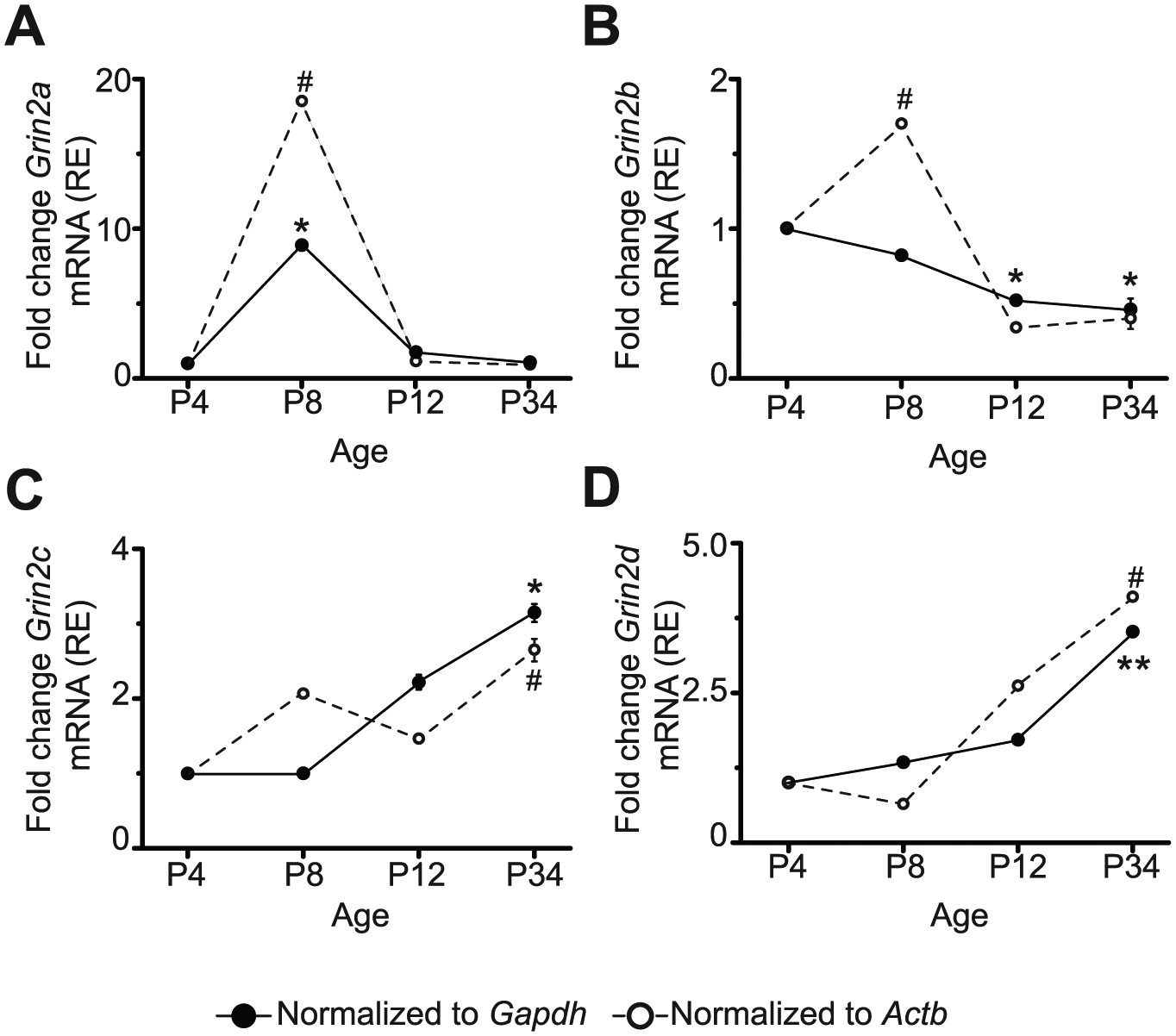
mRNA expression relative (RE) to P4 of the different NR2 subunits during the postnatal development of the rat. mRNA of Grin2a that encodes for NR2A (A), Grin2b for NR2B (B), Grin2c for NR2C (C), and Grin2d for NR2D (D) at postnatal ages 4, 8, 12, and 34. Data correspond to triplicate experiments of the same mRNA pool from 8 animals belonging to each analyzed postnatal age. *P < 0.05, **P < 0.01 vs. P4 normalized to Gapdh; #P < 0.05 normalized to Actb.
Developmental Expression of NR2 Subtypes and Scaffolding Protein PSD-95 in the Ventral SCN
Immunofluorescence intensities of NR2 subtypes were analyzed at different ages of postnatal development (Figs. 3A, 4A, 5A, and 6A). For NR2A, a higher fluorescence intensity was observed at P8 (79 ± 7 arbitrary units [a.u.]) and P34 (72 ± 8 a.u.) compared with P4 (49 ± 4 a.u.; F[3,56] = 5, P = 0.010; Fig. 3B). No significant differences in immunofluorescence were found for the NR2B subtype among all postnatal ages investigated (54 ± 6 a.u., 68 ± 13 a.u., 68 ± 15 a.u., and 63.4 ± 10 a.u. at P4, P8, P12, and P34, respectively; F[3,56] = 0.34, P = 0.8; Fig. 4B). The immunofluorescence intensities of the NR2C subunit were significantly increased at all time points (70 ± 5 a.u., 64 ± 3 a.u., and 69 ± 3 a.u. at P8, P12, and P34, respectively) compared with P4 (52 ± 3 a.u.; F[3,50] = 6.0, P = 0.0013; Fig. 5B). For the NR2D subtype, the immunofluorescence intensities were increased at all analyzed ages compared with that at P4 (47 ± 3 a.u., 71 ± 4 a.u., 67 ± 3 a.u., and 67 ± 2 a.u. at P4, P8, P12, and P34, respectively; F[3,60] = 3.7, P = 0.017; Fig. 6B). Similarly, the protein PSD-95 showed a development-dependent expression that was significantly higher at P8 and P12; although, the mean intensity at P34 did not differ compared with that at P4 (66 ± 4 a.u., 97 ± 4 a.u., 113 ± 8 a.u., and 78 ± 5 a.u. at P4, P8, P12, and P34, respectively; F[3,150] = 12, P < 0.0001; Fig. 7). Overall, these results suggest a differential developmental pattern of the protein expression of NMDA-NR2 subtypes as well as that of the NMDA scaffolding protein PSD-95.
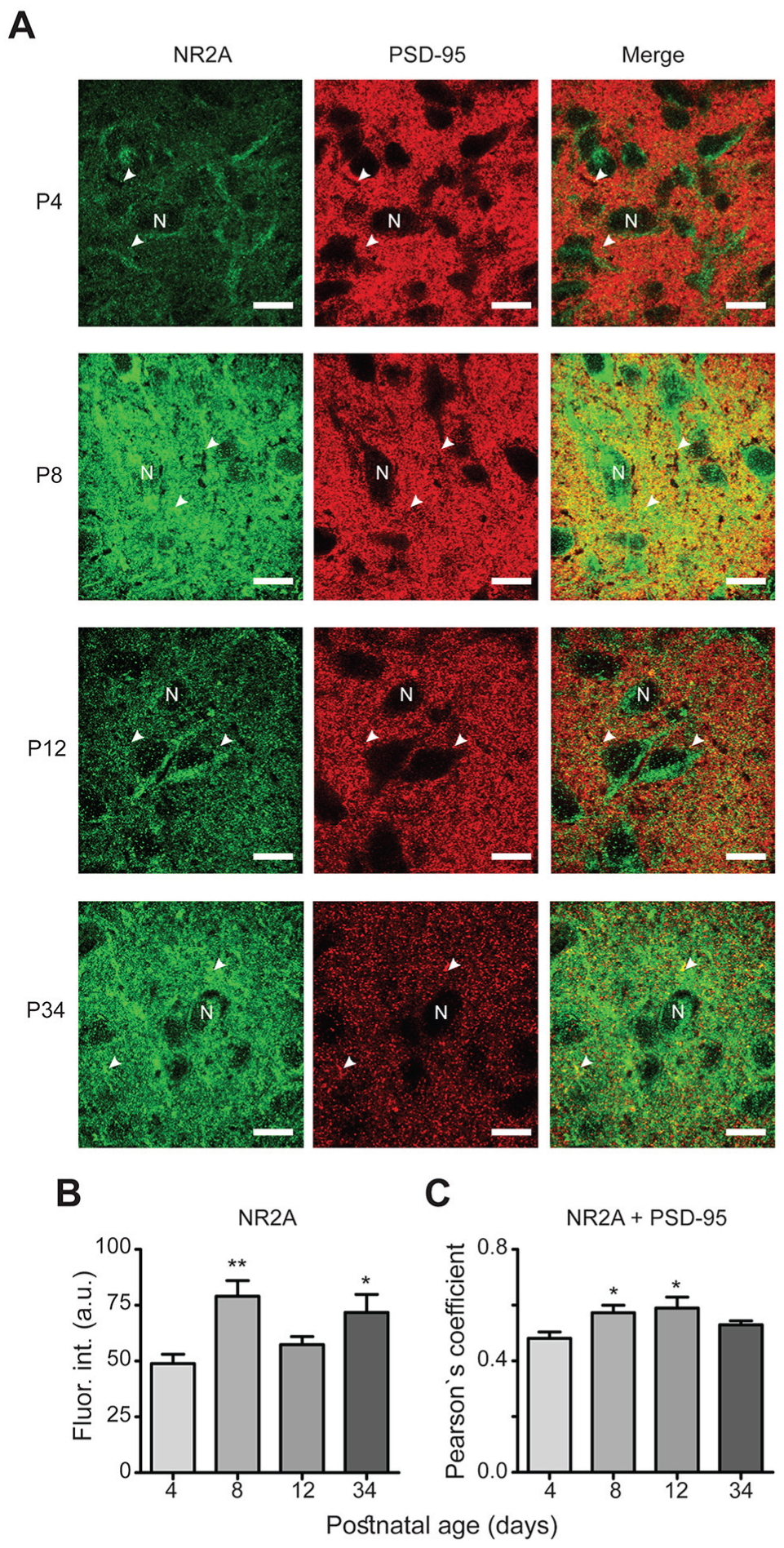
Developmental expression of the NR2A subtype. (A) For this and subsequent graphs in A panels from Figs. 3-6: green channel (first column) shows NR2 expression, NR2A in this case; red channel (second column) gives evidence for its corresponding PSD-95 immunoreactivity and the third column illustrates the merge of the two mentioned channels. A white letter N indicates a putative neuronal soma, and arrowheads depict co-localization dots of the NR2 subtype with PSD-95. Scale bars, 10 μm. (B) Fluorescence intensity (Fluor. int) in arbitrary units (a.u.) of the NR2A subtype at the 4 examined postnatal ages. (C) Co-localization of NR2A and PSD-95 denoted by Pearson’s coefficient. *P < 0.05, **P < 0.01 vs. P4.
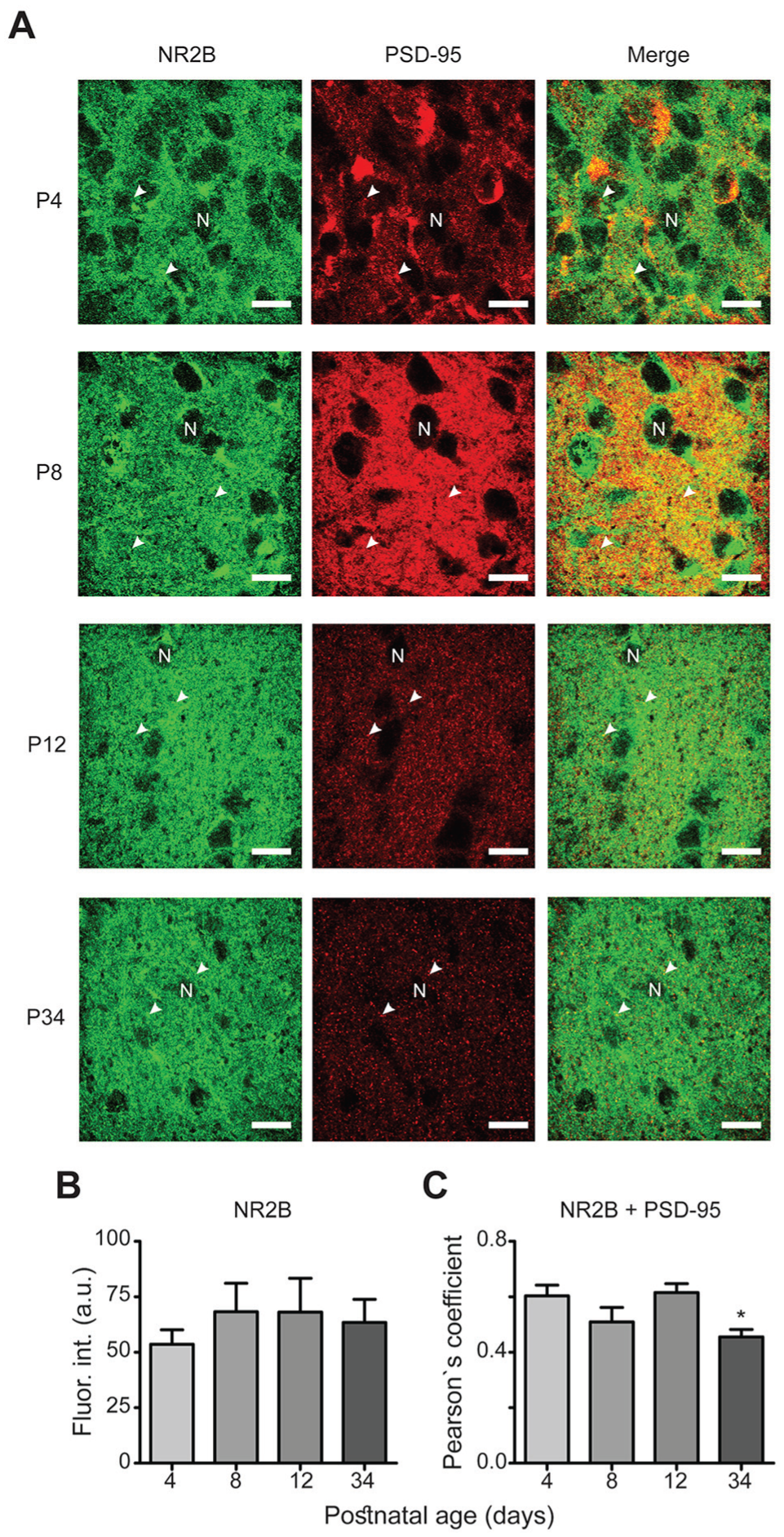
Expression of the NR2B subtype during postnatal development. (A) Similar micrographs as described for Fig. 3A (scale bars, 10 μm). (B) The fluorescence intensity of the NR2B subtype does not exhibit developmental changes at the analyzed postnatal ages. (C) Data from co-localization analyses of NR2B with the PSD-95 protein.
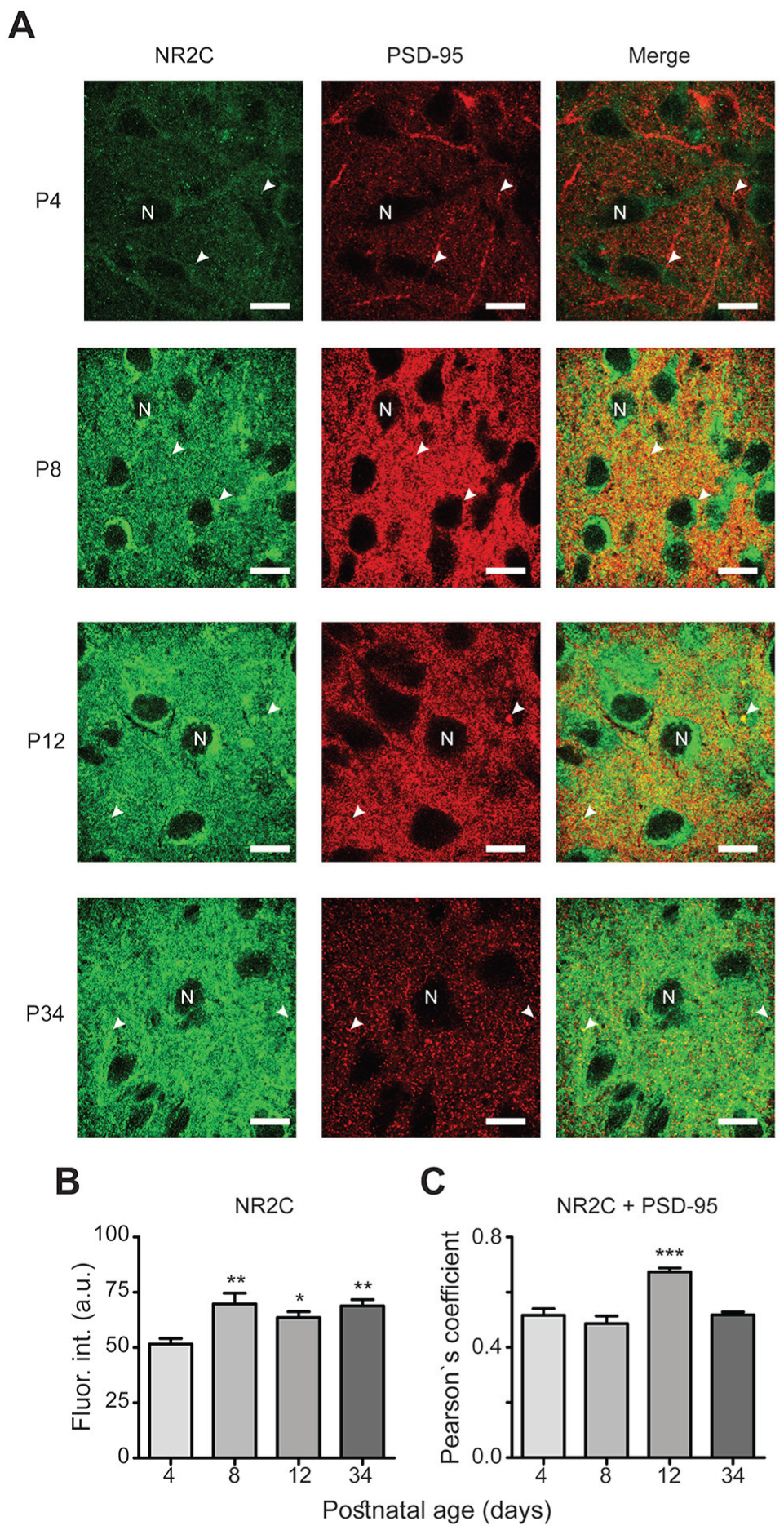
Development-dependent expression of the NR2C subtype. (A) Similar micrographs as described for Fig. 3A (scale bars, 10 μm). (B) Significant increase in NR2C immunofluorescence intensity at P8, P12, and P34. (C) Significantly augmented co-localization of NR2C and PSD-95 at P12. *P < 0.05, **P < 0.01, ***P < 0.001 vs. P4.
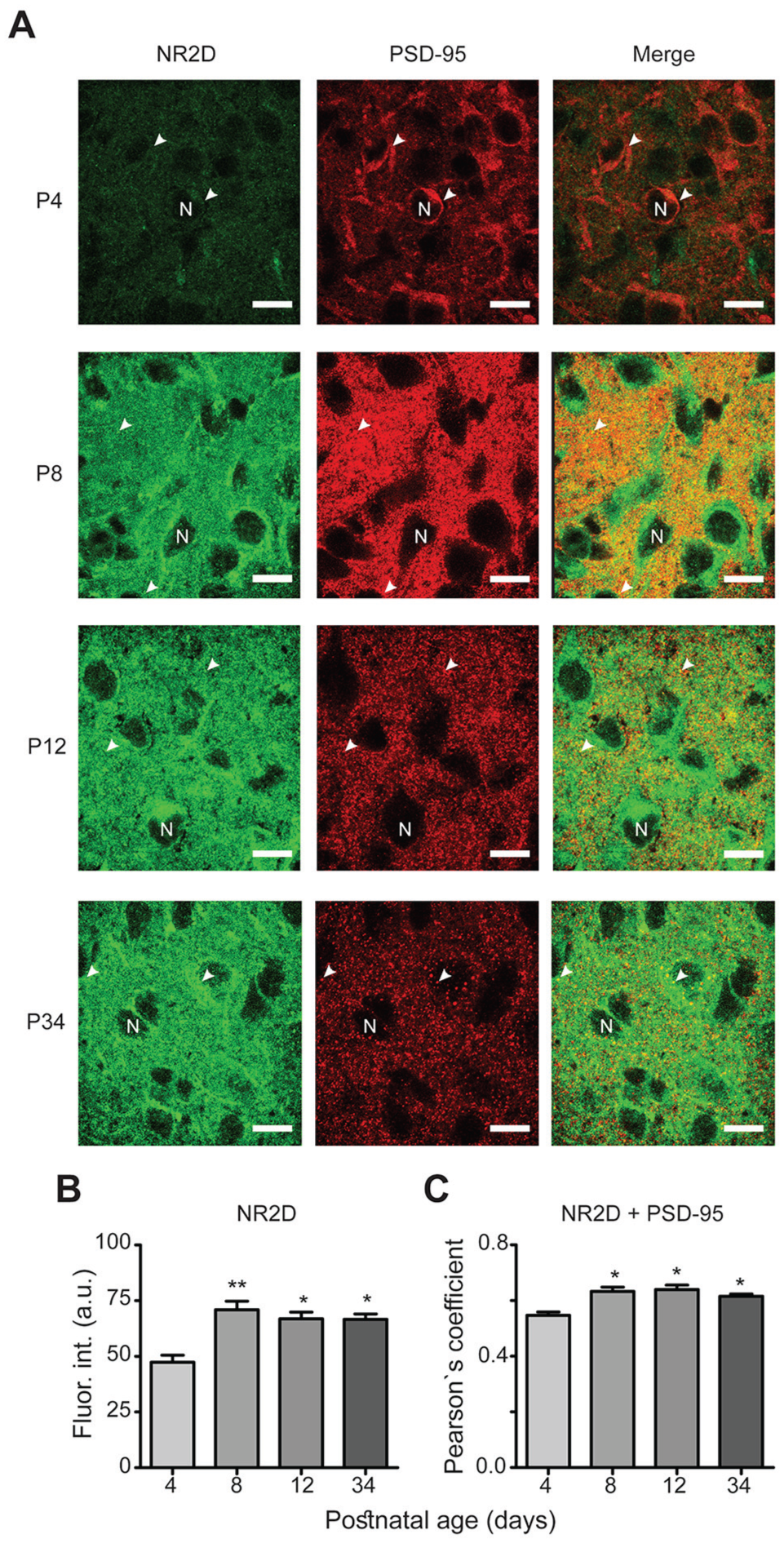
NR2D subtype expression during postnatal development. (A) Similar micrographs as described for Fig. 3A (scale bars, 10 μm). Immunofluorescence intensity is significantly increased after P8 (B), and similar situation in co-localization analyses (C). *P < 0.05, **P < 0.01 vs. P4.
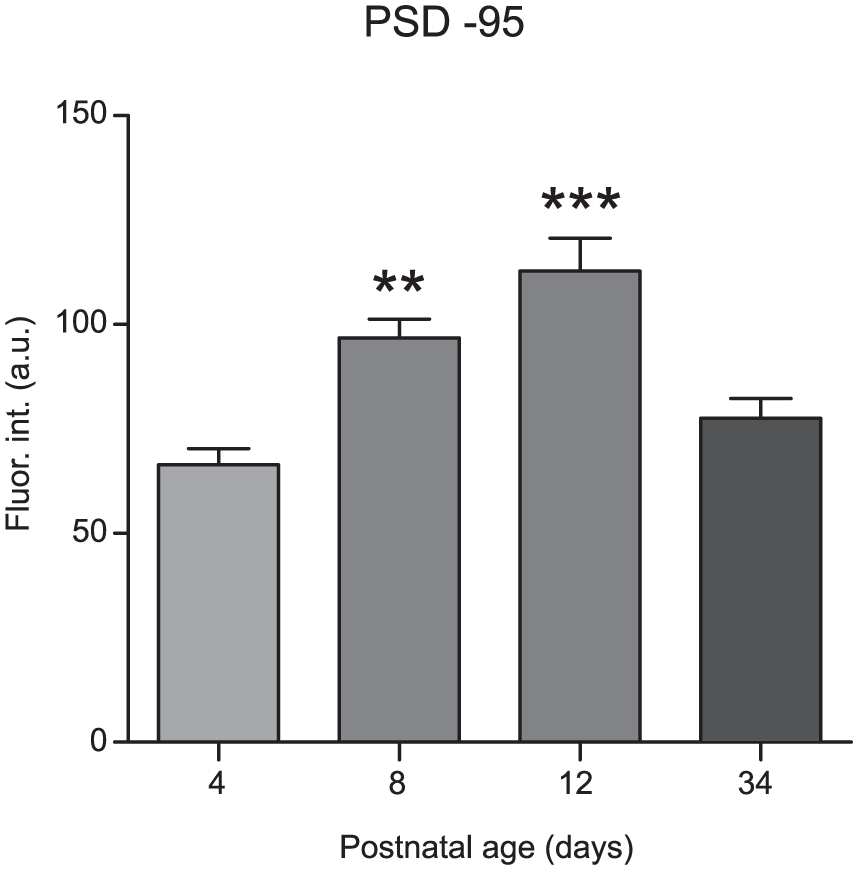
Co-Localization of NR2 Subunits and PSD-95 Protein
PSD-95 functions as a platform for NMDA receptors, and it is therefore expected that NR2 subtypes and PSD-95 are co-localized. We analyzed the degree of this co-localization, and our results revealed high co-localization between NR2 subtypes and PSD-95 (0.6 Pearson’s coefficient on average; Figs. 3-6, merge panels). Particularly, NR2A and PSD-95 showed an increased co-localization index at P8 and P12 (Pearson’s coefficient of 0.57 ± 0.02 and 0.59 ± 0.03, respectively) compared with that at P4 (0.48 ± 0.02) (Fig. 3C). NR2B showed a reduction at P34 (0.45 ± 0.02) compared with P4 (0.6 ± 0.04), whereas NR2C increased at P12 (0.67 ± 0.01) relative to P4 (0.51 ± 0.02). Similarly, co-localization of NR2D and PSD-95 was larger at all studied postnatal ages (0.63 ± 0.01, 0.64 ± 0.01, and 0.61 ± 0.01 at P8, P12, and P34, respectively) compared with P4 (0.54 ± 0.01) (Fig. 6C). These results suggest that NR2 subunits co-localize with PSD-95 in the developing ventral SCN, and the higher co-localization index at specific postnatal ages may imply that NR2 subtypes are recruited into postsynaptic densities.
Electrophysiological Changes of NR2 Subtypes during Development
Because we found developmental changes in mRNA and protein expression of the diverse NR2 subtypes, we hypothesized that these changes have an impact on the physiology of SCN ventral neurons receiving glutamatergic RHT inputs. Consequently, we analyzed the effects of a selective antagonist for the different NR2 subtypes and determined their contributions to the frequency and amplitude of spontaneous NMDA EPSCs recorded in ventral SCN neurons at the 2 developmental ages, P8 and P34. We selected these 2 postnatal ages because we wanted to elucidate if there are changes in the physiology of NMDA receptors between the age where the initial synchronization to light starts (P8) (Duncan et al., 1986) and an older juvenile age (P34). The NR2A subtype had a larger contribution at P8 compared with that at P34, with reductions in EPSC frequencies by PEAQX of 57% ± 11% and 18% ± 10%, respectively (t = 2.6, df = 15, P = 0.01, unpaired t-test) (Fig. 8A). No statistically significant differences in antagonist-induced changes were found in the frequencies of NMDA EPSCs attributable to the NR2B subunit at the 2 analyzed ages. The reductions in EPSC frequencies by Ro 25-6981 were 33% ± 11% and 51% ± 12% at P8 and P34, respectively (t = 1, df = 15, P = 0.3, unpaired t-test) (Fig. 8B). Similar results were found in NMDA EPSC frequencies attainable to the NR2C/D subtype participation because the PPDA-induced reductions in EPSC frequencies were 19% ± 10% at P8 and 34% ± 10% at P34 (t = 1, df = 12, P = 0.1, unpaired t-test; Fig. 8C).
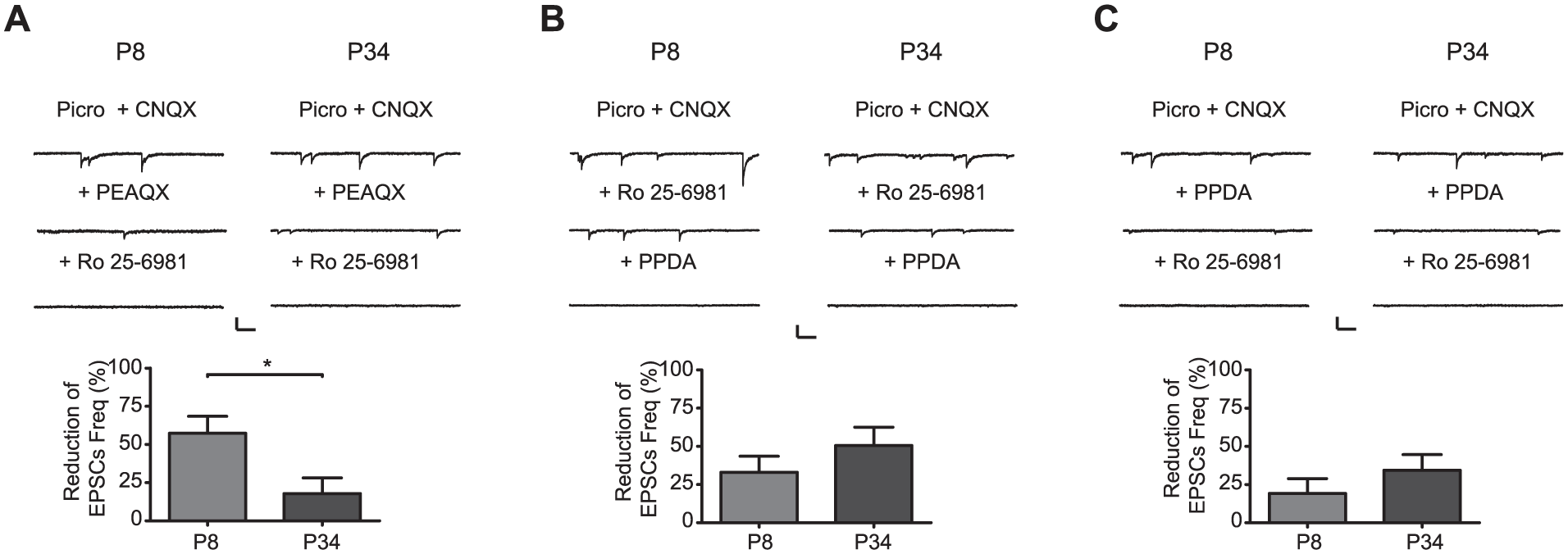
Contribution of the different NR2 subtypes on EPSC frequency (Freq) at 2 postnatal ages (P8 and P34). NMDA EPSCs recorded before (Control = Picro + CNQX) and after application of the NR2A antagonist PEAQX (A), the NR2B antagonist Ro 25-6981 (B), and the NR2C/D antagonist PPDA (C) at the 2 analyzed developmental ages. Scale bars: 40 pA, 200 ms. Bar graphs show the reduction in NMDA EPSC frequency induced by the respective NR2 antagonist. *P < 0.05.
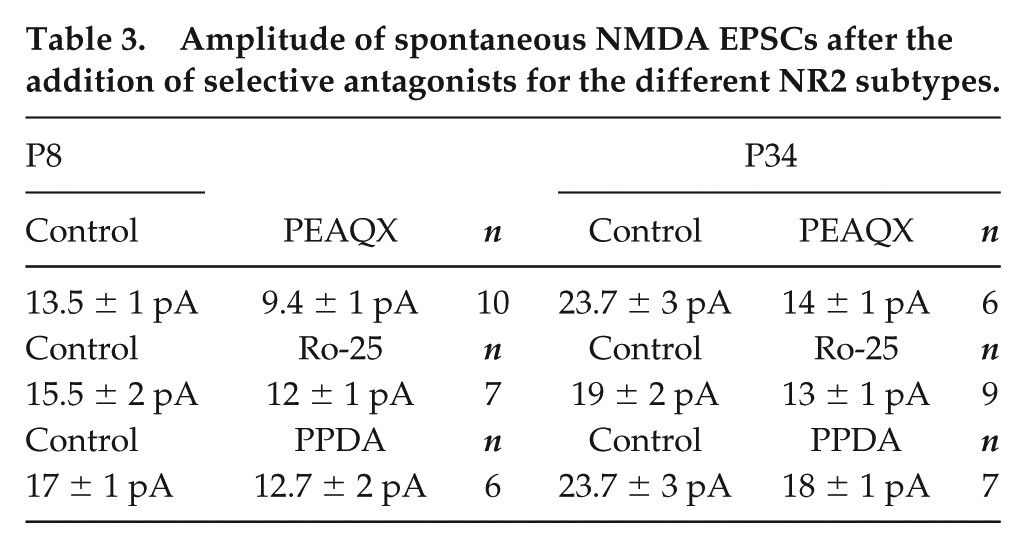
The analysis of the NMDA EPSC amplitudes indicated no developmental changes in NR2 subtype contributions at the examined time points P8 and P34 (Table 3). NR2A showed a similar participation at the 2 analyzed ages (30% ± 6% and 41% ± 14% at P8 and P34, respectively; t = 1.2, P = 0.11, unpaired t-test) (Table 3). The NR2B subtype displayed an age-independent performance (22% ± 7% and 31% ± 7% at P8 and P34, respectively; t = 0.9, P = 0.2, unpaired t-test; Table 3). Comparable results were also obtained for the NR2C/D subtypes (25% ± 11% and 24% ± 7% at P8 and P34, respectively; t = 0.1, P = 0.44, unpaired t-test; Table 3).
Amplitude of spontaneous NMDA EPSCs after the addition of selective antagonists for the different NR2 subtypes.
To investigate the effects of the 2 variables (drug administration and developmental ages) on NMDA EPSCs, a 2-way ANOVA test was carried out to analyze the contribution of NR2 subtypes on the frequency and amplitude of NMDA EPSCs. The interaction test indicated that the participation of NR2 subtypes on the reduction in EPSC frequency was dependent on age (P8 vs. P34; F[2, 90] = 4.6, p = 0.01; data not shown). Regarding the amplitude of EPSCs, the interaction test showed that the contribution of NR2 subtypes did not depend on the developmental age (F[2,90] = 0.43, p = 0.64; data not shown).
Taken together, our results indicate that NR2 subtypes, at least NR2A, exhibit a development-dependent contribution to the EPSC frequency of SCN neurons at P8 and P34.
Discussion
The main result of this study is that we found developmental changes in the expression of the NR2 subtypes of glutamatergic NMDA receptors both in mRNA content and protein expression, which were consistent with the findings in experiments measuring the postsynaptic electrical activity. To our knowledge, this is the first study focused on an investigation of postsynaptic changes in SCN neurons related to light synchronization that occurs at adult stages. Previous studies were aimed at analyzing presynaptic changes by examining the innervations connecting the retina with SCN neurons during postnatal development (Speh and Moore, 1993).
In the current study, we found that the NR2A subtype had a stronger influence on the frequency of NMDA EPSCs at P8, just when the pup starts synchronizing to light via its RHT, whereas the NR2A contribution is decreased afterwards at age P34. Behavioral studies performed in adult animals implicate the NR2A subtype in the synchronization to light, with knockout animals taking longer to synchronize to damped light conditions (Moriya et al., 2000). The NR2B subtype did not show developmental changes in protein expression at all of the examined postnatal ages; although, its mRNA expression progressively decreased with age when normalized to Gapdh. However, it peaked at P8 when normalized to Actb. The latter result is probably more reliable since the levels of this housekeeping gene remain very steady throughout development. The data regarding the expression of the NR2B protein and its co-localization with PSD-95 are consistent with our electrophysiological results. Several lines of evidence indicate that the NR2B subunit is already present at pre- and early postnatal ages (see Cull-Candy et al., 2001). NR2B plays an important role in the synaptic communication of the RHT–SCN pathway at adult stages (Clark and Kofuji, 2010). We believe that the presence of NR2B at all analyzed ages supports the notion of the main influence of this subtype on RHT–SCN synaptic transmission, from early postnatal ages until a young stage. Wang and colleagues (2008) elicited phase-shift reductions in animals by ifenprodil administration but not their complete abolition, which indicates that other NR2 subtypes could participate in the synchronization of SCN neurons to light.
As shown in knockout animals, the NR2C subtype does not seem to be involved in the synchronization to light at adult ages (Moriya et al., 2000). In the present study, we found larger mRNA levels and protein expression of NR2C at juvenile ages; i.e., P34, in which synchronization to light is completed. Similar results (major mRNA content and protein expression) were obtained for the NR2D subtype at the same age. The electrophysiological experiments measuring the EPSC frequencies did not show developmental changes between P8 and P34 according to the effects of an NR2C/D antagonist, which is in agreement with the immunohistochemical experiments because the protein expression of the NR2D subtype was increased at P8 and afterwards. To date, there are no specific antagonists for NR2C and NR2D subtypes, and thus there are no functional studies focused on the contribution of the NR2D subtype to the light synchronization of SCN neurons. Although there are reports indicating that NR2D is not present at the synapses (in a diheteromeric form) (see Cull-Candy, 2001), our co-localization analyses of NR2D and PSD-95 suggest that this subtype is present in the postsynaptic membrane. Clark and Kofuji (2010) proposed that, in SCN neurons, there is a subpopulation of triheteromeric NMDA receptors formed by NR2A and NR2C/D proteins, because the NMDA current remaining after application of the NR2B antagonist ifenprodil shows pharmacological properties of the NR2A subtype with a Mg2+ sensitivity more similar to that of the NR2C/D than the NR2A subtype.
Even though the developmental expression of NR2 subtypes in the brain indicates that NR2B/C and D are mainly expressed at early postnatal ages (P0-P12) while NR2A predominates at later developmental stages (Monyer et al., 1994), our results indicate that the NR2 subtypes of the NMDA receptors in ventral SCN neurons do not follow this pattern. In fact, the functional performance of NR2A showed a reduction at later postnatal ages, NR2B did not present any developmental modification regarding electrophysiology and protein expression, and NR2C and D expressions were not reduced but rather increased at more advanced developmental stages compared with the earlier postnatal age, P4.
PSD-95 has been described as a scaffolding protein for NMDA receptors, localized specifically at the postsynaptic membrane of glutamatergic synapses (Hunt et al., 1996). The increased number of synapses could explain the increase in PSD-95 expression that we found at P8 and P12. Moore and Bernstein (1989) have shown an increase in synaptogenesis between P6 and P10, which is in accordance with our results. Additionally, our analysis of the PSD-95 co-localization with the NR2 subtypes resulted in values higher than 0.5, suggesting that at least half of the immunofluorescence signal is found in the postsynaptic membrane, whereas the remaining fluorescence could be due to NR2 subtypes present in the presynaptic membrane or other cellular structures, such as glial cells (Bouvier et al., 2015; Steinhäuser and Gallo, 1996).
Moreover, our results point to P8 as the developmental stage where NMDA receptors, specifically the NR2 subunit, have reached an adequate level of maturation to allow SCN neuron synchronization to light, which is consistent with a pioneering study that shows that around P7 the rat pup is able to synchronize to the LD cycle without maternal influence (Duncan et al., 1986).
In conclusion, our results indicate that ventral SCN neurons present a development-dependent change in the mRNA content, protein expression, and postsynaptic electrical activity of NR2 subtypes of glutamatergic NMDA receptors that may be important for the synchronization to light at adult stages.
Supplemental Material
Sup_Fig_1 – Supplemental material for Development-Dependent Changes in the NR2 Subtype of the N-Methyl-D-Aspartate Receptor in the Suprachiasmatic Nucleus of the Rat
Supplemental material, Sup_Fig_1 for Development-Dependent Changes in the NR2 Subtype of the N-Methyl-D-Aspartate Receptor in the Suprachiasmatic Nucleus of the Rat by J. Manuel Herrera-Zamora, Luis A. Castro-Sánchez, Miriam Reyes-Mendez, Irving Aguilar-Martinez, Fernando Osuna-López, Eloy G. Moreno-Galindo, Ricardo A. Navarro-Polanco, Raul A. Aguilar-Roblero, Enrique Sánchez-Pastor and Javier Alamilla in Journal of Biological Rhythms
Supplemental Material
Sup_Fig_2 – Supplemental material for Development-Dependent Changes in the NR2 Subtype of the N-Methyl-D-Aspartate Receptor in the Suprachiasmatic Nucleus of the Rat
Supplemental material, Sup_Fig_2 for Development-Dependent Changes in the NR2 Subtype of the N-Methyl-D-Aspartate Receptor in the Suprachiasmatic Nucleus of the Rat by J. Manuel Herrera-Zamora, Luis A. Castro-Sánchez, Miriam Reyes-Mendez, Irving Aguilar-Martinez, Fernando Osuna-López, Eloy G. Moreno-Galindo, Ricardo A. Navarro-Polanco, Raul A. Aguilar-Roblero, Enrique Sánchez-Pastor and Javier Alamilla in Journal of Biological Rhythms
Supplemental Material
Sup_Fig_3 – Supplemental material for Development-Dependent Changes in the NR2 Subtype of the N-Methyl-D-Aspartate Receptor in the Suprachiasmatic Nucleus of the Rat
Supplemental material, Sup_Fig_3 for Development-Dependent Changes in the NR2 Subtype of the N-Methyl-D-Aspartate Receptor in the Suprachiasmatic Nucleus of the Rat by J. Manuel Herrera-Zamora, Luis A. Castro-Sánchez, Miriam Reyes-Mendez, Irving Aguilar-Martinez, Fernando Osuna-López, Eloy G. Moreno-Galindo, Ricardo A. Navarro-Polanco, Raul A. Aguilar-Roblero, Enrique Sánchez-Pastor and Javier Alamilla in Journal of Biological Rhythms
Footnotes
Acknowledgements
This work was supported by Consejo Nacional de Ciencia y Tecnología (CONACYT): CB-2015-01- 254887 to JA.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Note
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
