Abstract
The timing of the preovulatory surge of luteinizing hormone (LH), which occurs on the evening of proestrus in female mice, is determined by the circadian system. The identity of cells that control the phase of the LH surge is unclear: evidence supports a role of arginine vasopressin (AVP) cells of the suprachiasmatic nucleus (SCN), but it is not known whether vasopressinergic neurons are necessary or sufficient to account for circadian control of ovulation. Among other cell types, evidence also suggests important roles of circadian function of kisspeptin cells of the anteroventral periventricular nucleus (AvPV) and gonadotropin-releasing hormone (GnRH) neurons of the preoptic area (POA), whose discharge is immediately responsible for the discharge of LH from the anterior pituitary. The present studies used an ovariectomized, estradiol-treated preparation to determine critical cell types whose clock function is critical to the timing of LH secretion. As expected, the LH surge occurred at or shortly after ZT12 in control mice. In further confirmation of circadian control, the surge was advanced by 2 h in tau mutant animals. The timing of the surge was altered to varying degrees by conditional deletion of Bmal1 in AVPCre, KissCreBAC, and GnRHCreBAC mice. Excision of the mutant Cnsk1e (tau) allele in AVP neurons resulted in a reversion of the surge to the ZT12. Conditional deletion of Bmal1 in Kiss1 or GnRH neurons had no noticeable effect on locomotor rhythms, but targeting of AVP neurons produced variable effects on circadian period that did not always correspond to changes in the phase of LH secretion. The results indicate that circadian function in multiple cell types is necessary for proper timing of the LH surge.
The timing of the transition of the effects of estradiol (E2) from negative to positive feedback on the afternoon of proestrus critically determines the initiation of ovulation. Perhaps no finding was more important in founding the field of neuroendocrinology than the discovery by Everett and Sawyer (1950) that barbiturates, if administered to rats during a critical period on the day of proestrus, delay ovulation by 24 h. Not only did this implicate neural (specifically GABAergic) control, but it also implied an essential role of a light-regulated clock. Over the intervening decades, the circadian basis of the preovulatory luteinizing hormone (LH) surge has been amply demonstrated. Ovariectomized, estrogen-implanted (OVX+E) rats generate repeated daily LH surges (Legan and Karsch, 1975). Barbiturates delay LH surges by 24 h in this preparation (again, only if administered during a critical period in the early afternoon). Electrolytic lesions of the suprachiasmatic nucleus (SCN) of the hypothalamus (the site of a master circadian pacemaker) arrest the estrous cycle and LH surges (Wiegand and Terasawa, 1982), as do genetic manipulations that compromise the circadian clock (Miller et al., 2004; Chu et al., 2013). Splitting of circadian rhythms, induced by exposure to constant light, causes OVX+E hamsters to generate 2 surges per day (Swann and Turek, 1985), each of which is phase locked to activation of the ipsilateral SCN (de la Iglesia et al., 2003), and gonadotropin-releasing hormone (GnRH) cells receive appositions from SCN neurons in this species (de la Iglesia et al., 1995). The loci of circadian control remain uncertain, however, with evidence to support critical roles of clocks in several cell types (reviewed by Christian and Moenter, 2010; de la Iglesia and Schwartz 2006; Williams and Kriegsfeld, 2012; Tonsfeldt and Chappel, 2012; Simmoneaux and Bahougne, 2015). Within the SCN, vasopressinergic neurons may play a critical role in determining the timing of the LH surge (Palm et al., 1999, 2001; Kalamatianos et al., 2004; Miller et al., 2006; Smarr et al., 2012). These cells project to a kisspeptinergic population in the anteroventral periventricular nucleus (AvPV) that plays a critical role in estradiol positive feedback (Smith et al., 2006; Clarkson et al., 2008; Vida et al., 2010; Ronnekleiv et al., 2014; Yip et al. 2015). Nevertheless, it is not clear that vasopressin neurons are necessary or sufficient to ensure circadian control of the LH surge, and other SCN cell types may also make critical contributions. In particular, VIP neurons regulate LH secretion, project directly to GnRH cells, and make preferential synaptic connections with those that are activated at the time of the surge (Vijayan et al., 1979; Samson et al., 1981; Alexander et al., 1985; Kimura et al., 1987; Van der Beek et al., 1994, 1997, 1999; Horvath et al., 1998; Smith et al., 2000; Kriegsfeld et al., 2002; Christian and Moenter, 2008; Piet et al., 2016). Efferent projections of prokineticin 2 neurons may also play an important role (Xiao et al., 2014).
Clock function in cells outside the SCN may also be necessary to ensure proper timing of the LH surge. The interlocked transcriptional-translational feedback loops (TTFLs) that comprise principal components of cell-autonomous circadian clocks are expressed widely in the brain and periphery. This raises the possibility that circadian function not only of cells of the SCN but also of neurons elsewhere in the ovulatory circuit may have an important role in the generation of the LH surge. Evidence has been gathered for control of the timing of ovulation by circadian rhythms in kisspeptin cells of the AvPV (Xu et al., 2011; Smarr et al., 2013; Chassard et al., 2015) and in the GnRH cells themselves (Gillespie et al., 2003; Chao and Kriegsfeld, 2009; Hickok and Tishkau, 2010; Tonsfeldt et al., 2011; Williams et al., 2011).
Conditional knockout of clock genes in specific neuronal types has proven to be a powerful tool in the examination of circadian function (Storch et al., 2007; Husse et al., 2011; Smyllie et al., 2016; Van der Vinne et al., 2018; Weaver et al., 2018). The present experiments targeted Bmal1, a critical constituent of the TTFL, to test the importance for estrogen-induced LH surges of circadian function in arginine vasopressin (AVP), kisspeptin, or GnRH cells. In addition, the effects of altering circadian period (τ) selectively within AVP neurons were examined. The results indicate that circadian rhythmicity of AVP neurons is of particular, but not exclusive, importance in determining the timing of the LH surge. The operation of circadian clocks in kisspeptin and GnRH neurons is also critical. The findings suggest the importance of circadian resonance: the periods of clocks in these cell types must correspond for a normal LH surge to be generated.
Methods
Mice were maintained in 12L:12D unless noted otherwise and had ad libitum access to Purina chow (#5058) and water throughout these experiments. All procedures were approved by the Institutional Animal Care and Use Committee of the University of Massachusetts at Amherst.
Mouse Lines
Several Cre lines were used in these studies. AVP-IRES-Cre mice were obtained from Dr. Brad Lowell (Beth Israel Deaconess Medical Center, Boston, MA; Krashes et al., 2014). Kiss1CreBac mice were kindly provided by Drs. Anne Langjuin and Catherine Dulac (Harvard University, Boston, MA). The Cre cDNA sequences including a bGH polyA tail were recombined after the Kiss1 translational start ATG on BAC RP23-240P23 (Wang, 2012). GnRHCreBac mice, also known as Lhrh strain, were also provided by Dr. Dulac. These mice are available as Tg(Gnrh1-cre)1Dlc/J (Jackson lab stock No. 021207), and bear 1.8 kb of genomic DNA centered around the start codon of GnRH, introduced by homologous recombination into BAC RP23-22J8 (Yoon et al., 2005; Hoffmann et al., 2019). Both Kiss1CreBAC and GnRHCreBAC mice were on a C57BL/CBA mixed background.
Founder Bmal1fl/fl mice were obtained from Jackson Laboratories (stock No. 007668, B6.129S4[Cg]-Arntltm1Weit/J). Circadian rhythms are eliminated when this conditional allele is excised through the action of Cre recombinase (Storch et al., 2007). CnSK1etau mice, in which loxP sites flank mutant exon 4, were kindly provided by Dr. Andrew Loudon (University of Manchester, Manchester, UK). These mice are available from Jackson laboratories as B6.129-Csnk1etm1Asil/J. They carry a mutation of Csnk1e, which codes for a gain-of-function isoform of casein kinase. This allele becomes nonfunctional upon action of Cre recombinase (Loudon et al., 2007; Meng et al., 2008), causing reversion of the circadian period from approximately 20 h or 22 h (in homozygotes or heterozygotes carrying the floxed allele, respectively) to about 24 h. Combinations of mutant alleles were achieved by crossing mice of appropriate genotypes and verified by polymerase chain reaction (PCR). Primers for each transgene and cycle parameters are described in the supplementary methods.
Assessment of Locomotor Activity Rhythms
Mice were placed in individual cages in a light-tight cabinet. Each cage contained a running wheel (12-cm diameter) that tripped a magnetic read switch on each revolution. Activity was recorded and analyzed using a Clocklab Actimetrics package. Mice were maintained in 12L:12D for at least 7 days to assess the entrained phase angle and in DD for 10 days to assess the circadian period by fitting a linear regression line to activity onsets.
Assessment of Reproductive Function
Estrous cycles were assessed by vaginal smears taken during the light phase. After at least 12 days, animals were ovariectomized under isoflurane anesthesia. Buprenorphine (0.05 mg/kg) was administered at the time of surgery and used for up to 48 h thereafter as postoperative analgesia. To determine the capacity of each mouse to generate an LH surge, we adopted the 2-step injection procedure described by Ronnekleiv and colleagues (Ronnekleiv et al., 2014; Qiu et al., 2016). At about ZT4 on the fifth day following ovariectomy, females received a priming dose of estradiol benzoate (EB; 0.25 µg in 50 µL sesame oil, subcutaneously). The next day, a surge-inducing dose of EB (1.0 µg/50 µL sesame oil) was given at ZT4. On the following day, mice were restrained at 90-min intervals, and blood samples (1-2 µL) were taken from the tail vein using a GentleSharp probe (Actuated Medical, Bellefonte, PA) beginning at ZT9 and continuing until ZT15.
LH concentrations were determined using the method of Steyn et al. (2013). Whole blood was diluted (1 µL/50 µL assay buffer). The enzyme-linked immunosorbent assay used a primary monoclonal anti-LHβ (518B7, 1:1000, Janet Rosner, University of California, Davis), a polyclonal rabbit anti-LH (AFP240580, from the National Hormone and Pituitary Program), and a horseradish peroxidase–conjugated goat anti-rabbit (Agilent P044801-2). Mouse LH (AFP-5306A, NIDDK-NHPP) was used as standard. Intra- and interassay coefficients of variation were 9.6% and 5.8%, respectively.
Statistical Analysis
The period and phase of locomotor rhythms were determined by linear regression of activity onsets as previously described (Bittman, 2012). To assess the incidence of LH surges, Chi-squared tests were performed to determine the proportion of wild-type and of each conditional knockout strain showing elevated LH (>5 ng/mL) at each of the 6 sampling times. In addition, LH values of wild-type and Bmal1fl/fl mice carrying AVPCre, KissCreBAC, or GnRHCreBAC were used to assess genotype effects by 2-way analysis of variance (ANOVA), with repeated measures on the ZT factor. Time and strain were main effects. Effects of the tau mutation and its reversal in AVP cells were examined in a separate repeated-measures 2-way ANOVA. The Mann-Whitney test was used to assess differences between LH concentrations of groups of mice at ZT12.
Results
Behavioral Rhythms
Wild-type mice entrained to 12L:12D with activity onset at approximately the time of lights-off. When released into DD, these animals free ran with the expected period (τDD = 23.77 ± 0.10 h; Fig. 1). In contrast, conditional knockout of Bmal1 in AVP cells lengthened the free-running period, as previously reported by Mieda et al. (2015, 2016). Neither entrainment nor τDD was markedly affected by conditional knockout of Bmal1 in GnRH or Kiss cells (Suppl. Fig. S1).
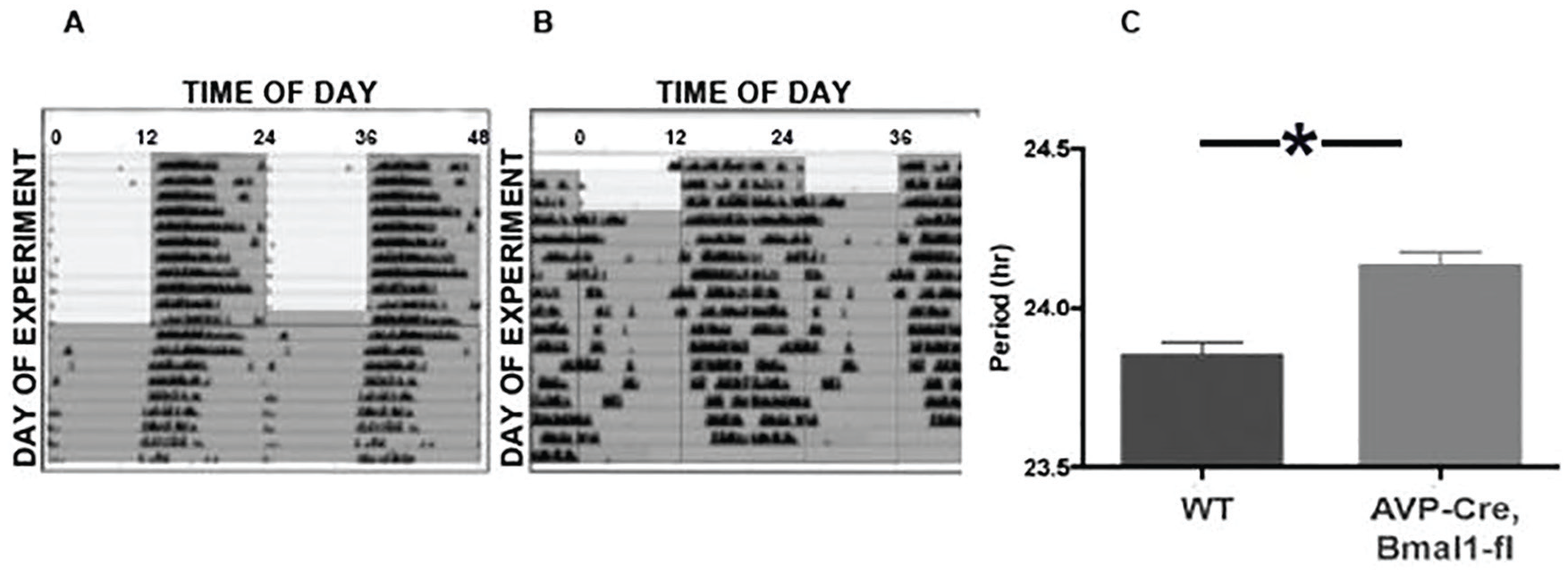
A, representative double plotted actogram of a wild type mouse that was initially maintained in 12L:12D (gray shading indicates lights off) before release into constant darkness (DD). B., Actogram of a representative AVPCre,Bmal1fl/fl mouse under the same conditions. C, mean (+SEM) free running period (τDD) of 14 mice studied in this experiment (* indicates p < 0.05).
Additional experiments used tau mutant mice. As previously reported (Meng et al., 2008), mice homozygous for the floxed Cnske1 (tau) allele in the absence of Cre showed a τDD of approximately 20 h. These mice were crossed with Cre animals to induce selective reversion to a wild-type circadian period in particular cell types. Most AVPCre, Cnsk1etau mice became active at about the time of lights-off, although the phase of locomotor onset upon release in DD indicated negative masking (Fig. 2). After transfer to DD, free-running patterns of activity varied: some showed no evidence of a change in τDD, while others experienced a partial lengthening of the free-running period, although none achieved the period of the wild-type. Still other AVPCre, Cnsk1etau mice showed unstable or disrupted locomotor activity (Fig. 2F,G). There was no apparent correlation between the locomotor pattern and homozygosity of the Cre allele.

Actograms of AVPCre/+, Cnsk1etau/tau mice that were maintained in 12L:12D (gray shading indicates dark phase) and then released into DD. In some cases (A-C), free running rhythms were similar to mice not bearing a Cre allele (τDD approximately 20h). (D, E), Other AVPCre mice showed a partial reversion to a longer period, although none of these mice free ran with as long a period as WT mice. Several mice (F, G) showed nocturnal activity in 12L:12D but unstable patterns of running wheel activity in DD.
Estrous Cycles and the LH Surge
Before ovariectomy, control mice showed regular 4-day estrous cycles. Estrous cycles were irregular in AVPCre, Bmal1fl/fl and GnRHCreBAC, Bmalfl/fl animals as the time spent in estrus was extended (Suppl. Fig. S2). KissCreBac, Bmal1fl/fl mice showed regular estrous cycles (Suppl. Fig. S3).
After ovariectomy, control mice showed a clear surge of LH either at dusk (ZT12, n = 6) or shortly thereafter (ZT13.5, n = 2) at the end of the light phase (Fig. 3A). In all but 1 of these mice, basal LH levels observed at ZT10 were at or below 5 ng/mL, indicating that the estrogen injection regimen effectively activated the negative feedback response in ovariectomized animals. There were no consistent differences between the LH patterns of wild-type females, Bmal1fl/fl and Bmalfl/+ animals lacking Cre, or Cre animals lacking a floxed allele.
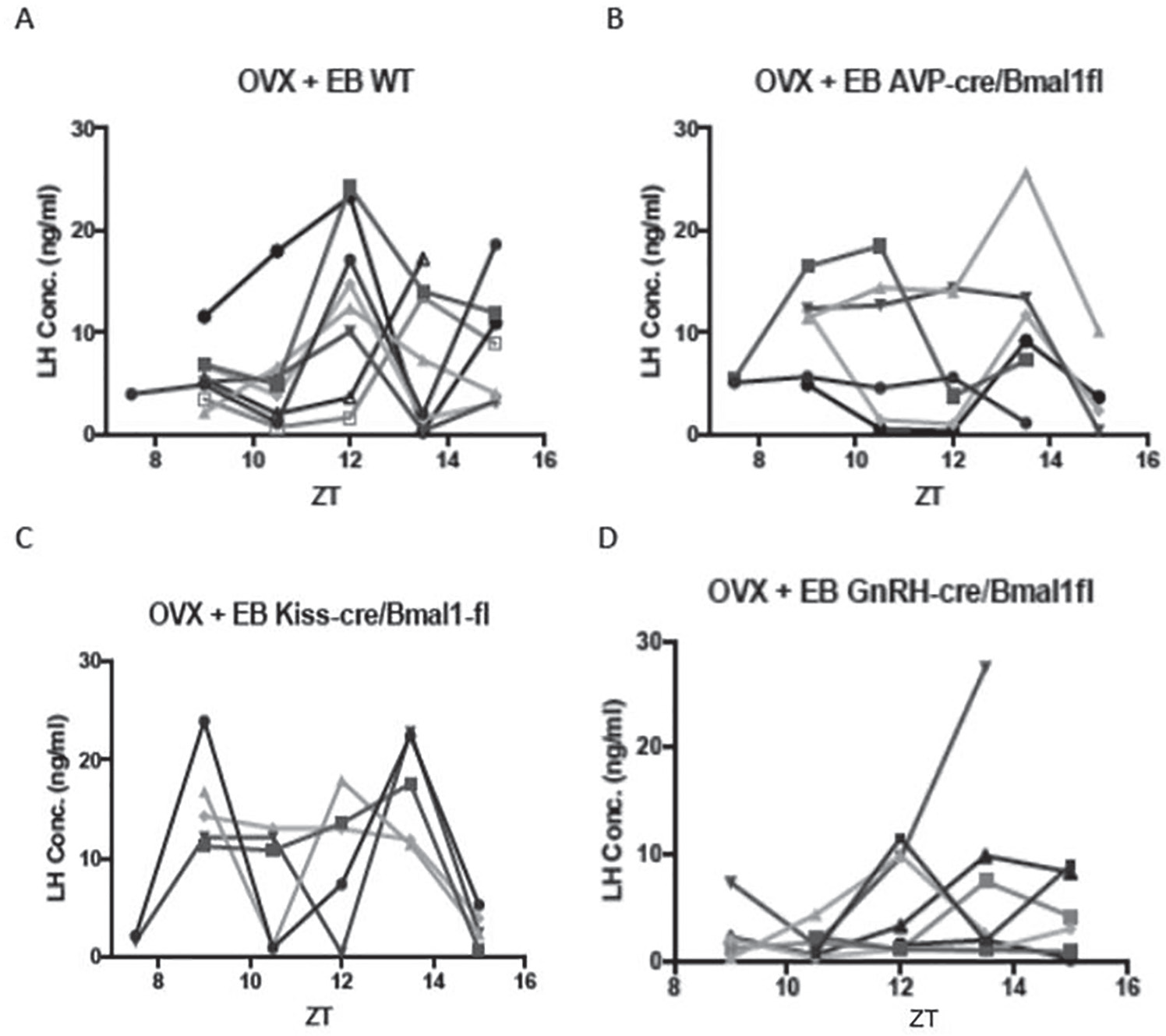
A, LH concentrations (ng/ml) in individual ovariectomized control mice given priming followed by surge-inducing doses of estradiol benzoate. B, C, D., LH values in similarly treated AVPCre/+,Bmal1fl/fl, KissCreBAC,Bmalfl/fl, and GnRHCreBAC,Bmalfl/fl mice, respectively.
Both genetic construct and ZT had significant effects on LH concentration (F = 4.07, p = 0.02 and F = 3.0, p = 0.03, respectively; Suppl. Fig. S4). The interaction between group and ZT did not reach significance (F = 1.99, p = 0.056), but LH concentration and the incidence of LH surges at ZT12 in each of the Cre groups was significantly altered from the wild-type controls (p < 0.05, Mann-Whitney test, and p < 0.001, χ2 test, respectively). None of the 6 AVPCre/+, Bmal1fl/fl mice showed an LH surge at ZT12 (Fig. 3B). Two mice in this group showed a low-amplitude rise at ZT13.5, and LH peaked at this point in 1 animal but from a high baseline. This was 1 of 3 cases in which basal LH was elevated at ZT9, indicating a defect in negative feedback.
Conditional deletion of Bmal1 in Kiss cells also altered the pattern of LH secretion (Fig. 3C). Several of these mice showed a bimodal pattern of LH concentrations, and only 1 showed a surge at ZT12. This animal, and 2 others that experienced a peak at ZT13.5, had high LH levels at ZT9 as well, suggesting an abnormality in negative feedback that affected baseline concentrations.
GnRHCreBAC, Bmal1fl/fl mice showed the most consistent disruption of the LH surge of all experimental groups (Fig. 3D). LH surges failed to occur at ZT12 in most cases, and elevations of LH observed at lights-off or at ZT13.5 were of lower amplitude than in controls.
To assess further the role of circadian function of AVP cells in determining the phase of the LH surge, Cnsk1etau females were compared with controls that lacked the Cnsk1etau allele and with AVPCre, Cnsk1etau animals (Fig. 4). Two-way ANOVA with repeated measures showed significant effects of genotype (F = 4.48, p = 0.03) and ZT (p = 0.001) and an interaction between these main factors (F = 26, p = 0.03; Suppl. Fig. S5). LH surges in Cnsk1etau mice tended to be advanced relative to lights-off, with 3 animals showing a clear surge at ZT10.5. This pattern was altered in Cnsk1etau mice bearing the AVPCre/+ allele. Three such animals experienced a reversion of the timing of the surge to the wild-type phase, with a peak at ZT12. Three others showed no discrete surge; 2 of these animals had continuously elevated LH concentrations between ZT9 and 13.5, suggestive of a disruption of negative feedback. Overall, LH concentrations at ZT12 did not differ between AVPCre, Cnsk1etau mice and controls that lacked the tau allele (Mann-Whitney test, UA = 27, p > 0.05). We did not observe any consistent correlation between the reversion of locomotor rhythms (Fig. 2) and the pattern of LH secretion (Suppl. Fig. S6). A small experiment involving 4 KissCreBac, Cnsk1etau mice indicated an effect of deleting the tau allele in kisspeptin cells as well, but this manipulation produced high baseline LH levels that complicated the assessment of the timing of the LH surge (Fig. 4C).
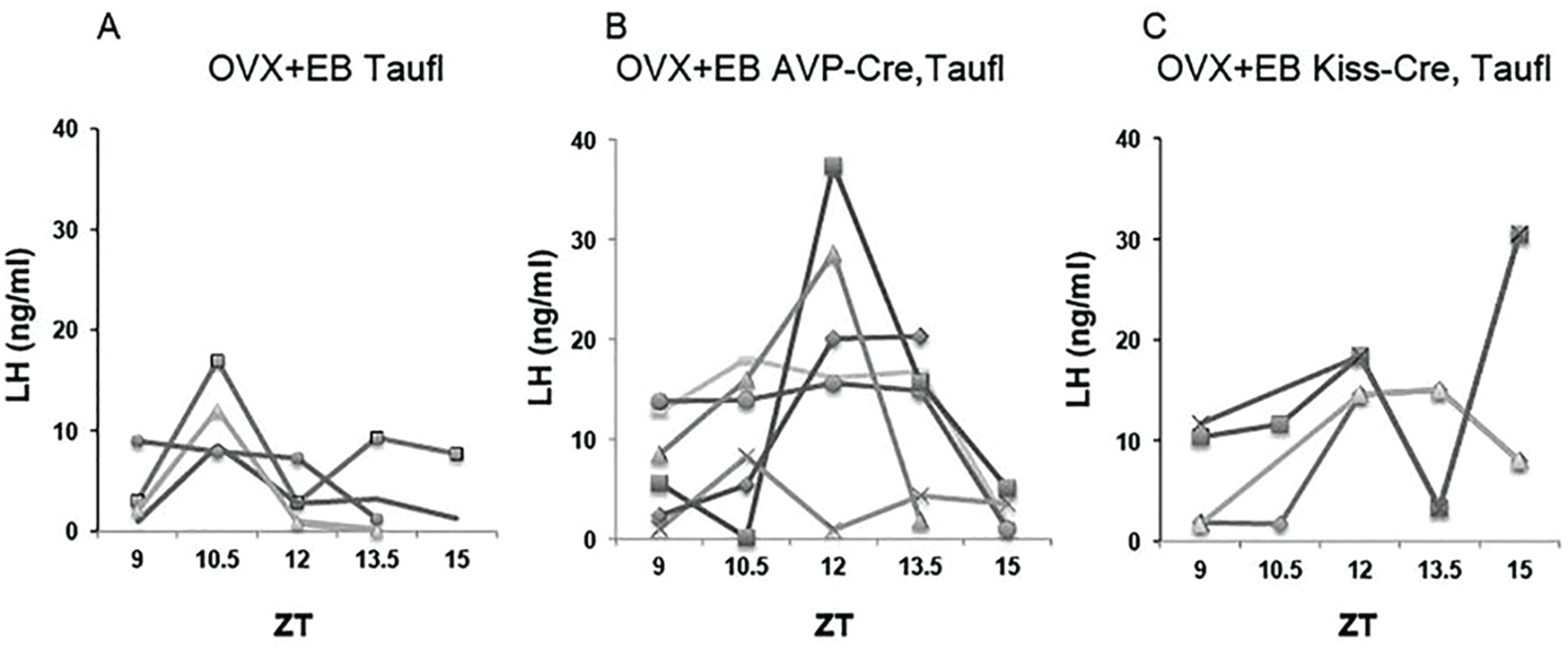
A, Circulating LH (ng/ml) in individual CnSK1etau ovariectomized control mice given priming followed by surge-inducing doses of estradiol benzoate. B, LH concentrations in identically treated AVPCre/+,Cnsk1etau/tau mice. C. LH concentrations in identically treated KissCreBAC,CnSK1etau/tau mice.
Discussion
The LH surge is disrupted by conditional knockout of Bmal1 not only in vasopressin cells, which may provide a critical output of the pacemaker, but also in Kiss or GnRH cells. Thus, proper timing of the LH surge requires operation of circadian clocks at 3 or more points in the circuit, including cells that are direct or indirect targets of SCN efferents. The importance of circadian function in Kiss and GnRH cells may be explained by their function as subordinate oscillators, which need SCN input to maintain phase coherence (Xu et al., 2011; Smarr et al., 2013; Chassard et al., 2015; Hickok and Tishkau, 2010; Williams et al., 2011). The traditional view is that SCN signals are critical to the generation of the LH surge because they acutely activate such targets. The present results support the alternative interpretation that the LH surge depends on the function of the circadian pacemaker to entrain them. Significantly, the effect of the conditional deletion of Bmal1 in any one of these cell types was not to eliminate the surge but to alter its timing. This may reflect loss of phase coherence within Kiss and/or GnRH cell populations. Such desynchronization may progress gradually in uncoupled neuronal populations, as cell-autonomous oscillations may differ only slightly in period so that they drift out of phase only after several cycles when deprived of critical input from vasopressinergic (and perhaps other) cells of the SCN. It is possible that disruption of circadian function in AVP, Kisspeptin, and/or GnRH cells would compromise the LH surge more severely in mice kept in constant conditions; the light:dark cycle to which mice were exposed in the present experiments may have provided a masking influence. The abrogation of LH surges upon destruction of the SCN (Wiegand and Terasawa, 1982) or global knockout of Bmal1 (Chu et al., 2013) may indicate redundancy in this multioscillatory system, in that ablation of the pacemaker or cessation of clock function in all cell types has more devastating effects than interruption of circadian rhythms of any single component of the circuit.
The finding that Bmal1 expression in AVP cells is necessary for a normally timed surge is consistent with evidence that vasopressinergic outputs from the dorsal SCN “shell” play an important role in circadian timing of behavioral and endocrine outputs (Kalsbeek et al., 2006; Evans et al., 2015; Mieda et al., 2015, 2016). The role of vasopressin in generation of the LH surge is supported by demonstrations that disruption of estrous cycles in Clock mutant mice is accompanied by reduced AVP expression in SCN and lower AVP1a receptor mRNA in hypothalamus (Miller et al., 2006), and with the finding that injection of AVP into this region can restore LH surges to SCN-lesioned rats (Palm et al., 1999, 2001). The present results indicate that baseline LH is elevated in these conditional knockout animals, suggesting that clock function of vasopressin neurons may also have a role in negative feedback actions of estradiol. Disruption of the tonic mode of LH secretion may contribute to the action of Bmal1 deletion to compromise the surge. It is important to note that these experiments do not exclude a role of vasopressin neurons outside the SCN in control of the LH surge, as the conditional knockout procedure used in these studies is expected to eliminate clock function in all AVP cells, including not only parvicellular neurons elsewhere in the forebrain but also magnocellular neurons in supraoptic and paraventricular nuclei. Evidence that conditional deletion of the mutant tau allele in vasopressin cells normalizes the timing of the LH surge, however, supports the results from the AVPCre/+, Bmal1fl/fl mice and suggests that the relevant site is the circadian pacemaker. This result supports a model in which the output of vasopressin cells is sufficient to entrain cell-autonomous oscillations in kisspeptin, GnRH, and perhaps other populations to determine the timing of the LH surge. This role of AVP in the surge circuit may be specific, as the altered pattern of the LH surge in AVPCre/+, Bmal1fl/fl mice could not be explained by a change in the entrained phase angle of locomotor activity rhythms. It will be necessary to restrict Bmal1 knockout, and/or conditional deletion of the tau allele, to vasopressin cells of the SCN to establish unequivocally the role of this particular population in surge generation. This may be achieved through strategies in which Bmal1 expression is compromised only in specific cell types of the circadian pacemaker (Atasoy et al., 2012; Tso et al., 2017).
Conditional knockout of Bmal1 in Kisspeptin neurons also altered the timing and amplitude of the LH surge. The results suggest that circadian clock function in Kiss neurons plays an important role in the regulation of LH secretion, which may extend to both negative and positive feedback actions of E2. As is the case with the AVPCre/+, Bmal1fl/fl mice, it is possible that disruption of clock function in Kiss neurons in multiple areas contributes to changes in the pattern of LH secretion. Although the vasopressinergic projection of the SCN to the AvPV drives the activation of Kiss cells to trigger the surge, the arcuate population of Kiss cells controls the tonic pulsatile mode of LH secretion and steroidal negative feedback (Han et al., 2015; Dubois et al., 2016). In a model of pulsatile stimulation using a luciferase reporter, the activation of GnRH cells by kisspeptin was reduced in Bmal1-deficient mice (Choe et al., 2013). Coordination of relaxation of negative feedback with activation of positive feedback systems may be important to mounting a normal LH surge (Russo et al., 2015). Thus, Kiss neurons in the arcuate nucleus may also participate in circadian regulation of the LH surge and could contribute to the finding that timing of the LH surge is altered in KissCreBAC, Bmalfl/fl mice (Helena et al., 2015; Mittelman-Smith et al., 2016). Again, targeted deletion of Bmal1 in Kiss cells of AvPV versus arcuate will help to determine the important sites of clock function in kisspeptinergic control of the LH surge. Inhibitory effects of the arcuate Kiss population on GnRH secretion likely occur through the release of dynorphin from KnDY cells (Lehman et al., 2010). For both the Kiss and the AVP cells, peptides are co-released with GABA and possibly other small classical neurotransmitters. Manipulation of circadian function in either or both cell types may alter LH secretion through effects on the release of either the peptide, the co-released signal, or both.
The LH surge is most profoundly compromised in GnRHCreBAC, Bmal1fl/fl mice. This is consistent with findings of antiphase Per1 and Bmal1 expression in GnRH cells (Hickok and Tischkau, 2010) and with evidence from hamsters that the efficacy of Kiss administration to the preoptic area (POA) to activate GnRH cells varies with time of day (Williams et al., 2011). As is the case for AVP and Kiss neurons, GnRH cells may be heterogeneous in distribution and function: they are found in a continuum running from the diagonal band to the anterior hypothalamus, may produce different isoforms of this decapeptide, and may not all be activated at the time of the LH surge (Lee et al., 1992; Herbison, 2014). Thus, circadian function in a particular subset of GnRH neurons may be of particular importance to surge generation. In light of noncircadian functions of Bmal1 in development, we cannot exclude the possibility that effects of Bmal1 deficiency in Kiss, GnRH, or perhaps AVP cells reflects disruption of sexual differentiation (Yang et al., 2016). It will be interesting to restrict conditional deletion of Bmal1 in these cell types to adulthood and/or to use chemogenetics or optogenetics to acutely silence AVP or Kiss cells to define a critical period for their depolarization.
Circadian function in cell types other than those examined in this study may also be important in determining the phase of the LH surge. A principal candidate is the VIP population of the SCN (Ohtsuka et al, 1988; van der Beek et al., 1993; Weick and Stobie 1995; Harney et al. 1996; Gerhold et al., 2005; Loh et al. 2014; Piet et al., 2015). These neurons project directly to GnRH cells and are thus positioned to regulate their discharge. Although conditional deletion of Bmal1 in VIP neurons may well affect the surge, it would be unclear whether such an effect arose from a change in their function as an SCN output or because of their role in entrainment to light and coupling of cell autonomous oscillators within the SCN (Aton et al., 2005). In addition to its function as an SCN output, vasopressin likely has intra-SCN functions (Mieda et al., 2015, 2016), and these could also contribute to the changes in the timing of the LH surge in AVPCre, Bmal1fl/fl mice described above. It is possible that direct (VIP) and indirect (AVP-kisspeptin) pathways from the SCN to the GnRH cells complement one another, perhaps having different roles in entraining subordinate oscillators versus triggering their depolarization. SCN projections to the subparaventricular region may also regulate the timing of the LH surge, as vasopressinergic projections to the dorsomedial subparaventricular zone may regulate neurons that in turn project to the medial preoptic area (Vujovic et al., 2015). The phase of LH secretion may also be regulated by circadian function of any of multiple peptidergic cell types including GnIH, hypocretin, melanin concentrating hormone (MCH), cocaine- and amphetamine-regulated transcript (CART), ß-Klotho, β-endorphin, and prokineticin 2 and be influenced by the timing of GABAergic and glutamatergic input to GnRH neurons (Christian and Moenter, 2010).
These experiments were restricted to the interval between ZT9 and ZT15, the time window in which LH surges occur in wild-type mice. It will be necessary to examine LH secretion at other times in order to determine whether LH surges occur in these Cre lines earlier in the day or later in the night. Despite the effects of these genetic constructs on the timing and pattern of the LH surge, all are fertile. Indeed, even mice with a global knockout of Bmal1 experience relatively subtle disruptions of the estrous cycle and ovulate, despite the absence of LH surges (Chu et al., 2013). Likewise, estrous cycles continue in mice in which kisspeptin neurons are eliminated by Kiss-Cre–driven diphtheria toxin (Mayer and Boehm, 2011). This may be attributable to compensation during development, as acute ablation of Kiss neurons in adulthood results in constant estrus. It is also important to recognize that the present experiments were restricted to ovariectomized animals in which the only gonadal steroid available to control LH secretion was estradiol. Circadian function in any or all of the cell types studied here may be less important when preovulatory progesterone is available to influence the timing and amplitude of the LH surge (DePaolo and Barraclough, 1979; Stephens et al., 2015; Leite et al., 2016). Finally, circadian rhythms in the ovary (Mereness et al., 2016), if not the pituitary (Chu et al., 2013), may contribute to the timing of ovulation. Despite this possible redundancy in the neural systems that regulate the LH surge, the present data indicate that circadian control of its timing is distributed among several cell types.
Supplemental Material
Supplementary_Fig_5_7-11-19 – Supplemental material for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge
Supplemental material, Supplementary_Fig_5_7-11-19 for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge by Eric L. Bittman in Journal of Biological Rhythms
Supplemental Material
Supp_Fig_1_7-11-19 – Supplemental material for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge
Supplemental material, Supp_Fig_1_7-11-19 for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge by Eric L. Bittman in Journal of Biological Rhythms
Supplemental Material
Supp_Fig_2 – Supplemental material for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge
Supplemental material, Supp_Fig_2 for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge by Eric L. Bittman in Journal of Biological Rhythms
Supplemental Material
Supp_Fig_3_1 – Supplemental material for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge
Supplemental material, Supp_Fig_3_1 for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge by Eric L. Bittman in Journal of Biological Rhythms
Supplemental Material
SUPP_FIG_4_7-11-19 – Supplemental material for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge
Supplemental material, SUPP_FIG_4_7-11-19 for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge by Eric L. Bittman in Journal of Biological Rhythms
Supplemental Material
Supp_Fig_6 – Supplemental material for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge
Supplemental material, Supp_Fig_6 for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge by Eric L. Bittman in Journal of Biological Rhythms
Supplemental Material
Supp_Table_1 – Supplemental material for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge
Supplemental material, Supp_Table_1 for Circadian Function in Multiple Cell Types Is Necessary for Proper Timing of the Preovulatory LH Surge by Eric L. Bittman in Journal of Biological Rhythms
Footnotes
Acknowledgements
These experiments were performed by Mr. Ajay Kumar. I thank Drs. Catherine Dulac, Michael Hastings, Andrew S. I. Loudon, and Bradley L. Lowell for generous supply of transgenic lines; Amanda Hageman for expert animal care; and Kevin Shen, Emma Sisson, and Yahaira Bermudez for assistance with PCR genotyping. This research was supported by National Institutes of Health grants R21HD078863, R21NS099473, and RO1HL138551 to E.L.B.
Conflict Of Interest Statement
The author has no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
