Abstract
In vertebrates, reproductive output often increases with age. Unlike older birds, first-year photoperiodic birds lack experience with the reproductively stimulatory effects of long day lengths (photostimulation). We examined whether age-related differences in annual reproductive development could be partially attributed to previous experience with photostimulation in the photoperiodic house finch (Carpodacus mexicanus). By manipulating photoperiod, we generated 2 groups of first-year females: a photo-experienced group that underwent 1 photoperiodically induced cycle of gonadal development and regression and a photo-naïve group exposed to long days since hatch. We transferred both groups from long to short days and then photostimulated and exposed them to male birdsong prior to sacrifice. Following concurrent photostimulation, both groups exhibited similar plasma luteinizing hormone surges and hypothalamic vasoactive intestinal polypeptide immunoreactivity. In contrast, hypothalamic gonadotropin-releasing hormone immunoreactivity and circulating vitellogenin levels were higher in photo-experienced birds, and yolk deposition occurred in only 2 females, both of which were photo-experienced. Our results demonstrate that photo-experience enhances some aspects of early photo-induced reproductive development and raise the hypothesis that photo-experience may account for at least some age-related variation in reproductive output.
Keywords
In most iteroparous animals, reproductive output increases with age (Clutton-Brock, 1988). In birds, older females generally initiate egg production earlier, lay more and sometimes larger eggs, and produce more fledglings than first-year females (Saether, 1990; Fowler, 1995). Furthermore, the reproductive output of individual females improves between first and second annual breeding seasons (Hannan and Cooke, 1987; Newton and Rothery, 1998).
For most temperate zone species, the most reliable cue for initiating breeding is the annual cycle of changes in day length (photoperiod) (Wingfield, 1980). Temperate-zone birds hatched in the spring and summer are unresponsive to the reproductively stimulatory effects of long photoperiods (photorefractory), which prevents premature reproductive development (Farner et al., 1983; Nicholls et al., 1988). Exposure to short day lengths during fall and winter dissipates photorefractoriness, resulting in hypothalamic responsiveness to reproductively stimulatory photoperiodic and social cues (photosensitivity). Increasing spring day lengths accelerate reproductive development (photostimulation) (Farner et al., 1983) by stimulating the hypothalamic release of gonadotropin-releasing hormone (GnRH-I), which induces secretion of gonadotropins, luteinizing hormone (LH), and follicle-stimulating hormone, from the anterior pituitary (Sharp and Ciccone, 2005). In females, elevated gonadotropin levels stimulate ovarian development and production of 17β-estradiol (E2) and progesterone (Williams, 1998). Elevated circulating E2 induces hepatic synthesis and secretion of egg yolk precursors, yolk-targeted very-low density lipoprotein (VLDLy) and vitellogenin, which are taken up by developing ovarian follicles (Bergink et al., 1973; Walzem, 1996).
Exposure of photosensitive birds to long day lengths also increases hypothalamic secretion of vasoactive intestinal polypeptide (VIP) (Mauro et al., 1992; Deviche et al., 2000), stimulating the production and release of prolactin from the anterior pituitary (Lea et al., 1991; El Halawani et al., 1997). Increased plasma prolactin is involved in the initiation and maintenance of incubation and parental care (Sockman et al., 2006) and the onset of photorefractoriness, gonadal regression, and postnuptial molt as the breeding season ends (Nicholls et al., 1988; El Halawani et al., 1997; Dawson and Sharp, 1998).
Whereas older birds have previous experience with photostimulation (photo-experience) and at least 1 cycle of photo-induced gonadal development and regression, first-year birds initiating their first breeding attempt are experiencing photostimulation for the first time (photo-naïve). Our goal was to examine whether prior experience with photostimulation affects early reproductive development and responses to reproductive cues, thereby contributing to the age-related differences observed in reproductive output. Using the photoperiodic female house finch (Carpodacus mexicanus), we investigated whether previous photo-experience, rather than age, per se, influences the integration of stimulatory environmental cues into the neuroendocrine signaling pathways that regulate early, photo-induced reproductive development. We designed the experiment based on a similar study in which female European starlings (Sturnus vulgaris) (Sockman et al., 2004) were initially exposed to a short day length (8 h L: 16 h D) for 12 weeks to ensure photosensitivity. The photo-naïve group was maintained on short days for 20 additional weeks while the photo-experienced group was transferred to long days for 12 weeks and then back to short days for 8 weeks to induce photosensitivity for the second time. Both groups were then photostimulated. However, the prolonged photosensitivity of the photo-naïve group may have (1) desensitized the birds’ pituitaries to GnRH, resulting in a dampened LH response to photostimulation compared with the photo-experienced group, and (2) increased hepatic storage of vitellogenin, resulting in the more rapid increase in circulating vitellogenin levels in photo-naïve females following photostimulation (Sockman et al., 2004). We designed the present study to avoid these potential problems by maintaining the photo-naïve group in a reproductively quiescent, photorefractory state from hatch by exposure to long days. After exposure to short days to induce photosensitivity, the first photoperiodic response of the photo-naive group was, therefore, more physiologically comparable to the photo-experienced group than in the earlier starling study. As conspecific song and mate availability are “supplementary” cues that female songbirds use to fine-tune the timing of early reproductive development (Wingfield, 1980), we housed females with males and, during the last day of the study, isolated and exposed the females to novel conspecific male song in an effort to maximize reproductive development, using heterospecific male song as a control.
We predicted that after photostimulation and exposure to conspecific male song, photo-experienced females would have higher circulating LH and vitellogenin levels, larger or more developed reproductive organs, and more immunocytochemically visible hypothalamic GnRH and VIP neurons than photo-naïve birds. If these predictions are correct, they would be consistent with the view that age-related differences in reproductive development are attributable, at least in part, to photo-experience.
Materials and Methods
Animals and Housing
We captured house finches between June and July 2006 in Chapel Hill, North Carolina (35.91°N 79.05°W), and transferred them to outdoor flight cages at the University of North Carolina at Chapel Hill, where we conducted the study. This study was approved by the university’s institutional animal care and use committee (protocol 07-260). For the entire study we provided the birds with food (Daily Maintenance, Roudybush; Woodland, CA) and water ad libitum. We identified hatch-year birds (new fledglings) by the presence of feather tufts on the head and new, unworn wing feathers (Hill, 2002) and, on 24 July 2006, moved them to indoor cages on a photoperiod (16 h L: 8 h D, referred to as long days) that maintained them in a nonreproductive, photorefractory state (Nicholls et al., 1988). Following completion of their annual molt, we identified males and females by the presence or absence, respectively, of yellow plumage on the head and later confirmed post mortem.
Photoperiod Manipulation
On 21 November 2006 (week 0), we randomly assigned and transferred 2 females and 1 male to each of 10 light-proof, foam-lined, sound-attenuation chambers located together in 1 room. Each chamber had a cage with 3 perches, an air intake and fan-driven exhaust, and a fluorescent light that maintained the chamber-specific photoperiod. We changed the photoperiod to 8 h L: 16 h D (short days) in 5 chambers in order to begin the process of instating sensitivity to reproductive stimuli (Fig. 1). On 16 January 2007 (week 8), we changed the photoperiod in these 5 chambers to 16 h L: 8 h D (long days), driving these birds first through a reproductive-like state, and then into a nonreproductive, photorefractory state (Nicholls et al., 1988) (photo-experienced group) (Fig. 1). Throughout this time, we maintained the original 16 h L: 8 h D photoperiod in the other 5 chambers, thereby maintaining the nonreproductive, photorefractory status of these birds (Nicholls et al., 1988) (photo-naïve group) (Fig. 1). We spatially interspersed the replicate chambers of the treatments to control for location effects. We exposed the birds to this long-day photoperiod until all of the birds in the photo-experienced group initiated molt, resulting in the photo-experienced group being exposed to long days for 17 weeks and the photo-naïve group for 25 weeks. On 16 May 2007 (week 25), we moved each triplet group of birds into each of 10 cages located together in 1 room and changed the photoperiod in the room to 8 h L: 16 h D, thereby beginning the process of instating sensitivity to reproductive stimuli for the first time in the photo-naïve group and for the second time in the photo-experienced group (Fig. 1). On 18 July 2007 (week 34), after exposing all birds to the 8 h L: 16 h D photoperiod for 9 weeks, we changed the photoperiod in the room to 16 h L: 8 h D, driving the photo-naïve birds into a reproductive-like state for the first time and the photo-experienced birds for the second time (Fig. 1).
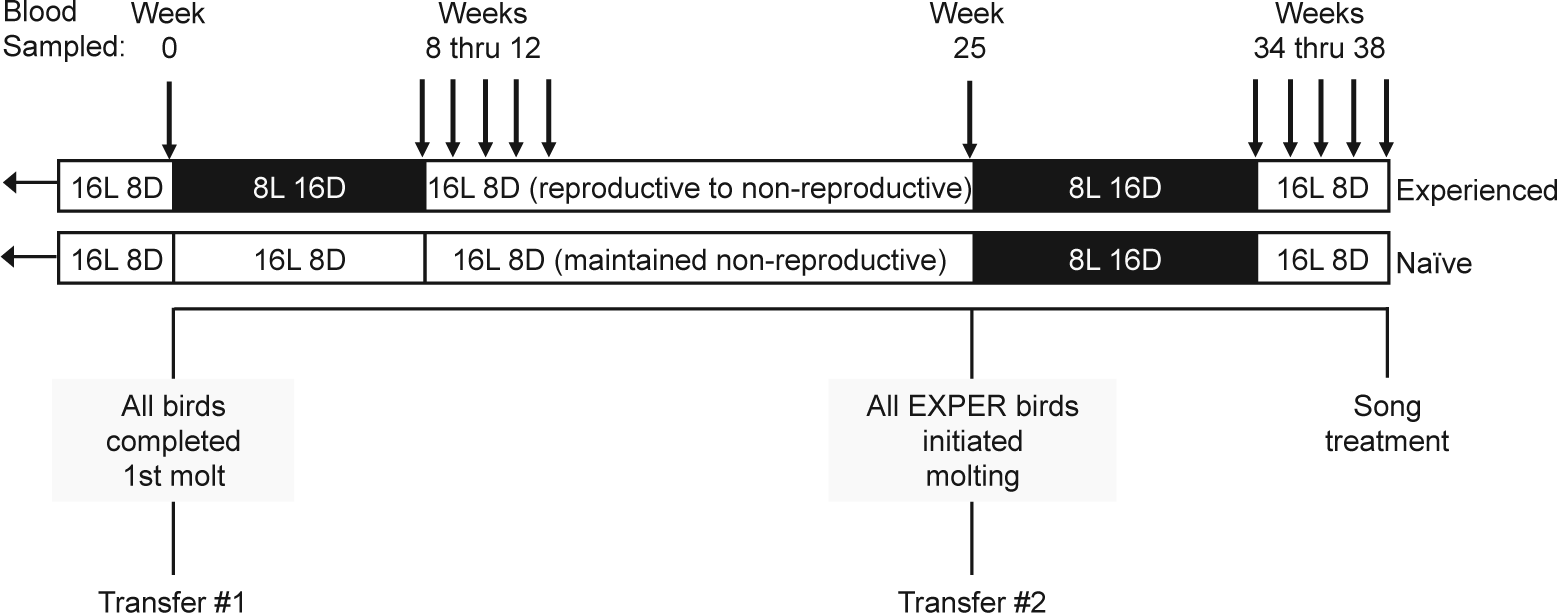
Experimental design. The photoperiod treatment is depicted at the top of the figure as white bands for long days (16 L: 8 D) and black bands for short days (8 L: 16 D). All birds were initially exposed to long days from the time of capture. At the end of week 0 (Transfer 1), we moved each triplet group (2 females, 1 male) into 1 of 10 sound-attenuation chambers in 1 room. From weeks 1 to 8, we exposed the birds in the photo-experienced group (Experienced) to short days, and we maintained the birds in the photo-naïve group (Naïve) on long days. We exposed all birds to long days from weeks 9 through 25. At the end of week 25 (Transfer 2), we moved each triplet group into 1 of 10 cages in 1 room and exposed all birds to short days from weeks 26 to 34 and then finally to long days for the last 4 weeks (weeks 35-38) of the study. Downward arrows indicate the weeks during which we took a blood sample from each bird.
Body Mass Measurements and Blood Sampling
Starting on 21 November 2006 (week 0), we measured the body mass of each bird once every week during the 38-week photoperiod manipulation. We took a blood sample (~150 µL) from each bird at predetermined time points throughout the study (Fig. 1) to measure temporal variation in circulating levels of LH and vitellogenin. We centrifuged the blood samples to separate the plasma, which we stored at −20°C until analysis. Only the data for female house finches are presented here.
Song Exposure, Sacrifice, and Tissue Collection
On the afternoon of 13 August 2007, at week 38 of the study, we weighed 7 females (n = 2 photo-experienced; n = 5 photo-naïve) from 5 cages, moved them individually into each of 7 light-proof, sound attenuation chambers (58 × 41 × 36 cm, Industrial Acoustics Company, New York, NY) located together in 1 room, and isolated the birds for 1 full day. Each chamber had a speaker (Pioneer Corp. TS-G1040R, Tokyo, Japan) and was otherwise equipped similarly to the photo-experience chambers. We powered the speakers by a daisy chain of monoblock amplifiers interfaced with a computer.
Beginning 1 h after the onset of the photophase on 15 August 2007, we exposed 1 bird to a song set recorded from either male house finches or male northern cardinals (Cardinalis cardinalis) (see the next section) for 30 min through the chamber’s speaker (hereafter termed song treatment). We played the song at approximately 80 dB at 5 cm from the speaker to approximate the amplitude of songs that a free-living bird would experience from a nearby male. Each female heard a unique set of songs from a unique set of male singers (i.e., no male’s song was played to more than 1 female). We staggered exposure to the song treatment by 30 min between females.
At 90 min after the onset of the song treatment, we weighed the birds, and after taking a blood sample (~150 µL) from a brachial vein, rapidly decapitated them and removed their brains. Using previously described protocols (Sockman and Salvante, 2008), we halved each brain using a midline sagittal cut, fixed 1 hemisphere (5% acrolein for 4.5 hours; alternating left and right hemispheres from successive birds), and stored the fixed hemispheres at −80°C after cryoprotection in 30% sucrose. We recorded the color and diameter of the 3 largest ovarian follicles from each bird.
We repeated these procedures for the remaining females (n = 4 photo-experienced; n = 4 photo-naïve) after moving them individually into each of 8 light- and sound-proof chambers on the afternoon of 15 August 2007. By balancing the song treatment levels between subjects from the same photoperiod experience group, we generated 4 female treatment groups: (1) photo-experienced, conspecific song (n = 3); (2) photo-experienced, heterospecific song (n = 3); (3) photo-naïve, conspecific song (n = 5); and (4) photo-naïve, heterospecific song (n = 4).
Song Recordings Used for Playbacks
We recorded the songs used for playback from free-living male house finches and Northern cardinals in the area surrounding the UNC–Chapel Hill campus using a short-shotgun microphone (Sennheiser ME-66/K6, Wedemark, Germany), connected to a digital recorder (Marandtz PMD 660, Mahwah, NJ) set to record uncompressed files sampled at 44.1 kHz. We then selected 2 songs from each of 24 male house finches and 24 male cardinals using Raven software (v.1.2.1, Cornell Laboratory of Ornithology). We matched conspecific and heterospecific songs based on individual song duration and created 8 sets of duration-matched house finch and northern cardinal songs composed of 6 songs (2 songs from 3 different males) arranged in a random order such that all 6 songs were repeated the same number of times within the 30 minutes. All song sets were 30 min long and contained a total of 15 min of song and 15 min of silence.
GnRH and VIP Immunocytochemistry and Quantification
We sectioned the fixed brain hemispheres in the sagittal plane at 40 µm on a cryostat and performed immunocytochemistry (ICC) for GnRH on every third section as previously described by Sockman and colleagues (Sockman et al., 2004). As part of another study, we initially labeled the tissue for ZENK immunoreactivity using a different chromogen. We quenched the tissue with 0.5% H2O2 before incubating with a 1:10,000 dilution of GnRH primary antibody (HU60 bleed H, provided by H.F. Urbanski, Division of Neuroscience, Oregon Regional Primate Center, Beaverton, OR). The details of the GnRH antibody, which has been validated for use in house finches (Cho et al., 1998), have been described previously (Urbanski, 1992). The rabbit-raised GnRH antibody recognizes intact, but not fragmented forms of GnRH-I and GnRH-II found in birds (Sharp et al., 1990; Sharp and Ciccone, 2005). We processed all of the tissue in 2 ICC batches. Given the uneven mortality between treatment groups, we counterbalanced the 4 photo-experience-song treatment groups as much as possible within each ICC batch.
We performed ICC for VIP on an alternate set of every third section as previously described for the transcription factor ZENK (Sockman et al., 2002) except that we incubated the sections with VIP primary antibody (Immunostar, Hudson, WI) at 1:10,000 dilution for 48 hours at 4°C. We processed all of the sections in 2 ICC batches, counterbalancing the 4 photo-experience-song treatment groups within each batch.
We conducted all quantification procedures blind to the experimental condition of each subject. Using a Leica DM4000 digital research microscope, we summed the number of GnRH-immunoreactive (GnRH-ir) cells with visible nuclei in the septo-preoptic area between the anterior commissure and the supraoptic decussation of every third-cut section (1 or 2 sections were quantified per subject) under 200x magnification and Köhler illumination. Although GnRH-ir cell bodies were not seen in both sections from some birds, GnRH-ir fibers were always present. Although the GnRH antibody recognizes both GnRH-I and -II, only GnRH-I and not GnRH-II cell bodies are present in the septo-preoptic area (Sharp and Ciccone, 2005). Previous studies have found that this region is innervated by central photoreceptors (Saldanha et al., 1994, 2001) and responds to photostimulation with increased fos-like immunoreactivity (Meddle and Follett, 1997) and increased GnRH-immunoreactivity (Sockman et al., 2004). We quantified VIP-immunoreactivity (VIP-ir) in every third section of tissue medially from the medial edge of the occipito-mesencephalic tract under 400× magnification and Köhler illumination. We counted the number of VIP-ir cell bodies with visible nuclei in 4 sections through the infundibular nuclear complex (INF) and the ventromedial nucleus (VMN). Previous studies have shown that these areas of the hypothalamus contain dense concentrations of VIP-ir cells and fibers (Péczely and Kiss, 1988; Mauro et al., 1992).
LH and Vitellogenin Assays
We assayed plasma LH concentrations using a modification (Caro et al., 2006) of a homologous chicken LH radioimmunoassay (Sharp et al., 1987) using LH antiserum 1/8 at 1:24,000 dilution and LH, code AE1a run 4, as iodinated label and standard. All samples were analyzed in 1 assay with a sensitivity of 0.45 ng/mL at 80% displacement and 1.55 ng/mL at 50% displacement of the iodinated label from the LH antibody.
Plasma samples were assayed for vitellogenin using the zinc method developed for the domestic hen (Zinc kit, Wako Chemicals, Richmond, VA) (Mitchell and Carlisle, 1991) and validated for passerines (Williams and Christians, 1997). This method measures total plasma zinc and then separates the zinc bound to serum albumen from that bound to vitellogenin and very-low density lipoprotein (VLDL) by depletion of vitellogenin and VLDL from the plasma sample by precipitation with dextran sulfate. The depleted plasma sample is then assayed for zinc. Vitellogenic zinc is equal to the difference between total and depleted zinc; VLDL accounts for only 2% of total plasma zinc (Mitchell and Carlisle 1991). The concentration of vitellogenic zinc is proportional to the plasma concentration of plasma vitellogenin (Mitchell and Carlisle, 1991). Intra- and interassay coefficients of variation determined for a laying hen plasma pool were 3% (n = 15 sample replicates) and 7% (N = 16 assays), respectively.
Statistical Analyses
The sample sizes of the relevant treatment groups for each analysis are displayed at the bottom of the corresponding figures. Uneven mortality between treatment groups occurred during the 38-week photoperiod manipulation, resulting in a decrease in sample size from 20 to 15 females (6 photo-experiences and 9 photo-naïve) by the end of the study (see Fig. 2). Because of tissue damage we were unable to analyze the appropriate sections from 1 photo-naïve, conspecific song female for GnRH-ir and 2 photo-naïve, conspecific song females for VIP-ir. The exclusion of these females from the analyses decreased the number of females in the photo-naïve, conspecific song group from 5 to 4 in the GnRH-ir analysis and to 3 in the VIP-ir analysis.
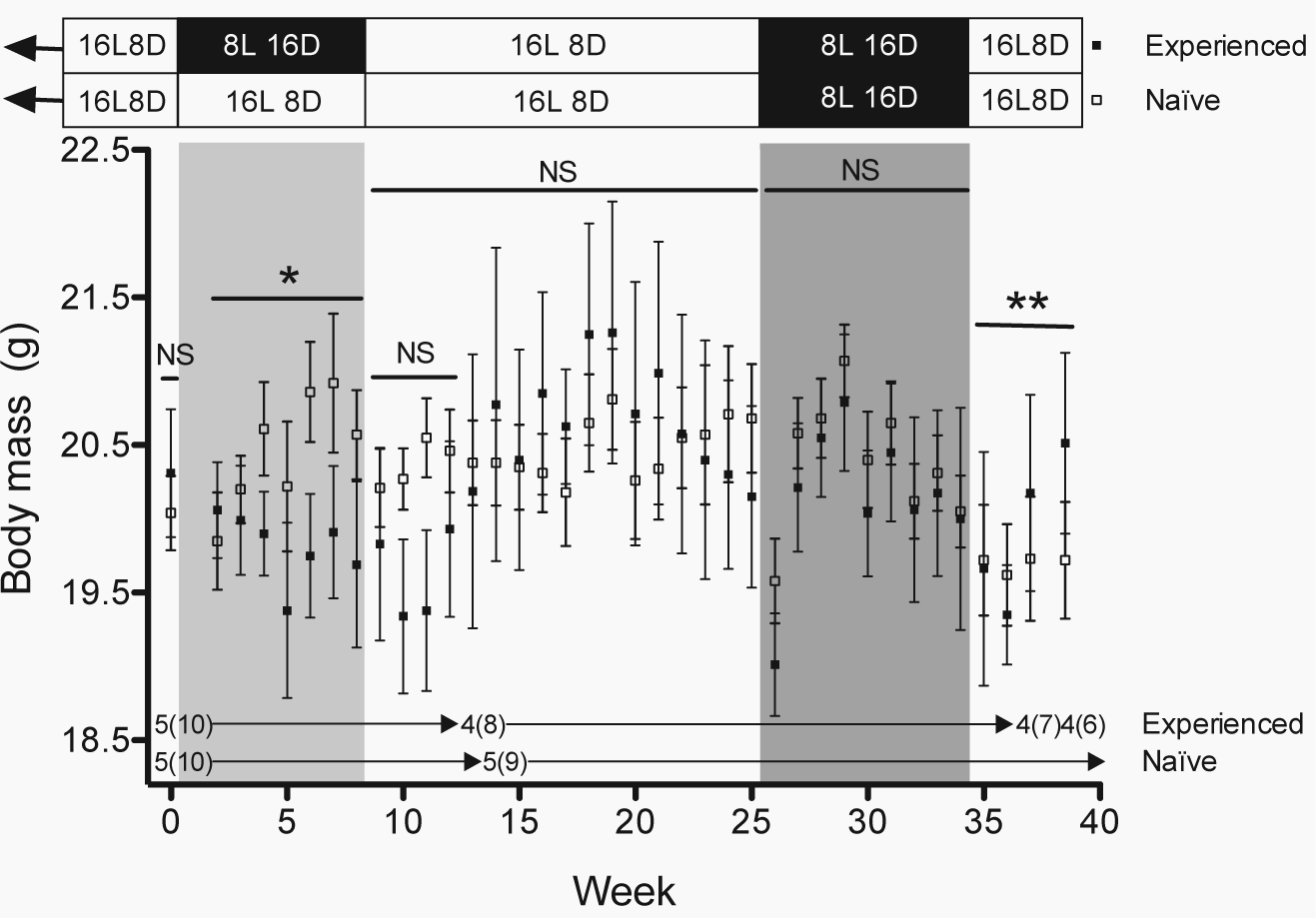
Body mass. Body mass (mean
Our data consisted of a combination of fixed (e.g., photo-experience, week) and hierarchically structured random (e.g., individual nested within triplet) effects, each of which may differ from the others in its correlation structure. In addition, the mortality mentioned above rendered our data set unbalanced. Therefore, we analyzed these data in a mixed, multilevel modeling framework using the software Stata IC 10.0 for the Macintosh (Stata Corporation, College Station, TX), which readily accommodates unbalanced, hierarchically structured combinations of fixed and random effects (Burton et al., 1998; Goldstein et al., 2002; Rabe-Hesketh and Skrondal, 2005). We used Stata’s command for multilevel mixed-effects linear regression but, for the GnRH-ir and VIP-ir cell count data, we instead used the command for multilevel mixed-effects Poisson regression because count data tend to follow Poisson distributions. These models estimated parameters with restricted maximum likelihood and used z-tests to test the null hypothesis that a coefficient equaled zero.
For analyses of GnRH-ir and VIP-ir, we counted the number of GnRH-ir or VIP-ir cell bodies and used photo-experience, song treatment, and their interaction as fixed factors, with observation (individual bird) nested within triplet as a random intercept and as a random coefficient for song treatment. For analyses of body mass and circulating LH and vitellogenin levels, we used photo-experience, week, and their interaction as fixed factors and nested observation (the individual bird’s measurement that week) within female as a random coefficient for week and nested female within triplet as a random intercept. For ovarian follicle size, we used photo-experience, female body mass, follicle order (from most to least developed), and the interaction between photo-experience and follicle order as fixed factors and nested observation (the individual bird’s measurement of an individual follicle) within female as a random coefficient for follicle order and nested female within triplet as a random intercept. As female body mass differed between photo-experience groups at the end of the study, it was included as a covariate to control for differences in ovarian follicle size due to body mass alone (Sockman et al., 2004). For comparison of circulating LH and vitellogenin levels in the 2 groups during the different rounds of photostimulation (photo-experienced: first and second rounds of photostimulation; photo-naïve: first round of photostimulation), we used photo-experience, number of weeks exposed to long days, their interaction, and the interaction between photo-experience and round of photostimulation (i.e., first or second) as fixed factors and nested observation (the individual bird’s measurement that week) within female as a random coefficient for week and nested female within triplet as a random intercept. The nesting structure we used for random effects follows the approach recommended by Schielzeth and Forstmeier (2009).
Results
Body Mass
At the start of the study, the 2 groups of female house finches had the same body mass (week 0: p > 0.5; Fig. 2), but this changed during the initial 8-week photoperiodic treatment period (weeks 2-8: photoperiodic treatment × week: z = −2.05, p < 0.05; Fig. 2). Although the body mass of birds that had experienced changing photoperiod did not change during the 8-week exposure to short days (weeks 2-8: week: p > 0.4), it increased in photo-naïve females retained on long days (weeks 2-8: week: z = 2.38, p < 0.02) (Fig. 2). However, body mass did not differ between photo-experienced and photo-naïve females during the following photoperiodic treatment period when both groups were exposed to long days (weeks 9-12: all p > 0.1; weeks 9-25: all p > 0.2) and subsequently exposed to short days (weeks 26-34: all p > 0.1) (Fig. 2). After both groups were returned to long days, photoperiodic treatment affected body mass over the last 4 weeks of the study (weeks 35-38: photoperiodic treatment × week: z = 2.31, p < 0.03; Fig. 2). During this period, the body mass of photo-experienced females increased (weeks 35-38: week: z = 2.49, p < 0.02) whereas that of photo-naïve females, which were being photostimulated for the first time, did not change (weeks 35-38: week: p > 0.7; Fig. 2).
GnRH and VIP Immunoreactivity
Although the photo-experience and song treatment main effects were not significantly, individually associated with GnRH-ir (p > 0.1 in both cases), photoperiodic experience influenced the way in which song treatment affected GnRH-ir in the hypothalamic septo-preoptic area of female house finches (photo-experience × song treatment: z = 2.80, p < 0.01; Fig. 3a). Within the photo-experienced group, females exposed to conspecific song had more GnRH-ir cells than females exposed to heterospecific song (song treatment: z = 2.46, p < 0.02; Fig. 3a). In contrast, we found no effect of song treatment on the number of GnRH-ir cells in photo-naïve females (song treatment: z = −1.55, p > 0.1; Fig. 3a). Furthermore, within the group of females exposed to conspecific song, the photo-experienced females had more GnRH-ir cells than photo-naïve females (photo-experience: z = 3.95, p < 0.001; Fig. 3a). However, for the females exposed to heterospecific song, we found no relationship between GnRH-ir cell and photo-experience (photo-experience: z = −0.86, p > 0.3; Fig. 3a).
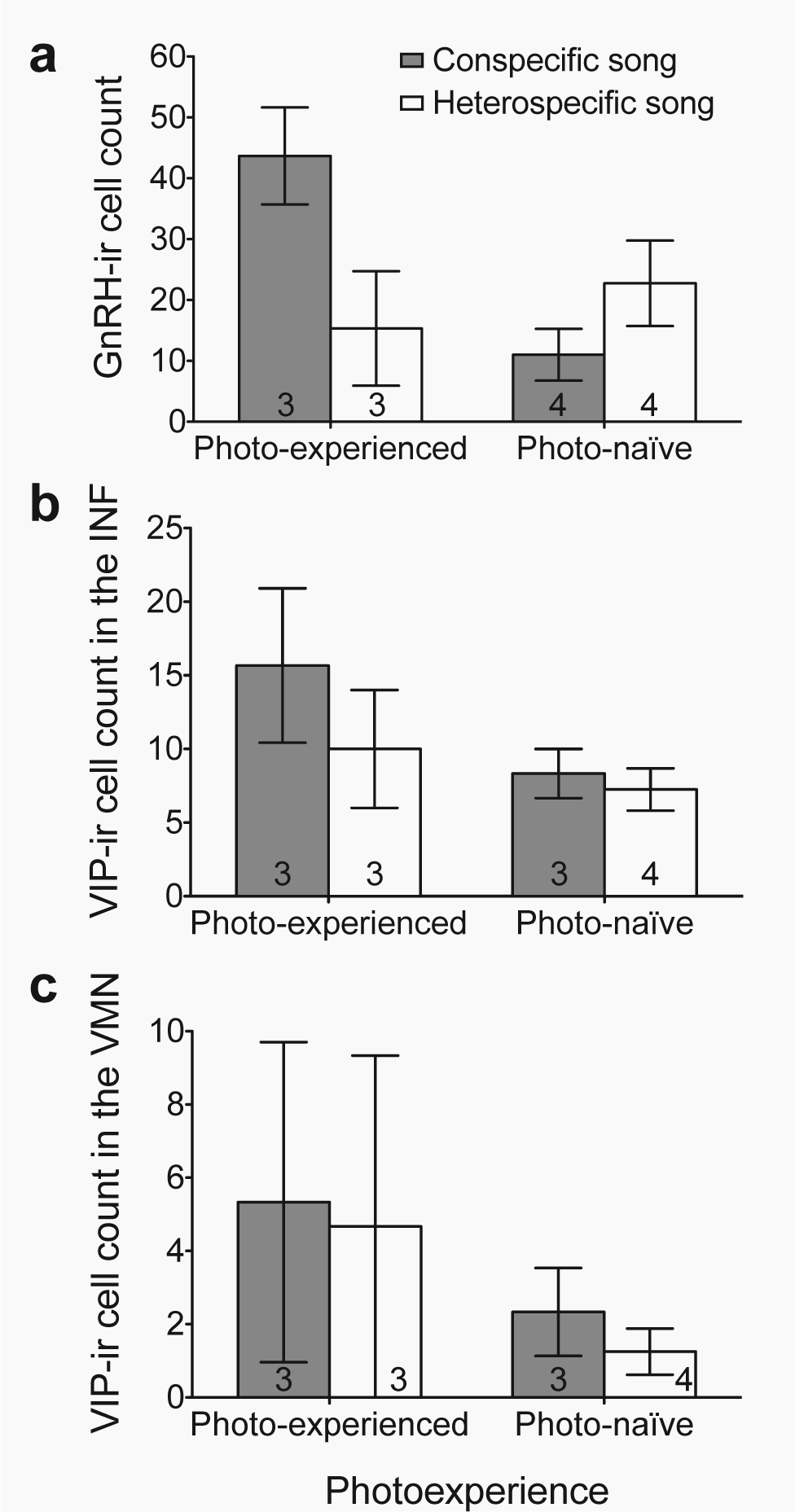
Gonadotropin releasing hormone and vasoactive intestinal polypeptide immunoreactivity. Number of cells immunoreactive for (a) gonadotropin releasing hormone (GnRH-ir) (mean
Neither photo-experience nor song treatment nor their interaction influenced the number of VIP-ir cells in the INF (Fig. 3b) or the VMN (Fig. 3c) of the hypothalamus (all p > 0.3). Even when song treatment and the interaction between photo-experience and song treatment were removed from the model, VIP-ir cell count in the INF and VMN were not related to photo-experience (both p > 0.1).
Plasma LH and Vitellogenin
At the beginning of the study and at the end of the 8-week short day photo-experience treatment, both groups of females had similar, low levels of circulating LH (weeks 0 and 8: both p > 0.5; Fig. 4). Photo-experience determined the way circulating LH levels changed during the 4 weeks immediately following photo-experience treatment (weeks 9-12: photo-experience × week: z = −10.34, p < 0.001; Fig. 4). During this time plasma LH levels remained low in photo-naïve females, who maintained their nonreproductive state (weeks 9-12: week: p > 0.8; Fig. 4). In contrast, plasma LH levels in photo-experienced females photostimulated for the first time increased almost 15-fold after 1 week of exposure to long days and declined to levels that were still 5-fold higher than those of photo-naïve females after 4 weeks on long days (weeks 9-12: week: z = −10.55, p < 0.001; Fig. 4). Plasma LH levels were low in both groups at week 25 (photo-experience: p > 0.3) and week 34 (photo-experience: p > 0.7) of the study, when both groups of females were in a nonreproductive state (Fig. 4).
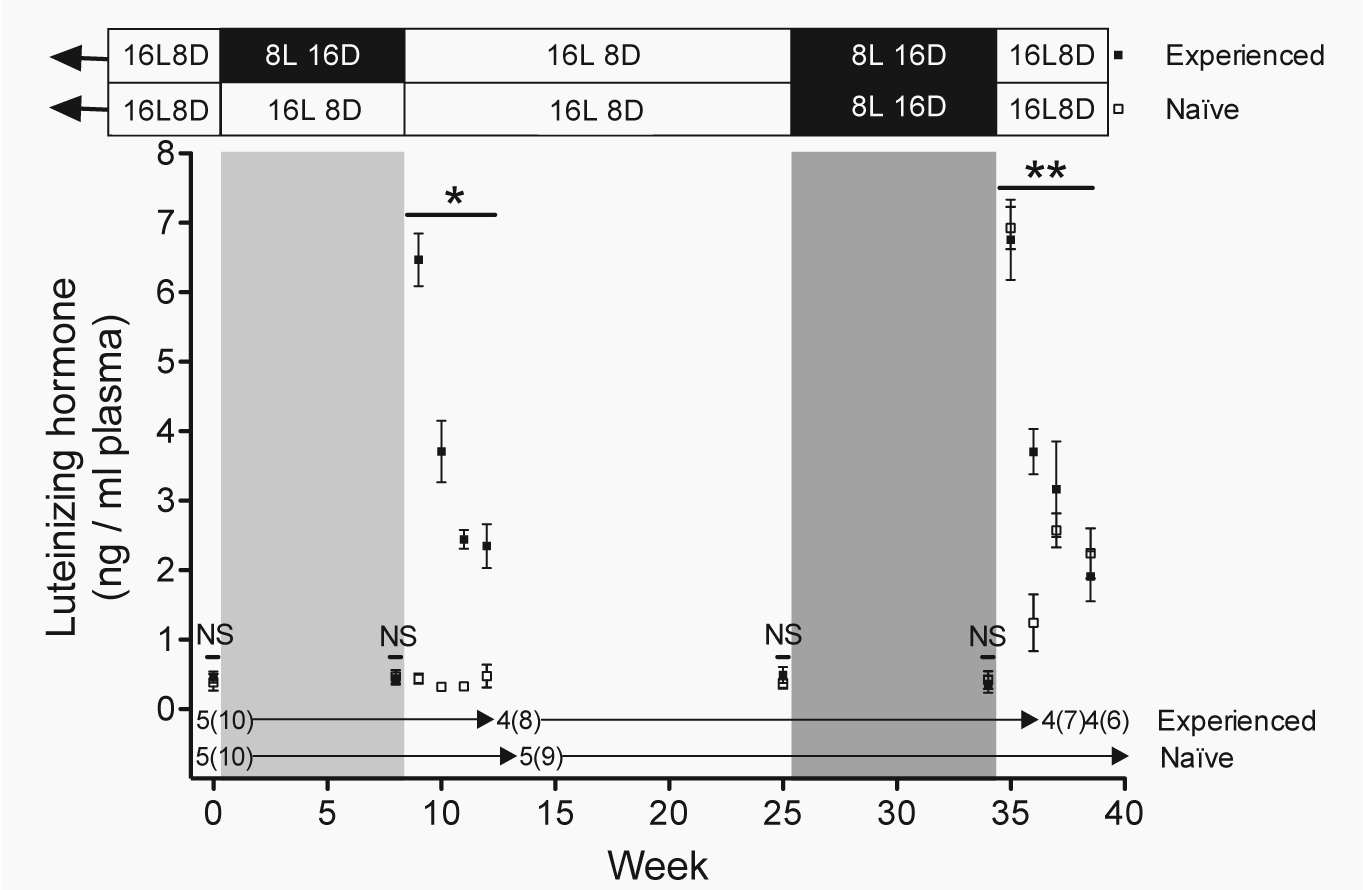
Luteinizing hormone. Plasma luteinizing hormone (LH) levels (mean
During the last 4 weeks of the study, both groups were photostimulated, and circulating LH levels in both groups increased almost 15-fold after exposure to long days for 1 week and then declined approximately 3-fold by the end of the study (weeks 35-38: week: z = 7.51, p < 0.001; photo-experience and photo-experience × week: p > 0.9; Fig. 5). This photo-induced surge in LH was similar to the LH surge observed in photo-experienced females undergoing photo-induced early reproductive development for the first time (photo-experienced weeks 9-12 vs. photo-experienced weeks 35-38 vs. photo-naïve weeks 35-38: weeks exposed to long days: z = −7.76, p < 0.001; photo-experience and photo-experience × weeks exposed to long days (1-4): p > 0.5, photo-experience × round of photostimulation: p > 0.8; Fig. 4).
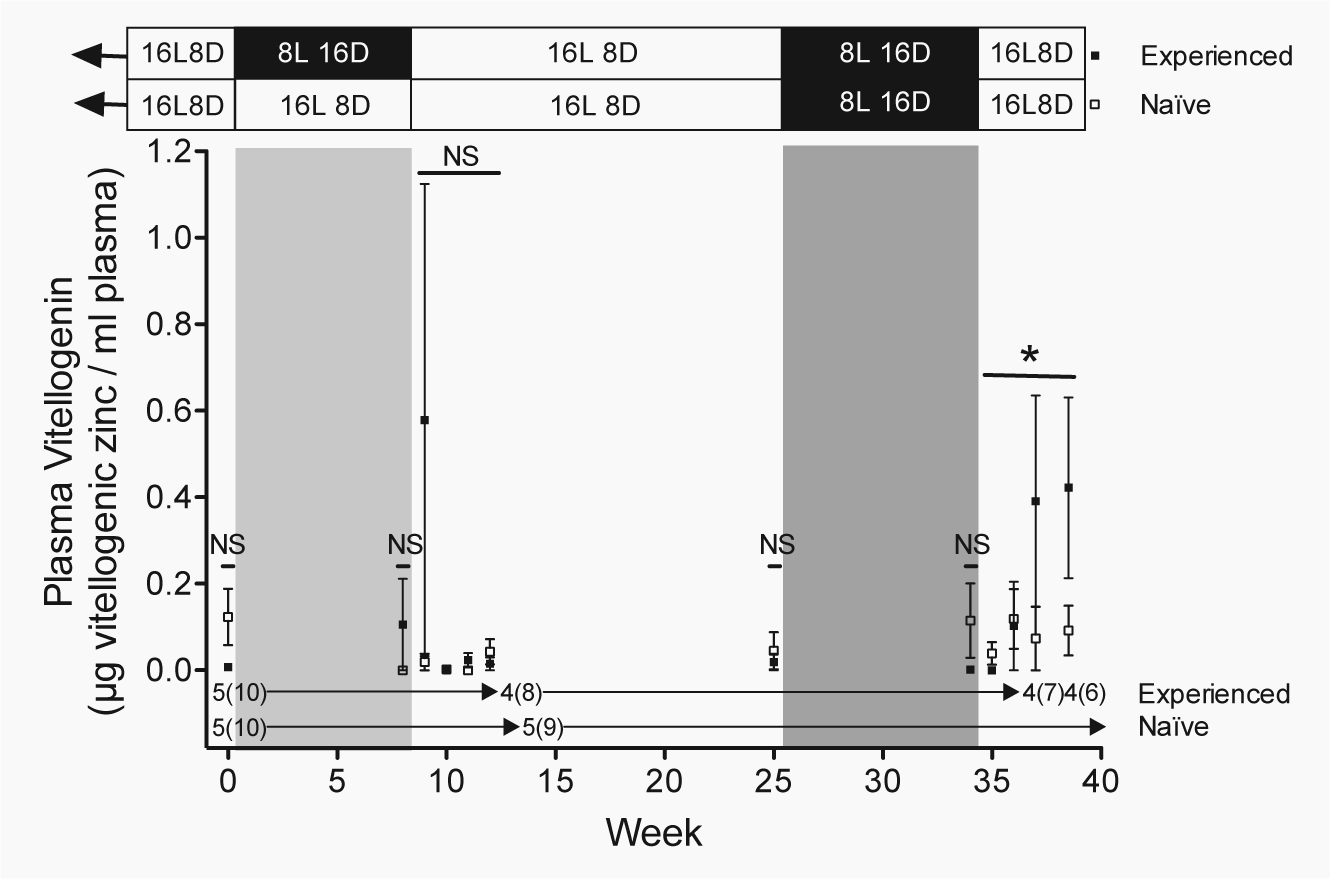
Vitellogenin. Plasma vitellogenin levels (mean
Both groups of females had similar, low levels of circulating vitellogenin at the beginning of the study (week 0: z = −1.53, p > 0.1) and at the end of the 8-week photoperiodic treatment (week 8: z = 1.00, p > 0.3; Fig. 5). Plasma vitellogenin levels also did not differ between the groups in the 4 weeks following photoperiodic treatment, despite the fact that photo-experienced females were undergoing photostimulation for the first time and the photo-naïve females remained in a nonreproductive state (weeks 9-12: all p > 0.3; Fig. 5). There was, however, 1 photo-experienced female that had elevated vitellogenin levels after 1 week of exposure to reproductively stimulatory long days, resulting in large variation in this group, but her vitellogenin levels were undetectable in the following week (Fig. 5). Still, this heteroscedasticity may have contributed to our inability to detect a difference between treatment groups during this time period. Similar to plasma LH, circulating vitellogenin levels were low in both groups at week 25 (photo-experience: p > 0.6) and week 34 (photo-experience: p > 0.2) of the study, when both groups of females were in a nonreproductive state (Fig. 5).
When both groups were returned to long days, photo-experience affected how plasma vitellogenin changed over the last 4 weeks of the study (weeks 35-38: photo-experience × week: z = 2.00, p < 0.05; Fig. 5). Photo-experienced females undergoing photo-induced early reproductive development for the second time exhibited an increase in circulating vitellogenin levels over the last 4 weeks of the study (weeks 35-38: week: z = 2.18, p < 0.03; Fig. 5). In contrast, the vitellogenin levels of females in the photo-naïve group, who were undergoing photostimulation for the first time, did not change during this time (weeks 35-38: week: p > 0.5; Fig. 5). This pattern was similar to that of photo-experienced females during their first round of photostimulation (photo-experienced weeks 9-12 vs. photo-naïve weeks 35-38: photo-experience: p > 0.1; weeks exposed to long days: p > 0.8; interaction p > 0.1; Fig. 5).
Follicular Development
Photo-experienced females undergoing photo-induced early reproductive development for the second time had larger ovarian follicles than photo-naïve females being photostimulated for the first time (photo-experience: z = 1.97, p < 0.05; Fig. 6a). The photo-experienced group exhibited large interindividual variation in ovarian follicle diameter that can be explained by variation in female body mass (body mass: z = 3.91, p < 0.001). When ovarian follicle diameters were adjusted for body mass, the marked interindividual variation in the follicle diameter of photo-experienced females decreased (Fig. 6b). By the end of the study, following 4 weeks of photostimulation, 2 of the 6 photo-experienced females had yellow ovarian follicles that had begun to take up yolk, and 1 of these females laid an egg on the last day of the study. In contrast, none of the 9 photo-naïve females had any yellow, yolky follicles.
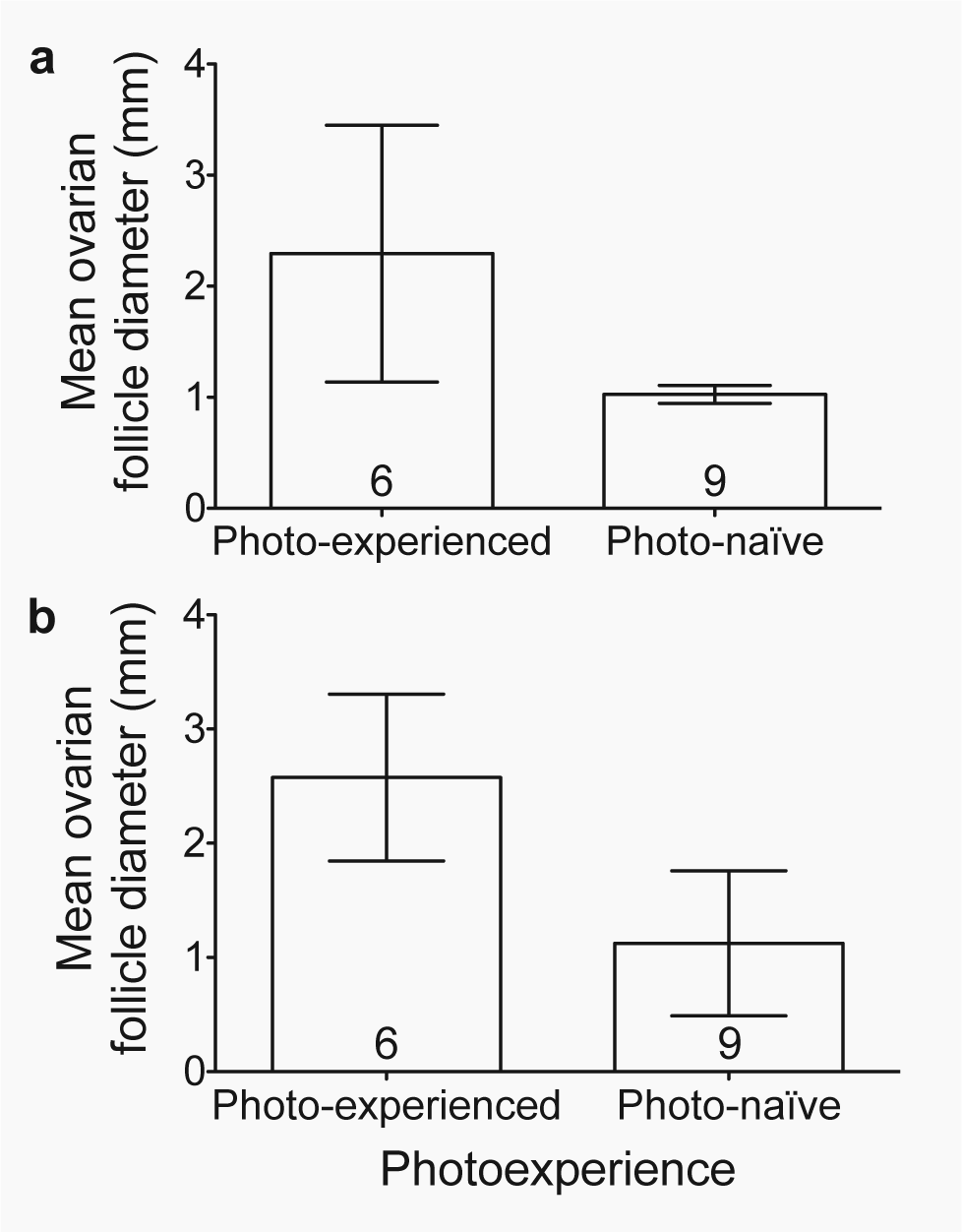
Ovarian follicle size. Effect of photo-experience on the average diameter (mean
Discussion
Our results suggest that previous photo-experience sensitizes the neuroendocrine system to the reproductively stimulatory effects of increasing photoperiod and changes the way the neuroendocrine system responds to the supplementary cue of male song. These effects of photo-experience may be responsible, at least in part, for the age-related advance in the early stages of reproductive development.
Body Mass
The photo-induced increase in the body mass of photo-experienced female house finches observed over the last 4 weeks of the study is consistent with the increase in body mass associated with early reproductive development and egg production. Sockman and colleagues observed similar photo-experience-dependent patterns of changes in body mass in female European starlings (Sockman et al., 2004). Visual observations of pectoral muscle shape and furcular and abdominal fat deposits were unchanged during this period (Salvante and Sockman, unpublished data), and thus the 1-g difference in body mass between photo-experienced and photo-naïve females in our study was likely due to the additional mass of the newly regrown reproductive tissues, including the ovary, ovarian follicles, and associated yolk and oviduct, of the photo-experienced females (Vézina and Salvante, 2010). Egg-producing female passerines of similar size to house finches display similar gains in body mass above nonbreeding values (e.g., great tits, Parus major [Silverin, 1978]; zebra finches, Taeniopygia guttata [Salvante et al., 2010]).
VIP
We did not find an effect of photo-experience on VIP-ir in the INF or the VMN of the hypothalamus following 4 weeks of concurrent photostimulation. This may be due to large variation in the number of VIP-ir cells observed in all treatment groups and small sample sizes resulting in low statistical power. However, photostimulation of previously photo-naïve, male European starlings had no effect on basal hypothalamic VIP levels (measured every 2 weeks through week 8, then every 4 weeks through week 24 of exposure to long days) when compared with levels measured while the birds were exposed to short days (Dawson et al., 2002).
GnRH
The elevated GnRH expression observed in photo-experienced females exposed to conspecific but not heterospecific song suggests that previous experience with photostimulation “primes” the brain to increase its responsiveness to socially relevant environmental stimuli during subsequent breeding seasons. Although our results are based on small sample sizes within each photo-experience-song treatment group, similar effects of reproductively relevant and stimulatory social cues and previous photo-experience on GnRH expression or its priming effects on the brain have been reported by others. Exposure to reproductively relevant and stimulatory social cues (e.g., conspecific vocalizations, removal of competitors for mates, and presence of potential mates) has been associated with increased GnRH expression and enhanced reproductive physiology (e.g., circulating LH levels), morphology (e.g., male coloration), and behavior (e.g., dominance behaviors) in a variety of vertebrates including female ring doves (Streptopelia risoria) undergoing egg formation (Cheng et al., 1998), male cichlid fish (Astatotilapia (Haplochromis) burtoni) transitioning from subordinate to dominant reproductive status (Burmeister et al., 2005), and photostimulated male European starlings (Stevenson and Ball, 2009). Similarly, photorefractory adult European starlings with prior photo-experience, and thus prior exposure to elevated levels of GnRH, exhibited a larger LH response following exogenous GnRH administration than photorefractory juveniles that lack previously exposure to elevated GnRH levels (McNaughton et al., 1995).
Circulating LH
The surge in circulating LH levels observed in both photo-experience groups at week 35 was similar to the LH surge observed in photo-experienced females undergoing photostimulation for the first time at week 9 and in photo-experienced European starlings undergoing photostimulation for the first and second times (Sockman et al., 2004). Interestingly, the marked photo-induced LH surge was absent in photo-naïve starlings exposed to reproductively stimulatory long days for the first time (Sockman et al., 2004). That study suggested that the lack of LH response may have been due to desensitization of the pituitary to GnRH by negative feedback of chronic, low-level gonadal steroid activity associated with the prolonged time (32 weeks) that photo-naïve females spent on an 8 h L: 16 h D photoperiod. In contrast, our photo-naïve females spent only 9 weeks exposed to short day lengths and did respond to photostimulation with a surge in LH. Therefore, the lack of an effect of photo-experience on the LH response to photostimluation suggests that LH may not play a direct role in the physiological mechanisms underlying age-related variation in early reproductive development. However, as the up-regulation of LH receptors is hormone-dependent (You et al., 2000; Johnson and Bridgham, 2001), if the expression of these regulatory hormones is dependent on photo-experience, then LH activity may also vary with photo-experience and potentially contribute to the age-related variation observed in early reproductive development.
Circulating Vitellogenin and Follicular Development
In passerine birds, the onset of vitellogenin production is tightly coupled with the onset of follicular yolk deposition (Challenger et al., 2001; Salvante and Williams, 2002). As both groups of females had relatively stable and low vitellogenin levels during the first 4 weeks of their first round of photostimulation, this relationship suggests that neither group had begun follicular yolk deposition during this time. This, together with the elevated plasma vitellogenin levels and the presence of an egg and yolky follicles in photo-experienced females, but not in age-matched photo-naïve females by the end of the study, is consistent with the hypothesis that photo-experience influences the age-related advancement of egg production and laying date.
One major difference between the 2 groups at the end of the study was that the photo-experienced females had previously been exposed to elevated circulating levels of E2 during their first experience with photostimulation and gonadal development. Primary exposure of the avian liver to E2 induces genomic changes to the regulatory sites of the genes coding for vitellogenin and apoVLDL-II, the VLDLy-specific surface protein, including demethylation of the E2-receptor complex binding site at the 5′ end of the vitellogenin gene, and changes to the chromatin of the vitellogenin and apoVLDL-II genes resulting in nuclease-hypersensitive sites (Wilks et al., 1982; Kok et al., 1985). These and other E2-induced genomic changes may contribute to the earlier induction and more rapid synthesis of vitellogenin and apoVLDL-II mRNA and circulating vitellogenin and VLDLy following secondary estrogen exposure (Jost et al., 1978). Previous exposure to elevated levels of E2 may also contribute to the advance in egg formation in photo-experienced females via the stimulatory effect that E2 has on the synthesis of its own receptors (Sutherland and Baulieu, 1976; Cidlowski and Muldoon, 1978) in the avian oviduct. Secondary estrogen administration has also been associated with rapid increases in both nuclear binding of progesterone receptor (Boyd-Leinen et al., 1984) and ovalbumin mRNA transcription (Swaneck et al., 1979) in the avian oviduct. If these priming effects of E2 persist until the next breeding season, they could contribute to the advancement of oviduct growth and development, egg formation, and laying date in photo-experienced females.
We found that female house finches with prior photo-experience exhibited advanced early reproductive development in comparison with age-matched birds with no prior experience with reproductively stimulatory long days. Our results suggest that photo-experience and not age, per se, may explain at least in part the advancement in laying date and enhanced reproductive output observed in older seasonally breeding birds compared with first-year females (Saether, 1990; Fowler, 1995).
Footnotes
Acknowledgements
We thank Kendra B. Sewall, Danielle M. Racke, and C. Ryan Campbell for their help with data collection; Sachi Vora, Katie Suppler, Kristina Simmons, and Adam Byerly for their help with bird care; and Tony D. Williams for logistical support. This study was supported by NIH R01 NS055125 to K.W.S.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
