Abstract
The human circadian timing system is most sensitive to the phase-shifting effects of light during the biological nighttime, a time at which humans are most typically asleep. The overlap of sleep with peak sensitivity to the phase-shifting effects of light minimizes the effectiveness of using light as a countermeasure to circadian misalignment in humans. Most current light exposure treatments for such misalignment are mostly ineffective due to poor compliance and secondary changes that cause sleep deprivation. Using a 16-day, parallel group design, we examined whether a novel sequence of light flashes delivered during sleep could evoke phase changes in the circadian system without disrupting sleep. Healthy volunteers participated in a 2-week circadian stabilization protocol followed by a 2-night laboratory stay. During the laboratory session, they were exposed during sleep to either darkness (n = 7) or a sequence of 2-msec light flashes given every 30 sec (n = 6) from hours 2 to 3 after habitual bedtime. Changes in circadian timing (phase) and micro- and macroarchitecture of sleep were assessed. Subjects exposed to the flash sequence during sleep exhibited a delay in the timing of their circadian salivary melatonin rhythm compared with the control dark condition (p < 0.05). Confirmation that the flashes penetrated the eyelids is presented by the occurrence of an evoked response in the EEG. Despite the robust effect on circadian timing, there were no large changes in either the amount or spectral content of sleep (p values > 0.30) during the flash stimulus. Exposing sleeping individuals to 0.24 sec of light spread over an hour shifted the timing of the circadian clock and did so without major alterations to sleep itself. While a greater number of matched subjects and more research will be necessary to ascertain whether these light flashes affect sleep, our data suggest that this type of passive phototherapy might be developed as a useful treatment for circadian misalignment in humans.
The human circadian system controls the timing of most physiological systems including the endocrine, immune, and neurologic systems. Proper alignment of the circadian system with the external day is important for good health (Vogel et al., 2012). The primary manner by which the internal circadian system remains entrained with the external day is through regular exposure to light and dark (Duffy and Czeisler, 2009). Circadian misalignment can lead to health disruptions from mundane (e.g., jet lag) to serious (e.g., cancer) (Stevens, 2005). A number of sleep disorders (advanced and delayed sleep phase disorder, non-24-h sleep-wake disorder, shift work sleep disorder) are also directly attributable to an improper alignment between the circadian system and an individual’s social schedule (Morgenthaler et al., 2007). While bright light is a useful tool for maintaining proper alignment, the human circadian timing system is most sensitive to light during times at which people are normally asleep (St. Hilaire et al., 2012). Administration of light during sleep is plausible, given the penetrance of light through closed eyelids (which act as a red-pass filter with approximately twice as much of the longer wavelengths passing through as the shorter wavelengths) (Moseley et al., 1988; Robinson et al., 1991), and preliminary evidence indicates that such treatment is effective in shifting circadian rhythms (Cole et al., 2002; Figueiro and Rea, 2012). Continuous bright light or long-duration light pulses delivered during this most sensitive phase would, however, disrupt sleep as these would be likely to either cause or sustain awakenings (Cajochen et al., 2000). We have recently demonstrated that a train of millisecond flashes of light presented to subjects who are awake has the capacity to elicit phase delays of the human circadian system (Zeitzer et al., 2011). Bringing these 2 streams of evidence together, we examined whether a train of millisecond flashes of light during sleep had the capacity to change the phase of the circadian system and to do so without affecting sleep.
Materials and Methods
The protocol was approved by the Stanford University Institutional Review Board and was conducted under the principles outlined in the Declaration of Helsinki. Written informed consent was received from all participants prior to inclusion in the study. This study was registered with ClinicalTrials.gov #NCT01119365 (http://www.clinicaltrials.gov/ct2/show/NCT01119365).
Participants
Thirteen healthy young (26.9 ± 4.8 years) male (n = 6) and female (n = 7) volunteers participated in a parallel-group, 16-day study. Subjects were studied between November 2010 and July 2012. Sample size was calculated a priori based on estimated variance of phase change measures and a minimal detectable difference in phase change of 30 min between groups. All volunteers self-reported good physical and mental health, they were not depressed (Radloff, 1977), they had no evidence of sleep disorders (Buysse et al., 1989) nor did they use any medications that could affect sleep (e.g., antihistamines, antidepressants, benzodiazepine agonists), they did not abuse alcohol (Babor et al., 2001), they had normal color vision (Ishihara, 2007), they were nonsmokers, and they were of moderate chronotype (Horne and Östberg, 1976). All female subjects began the in-laboratory portion of their stay within 5 days from the onset of menses (Baker and Driver, 2007).
At-Home Sleep/Circadian Stabilization Protocol
For 2 weeks prior to entry into the laboratory, subjects participated in an at-home sleep-circadian stabilization protocol during which they were required to keep a strict sleep schedule. Subjects self-selected bedtimes and wake times that had to be 7 to 9 h apart, and each bedtime and wake time had to be within ±30 min of this target time. Verification was accomplished using self-reported sleep-wake logs and wrist actigraphy (Ancoli-Israel et al., 2003). Such a schedule is useful for normalizing the amplitude and phase angle of the circadian system (Zeitzer et al., 2011; Jewett et al., 1994; Duffy et al., 1999).
In-Laboratory Light Exposure Protocol
Subjects stayed in the laboratory for a 35-h protocol starting 7 h after habitual wake time (Figure 1). All events were timed relative to the subjects’ at-home sleep patterns (Zeitzer et al., 2011). In the laboratory, subjects were exposed to less than 10 lux during scheduled wake and complete darkness (<0.05 lux) during scheduled sleep. Low illuminance is necessary to unmask the endogenous rhythm of melatonin (Zeitzer et al., 2000). For 9 h prior to bedtime, subjects lay in bed in a constant, semirecumbent posture (Deacon and Arendt, 1994; Duffy and Dijk, 2002). Instead of receiving dinner, subjects were given hourly equicaloric aliquots of food and isovolumetric (90 mL) water (Mifflin et al., 1990) in order to minimize any possible effects of meals on metabolism and subsequent effects on circadian rhythms. Saliva samples were obtained every 30 min, immediately before or midway between snacks, using untreated Salivettes (Sarstedt, Newton, NC). Saliva samples were spun and immediately frozen. Prior to bedtime, subjects were fitted with electrodes on their faces (bipolar electro-oculogram, chin electromyogram) and scalps (electroencephalogram [EEG] C3/C4/O1/O2) (Klem et al., 1999). Sleep data were recorded at a 256-Hz sampling frequency on a polysomnograph (PSG) (Siesta, Compumedics, Cary, NC) and scored according to standard clinical criteria (Rechtschaffen and Kales, 1968) by a single expert PSG technician blind to group allocation. Sleep data were unavailable from 2 of the subjects exposed to a dark stimulus due to equipment failure.
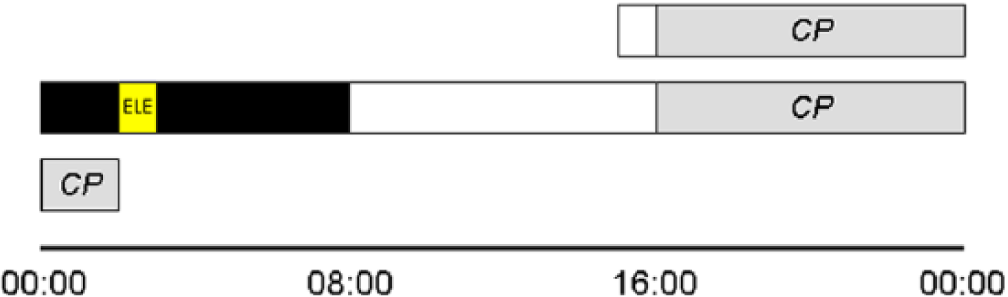
Diagram showing the protocol flow for a subject who had habitually slept from 0000 h until 0800 h during the 2 weeks prior to laboratory entry. One hour after entry, subjects had an initial constant posture (CP). For an hour, starting 2 h after lights out, subjects were exposed to an experimental light exposure (ELE), consisting of either a flash sequence or darkness, during sleep. Subjects were ambulatory in the dimly lit laboratory until the start of a second CP. Phase change in salivary melatonin phase was determined between CP1 and CP2. Sleep during the ELE was compared with that which occurred during the prior hour on the same night.
From hours 2 to 3 after bedtime, subjects were exposed to 1 of 2 lighting regimes: 60 min of darkness (i.e., no change in lighting) (n = 7) or 60 min of a flash sequence (n = 6). The flash sequence consisted of 1 moderately bright (~2995 lux at corneal level) 2-msec flash of white light delivered every 30 sec. The flash was generated by a wide-spectrum xenon flash bulb (ColorDome, Diagnosys, Lowell. MA) placed directly above the subject’s head. During sleep, subjects lay supine, confirmed with a PSG position sensor, with their head between 2 foam bolsters. The ColorDome was controlled from outside the room and the subject was not intentionally awakened at any point during this procedure. Following the stimulus, subjects were allowed to continue to sleep in darkness for an additional 5 h and were awakened into dim light at their habitual wake time. For the first 8 h after wake time on the second day in the laboratory, subjects were ambulatory en suite, after which they got into bed for an 11-h constant posture protocol as described for Day 1. This second day was used to make a second, post–stimulus phase determination. Unintentionally, the distribution of male and female subjects was not equal across the 2 study groups. All 6 of the participants exposed to flashes were female and only 1 of the 7 participants exposed to dark was female. Males have, on average, a circadian period length that is approximately 6 min longer than that of women (Duffy et al., 2011). As such, we would expect that the group exposed to dark (primarily male) would have a greater change in circadian phase due to their intrinsic period. This would tend to reduce the difference between the dark group and the light-exposed group, making a light-induced phase shift more difficult to detect.
Salivary Melatonin
Saliva samples were defrosted and assayed for melatonin concentrations using a commercially available enzyme-linked immunosorbent assay (ALPCO, Salem, NH) according to the manufacturer’s instructions (intra-assay variability 12.6%, interassay variability 22.9%, sensitivity 0.5 pg/mL, per the manufacturer). Single kits were used for each subject. Melatonin phase was specified as the time at which salivary melatonin concentrations rose above a subject-specific threshold, calculated as the average of the first 3 daytime concentrations plus twice the standard deviation of these values (Voultsios et al., 1997).
Electroencephalography
EEG data were processed for examination of spectral content and for evoked EEG responses associated with the light flashes. For spectral analysis, signals from each cortical electrode (C3-A2, C4-A1, O1-A2, O2-A1) were deconvoluted into component frequencies (PRANA, PhiTools, Chicago, IL). Using this method for each 30-s bin, we calculated the absolute power for the frequency bands: delta (0.5-4 Hz), theta (4-7.5 Hz), alpha (8-12.5 Hz), sigma (12.5-14 Hz), beta (14-29 Hz), and gamma (30-40 Hz). Data were converted to relative power by normalizing to the total spectral power. Using automatic detection algorithms, EEG data with artifact were removed from the analyses. Only data from periods of scored NREM were used for power spectral analysis. For examination of evoked responses, we folded the EEG data at 30-sec intervals during the 1 h of light stimulus. This was done separately for each of the 4 derivations (O1-A2, O2-A1, C3-A2, C4-A1). Average waveforms were generated (i.e., each 30-sec waveform was the average of 120 of the 30-sec intervals). The O1-A2 event-related potential (ERP) has data from only 5 subjects, as 60-cycle noise rendered these channels unusable in 2 subjects. These ERPs were not excluded from the power spectral analysis of sleep.
All data are presented as mean ± SD. To determine the effect of the experimental stimuli on circadian timing, we examined the change in the onset of salivary melatonin onset during the first and second constant postures. To examine the effect of the experimental stimuli on sleep, we compared both EEG power spectral data and sleep staging during the hour of the experimental stimulus with the prior hour of sleep. We also examined EEG power spectral data and sleep staging during the 5 h following the experimental stimuli. Within-group differences were assessed with paired 2-tailed t tests, while between-group differences were assessed with unpaired 2-tailed t tests adjusted for scedasticity. When we compared multiple EEG bands, to keep the overall test α′ = 0.05, individual test α values were set to 0.008 incrementing upward to 0.05 for each test, according to the method of Holm-Bonferroni (Holm, 1979).
Results
Timing of the Stimuli
To examine the effectiveness of brief flashes of light in changing the timing of the human circadian system, we exposed a group of young, healthy volunteers (n = 13) to either 1 h of darkness or a 1-h sequence of light flashes (a 2-msec, ~2995-lux flash every 30 sec) during sleep. Both exposures were scheduled to occur approximately 4 h after the onset of melatonin secretion, which typically occurs about 2 h before habitual bedtime. As the effects of light on the human circadian timing system are dependent on the phase of the circadian cycle during which the light stimulus is presented (St. Hilaire et al., 2012), it is important to deliver light at the same phase in all subjects. At the targeted circadian phase, light was expected to delay the phase (timing) of the circadian rhythm (St. Hilaire et al., 2012). Of the 6 subjects exposed to the light flashes, 5 were clustered at the same phase (beginning 4.3 ± 0.85 h after melatonin onset, determined retrospectively). One subject (light stimulus started 6.8 h after melatonin onset) was excluded due to the mistimed light exposure. Of the 7 subjects exposed to the control dark stimulus, all were clustered at the same phase (stimulus beginning 4.5 ± 0.78 h after melatonin onset), which was indistinguishable from the phase at which the flash stimulus was presented (p = 0.71, t test). Thus, subjects exposed in both the control and the flash conditions were given experimental stimuli at the same circadian phase.
Circadian Response to the Stimuli
In response to the dark condition (equivalent to no change in regular sleep conditions), subjects exhibited a nonsignificant 0.01 ± 0.48 h phase change (advance) of their circadian rhythm of salivary melatonin (p = 0.95, paired t test). In response to the flash stimulus, subjects exhibited a significant −0.48 ± 0.15 h phase change (delay) in their circadian rhythm (p < 0.01, paired t test). The phase change exhibited by subjects in response to the flash stimulus was significantly different from that exhibited by subjects in response to the control (darkness) stimulus (p < 0.05, t test) (Figure 2). Thus, despite the approximately 90% attenuation of light by the eyelid (Moseley et al., 1988; Robinson et al., 1991), this flash stimulus was able to change the circadian phase in our volunteers while they were asleep.
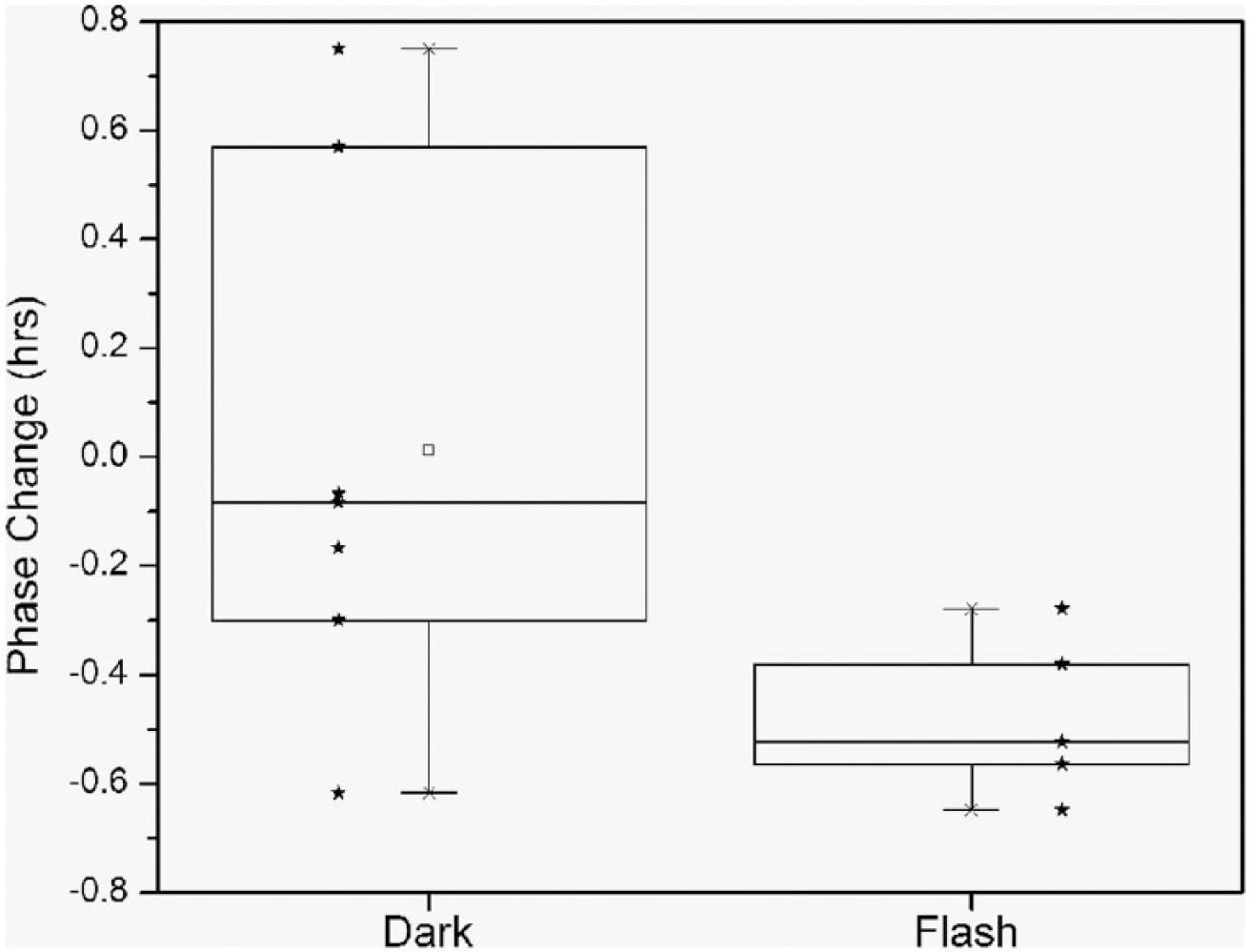
Box chart of circadian phase changes (timing of melatonin onset) after exposure to the dark and flash stimuli. No significant phase change was exhibited under the dark condition (p = 0.95, paired t test), while the flash condition induced a significant 30-min phase change (p < 0.01, paired t test) that was also significantly different from that observed in the dark condition (p < 0.05, t test). Individual data are shown as stars.
Event-Related Potential
We used an independent measure to determine whether flashes of light were indeed passing through the eyelids and evoking a change in the brain. We analyzed the EEG for an ERP occurring at the time of the stimulus. We averaged 30-sec windows of the EEG to examine the impact that a repeated stimulus might have. In doing so, we detected an ERP in each of the 4 EEG channels that we recorded (bilateral central leads and occipital leads) (Figure 3). This ERP, a short-lived 0.5-Hz change in the EEG, confirms that the cortex is actually receiving information about the light flashes. We are, therefore, convinced that the flashes that we delivered were able to penetrate the eyelids and impact brain function.
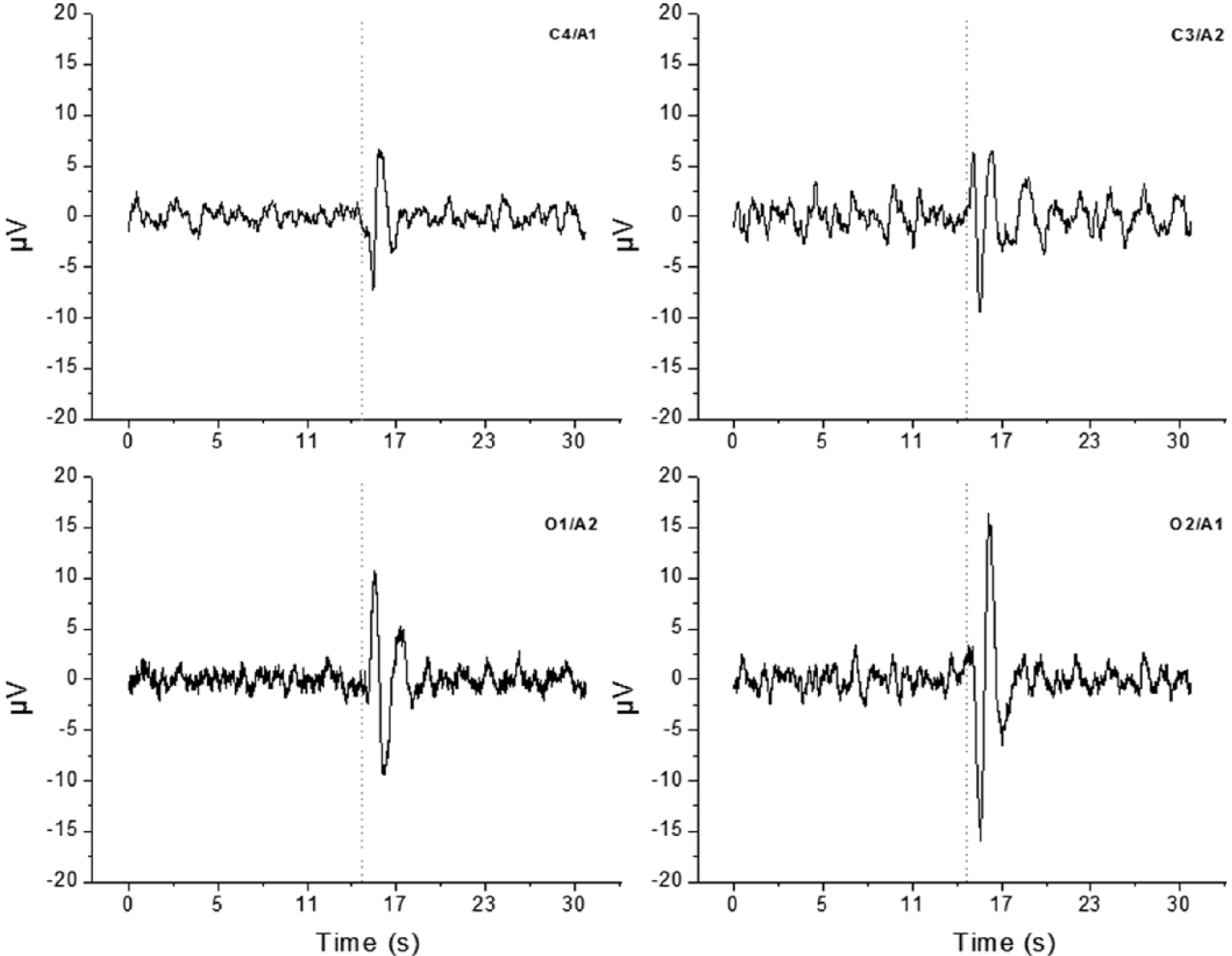
The ERP generated from subjects exposed to the flash sequence during sleep. Each ERP represents the average 30-sec EEG waveform during the 1-h exposure. Data were averaged within and then between subjects for each of the 4 derivations (2 central, C4 and C3; 2 occipital, O1 and O2). Data are centered on the timing of the flash (dotted line). Robust changes are observable in both of the occipital (primary visual) cortex channels with lesser but still obvious changes in both of the central cortical channels, confirming that the flash signal is passing through the eyelids and affecting brain firing patterns.
Effects on Sleep
We also examined whether subjects were awake during the flashes or whether the flashes caused an alerting response or any other change in sleep. We compared sleep during the hour of exposure to the flashes with sleep the previous hour, during which no flash stimuli were present. There were no differences in the change in wake (10.5 ± 18.0 min, p = 0.17, paired t test), stage N1 (1.07 ± 18.0 min, p = 0.88, paired t test), stage N2 (–8.36 ± 23.5 min, p = 0.38, paired t test), stage N3 (–2.64 ± 9.81 min, p = 0.50, paired t test), or REM (–0.429 ± 6.75 min, p = 0.87, paired t test) sleep between the 2 h, nor was there a difference in the number of state transitions (-0.14 ± 13, p = 0.98, paired t test). Thus, from a macroarchitectural level, there were no changes in sleep that could be attributed to the flash stimulus. We then compared the spectral power present in the commonly parsed frequency bands during the hour of flashes to the previous hour. In none of the predefined spectral power bands—delta (10.9% ± 19.9%, p = 0.65, paired t test), theta (44.8% ± 79.1%, p = 0.88, paired t test), alpha (74.4% ± 166%, p = 0.30, paired t test), sigma (65.6% ± 182%, p = 0.68, paired t test), beta (173% ± 309%, p = 0.53, paired t test), gamma (206% ± 383%, p = 0.75, paired t test)—was there a difference between the 2 h. Thus, from the level of EEG spectrum, there were no changes in sleep that could be attributed to the flash stimulus. Therefore, we observed no direct effect of the flashes on sleep.
Finally, we examined whether the flash stimulus caused a change in sleep that outlived the flashes themselves. We examined sleep during the 5 h after experimental intervention, either the flash or dark stimulus, and before lights-on in the morning. This poststimulus sleep was indistinguishable between the subjects exposed to the flashes and those exposed to darkness. In comparison of these 5 h in subjects exposed to dark versus wake, the amounts of wake (43.7 ± 30.0 min vs. 59.3 ± 53.0 min, p = 0.58, t test), N1 (142 ± 28.6 min vs. 148 ± 32.3 min, p = 0.75, t test), N2 (57.9 ± 20.3 min vs. 62.5 ± 21.1 min, p = 0.73, t test), N3 (13.5 ± 12.1 min vs. 1.90 ± 4.25 min, p = 0.07, t test), and REM (43.3 ± 5.90 min vs. 28.3 ± 22.9 min, p = 0.22, t test), as well as the number of transitions (128 ± 10.2 vs. 123 ± 38.0, p = 0.79, t test), were statistically similar between the groups. Relative amounts of delta (55.7 ± 2.29 vs. 46.2 ± 14.9, p = 0.23, t test), theta (11.8 ± 2.37 vs. 16.9 ± 18.4, p = 0.57, t test), alpha (5.52 ± 1.21 vs. 4.64 ± 2.14, p = 0.45, t test), sigma (2.78 ± 1.08 vs. 2.83 ± 0.976, p = 0.94, t test), beta (3.31 ± 0.675 vs. 3.37 ± 0.661, p = 0.89, t test), and gamma (1.10 ± 0.197 vs. 1.73 ± 1.18, p = 0.23, t test) power also were statistically similar between the groups. Thus, not only did the light stimulus not change sleep during the administration of the stimulus, but it did not appear to affect sleep during the 5 h after the stimulus, indicating that the flash intervention was able to simultaneously change circadian phase and not affect sleep.
Discussion
We have demonstrated that brief, millisecond flashes of light have the capacity to change circadian phase in people while they are asleep. Given the relatively small sample size and the sex imbalance in the protocol, it is impossible to rule out an effect of the light on sleep, but we could find no gross objective or subjective changes in sleep that were related to the light exposure during sleep. Light as a therapeutic intervention for circadian-based sleep and medical disorders (“phototherapy”) is most commonly prescribed to occur before normal wake time or after normal bed time (Morgenthaler et al., 2007). While highly efficacious in theory, this type of therapy has poor effectiveness because compliance is low and, when compliance is adhered to, the therapy leads to chronic sleep deprivation (Bjorvatn and Pallesen, 2009; Barion and Zee, 2007). Our demonstration of effective light treatment during sleep without gross interference of sleep opens new avenues of therapy because the light treatment is nonintrusive and requires no change in behavior from the recipient. This could lead to novel treatments for highly prevalent disorders, such as delayed sleep phase syndrome (adolescents), shift work sleep-wake disorder, and advanced sleep phase syndrome (senescence), or the nuisance of jet lag. Because phase-delaying light is normally administered in the evening prior to sleep, this type of light could be used to supplement this light exposure (i.e., continue such light exposure into the hours of sleep) or to allow for subjects to go to sleep at an earlier circadian phase, such as might be necessary when adapting for particular shift schedules. A recent study examining the use of light pulses 1000 times longer (2 sec) confirmed the possibility that light administration during sleep has a significant impact on circadian physiology without disrupting sleep (Figueiro et al., 2013). It remains to be determined whether application of light flashes at all times of the sleep cycle would have equally minimal effects on sleep. Future work on the administration of light flashes during sleep should focus on further confirming these findings and optimization, in terms of spectral content of the flashes (i.e., which photoreceptors are responsible for converting the light flashes to a neural signal), spacing of flashes, and flash duration such that the sequence should be primed to produce the maximal shift with minimal time. Future work should better address whether there is any impact on sleep and the extent of this impact. Our relatively small sample size did not allow us to explore all possible changes in sleep without the risk of type II error, and there are many other analyses that could be used to examine possible changes in sleep. As there is an ever-increasing number of recognized circadian-related morbidities, this type of passive intervention could be of great societal benefit.
Footnotes
Acknowledgements
The authors thank Mr. Ban Ku, Ms. Chun-Ping Liao, and Mr. Daniel East for help in data collection. This work was supported by the National Heart Lung and Blood Institute (1R01HL108441-01); Air Force Office of Scientific Research (F2-4506); and Department of Veterans Affairs Mental Illness Research, Education, and Clinical Center.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
