Abstract
Recently, we reported differences in the expression pattern of the blue light-sensitive flavoprotein cryptochrome (CRY) and the neuropeptide pigment-dispersing factor (PDF) in the neuronal clock network of high-latitude Drosophila species, belonging to the Drosophila subgenus (virilis-repleta radiation), compared with cosmopolitan D. melanogaster flies, belonging to the Sophophora subgenus. Alterations in rhythmic patterns of activity due to these differences might have adaptive significance for colonizing high-latitude habitats and, hence, adjusting to long photoperiods. Here, we show that these differing CRY/PDF expression patterns are only present in those species of the virilis-repleta radiation that colonized high latitudes. The cosmopolitan species D. mercatorum and D. hydei have a D. melanogaster-like clock network and behavior despite belonging to the virilis-repleta radiation. Similarly, 2 species of the holotropical Zaprionus genus, more closely related to the Drosophila subgenus than to the Sophophora subgenus, retain a D. melanogaster-like clock network and rhythmic behavior. We therefore suggest that the D. melanogaster-like clock network is the “ancestral fly clock phenotype” and that alterations in the CRY/PDF clock neurochemistry have allowed some species of the virilis-repleta radiation to colonize high-latitude environments.
Keywords
The Drosophilidae dipteran family accounts for about 4,000 described species, which differ in morphology, behavior, ecology, and biogeography (Brake and Bächli, 2008; O’Grady and DeSalle, 2018). Drosophilidae are thought to have originated in the tropics about 50 million years ago (Markow and O’Grady, 2007; Obbard et al., 2012; O’Grady and DeSalle, 2018; Russo et al., 2013; Throckmorton, 1975) and to have subsequently colonized virtually all environments (Fig. 1A). These elements make the Drosophilidae family an unprecedented model for studying the evolutionary processes that underlie species adaptation to different environments (Adrion et al., 2015). Studies on adaptation to spatially varying environments carried out in Drosophila report correlations between latitude and morphological traits, such as body size (James et al., 1997), wing morphology (Haas and Tolley, 1998), latitude and behavior (for example, egg laying; Allemand and David, 1976), and eclosion rhythm (Lankinen et al., 1986). The idea underlying these studies is that morphological and behavioral traits vary to maximize fitness (Birch, 2016).
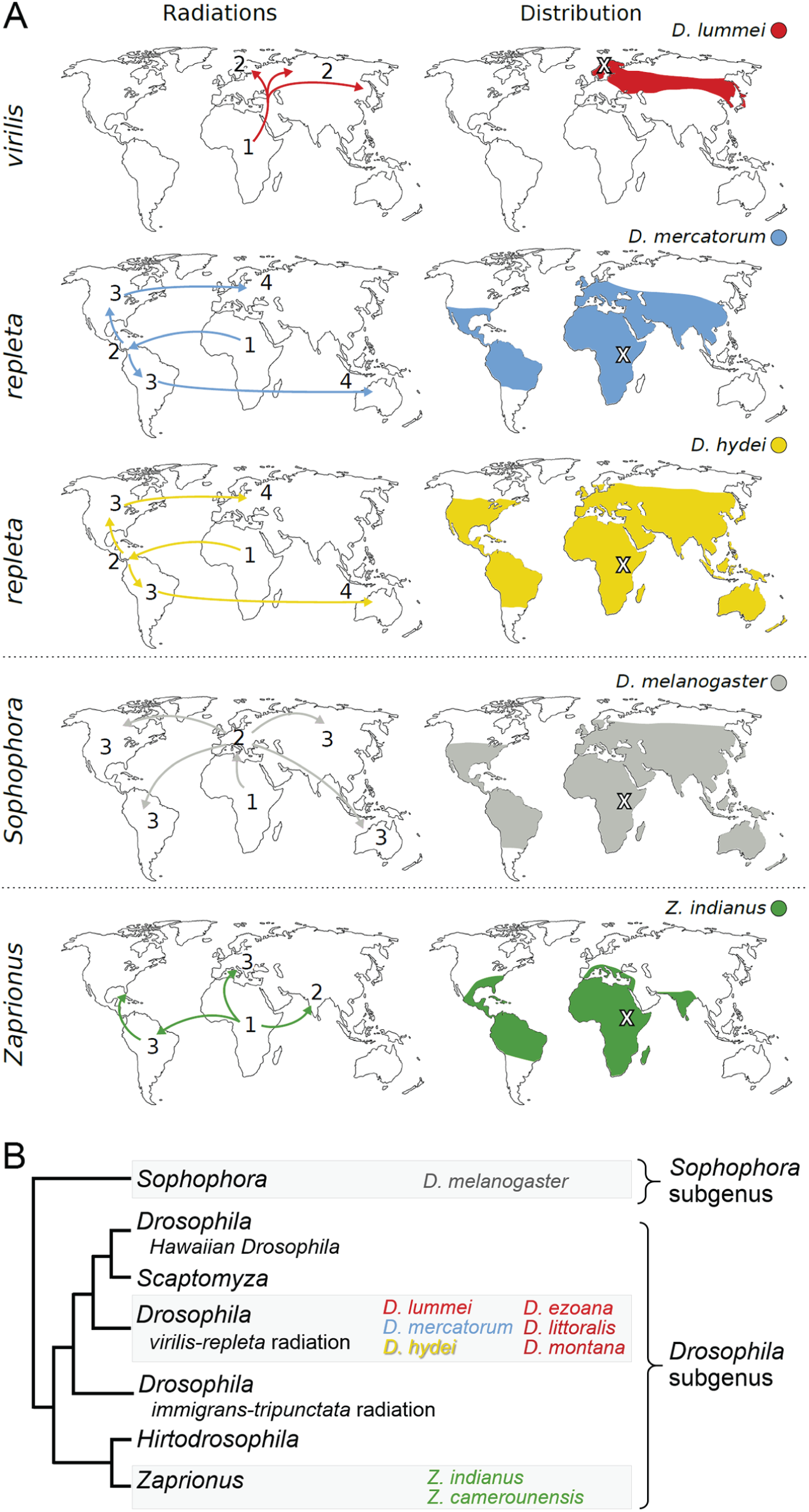
Phylogeny and biogeography of selected Drosophila species. (A) Radiations and distribution of the species studied here. Numbered arrows (left maps) illustrate the order in which the species radiated from their ancestors. Colored areas (right maps) depict the current geographical distribution of the species (based on data from www.taxodros.uzh.ch; G. Baechli). Crosses (right maps) indicate the collection sites of our experimental species. D. melanogaster, originated in Africa and subsequently colonized Europe, the new World, and became a cosmopolitan species. It can be found virtually anywhere but it is unable to overwinter at high latitudes. D. hydei and D. mercatorum underwent a similar radiation as D. melanogaster and are respectively cosmopolitan species, whereas the high-latitude species D. lummei uniquely inhabits northern Europe and Asia. In contrast, Z. indianus (and Z. camerounensis) inhabit mainly tropical regions and are therefore considered as holotropical (Morrone, 2015). (B) Simplified phylogenetic tree illustrating the relationship between the species under investigation (adapted from O’Grady and Markow, 2009). D. lummei is closely related to D. hydei and D. mercatorum. These 3 species, together with the previously investigated species D. ezoana, D. littoralis and D. montana (Kauranen et al., 2012; Kauranen et al., 2016; Menegazzi et al., 2017), belong to the virilis-repleta radiation within the Drosophila subgenus. They are distantly related to D. melanogaster, a member of the Sophophora subgenus whose separation from the Drosophila subgenus took place more than 30 million years ago (Obbard et al., 2012; Russo et al., 2013; van der Linde et al., 2010). The Zaprionus species also belongs to the Drosophila subgenus and is therefore more closely related to the flies of the virilis-repleta radiation than to D. melanogaster.
The ability to anticipate rather than to merely react to daily changes in light and temperature also contributes to fitness (Beaver et al., 2001; Emerson et al., 2008). Living organisms achieve this anticipation thanks to a timekeeping system, the circadian clock, which allows specific biological activities to occur at precise times of day (Daan, 2010). A number of chronobiology studies have investigated the impact of latitude on the circadian clock of phylogenetically distant Drosophila species (Bahn et al., 2007; Hermann et al., 2012; Kauranen et al., 2012; Kauranen et al., 2016; Menegazzi et al., 2017). They reported differences in clock network neuroanatomy and locomotor activity between D. melanogaster and distantly related Drosophila species. In particular, high-latitude species belonging to the virilis-repleta radiation lack the blue-light sensitive flavoprotein cryptochrome (CRY) in the large lateral ventral neurons (l-LNvs), which are responsible for light input and arousal (summarized in Helfrich-Förster, 2017). In addition, the species lack the neuropeptide pigment-dispersing factor (PDF) in the small lateral ventral neurons (s-LNvs), which are responsible for the flies’ morning activity (summarized in Yoshii et al., 2012). Instead, abundant and dense PDF arborizations originating from the l-LNvs are found in the central brain and close to the lateral dorsal neurons (LNds) that control evening activity (Menegazzi et al., 2017). Regarding locomotor activity rhythms, the morning activity in high-latitude species is low to absent, whereas evening activity is delayed and prolonged until dusk on long days (Bahn et al., 2007; Kauranen et al., 2012; Kauranen et al., 2016; Menegazzi et al., 2017). In marked contrast, D. melanogaster flies exhibit morning activity followed by a deep siesta and a pronounced and short evening activity starting and ending before dusk under long-day conditions. Under constant illumination, typical of northern summers, D. melanogaster flies become arrhythmic, whereas high-latitude species retain some rhythmicity (Kauranen et al., 2012; Menegazzi et al., 2017).
In D. melanogaster, CRY is a circadian photoreceptor that induces, upon light activation, degradation of the clock protein Timeless (TIM) and, by doing so, synchronizes the clock to LD cycles (Ceriani et al., 1999; Emery et al., 1998; Emery et al., 2000). Constant illumination consequently leads to a progressive degradation of TIM and, eventually, to arrhythmic behavior. PDF, on the other hand, delays evening activity by inhibiting the electrical activity of the evening neurons (LNds) (Liang et al., 2016; 2017); there are first indications that, under long summer days, these phase-delaying effects come from PDF in the l-LNvs (Schlichting et al., 2016). The current model thus identifies the late and broad evening activity of high-latitude Drosophila species to be provoked by the extra l-LNvs arborizations in the central brain, and the partial rhythmicity observed under constant light to be dependent on the absence of CRY in the same neurons (Menegazzi et al., 2017). Furthermore, the strongly reduced morning activity is caused by the absence of PDF in the s-LNvs.
Based on this description, it is nevertheless not possible to discern whether these differences result from adaptation to a natural environment or are characteristic traits of the species (Kyriacou, 2017; Menegazzi et al., 2017). All high-latitude Drosophila species investigated to date belong to the Drosophila subgenus (virilis-repleta radiation), which separated from the Sophophora subgenus—to which D. melanogaster belongs—more than 30 million years ago (Fig. 1B; Obbard et al., 2012; Russo et al., 2013; van der Linde et al., 2010). Thus, the observed traits may not be of adaptive significance at high latitudes but simply a phylogenetic feature. In this scenario, such traits would be retained across species within the virilis-repleta radiation (Fig. 1B). To tackle this issue, we compared the neuronal clock network and the locomotor activity rhythms of other species belonging to the virilis-repleta radiation but having different geographical distributions (D. lummei, D. mercatorum and D. hydei). D. mercatorum and D. hydei have a similar cosmopolitan distribution as D. melanogaster, whereas D. lummei is restricted to high latitudes (Fig. 1A). In addition, we investigated 2 species of the Zaprionus species group (Z. indianus and Z. camerounensis) that also separated more than 30 million years ago from the Sophophora subgenus (Russo et al., 2013; van der Linde et al., 2010) but remained holotropical (Fig. 1A). To analyze the neurochemistry of the clock circuit, we focused on the aforementioned molecular components of the cellular clock network, PDF and CRY. To analyze rhythmicity in their behavior, we recorded locomotor activity of the flies under different day lengths and under constant light and darkness.
Materials and Methods
Fly Species and Rearing
The experimental species were caught in the wild at two different sites: the high-latitude species D. lummei was caught in Finland near the Oulanka research station Kuusamo (65°57’ N, 29°10’ E), and the cosmopolitan species, D. mercatorum and D. hydei, were caught in Tanzania, near the Nkweseko research station, Machame (3°11’ S, 37°14’ E). The holotropic species, Z. indianus and Z. camerounensis, were acquired from the Drosophila Species Stock Center (UC; San Diego). These species stem from the following locations in Africa: Z. indianus, Alexandria, Egypt, 31°13’ N, 29°58’ E; Z. camerounensis, East Usambara Montains Amani, Tanzania, 5°05’ S, 38°40’ E. For comparison, the cosmopolitan species D. melanogaster was investigated in parallel. The strain we used came from the same region in Tanzania as the D. mercatorum and D. hydei strains and was described in Menegazzi et al. (2017). All wild species were caught in 2014 and kept in the laboratory from that time onward. The experiments were performed between 2016 and 2017.
All flies were fed on cornmeal/agar medium supplemented with yeast. D. mercatorum, D. hydei and the Zaprionus species were reared at 25°C, 60% humidity, under LD12:12, whereas D. lummei were reared at 20°C and 60% humidity under LL.
Immunocytochemical Staining
Immunocytochemistry was performed on young male fly brains. For each experimental group, 12 whole flies were fixed in 850 µl of 4% paraformaldehyde, dissolved in phosphate-buffered saline containing 0.5% Triton X-100 (PBST) (3 h for D. mercatorum, D. hydei, Z. indianus, and Z. camerounensis; 3.5 h for D. lummei) and dissected after 3 × 10 min washes in PBS. Brains were blocked in 5% normal goat serum (NGS) in PBS overnight at 4°C and then incubated overnight at room temperature with rabbit anti-CRY (dilution 1:1000; Yoshii et al., 2008) or mouse anti-PDF (dilution 1:2000; Developmental Studies Hybridoma Bank (DHSB), Iowa USA) diluted in PBST with 5% NGS and 0.05% NaN3. After 5 × 10 min washes in PBST, brains were incubated with goat anti-rabbit Alexa Fluor 488 or goat anti-mouse Alexa Fluor 635 (Molecular Probes; Carlsbad, CA) diluted 1:200 in PBST containing 5% NGS for 3 h at room temperature. Brains were then washed and mounted on glass slides using Vectashield mounting medium (Vector Laboratories; Burlingame, CA). Imaging was performed using a laser scanning confocal microscope (TCS SP8; Leica, Wetzlar, Germany), with appropriate laser diodes to excite the secondary antibody fluorophore at 488 nm and 635 nm for stacks of 2-μm-thick images. Images were processed using Fiji, an open-source platform for biological-image analysis (Schindelin et al., 2012).
Monitoring Locomotor Activity
Behavioral experiments were conducted on young male flies at 20°C. Individual flies were confined to glass tubes containing an agar and water-based food source and placed in Drosophila activity monitors (DAM, Trikinetics; Waltham, MA). Laser beam crosses were considered to reflect a single activity bout, and these counts were recorded at the resolution of 1 min. For all photoperiodic regimes, flies were exposed to artificial LD cycles (white light at 100 lux) with gradual transitions from light to darkness, and vice-versa, in the time span of 1.5 h, as described in Rieger et al. (2007). To test behavioral responses to progressively increasing photoperiods, we employed the following routine: LD 12:12 on days 1 to 6; LD 16:08 on days 7 to 12, and LD 20:04 on days 13 to 18 at constant 20°C. To test behavioral responses to constant conditions, we first entrained flies to LD 12:12 for 6 days and then exposed them to constant darkness (DD) or constant light (LL, 100 lux) for 10 days at constant 20°C. Individual fly activity across a 24-h period was visualized and represented in double-plotted actograms generated using ActogramJ (Schmid et al., 2011). Average activity profiles were obtained by averaging across 3 LD cycles of recording for flies that survived the entire photoperiod. Activity in the average activity profiles was plotted in 15-min bins. Rhythmicity of individual flies was determined using the Chi square periodogram analysis tool implemented in ActogramJ, over 10 days of constant conditions, in combination with visual inspection of the activity. Since many flies showed very low activity levels/amounts, we performed the periodogram analysis over locomotor activity data binned into 30-min intervals. Morning (M) and evening (E) activity maxima were manually/visually determined for individual flies, as described in Schlichting and Helfrich-Förster (2015).
Statistical Analysis
The number of rhythmic and arrhythmic flies in DD and LL was statistically compared by the χ2 test. The free-running periods under these conditions were compared by a 2-way ANOVA followed by a Tukey multiple comparisons post-hoc test (Systat 11). The phase angles between M and E activity peaks under LD cycles with increasing photoperiod were compared by a one-way ANOVA followed by a Tukey multiple comparisons post-hoc test.
Results
Clock Neuroarchitecture in Different Species
In all investigated fly species, PDF staining was present in the l-LNvs and in their projections to the ipsilateral medulla and via the posterior optic commissure (POC) to the contralateral medulla (Fig. 2). As described previously for the other high-latitude species of the virilis group (Hermann et al., 2013; Menegazzi et al., 2017), in D. lummei, supplemental projections of the l-LNvs extended into the central brain (arrowheads in Fig. 2A) and additional PDF-positive neurons were found in the dorsal protocerebrum (asterisks in Fig. 2A). No PDF was visible in the s-LNvs (Fig. 2, B and D) and no CRY was present in the l-LNvs (Fig. 2, C-D). In contrast, we found PDF in the s-LNvs and their projections into the dorsal protocerebrum, and CRY in the s-LNvs and l-LNvs in the cosmopolitan and holotropical species D. hydei, D. mercatorum, Z. indianus, and Z. camerounensis (Fig. 2, E-T), exactly as reported for D. melanogaster. The PDF staining profile of D. hydei closely resembled that of D. melanogaster (Fig. 2I): the s-LNvs and l-LNvs were the only PDF-positive neurons in the brain and their projections were of similar shape and density as those described for D. melanogaster. D. mercatorum possessed additional PDF-positive neurons in the central brain (asterisks in Fig. 2B), the projections of the s-LNvs in the dorsal protocerebrum were “less bent”, and the PDF-positive network on the medulla was denser than that in D. melanogaster and D. hydei. Similarly, the two Zaprionus species possessed a dense network of PDF fibers on the medulla and had less-bent dorsal projections of the s-LNvs, but no additional PDF-positive neurons or fibers in the central brain (Fig. 2, M and Q).
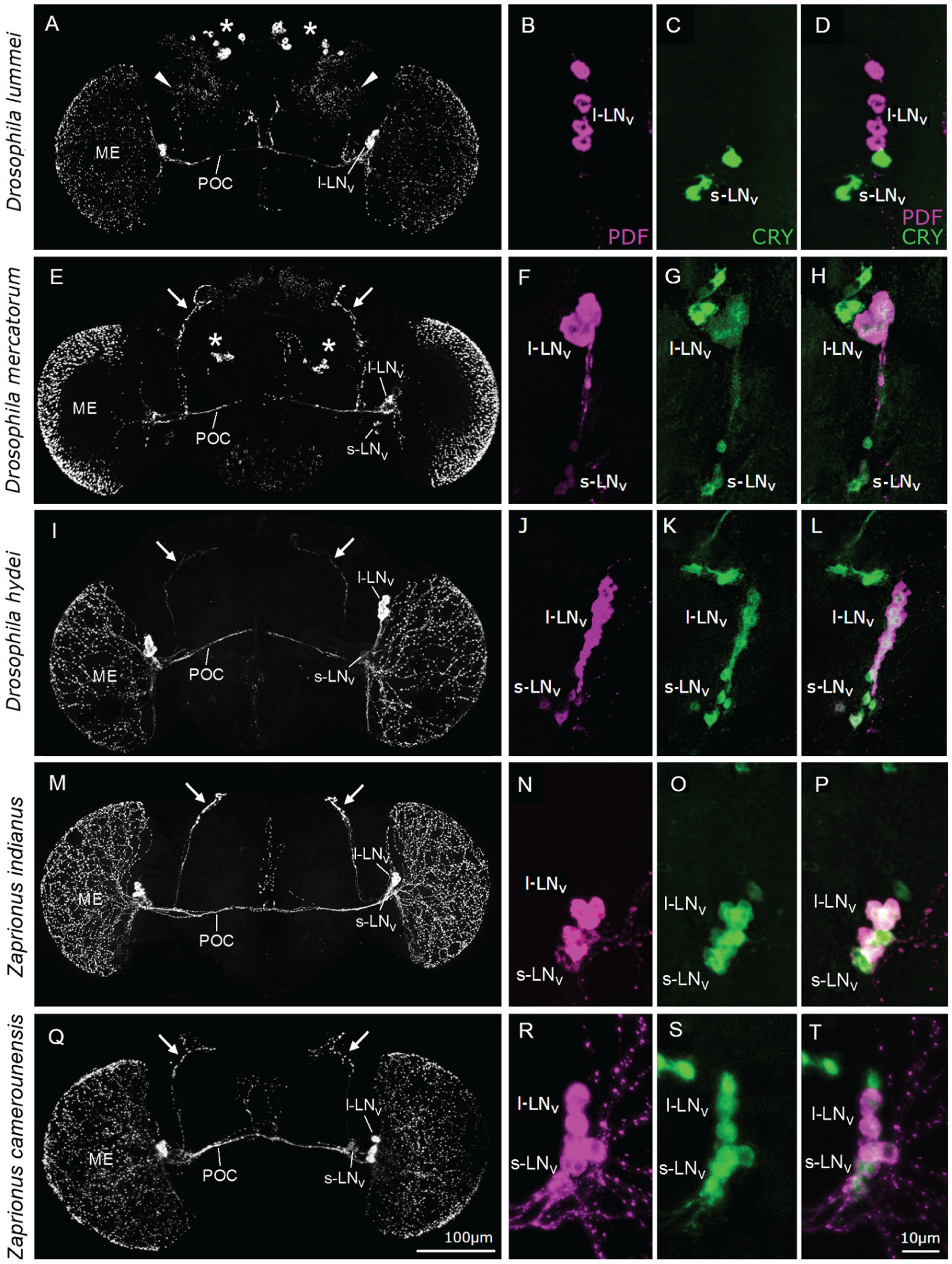
Localization of the neuropeptide PDF and the photopigment CRY in the cells of the clock network of D. lummei, D. mercatorum, D. hydei, Z. indianus and Z. camerounensis. PDF is shown in magenta and CRY in green. Images on the left (A, E, I, M, Q) represent full-brain PDF staining, whereas the colored images on the right provide a magnified view of the lateral neurons (l-LNvs and s-LNvs) double-labelled with anti-PDF (B, F, J, N, R) and anti-CRY (C, G, K, O, S), and the overlay of PDF and CRY (D, H, L, P, T). (A) In D. lummei, PDF is expressed only in the l-LNv and their projections into the medulla (ME), via the posterior optic commissure (POC) into the other brain hemisphere and in projections into the lateral central brain (arrowheads). Additional PDF staining is seen in numerous dorsal and central brain neurosecretory cells (asterisks), which are not part of the clock network. (B-D) PDF-CRY double-labelling shows PDF in the l-LNvs (B) and CRY (D) in the s-LNvs, with no overlap between the 2 stainings (D). (E) In D. mercatorum, PDF is present in the l-LNvs and s-LNvs and in a few additional PDF neurons in the central brain (asterisks). Weakly stained fibers arising from the additional PDF neurons are present in the middle and dorsal brain. The s-LNvs show characteristic projections into the dorsal brain (arrows), whereas the l-LNvs project densely on the medulla (ME). The most anterior network on the medulla is cut off in this confocal stack and the medulla is therefore not completely stained. (F-H) PDF-CRY double-labelling shows PDF and CRY in the l-LNvs and in the s-LNvs. (I) The PDF staining pattern of D. hydei is almost identical to that of D. melanogaster, with PDF and CRY present in the l-LNvs and in the s-LNvs (J-L). (M-Q) PDF and CRY staining of the two Zaprionus species was also almost identical to that of D. melanogaster, except for a denser network of PDF fibers on the medulla (ME in M and Q).
Rhythmicity under Constant Conditions
To study the free-running properties of the circadian clock, we recorded the locomotor activities of flies exposed to DD and LL following entrainment to LD 12:12 at 20°C (Fig. 3A). D. lummei flies showed almost no rhythmic activity under DD (only 12.5% of flies were rhythmic) but were significantly more rhythmic under LL (62.5% of flies were rhythmic) (χ2 = 17.07, df = 1, P < 0.001) (Fig. 3B). In contrast, D. mercatorum and D. hydei were highly rhythmic under DD (100% rhythmic) but largely arrhythmic under LL (12.5% and 20% of the flies were rhythmic per group, respectively) (Fig. 3B). The difference between rhythmicity under DD and LL was highly significant in both cases (χ2 > 41.77, df = 1, P < 0.001). Similarly, the two Zaprionus species were significantly more rhythmic in DD than in LL (Z. indianus: χ2 = 10.24, df = 1, P = 0.001; Z. camerounensis: χ2 = 25.77, df = 1, P < 0.001), although differences in rhythmicity between DD and LL appeared less pronounced than in D. mercatorum and D. hydei (Fig. 3B). In summary, the rhythmic behavior of the cosmopolitan and holotropical flies in DD and LL largely resembled that of D. melanogaster (Fig. 3B), whereas the rhythmic behavior of the high-latitude species D. lummei was similar to that reported for other high-latitude species (Kauranen et al., 2012; Menegazzi et al., 2017; Vaze and Helfrich-Förster, 2016).
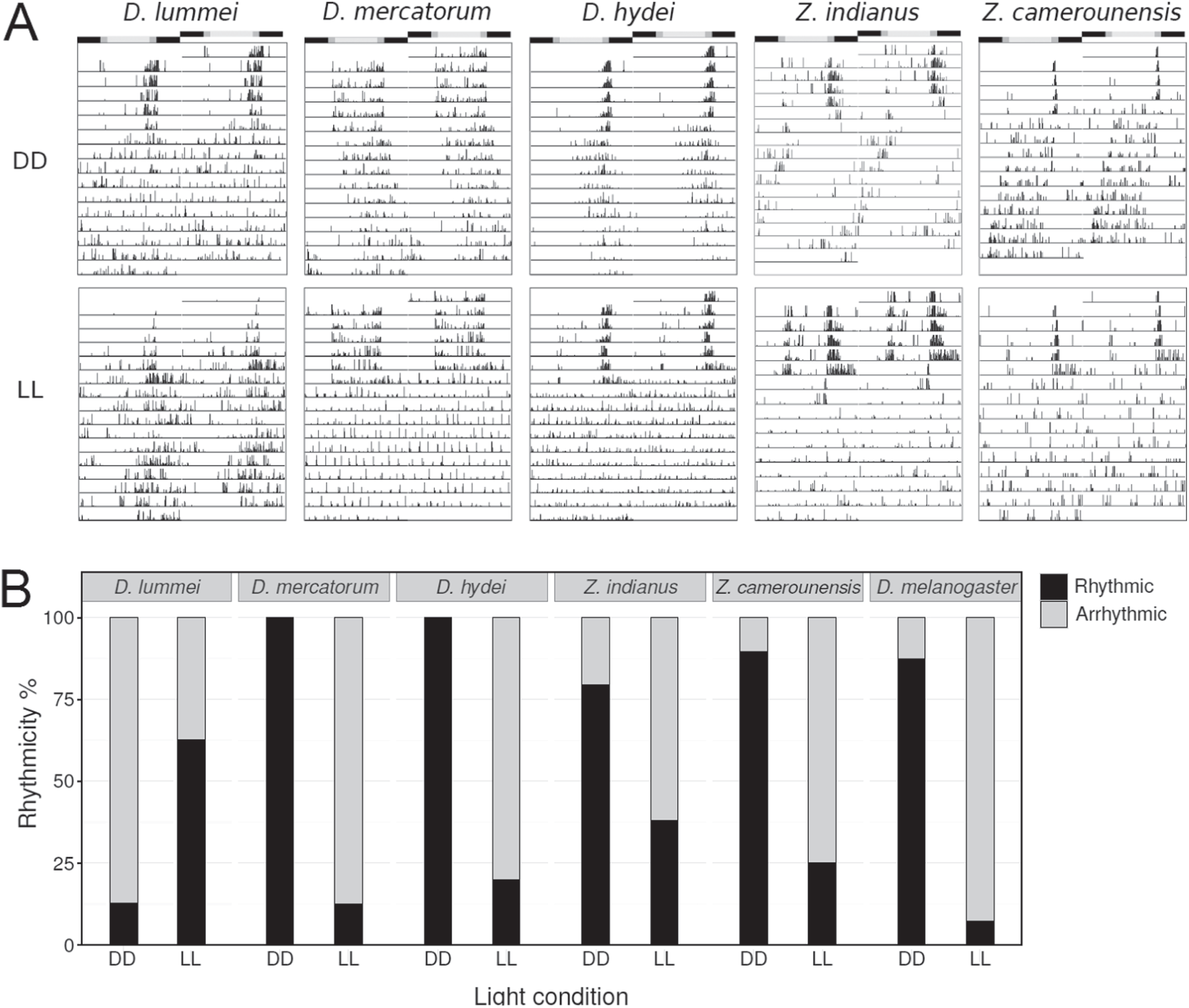
Locomotor activity of flies released in constant conditions. (A) Representative actograms of individual flies (one for each species) exposed to constant darkness (DD) or constant light (LL) after entrainment to LD 12:12. In the top 5 actograms, D. mercatorum, D. hydei, Z. indianus, and Z. camerounensis maintain a rhythmic activity under DD, whereas D. lummei does not. Conversely, D. mercatorum, D. hydei, Z. indianus, and Z. camerounensis are arrhythmic in LL whereas D. lummei maintains rhythmicity partly in this condition. (B) The quantification of these phenomena is pictured in the bar charts expressing the percentage of rhythmic and arrhythmic flies under DD and LL.
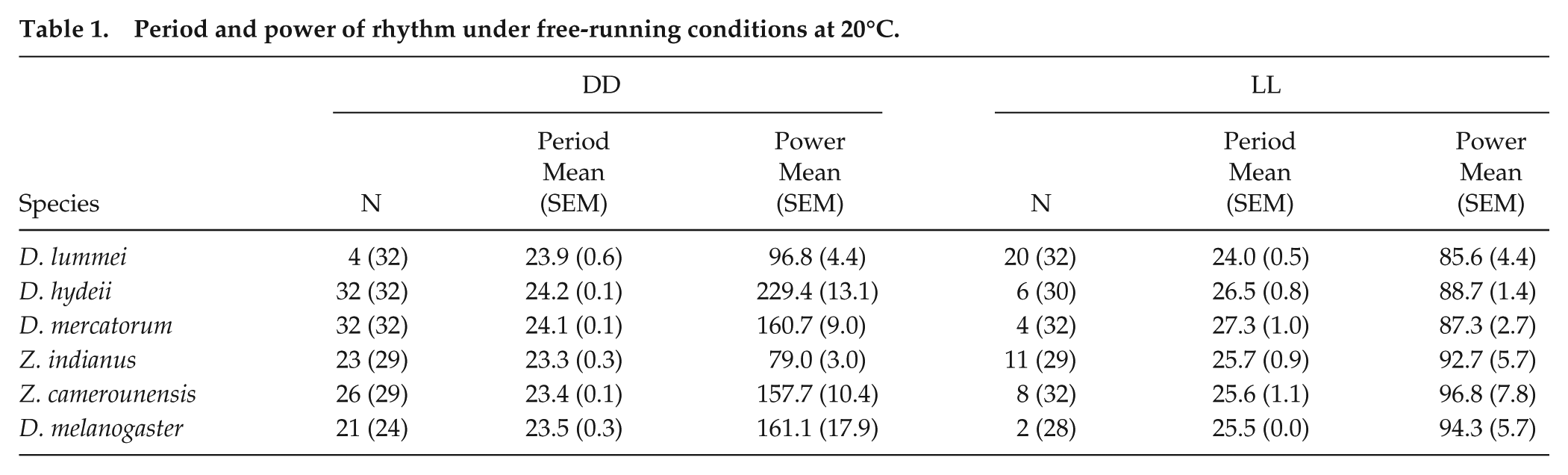
A 2-way ANOVA revealed that the free-running period of flies strongly depended on the light condition (F(1,177) = 41.47; P < 0.001) and on the species (F(5,177) = 3.02; P = 0.012) without any interaction between the two factors (F(5,177) = 1.64; P = 0.152). Tukey’s multiple comparisons post-hoc test showed that, in D. lummei, the free-running period of the rhythmic flies was not significantly different between DD and LL (P = 1.0), whereas, in the holotropical and cosmopolitan flies, period was always significantly longer in LL than in DD (P < 0.05). The corresponding rhythm powers were lower in LL than in DD in most cases, with the exception of D. lummei and Z. indianus flies. In D. lummei, power was rather low under both conditions and Z. indianus showed an unusual low-power rhythm in DD (Table 1). The low-power rhythm of Z. indianus may be caused by the rather low activity of these flies, which is consistent with an earlier report about their activity rhythm under semi-natural conditions (Prabhakaran and Sheeba, 2013).
Period and power of rhythm under free-running conditions at 20°C.
Locomotor Activity under Increasingly Longer Photoperiods
To test how flies respond to increasing photoperiods, we calculated average day activity profiles and determined the phases of morning (M) and evening (E) activity peaks. We were interested in observing the extent to which flies could adjust their morning and evening activity bouts to dawn and dusk. We hypothesized that the holotropical and cosmopolitan species would do less well than the high-latitude species D. lummei because they do not experience extremely long photoperiods in nature and because they differ in clock neuron anatomy (Fig. 2).
The average activity profiles revealed that all species exhibited M and E activity bouts (Fig. 4). Except for the two Zaprionus species, which were almost equally active in the morning and evening, the others exhibited most activity in the evening. An additional peculiarity of the two Zaprionus species was that they responded with much higher activity to the dawn and dusk simulations than the other species. This became most evident at LD 20:04, where the major M and E activity bouts occurred later and earlier, respectively, than dawn and dusk (Fig. 4).
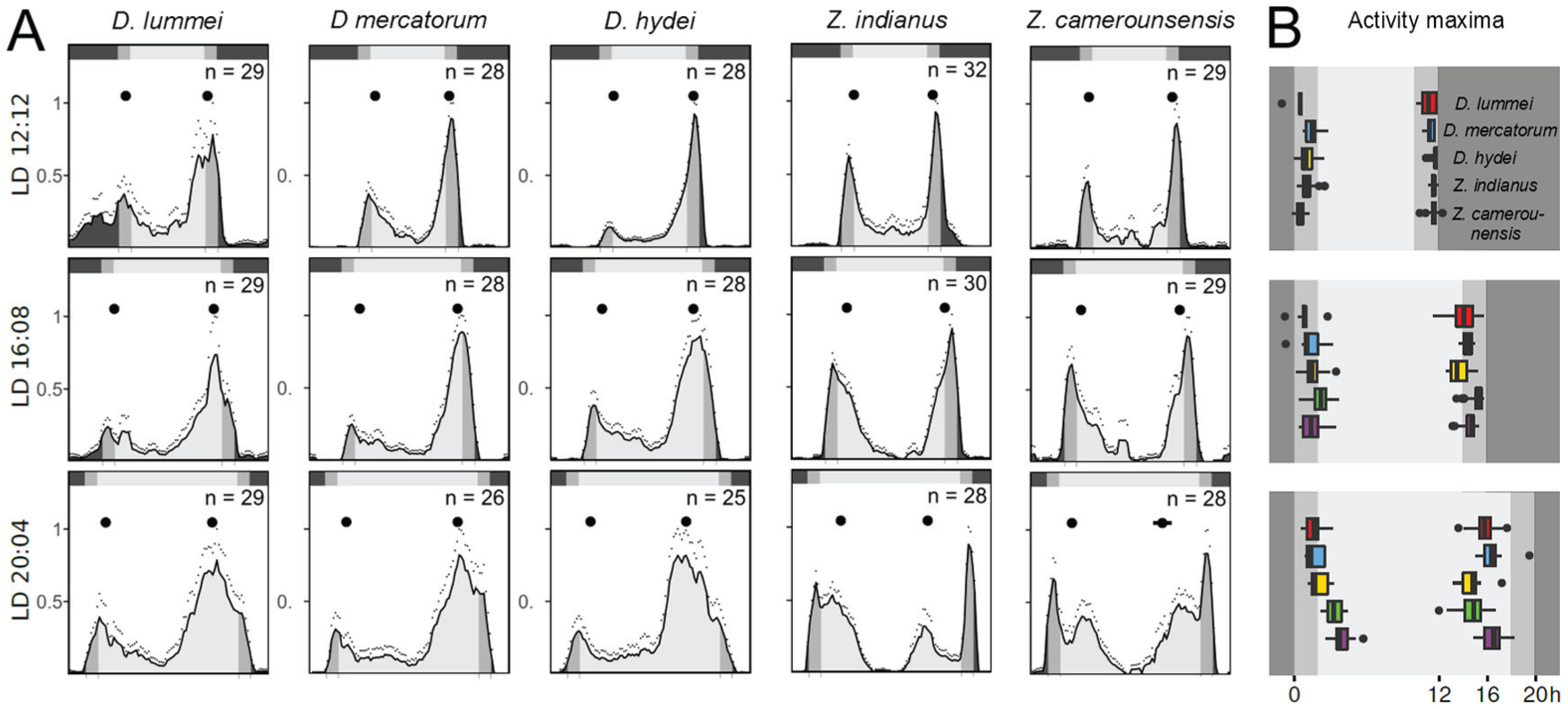
Average daily activity profiles and calculated phases of morning (M) and evening (E) peaks. (A) Average activity profiles of flies recorded under a 12-h photoperiod (LD 12:12) followed by a 16-h photoperiod (LD 16:08) and then a 20-h photoperiod (LD 20:04). All LD cycles included a simulation of dawn and dusk (see Rieger et al., 2007). Standard errors of the mean are plotted as small dots above the bars. The reference top bar indicates the light regime; i.e. dark phase (black), twilight phases (grey), and light phase (light grey). Average M and E peaks are plotted as large dots below the reference top bar. (B) Whisker box plots illustrating the timing of M and E peaks for all species across the 3 photoperiods.
As the photoperiod extended from LD 12:12 to LD 20:04, flies broadened the phase angle between morning and evening activity; however, the extent to which this occurred varied among the species. Under LD 12:12 and LD 16:08, locomotor activity peaked around dawn and dusk, with minor differences among species. However, under LD 20:04, M and E peaks no longer coincided with dawn and dusk but instead occurred within the light phase. Hereby, the phase angle between the M and E peak was dependent on the species (ANOVA: F(4,91) = 26.54, P < 0.001). D. mercatorum and, even more so, D. lummei flies appeared to be the best at tracking dawn and dusk; D. lummei flies displayed the largest phase angle between M and E peaks. Tukey’s multiple comparisons post-hoc test showed that this was significantly larger than that for D. hydei, Z. indianus and Z. camerounensis (P < 0.001) but not significantly larger than that for D. mercatorum (P = 0.482; Fig. 4, right panel).
Discussion
Our results show that the clock architecture and the locomotor activity patterns are different among phylogenetically related flies living at different latitudes but similar among distantly related species living at the same latitude. These findings expand a picture so far based on experiments that only involved distantly related species inhabiting different latitudes (Menegazzi et al., 2017).
Clock Neuroarchitecture Is Similar in Flies Living in the Same Environment
In the high-latitude species D. lummei, we observed a parceling of PDF and CRY expression in the LNvs: whereas PDF was only present in the l-LNvs, CRY was only expressed in the s-LNvs. This is in line with previous reports using other high-latitude species (Hermann et al., 2013; Kauranen et al., 2012; Menegazzi et al., 2017). In addition, PDF fibers stemming from the l-LNvs extended into the central brain. In contrast, the cosmopolitan and holotropical species possessed both CRY and PDF in all LNvs and no extra PDF in the central brain, resembling the expression pattern of the cosmopolitan D. melanogaster. Because this expression was seen in flies from the Drosophila and Sophophora subgenera, it may represent the ancient clock phenotype.
Rhythmic Behavior Is Similar in Flies Living in the Same Environment
Under LL conditions, most cosmopolitan and holotropical species became arrhythmic, whereas most of the flies of the high-latitude species, D. lummei, remained rhythmic. This may be explained by the expression of CRY in all clock neurons of the cosmopolitan and holotropical species and its absence from the l-LNvs of D. lummei. In D. melanogaster, CRY activation by light leads to TIM degradation, which first slows down the circadian clock and finally stops it (Busza et al., 2004; Ceriani et al., 1999; Emery et al., 2000; Konopka et al., 1989; Rieger et al., 2006; Rosato et al., 2001).
Consistent with this hypothesis, the few cosmopolitan and holotropical flies that remained rhythmic under LL exhibited long free-running periods, whereas the rhythmic D. lummei flies retained periods close to 24 h in LL (Table 1). The absence of CRY in the l-LNvs of D. lummei might partly prevent the slowing down of the clock and its stopping under LL even though the l-LNvs are not the main circadian pacemaker neurons (see also Discussion in Menegazzi et al., 2017). Most likely, the light-sensing properties of CRY are different in D. lummei and other high-latitude species. We have preliminary results showing that the CRY molecule of several high-latitude species carries specific amino acid changes close to the first tryptophan involved in the photoreduction cascade as well as in the C-terminal tail, which might reduce its light-sensitivity (Deppisch et al., unpublished observations). In any case, all of the high-latitude species investigated to date appear to be at least slightly more rhythmic under LL than the cosmopolitan and holotropical species (Kauranen et al., 2012; Menegazzi et al., 2017); this speaks for an adaptation of their circadian system to the extreme long summer days to which they are exposed in nature. This adaptation appears specific to the high-latitude species and not to the virilis-repleta group.
Reciprocally, rhythmicity under DD conditions appears to be a unique feature of cosmopolitan and holotropic flies, as we found it in D. melanogaster, D. mercatorum, D. hydei, Z. indianus and Z. camerounensis; rhythmicity under DD was not present in the high-latitude species that have been examined to date (present study findings, and Kauranen et al., 2012; Menegazzi et al., 2017; Vaze and Helfrich-Förster, 2016). The reason for the lack of rhythmicity under DD appears to be the lack of PDF in the s-LNvs (Menegazzi et al., 2017), which is reminiscent of the PDF mutants of D. melanogaster or flies with down-regulated PDF in the s-LNvs (Renn et al., 1999; Shafer and Taghert, 2009). Arrhythmic behavior under constant conditions has also been reported for other high-latitude species (Lankinen and Forsman, 2006; Riihimaa and Lankinen, 1997). In the present study, the rhythm power of the D. lummei flies that were rhythmic under DD and LL was rather low, which indicates a weak clock (Table 1); such weak clocks are generally more plastic than strong clocks (Abraham et al., 2010; Vitaterna et al., 2006), which may partly explain why they can easily adapt to extreme changes in day length that prevail in the North (see below).
High-Latitude Species Adjust their Morning (M) and Evening (E) Activity to Dawn and Dusk Better Than Cosmopolitan and Holotropical Species
A key task of endogenous clocks is to adapt activities to different environmental conditions; for example, in response to seasonal changes that are accompanied by different photoperiods. This adaptation can nicely be followed in animals showing bimodal activity patterns, in which the timing of M and E activities correlate with dawn and dusk, with M activity occurring earlier and E activity later under long summer days (Aschoff, 1966; Aschoff and Wever, 1965; Daan et al., 2001; Pittendrigh and Daan, 1976). M and E activity peaks of D. melanogaster flies can also track dawn and dusk (Majercak et al., 1999; Rieger et al., 2003; Shafer et al., 2004); strains collected at high latitudes show better tracking than strains collected at the equator (Menegazzi et al., 2017; Rieger et al., 2012). The E activity bout of high-latitude species, such as D. ezoana and D. littoralis, tracked dusk even on very long days (LD 20:04; Menegazzi et al., 2017); however, the M activity was virtually absent, which correlates to the lack of PDF in the M peak-controlling s-LNvs (Menegazzi et al., 2017). Here, we confirmed that the high-latitude species D. lummei tracked dusk better than most of the cosmopolitan and holotropical species (D. hydei, Z. indianus, and Z. camerounensis). Only D. mercatorum flies seemed to be an exception (see Fig. 4 and below). The late E peak of the high-latitude species at LD 20:04 is thought to be caused by PDF from the l-LNvs that project into the central brain and terminate close to the lateral dorsal neurons (LNds) (Menegazzi et al., 2017; see arrowheads in Fig. 2A). In D. melanogaster, PDF has phase-delaying (decelerating) effects on lateral clock neurons expressing the PDF-receptor and control the E peak (Im and Taghert, 2010; Liang et al., 2016, 2017; Schlichting et al., 2016; Yao and Shafer, 2014). Nevertheless, PDF also has phase-advancing (accelerating) effects in some clock neurons in the dorsal brain (Wülbeck et al., 2008; Yoshii et al., 2009). Most interestingly, the PDF fibres from the l-LNvs extend into the dorsal brain of D. lummei and additional PDF-positive, non-clock neurons are located in this area, making it likely for PDF to be secreted into this area. Unlike the other high-latitude species (e.g., D. ezoana, D. littoralis, and D. montana), D. lummei flies also show M activity that nicely tracks dawn (Fig. 4). Speculatively, other clock neurons have overtaken the role of the s-LNvs in controlling morning activity. Good candidates are specific dorsal clock neurons (DN1ps), which contribute to the control of M activity even in D. melanogaster (Yoshii et al., 2012; Zhang et al., 2010). These DN1ps may be phase-advanced by PDF released into the dorsal brain, and this might explain why M activity can track dawn in D. lummei.
D. mercatorum is the only cosmopolitan species in our study capable of tracking dawn and dusk at LD 20:04 and the only cosmopolitan species to possess PDF-positive, non-clock neurons in the central brain (Fig. 2E). Possibly, these extra PDF-positive neurons release PDF close to the DN1ps and LNds and advance/delay their clock, respectively, so that the flies can adapt M and E activities to long days. Overall, this underlines the role of PDF in behavioral adaptation to long photoperiods (see also Yoshii et al., 2009).
Footnotes
Acknowledgements
We thank Lilli Bähr and Hannele Kauranen for collecting flies in Finland and members of the KiLi project (DFG research unit FOR1246) for collecting flies from Tanzania. We are grateful to Takeshi Todo (Osaka University, Osaka, Japan) for the CRY antibody and to Gerhard Bächli, David Grimaldi and Pekka Lankinen for help with determining the identity of the caught species. This study was initiated by the SFB 1047 “Insect Timing”, Project A1 and C2 (German Research Foundation (DFG)) and further funded by DFG grants FO 207-15 to C.H.F and ME 4866-1 to P.M.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
