Abstract
The circadian clock of social insects has become a focal point of interest for research, as social insects show complex forms of timed behavior and organization within their colonies. These behaviors include brood care, nest maintenance, foraging, swarming, defense, and many other tasks, of which several require social synchronization and accurate timing. Ants of the genus Camponotus have been shown to display a variety of daily timed behaviors such as the emergence of males from the nest, foraging, and relocation of brood. Nevertheless, circadian rhythms of isolated individuals have been studied in few ant species, and the circadian clock network in the brain that governs such behaviors remains completely uncharacterized. Here we show that isolated minor workers of Camponotus floridanus exhibit temperature overcompensated free-running locomotor activity rhythms under constant darkness. Under light-dark cycles, most animals are active during day and night, with a slight preference for the night. On the neurobiological level, we show that distinct cell groups in the lateral and dorsal brain of minor workers of C. floridanus are immunostained with an antibody against the clock protein Period (PER) and a lateral group additionally with an antibody against the neuropeptide pigment-dispersing factor (PDF). PER abundance oscillates in a daily manner, and PDF-positive neurites invade most parts of the brain, suggesting that the PER/PDF-positive neurons are bona fide clock neurons that transfer rhythmic signals into the relevant brain areas controlling rhythmic behavior.
Keywords
Colonies of most social insects, like those of the carpenter ant Camponotus floridanus (Buckley), show division of labor in the form of temporal and physical castes (Hölldobler and Wilson, 1990; Endler et al., 2005). Apart from the reproductive caste composed of females and males, the occurrence of different, sterile worker castes may depend on age and body size, or a combination of both, for instance in the ants Solenopsis invicta (Tschinkel, 2006) and Atta sexdens (Wilson, 1980). In the ant C. floridanus, two distinct morphological castes have been found that seem to have a different bias toward becoming foragers (Simola et al., 2016). Whereas most foraging and scouting activities are executed by minor workers, major workers do not scout and hardly forage. The tendency to forage increases in both morphological castes with age and is thus dependent on both morphology and age. Most foraging takes place during the dark period, when ants are subjected to a 12:12-h light-dark cycle (LD) (Simola et al., 2016). Although it has not been shown yet whether this behavior persists in constant conditions, other forms of daily and circadian behavior have been described for ants belonging to the genus Camponotus. Workers of Camponotus compressus display caste-specific locomotor rhythms (Sharma et al., 2004); that is, major workers and most media workers are nocturnal under LD regimes, whereas minor workers do not entrain at all. Furthermore, major workers show a single stable circadian rhythm under constant darkness (DD), whereas media workers change the period of their circadian rhythm after several days of DD, and minor workers are not rhythmic at all under constant conditions (Sharma et al., 2004). Nurse workers of the ants Camponotus mus have a circadian preference for different temperatures to place the brood (Roces and Núñez, 1989; Falibene et al., 2016), and the males of Camponotus clarithorax show a preference for emergence from the nest in the early morning hours (McCluskey, 1965). In Camponotus rufipes, both forager and nurse workers exhibit entrainment of circadian rhythms in locomotor activity by a 12:12-h light-dark cycle and free-run under constant conditions (Mildner and Roces, 2017). Moreover, activity rhythms can be entrained by nonphotic zeitgebers such as feeding times, since most foragers showed circadian foraging behavior synchronized with food availability, at either daytime or nighttime (Mildner and Roces, 2017). In addition, C. rufipes workers have the ability to estimate short time intervals while visiting nectar sources and to adjust their visiting times at food patches accordingly (Schilman and Roces, 2003). Finally, workers of some Mediterranean Camponotus ant species shift their activity patterns on a seasonal basis as a response to changing environmental conditions and also if food competitors are present (Cros et al., 1997). This daily asynchrony can have positive effects on the stability of ecosystems (Houadria et al., 2016). Timed, complex behaviors as those described above may be exogenously driven or require an endogenous time-keeping mechanism: for example, a circadian clock. The molecular basis and neuronal pathways comprising such a clock have been studied extensively in the fruit fly Drosophila melanogaster (reviewed in Peschel and Helfrich-Förster, 2011; Helfrich-Förster, 2017) and to some extent in other social hymenopteran species like the fire ant S. invicta and the honey bee Apis mellifera (Rubin et al., 2006; Ingram et al., 2009, 2012). In all these organisms, the PERIOD (PER) protein has been found to be a core player of the translational-transcriptional feedback loop comprising the molecular basis of the clock and to be expressed in the brain (Hall, 1995; Helfrich-Förster, 1995; Rubin et al., 2006; Ingram et al., 2009; Bertossa et al., 2014). PER is a negative factor blocking its own transcription through interaction with its transcription factor, the CLOCK/CYCLE heterodimer, thus leading to a cyclic fluctuation in PER protein levels (Hall, 1995). The neurons in the brain in which PER oscillates are usually called clock neurons, and in fruit flies, they have been shown to be important for controlling behavioral rhythmicity. A neuropeptide that is also connected to the endogenous clock of many arthropods is the pigment-dispersing hormone (PDH) or pigment-dispersing factor (PDF) (Rao, 2001). In fruit flies and honey bees, some PER expressing neurons in the lateral brain coexpress PDF (Helfrich-Förster, 1995; Fuchikawa et al., 2017), but many other insects do not appear to coexpress PER and PDF (Závodská et al., 2003). PDF fibers form a complex network in the brain and the optic lobes that has been described in numerous insects including two bee species (Homberg et al., 1991; Helfrich-Förster and Homberg, 1993; Stengl and Homberg, 1994; Helfrich-Förster, 1997; Závodská et al., 2003; Bloch et al., 2001; 2003; Weiss et al., 2009; Wei et al., 2010; Beer et al., 2018). PDF is thought to mediate light input to the circadian clock neurons in the brain; to communicate between the different clock neurons and adjust their cycling amplitude, period, and phase; and to mediate output from the clock to photoreceptor cells in the eye and to downstream neurons in the central brain (Petri and Stengl, 1997; Peng et al., 2003; Saifullah and Tomioka, 2003; Singaravel et al., 2003; Lin et al., 2004; Yoshii et al., 2009; Schendzielorz et al., 2014; Liang et al., 2016, 2017). In honey bees, the PDF network is extraordinarily widespread and dense, and it appears well suited to mediate time-of-day information to the brain centers that are involved in sleep activity rhythms, time-place memory, and sun-compass orientation (Beer et al., 2018). Ants are central-place foragers like bees. Consequently, ants possess a similarly good time-place memory compared with bees (Schatz et al., 1999), and several ant species rely on sun-compass orientation (Wehner, 1997). However, so far, the neuronal basis of the circadian clock has not been revealed in any ant species.
Here, we recorded locomotor activity rhythms of isolated C. floridanus workers under LD to investigate their entraining abilities as well as under DD to demonstrate the endogenous nature of the clock. Furthermore, workers were monitored under different constant temperatures to demonstrate temperature compensation as another fundamental property of the circadian clock. In addition, we immunostained ant brains with antibodies against PER and PDF to reveal the circadian clock network in the brain and to demonstrate that PER levels in the different clock neurons oscillate in a daily manner.
Material and Methods
Ant Colonies
Minor workers of the species C. floridanus stemming from two colonies (C79 and C152) were used for these studies. Small colonies were collected on the Orchid Islands, Florida, in July 2002 and reared in artificial plaster nests in plastic boxes at 25 °C, an ambient humidity of 50%, and a light-dark cycle of 12h:12h (light onset at 0600 h, light offset at 1800 h). Colonies were provided with water and fed ad libitum with diluted honey and freshly killed cockroaches. At the time of the experiments, 15 years after their collection, each colony still contained the founding queen, approximately 3000 workers, and plenty of brood in all stages. The laboratory nests offered brood chambers and a foraging area, so that workers distributed themselves based on their preferred tasks, and no behavioral biases due to the long rearing in the laboratory were apparent.
Monitoring of Individual Locomotor Rhythms
For preentrainment, individual minor workers of unknown age were collected randomly from colonies C79 and C152 and placed in subcolonies into a temperature-controlled incubator (~60% humidity, light-dark cycle 12 h of light and 12 h of darkness [LD12:12], and constant temperature (either 20, 25, or 30 °C). They were fed ad libitum with 5% sucrose. After at least 7 days of acclimatization to the environmental conditions of the experiment, individual ants were transferred to the recording tubes (100 mm length, 16 mm diameter) of the Locomotor Activity Monitoring System (LAM) available from TriKinetics Inc. (Waltham, MA). As described in previous studies (Sharma et al., 2004; Mildner and Roces 2017), individual Camponotus ant workers show consistent locomotor rhythms over 20 days in single recording tubes, despite isolation. Inside the tubes, ants were fed using either feeding devices developed by Mildner and Roces (2017) or a newly developed feeding device (Fig. 1). Both devices were located at both ends of the tubes, sealing the tube and providing 5% sucrose-solution ad libitum. The sucrose solution had to be refilled every second day in the feeding device of Mildner and Roces (2017) and every fifth day in the new feeding device. Each LAM monitor contained 32 tubes that were equipped with infrared light beams (Fig. 1). Raw data of individual light beam crosses were collected in 1-min bins by the LAM System Collection Software (TriKinetics Inc.). For the first 5 to 8 days, ants were recorded under the same LD12:12 cycle as during preentrainment, and subsequently their activity was monitored for at least 10 more days under DD. Since periodogram analysis requires time series that are longer than 1 week to yield reliable results, only the activity data of ants surviving at least 7 days under DD conditions were analyzed. Actograms were generated using the ActogramJ Plugin (Schmid et al., 2011) in Fiji (Schindelin et al., 2012). ActogramJ was also used to test for rhythmicity and to determine period length under LD and DD (χ2 periodogram analysis). On the basis of actograms and periodogram analysis, animals were classified as diurnal, nocturnal, or arrhythmic. Animals were considered diurnal when they were significantly rhythmic under LD and DD, they were entrained to the LD (period = 24 h), and their free-running activity in DD continued from the light phase (day) of the LD. Animals were considered nocturnal when they were significantly rhythmic under LD and DD, they were entrained to the LD, and their free-running activity continued from the dark phase (night) of the LD. Animals were considered arrhythmic when periodogram analysis revealed no significant rhythmicity under DD. For generating average daily activity profiles for diurnal, nocturnal, and arrhythmic ants, the first raw data of the last 3 days in LD were averaged for each single ant. Thereafter, single activity profiles were averaged across all ants of each group and smoothed by applying a moving average of 11. For all animals, average activity levels (average number of beam crosses per minute) in LD were determined during the entire day as well as during day and night separately.

Device for recording locomotor activity of individual ants. Single ants were kept in glass tubes (16 mm of diameter) in the middle of which (arrow) an infrared light beam recorded their activity (commercial activity recording, TriKinetics). Food was provided at both ends of the tubes through a capsule (b) with small holes for air ventilation for air ventilation that was connected to a Plexiglas reservoir (volume ~5 mL) containing 5% sucrose-solution (a). The ants had access to the sucrose-solution via a sponge (c). The arrow head points to the hole that was used to refill the sugar solution.
Statistical Analysis
The numbers of nocturnal, diurnal, and arrhythmic ants were statistically compared by use of the χ2 test. Effects of colony, temperature, and activity pattern (diurnality, nocturnality, or arrhythmicity) on the period length of the free-running rhythms in DD were evaluated by a 3-way ANOVA followed by a Bonferroni post hoc test after the data had been tested for normal distribution by the Kolmogorov-Smirnov test (SYSTAT Version 11). Similarly, effects of colony, temperature, and activity pattern on activity levels in LD were determined by a 3-way ANOVA followed by a Bonferroni post hoc test.
Immunohistochemical Staining
All staining was performed on whole mount brains.
PER/PDH Double Staining
Nocturnal ants from colony C79 that had been recorded over 6 days under LD12:12 at 25 °C were collected at zeitgeber times (ZT) 0, 3, 6, 9, 12, 15, 18, and 21. At collection, ants were decapitated, a window was cut, and only the head was quickly fixed in 4% paraformaldehyde (PFA) in phosphate-buffered saline (PBS; Merck, Darmstadt, Germany; pH = 7) with 0.1% Triton X-100 (PBST 0.1%) at room temperature in the dark. After 3 h of prefixation, the heads were washed 3 times in PBS for 15 min, and the brains were dissected in PBS. The brains were postfixed in 4% PFA in PBS with 0.5% Triton X-100 (PBST 0.5%) for 45 min more at room temperature and then washed in PBS 3 times for 15 min. Afterward, the brains were dehydrated in 30%, 50%, 70%, and 90% methanol in PBS for 15 min each at room temperature, and finally they were rinsed in 100% methanol 2 more times for 15 min and then stored in methanol at −20 °C at least overnight. For rehydration, the steps were reversed; the brains were rinsed in PBS 3 times for 10 min and transferred to sodium citrate (pH 0 8.5) to incubate for 40 min at 80 °C. After 3 washes in PBS for 10 min at room temperature, the brains were blocked in 5% normal goat serum (NGS) for 2 h at room temperature. This was followed by incubation in primary antibodies (anti-βPDH 1:1000, and anti-amPER 1:1000 in PBST containing 5% NGS and 0.02% NaN3).
The polyclonal anti-βPDH antibody (kindly provided by Heinrich Dircksen) was raised against a glutaraldehyde conjugate of synthetic crab β-pigment-dispersing hormone (βPDH) in rabbits (Dircksen et al., 1987). Anti-βPDH reliably stains the PDF-positive neurons of insects, including honey bees (Homberg et al., 1991; Helfrich-Förster, 1995; Fuchikawa et al., 2017). The polyclonal anti-amPER (kindly provided by Eva Winnebeck) was also raised in rabbits against a large part of crystallized honey bee PER and has been characterized by Fuchikawa et al. (2017). Since both primary antibodies were raised in rabbits, we performed two separate staining procedures, first against PER and then against PDF, with an additional fixation in PFA in between (for details, see Fuchikawa et al., 2017). This additional fixation of the brains denatured the already bound antibodies so that the antibodies applied in the second staining procedure could no longer recognize the PER antibody that had bound in the first staining procedure, allowing us to unequivocally distinguish between PER and PDF staining. The incubation in anti-amPER solution lasted 2 days at room temperature and 5 days at 4 °C. Subsequently, brains were washed 6 times for 10 min in PBST, and the secondary fluorescence-labeled antibody (Alexa Fluor 488, goat-anti rabbit; Molecular Probes at Thermo Fisher Scientific Inc, Germany) was applied overnight at 4 °C (at a 1:200 dilution in PBST containing 5% NGS). After thorough rinsing (every hour for 1 day at room temperature with PBST) and the second round of fixation, anti-βPDH was applied for another 7 days (same procedure as described above). For detecting PDF, Alexa Fluor 635 (goat-anti rabbit) was used (1:200 in PBST containing 5% NGS). After incubation in the secondary antibody, brains were washed 3 times with PBST and 3 times with PBS and subsequently embedded in VectaShield mounting medium (Vector Laboratories, Burlingame, CA) between 2 cover slips so that both the anterior and posterior sides of the brain could be scanned.
PDH/SYN Staining
Ants were taken randomly from colony C152 (entrained to an LD12:12 cycle at 25 °C) and dissected at ZT4. Whole brains were fixed in 4% PFA in PBST for 3 h, washed twice with PBS and twice with PBST at room temperature, and blocked overnight at 4 °C with 5% NGS in PBST. Incubation in the primary antibody followed subsequently at room temperature for 5 days (1:500 rabbit anti-PDF, 1:50 mouse anti-3c11, 5% NGS, 0.02% NaN3 in PBST). The monoclonal antibody 3c11 is directed against Drosophila SYNAPSIN (SYN, provided by the Hofbauer-Buchner Collection of monoclonal antibodies, Würzburg, Germany). SYN is a synaptic protein present in all synaptic terminals, and the antibody against it reliably marks all neuropils of insects (Groh et al., 2004; Hofbauer et al., 2009). After incubation in the primary antibody, brains were washed 6 times for 10 min in PBST, and secondary fluorescence-labeled antibodies (Alexa Fluor 635 [goat anti-rabbit] and Alexa Fluor 488 [goat anti-mouse] at 1:200 in PBST containing 5% NGS) were applied overnight at room temperature. The preparations were washed 3 times in PBST and 3 times in PBS and subsequently mounted in VectaShield as described above.
Microscopy and Image Analysis
Microscopy of immunofluorescent brains was performed with a laser-scanning confocal microscope (Leica TCS SP-8 for PER/PDF, Leica TCS SP-E for PDF/SYN; Wetzler, Germany) and a 10× objective. Confocal stacks of 2-µm thickness were obtained by sequential scanning to prevent bleed-through. For visualizing the location of PER-positive cells and the PDF arborization patterns, Z stacks of merged images were generated in Fiji using maximal intensity. Brightness and contrast were adjusted separately for the 2 channels, and the pictures were overlaid either in Fiji or in Corel Photo-Paint (Corel Corp, Ottawa, Canada).
For quantification of PER staining intensity, the laser settings in the confocal were kept constant, and brightness and contrast of the pictures were not changed at all. The measurement of pixel intensity in the different cell clusters was performed on single confocal sections in Fiji (ImageJ) in 5 hemispheres per time point as described in Fuchikawa et al. (2017). The effect of time on PER staining intensity was calculated by the nonparametric Kruskal-Wallis test for each cell cluster. The number of PER-positive cells per cluster was determined at ZT0 and ZT3.
Results
C. floridanus Ants Are Active during Day and Night with a Preference for the Night
Under the 3 tested temperatures, the great majority of ants entrained to the LD cycle and were rhythmic under DD. Nevertheless, at 20 °C, most ants died before the transfer to DD, and the few that allowed an assessment of rhythmicity lived only for 2 to 5 days in DD. Therefore, we had to exclude the experiment at 20 °C from further analysis. At 25 and 30 °C, most ants survived until the end of the experiment. In the majority of ants (60%-80%) the free-running rhythm started from the previous dark phase, which indicates that they were nocturnal (Fig. 2, A and D). Still, most of these ants exhibited considerable activity also during the light phase of the LD (see also Fig. 2G). In a few cases, main activity in DD started from the dark phase and an additional smaller activity bout continued from the light phase (Fig. 2C). These ants were also regarded as nocturnal. In about 15% of the individuals, the free-running rhythm in DD started from the previous light phase (Fig. 2, B and E), and these were regarded as diurnal. Between 8% and 18% of the animals lacked any evident rhythm under DD (Fig. 2F), and they were therefore regarded as arrhythmic. There were no significant differences in the percentages of nocturnal, diurnal, and arrhythmic animals between the 2 colonies (χ2 = 3.00; p = 0.22). In addition, temperature had no effect on the percentages of nocturnal, diurnal, and arrhythmic animals of both colonies (χ2 = 1.78; p = 0.41; Fig. 2H).
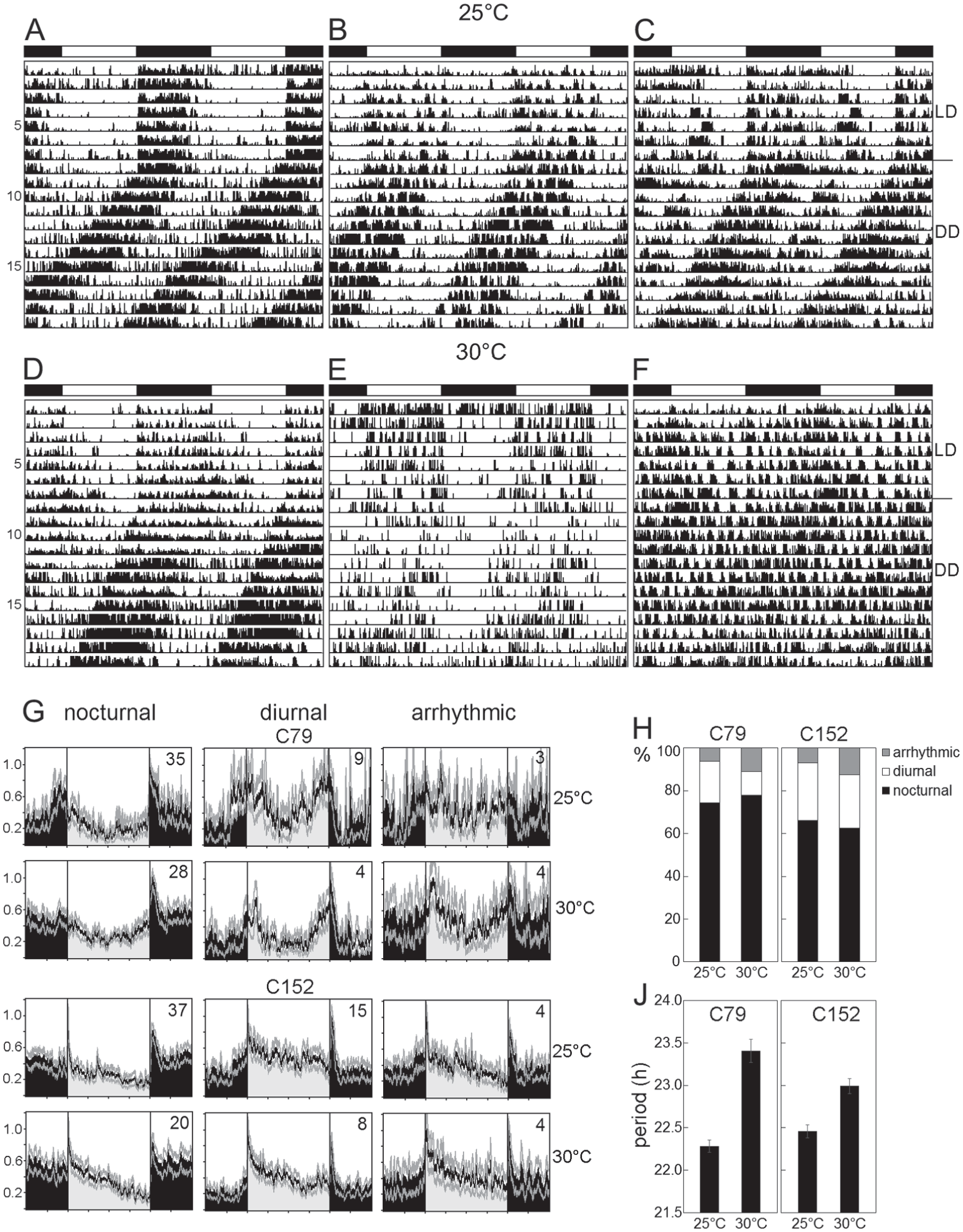
Locomotor activity rhythms of minor workers at 25 and 30 °C. (A-F) Exemplary actograms of single individuals (from colony C152) showing different types of activities. All actograms are double-plots. The white-black bars on top of each actogram indicate the light-dark phases during the LD cycle. On day 8, the animals were transferred to constant darkness (DD). (A, D) Nocturnal individuals with the free-running activity rhythm in DD continuing from the dark phase. (B, E) Diurnal individuals with the free-running activity rhythm in DD continuing from the light phase. (C) Nocturnal animal with some activity in the light phase that continued as an extra activity bout in DD. (F) Arrhythmic individual in LD and DD. (G) Average activity profiles under LD for nocturnal, diurnal, and arrhythmic ants of colonies C79 and C152 at 25 and 30 °C, respectively (numbers in the graphs indicate the number of animals). (H) The percentage of animals displaying diurnal, nocturnal, or arrhythmic behavior at 25 and 30 °C for the colonies C79 and C152, respectively. (J) Mean period length (±SEM) at 25 and 30 °C in C79 and C152.
The general activity pattern differed slightly between the colonies, as can be seen in the average activity profiles of nocturnal, diurnal, and arrhythmic ants (Fig. 2G): Ants of colony C79 exhibited 2 main activity bouts, one around lights-on and the other around lights-off, whereas ants of colony C152 showed only 1 main activity bout that was highest at lights-on in diurnal animals and at lights-off in nocturnal animals.
Besides these differences, the ants of both colonies had similar overall activity levels (Table 1), and ANOVA did not reveal any significant effects of colony on activity levels (F1,164 = 0.8549, p = 0.356). In both colonies, the average activity level was significantly higher during the night than during the day in nocturnal animals (Table 1; ANOVA followed by a post hoc test: p < 0.001) and in most cases higher during the day than during the night in diurnal animals (Table 1; p < 0.05 with the exception of C79 ants at 30 °C). The post hoc test furthermore revealed that arrhythmic animals were significantly more active than both diurnal (p = 0.003) and nocturnal (p = 0.026) animals. As can be seen in Table 1 and Figure 2G, arrhythmic animals spent comparably high amounts of activity during day and night. Although arrhythmic animals showed no clear preference for diurnal or nocturnal activity in LD, their average activity profiles still revealed some rhythmic patterns (Fig. 2G) that were difficult to see in the actograms of individual ants (compare Fig. 2F).
Mean activity levels (beam crosses per minute ± SEM) of C79 and C152 ants with different activity patterns at different temperatures.
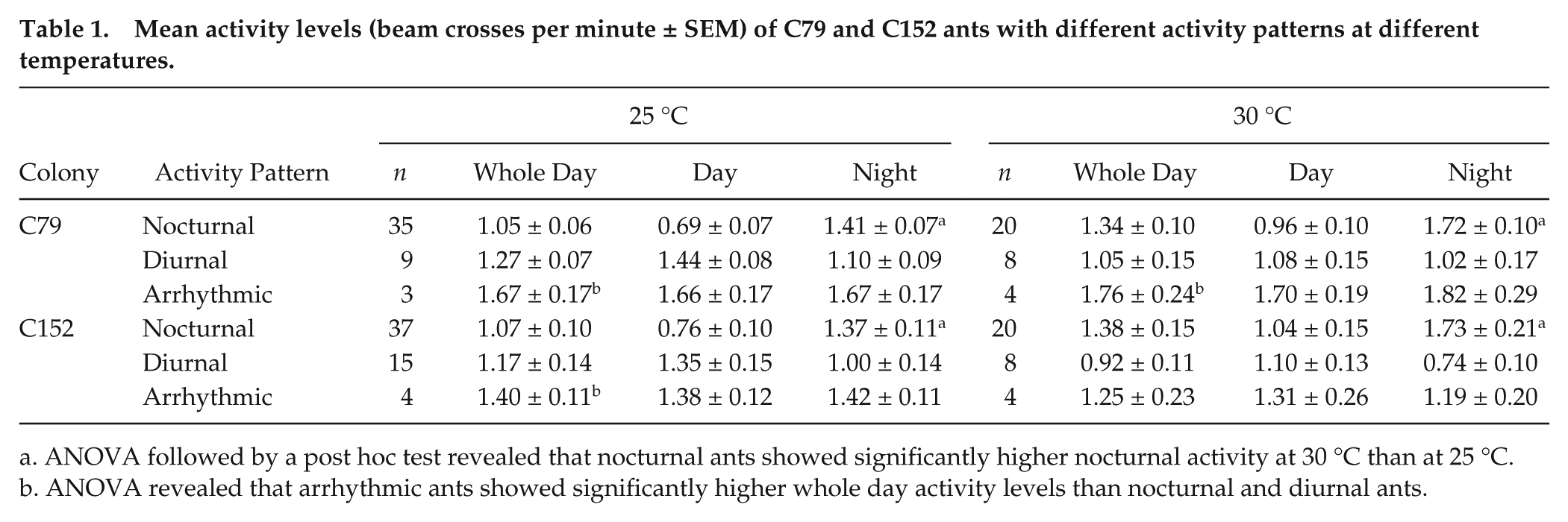
ANOVA followed by a post hoc test revealed that nocturnal ants showed significantly higher nocturnal activity at 30 °C than at 25 °C.
ANOVA revealed that arrhythmic ants showed significantly higher whole day activity levels than nocturnal and diurnal ants.
ANOVA also did not reveal significant general effects of temperature on activity levels (F1,164 = 0.0219, p = 0.883) but showed a significant interaction between temperature and activity pattern (F2,164 = 6.0561, p = 0.003). The post hoc test revealed that nocturnal animals had significantly more activity during the night at 30 °C than at 25 °C (p = 0.034). No such effects were present in diurnal animals (p = 0.901).
C. floridanus Ants Have a Temperature Overcompensated Clock
Under DD, the ants of both colonies free-ran with a period length shorter than 24 h. There was no difference in period length between diurnal and nocturnal ants (p = 0.100 for colony C152; p = 0.673 for colony C79), but we noted a significant effect of temperature on period (p = 0.006 for colony C152; p < 0.001 for colony C79). At 30 °C, average period length τ of the endogenous rhythm was significantly longer than at 25 °C in both colonies (Fig. 2J). This speaks for a temperature “overcompensated” clock.
PER Is Present in Lateral and Dorsal Neurons of the Ant Brain, and PDF Partly Colocalizes with a Subgroup of Lateral Neurons
Double labeling of PER and PDF revealed nuclear PER staining in 4 main cell clusters per brain hemisphere. These PER positive clusters were located in the dorsal and lateral regions of the protocerebrum and largely resembled the 4 neuronal PER clusters found in the honey bee, A. mellifera (Fig. 3) (Fuchikawa et al, 2017). Therefore, we named them accordingly. In the following, we describe the PER clusters from anterior to posterior. The most anterior dorsal cluster consisted of ~5 neurons that were located in the cell body cortex between the medial-lateral calyces and the vertical lobes of the mushroom bodies (Fig. 3A). This cluster continued posteriorly into ~6 further neurons (Fig. 3B) that were found in exactly the same place in the honey bee and were named “dorsal neurons” (DN) (Fuchikawa et al, 2017). We assume that both clusters found in C. floridanus belong to the DN. To consider their slightly different position, we named the more anterior cluster DNanterior (DNa) and the more posterior one DNposterior (DNp). Slightly more lateral and posterior of the DNp cluster, ~30 “dorsal-lateral neurons” (DLN) were located (Fig. 3, B and C). These cells were brightly stained by anti-PER. In a clearly more lateral place, adjacent to the medulla, we found the large group of “lateral neurons 1” (LN1) (Fig. 3, C-D). This cluster consisted of ~40 neurons that were distributed along the dorsal medulla and reached from anterior-distal (Fig. 3D) to more posterior-medial positions (Fig. 3E). At the posterior-medial edge of the LN1, we saw a small cluster of always 6 neurons. This cluster had a similar size and PER staining intensity as the LN1 and might belong to the LN1. However, since this cluster was always separated from the LN1 by 5 to 6 µm (see white arrow in the inset of Fig. 3E), we treated it as separate cluster and named it LN1′. Ventral of the distal LN1, ~3 “lateral neurons 2” (LN2) with slightly larger nuclei and rather weak labeling were detected. All of these neurons coexpressed PDF (Fig. 3, C and D; Fig. 4). PDF staining was cytoplasmic, while PER was generally found in the nucleus. Only in a few cases, we found weak cytoplasmic staining of PER in the LN2 (Fig. 4). The precise numbers of cells in the different PER clusters are given in Table 2. Anti-PER staining did not clearly label putative glia cells in the brain of C. floridanus, but in several places weak PER staining was found that resembled the glial staining found in honey bees (small green arrow in Fig. 3).
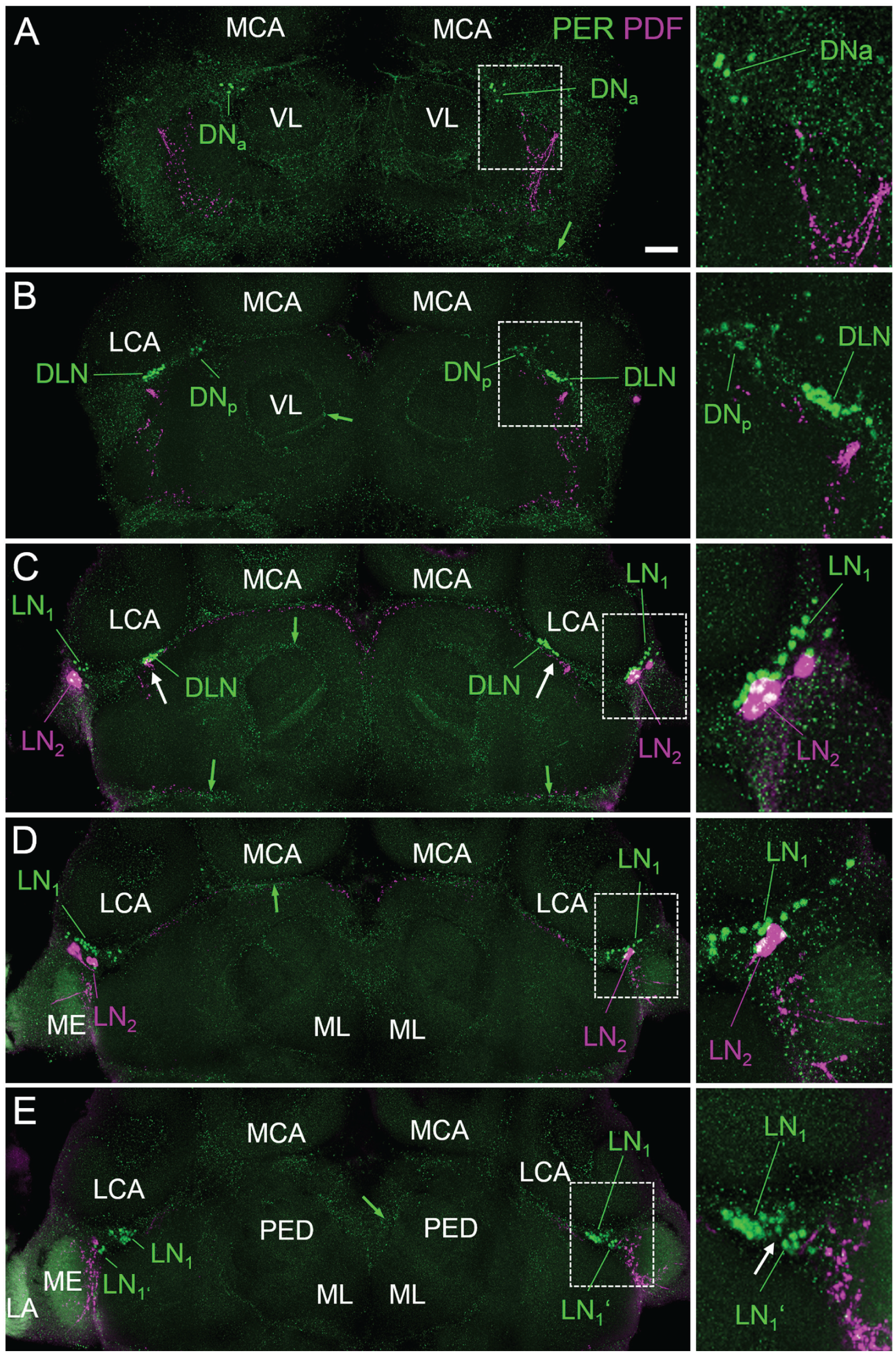
PER-positive cells in C. floridanus. Overlays of confocal sections of one representative whole mount brain labeled for PER (green) and PDF (magenta) at ZT0 are shown from anterior (A) to posterior (E). Each picture represents the overlay of 5 confocal stacks of 2 µm thickness. Specific regions of the brains (indicated by rectangles with broken white lines in the large picture) are magnified to the right. The PER-positive neuronal clusters are designated dorsal-lateral neurons (DLN), dorsal neurons anterior (DNa), dorsal neurons posterior (DNp), lateral neurons 1 (LN1 and LN1′), and lateral neurons 2 (LN2). For further explanations, see text. The small green arrows point to putative PER-positive glia cells. The white arrows in (C) indicate the close vicinity of PDF fibers to the DLN, and the white arrow in (E) indicates the small space that is free of PER-positive neurons between the LN1 and LN1′. LCA = lateral calyxes, MCA = medial calyces, VL = vertical lobes, and ML = medial lobes of the mushroom bodies. LA = lamina; ME = medulla. Scale bar: 100 µm.
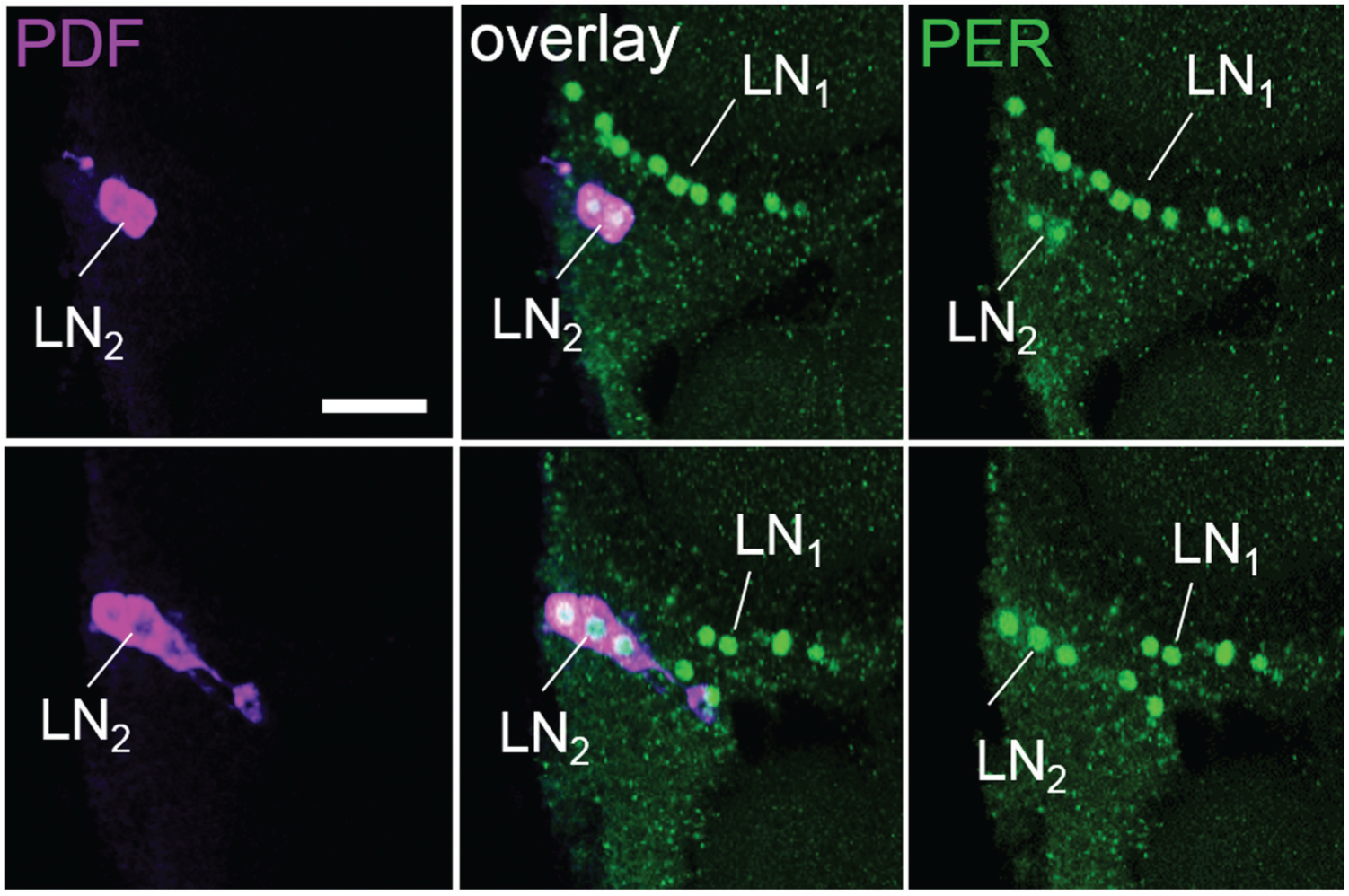
Example of PER/PDF double-labeling in the lateral neurons 2 (LN2). In this brain hemisphere, 5 of the 7 PDF-positive neurons contained PER. Each picture represents the overlay of 3 consecutive confocal stacks of 2 µm. The lower row is 10 µm more posterior than the upper one. PER labeling in the LN2 is unusually strong in this brain, and 2 of the LN2 clusters show weak cytoplasmic PER staining (upper panels). Scale bar: 50 µm.
Mean number of neurons (±SEM) in the PER-positive cell clusters (14 brain hemispheres evaluated).

To investigate whether PER staining intensity varied rhythmically, we collected brains every 3 h throughout the 24-h day. We found that PER staining intensity varied throughout the day (Fig. 5). All PER-positive clusters showed a maximum in staining intensity around ZT0 and a minimum around ZT15 (Fig. 5B). As already seen in Fig. 3, the highest PER staining at ZT0 was present in the DLN, the lowest in the LN2. Nevertheless, the Kruskal-Wallis test revealed significant differences in PER staining intensity in all clusters, also in the LN2 (DN1a: H7 = 94.44,p < 0.001; DLN: H7 = 622.47, p < 0.001; LN1: H7 = 644.88, p < 0.001; LN1′: H7 = 165.87, p < 0.001; LN2: H7 = 14.93, p = 0.037).
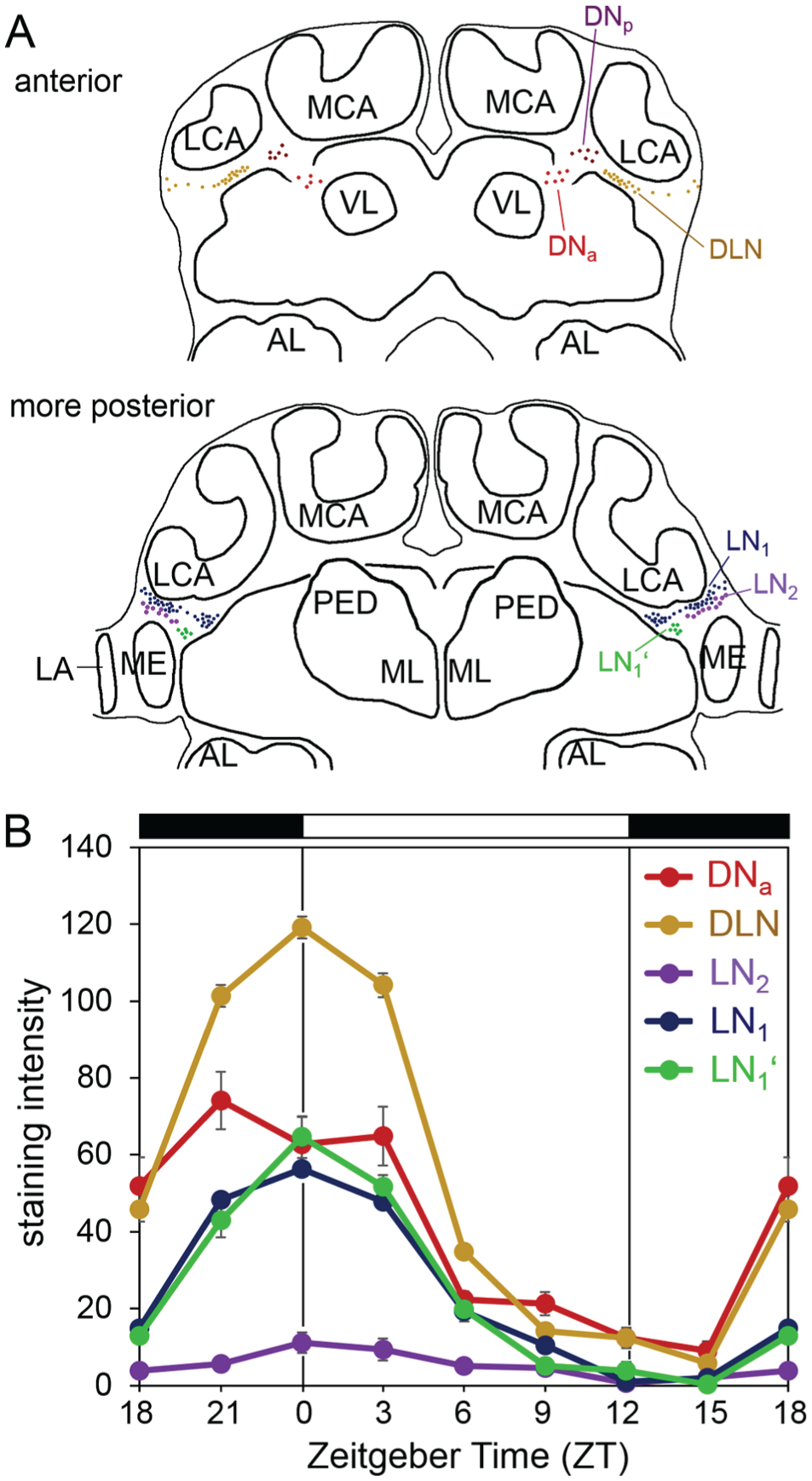
Diurnal oscillations in PER staining intensity in the PER-positive cell clusters. (A) Schematic representation of the different PER clusters. (B) Mean PER staining intensity (±SEM) in these clusters over the 24-h day (staining intensity is given in gray values ranging from 0 [no staining] to 255 [maximal staining]). At each zeitgeber time, 5 brains were evaluated. LCA = lateral calyxes, MCA = medial calyces, VL = vertical lobes, and ML = medial lobes of the mushroom bodies. LA = lamina; ME = medulla.
The PDF-positive Neurons Show Widespread Arborizations in the Ant Brain
As mentioned above, we found PER and PDF co-labeling in ~3 LN2 with a maximum of 5 PER/PDF-positive cells. The number of PDF-positive neurons was larger (Table 1) and represented 7 cells at maximum. The PDF-positive somata lay close together and their neurites overlapped, so that it was not possible to distinguish the fibers of individual neurons. Altogether, they projected into the optic lobes and many parts of the central brain (Fig. 6A). Of the optic lobes, the PDF fibers densely innervated the medulla and the lamina, but not the lobula. The lamina and the proximal margin of the medulla showed the densest PDF network. The PDF network at the proximal margin of the medulla reminds us of the accessory medulla (AMe) that houses the circadian pacemaker center in several insects, but we could not distinguish the AMe from the medulla as a separate neuropil. The dense PDF fibers appeared rather fused with the medulla. The PDF fibers leading to the central brain formed a dense bundle that bifurcated anteriorly into a dorsal and ventral fiber tract (red arrow in Fig. 6). The dorsal fiber tract ran ventrally of the mushroom body calyces and crossed the midline of the brain in an anterior commissure. The ventral fiber tract dissolved into a network of fibers that arborized on the surface of the lateral medial protocerebrum (Fig. 6B). Other fibers left the dense bundle of PDF fibers before and after its bifurcation and invaded more posterior parts of the protocerebrum (blue fibers in Fig. 6A; Fig. 6C). From there, single PDF fibers ran ventrally toward the subesophageal ganglion (black arrows in Fig. 6A), and in 1 of 20 brains, a PDF fiber entered the antennal lobes from behind (not shown). The double staining with the synaptic marker Synapsin revealed a close vicinity of the PDF fibers to the mushroom bodies (Fig. 6C) but not to the central complex (Fig. 6D).
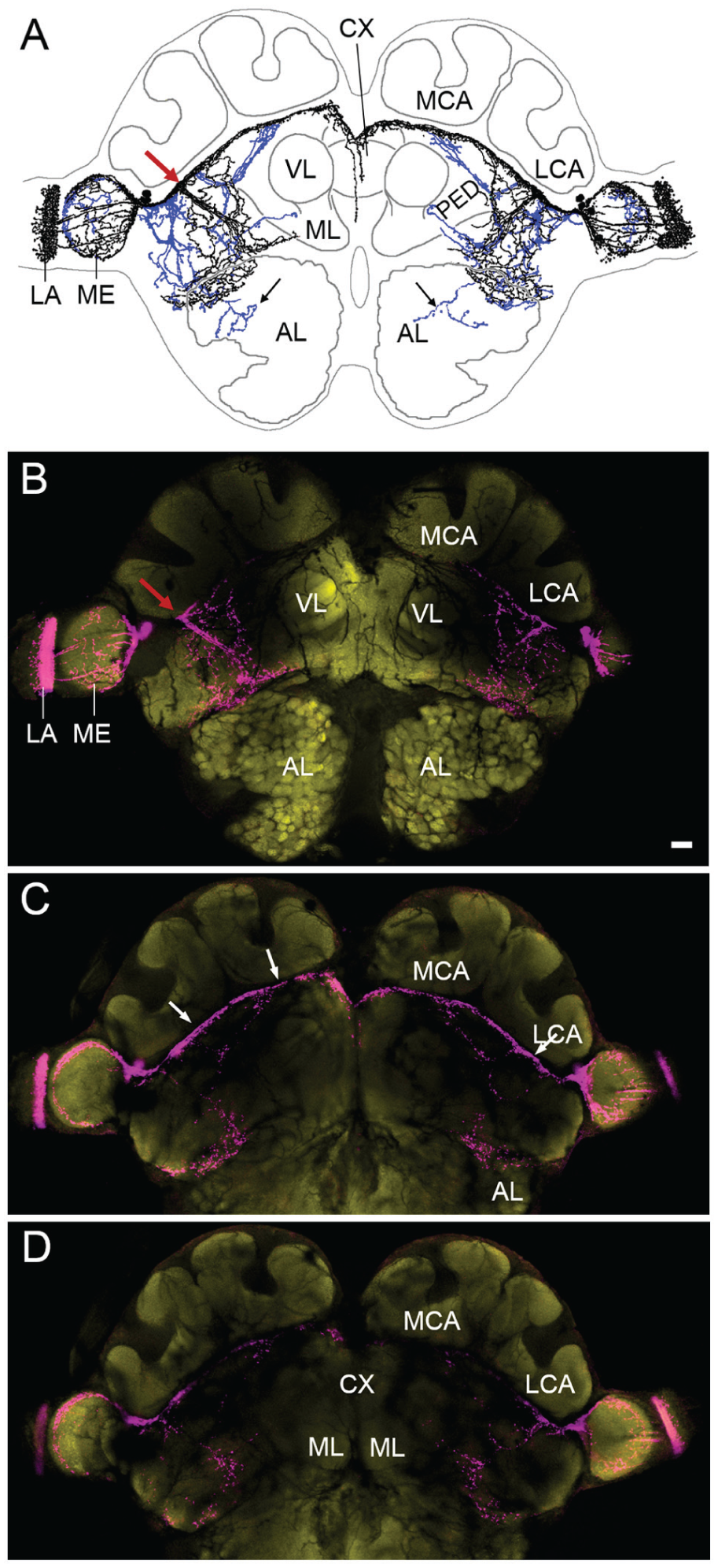
The PDF network in minor workers of C. floridanus. (A) Drawing of the PDF fibers in a representative brain. Anterior fibers are depicted in black, posterior ones in blue. The red arrow points to the bifurcation in the anterior brain. (B-D) Confocal pictures of the same brain stained with anti-βPDH (magenta) and anti-synapsin (yellow) from anterior (A) to posterior (C). Each picture represents an overlay of ten 2-µm-thick confocal stacks. The white arrows in (C) point to the PDF fibers that are in close vicinity to the calyces of the mushroom bodies. LCA = lateral calyxes, MCA = medial calyces, VL = vertical lobes, and ML = medial lobes of the mushroom bodies. CX = central complex; LA = lamina; ME = medulla. Scale bar: 100 µm.
Discussion
In this study, we demonstrate that individual minor workers of the ant C. floridanus possess a circadian clock, and we provide the first description of its putative neuronal basis.
Locomotor Activity Rhythms
Isolated individuals of C. floridanus minor ants entrained to artificial LD cycles of 12:12 h, and the great majority continued to be rhythmic in DD. In most cases, the free-running activity in DD continued from the dark phase, indicating that most individuals are nocturnal; however, we also found some diurnal animals, in which the free-running activity continued from the day phase. Furthermore, a small number of animals did not show any rhythmic activity patterns under DD, although rhythmic patterns were still visible in their average activity profiles under LD. Altogether, the activity rhythms of C. floridanus were variable, which suggests that their behavior shows some plasticity.
In honey bees, plasticity in circadian rhythms is linked to division of labor, which is important for the social organization of the colony (Shemesh et al., 2010). Young bee nurses are arrhythmic, whereas older foragers show robust diurnal rhythms. Since we collected ant workers randomly from the laboratory colonies, we do not know whether the nocturnal or diurnal animals have been foragers or whether the arrhythmic ones were the ones who took care of the brood. Nevertheless, it appears unlikely that ant nurses are arrhythmic, because in the species C. mus and C. rufipes, they rhythmically relocate and expose their brood to different temperatures on a daily basis (Roces and Núñez, 1989; Roces, 1995; Weidenmüller et al., 2009). In fact, nurses of the species C. rufipes showed circadian locomotor rhythms in isolation (Mildner and Roces, 2017). The observed rhythmicity in thermoregulatory brood-tending behavior in ants appears intuitively adaptive, since Camponotus ants are clearly different from Apis bees regarding the temperature environment of their brood. Whereas honey bees try to keep brood temperature as constant as possible (Bloch et al., 2001), the exposure of brood to daily temperature cycles appears to be important for brain development in Camponotus ants (Falibene et al., 2016). So far, there is no evidence that Camponotus ants are arrhythmic at any stage of their development, but ant workers that fulfill different tasks can show arrhythmic behavior depending on their social context (Mildner and Roces, 2017). The arrhythmic ants in the present study had high activity levels during the day and the night, suggesting that their high activity may have obscured any potential rhythmicity. Thus, their circadian clock may run, although their behavior looks arrhythmic.
Even the rhythmic individuals of C. floridanus show activity during the day and the night. Such a behavior is well described in primates and is called cathemeral, deriving from the greek kata (“through”) and hemera (“day”) (Tattersall, 1987). It means that activity is distributed approximately evenly throughout the 24 h of the daily cycle, or that significant amounts of activity, particularly feeding and/or searching behavior, occur within both the light and dark portions of that cycle. Our recording setup does not allow us to distinguish between feeding and searching, but Mildner and Roces (2017) showed for C. rufipes that foraging activity can occur during the night or the day depending on food availability. Even in nature, C. rufipes ants are active during day and night (Del-Claro and Oliveira, 1999), suggesting that they either shift a given activity to different time windows or perform different tasks at different times of the day.
Despite the fact that C. floridanus ants show cathemeral activity, we could distinguish nocturnal and diurnal individuals. This raises the question whether nocturnality and diurnality are “hardwired” in this species or whether even individual ants could switch between nocturnality and diurnality depending on the environmental conditions. In a number of ant species, activity rhythms in foraging activity change from nocturnality to diurnality on a seasonal basis, with foraging workers being diurnal in the colder months and nocturnal in the hottest periods (Fowler and Robinson, 1979). Switches between diurnality and nocturnality are also reported for small mammals and seem to strongly depend on their energy balance (Hut et al., 2012; van der Vinne et al., 2014a; van der Vinne et al., 2014b). Coldness and hunger induce diurnality in nocturnal mice (Hut et al., 2011; van der Vinne et al., 2014a). In C. floridanus, we did not see a reduction in the number of diurnal ants at 30 °C, but we saw an increase of nocturnal activity at 30 °C in the group of nocturnal ants (Table 1), suggesting that higher temperatures favor activity at night. Nevertheless, our study allows only limited conclusions, because 25 and 30 °C are at the upper temperature range to which C. floridanus ants are exposed in nature. Furthermore, we did not see an increase in nocturnal activity at 30 °C in diurnal and arrhythmic animals. Although the latter might be due to the relatively small number of animals showing these activity patterns, it will be rewarding to repeat the experiments at 20 °C or even at lowertemperatures.
Circadian clocks are typically temperature compensated within a certain physiological temperature range (Edmunds, 1988). Temperatures of 20 to 30 °C are certainly within the physiological temperature range of C. floridanus, but for unknown reasons the animals did not survive long enough in our 20 °C experiment to allow determination of the free-running period. Nevertheless, we could determine the free-running period at 25 and 30 °C and found that it was significantly longer at 30 °C than at 25 °C. This means that within this small temperature range, the circadian clock slows down at the higher temperature, called temperature “overcompensation.” It will be interesting to test in future studies whether the same overcompensation applies also for the lower temperature range.
Comparison of PER-positive Neurons inC. floridanus Ants and A. mellifera Bees
We could reliably stain 4 main clusters of PER-positive neurons in the C. floridanus brain, 2 in the dorsal protocerebrum and 2 in the lateral protocerebrum. These clusters largely resembled the PER clusters described recently in the A. mellifera brain (Fuchikawa et al., 2017), although differences are evident in the number of neurons in each cluster as well as in the presence of putative subgroups within the clusters. We start our description with the DN clusters that seemed to be composed of a more anterior and a more posterior subgroup in C. floridanus, each consisting of about 6 to 7 neurons. In A. mellifera, the DN clusters also extend from anterior to posterior, but there was no clear separation between anterior and posterior DNs and therefore anterior and posterior DNs were included in a general DN group of 15 neurons (Fuchikawa et al., 2017). Nevertheless, in both insects the DN cluster consists of anteriorly and posteriorly located neurons, strongly reminiscent of the situation in the fruit fly D. melanogaster, which also possesses anteriorly located DN1 neurons (called DN1a) that are distinct in their neurotransmitter composition and projection patterns from the posteriorly located siblings, the DN1p (Shafer et al., 2006). Future studies are required to reveal which neurotransmitters the DN1a and DN1p use in C. floridanus and A. mellifera. The DLN cluster was the strongest stained cluster in C. floridanus and consisted of ~30 neurons. In A. mellifera, the DLNs are less strongly stained and are composed of 60 to 75 neurons, which clearly exceeds the number of DLNs in C. floridanus (Fuchikawa et al., 2017). The number of LN1 in A. mellifera (105-120 neurons) was again larger than that in C. floridanus (40-50 neurons). Six of the LN1 were clearly separated from the main group and were therefore called LN1′. Such a separation was not evident in the LN1 of A. mellifera, but we cannot exclude that even there subgroups of the LN1 exist. Again, there are parallels in D. melanogaster, in which the putatively homologous LNd group consists of 2 different subgroups, although they are located closely together (Helfrich-Förster et al., 2007; Johard et al., 2009). Finally, we found 2 to 5 LN2 cells in C. floridanus that had larger nuclei, were clearly less intensively stained than the other PER-positive neurons, and coexpressed PDF. In A. mellifera, 14 to 16 such cells were found (Fuchikawa et al., 2017). In contrast to C. floridanus, in A. mellifera the number of PDF- and PER-positive LN2 neurons was identical. In other words, all PDF neurons also expressed PER in A. mellifera, whereas maximally 5 of the 7 PDF neurons were PER-positive in C. floridanus. Since PER staining intensity in the LN2 was very low, it is possible that we could not detect all PER-positive cells. Staining of PER and PDF on sections of C. floridanus brains, instead of whole mounts, might shed further light on this issue. PER staining intensity of the LN2 group was also very low in honey bees (Fuchikawa et al., 2017). Future studies are needed to reveal whether this has any functional reason: for example, whether these neurons play a less important role in the circadian circuit. Since the LN2 show a significant cycling in PER staining intensity and express PDF, which has been shown to be an important clock factor in a wide variety of insects, we do not think that the LN2 are less important than the other clock neurons (see discussion below).
In contrast to A. mellifera and D. melanogaster brains, in which many glia cells express PER, we did not see clear PER staining in glia cells of C. floridanus brains. Again, we do not think that there is a fundamental difference between ants, bees, and flies in the presence of PER in glia cells. More likely, the staining intensity with the antibody raised against PER of honey bees might be too low to be detected in all PER-positive cells of the C. floridanus brains. The glimpse of putative PER glia staining in brain regions of C. floridanus, in which we have typically seen prominent glia staining in honey bees, speaks for this hypothesis.
In summary, the PER staining pattern of C. floridanus brains strongly resembles the pattern observed previously in bees and flies and makes us confident that we indeed detected C. floridanus PER with the antibody raised against A. mellifera PER.
Comparison of PER Oscillations in Nocturnal C. floridanus Ants and Diurnal A. mellifera Bees and D. melanogaster Flies
We showed that PER staining intensity exhibited a significant daily fluctuation in all 5 measured neuronal subgroups. PER staining intensity in all clusters was maximal around lights-on and minimal after lights-off. Although the immunolabeled C. floridanus ants were clearly nocturnal, their phase of PER cycling exactly matches the phases of PER cycling in diurnal flies and bees (Zerr et al., 1990; Shafer et al., 2002; Yoshii et al., 2009; Fuchikawa et al., 2017). These diurnal insects also showed maximal PER intensity around lights-on and a trough at the beginning of the night. Thus, we show for the first time that there is no difference in PER cycling in diurnal and nocturnal insects. This is not surprising, given that PER of diurnal and nocturnal mammals cycles in the same phase in the circadian clock neurons of the mammalian pacemaker, the suprachiasmatic nucleus (SCN) (for reviews, see Smale et al., 2003; Challet, 2007). Even within one species, PER cycling appears not to switch its phase when the animal switches its behavior from nocturnal to diurnal, as was recently shown for mice (reviewed in Riede et al., 2017; van der Veen et al., 2017). It will be most interesting to test whether the same is true for nocturnal and diurnal C. floridanus ants.
Another interesting parallel between ants, bees, and mice is the almost exclusive presence of PER in the nuclei of the neurons. Ants, bees, and mice possess a mammalian type molecular clock with the clock proteins PER and CRY in the negative limb of the feedback loop, which is in contrast to the Drosophila clock that uses PER and TIM in this negative limb (Rubin et al., 2006; Ingram et al., 2012; Yuan et al., 2007). In fruit flies, PER accumulates in the cytoplasm and shows a gated translocation into the nucleus in the middle of the night (Curtin et al., 1995; Shafer et al., 2002). This is very different in mice and bees, in which PER seems to be transferred to the nucleus immediately after its synthesis, and therefore almost no cytoplasmic PER can be observed (Smyllie et al., 2016; Fuchikawa et al., 2017). The same seems to be true for C. floridanus ants: Except for very little cytoplasmic PER in two LN2 neurons of one brain, we did not observe PER in the cytoplasm at any time point. Thus, PER seems to accumulate in the nucleus as in mice and bees.
Comparison of PDF Arborization Patterns between C. floridanus Ants and A. mellifera Bees
Neurons that express the neuropeptide PDF play crucial roles in the clock network of many insects and, due to their global branching pattern in the insect brain, are suitable candidates to provide time-information to brain centers controlling rhythmic behavior. PDF neurons were initially described in cockroaches (Stengl and Homberg, 1994), crickets (Homberg et al., 1991), and flies (Nässel et al., 1991; Helfrich-Förster and Homberg, 1993; Nässel et al., 1993; Shiga et al., 1993) and later in a variety of other insects including bees (Bloch et al., 2003; Závodská et al., 2003; Weiss et al., 2009; Sumiyoshi et al., 2011). In the honey bee A. mellifera, the PDF projections invade most areas of the protocerebrum (Bloch et al., 2003; Beer et al., 2018). They are particularly dense around the mushroom bodies, centers for olfactory memory in insects. Since A. mellifera bees have a remarkable sense of time and can learn up to 9 different time points at which different flowers yield maximal nectar and pollen (Kleber, 1935), the PDF neurons have been hypothesized to transfer the relevant time information to the mushroom bodies (Beer et al., 2018). The information about time is similarly important for time-compensated sky compass orientation. The centers of sky compass orientation are located in the central and lateral complexes that receive input of skylight polarization from a specialized area of the compound eyes (Homberg et al., 2011; Homberg, 2015). In A. mellifera, the PDF arborizations are in close vicinity to several regions along the sky compass network (Zeller et al., 2015; Beer et al., 2018) and appear suited to give circadian input into this network.
Here, we show that the PDF neurons arborize in close vicinity to the mushroom bodies but not to the central complex of C. floridanus. It has not yet been tested whether C. floridanus ants possess a time memory, but Ectatomma ruidum ants show time-place learning abilities (Schatz et al., 1999), and C. rufipes is at least capable of interval timing (Schilman and Roces, 2003). Thus, it is quite likely that C. floridanus ants also possess a time memory. This fits with the neuroanatomy of the PDF-positive clock neurons that arborize close to the mushroom bodies. Regarding sky compass orientation, it is questionable whether any Camponotus species is capable of it. These ants are known to use external landmark orientation and to follow trail pheromones that nestmates have laid before (Hölldobler 1971; Hartwick et al., 1997; Klotz, 1987; Klotz and Reid, 1993), indicating that they normally do not rely on sky compass orientation. Thus, C. floridanus ants would not depend on the transfer of time information to their central complex, which fits with the lack of PDF fibers close to the central complex. It will be most interesting to investigate whether PDF fibers extend to the central complex in other ant species that use time compensated sky compass orientation, such as desert ants (Müller and Wehner, 2007).
In A. mellifera, it was furthermore shown that PDF fibers arborize in close vicinity to the other clock neurons and that the PDF fibers are often accompanied by PER-positive glia cells (Fuchikawa et al., 2017; Beer et al., 2018). In C. floridanus ants, the PDF fibers are in similarly close vicinity to some of the other PER neurons, such as the DLNs (Fig. 3C). Nevertheless, a more detailed histological analysis of PER and PDF on sections is warranted to reveal the precise connection between PDF fibers and other PER cells.
Although many questions remain about the circadian clock of the carpenter ant C. floridanus, we were able to shed the first light on its working mechanisms, its anatomic location, and its putative role in controlling behavioral rhythmicity. Since social insects and their timing of coordinated behavior could offer many interesting insights in chronobiological research, we hope this study may lay the foundations for further investigation of the clock of C. floridanus and other ants.
Footnotes
Acknowledgements
We thank Katharina Beer for discussions and comments on the manuscripts. The study was funded by the German Research Foundation (DFG), SFB 1047 Insect Timing, Project A1 and C1.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
