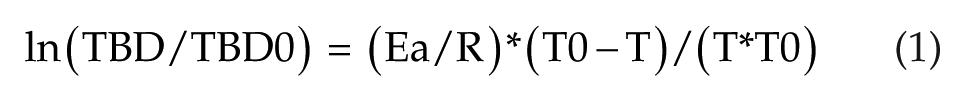Abstract
Syrian hamsters may present 2 types of torpor when exposed to ambient temperatures in the winter season, from 8°C to 22°C (short photoperiod). The first is daily torpor, which is controlled by the master circadian clock of the body, located in the SCN. In this paper, we show that daily torpor bout duration is unchanged over the 8°C to 22°C temperature range, as predicted from the thermal compensation of circadian clocks. These findings contrast with the second type of torpor: multi-day torpor or classic hibernation. In multi-day torpor, bout duration increases as temperature decreases, following Arrhenius thermodynamics. We found no evidence of hysteresis from metabolic inhibition and the process was thus reversible. As a confirmation, at any temperature, the arousal from multi-day torpor occurred at about the same subjective time given by this temperature-dependent clock. The temperature-dependent clock controls the reduced torpor metabolic rate while providing a reversible recovery of circadian synchronization on return to euthermy.
When exposed to adverse conditions, many mammals may undergo torpor, in which their body temperature (Tb) and metabolic rate (MR) both sharply decrease. According to the recent review by Ruf and Geiser (2015), daily torpor and multi-day torpor (classic hibernation) are clearly distinguishable by cluster analysis. In both, episodes (or “bouts”) of hypothermia and reduced MR are intercalated with short returns to euthermy (arousals). In daily torpor, bout duration is less than a day, Tb drops about 10°C on average, and MR is reduced to about 35% of the minimal euthermic value. Time control is achieved by the temperature-compensated circadian clock of the SCN. In contrast, in multi-day torpor, bout duration may be much longer, and Tb often drops close to ambient temperature (Ta). Yet it is unclear how bout duration is controlled.
Various hypotheses have been proposed to explain bout duration, but none has convincingly established its control. The context is now changing (see Discussion). First, the circadian system is now viewed as a complex network of peripheral oscillators or clocks, which receive synchronizing signals (nervous or endocrine) from a clock located in the SCN (Golombek et al., 2014; McGinnis and Young, 2016; Schibler et al., 2015) The SCN clock in turn is synchronized to astronomical time by photoperiod-dependent signals generated in the retina. Second, the circadian system is intricately interfaced with the control of metabolism (Kumar Jha et al., 2015; Ray and Reddy, 2016): the synthesis or degradation of key metabolic enzymes is controlled by clock output genes and, reciprocally, metabolic factors impinge on the functioning of the clock. Additional regulations handle the instantaneous changes of substrate utilization that are needed in the absence of de novo protein synthesis; e.g., to sustain a flight response to avoid a predator, or for the arousal from deep torpor. Metabolic scope—i.e., the ratio of maximum to minimum MR observed in resting conditions—can thus reach 10- to 15-fold (average) in small mammals in the absence of major changes to Tb (Bishop, 1999). The timing of multi-day torpor bouts of classic hibernation should provide new insights as to their regulation.
When Tb changes during a torpor bout, one might expect changes in the clock machinery. In particular, fluctuations in the period of the SCN circadian clock could exceed the range that allows synchronization to day/night alternations. In daily torpor, such fluctuations are constrained by a process called temperature compensation, which is a fundamental property of circadian clocks (Pittendrigh, 1954). In a previous paper, however, one of us (Malan, 2010) observed that this was insufficient to explain the temporal patterns of the multi-day torpor bouts of classic hibernators. Instead, it was proposed that those patterns might be controlled by a non-temperature-compensated, but circadian-compatible, clock. The present paper presents an experimental verification of this hypothesis.
To this end, we have chosen the Syrian hamster (Mesocricetus auratus), which may present both kinds of torpor over a wide range of ambient temperatures (8°C to 22°C), to compare the characteristics of torpor as a function of temperature.
Materials and Methods
Animals
This study aimed to determine the effects of Ta and Tb on the timing of torpor bouts over a full hibernation season in the Syrian hamster, Mesocricetus auratus. This species was chosen because it may spontaneously present either daily or multi-day torpor bouts over a wide range of ambient temperatures. Three groups of 15 adult male Syrian hamsters were born and reared in the Chronobiotron animal facility (UMS 3415, CNRS and University of Strasbourg). The groups were assigned to Ta of 8°C, 15°C, or 22°C (±1°C). Group allocation was randomly selected by the animal number in Chronobiotron records, to which the investigators were blinded. Homogeneity of body mass was confirmed. Group size was dictated by the availability of animals and space in temperature-controlled units. The photoperiod was maintained at light-dark 10:14 (LD 10:14); this mimics a winter-like schedule and leads to complete gonadal regression in the Syrian hamster (Gaston and Menaker, 1967; Powers et al., 1997). Food pellets and water were provided ad libitum. In addition to standard bedding, the animals were provided hay as hibernaculum-building material.
Timing of Torpor Bouts
The first objective was to analyze the timings of spontaneous torpor bouts throughout a full hibernation season. A torpor bout was characterized by 2 successive crossings of a Tb threshold of 34°C: downwards (entry into torpor) followed by upwards (arousal). This threshold, located just below the range of spontaneous fluctuations of Tb in euthermic resting conditions, maximized the range of Tb to study. Two intraperitoneal data loggers (Thermochron i-buttons) were implanted under isoflurane anesthesia (start 4%, then 2.5%) preceded with analgesia: local (i.p.) anesthetic treatment with 2.8 mg/kg lidocaine at the site of incision, and non-steroidal anti-inflammatory treatment with 2 mg/kg (s.c.) metacam. During the post-recovery period, the animals received 1 mg/kg metacam in their drinking water for 3 days. The data loggers were calibrated against a precision mercury thermometer (±0.1°C) before implantation. To obtain data over the full hibernation season, the acquisition period was set to 20 min, and the time-scale of the second data logger was shifted with respect to the first one for complete coverage. At the end of the experiment, data were transferred to a computer. The experiment was terminated when all animals had stopped presenting torpor bouts for 2 weeks. Tb was recorded every 20 min. The period between the first and last torpor bouts was recorded as the overall hibernation period. Animals that never entered torpor within this period were excluded from the study.
All experiments were performed in accordance with the NIH Guidelines for the Care and Use of Laboratory Animals (1996) and the French National Law (implementing the European Union Directive 2010/63/EU), and were approved by the Regional Ethical Committee of Strasbourg for Animal Experimentation (CREMEAS) and the French Ministry of Higher Education and Research.
Statistics (General)
Standard statistical methods were used as described in Zar (1984). Non-linear least squares regression was performed with Sigmaplot software (Systat Software; San Jose, CA). If not otherwise stated, the confidence limit was set to P < 0.05. Calculations of torpor bout durations in subjective time, as described in the Discussion, were completed with Delphi (Embarcadero; Austin, TX) to benefit from the high precision of the internal representation of variables in double precision (±2 gigabits). The simplicity of the algorithms did not necessitate the use of a programming language (see below).
Calculation of Torpor Bout Duration in Subjective Time
The next step was to select an equation to relate duration of long torpor bouts to Tb. When the MR is reduced in torpor, the Tb to Ta temperature gradient is sharply reduced in a small mammal and may be ignored as a first approximation. In the absence of temperature compensation, we have assumed that Arrhenius (Boltzmann) thermodynamics of the velocity of catalytic reactions (Laidler and Peterman 1979) can be applied to the set of catalytic reactions involved in the clock or, equivalently, to the rate-limiting process. Any duration measured by the clock, such as torpor bout duration (TBD) or the period tau of the clock oscillation, will then follow equation (1), when the absolute temperature, T (Kelvin), varies from T0 to T:
TBD and TBD0 are the torpor bout durations at absolute temperatures T and T0; T0 is the reference temperature (310.15 K, equivalent to 37°C); Ea is the apparent enthalpy of activation; and R is the constant of perfect gases. The same equation applies to the period tau of the clock by substituting tau to TBD in equation (1). The durations in subjective time of all bouts can then be calculated individually. This was done by summing the interval durations in subjective time corresponding to each of the 60 to 600 intervals of 20-min astronomical time comprising the bout. Precisely, for each 20-min interval, the values of the period tau for the beginning and end of the interval were calculated from equation (1). The geometric mean of the 2 tau values was then used to calculate the duration in subjective time corresponding to 20 min of astronomical time: 20 min / (mean tau). The sum of the individual subjective durations was taken as an estimate of the total duration of the bout, as measured by the subjective clock.
A note of warning to readers who would like to apply similar calculations to other data: our original calculations were performed in Delphi (Embarcadero; Austin, TX). The algorithms used here were too simple to justify the use of a programming language, but Delphi provided an accurate representation of double-precision variables (±2 gigabits). From our experience, running the repeated matrix inversions involved in non-linear least squares regressions with a less-precise format may lead to incorrect results due to the accumulation of rounding errors. In case of doubt, performing calculations with double-precision integers should give more reliable results.
Results
Torpor occurred in 14/15 animals at 8°C, 8/15 animals at 15°C, and 8/15 animals at 22°C (Fig. 1). Within the hibernation season, these figures comprised both daily and multi-day torpor bouts, with variations among individuals, Ta, and date. There was no indication of bi-circadian cycles, such as observed in human studies (Gleit et al., 2013; Phillips et al., 2011). The number of torpor events for each animal or within a temperature group were too small and variable for specific categorization. Torpor bouts were therefore pooled for each Ta: 110 bouts at 8°C, 94 bouts at 15°C, and 181 bouts at 22°C; the distributions of bout durations in astronomical time are shown as histograms (Fig. 2). Whatever the temperature, the distribution was bimodal. Because the width of the right peak increased as Ta decreased, a larger time interval was utilized for the right peak of the histogram than for the left one at 15°C and 8°C. All histograms could be significantly fitted to a double Gaussian distribution by a non-linear least squares regression (Sigmaplot, Systat Software; San Jose CA). P values were respectively P < 0.01 at 22°C, P = 0.01 at 15°C, and P < 0.01 at 8°C. Normality (Shapiro-Wilk) and constant variance (Kolmogorov-Smirnov) tests were passed. The mean (SD) values of the left peaks were 0.37 (0.15) days at 22°C (n = 11), 0.18 (0.04) days at 15°C (n = 8) and 0.51 (0.51) days at 8°C (n = 14). For the right peaks, the values were 0.91 (0.18) days at 22°C (n = 11), 1.39 (0.58) days at 15°C (n = 8) and 3.35 (0.68) days at 8°C (n = 14). The data at 15°C should be interpreted with caution because of the skewedness of the peak. As a confirmation (in case the results might have been affected by the non-uniform bin spacing at 15°C and 8°C), single Gaussian curves were also fitted individually to the left and right peaks. The fitting just failed to reach significance for the left peaks at 15°C (P = 0.056) and 8°C (P = 0.057) but was significant for the right peak at both temperatures (P = 0.03 for both). A similar double peak was observed at 15°C, but there were too few multiday torpor bouts to obtain an un-skewed left histogram. Thus, except for the short bouts at 15°C, all bout durations were normally distributed.
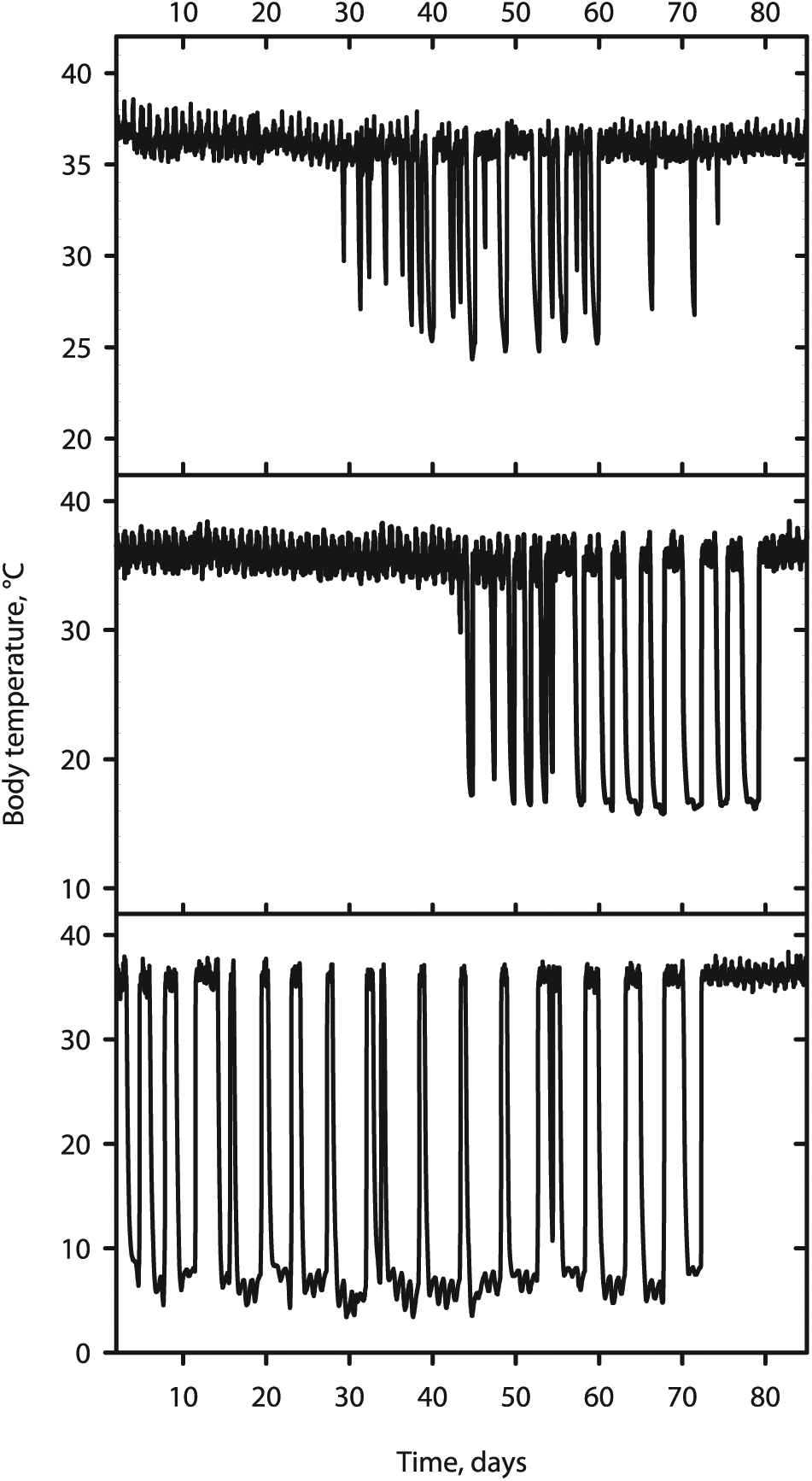
Representative body temperature (Tb) recordings of full hibernation seasons of individual Syrian hamsters. From top to bottom: Animals housed at ambient temperatures (Ta) of 22°C (top), 15°C (middle), and 8°C (bottom). At all Ta, both shorter and longer torpor bouts were observed, with no obvious seasonal distribution at 15°C or 22°C. Irrespective of the Ta and bout duration, all torpor bouts started from a common body temperature of ~37°C, and ended up at this same temperature.
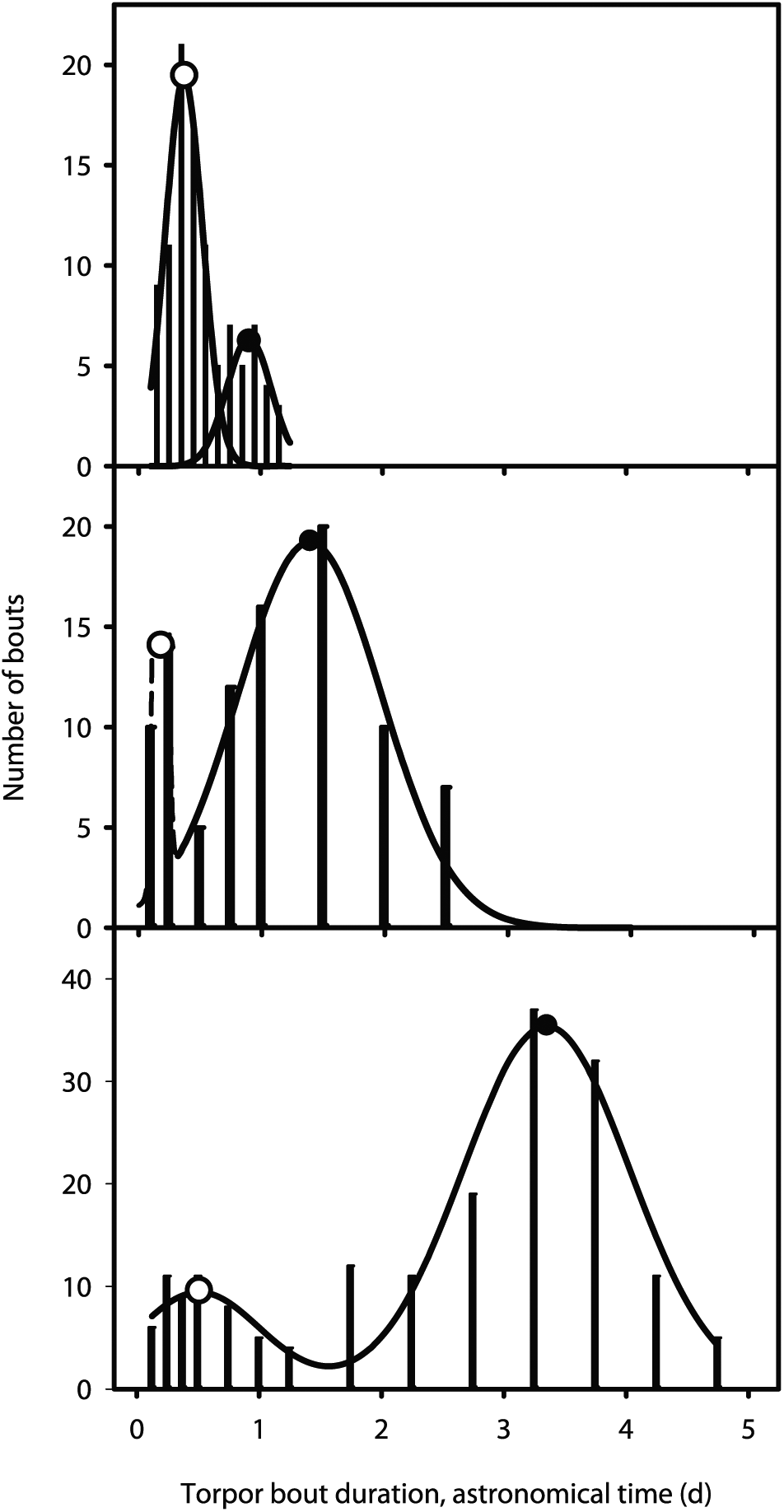
Double-peak distributions of torpor bout durations in Syrian hamsters at 22°C, 15°C, and 8°C. Data were pooled for animals that had undergone torpor bouts at 22°C (top: n = 8; 100 bouts), 15°C (middle: n = 8; 94 bouts), or 8°C (bottom: n = 12; 181 bouts). Curves: Bimodal Gaussian curves fitted by non-linear least squares regression (see “Statistics” section). Left peaks and open circles (mean): daily torpor. When the temperature decreased, the mean duration was relatively unchanged. For daily torpor, this is known as “temperature compensation”. Right peaks and closed circles (mean): multiday torpor (classic hibernation). At all temperatures, the distribution was smooth, indicating that it corresponded to single bouts and not to the fusion of a variable number of bouts. The shapes of the curves were similar to those of daily torpor, except that the means were highly temperature-dependent.
The double Gaussian distribution resulted from a purely descriptive approach and involved no assumption on underlying mechanisms. It confirmed the distinction between daily torpor and multi-day torpor made by Ruf and Geiser (2015). The distribution also provided an objective criterion for short and long torpor bouts to be distinguished at any Ta by their abscissa with respect to the minimum difference between the 2 peaks. This might have ignored “test drops”—i.e., shorter forerunners of long bouts at the beginning of the hibernation season (Strumwasser, 1959)—which could thus have been pooled with bouts of circadian torpor. However, such test drops were observed only at 8°C and for few animals: among the 13 animals, 8 presented no such early shorter bout, 2 showed one bout, and 3 showed two bouts. At 15°C and 22°C, no seasonal repartition of short vs. long bouts could be readily identified. This point was therefore ignored in further calculations.
The durations of short bouts at 8°C and 22°C did not differ. Because this fitted with data in the literature for daily torpor (see Discussion) no further tests were run to verify that the homogeneity applied to all Tau’s, as predicted for durations controlled by the temperature-compensated master circadian clock. The distributions of long bouts were Gaussian irrespective of Ta, but the means and standard errors increased as Ta decreased (Fig. 2).
The relationship between multi-day torpor bout duration and temperature was then fitted to a linear form of the Arrhenius equation (Fig. 3). Of note, this is a standard equation of chemical thermodynamics. It implies no other assumption than the involvement of a catalytic mechanism, a common feature of biochemical systems. The fit was highly significant: F1,220 = 495; P < 10-10; R² = 0.692; hence, the regression explained more than two-thirds of the total variance, leaving little room for additional factors, such as metabolic inhibition. At 310.15 K (37°C), the line extrapolated near the middle of the confidence interval (CI) of the distribution of the mean on the log scale: CI = (−2.70 to 0.387) (Fig. 3).
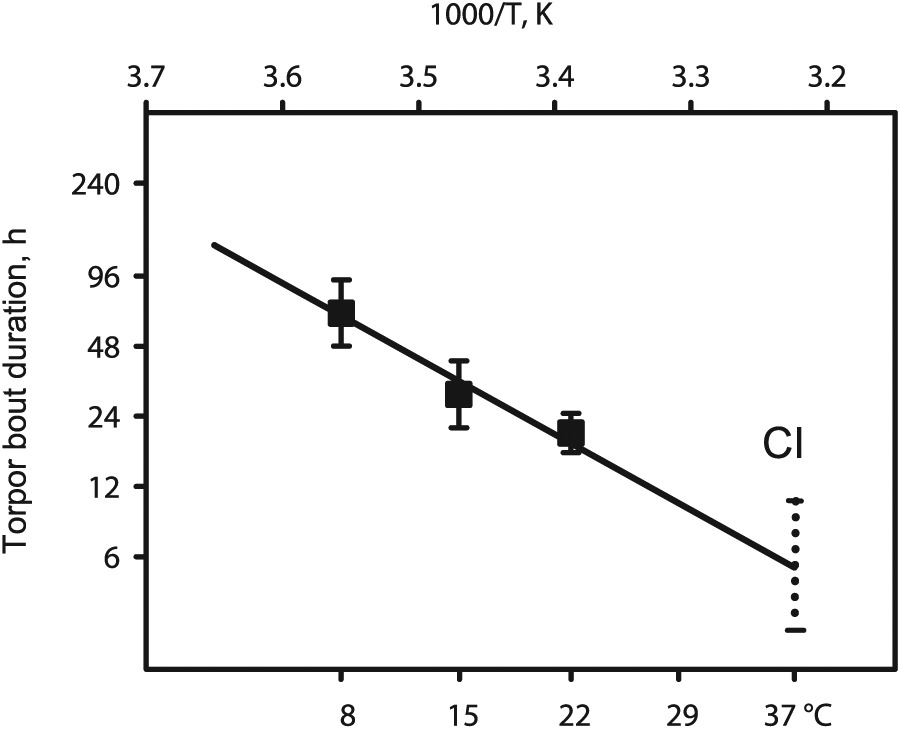
The relationship between torpor bout duration and temperature complies with Arrhenius (Boltzmann) thermodynamics. Squares: Durations (mean ± SD) of multi-day torpor bouts of Syrian hamsters at 8°N, 15°C or 22°C. Line: Arrhenius (Boltzmann) thermodynamics predict a linear relationship between, on the one hand, the reciprocal of absolute temperature (abscissa, top), and on the other hand the natural logarithm of the rate of a biochemical reaction, taken here as determining torpor bout duration (ordinate). The bottom abscissa (°C) is given as a practical reference. The quality of the fitting practically excludes any additional factors. CI, confidence interval of daily torpor bouts measured independently at 22°C (Fig. 2). At 37°C the extrapolation of of the Arrhenius line for multi-day torpor bouts falls within this CI.
We questioned whether a clock was involved in the duration of multi-day torpor bouts. This could not be the SCN clock, because there was no temperature compensation (Fig. 3). A more compelling argument (in favor of a clock) would be that whatever Tb torpor bout durations measured on the clock would all occur within the same angular range on a 24-h dial of the clock (Fig. 4) or, in other terms, within the same range of subjective time. This was tested as follows. For each of the successive 20-min intervals of astronomical time comprising a torpor bout, the duration of subjective time read on the temperature-dependent clock was calculated from equation (1). For instance, if the period of the clock at a given Tb was 4 days (96 h)—i.e., if it was 4 times slower than the SCN clock—then the 20 min-period in astronomical time would show as 5 min (20/4) of the subjective time of the temperature-dependent clock. By the same token, if the period of the temperature-dependent clock increased to 10 days, the 20-min interval would appear as 2 min on the temperature-dependent clock, and so on. Starting from zero at the beginning of a torpor bout and adding the successive intervals in subjective time calculated in this fashion, we could obtain the total subjective time spent torpid at the end of the bout. The torpor bout durations in subjective time observed at any Ta could then be directly compared using a common polar plot with a circumference of 24 h of subjective time (angular scale) and a radial scale showing the periods of the clock in astronomical time for various temperatures (Fig. 4). At all temperatures, most torpor bouts lasted between 6 and 11 h of subjective time. No differences were found between bouts at 8°C and 15°C but both were shorter than the bouts at 22°C. However, at 22°C, the base of the curve of a bout was shorter than that at the higher Tb (Fig. 5). This increased the weight of higher Tb’s in the calculated mean and probably explained most of the difference. If any remaining difference may be ignored, then, irrespective of Ta, all torpor bouts took place during the same time interval of subjective time of the temperature-dependent clock. This suggests that the durations of long (i.e., multi-day) torpor bouts were controlled by the temperature-dependent clock, and provides an answer to the ancient problem of the duration of multi-day torpor bouts.
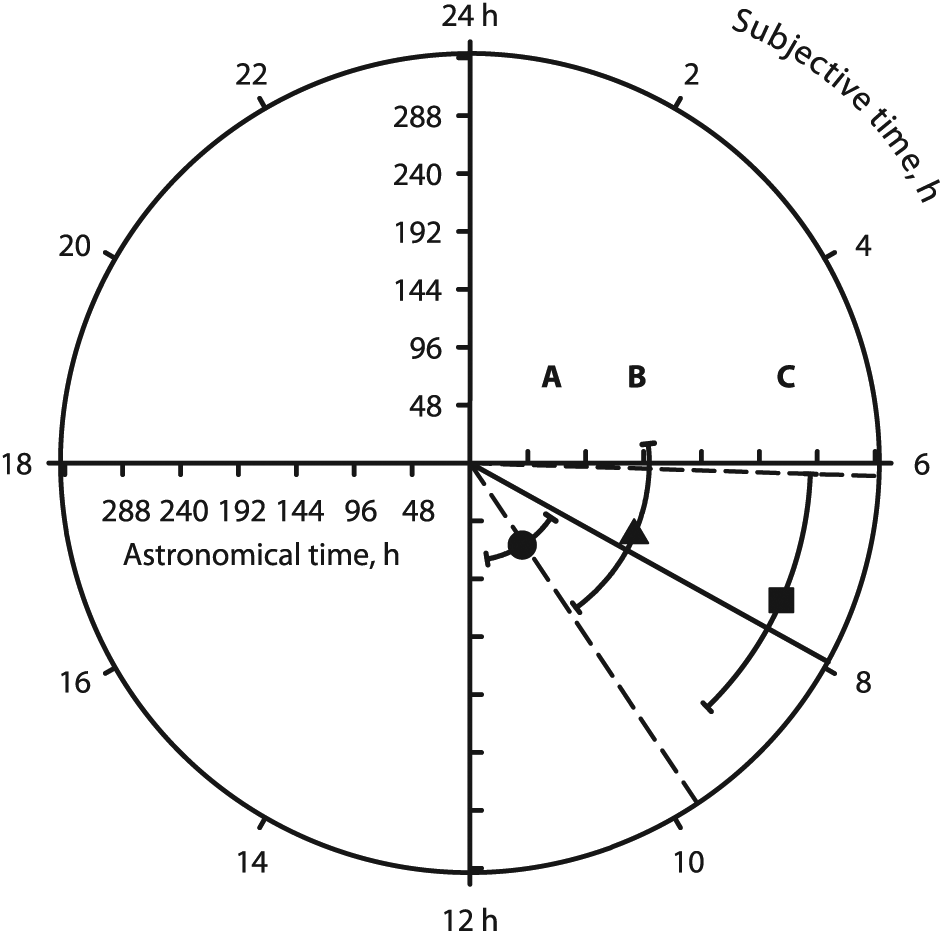
Torpor bout duration measured by subjective time of the temperature-dependent clock at 22°C, 15°C or 8°C. Figures 2 and 3 have shown that torpor bout duration measured in astronomical time increases considerably as temperature decreases. Here, the bout durations are expressed in “subjective time” and are all represented on a single, 24-h dial of the temperature-dependent clock (common 24 h angular scale). This scale corresponds to the common starting and ending temperatures and periods of all torpor bouts, as shown in Figure 1. Pooled torpor bouts (mean ± SD) are shown for 22°C (A and circle, n = 36), 15°C (B and triangle; n = 59) and 8°C (C and square; n = 125). Solid and dashed radial lines indicate the grand angular mean ± SD. Most bouts occurred within 6 h and 11 h of the clock subjective time. This suggests that, at all temperatures, torpor bout duration was controlled by the temperature-dependent clock.
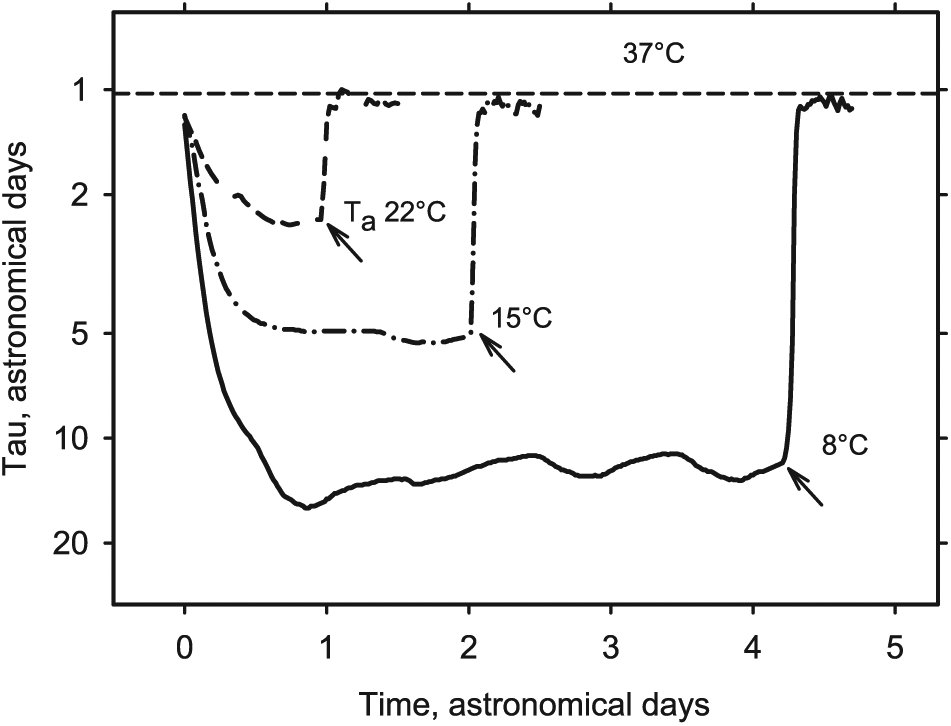
The reversibility of the temperature-related excursions of the subjective period tau at 22°C, 15°C and 8°C. The statistical aspects of multi-day torpor bouts are shown in previous figures. The present figure serves only to illustrate the reversibility of function of the temperature-dependent clock. Three representative bouts of multi-day torpor are shown. Ordinate: log scale (inverted to comply with usual view of body temperature). The subjective tau increased considerably as temperature decreased. The control, by a purely thermodynamic phenomenon, ensured that, at the end of a bout (arrows) and irrespective of the minimum temperature reached in torpor, Tb always returned to its euthermic, circadian value compatible with resynchronization to astronomical time. Notice that, at 22°C, owing to the smaller (Tb – Ta) gradient, temperature equilibrium was achieved only later in the bout. As a result, the subjective Tb, calculated as the mean of all data within the torpor bout, was over-estimated. The differences in the data for 22°C, as well as for 8°C and 15°C in Figure 4, might thus essentially result from this mathematical artefact.
Discussion
At the 3 ambient temperatures studied (8°C, 15°C, and 22°C), Syrian hamsters presented both kinds of torpor: daily torpor and multi-day torpor. This allowed the time controls of these 2 phenomena to be compared. Daily torpor is controlled by the main clock of the body, located in the SCN. This clock is circadian: its period τ (tau) is close to 24 h, and it is temperature-compensated; i.e., it is nearly independent of temperature. Deviations in τ with temperature were thus constrained to remain within a range compatible with synchronization to 24 h (Aschoff, 1965; Gaston and Menaker, 1967; Hastings and Sweeney, 1957) (Fig. 2). This contrasts with multi-day torpor bouts, where the distribution of torpor bout durations was also normal (Gaussian) but the period increased as Tb decreased (Fig. 3), following a curve similar to the Arrhenius curve of a temperature-dependent, enzyme-controlled chemical reaction. A similar observation had been made previously by Twente et al. (1977) (data analyzed by Malan, 2010) on the multi-day torpor bout durations of 3 species of ground squirrels: Urocitellus columbianus, Ictidomys tridecemlineatus and Callospermophilus lateralis (studied individually). Together, the Arrhenius pattern has thus been documented in 4 species of hibernating mammals, notwithstanding partial data obtained in other mammalian species, including Marsupials and Echidnas in our earlier study (Malan, 2010). The time control of multi-day torpor bout duration would thus involve a catalytic process (or processes) governed by an Arrhenius equation.
Earlier Explanations of Torpor Bout Durations
The control of classic (multi-day) torpor bout duration had remained inconclusive since posited by Dubois in 1896. It was first ascribed to an hourglass mechanism, in sensing the accumulation or depletion of metabolic substrates (French, 1985) or end-products (Beckman and Stanton, 1976; Nemeth et al., 2009; and as reviewed in Ruf and Geiser, 2015). Another line of evidence concerned the time- dependent increase in the sensitivity of various brain areas to the injection of drugs or neuromediators (Beckman and Stanton, 1976; Jinka et al., 2012; Twente and Twente, 1968). In addition, many processes show reductions in energy expenditure during torpor (Storey and Storey, 2011): protein synthesis and degradation are inhibited (Storey and Storey, 2011) and essential proteins are replaced at the end of a torpor bout. By the time of arousal, synapse connectivity, immune function, coagulation, levels of hormones and acid-base balance are all restored to their euthermic levels (Carey et al., 2003; Malan, 2014; Prendergast et al., 2002; von der Ohe et al., 2007). How a single hourglass mechanism could integrate all this information and then distribute an arousal signal to such a variety of functional units remained unsolved.
The Circadian System: Hourglasses Versus Clocks
Recently, considerable progress has been made in terms of our understanding of the control of circadian time in the body. Conservation of circadian rhythmicity in classic hibernation had already been hinted at in 1994 (Canguilhem et al., 1994; Grahn et al., 1994). Clocks based on transcription-translation feedback loops (TTFL) have since been found nearly universally in nucleated cells (Adamovich et al., 2014; McGinnis and Young, 2016). Their oscillations are synchronized within each organ (Jaeger et al., 2015) and functioning is synchronized at the organismal level by a master oscillator located in the SCN. The SCN clock itself is synchronized to astronomical time by the photoperiod and distributes this information through neural and endocrine signals (Zheng and Sehgal, 2015). Circadian clocks are intimately and reversibly coupled to metabolic control (Brown, 2016; McGinnis and Young, 2016; Sancar and Brunner, 2014; Zheng and Sehgal, 2015). The well-known effects of free fatty acids on torpor depth and duration (Adamovich et al., 2014; Geiser, 1991) may intervene at this stage. The dual system of clocks and metabolic control also operates in the sleep-wake cycle (Ray and Reddy, 2016). Together with the intricate interactions between clock and metabolic genes (Ray and Reddy, 2016), the distinction between hourglass and clock time control is losing ground.
Control of the Time Spent in Torpor by the Temperature-dependent Clock
As shown in Figures 4 and 5, in classic hibernation, torpor bout durations, expressed in terms of subjective time of the temperature-dependent clock, do not occur randomly within the subjective 24-h period of that clock. At any Ta, torpor bout durations in subjective time could be directly compared by plotting a common polar plot with a circumference of 24 h of subjective time (angular scale) and a radial scale showing the periods of the clock in astronomical time for the various temperatures (Fig. 4). In this study, most torpor bouts lasted between 6 and 11 h of subjective time at all temperatures. At 22°C, bouts were apparently longer than those at 8°C or 15°C, but at 22°C, the base of the curve was shorter than that at lower temperatures (Fig. 5). This increased the weight of the measurements taken at higher temperatures in the calculated mean and explains most if not all of the apparent difference. Ignoring this artefact, then, it seems that torpor bout durations occurred within a relatively short range of subjective temperature, irrespective of the Ta at which the torpor bouts had been observed. This confirms that torpor bouts were controlled by the temperature-dependent clock.
The Reversibility of the Clock
An important feature of the temperature-dependent clock is the lack of its significant effect on metabolic inhibition (Fig. 3). Because temperature dependence results from a thermodynamic phenomenon—not a complex biochemical process—there is no time delay. This allows for a full and instantaneous reversal of metabolic inhibition at the time of arousal (Fig. 5, arrows). Such reversibility would guarantee a strict return to the period that prevailed at the previous euthermy (Fig. 5), which, in turn, would restore the 24-h periodicity if a suitable synchronizer were available.
The Constraints Imposed by Periodic Arousals: Controlling Metabolic Scope
In euthermy in small mammals, the highest instantaneous increases in MR over the resting rate are observed in running (e.g., to escape a predator) and in cold thermogenesis. In such conditions, time is insufficient for the recruitment of additional enzyme units and the increased level of MR is achieved with preexisting enzymes. The metabolic scope, i.e., the ratio of the maximum MR over the resting MR, is about 10-fold in mammals, with higher exceptions in dogs and horses (Bishop, 1999). In daily torpor, MR is reduced about 4-fold compared with the euthermic resting MR (Ruf and Geiser, 2015). Metabolic scope is thus multiplied by 4, and this is handled with the help of metabolic inhibition (Carey et al., 2003; Storey and Storey, 2011). By contrast, in the multi-day torpor of deep hibernation, torpor MR is reduced 20- to 50-fold compared to euthermic resting MR (Ruf and Geiser, 2015) and this again multiplies the range of reaction rates over which metabolic control has to operate. During sharp increases in MR, such as in insect flight and perhaps in the arousal from torpor, there may be an increase in substrate cycling (“futile cycles”) (Newsholme et al., 1983). But this does not work in reverse, when the animal enters torpor from a resting state; substrate cycling is probably already very low and cannot be further reduced. Metabolic control by the circadian system comprises successive steps (from eating to the final processing of food), each of which occurs within a restricted time interval (Kumar Jha et al., 2015; McGinnis and Young, 2016; Schibler et al., 2015). The circadian clock would thus control the rate at which each step proceeds. Consequently, slowing the (subjective) time of the clock would automatically reduce the rate of all metabolic processes. Heart rate is a good predictor of overall metabolic rate (Green, 2011) and is directly controlled by the circadian system (Buijs et al., 2016; Pittman-Polletta et al., 2013; Wu et al. 2011). When Tb decreases from 37°C to 8°C, the heart period of the Syrian hamster is multiplied by about 40 (Horwitz et al., 2013). This would be explained as the product of a factor of 10 resulting from thermodynamics (Fig. 5), while the remaining factor of 4 would correspond to metabolic inhibition, for which 75% is a reasonable estimate (75% = 100% − 25%) (Brown, 2016; Carey et al., 2003; Ruf and Geiser, 2015; Storey and Storey, 2011). And because of its thermodynamic dependence on temperature, the temperature-dependent clock would follow a path close to that of the heart rate (and overall metabolic rate) to control reaction rates.
The temperature-dependent clock might substitute for the SCN clock at low Tb when transcription stops (Ikeno et al., 2017; Revel et al., 2007). It might involve a peroxiredoxin redox rhythm, such as found in red blood cells (O’Neill and Reddy, 2011). Redox rhythms are better conserved than transcription-translation rhythms at low Tb (Ray and Reddy, 2016).
When the hibernaculum temperature decreases below a critical threshold, freezing can be avoided by increasing metabolic heat production. The animal stabilizes Tb at the expense of a considerable increase in MR (Buck and Barnes, 2000; Ortmann and Heldmaier, 2000). This confirms that Tb and MR may vary independently of each other.
Finally, temperature-dependent clocks and circadian clocks should similarly facilitate synchronization to astronomical time: the lack of influence of body size eliminates a potential source of period variability (Geiser, 2004; Malan, 2010; Ruf and Geiser, 2015; Twente et al., 1977). Woodchucks would apparently differ, because bigger individuals spend less time torpid over the winter, but their shorter bouts are probably explained by their higher Tb (Fig. 3) (Zervanos et al., 2014; Zhou et al., 2015). The case of bears is probably considerably different and will not be addressed here.
Temperature Dependence Versus Temperature Compensation
The mechanism of temperature compensation for the circadian clock has recently been identified. The stability of the PER2 clock protein is regulated by 2 competing casein kinases with different temperature coefficients. This “phosphoswitch” results in a variable balance between a stable and an unstable state of the protein, which provides a temperature compensation (Zhou et al., 2015). Thus, temperature compensation is a design feature of the core SCN clock machinery. Of note, the binding of casein kinase CK1ε to the PER2 protein may be affected in Syrian hamsters by the tau mutation. In tau-/- hamsters, the circadian period is reduced (Ralph and Menaker, 1988; Tosini and Menaker, 1998), but multi-day torpor bout duration is unaffected (Oklejewicz et al., 2001). In cultured retinas from these mutants, temperature compensation of the circadian period is partially restored but the apparent enthalpy of activation, 24.9 ± 2.8 kJ/mole (calculated from Tosini and Menaker, 1998), is far different than that of the temperature-dependent clock, 61.6 ± 2.8 kJ/mole. It is thus likely that the temperature-dependent clock incorporates an older variant of PER2 that lacks this phosphoswitch and is unaffected by the tau mutation, and that the tau mutation affects more recent PER2 variants.
Why Keep the Old Pattern? The Primitive (Plesiomorphic) Character of Hibernation
Finding an old clock pattern in extant hibernating mammals is less surprising in the context of the recent concepts of the evolution of mammalian hibernation. Hibernation is no longer viewed as a polyphyletic set of converging adaptations, which would have occurred in parallel in many clades of mammals. It is now considered as a plesiomorphic trait (Lovegrove, 2012); i.e., the conservation of primitive characters (Malan, 1996; 2014) that would have helped ancestral mammals survive the predation of Dinosaurs throughout the Cenozoic era (Lovegrove, 2012; Lovegrove et al., 2014).
More Efficient Mechanisms for On and Off Transitions
Conserving an old version of a temperature-dependent clock over the geological times requires a periodic revision of its advantages or drawbacks. The ancestors of extant mammalian hibernators have benefitted from the reduced energy requirements of prolonged and deeper multi-day torpor. These implied increased durations of the transition phases, entry and arousal. Both phases had to be kept short because of the risks of predation and of the competition for reproduction. De novo synthesis of proteins is relatively inefficient, especially below the cutoff temperatures for transcription and translation (van Breukelen and Martin, 2001). The development of brown adipose tissue had provided a partial solution (Nedergaard and Cannon, 1984) as well as, presumably, a more extensive role in oxido-reduction (peroxiredoxins) in metabolic control (Reddy and Rey, 2014). The ventilatory control of CO2 (loading on entry, unloading on arousal) (Malan, 2014) avoids a possible conflict between clocks by inhibiting the circadian clock (Walton et al., 2018). In extant hibernating mammals, all these new possibilities would have been subserved by the rapid and reversible control of subjective time supplied by the Arrhenius thermodynamics of a conserved primitive clock.
Perspectives
Further research will be needed to localize the temperature-dependent clock. In deep hibernation, the temperature-dependent clock operates in the presence of respiratory acidosis (Malan, 2014). Acidosis is inhibitory for the SCN clock (Walton et al., 2018). Running experiments at an acidic pH would thus allow the temperature-dependent clock to be selected. Insensitivity to an acidic pH might result from the presence of identifiable components, such as buffers or Na+/H+ antiporters. Although the final confirmatory experiments will need to be conducted at hibernation temperature, the very long period may make the process rather lengthy. But the clock also operates at higher temperatures and some experiments may be run at 37°C or at room temperature once the SCN clock has been inhibited.
Supplemental Material
Malan_Ciocca_Challet_Pevet_Torpor_The_temperature_dependent-clock_(1) – Supplemental material for Implicating a Temperature-Dependent Clock in the Regulation of Torpor Bout Duration in Classic Hibernation
Supplemental material, Malan_Ciocca_Challet_Pevet_Torpor_The_temperature_dependent-clock_(1) for Implicating a Temperature-Dependent Clock in the Regulation of Torpor Bout Duration in Classic Hibernation by André Malan, Dominique Ciocca, Etienne Challet and Paul Pévet in Journal of Biological Rhythms
Footnotes
Acknowledgements
The CNRS and the University of Strasbourg are gratefully acknowledged for financing the publication. No extra funding was provided.
Author Contributions
P.P. and A.M. designed the research. D.C. performed the research. E.C. provided fruitful discussions. A.M. analyzed the data and wrote the paper. All authors critically read the manuscript.
Conflict of Interest Statement
The authors have no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.