Abstract
Most organisms have a circadian system, entrained to daily light-dark cycles, that regulates 24-h rhythms of physiology and behavior. It is unclear, however, how circadian systems function in animals that exhibit seasonal metabolic suppression, particularly when this coincides with the long-term absence of a day-night cycle. The arctic ground squirrel, Urocytellus parryii, is a medium-sized, semi-fossorial rodent that appears above-ground daily during its short active season in spring and summer before re-entering a constantly dark burrow for 6 to 9 months of hibernation. This hibernation consists of multiple week-long torpor bouts interrupted by short (< 20 h) arousal intervals when metabolism and body temperature (Tb) return to normal levels. Here, we used immunohistochemistry to measure the expression of daily or circadian rhythms of the protein products of 3 circadian clock genes, PER1, PER2, BMAL1, and the neural activity marker c-FOS in the suprachiasmatic nucleus (SCN) of arctic ground squirrels before, during, and after the first torpor bout of hibernation. Before torpor, while under 12:12-h light:dark conditions, animals showed significant daily rhythms in their Tb, as well as in protein expression levels of PER1 and PER2, but not BMAL1. Upon entering first torpor (Tb < 30°C), animals were moved into constant darkness. When sampled at 6-h intervals—beginning 24 h after the last light out, with Tb 3°C to 4°C—there were no circadian oscillations in PER1, PER2, or c-FOS expression. Sampling across 24 h during the first spontaneous arousal interval, c-FOS expression was elevated only when Tb reached 20°C and PER1 and PER2 expression did not show any Tb- or time-dependent changes. These results suggest that the central circadian clock might have stopped functioning during hibernation in this species, and the timing of arousal from torpor in arctic ground squirrels is unlikely to be controlled by the circadian clock within the SCN.
The circadian system coordinates daily rhythms in physiology and behavior, allowing organisms to anticipate and synchronize with day-night fluctuations in their environment (Klein et al., 1991). In mammals, the central master clock is in the suprachiasmatic nucleus (SCN) of the hypothalamus and composed of approximately 20,000 neurons that integrate the rhythmic output of neuronal activities and hormonal secretions. Oscillation of the circadian clock is based on transcriptional/translational feedback loops composed of clock genes and their protein products. The circadian clock—molecular gears that do not measure an exact 24 h—relies on external cues to entrain/synchronize its oscillation to the phase of the earth’s rotation. The most salient cue for synchronizing and entraining the SCN clock is the environmental light-dark (LD) cycle.
In hibernating mammals, it is currently unclear the adaptive value of maintaining circadian rhythmicity during prolonged seasonal metabolic suppression and the stage at which animals are sequestered below ground in the absence of a LD cycle. The persistence and function, if any, of circadian rhythms during mammalian hibernation is not well understood and may vary among species (Körtner and Geiser, 2000; Heller and Ruby, 2004). During hibernation, a physiological state characterized by profound suppression of metabolic rate and body temperature (Tb) for energy conservation, there is a global depression of multiple cellular processes, including transcription and translation (Storey and Storey, 2004). In small mammals, hibernation consists of multiday torpor bouts of hypometabolism and low Tb that alternate with arousal episodes when Tb returns to euthermic levels for a few hours to about one day depending on the species (Ruf and Geiser, 2015). Although entries into and arousals from torpor have been observed to be precisely timed in some rodents (Daan, 1973; French, 1977; Zervanos et al., 2009), whether this is driven by a functional circadian clock or environmental cues is unclear. No circadian rhythmicity in Tb during torpor has been detected in several rodent species (Hut, Barnes, et al., 2002; Gür et al., 2009; Williams, Barnes, Buck, 2012); however, very low amplitude (0.1–0.2°C) and irregular circadian rhythms in Tb during torpor have been described in golden-mantled ground squirrels, Callospermophilus lateralis, housed in constant dark conditions (Grahn et al., 1994; Larkin et al., 2002). The irregular rhythms may indicate that the period of the rhythm differs between individual bouts of hibernation. Since the division of the length of each bout by the period of that bout is a whole integer, it appears as if the circadian system is timing the arousal intervals in golden-mantled ground squirrels. It has also been reported that SCN lesions eliminate circadian Tb rhythms, as observed in torpid squirrels under constant light, supporting the role of the SCN in maintaining circadian rhythms during torpor in this species (Ruby et al., 2002).
The SCN maintains a relatively high tissue-specific metabolism as compared with other brain regions during hibernation (Kilduff et al., 1990), and mRNA expression of early immediate genes, including c-fos, is especially elevated in the SCN during the transition between torpor and arousal, with a dramatic peak when Tb reaches 20°C (Bitting et al., 1994; O’Hara et al., 1999; Bratincsák et al., 2007). The expression of c-fos in the SCN is also rapidly induced by a photic signal during the subjective night, accompanied by induction of a circadian clock gene Period1 (Per1) and leading to phase shifts of circadian activity rhythms (Kornhauser et al., 1990; Albrecht et al., 1997). Together with evidence showing that c-fos expression in the SCN is circadian regulated (Guido et al., 1999), the elevation in c-fos during arousal is assumed to be associated with an activation of transcription and a continuing circadian function of the SCN clock during hibernation (Bitting et al., 1994; Bratincsák et al., 2007; van der Veen et al., 2008). This is not supported, however, by the finding that rhythmic mRNA expression of circadian clock genes, Per1, Per2, and Bmal1, is abolished in the SCN during hibernation in European hamsters, Cricetus cricetus (Revel et al., 2007).
The arctic ground squirrel, Urocitellus parryii, the farthest north hibernator, is a semi-fossorial Sciurid rodent patchily distributed throughout NE Siberia in Eurasia and from Alaska to Hudson Bay in North America. They show entrained, diurnal patterns in above-ground activity and Tb during their short, above-ground active season that lasts from May to August, a period that includes > 6 weeks of constant daylight (Long et al., 2005; Williams et al., 2014). Beginning between early August and early October, they isolate themselves in a frozen burrow under constant darkness and hibernate for 6 to 8 months until early spring (Buck and Barnes, 1999b). Hibernation consists of 14 to 17 torpor bouts, each lasting 2 to 3 weeks, with minimum Tb of −2.9°C; torpor bouts are shorter in early and late hibernation (Barnes, 1989; Buck et al., 2008). Torpor bouts are regularly interrupted by arousal episodes when Tb returns to euthermic levels (35°C to 36°C) for 14 to 20 h (Buck et al., 2008). In spring, females emerge from their burrows within 1 to 2 days of ending torpor and return to constant high Tb (Sheriff et al., 2011), whereas males end torpor 4 to 6 weeks earlier and remain underground for several weeks at euthermic Tb while undergoing reproductive maturation before first emergence (Barnes, 1996). In free-living, arctic ground squirrels, circadian rhythms in patterns of Tb have not been detected during hibernation (Williams, Barnes, Richter, 2012), though weak rhythms can occur during post-torpor euthermia before emergence (unpublished data). Significant and robust diurnal rhythms of Tb immediately resume when animals first emerge to the surface and are exposed to light (Williams, Barnes, Richter, 2012). These observations suggest that circadian rhythms do not persist during hibernation, and that light exposure potentiates/accelerates the resumption of the circadian function in this species (Williams, Barnes, Richter, 2012). A lack of circadian rhythmicity in Tb, however, does not necessarily indicate that the central circadian clock is arrested, since it is still possible that molecular oscillations continue within the SCN, but the cellular clocks are desynchronized or output pathways are disconnected during this unique physiological state.
Here, we continuously measured Tb in captive arctic ground squirrels, taking samples in LD conditions before and after constant darkness during the first bout of torpor and then within the subsequent spontaneous arousal interval. We used these samples to examine whether diurnal or circadian rhythmic patterns occurred with the expression of circadian clock proteins, PER1, PER2, and BMAL1, and the neural activity marker, c-FOS, in the SCN. Our results suggest that circadian molecular oscillations cease or become desynchronized once animals enter torpor, and that arousal from torpor is likely not controlled by the circadian clock within the SCN in this species.
Materials and Methods
Animals
Sixty-eight arctic ground squirrels (28 males and 40 females) were captured in the vicinity of the Toolik Field Station (TFS; 68º 38”N, 149º 38”W) located in the northern foothills of the Brooks Range in Alaska in July (2012 to 2014) and transported to the University of Alaska Fairbanks. All animals were initially housed at 20 ± 2°C in 12:12 h light:dark (12:12 LD) conditions. Food (Mazuri Rodent Chow, Land O’Lakes, Inc., Arden Hills, MN, USA) and water were available ad libitum. In early August, animals were implanted with calibrated, temperature-sensitive radiotransmitters (Data Science International, New Brighton, MN, USA) in their abdominal cavity (Richter et al., 2015) and, after 2 weeks of recovery, were transferred into environmental chambers held at 2 ± 1°C in 12:12 LD with food and water provided ad libitum. Tb and activity were continuously monitored using an automated data collection system that records Tb (± 0.1°C) and movement in 10-min bins. In this study, males and females were randomly used for the analysis. All of the procedures used were approved by the Institutional Animal Care and Use Committee of University of Alaska Fairbanks.
Pre-hibernation phase
In late August to early September, brains were collected from a total of 8 males and 16 females, housed at 2°C ± 1°C in 12:12 LD. The brains were collected before the first torpor at 6-h intervals (n = 6 per time point) for 18 h, beginning at light onset (zeitgeber time 0: ZT0; Fig 1).
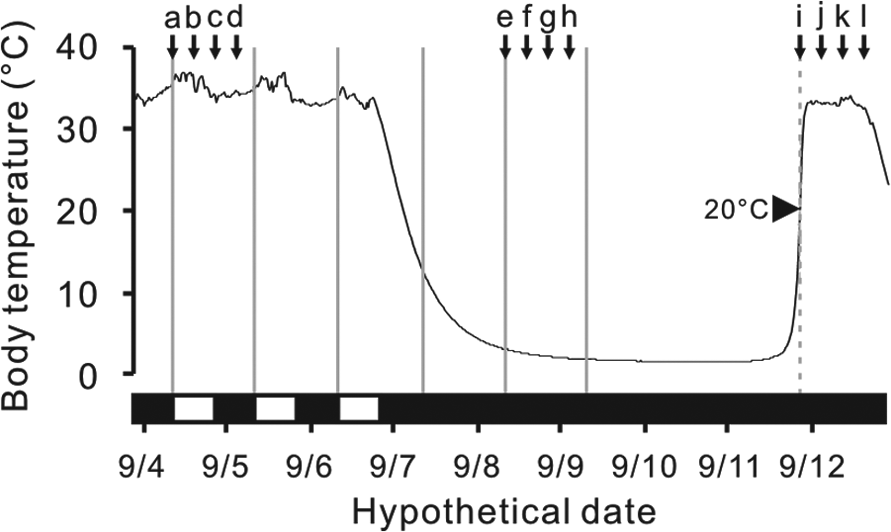
An example of body temperature changes of an arctic ground squirrel and the sampling schedule. Horizontal black bars indicate periods of dark and white bars indicate periods of light. Prior to the first torpor bout, animals were sampled at 6-h intervals after lights-on at zeitgeber time (ZT) 0 (a), ZT6 (b), ZT12 (c) and ZT18 (d). During the first torpor bout, animals were sampled at 6-h intervals after subjective lights on at circadian time (CT) 0 (e), CT6 (f), CT12 (g) and CT18 (h). During spontaneous arousal episodes, animals were sampled at 6-h intervals after body temperature reached 20°C at arousal time (AT) 0 (i), AT6 (j), AT12 (k) and CT18 (l).
Hibernation phase
As animals spontaneously entered their first torpor bout (Tb < 30°C), they were moved to an adjacent environmental chamber kept at 2°C ± 1°C, where disturbance and light exposure were eliminated (constant darkness: DD). Beginning 23 h after entering torpor, brains were collected as described below from animals (13 males and 7 females) at 6-h intervals (n = 5/time point) for 18 h, starting at projected circadian time 0 (CT0). CT0 is the same time of day as ZT0, except lights do not come on, as animals are maintained in constant darkness following entry into torpor (Fig. 1).
Arousal phase
Animals underwent a complete torpor bout and initiated a spontaneous arousal episode in DD. Spontaneous arousals were detected by continuously monitoring Tb. Brains were collected as described below from animals (8 males and 16 females) at 6-h intervals (n = 6/time point) for 18 h, beginning when Tb first reached 20°C (arousal time 0, AT0). The mean number of days in torpor before arousal was 9.2 ± 0.5 days.
Histological Analysis
Animals were euthanized with intraperitoneal sodium pentobarbital (200 mg/kg) and perfused intracardially using 100 ml saline followed by 200 ml 4% paraformaldehyde in 0.1 M phosphate buffer. Brains were post-fixed for 12 to 18 h, cryoprotected in a 20% sucrose solution, and shipped to Michigan State University, where 4 alternate sets of 30-μm sections were cut through the entire SCN from each brain using a cryostat.
Immunohistochemistry (IHC)
Each set of the free-floating sections was incubated in primary antibody against mouse PER1 (raised in rabbit, 1:1,000, AB2201, Millipore; Billerica, MA, USA), mouse PER2 (raised in rabbit, 1:1,000, AB2202, Millipore), BMAL1 (raised in Guinea pig, 1:1,000, NG1544109, Millipore), or c-FOS (raised in rabbit, 1:20,000, ab11959, Abcam; Cambridge, MA, USA), and then incubated in biotinylated goat anti-rabbit IgG secondary antibody (1:1000, BA-1000, Vector Laboratories; Burlingame, West Grove, PA, USA) for PER1 and PER2, biotinylated goat anti-Guinea pig IgG secondary antibody (1:1000, BA-7000, Vector Laboratories) for BMAL1, or biotinylated donkey anti-rabbit IgG secondary antibody (1:1000, 711-066-052, Jackson Immunoresearch Laboratories Inc.) for c-FOS. Sections were processed using the avidin-biotin-immunoperoxidase technique with 3, 3-Diaminobenzidine tetrahydrochloride (DAB, Sigma-Aldrich; St Louis, MO, USA) enhanced with 4% nickel sulfate as the chromogen. Animals that were compared directly were processed together with a unique cut to mark each brain. Following the IHC reaction, the sections were mounted on slides, rinsed with ethanol and xylene, and coverslipped with Permount (Thermo Fisher Scientific; Waltham, MA, USA).
IHC data analysis
Images of mid-SCN sections were captured using a light microscope (Zeiss; Gottingen, Germany) and a CCD video camera (CX9000, MBF Bioscience; Williston, VT, USA). The immunoreactive nuclei were counted in the SCN using NIH Image J program (NIH, Bethesda, MD), as described previously (Yan and Silver, 2004; Yan, 2011). The immunoreactive nuclei were counted bilaterally within the area, which was set consistently across the sections, and the average was used as the value for each animal.
Statistical Analysis
Temporal changes in Tb and protein expression were analyzed by a one-way analysis of variance (ANOVA) followed by the Tukey HSD test. When the variances among the groups were not equal (Bartlett’s test, p < 0.05), the data were analyzed by the Kruskal-Wallis test, and the Steel-Dwass test was used to further evaluate group differences. Tb data in arousal animals were compared among AT6, AT12, and AT18. Mean differences were considered statistically significant when p < 0.05.
Results
Pre-hibernation Phase
Before entering hibernation, arctic ground squirrels housed in LD cycles showed a clear diurnal rhythm in Tb (effect of time, p < 0.01, one-way ANOVA), peaking at the middle of the day and with an amplitude of approximately 2°C (Fig. 2A). In the SCN, PER1 was rhythmically expressed (effect of time, p = 0.037, one-way ANOVA) with the highest value at the beginning of the dark phase (Fig. 2B). PER2 expression also showed diurnal variations (effect of time, p = 0.021, one-way ANOVA) with the highest value at ZT12 (Fig. 2C). However, no diurnal variations were detected in BMAL1 expression (effect of time, p = 0.86, one-way ANOVA) (Fig. 2D).
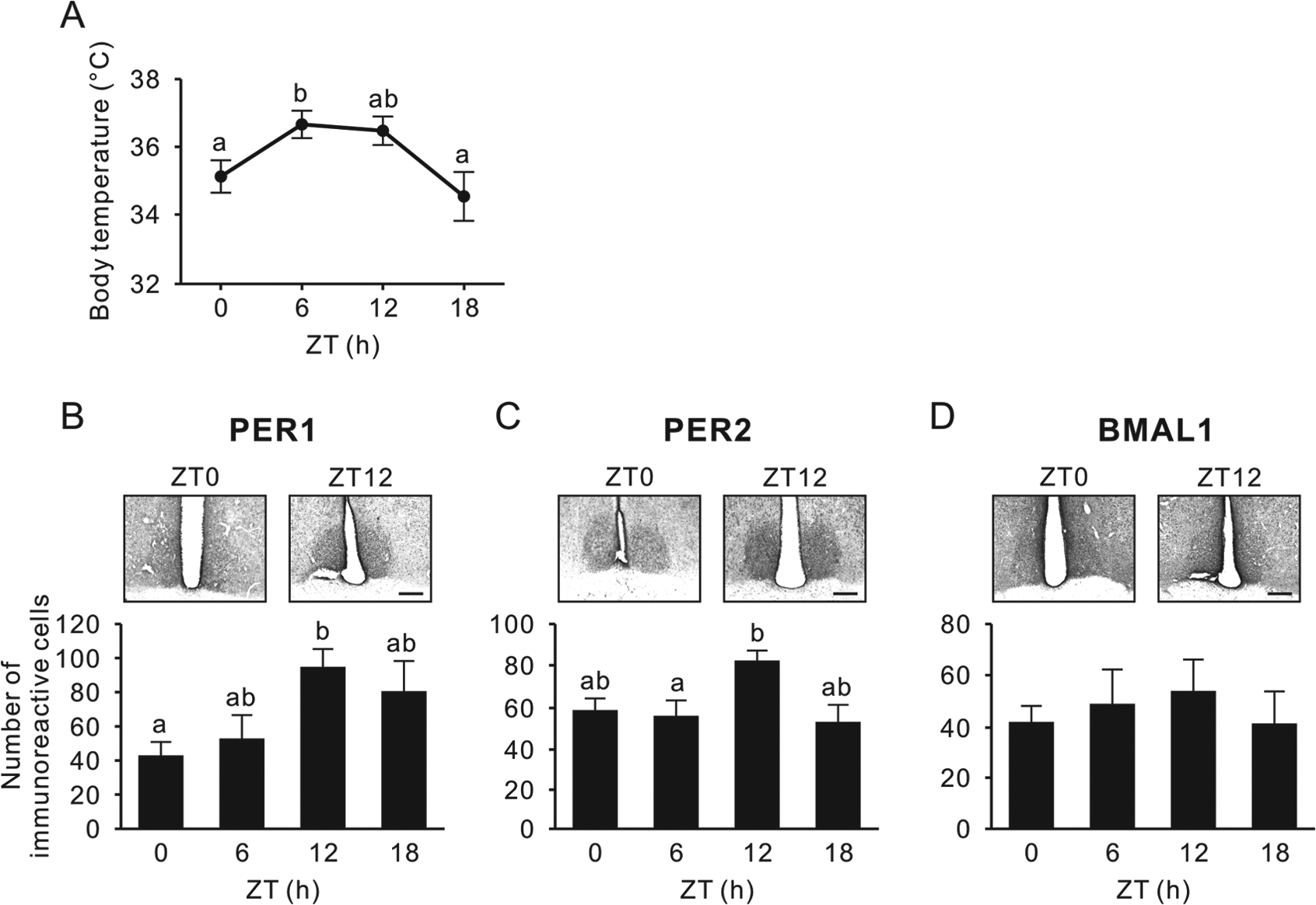
Body temperature and PER1, PER2, and BMAL1 expression in the SCN before entering hibernation. (A) Body temperature when the animals were euthanized at zeitgeber time (ZT) 0, 6, 12, and 18 in 12:12 LD before initiating their first torpor bout. (B-D) Representative photographs depicting PER1 (B), PER2 (C), and BMAL1 immunoreactivity (D); the respective number of immunoreactive nuclei (mean ± SEM) in the SCN are shown. Values with different letters are significantly different (p < 0.05, Tukey’s post hoc test). Scale bar, 200 µm.
Hibernation Season
As each animal initiated its first torpor bout (Tb decreasing below 30°C), it was transferred to DD. At 23 h after entering torpor, Tb was 3°C to 4°C and showed no significant variation over the subsequent 18 h, as animals were sampled each 6 h (p = 0.051, Kruskal-Wallis test) (Fig. 3A). Similarly, there were no significant differences in levels of PER1 (Fig. 3B), PER2 (Fig. 3C), or c-FOS expression (Fig. 3D) in the SCN over this sampling interval (p = 0.36, p = 0.076, and p = 0.16, respectively, one-way ANOVA). There was no correlation between time elapsed after torpor initiation and Tb, or PER1, PER2 and c-FOS (p = 0.18, p = 0.16, p = 0.19, and p = 0.21, respectively, Pearson’s correlation test) (Fig. 4).
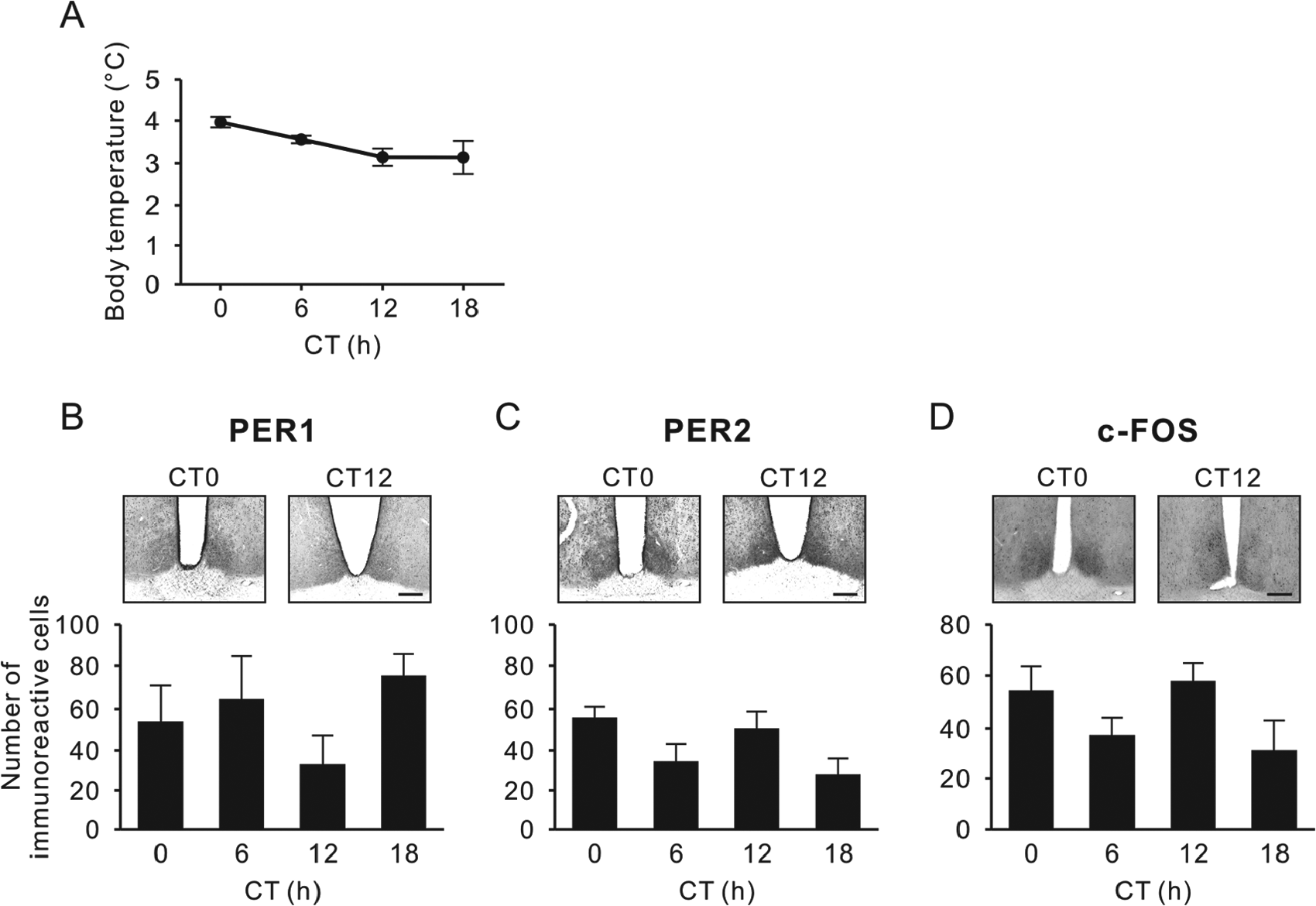
Body temperature and PER1, PER2, and c-FOS immunoreactivity in the SCN during torpor. (A) Body temperature when the hibernating animals were euthanized at projected circadian time (CT) 0, 6, 12, and 18 in constant darkness. No significant differences were found for each protein across the 4 time points (p > 0.05, Steel-Dwass post hoc test). (B-D) Representative photographs depicting PER1 (B), PER2 (C), and c-FOS immunoreactivity (D), also showing the respective number of immunoreactive nuclei (mean ± SEM) in the SCN. Scale bar, 200 µm.
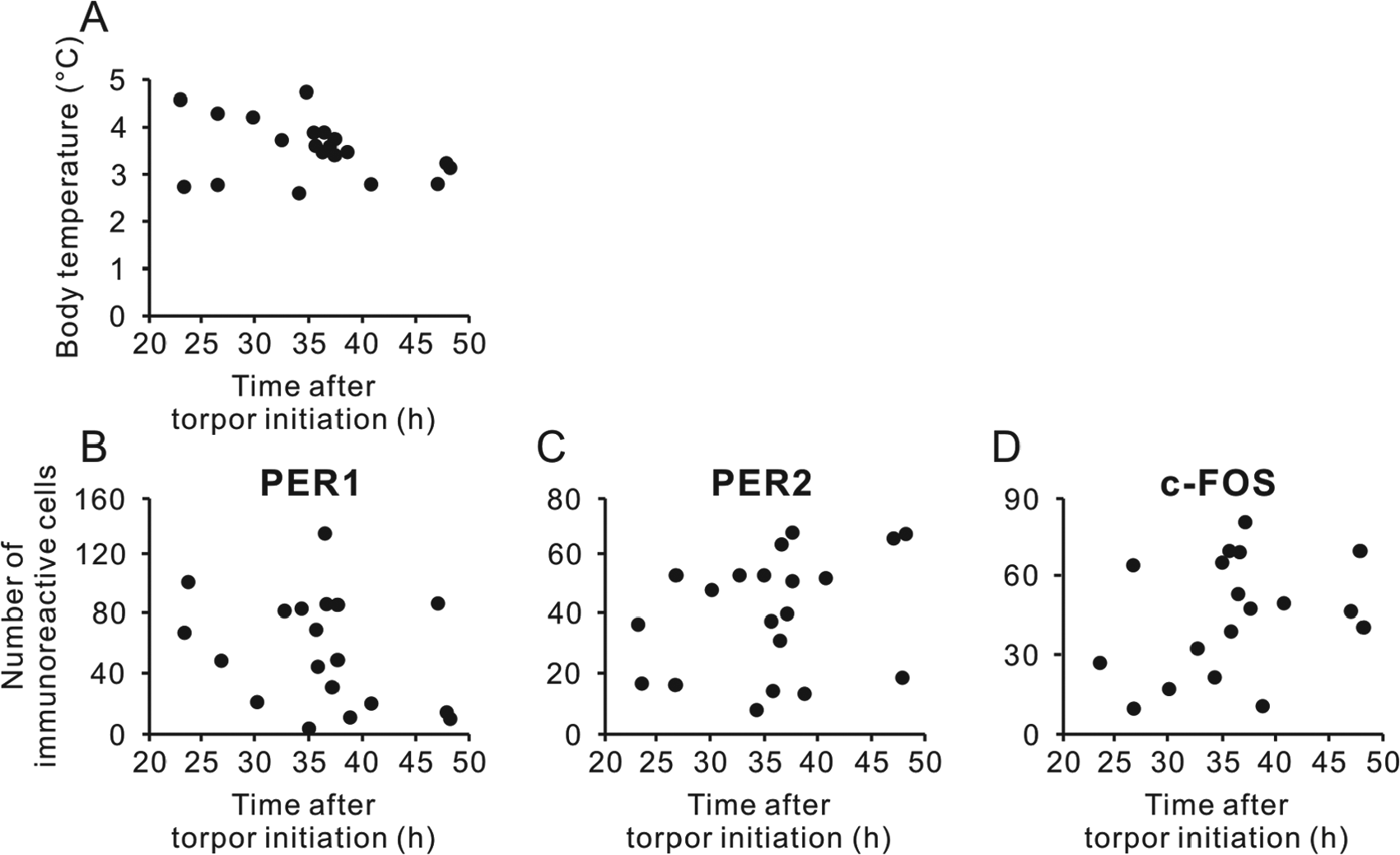
Time after torpor initiation and body temperature and immunoreactivity of PER1, PER2, and c-FOS in the SCN. Individual data of body temperature (A) and number of PER1- (B), PER2- (C), and c-FOS-immunoreactive nuclei (D) are plotted.
Arousal Phase
The completion of the first torpor bout was marked by a spontaneous arousal episode, detected by steadily increasing Tb that returned to euthermic levels within 6 h and then did not significantly change over the next 12 h (p = 0.41, one-way ANOVA) (Fig. 5A). In the SCN, PER1 and PER2 expression also did not show significant arousal time (AT)-related changes over 18 h (p = 0.98 and p = 0.068, respectively, one-way ANOVA) (Fig. 5B, C). A greater than 2-fold upregulation of c-FOS expression was detected in the dorsal part of the SCN at AT0 (p < 0.01, one-way ANOVA) (Fig. 5D). There were no correlations between Tb or protein expression in the SCN and external time when animals were euthanized (Supplementary Online Material; Suppl. Fig. S1).
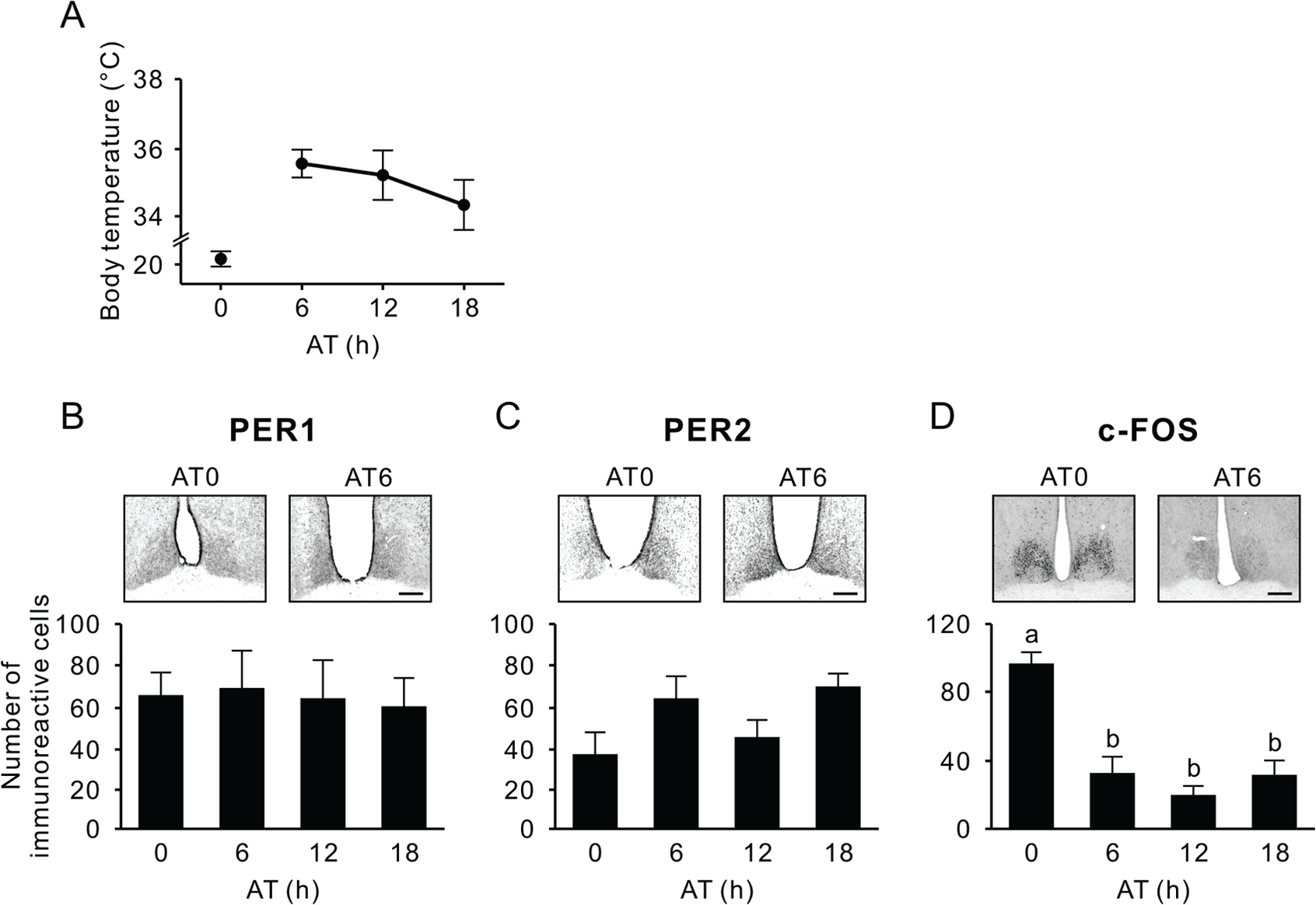
Body temperature and PER1, PER2, and c-FOS immunoreactivity in the SCN during the arousal phase. (A) Body temperature when the animals were euthanized at arousal time (AT) 0, 6, 12, and 18 in constant darkness. (B-D) Representative photographs depicting PER1 (B), PER2 (C), and c-FOS immunoreactivity (D), also showing the respective number of immunoreactive nuclei (mean ± SEM) in the SCN. Values with different letters are significantly different (p < 0.05, Tukey’s post hoc test). Scale bar, 200 µm.
Discussion
We found that, whereas Tb and clock proteins in the SCN showed diurnal variation under an LD cycle in euthermic arctic ground squirrels, no significant circadian oscillations were observed in Tb or clock protein expression during their first multi-day torpor bout. Moreover, we found that arousal from torpor did not trigger or coincide with molecular oscillations of clock proteins in the SCN; although, c-FOS expression was acutely induced during rewarming. Our results suggest that circadian function of the SCN may not persist once torpor is initiated and induction of c-FOS in the SCN during early arousal is likely independent of the circadian clock in this species.
Circadian clocks allow animals to coordinate and synchronize the phasing of physiological and behavioral processes with external daily fluctuations. Circadian function appears to be important to arctic ground squirrels, as they maintain entrained daily rhythms of Tb and behavior throughout their summer active season, which includes over 6 weeks of continuous daylight (Long et al., 2005; Williams, Barnes, Buck, 2012). This contrasts with the activity patterns of 2 other indigenous arctic animals, the Svalbard ptarmigan (Lagopus mutus) and reindeer (Rangifer tarandus). In ptarmigans, daily rhythms in feeding activity were not observed in summer and mid-winter when the sun was constantly above or below the horizon (Reierth and Stokkan, 1998). In reindeer, daily activity rhythms were lost during constant lighting conditions in summer and winter (van Oort et al., 2005). These data should be interpreted cautiously, as dampened rhythms were still detected in some animals. In the present study, we found that diurnal rhythmicity in Tb occurs in captive arctic ground squirrels housed in 12:12 LD cycles, together with PER1 and PER2 expression, which peaks in their SCN at the beginning of the night. This temporal profile of PER1 and PER2 in the SCN is similar to that found in a variety of diurnal and nocturnal rodent species kept in LD cycles or DD conditions (Field et al., 2000; Ramanathan et al., 2006; Harbour et al., 2013); although, the amplitude of PER1 and PER2 expression levels in our study over a 24-h cycle was low (less than 2-fold) as compared with that measured in other non-arctic diurnal or nocturnal rodents (Field et al., 2000; Ramanathan et al., 2006; Harbour et al., 2013). This discrepancy could be due to reduced synchronization of cellular oscillators within the SCN, and thereby weaker expression in this species, a trait that may be characteristic of arctic animals (van Oort et al., 2005). BMAL1 expression in the SCN was not rhythmic in arctic ground squirrels, consistent with studies of mouse SCN (Von Gall et al., 2003; Ansari et al., 2009; but also see Shearman et al., 2000; Wyse and Coogan, 2010). These results show that, at least in the presence of LD cycles, the molecular clockwork in the arctic ground squirrel SCN is conserved and drives rhythmic outputs.
Each year under natural conditions, arctic ground squirrels are sequestered below ground under constant darkness over a 6- to 9-month hibernation season (Buck and Barnes, 1999a; Williams, Barnes, Richter, 2012). Consistent with our previous findings of a lack of circadian rhythmicity in Tb during hibernation (Williams, Barnes, Buck, 2012; Williams, Barnes, Richter, 2012), we found no evidence for rhythmic, time-dependent changes in PER1, PER2 or c-FOS expression in the SCN of captive ground squirrels during steady-state torpor. There are a few possible explanations that could account for the absence of circadian rhythms observed in the present study. First, it should be noted that, due to the methodological limitation of a relatively small sample size (n = 5) and individual differences, the power of our analysis was limited in detecting rhythms of low or dampened amplitude. Next, it is possible that the lack of observed rhythmicity at the group level was due to individual differences in circadian phase, with persistence in the oscillations of protein expression occurring in individual animals that are out of phase with each other. However, ground squirrels were entrained together under the 12:12 LD cycle just before first entering torpor and had experienced DD for only 23 h to 53 h before samples were taken; this is a short interval for their circadian clocks to have drifted substantially away from one another. In other rodent species, internal, free-running periods of wheel-running activity in DD are 23.68 h ± 0.06 h for mice (Refinetti, 2004), 24.05 h ± 0.07 h for golden hamsters (Weinert et al., 2001), and 23.87 h ± 0.04 h for diurnal Nile grass rats (Refinetti, 2004); under these conditions, de-synchronization among free-running individuals does not become evident within 2 days due to these small variations in period length among individuals. In arctic ground squirrels kept in constant darkness, we have measured free-running periods of 23.35 h to 24.05 h in captivity (unpublished data). Thus, we conclude that it is unlikely that differences in free-running periods among individual arctic ground squirrels led to the lack of measurable group differences in PER1 or PER2 expression within 2 days of initiating torpor; although, this remains a possibility if low brain temperatures altered the period of the rhythm of protein expression in an irregular manner.
We also consider that a subset of SCN cells continued circadian oscillations in protein expression, but that analyzing the entire SCN, as done in this study, may not be sensitive enough to reveal such an effect. De-synchronization among multiple oscillators anatomically separated within the SCN, such as left-right lobes and dorsal-ventral subregions, has been reported in some rodents under specific lighting conditions, including constant light, 22-h LD cycles or 24-h LDLD cycles (de la Iglesia et al., 2000; de la Iglesia et al., 2004; Ohta et al., 2005; Yan et al., 2005; Yan et al., 2010). We did not detect any regional differences, at least between left and right or dorsal and ventral SCN, in the present study. However, if circadian rhythms with highly irregular periods are generated as a result of variability in the subset of neurons that remain coupled during torpor, this would not be detectible using our methodology. Last, it is also plausible that circadian oscillations of circadian clock protein expression are arrested during torpor in arctic ground squirrels, in which case, the immunoreactivity observed during torpor or arousal was from proteins accumulated before entering torpor. An absence of rhythms in clock gene expression was also reported in the SCN of hibernating European hamsters over multi-day torpor (Revel et al., 2007). While the low individual variability in clock-gene mRNA in the study by Revel et al. (2007) suggests that the circadian clock was arrested at a particular phase, we did not find support for this, as protein expression in the SCN of arctic ground squirrels varied widely among torpid (Fig. 3, 4) and aroused individuals (Fig. 5, S1), both within and across groups. This variation could reflect differences in when the synthesis of clock proteins was arrested or differences in the rates of protein degradation among individuals as animals entered torpor; albeit, rates of Tb cooling into torpor were similar, and rates of proteolysis are greatly reduced during torpor in ground squirrels (Velickovska et al., 2005). Indeed, we did not find any correlation between protein levels and time after entering torpor (Fig. 4). Thus, we suggest the persistence of immunoreactive cells during torpor stems from the lack of protein breakdown within the SCN. This would be similar to what occurs in synapses: Even though entry into torpor is associated with a 50% to 65% loss of synapses, synaptic protein is not lost but instead becomes dissociated and forms a reservoir for reforming synapses during the animal’s subsequent return to euthermia (von der Ohe et al. 2007).
An absence of circadian rhythms under constant darkness or constant light has been demonstrated in polar animals, such as Svalbard reindeer (van Oort et al., 2005), Svalbard ptarmigan (Reierth and Stokkan, 1998), and Antarctic midges (Kobelkova et al., 2015). However, it is likely that the lack of rhythmicity in the SCN of arctic ground squirrels held under DD is attributed to torpor state and not to constant conditions, because this species continues clear Tb rhythms throughout their active season, including during the several weeks with constant sunlight in summer (Williams, Barnes, Buck, 2012). Both transcription and translation appear to be globally depressed during torpor in mammals (Storey and Storey, 2004). In torpid golden-mantled and arctic ground squirrels, polysome disaggregation begins and translation is suppressed when Tb decreases below 18°C (Knight et al., 2000; van Breukelen and Martin, 2001). Nevertheless, we clearly observed protein expression in the SCN of torpid squirrels at a comparable level to that seen in euthermic animals, probably because factors involved in protein degradation were also not functional and therefore proteins in the SCN were preserved during torpor (Velickovska et al., 2005).
During their first spontaneous arousal from torpor, the Tb of arctic ground squirrels returned to euthermic levels within 6 h, and for 18 h thereafter, there were no time-related changes in Tb. Although circadian rhythmicity in Tb and activity patterns gradually reappear 5 to 15 days after hibernation ends in European ground squirrels (Spermophilus citellus) kept under dim light conditions (Hut, Van der Zee, et al., 2002), field recordings have shown that, in arctic ground squirrels, circadian Tb rhythms are very weak or absent after the termination of hibernation, and the development of robust Tb rhythms coincides with their first emergence to the surface (Williams, Barnes, Buck, 2012; Williams, Barnes, Richter, 2012); this, too, has been reported in free-living golden mantled ground squirrels (Healy et al., 2012). Collectively, this suggests that light exposure either triggers oscillations in the SCN or potentiates circadian Tb rhythms by aiding in the synchronization of oscillating cells within the SCN.
Although a role for the SCN in maintaining Tb rhythms and regulating the timing/frequency of arousal intervals has been suggested by previous studies in the golden-mantled ground squirrel (Grahn et al., 1994; Larkin et al., 2002; Ruby et al., 2002), the results in the present study support the view that arousal episodes in DD are not temporally regulated by the circadian clock within the SCN in arctic ground squirrels. However, it is possible that arousal is gated by circadian-independent mechanisms within or beyond the SCN.
During arousal from torpor in golden-mantled and 13-lined ground squirrels, c-fos mRNA is acutely elevated in the SCN when Tb reaches 20°C (Bitting et al., 1994; O’Hara et al., 1999; Bratincsák et al., 2007), consistent with our result showing strong immunoreactivity of c-FOS in the SCN only at AT0, when Tb reached 20°C. Rapid induction of c-fos expression is also observed when rodents are exposed to light during the subjective night, resulting in phase shifts in circadian activity rhythms (Kornhauser et al., 1990; Albrecht et al., 1997). Thus, it has been assumed that c-fos induction by arousal is associated with circadian function of the SCN during this phase (Bitting et al., 1994; Bratincsák et al., 2007). The present study, however, revealed that c-FOS expression during arousal was mostly restricted to the dorsal SCN, whereas a photic activation of c-fos is observed in the ventral SCN (Albrecht et al., 1997). Moreover, Per1 is also known to be acutely induced by light in the SCN (Albrecht et al., 1997) but PER1 expression was not elevated during arousal in the present study. These results suggest that underlying mechanisms of c-FOS induction are not identical between arousal and light stimulus. During the first arousal episode, the expression of both PER1 and PER2 in the SCN did not show any temporal changes related to time after the Tb reached 20°C or external or subjective time. Taken together, we conclude that it is unlikely that the circadian molecular oscillation resumes after arousal, and that c-FOS induction during the arousal phase is independent of the clock function of the SCN. Once the Tb returned to the euthermic level, arctic ground squirrels enter the next bout of torpor, usually within 15 h (Barnes and Ritter, 1993), and this cycle is repeated for more than half a year (Buck and Barnes, 1999b).
In summary, we demonstrate that, although clearly entrained daily rhythms in PER1 and PER2 were evident before animals entered hibernation, circadian oscillation of these clock proteins was not observed in the SCN during initial torpor bouts or during arousal intervals in hibernating arctic ground squirrels. This suggests that the SCN clock may have turned off during hibernation in arctic ground squirrels, and that the timing or interval of arousal episodes during torpor may not be controlled by the clock function of the SCN. This outcome is consistent with the adaptive significance of hibernation, during which a regulated minimization of systemic energy expenditure might be compromised by a persistent daily alerting influence of the circadian clock.
Footnotes
Acknowledgements
The authors thank Jeanette Moore and Widya Adidharma for excellent technical assistance. This work is supported by National Science Foundation grant IOS-1147187 to CTW and CLB and IOS-1147232 to BMB and LY.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
