Abstract
The safety and efficacy of chemotherapeutics can vary as a function of the time of their delivery during the day. This study aimed to improve the treatment of glioblastoma (GBM), the most common brain cancer, by testing whether the efficacy of the DNA alkylator temozolomide (TMZ) varies with the time of its administration. We found cell-intrinsic, daily rhythms in both human and mouse GBM cells. Circadian time of treatment affected TMZ sensitivity of murine GBM tumor cells in vitro. The maximum TMZ-induced DNA damage response, activation of apoptosis, and growth inhibition occurred near the daily peak in expression of the core clock gene Bmal1. Deletion of Bmal1 (Arntl) abolished circadian rhythms in gene expression and TMZ-induced activation of apoptosis and growth inhibition. These data indicate that tumor cell-intrinsic circadian rhythms are common to GBM tumors and can regulate TMZ cytotoxicity. Optimization of GBM treatment by timing TMZ administration to daily rhythms should be evaluated in prospective clinical trials.
Glioblastoma (GBM) is the most common and aggressive malignant primary brain tumor in adults. Despite extensive research and clinical trials, median survival remains about 15 months (Stupp et al., 2009). Therefore, all opportunities to improve outcomes should be pursued. In 2005, a landmark paper demonstrated a 2.5-month increase in median survival and an increase in the 2-year survival rate (10% to 27%) by treating GBM with TMZ chemotherapy during and after radiotherapy (Stupp et al., 2005). Since then, TMZ has been a standard component of GBM treatment. Investigating opportunities to enhance the antitumor efficacy of TMZ has significant potential for expedited translation to patients.
The efficacy and tolerability of cancer chemotherapeutics at different times of day have been investigated. Drug administration based on daily biological rhythms is known as chronotherapy. Chrono-chemotherapy has increased 5-year survival rates in acute lymphoblastic leukemia and increased the objective response rate in colorectal cancer while reducing toxicities (Levi et al., 1995; Schmiegelow et al., 1997). Chronotherapy has never been applied to the treatment of brain tumors. Standard administration of TMZ is a single, daily dose for 5 consecutive days at the start of every 28-day treatment cycle (Newlands et al., 1992; Stevens et al., 1987). The time of day of TMZ administration has not been tested as an independent variable in analyses of outcome or toxicity. Its oral administration and 1.8-h half-life in plasma (Beale et al., 1999) make TMZ an ideal candidate for chronotherapy.
Daily rhythms in sleep, hormone release, and other processes can affect drug efficacy and toxicity (Vitaterna et al., 2001). The foundation of daily rhythms is a molecular clock that generates near 24-h oscillations in gene expression through BMAL1- and CLOCK-mediated transcription of circadian genes including the Period (Per1, Per2, and Per3) and Cryptochrome (Cry1 and Cry2) loci. With a delay of about 12 h, the PER and CRY proteins accumulate and repress BMAL1/CLOCK-dependent transcription of genes including Per and Cry (Reppert and Weaver, 2002). This feedback loop creates daily oscillations in approximately 50% of genes (Zhang et al., 2014). Transcription of Bmal1 (Arntl) peaks in anti-phase to the Period genes (Nakajima et al., 2004; Preitner et al., 2002; Sato et al., 2004), and loss of Bmal1 abolishes molecular and cellular circadian rhythms (Bunger et al., 2000).
We aimed to test whether GBM cells exhibit intrinsic circadian rhythms in gene expression and their response to chemotherapy. Using an in vitro model of GBM, we examined the role of Bmal1 in daily rhythms in Per2 expression and TMZ-induced DNA damage.
Materials and Methods
Human GBM Cell Culture
Low-passage primary human GBM cells were obtained and used in accordance with a Washington University Institutional Review Board (IRB)–approved Human Studies Protocol (#201102299). They were maintained as adherent cultures on laminin (Sigma L2020, St Louis, MO) coated tissue culture plates in RHBA media (Clonetech, Mountain View, CA), supplemented with 20 ng/mL epidermal growth factor (EGF; Sigma) and 20 ng/mL basic fibroblast growth factor (bFGF; Chemicon, Temicula, CA) as described previously (Barone et al., 2014).
The Cancer Genome Atlas (TCGA) Queries
Details of core clock gene mutations in GBM were obtained by querying TCGA through cbioportal.org. Three datasets were queried: TCGA provisional with 604 samples, TCGA 2013 with 206 samples, and TCGA 2008 with 580 samples. We searched for mutations-only in the following genes: CLOCK, ARNTL, ARNTL2, NPAS2, CRY1, CRY2, PER1, PER2, PER3, CSNK1D, CSNK1E, RORA, RORB, RORC, NR1D1, and NR1D2.
Clock Gene Sequencing
Total RNA was isolated using the RNeasy Mini system and then treated with DNase I according to the manufacturer’s instructions (Qiagen, Germantown, MD). Then 1.5 µg of total RNA from cultured human GBM cells was reverse transcribed with the SuperScript III using random hexamers and Oligo(dT) (Invitrogen); 50 ng of this reaction served as template for quantitative real-time RT-PCR analysis using iTaq Universal SYBR Green Supermix PCR reagents (Biorad). Primers (Suppl. Table S1) were designed by DS Gene software (Accelyrs, Inc., San Diego, CA) to include the specific mutation of interest identified from the analysis of clock gene mutations in the TCGA database and had the following parameters: 18-27 bases, product 100-200 bp, and product melting temperatures within 66-78 °C. PCR reaction products were cleaned using the QIAquick PCR Purification Kit according to the manufacturer’s instruction. Gene amplicons were verified by size on a DNA agarose gel. Samples were then sequenced for mutation(s) using Genewiz services (South Plainfield, NJ).
Animals
Animals were used in accordance with National Institutes of Health guidelines following protocols approved by the Washington University Animal Studies Committee. NCR nude mice (Taconic Farms, Inc., NY) and Nf1flox/flox;GFAP-Cre were housed under a 0700 h lights-on, 1900 h lights-off schedule.
Male Astrocyte Cultures
As a cellular model of mesenchymal GBM (mes-GBM), primary cultures of astrocytes were prepared from male, postnatal day 1 Nf1flox/flox;GFAP-Cre mice and rendered null for p53 function as previously described (Sun et al., 2014; Warrington et al., 2007). Due to increased risk for mesenchymal GBM in male mice and humans (Sun et al., 2014), we limited our studies to male mes-GBM astrocytes.
Expression of CRISPR-Cas9 Expression Vectors
The Genome Engineering Center at Washington University in St. Louis designed and cloned 6 guide RNAs targeting and disrupting the Bmal1 locus (Bmal1 KO). Bmal1 WT and KO cultures were used at the same passage number in parallel experiments.
Expression of Per2-luc and Bmal1-luc Reporters
We infected astrocyte cultures with lentiviral reporter constructs expressing firefly luciferase driven by the mouse Bmal1 (Bmal1-luc) (Liu et al., 2008; Zhang et al., 2009) or Period2 (Per2-luc) (Ramanathan et al., 2012) promoters (generous gifts of Dr. Andrew Liu, University of Memphis).
Expression of Casp-luc Reporter
Mes-GBM astrocyte cultures were transfected with the Caspase-3/7-luciferase plasmid (Casp-luc; generous gift of Dr. Alnawaz Rehemtulla, University of Michigan) (Galban et al., 2013) using Fugene 6 (Promega, Madison, WI). Stable lines were selected with 400 µg/mL G418 (Santa Cruz Biotechnology, Dallas, TX) and maintained in CO2-buffered DMEM supplemented with 10% FBS, 1% penicillin/streptomycin. Cultures were plated and entrained to temperature cycles of 30 to 34 °C every 12 h for 48 h prior to initiation of treatment. We performed 4 independently plated experiments with Bmal1 WT and Bmal1 KO cultures from 2 lots of mes-GBM cells. Of the 8 independent cultures recorded, 1 Bmal1 WT and 1 Bmal1 KO cultures were excluded because they did not respond to TMZ. Bioluminescence signals from TMZ-treated cells were first normalized to their DMSO control by subtraction. To compare caspase activity across treatment time points, all cultures were normalized to the first phase of treatment (falling phase).
γH2AX Immunofluorescence Staining and Quantification
γH2AX staining was used to measure DNA repair response in mes-GBM astrocytes. Cells plated on poly-D-lysine-coated glass coverslips were fixed with 4% paraformaldehyde, permeabilized with Triton-X-100, incubated with mouse anti-phospho-S139 H2AX primary antibody (1:800; Molecular Probes, Waltham, MA) for 3 h at 37 °C, and incubated with Alexa Fluor 568 Donkey anti-mouse IgG (1:10,000; Life Technologies, Carlsbad, CA) for 1 h at room temperature. Astrocyte nuclei were stained with DAPI (Life Technologies). Nuclear γH2AX staining intensity was quantified by 2 individuals blinded to treatment conditions by ImageJ analysis of integrated density of fluorescent images. Staining across 5 high-powered fields of view per coverslip was averaged across 2 coverslips per treatment condition. Thresholds for positive staining were defined by the intensity distributions of TMZ- versus DMSO-treated cells.
Bioluminescence Recordings In Vitro
We detected light from clock gene reporters (Bmal1-luc or Per2-luc) with photomultiplier tubes (HC135-11; Hamamatsu Corp., Bridgewater, NJ) in light-tight incubators (Beaule et al., 2011; Marpegan et al., 2009; Prolo et al., 2005). We integrated bioluminescence every 6 min over a 4- to 5-day experimental period. During recordings, we sealed lids with vacuum grease and maintained cultures at 34 °C in bioluminescence recording medium (HEPES-buffered DMEM supplemented with 10% FBS), B27 (1X; Gibco/Life Technologies, Carlsbad, CA), and 0.1 mM D-luciferin (Xenogen, Alameda, CA), as previously reported (Marpegan et al., 2009). Bioluminescence from GBM cultures was recorded with a low-light imaging system (Stanford Photonics, Palo Alto, CA) consisting of a light-tight incubator coupled to an ICCD camera (XR/Mega10-Z, Stanford Photonics) controlled with Micro-Manager software (Edelstein et al., 2014). Cells were plated in laminin-coated 96-well plates (2.5 × 104 cells per well) and maintained in a mixture of 50% bioluminescence recording medium and 50% RHBA supplemented with 20 ng/mL EGF and 20 ng/mL bFGF. Images were obtained by integrating light every 3 min and then processed using ImageJ software to obtain the average signal intensity for each well every half hour. In Casp-luc experiments, we entrained cultures by shifting the temperature between 30 °C and 34 °C every 12 h for 48 h (Buhr et al., 2010).
Statistical Analysis
Comparisons between treatments and genotypes (GraphPad Prism version 6.0; GraphPad, San Diego, CA) considered astrocytes derived from a single litter of mice as a single biological replicate. We performed statistical analyses on at least 3 biological replicates per experiment, with each lot derived from an independent litter of pups. Circadian period of bioluminescence recordings was analyzed with Chronostar V2.0 software (gift of A. Kramer and B. Maier, Charite, Berlin).
Results
Human GBM Cells Are Circadian
To determine whether GBM cells have intrinsic daily rhythms, we characterized expression of the core circadian gene, BMAL1 (also called Arntl), in human primary low-passage GBM cell lines. Using a transgenic bioluminescent reporter, we recorded BMAL1 promoter activity continuously for 5 days from GBM cells cultured from 5 patients (Fig. 1). We found daily rhythms in BMAL1 expression from all 5 lines with circadian periods ranging from 22.5 to 27.8 h. Because altered clock gene expression in human cancers has implicated circadian rhythms in tumor progression (Stevens, 2005; Wang et al., 2014), we used the Cancer Genome Atlas database to explore known mutations in core clock genes of human GBM tumors (Brennan et al., 2013). Twenty-six missense, 5 nonsense, and 1 in-frame deletion were identified in 16 genes critical for circadian timing (CLOCK, ARNTL, ARNTL2, NPAS2, CRY1, CRY2, PER1, PER2, PER3, CSNK1D, CSNK1E, RORA, RORB, RORC, NR1D1, and NR1D2) in 1390 GBM samples (Suppl. Fig. S1). Thus, approximately 2% of GBM cases had mutations in at least 1 of the core clock genes. We then sequenced the DNA of our 5 GBM cell lines and found that only 1 line (B18) carried 1 of the known clock gene mutations (PER1S784F). This line, however, possessed strong circadian rhythms. We conclude that human GBM cells are intrinsically circadian and mutations that affect their circadian timing are rare.
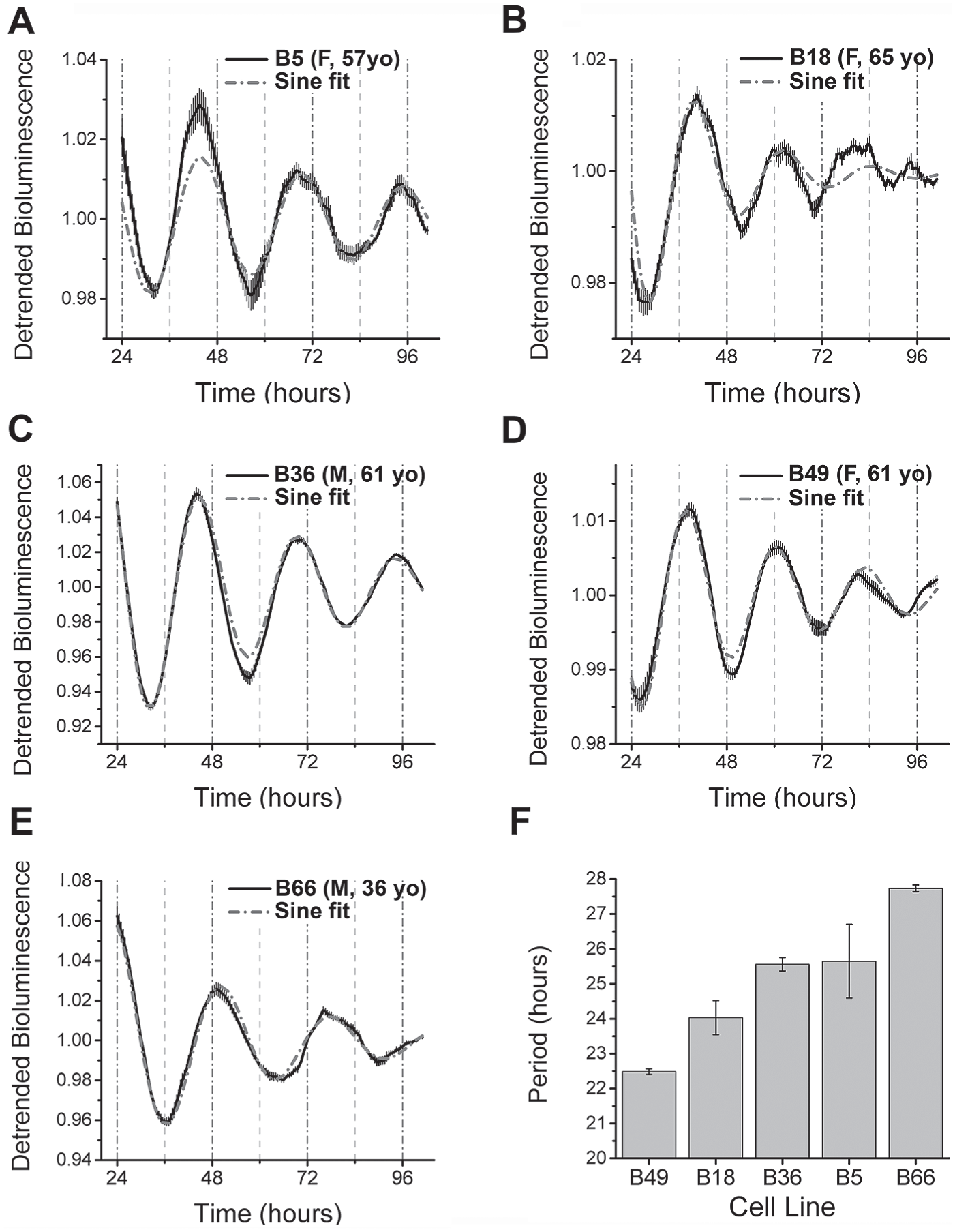
Human GBM cells are circadian. (A-E) Representative bioluminescence traces of Bmal1-luc expression in the 5 human GBM cell lines (B05, B18, B36, B49, and B66). Each trace shows the mean (solid line) and SEM (gray error bars) of 4 replicate cultures fitted by a sine function (dashed line). Each recording was made in cells at less than 30 passages from the time of initial isolation. The sex and age of each patient from which the cell lines were derived are presented in parentheses. Note that all cultures expressed intrinsic daily rhythms in Bmal1-luc. (F) Circadian period of the 5 GBM lines (mean ± SD, n = 4 independent platings).
Murine mes-GBM Astrocytes Exhibit Circadian Rhythms in TMZ Response
To further test the role of circadian timing in GBM biology, we generated an in vitro model of GBM. Glioblastoma has been successfully modeled in mice by deletion of the neurofibromin gene and abrogation of p53 function (Zhu et al., 2005). This model, which most closely resembles the mesenchymal subtype of GBM (mes-GBM) (Verhaak et al., 2010), has yielded new insights into tumor biology and been a productive model for preclinical evaluations of novel therapeutics (Reilly et al., 2000; Sun et al., 2014). Mouse cortical astrocytes rendered null for Neurofibromin (Nf1) and P53 function (mes-GBM) were transduced with a real-time luciferase reporter for either Bmal1 (Bmal1-luc) or Period2 (Per2-Luc) (Liu et al., 2008; Ramanathan et al., 2012; Zhang et al., 2009). Cultured mes-GBM astrocytes expressed circadian, anti-phase rhythms for at least 4 days in Per2-luc and Bmal1-luc with periods of 23.6 ± 3.2 h and 23.1 ± 2.6 h, respectively (mean ± SD; Fig. 2A), consistent with their expression patterns in other cell types (Reppert and Weaver, 2002). Thus, endogenous circadian rhythms are conserved in human and mouse GBM cells.
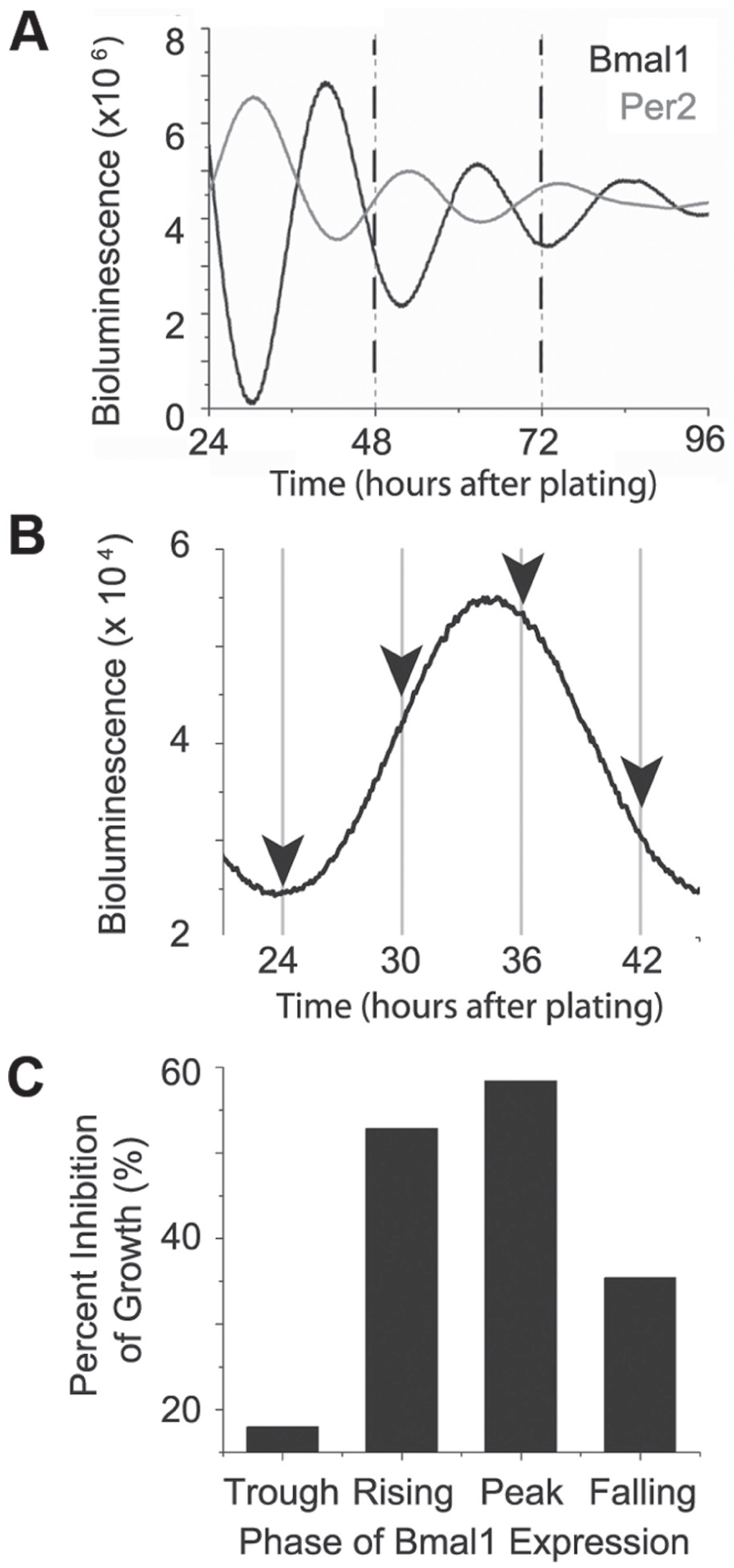
Mes-GBM astrocytes have rhythmic sensitivity to temozolomide in vitro. (A) Mes-GBM astrocytes express Bmal1-luc (black) and Per2-luc (gray) in anti-phase, with rhythmic periods of 25.3 h and 22.2 h, respectively, in these representative traces. (B) A representative trace shows how mes-GBM astrocytes were treated with TMZ or DMSO at 1 of 4 times (arrows) in their daily Bmal1 expression. (C) TMZ-induced growth inhibition varied with time of treatment (1 representative experiment shown), peaking near the peak of Bmal1-luc expression in 3 independent biological replicates.
To test whether the presence of circadian rhythms in gene expression affects GBM response to chemotherapy, we treated mes-GBM astrocytes with temozolomide (TMZ), the first-line chemotherapeutic for GBM, based on circadian time. Cells received either 1 mM TMZ or vehicle (DMSO) for 6 h at 1 of 4 phases of Bmal1-luc expression during the day after plating (Fig. 2B). We counted viable cells by their ability to exclude trypan blue 72 h after TMZ or DMSO addition. We defined growth inhibition as the number of TMZ-treated living cells divided by the number of DMSO-treated living cells at each treatment time (i.e., the death of all TMZ-treated cells would equal 100% growth inhibition). The greatest TMZ-induced growth inhibition occurred near the peak of Bmal1-luc expression (Fig. 2C). Thus, TMZ sensitivity of mes-GBM astrocytes varied with the phase of Bmal1 expression.
To assess whether the rhythm in growth inhibition was due to rhythms in response to TMZ-induced DNA damage, we treated mes-GBM astrocytes with 1 mM TMZ or DMSO at 1 of 4 phases of Bmal1-luc expression beginning 48 h after plating. To measure the extent of DNA damage, we stained for phosphorylation of histone H2AX (γH2AX), an early step in DNA damage response (DDR) and a commonly used marker for DNA double-strand breaks (Bonner et al., 2008; Rogakou et al., 1999) (Fig. 3A). We exposed cells to TMZ or vehicle for 6 h, changed the media, and fixed the cells 12 h later. We quantified nuclear staining for γH2AX (p-Ser139; example staining in Suppl. Fig. S2) and calculated the fraction of γH2AX-positive cells divided by the total DAPI-stained nuclei per field of view (Fig. 3B). TMZ-induced γH2AX (γH2AXTMZ – γH2AXDMSO) was maximal near the peak of Bmal1-luc expression (Fig. 3C). There was a mean 2.8-fold difference in TMZ response at the peak versus trough of Bmal1-luc expression across 3 independent experiments. These results demonstrate a time-of-day-dependent rhythm in the response of mes-GBM astrocytes to TMZ-induced DNA damage.
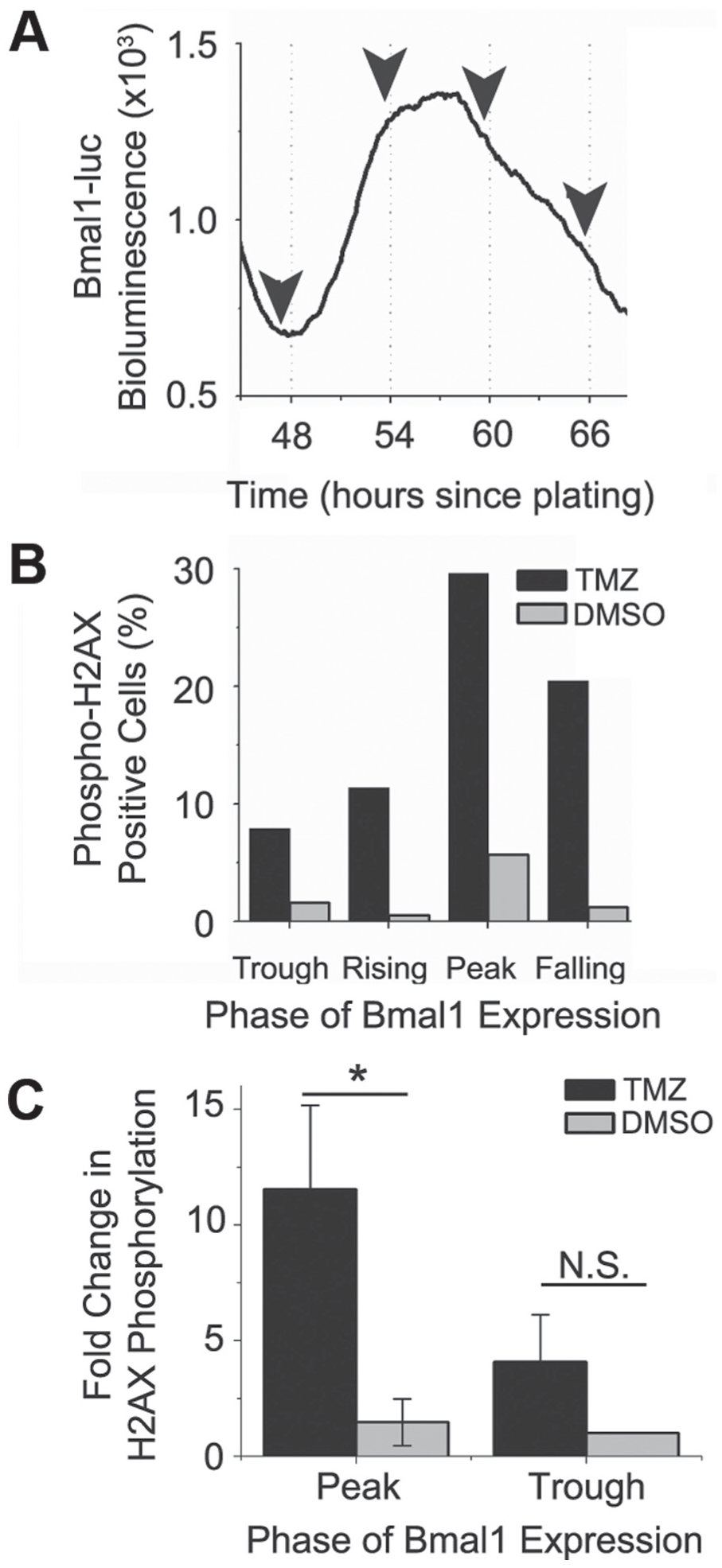
Phosphorylation of histone H2AX varies with time of treatment in vitro. (A) Bmal1-luc reporter mes-GBM cells showed oscillation of bioluminescence over time (1 representative experiment, n = 3). Arrows indicate times of TMZ or DMSO treatment for different mes-GBM cultures starting 48 h after plating. (B) Percentage of phospho-H2AX (γH2AX) positive cells varied with time of 1 mM TMZ treatment (1 representative experiment, n =3). (C) Relative to γH2AX staining of mes-GBM astrocytes treated at the trough of Bmal1 expression, TMZ induced a response at the peak, but not the trough, of Bmal1 expression (2-way ANOVA, Tukey’s multiple comparisons, *p < 0.05, n = 3).
Daily rhythms in TMZ-induced γH2AX and growth inhibition led us to test whether there was a time-of-day-dependent rhythm in TMZ-induced apoptosis. Mes-GBM astrocytes stably expressing a luciferase reporter of caspase 3 and 7 activities (Casp-luc) (Galban et al., 2013) were treated with TMZ or DMSO for 6 h at 1 of 4 phases of Per2-luc expression beginning 48 h after plating (Fig. 4A). TMZ-induced activation of Casp-luc bioluminescence above the levels evoked by DMSO was calculated from measurements taken 48 h after treatment. We expressed Casp-luc activation as fold change relative to the response at the Per2-luc falling phase (Fig. 4B). There was a significant increase in Casp-luc bioluminescence when mes-GBM astrocytes were treated at the trough of Per2-luc, which in parallel dishes corresponded to the peak of Bmal1. Thus, TMZ-induced activation of apoptosis in the mes-GBM astrocytes had a circadian rhythm.
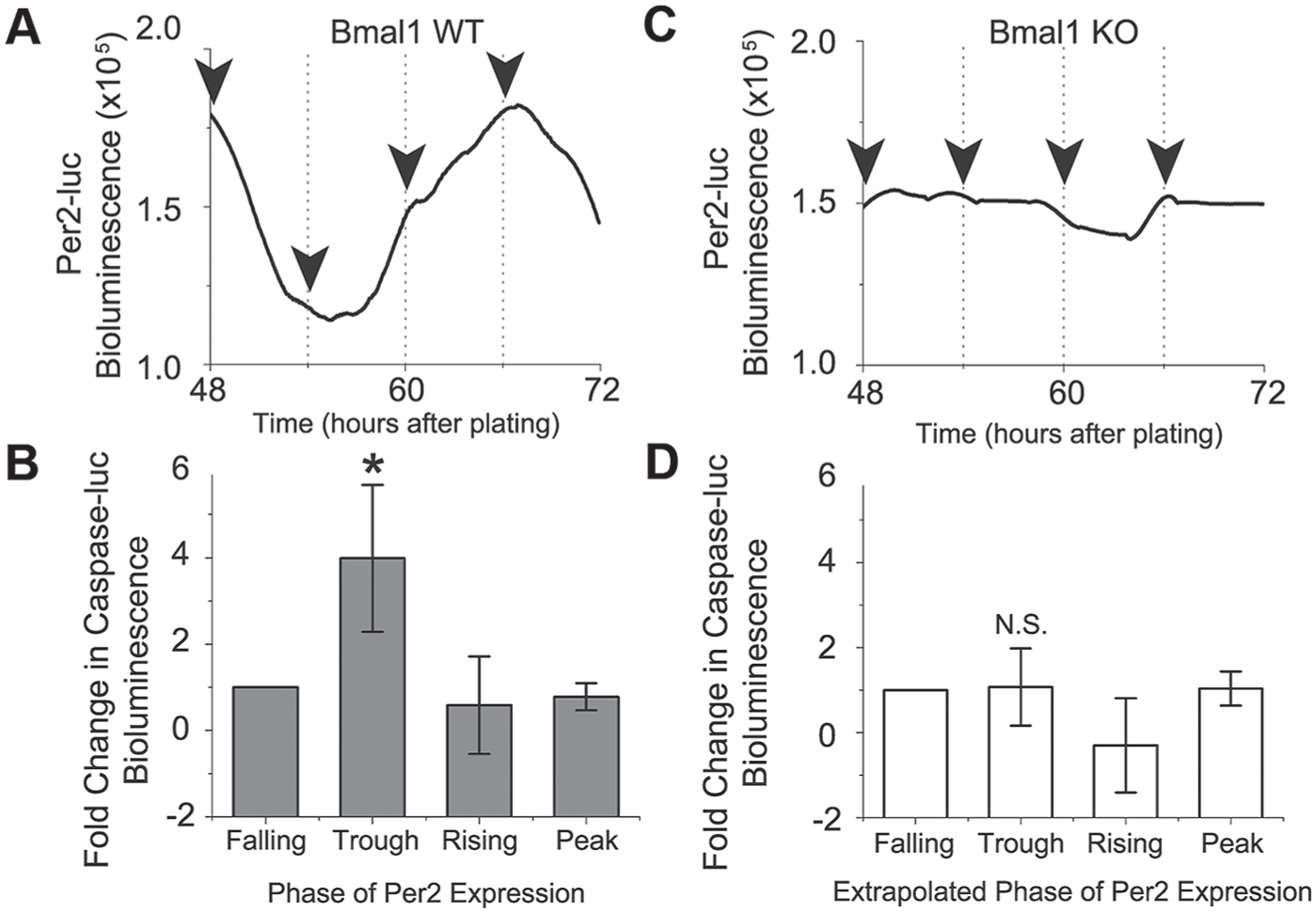
Rhythmic Per2-luc expression and activation of apoptosis depend on Bmal1 in vitro. (A) A representative culture of mes-GBM astrocytes showing circadian Per2-luc bioluminescence starting with the first treatment 48 h after plating. (B) TMZ-induced activation of a bioluminescent caspase reporter was highest when delivered at the trough of Per2-luc (i.e., peak of Bmal1-luc) (Kruskal-Wallis test, Dunn’s multiple comparisons test, p < 0.05). (C) CRISPR-mediated loss of Bmal1 resulted in arrhythmic Per2-luc expression in mes-GBM cells. (D) Caspase activation did not depend on the time of TMZ application in Bmal1 KO mes-GBM astrocytes (1-way ANOVA, Dunn’s multiple comparisons test, p > 0.05).
Circadian Rhythms in TMZ-induced Apoptosis Are Bmal1-dependent
The correlation between high Bmal1 expression and high TMZ sensitivity led us to hypothesize that increased TMZ sensitivity is Bmal1-dependent. We used CRISPR-Cas9 genome editing to disrupt the Bmal1 gene (Bmal1 KO) and abrogate BMAL1 protein expression (Suppl. Fig. S3). Loss of Bmal1 abolished circadian rhythms in Per2-luc expression (Fig. 4C). These data demonstrate disruption of the molecular clock in Bmal1 KO mes-GBM astrocytes.
To determine the necessity of Bmal1 expression to generate rhythms in TMZ-induced apoptosis, Bmal1 KO mes-GBM astrocytes stably expressing Casp-luc were treated with TMZ at 1 of 4 times 48 to 72 h after plating according to the Per2-luc rhythm of a WT culture (Fig. 4A). Loss of Bmal1 expression abolished the rhythm in TMZ-induced caspase activity (Fig. 4D), with similar activation of caspase-luc across all time points in Bmal1 KO cultures. Loss of Bmal1 also reduced the TMZ sensitivity of cultures, with the ratio of TMZ to vehicle Casp-luc activity ranging from 15.3% to 42.3% of that observed in Bmal1 WT across 3 independent experiments.
Discussion
Our study uniquely demonstrates endogenous circadian rhythms in human and murine GBM cells and their response to TMZ chemotherapy. We found cell-intrinsic daily rhythms in DNA repair, apoptosis, and growth inhibition, with maximum TMZ sensitivity of all 3 measures occurring near the peak of Bmal1 expression. Furthermore, our studies indicate that tailoring TMZ administration to the peak of Bmal1 (trough of Per2) expression in tumor cells can enhance TMZ efficacy. Taken together, these data support a rhythm in tumor response to TMZ at the level of DNA repair that leads to a rhythm in programmed cell death.
These experiments build upon research that has previously demonstrated regulation of the DDR by the circadian clock. Through direct protein-protein interactions, PER1 and PER3 enhance activation of cell cycle arrest in response to DNA double-strand breaks (Gery et al., 2006; Im et al., 2010). Our study is the first to test circadian regulation of the DDR in glioma cells. It will be important to determine whether other cell types exhibit circadian rhythms in their repair of DNA damage.
The correlation between the peak of Bmal1 expression and the greatest sensitivity to TMZ combined with the loss of circadian rhythm in TMZ-induced apoptosis in Bmal1 KO mes-GBM astrocytes suggests an important role for BMAL1 in regulating response to DNA damage. These findings are consistent with the daily variations observed in colon cancer cell sensitivity to irinotecan, achieving peak sensitivity at the peak of Bmal1 transcription (Dulong et al., 2015). Overexpression of Bmal1 increased sensitivity of colon cancer cells to oxaliplatin (Zeng et al., 2014). Bmal1 knockdown reduced etoposide-induced apoptosis of colon cancer cells (Zeng et al., 2010). These data are consistent with our findings that loss of Bmal1 leads to ablation of the rhythm in caspase activation and a reduction in maximal TMZ efficacy. There are no data showing a direct interaction of BMAL1 with proteins involved in apoptosis, but there are reports of Per2 overexpression altering expression of apoptotic genes (Hua et al., 2006). Thus, BMAL1 may act as an indirect regulator of apoptosis through its role as a transcriptional activator. Irinotecan is also reported to induce greater cytotoxicty at the peak of Bmal1, suggesting circadian regulation of a common DNA repair pathway in response to both of these chemotherapies.
Our results indicate that the circadian time of TMZ treatment affects the magnitude of the DDR and, ultimately, the fraction of tumor cells that survive. Because DNA damage itself (e.g., following doxorubicin treatment) can shift the circadian clock in HEK293T cells (Papp et al., 2015), we considered whether the changes in caspase activation and cell viability might actually reflect a resetting of GBM daily rhythms. In this scenario, TMZ might shift a circadian rhythm in phospho-H2AX, caspase activity, and cell viability. This seems unlikely given that we found no intrinsic rhythms in the regulation of the DDR or in apoptosis in control cultures (which were also treated for 6 h, but with DMSO rather than TMZ). Furthermore, the rhythm in the TMZ responses was unchanged in preliminary experiments where the time since plating was held constant, but the phase of Bmal1 expression was shifted by almost 12 h by treating GBM cultures with antiphase temperature cycles (data not shown). Future experiments should examine this further and establish whether the daily rhythms described here could also result from a daily rhythm in TMZ-induced DNA damage rather than in the DDR.
Future studies should test whether Bmal1 is playing a direct or indirect role in regulating the DDR (Jiang et al., 2016). Identifying rhythmic transcripts of DDR-related genes that lose rhythmicity in Bmal1 KO cells would provide a list of candidate regulators. In contrast, identifying DDR-related proteins in complex with Bmal1 would suggest a more direct role. At present, the Bmal1-luc and Per2-luc reporters serve as useful tools to help us understand circadian clock regulation within tumor cells and to guide the search for the mechanism of circadian regulation of the tumor cell-intrinsic response to TMZ.
Footnotes
Acknowledgements
This work was supported by the Children’s Discovery Institute (J.B.R. and E.D.H.) and NIGMS 9687304 (E.D.H.).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
