Abstract
Among nonphotic stimulants, a classic cholinergic agonist, carbachol, is known to have a strong and unique phase-resetting effect on the circadian clock: Intracerebroventricular carbachol treatment causes phase delays during the subjective early night and phase advances in the subjective late night, but the effects of this drug on the suprachiasmatic nucleus (SCN) in vivo and in vitro are still controversial. In the present study, we succeeded in reproducing the biphasic phase-shifting effect of carbachol on clock gene expression in organotypic SCN slices prepared from mice carrying a Per1-promoter fused luciferase gene (Per1-luc). Since this biphasic effect of carbachol in Per1-luc SCN was prevented by atropine but not by mecamylamine, we concluded that these phase shifts were muscarinic receptor–dependent. Next, we analyzed the expression of muscarinic receptors in the SCN by in situ hybridization and found that M3 and M4 subtypes were expressed in SCN cells. These signals appeared neonatally and reached adult levels at postnatal day 10. Together, these findings suggest that carbachol has a phase-dependent phase-shifting effect on the SCN clock through muscarinic receptor subtypes expressed in the SCN.
Many aspects of mammalian physiology and behavior, including sleep-wake cycles, core body temperature rhythms, and hormone secretion, are dominated by 24-h rhythms. In mammals, internal time is orchestrated by the master clock in the hypothalamic suprachiasmatic nucleus (SCN), whose coherent output signals synchronize cell clocks throughout the body (Yamaguchi et al., 2003; Mohawk and Takahashi, 2011; Aton and Herzog, 2005; Butler and Silver, 2009; Maywood et al., 2011; Cao et al., 2013). Not only required for rhythm generation, the SCN is also necessary for the entrainment of the circadian clock to environmental cycles. The major environmental “time cue” is the 24-h light-dark cycle generated by the earth’s rotation, information that is conducted to the SCN from the melanopsin-expressing cells in the retina via the glutamatergic retinohypothalamic tract (RHT) (Moore and Lenn, 1972; Hattar et al., 2002). Nonphotic time cues such as meals and temperature are also able to entrain the clock (Mistlberger and Skene, 2005), although they are comparatively weak synchronizers because the circadian rhythms of many totally blind individuals “free run” even when they maintain a 24-h activity-rest schedule (Lockley et al., 2007; Flynn-Evans et al., 2014). However, for clinical reasons these patients need to maintain normal social activity, and thus the development of effective novel drugs that entrain the clock would be beneficial.
Among reported agents, a classic cholinergic agonist, carbachol, has a strong resetting effect on the circadian rhythms. Carbachol administered into the lateral ventricle induces phase-dependent phase shifts of wheel-running activity rhythms in mice and hamsters (Zatz and Herkenham, 1981; Earnest and Turek, 1985; Meijer et al., 1988), possibly through muscarinic receptors (Liu and Gillette, 1996; Bina and Rusak, 1996). Interestingly, the phase response curve (PRC) for carbachol injection resembles the PRC for 1-h light pulses in hamsters maintained in constant darkness (DD) (Earnest and Turek, 1985), which indicates that the SCN is a target of carbachol action. Indeed, behavioral studies showed that carbachol microinjected into the hamster SCN similarly induced the phase-dependent phase shifts (Wee et al., 1992; Bina and Rusak, 1996). However, data are not consistent because Gillette and her colleagues reported that carbachol induces only phase advances and not phase delays in isolated rat SCN slice in vitro (Liu and Gillette, 1996) and by in vivo intra-SCN injection in mice (Buchanan and Gillette, 2005). Moreover, carbachol application is not accompanied by c-fos induction in hamster SCN, in contrast to being massively induced after light exposure (Colwell et al., 1993), suggesting mechanistic differences between these two stimuli. The action of the carbachol on the SCN remains somewhat an enigma.
It is known that the circadian expression profile of Per1 gene in the SCN mirrors behavioral rhythms of the circadian clock (Tei et al., 1997; Shigeyoshi et al., 1997). In previous studies, we succeeded in mimicking light-induced phase-dependent phase shifts of behavioral rhythms in vitro using organotypic Per1-luc SCN slice cultures by application of the glutamatergic agonists NMDA (Asai et al., 2001) or AMPA (Mizoro et al., 2010). Here we extended this real-time technique to examine the effect of carbachol, and we observed biphasic phase-dependent phase delays and phase advances. Moreover, these biphasic phase shifts were inhibited by simultaneous application of atropine, a muscarinic receptor antagonist, but not by mecamylamine, a nicotinic receptor antagonist, suggesting a dominant role for muscarinic receptors. We further characterized the expression of muscarinic receptors in the SCN and found that M3 and M4 subtypes are expressed in SCN neurons. From these findings, we speculated that muscarinic receptors inside the SCN play a dominant role for the phase-dependent phase shift.
Materials and Methods
Animals
We used Per1-promoter-luciferase (Per1-luc) transgenic mice that carried a firefly luciferase reporter gene linked to a 7.2-kbp DNA fragment covering most of the 5′ upstream region of the Per1 gene (Yamaguchi et al., 2000). These mice were housed and bred under light-dark cycles (light for 12 h, dark for 12 h), and their neonatal pups were used for the slice culture study. For the in situ hybridization study of muscarinic receptors, we used 8-week-old C57BL6 males (JAPS, Osaka, Japan) housed for at least 1 week in an environment with light-dark cycles. To examine the developmental expression of these receptors in the brain, we also used pups at postnatal days 5 and 10. All animal procedures described in this study were approved by the Animal Research Committee of Kyoto University (2015-43).
SCN Slice Culture
Coronal bilateral brain slices (400 µm thick) were prepared from 4- to 7-day-old neonates (Asai et al., 2001), and the SCN explants (approximately 0.7 mm long and 0.7 mm wide) were maintained on a membrane (Millicell-CM, Millipore, Bedford, MA) in 24-well cell culture plate, with 240 µL of culture medium (50% minimum essential medium, 25% Hank’s balanced salt solution, 25% horse serum, 36 mM glucose, and penicillin/streptomycin) at 35 °C. The SCN slices were cultured at least for 1 week before bioluminescence recordings, which were started by changing to medium containing 1 mM luciferin. The bioluminescence from the SCN cultures was measured with a highly sensitive CCD camera (850S42-40B; Spectral Instruments, Tucson, AZ) equipped with a specially designed macro-imaging lens system (Fujii-Optical Co., Yachio, Japan). Integrated bioluminescence for every 20 min was analyzed by Image J. With this system, up to 16 SCN slices can be simultaneously imaged and measured on the same field of view.
Drug Administration to SCN Slice Culture
Carbachol was applied to Per1-luc SCN slices between the second peak and third peak of their bioluminescence rhythms. Namely, 2.4 µL of carbachol stock in ddH2O (final concentration, 100 µM) or vehicle was added directly to the culture medium (240 µL) at various phases of Per1-luc rhythms. To determine whether the phase-shifting effects of carbachol are mediated through nicotinic or muscarinic receptors, carbachol was also administered in combination with either atropine (10 µM final) or mecamylamine (100 µM final). To minimize the technical artifacts of bioluminescent rhythms due to changes in temperature and mechanical stress, we performed bath application of the drug to the medium. In another series of experiments, we also applied carbachol transiently: The Millicell bearing the SCN slice was transferred to new culture medium containing carbachol (100 µM) or the same volume of solvent water, was kept in this medium for 60 min, and was then washed 3 times for 30 min by changing culture medium. After the washing steps, the SCN slices were returned to the original culture medium (before carbachol). To decrease the autoluminescence of plastic culture plates when exposed to fluorescent lights, we performed this application under dim light (<1 lux). Reagents were obtained from Nacalai tesque (Kyoto, Japan) unless otherwise stated.
To calculate phase shifts, we determined the timing of the first, second, third, and fourth peaks of each slice. We measured the interval between the first and second peaks and defined it as the pretreatment period length. We then compared this first interval with the interval between the third and fourth peaks (posttreatment) and confirmed that the durations of these 2 intervals were not significantly different. Last, we measured the interval between the second and third peaks (when treatment occurred) and calculated the difference between the first and second intervals, which was defined as the phase shift.
In Situ Hybridization
To visualize muscarinic receptor subtypes, we generated transcript-specific probes as follows: mAchR1 antisense probe covering nucleotides 1961-2520 (Genbank, NM_001112697.1) of mAChR1 (M1); nucleotides 1101-1620 (NM_203491.3) for mAChR2 (M2); nucleotides 2561-3100 (NM_033269.4) for mAChR3 (M3), nucleotides 851-1360 (NM_007699.2) for mAChR4 (M4), and nucleotides 763-1301 (NM_205783.2) for mAChR5 (M5).
The corresponding cDNA fragments were cloned and used as templates for riboprobe synthesis. The riboprobes were radiolabeled with [33P]UTP (PerkinElmer, Waltham, MA) using a standard protocol for cRNA synthesis. Two days before sacrifice, mice that had been housed for at least 1 week in an environment with a LD cycle were transferred to constant darkness. Brain sampling was performed at CT4 (4 h after the subjective dawn) and CT16 (4 h after the subjective dusk). In situ hybridization was performed according to the method detailed previously (Shigeyoshi et al., 1997). To characterize developmental changes in cholinergic receptor expression in the SCN, we also performed in situ hybridization at postnatal days 5 and day 10 (n = 6 at each time point). To minimize technical variations throughout the hybridization procedure, all sections from different experimental conditions were gathered into 1 group and processed simultaneously (floating sections). For the detection of isotope signals, we exposed X-ray film (BioMax film, Kodak, Rochester, NY) to the air-dried sections.
To visualize muscarinic subtypes at the cellular level, we further performed histochemistry by using digoxigenin-labeled antisense and sense cRNA probes (Roche Diagnostics GmbH, Mannheim, Germany) following standard protocol (Mizoro et al., 2010). The sections were processed for immunochemistry using a nucleic acid detection kit (Roche Diagnostics GmbH). Signals were visualized in a solution containing nitroblue tetrazolium salt (0.34 mg/mL, Roche Diagnostics GmbH) and 5-bromo-4-chloro-3-indolyl phosphate toluidium salt (0.18 mg/mL, Roche Diagnostics GmbH). Stained sections were observed under light microscopy (Zeiss, Oberkochen, Germany).
Results
The use of a cryogenic CCD camera coupled to a macroimaging lens allowed us to simultaneously measure luminescence of up to 16 SCN slices in a 24-well plate on the same field of view (Fig. 1, A and B), which is a dramatic improvement over our previous system, which was limited to 1 SCN slice in a 35-mm dish at a time (Asai et al., 2001).
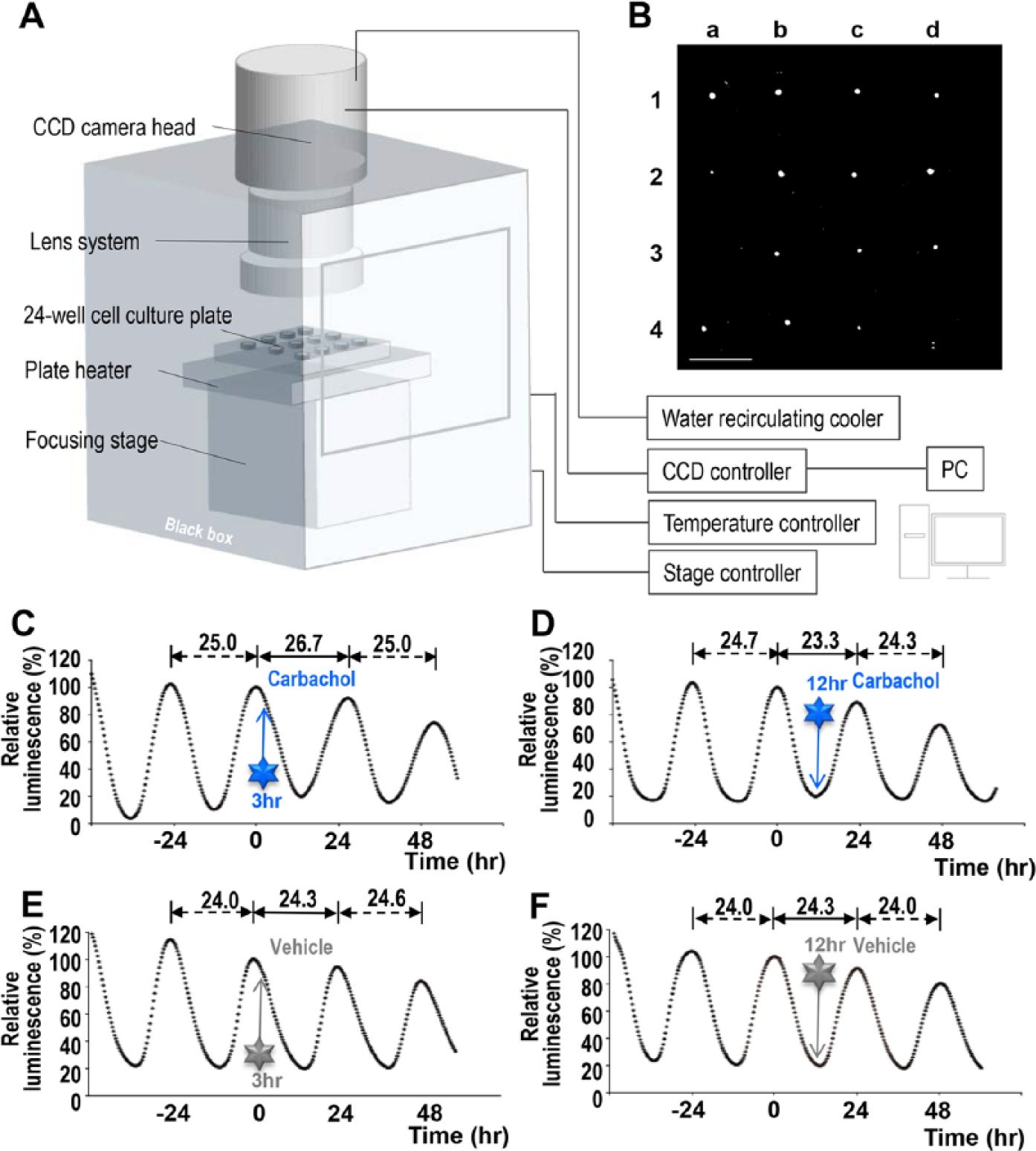
(A) Schematic illustration of experimental setup. (B) Images of a representative result of Per1-luc luminescence in SCN slice culture in 24-well plates. We measured 14 slices out of 16, discarding a-3 (dead slice) and d-4 (a slice attached to the wall of Millicell). Bar = 1 cm. (C) Exposure to carbachol at 3 h after the second peak of the luminescence induced a phase delay. (D) Carbachol applied at 12 h after the second peak induced a phase advance. Vehicle (ddH2O) application at (E) 3 h and (F) 12 h after the peak did not change the phase. (C-F) Graphs of representative results are shown; peak values at time 0 (second peak) equaled to 100%. The times of injection are indicated by arrows.
Per1-luc luminescence of the SCN slices showed a clear circadian oscillation of bioluminescence, as previously reported (Asai et al., 2001; Mizoro et al., 2010). When carbachol was applied 3 h after the second peak, the Per1-luc luminescence was phase delayed, while application 12 h after the second peak caused a phase advance (Fig. 1, C and D). Phase shifts were not observed in vehicle-administered slices (Fig. 1, E and F). To further examine the effect of carbachol on phase shifts, we built a PRC with 107 SCN slices (Fig. 2). The directions and magnitude of carbachol-induced phase shifts were dependent on the circadian phase at the time of treatment. Carbachol applications at 2 to 4 h after the peak caused phase delays, whereas the applications at 8 to 12 h caused phase advances. From 14 to 24 h, no prominent effects were observed. However, these data were obtained by bath-application of carbachol to the culture medium, so there was a possibility that carbachol had a long-term effect on the SCN slices. To eliminate this possibility, we transiently applied carbachol for 1 h and observed similar phase-dependent phase shifts: phase delay at 2 to 4 h after the peak, and phase advance at 8 to 12 h after the peak (Suppl. Fig. S1).
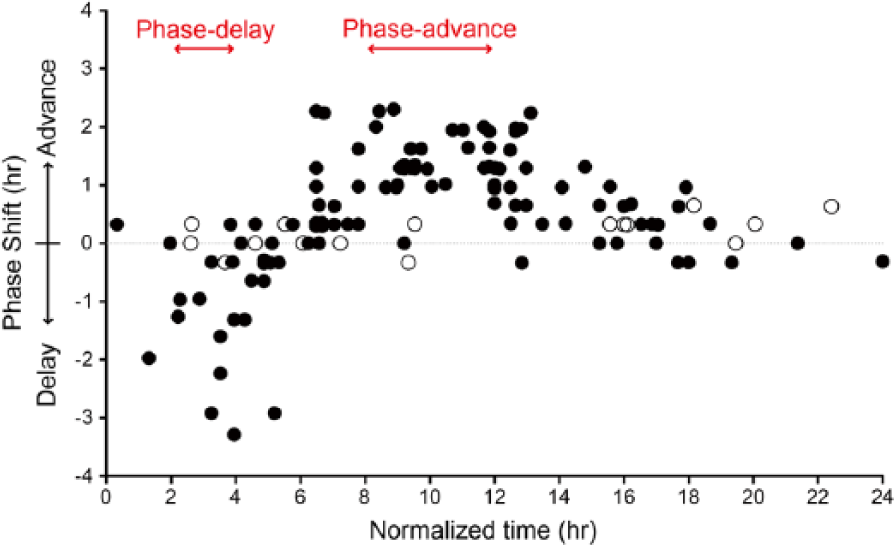
Phase response curve (PRC) obtained with SCN slice cultures stimulated by carbachol. Carbachol (closed circle; n = 107) and vehicle (open circle; n = 16) were applied at various times in 24 h, and resultant phase shifts were measured. The x axis represents normalized time after the second peak (1 normalized hour = free running period/24 h). Normalized time 0 was defined as the time of the second peak of Per1-luc luminescence. The y axis represents the magnitude of phase shifts normalized by multiplying each shift in hour by the factor of 24 h per free-running period. Plus and minus values mean phase advances and delays, respectively.
Since carbachol is a nonselective cholinergic agonist that acts on both muscarinic and nicotinic receptors, we next examined which receptor type is involved in carbachol-induced phase shift of Per1-luc. We applied carbachol simultaneously with atropine, a muscarinic antagonist (Rang et al., 2003), or with mecamylamine, a nicotinic antagonist (Martin et al., 1989). The carbachol-induced phase advance was completely suppressed by atropine but not by mecamylamine (Fig. 3, A and C). These findings suggest that the effects of carbachol are mediated by muscarinic receptors.
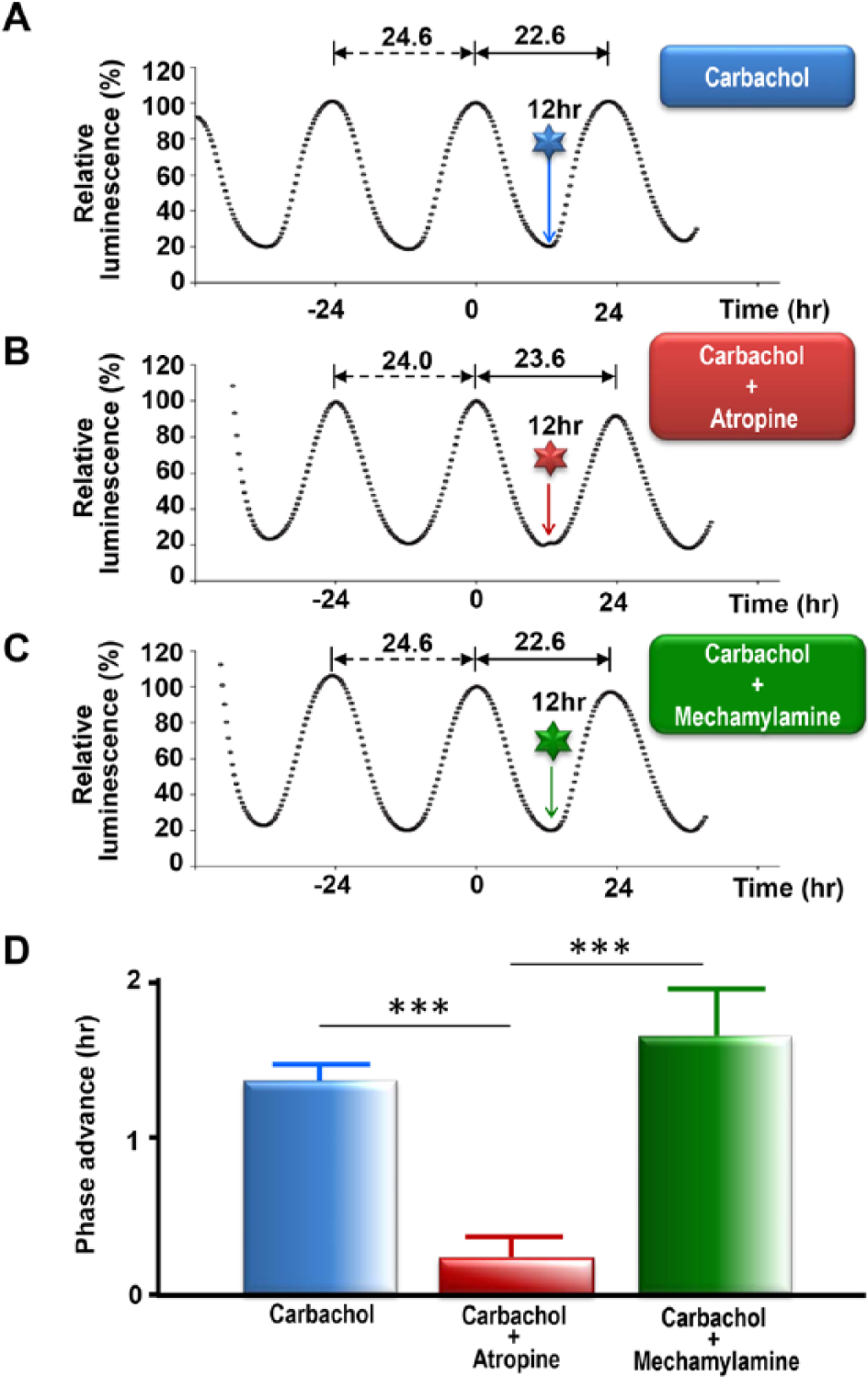
The effect of cholinergic receptor antagonists on carbachol-induced phase shift of Per1-luc SCN slice cultures at advance phase. Representative data of circadian rhythms of Per1-luc bioluminescence injected at 12 h with (A) carbachol, (B) carbachol with atropine, and (C) carbachol with mecamylamine. The times of injection are indicated by arrows. (D) Mass analysis of carbachol-induced phase difference at advance phase (8-12 h). Single treatment of carbachol (n = 30), carbachol and mecamylamine (n = 7), and carbachol and atropine (n = 8). Significant differences were observed between each group, analyzed by Tukey-Kramer multiple comparison method (***p < 0.001).
Pharmacologically identified muscarinic receptors are subdivided into 5 subtypes (Caulfield and Birdsall, 1998). To address which subtype of muscarinic receptors are involved in carbachol-induced phase shifts, we examined what subtypes of muscarinic receptors are expressed in the mouse SCN. We performed in situ hybridization and found that M3 and M4 receptors, but not M1, M2, and M5, were detectably expressed in the SCN (Fig. 4A). No apparent differences in expression between subjective day (CT4) and subjective night (CT16) were observed. We further analyzed M3 and M4 signals at the cellular level by digoxigenin-labeled probes and found moderate levels of M3 mRNA signals in neurons of the central part of the SCN and weak levels of M4 mRNA in the whole SCN (Fig 4B). Last, we examined the development of M3 and M4 receptors in the SCN and found that these receptors were not detected at postnatal day 5 but had increased to adult levels by day 10 (Suppl. Fig. S2). These findings suggest that carbachol-induced phase shifts are mediated by M3 and M4 receptor subtypes.
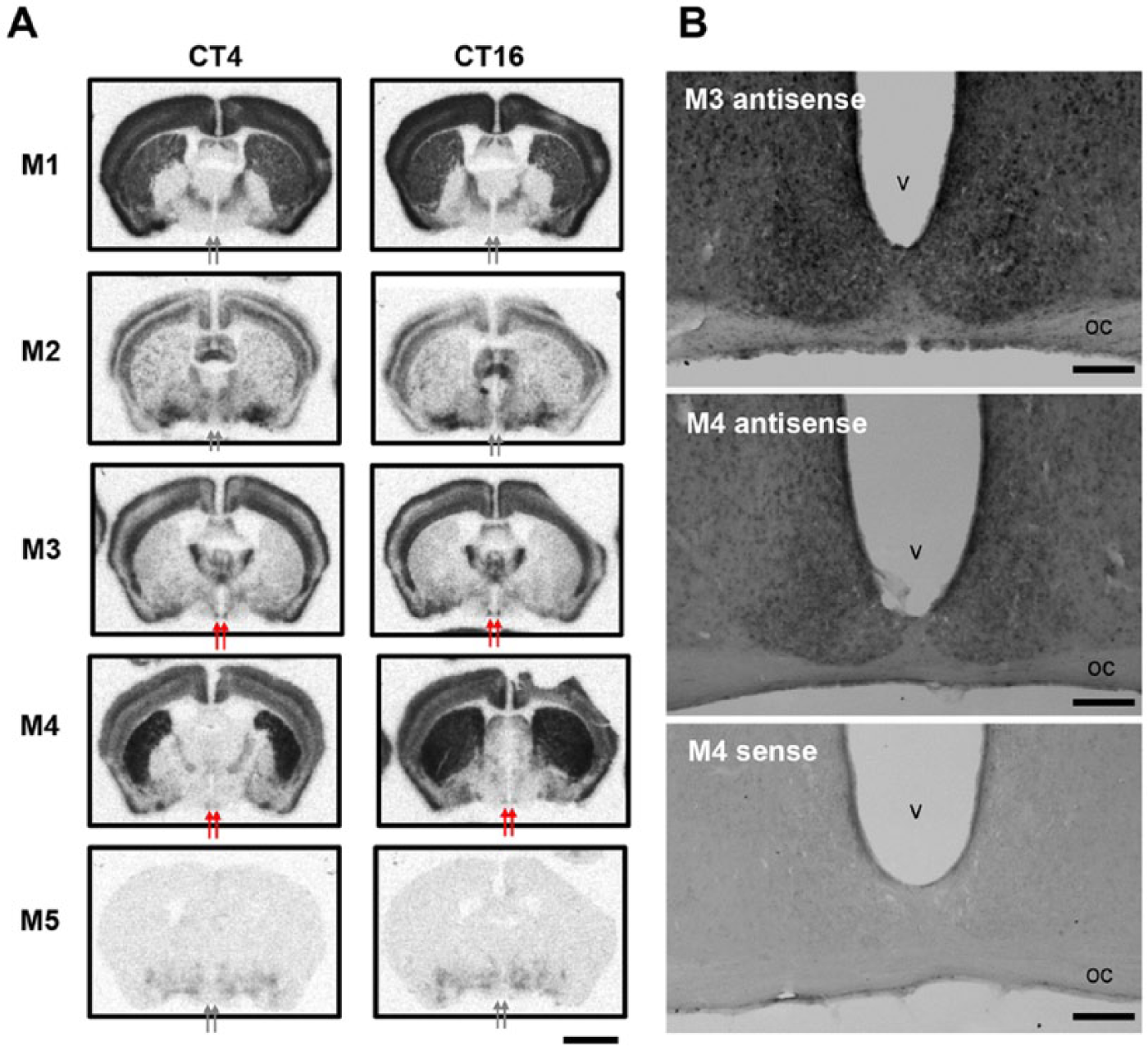
(A) Topographic analysis of muscarinic receptor mRNA (M1-5) in the mouse SCN by in situ hybridization using isotope-labeled riboprobes at CT4 and CT16. Arrows indicate SCN. (B) In situ hybridization histochemistry by using digoxigenin-labeled M3-antisense (upper), M4-antisense (middle), and M4-sense (lower) probes. oc, optic chiasma; v, ventricle. Scale bars, 2 mm in A; 100 µm in B.
Discussion
In this paper, carbachol induced both phase delays and advances in SCN slices depending on the circadian phase: Phase delays occurred at 2 to 4 h and phase advances at 8 to 12 h after the peak of Per1-luc. In the case of NMDA and AMPA we previously reported, phase delays occurred at 4 to 7 h and phase advances at 9 to 13 h (Asai et al., 2001; Mizoro et al., 2010). Phase delays induced by carbachol occurred earlier than those induced by NMDA or AMPA applications, although the timing of phase advances is similar. It is known that glutamatergic and cholinergic inputs innervate the SCN from completely different anatomical regions: Glutamatergic input to the SCN comes from the RHT (Moore and Lenn, 1972), and cholinergic input to the SCN from the basal forebrain (Bina et al., 1993). Our present study suggests that brain-derived cholinergic inputs phase-delay the clock earlier than do light-induced glutamatergic retinal inputs, and our findings also indicate that both brain-derived cholinergic and light-mediated glutamatergic inputs cause phase advance at subjective late night.
The involvement of the SCN in carbachol-induced phase shifts in vivo is still controversial: Carbachol microinjected into the hamster SCN induced phase-dependent phase shifts (Wee et al., 1992; Bina and Rusak, 1996), but microinjections in mouse SCN elicited only phase delays (Buchanan and Gillette, 2005). Our present real-time recording of mouse Per1-luc in vitro supports the idea that the SCN is the site of carbachol action that can produce characteristic bidirectional phase-dependent phase shifts, but many contributing factors such as species-specific differences of circadian characteristics may explain in part these discrepancies, similar to light-pulse stimulation having a large phase advance for hamsters but a large phase delay for mice.
Among other factors to explain the discrepancy in previous reports, one critical factor is probably the dose of carbachol used. Reports of bidirectional phase shifts using carbachol administration to the lateral ventricle of hamster and mice in vivo (Zatz and Herkenham, 1981; Earnest and Turek, 1985; Wee et al., 1992) were based on relatively high doses. Intra-SCN applications using relatively high doses (0.5 µL of a 10 mM solution) induced bidirectional phase shifts (Bina and Rusak, 1996), in contrast to unidirectional shifts obtained with lower doses (0.3 µL injection of a 50 µM carbachol solution) (Buchanann and Gillette, 2005). In our case, we applied a relatively high dose of carbachol (final 100 µM) into the culture medium. Thus, it is possible that higher doses of carbachol will affect different receptor subtypes in the SCN. Indeed, our present study identified 2 types of receptors expressed (M3 and M4) in the mouse SCN. Recently, it was demonstrated that the M1/M4 agonist McN-A-343, but not the M2/M3 agonist bethanechol, produced significant phase advances when injected into the SCN during the mid-subjective day in hamsters (Basu et al., 2016). It was also reported that the affinity of carbachol for the M4 receptor is approximately 10-fold higher than for M3 receptors (Brann et al., 1988). Combining these lines of evidence with our own observations, we speculated that M4 receptors in the SCN might be involved in phase advances and M3 receptors in phase delays. However, a previous RT-PCR study reported the expression of all subtypes of muscarinic receptors in the rat SCN (Yang et al., 2010).
We also examined the developmental regulation of M3 and M4 receptors in the SCN. In situ hybridization analysis has demonstrated that M3 and M4 were not detected at postnatal day 5 but reached adult levels by day 10. These findings suggest that cholinergic receptors gradually mature postnatally in the SCN. Moreover, many cholinergic systems operate in the SCN (Hut and van der Zee, 2011), and nicotinic receptors are also expressed (O’Hara et al., 1999; Yang et al., 2010). Future studies will be needed to understand the complexity of cholinergic signaling in the SCN.
Footnotes
Acknowledgements
K.D. is supported by the Training Program of Leaders for Integrated Medical System for Fruitful Healthy-Longevity Society, Kyoto University. This research was supported by grants from Kitsuen Research Foundation, Kobayashi International Scholarship Foundation, Core Research for Evolutional Science and Technology (CREST), and scientific grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan to H.O.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
