Abstract
Many physiological and behavioral processes such as sleep and wakefulness, hormone secretion, and olfactory sensitivity exhibit a 24-h rhythmicity that persists in constant conditions with a period close to (circa) 24 h. These circadian rhythms are driven by a network of endogenous clocks residing in various tissues, including the olfactory system, and are synchronized to the outside world by environmental time cues such as light, temperature, and food. In addition to having these well-known zeitgebers of circadian clocks, most environments consist of a multitude of odors that report, for example, the availability of food or the presence of predators—and often, they do so in a time-of-day-dependent manner. Considering the evolutionary significance of odors for various fitness-related behaviors such as mate choice, predator avoidance, and foraging strategies, we asked whether odors—similar to light, temperature, or food—might act as a circadian time cue able to influence circadian locomotor behavior in mammals. Administering individual air flow, periodically saturated with an artificial odor mix, to running wheel-equipped mouse cages, we found that rhythmic odor administrations significantly lengthened the period of circadian activity rhythms. Additionally, odor cues led to partial reemergence of circadian locomotor rhythmicity in suprachiasmatic nuclei (SCN)–lesioned mice, suggesting that the SCN as the central circadian pacemaker are not immediately required for odor-mediated effects on circadian behavior. However, odor-based modulation of circadian behavior did not occur in clock mutant (cry1−/−/cry2−/−) mice, indicating an odor-mediated mechanism that involves extra-SCN canonical clocks, such as the olfactory clock itself. Our results indicate not only that odor stimuli can act as a circadian time cue modulating circadian behavior but also that odor effects are even more pronounced in the absence of the SCN but nevertheless require the presence of a functional canonical clock.
Mammalian circadian rhythms are synchronized (or “entrained”) by different environmental time cues, called zeitgebers, including light (Pittendrigh and Daan, 1976b), temperature (Brown et al., 2002), and food (Stokkan et al., 2001). Under natural conditions, light is indirectly perceived by the master circadian pacemaker in the suprachiasmatic nuclei (SCN) and appears to be the strongest of all zeitgebers. The SCN are believed to be on top of the circadian hierarchy, coordinating rhythmicity in a variety of peripheral and neural extra-SCN clocks regulating circadian physiology and behavior (Maywood et al., 2007). However, there seem to be a few exceptions to this rule: For example, perception of the light zeitgeber involves retinal photoreceptors and intrinsically photosensitive retinal ganglion cells (Freedman et al., 1999; Berson et al., 2002) that contain an autonomous circadian clock (Ruan et al., 2008). The retinal clock appears unique among extra-SCN oscillators in that it modulates rhythmic gene expression in the SCN (Lee et al., 2003) as well as circadian behavior (Yamazaki et al., 2002). Like the retinal clock, the mammalian olfactory clock is autonomous and SCN-independent, as has been demonstrated by the persistence of olfactory bulb (OB) oscillations in SCN-lesioned mice in vivo (Granados-Fuentes et al., 2004; Abraham et al., 2005). In line with that, the OBs, but not the SCN, are required for rhythmic clock gene expression in another olfactory center, the piriform cortex, and OB removal modulated circadian behavior (Granados-Fuentes et al., 2006; Pieper and Lobocki, 1991; Perret et al., 2003; Jechura et al., 2006). Given the predominance of environmental odors, their vitality for survival, and the highly conserved olfactory pathways, system morphologies, and signal coding strategies among different species (Ache and Young, 2005), we hypothesized that the circadian system evolved to use odor prevalence as a time cue, that is, odors function as zeitgebers for the circadian system (Governale and Lee, 2001). While in laboratory experiments the environment is usually strictly controlled (i.e., constant odor levels), in this study we specifically investigated odor as a potential time cue for circadian clocks and found that odor indeed modulates circadian behavior in intact mice. Although odor effects do not require the presence of the SCN, they still require canonical clock function.
Materials and Methods
Animals and Housing
Male C57Bl/6 mice (Jackson Laboratories strain) and BALB/c mice were purchased from our animal facility (FEM, Berlin, Germany) at 6 to 8 weeks of age, housed individually in Makrolon cages equipped with running wheels, and provided food and water access ad libitum. Running wheel revolutions were recorded in 1-min bins and stored by a data acquisition system (ClockLab, Actimetrics, Wilmette, IL). Cages of the same experimental group (n = 6) were placed in light-tight recording chambers with controlled ambient lighting and individual ventilation.
Heterozygous and homozygous male and female PER2::LUC mice (Yoo et al., 2004), as well as homozygous male and female cry1/cry2 double knockout mice (van der Horst et al., 1999), bred and raised in our animal facility (FEM, Berlin, Germany) were used for behavioral assays at about 4 to 10 months of age. All procedures were authorized by and performed in accordance with guidelines and regulations of the German animal protection law (Deutsches Tierschutzgesetz).
Behavioral Experiments
C57Bl/6 mice were kept in a 12 h light: 12 h dark cycle (LD 12:12) with daily 30-min odor presentations (odor, n = 4-5) or air only (air-only, n = 4-5) at zeitgeber time (ZT) 16 (4 h after lights-off) and ZT23 (1 h before lights-on) for at least 7 days, while locomotor activity was recorded continuously. On day 8, the light schedule in both groups was switched to constant darkness (DD), whereas daily odor presentations persisted for another 17 days (DD + odor/air). On day 18, odor pulses were stopped, but locomotor activity registration continued for another 7 days (DD after odor/air). Two independent experiments were performed with n = 4 to 5 per group. To exclude confounding group effects, C57Bl/6 mice (n = 3 per experimental group) were subjected to a crossover design. Mice previously exposed to odor pulses received air only in the second experiment, and vice versa. To exclude the potential additive effects of light, an additional 10 C57Bl/6 mice received odor presentations (n = 4) or air only (n = 6) at ZT16 and ZT23 (with respect to the previous light-dark cycle) starting in DD only. To exclude strain-specific effects, we additionally tested PER2::LUC (n = 24), and balb/C mice (n = 12). SCN-lesioned C57Bl/6 mice (arrhythmicity was confirmed by locomotor activity registration for at least 1 month; completeness of SCN lesion was histologically confirmed after completion of the behavioral experiments) were exposed to a 1:10:1:12 h photoperiod for 1 week to exclude residual entrainment to the light-dark cycle. Starting on day 8, three mice received daily odor presentations at ZT16 and ZT23 for 7 days, whereas four mice received air only. Ten days later, the light schedule was switched to DD with periodic odor presentations/air only persisting for 18 days (DD + odor/air). Thereafter, odor presentations/air-only stopped while behavior was recorded for another 7 to 10 days (DD after odor/air). To exclude confounding group effects, 3 of the above-mentioned SCN-lesioned mice were subjected to a crossover design (n = 1 and 2 per experimental group). Mice previously exposed to odor presentations received air only in the second experiment (n = 1), and vice versa (n = 2). The cry1/cry2 double knockout mice were treated similarly: upon synchronization to a light-dark cycle (LD 12:12), mice received 30-min odor presentations at ZT16 (4 h after lights-off) and ZT23 (1 h before lights-on, n = 6) or received air only (n = 6). After 8 days, mice were transferred into constant darkness, while odor pulses and air administration persisted for another 27 days (DD with odor/air). After that, odor presentations/air-only stopped while behavior was recorded for another 10 to 14 days (DD after odor/air). To exclude confounding group effects, all cry1/cry2 double knockout mice were subjected to a crossover design. Mice previously exposed to odor presentations received air only in the second experiment, and vice versa.
During the course of a behavioral experiment, to avoid disturbances and introduction of novel odors, cages were not changed or cleaned. Drinking water was renewed once a week.
Odor Mix
The odor mix consisted of vanillin (saturated DMSO-solution), ethyl butyrate (“pineapple”), dimeth-ylpyrazine (“nuts”), isoamyl acetate (“banana”), and phenethyl alcohol (“floral, rose”) (all Fluka), respectively. We mixed 500 µL of each odor compound in a glass flask that was hooked up to the odor/air presentation system and diluted in an additional 2 mL of DMSO (AppliChem GmbH) resulting in a final volume of 4.5 mL and a 1:1.8 dilution of the odor mix. Halfway through a behavioral experiment, the odor mix was renewed to avoid attenuation of the administered odor pulses by evaporation. Previous studies found functional specific olfactory receptors for each of the compounds in the OE (Zou and Buck, 2006).
Odor/Air Presentation System
Cages were individually connected to a TiCODS-1 mini-pump (Time-Controlled Odor Delivery System, engineered by the Medical-Technical labs, Charité-Universitätsmedizin Berlin) to administer pure air to each cage continuously. At the indicated pulse times (see below), a timer-regulated valve opened to redirect the air flow to the experimental group through a glass flask containing odor mix (see above). Hence, for the duration of the odor presentations, odorant-filled air was directed to the experimental group, while pure air was directed to the control group. Experimental and control groups were separated spatially.
SCN Lesions
SCN lesions were performed using the following stereotactic coordinates: 0.4 mm posterior to, ±0.1 mm lateral to, and 5.7 mm below bregma. A direct current of 1.44 mA was passed through the electrode for 6 sec to destroy each of the bilateral SCN, respectively. Wheel-running activity was recorded for up to 1 month following surgery, and arrhythmicity was confirmed using χ2-periodogram analysis at the 0.01% confidence level (ClockLab, Actimetrics). The extent of SCN destruction was verified postmortem by histological inspection.
Analyses of Behavioral Data
Locomotor activity periods of intact mice in DD + odor/air and DD after odor/air were determined by χ2-periodogram analysis (ClockLab, Actimetrics). The degrees of residual rhythmicity in SCN-lesioned and cry1/cry2 double knockout mice were determined using χ2-periodogram analysis (ClockLab, Actimetrics), which calculates the probability of any given period to occur in a time series and displays them as amplitude values. The amplitudes are measured as the height of the peak above the χ2-periodogram significance line and are provided in arbitrary units (usually reported as power of rhythmicity). Arrhythmic systems display a multitude of periods with low amplitudes. To score for circadian rhythm strength/residual circadian rhythmicity in arrhythmic mice, only peaks crossing the significance line at the 0.001 confidence level were accounted for. The highest period peak in the circadian range between 18 and 30 h was defined as main peak. Rhythm strength was then calculated by dividing the amplitude value of the main peak by the number of secondary peaks. The higher the resulting amplitude value, the higher the strength of the residual circadian rhythmicity. To ensure comparability between experiments, locomotor activities in equal time periods were analyzed. Data on period length and circadian rhythm amplitudes were statistically analyzed using Graph Pad Prism 4 software (Graph Pad Software Inc.). The level of significance alpha was set at 0.05; all tests performed were 1-tailed.
Results
Rhythmic Odor Pulses Lengthen Circadian Behavior in Mice
If odor constitutes a zeitgeber for the circadian system, periodic odor application should be able to either synchronize or at least modulate circadian behavior, similar to what has been observed for other nonphotic zeitgebers, like food, social interactions, or drugs (Stephan, 2002; Mistlberger and Skene, 2004; Yannielli and Harrington, 2004). Consequently, timing information derived from odor pulses would have to act on the SCN itself or on other extra-SCN oscillators that are able to influence circadian behavior. This is conceivable since the OB, the primary cerebral center processing olfactory information, possesses a multisynaptic neural connection with the SCN (Krout et al., 2002). To test the effects of odor stimuli on circadian behavior, we exposed C57Bl/6 mice to a 12 h light: 12 h dark cycle with simultaneous periodic gaseous odor presentations and, subsequently, to periodic odor presentations during constant darkness (DD) while recording their locomotor activity. Based on traditional entrainment theory (Pittendrigh, 1981) and on previous studies in which single odor pulses had no effect on behavior in rats (Amir et al., 1999), we selected 2 times of daily odor presentations, namely zeitgeber times 16 (ZT16 = 4 h after lights-off) and 23 (ZT23 = 1 h before lights-on). Light pulses administered at these time points are predicted to cause maximal advance and delay phase shifts of locomotor behavior. Consequently, since odor potentiates light effects (Governale and Lee, 2001), we reasoned that odor presentations at these 2 time points are most likely to exert maximal effects on locomotor behavior as well. Odor presentations were administered for 30 min (1) to allow for temporal accumulation of odor within the mouse cage to maximize the chance that every mouse received a sufficient amount of odor to experience at least 1 olfactory response around the selected time windows and (2) to avoid desensitization of olfactory sensory neurons by exposure to the same odors for several hours. The times between odor presentations (6.5 and 16.5 h) were long enough to ensure complete recovery of olfactory sensory neurons after adaptation (Zufall and Leinders-Zufall, 2000).
Odor-treated animals did not exhibit immediate decrease or increase of locomotor activity in response to the odor stimulus (Fig. 1, A-C) but rather free-running locomotor activity rhythms with periods that were significantly longer by about 20 min compared with control mice (Figs. 1 and 2A) (odor, 23.6 ± 0.07 h [n = 11]; air-only, 23.28 ± 0.09 h [n = 11]; unpaired t test, p < 0.01; data expressed as mean ± SEM). The significant period difference persisted even after the odor administration had stopped (Figs. 1 and 2A) (after odor, 23.6 ± 0.07 h [n = 11]; after air-only, 23.26 ± 0.1 h [n = 11]; unpaired t test, p < 0.01). To exclude random group effects, we subjected 3 mice to air only first and, in a second experiment—including initial light entrainment—to odor (Fig. 1D, individuals a-c). Another 3 mice received the reverse treatment (Fig. 1D, individuals d-f). In all 6 mice, the individual free-running periods lengthened upon odor treatment or shortened in air-only, respectively, suggesting that the observed effects were treatment rather than group-specific. Furthermore, the individual lengthening/shortening effect of the treatment still persisted in 4 of 6 animals after odor/air administration had stopped (Fig. 1E), consistent with what was observed on the group level (Fig. 2A). The 2 out of 6 animals that did not show persistent period lengthening belonged to the group of animals that received air only first and odor later (Fig. 1D, individuals a and b). Hence, we cannot exclude that the lack of persistence in these mice was due to a mild age effect, resulting in reduced olfactory sensitivity.
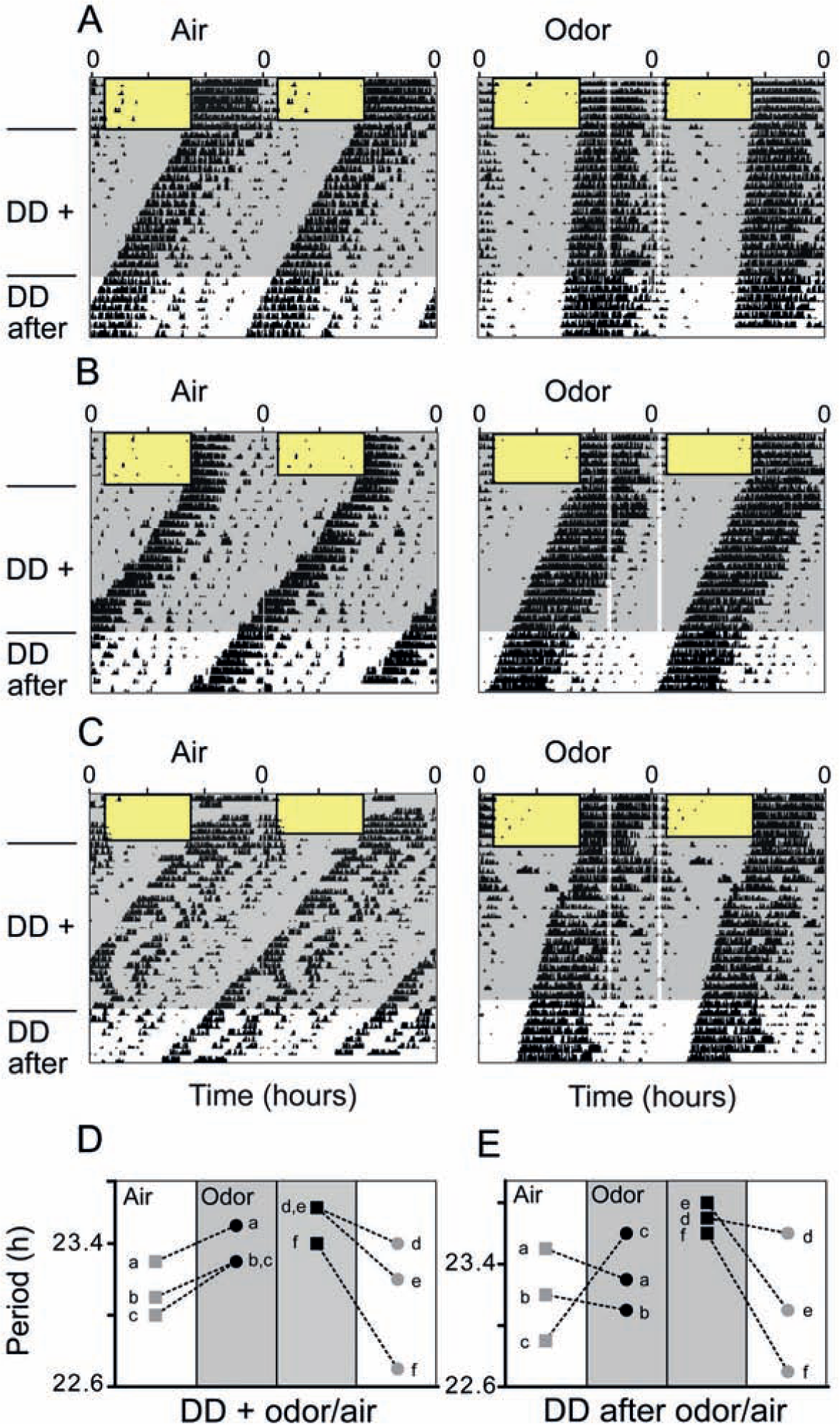
Periodic odor administration lengthens the activity period of mice. (A-C) Representa-tive double-plotted actograms showing wheel-running activity at different times of day (wheel revolutions per 6 min = black vertical bars; rectangles with black borders = light phases). Control mice (“Air”) were subjected to constant air flow (shaded gray areas, left panels), while the experimental group (“Odor”) was exposed to constant air flow (shaded gray areas, right panels) enriched by 30-min presentations of a gaseous odor mix, administered daily at zeitgeber times (ZT) 16 (4 h after lights-off) and 23 (1 h before lights-on; vertical transparent-white bars, right panels). Upon release into constant darkness with persisting air/odor conditions (DD+), the free-running activity rhythm of mice receiving odor displayed a longer period than that of mice receiving air only (A-C). This effect persisted after air/odor administration had stopped (DD after). (C-E) Results of a crossover design. (C) Double plotted actograms of the same mouse receiving air only in 1 set of experiments (left panel, “Air”) and odor presentations in a separate set of experiments (right panel, “Odor”). Consistent with the independent experiments displayed in A and B, the same individual also showed a longer free-running activity period upon odor presentations compared with when it received air only (depicted as mouse e in 3D,E). (D) Individual free-running periods of 3 mice (a, b, c, gray squares) that were measured in DD with air only and, in a second experiment, in DD with odor presentations (a, b, c, black dots). In an additional experiment, the free-running periods of 3 different mice (d, e, f, black squares) were first measured in DD with odor presentations and, in a second experiment, in DD with air only (d, e, f, gray dots). Periods of the same individuals are connected by dashed lines. (E) Period developments of the same 6 mice as in C were determined after odor/air had been switched off (DD after odor/air). The crossover-design revealed that all 6 mice displayed longer free-running periods when subjected to odor presentations and shorter periods in air only, independently of the sequence of the treatments. Hence, we can exclude a mere cohort effect. This phenomenon was retained in 4 of 6 mice after odor/air had been switched off. The actograms of mouse e are displayed in 3C.
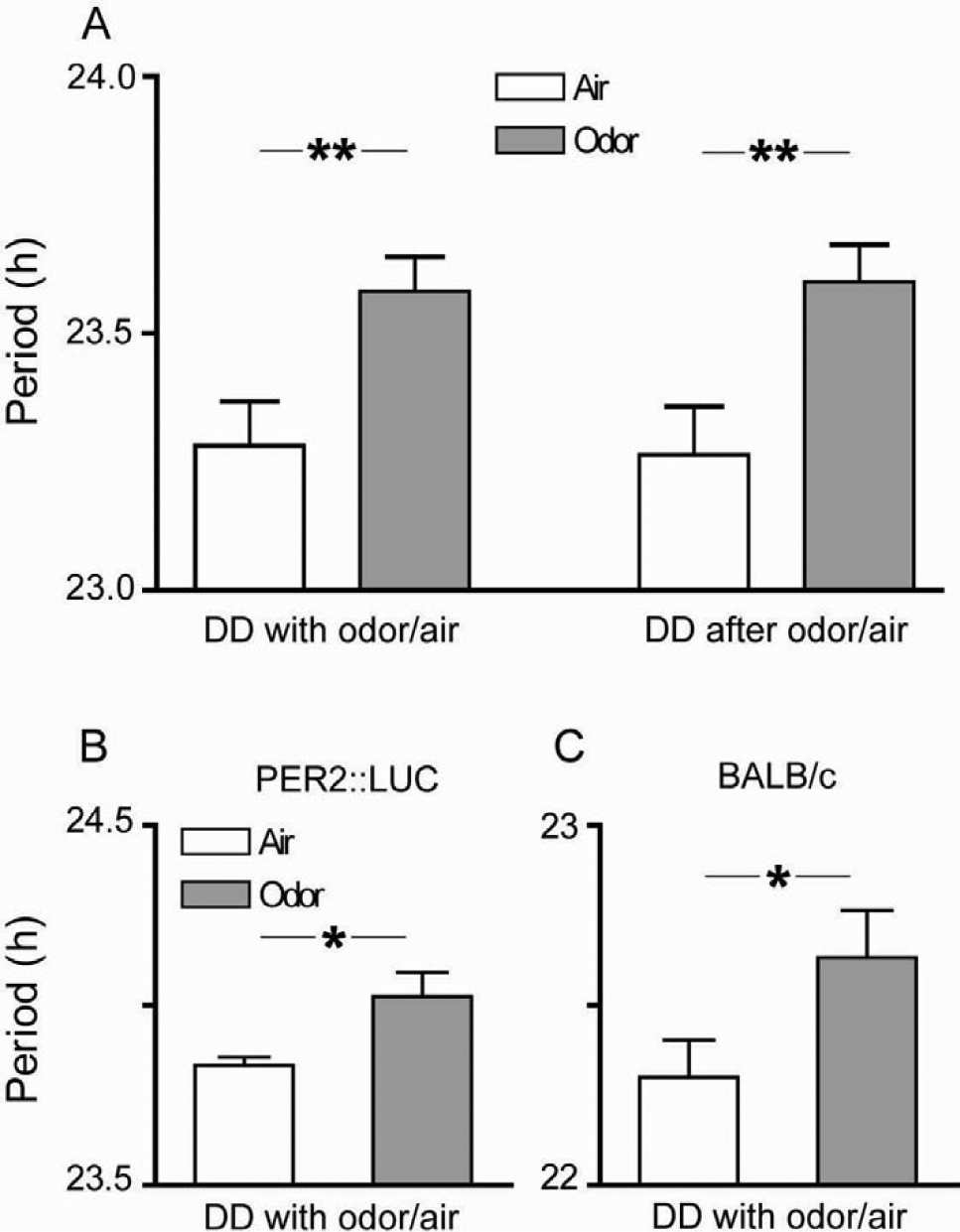
Periodic odor administration lengthens the activity period of mice and increases rhythmicity. (A) Mice that received daily 30-min presentations of odor mix at ZT16 and ZT23 in DD with odor/air displayed free-running behavioral activity with a period that was about 20 min longer than that of mice that received air only (unpaired t test, **p < 0.01, n = 11). The difference in periods persisted after the odor administration apparatus had been switched off (unpaired t test, **p < 0.01, n = 11). (B, C) Different mouse strains also showed a significant lengthening of the circadian locomotor activity rhythm upon odor treatment: PER2::LUC mice significantly lengthened their free-running period by about 14 min (B, Mann-Whitney test, *p < 0.05, n = 11-12 per group) and BALB/c mice by 18 min (C, Mann-Whitney test, *p < 0.05, n = 6 per group). Displayed are means
To further support our results on odor-induced period lengthening and to exclude effects brought along by subtle genetic differences in mice, we repeated the experiment with PER2::LUC mice, which genetically differ from C57Bl/6 mice only by a knockin of luciferase (Yoo et al., 2004). Consistent with odor effects being independent of minor genetic modifications, we found similar results as for C57Bl/6 mice: PER2::LUC mice lengthened their periods upon odor treatment by about 14 min (Fig. 2B) (odor-treated, 24.04 ± 0.1 h [n = 11]; air-only, 23.8 ± 0.02 h [n = 12]); Mann-Whitney test, p = 0.01). To also test whether odor effects are mouse strain-specific, we further investigated BALB/c mice and found that they, too, lengthened their activity periods by 18 min in response to odor treatment (Fig. 2C) (odor-treated, 22.6 ± 0.1 h [n = 6]; air-only, 22.3 ± 0.1 h [n = 6]; a free-running locomotor activity period of BALB/c mice that is about 1 h shorter than that of C57Bl/6 mice has already been reported by Schwartz and Zimmerman, 1990; Mann-Whitney test, p < 0.05). In contrast to C57Bl/6 mice, significant period lengthening in BALB/c mice did not persist after termination of odor treatment (after odor, 23.0 ± 0.15 h [n = 6]; after air-only, 22.8 ± 0.2 h [n = 6]; Mann-Whitney test, p > 0.05).
To test whether simultaneous administration of odor and light is necessary for the observed odor effects, we exposed C57Bl/6 mice to daily odor administration without simultaneous light treatment. Odor was given in constant darkness at times corresponding to ZT16 and ZT23 of the previous light-dark cycle. In this experimental setup, we did not detect a lengthening of the free-running periods upon odor administration (odor: 23.5 ± 0.09 h [n = 4]; air-only, 23.7 ± 0.07 h [n = 6]; Mann-Whitney test, p = 0.13). This suggests that simultaneous light and odor treatments are required for odor presentations to have a sustained effect on circadian behavior.
Odor Promotes Reemergence of Circadian Rhythmicity in Some SCN-lesioned Mice
We hypothesized that odor-mediated effects on circadian behavior might be brought about by the OB clock itself, to some extent independent of the SCN. This assumption is supported by the fact that primary olfactory information reaches the OB via a single neuronal projection from the olfactory epithelium. This is in contrast to other sensory inputs, which are relayed in clock-controlled hypothalamic and thalamic nuclei before they finally reach cortical areas (Butler and Hodos, 1996). Hence, olfactory inputs, and perhaps other nonphotic sensory signals, are processed without the immediate influence of the central circadian clock. Given that timed odor pulses very well do carry temporal information (see circadian odor effects), it is conceivable that these effects are mediated by the self-sustained, SCN-independent OB clock rather than by the SCN (Abraham et al., 2005). To test this hypothesis, we exposed SCN-lesioned and, consequently, behaviorally arrhythmic mice to either odor or air. Initially, we exposed mice to a skeleton photoperiod (1 h of light: 10 h of darkness: 1 h of light: 12 h of darkness) to avoid masking of behaviorally arrhythmic mice by a 12-h light phase. The skeleton photoperiod usually causes intact mice to synchronize their activity phase with the 12-h dark phase (i.e., the mice interpret this phase as “night”), while SCN-lesioned mice are no longer able to entrain their behavior (representative examples in Fig. 3, A-C). Daily odor presentations at ZT16 and ZT23 were applied during the skeleton photoperiod and persisted after transfer to DD. If odor acts as a circadian zeitgeber for an extra-SCN clock that can modulate behavioral rhythms, we expect a reconstitution of behavioral rhythmicity, at least to some extent. Indeed, the median relative amplitudes of the circadian component in SCN-lesioned mice were higher for mice treated with odor (Figs. 3 and 4A) (odor, 1.35 [min: 0.31, max: 4.12]; air-only, 0.43 [min: 0.3, max: 2.24]; after odor, 1.02 [min: 0.31, max: 3.93]; after air-only, 0.57 [min: 0.24, max: 1.1]; n = 5). The high variability in odor responses (Figs. 3 and 4A) becomes also apparent upon investigation of individual actograms: Whereas some mice respond weakly to odor presentations (Fig. 3A, right panel), other mice respond with reemergence of free-running (Fig. 3B, right panel) or even entrained (Fig. 3C, right panel) behavioral rhythms. The phasings of these reemerged rhythms, where detectable, were not random: The initial onsets of activity can be located at ZT4 (Fig. 3B, C; in reference to the skeleton photoperiod) in 2 of 5 SCN-lesioned mice. To exclude random group effects, 3 SCN-lesioned mice were subjected to 2 subsequent experiments, where 2 mice were treated with air only first and, subsequently, with odor, and 1 mouse received the reverse treatment. Also in this crossover design, median rhythm strength was higher in odor-treated SCN-lesioned mice than in mice receiving air only (odor, 1.35 [min: 0.44, max: 2.56]; air-only, 0.42 [min: 0.31, max: 0.48]; after odor, 1.47 [min: 0.66, max: 3.93]; after air-only, 0.25 [min: 0.24, max: 1.1]). Hence, odor effects in SCN-lesioned mice were specific and independent of the age of the animals.
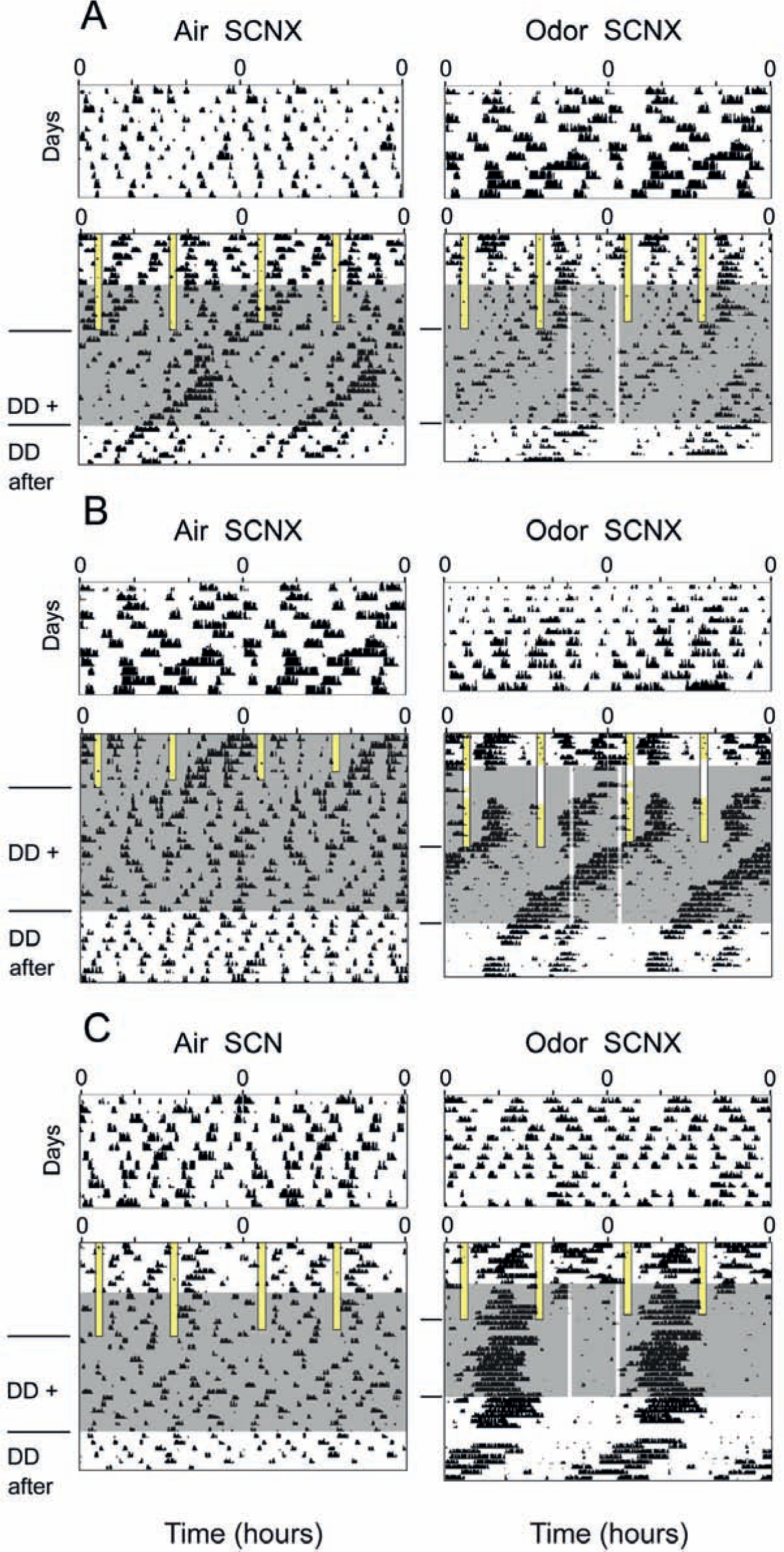
Odor presentations promote the reemergence of circadian rhythmicities in SCN-lesioned, arrhythmic mice. Double-plotted actograms show wheel-running activities of SCN-lesioned mice at different times of day. Wheel running activity did not synchronize with the skeleton photoperiod (two 1-h light pulses administered daily [LD 1:10:1:12], vertical bars with black borders = light phases), which is indicative of arrhythmicity. Control mice (“Air SCNX”) were subjected to constant air flow (shaded blue areas, left panels), while the experimental group (“Odor SCNX”) was exposed to constant air flow (shaded gray areas, right panels) enriched by 30-min presentations of a gaseous odor mix, administered daily at ZT16 and ZT23 (in reference to the skeleton light cycle, vertical transparent-white bars, right panels). Upon release into constant darkness with persisting odor/air conditions, in some individuals (B, C, right panels) residual rhythmicity emerged and consolidated into robust free-running circadian rhythmicity (B, right panel). (C) The period of the reemerged circadian rhythmicity was synchronized with the 24-h odor presentations, with an activity phase between ZT4 and ZT10 (with reference to the skeleton photoperiod). After the odor presentations had stopped, behavioral rhythmicity free-ran with a period shorter than 24 h (DD after, right), its phase consistent with entrainment by odor presentations in DD+. Residual behavioral rhythmicity was more likely to appear in odor-treated compared with air-treated mice (compare left and right panels), and, if so, its phase originated from activity during the subjective day (in reference to the skeleton photoperiod), between ZT4 and ZT10 (B, C, right panels). Actograms shown in A, right panel, and B, left panel, were obtained from the same mouse, subjected to a crossover design: The individual was first exposed to air only (B, left panel) and then, in a subsequent experiment, was exposed to odor (A, right panel). To demonstrate that all actograms were obtained from previously arrhythmic mice, we show on top of each actogram a locomotor activity recording of the respective mouse that was recorded in constant conditions, prior to the experiment. Postexperimental histological analysis also revealed that all mice displayed here had complete SCN lesions. Residual rhythmicities quantified from the behavioral data shown above are highlighted in Fig. 4A.
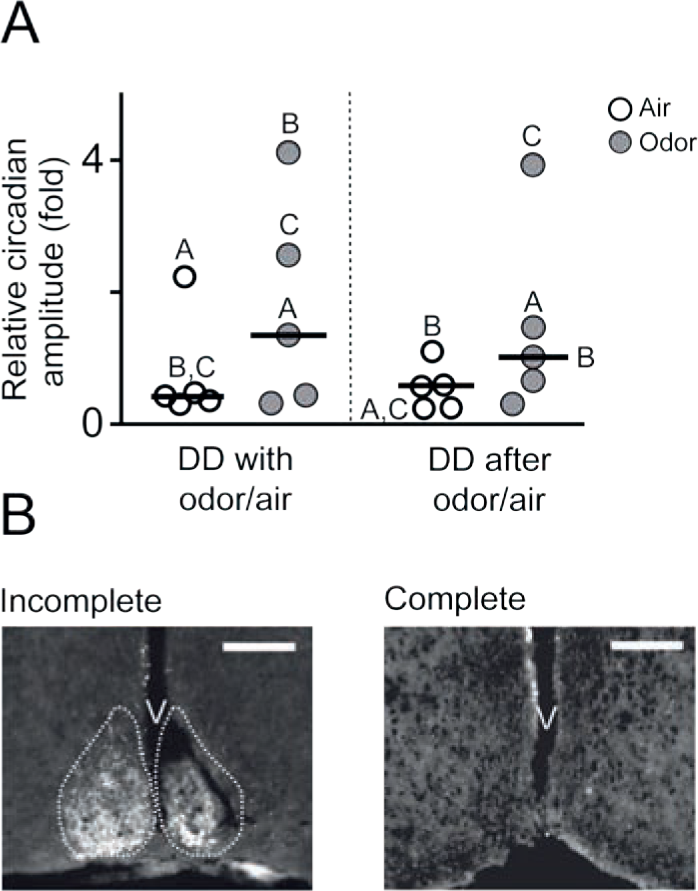
(A) χ2-periodogram analysis revealed that odor presentations yielded higher relative circadian amplitudes in SCN-lesioned mice than air only. Displayed are medians (black lines) and individual values of all mice bearing complete SCN lesions. Amplitude values that were derived from the behavioral data shown in Fig. 3 are marked by the responding letters (A-C for “with odor/air” and A-C for “after odor/air”). (B) Examples of incomplete and complete SCN lesions. Incomplete: immunofluorescence staining (white) against vasoactive intestinal polypeptide (VIP) shows 1 intact nucleus (left) and an extensive amount of surviving SCN cells in the other nucleus (cross-section, medial SCN, scale bar = 200 µm). The approximate borders of an intact SCN are outlined by dashed lines. Complete: the lack of immunofluorescence staining (white) against VIP demonstrates the absence of living SCN tissue (cross-section, medial SCN, scale bar = 200 µm; staining was obtained from the odor-treated mouse in Fig. 3B, right panel). V = third ventricle.
All animals included in the behavioral experiments were clearly arrhythmic prior to the experiments and did not entrain to the skeleton photoperiod. To additionally exclude that odor effects were compromised by incomplete SCN lesions, we immunohistochemically visualized the extents of the lesions. Only mice with complete SCN lesions (see Fig. 4B) were included in the behavioral analysis. Thus, the effects of odor on circadian behavior seem to be partially independent of the SCN and might involve yet unknown extra-SCN circadian oscillators.
Odor-driven Reemergence of Rhythmicity Requires Canonical Clock Genes
Although it is difficult to anatomically identify the extra-SCN area that might be involved in odor-driven rhythm regulation, we can narrow down our search by testing whether this area requires the presence of a functioning circadian clock. In other words, we asked whether circadian oscillations of canonical clock genes are needed for the re-emergence of rhythmicity upon odor pulses in otherwise behaviorally arrhythmic mice. To answer this question, we used cry1/cry2 double knockout mice, which have been shown to exhibit immediate behavioral arrhythmicity in constant conditions (van der Horst et al., 1999) as well as arrhythmic patterns of clock gene expression at the cell and tissue level (Yamaguchi et al., 2003). These mice were subjected to a similar experiment as the SCN-lesioned mice, but, in contrast to those, odor pulses did not promote reemergence of circadian rhythmicity in cry1/cry2 double knockout mice (Fig. 5, A and B) (median circadian amplitude with odor, 0.17 [min: 0.07, max: 1.1], n = 12; median circadian amplitude with air-only, 0.14 [min: 0.07, max: 0.4], n = 12; Wilcoxon signed-rank test, p > 0.05; median circadian amplitude after odor, 0.15 [min: 0.0, max: 1.1], n = 12; median circadian amplitude after air only, 0.2 [min: 0.0, max: 1.1], n = 12; Wilcoxon signed-rank test, p > 0.05). This suggests that canonical clock function is necessary for odor to affect circadian behavior.
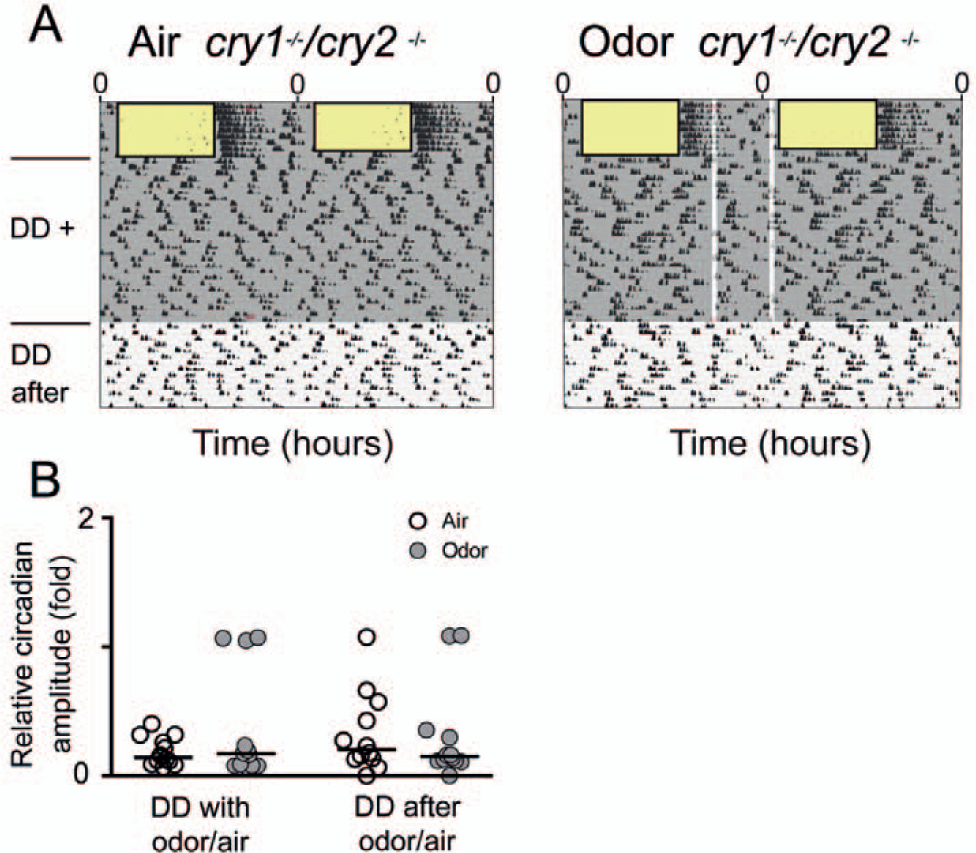
Odor presentations did not promote reemergence of circadian rhythmicities in cry1/cry2 double knockout mice. (A) Representative double-plotted actograms showing wheel-running activities per 6 min of cry1/cry2 double knockout mice at different times of day. Wheel-running activity completely synchronized with the light-dark cycle (rectangles with black borders = light phases), suggesting strong masking by light. Control mice were subjected to constant air flow (shaded gray area, left panel), whereas the experimental group was exposed to constant air flow (shaded gray area, right panel) enriched by 30-min presentations of a gaseous odor mix, administered daily at ZT16 and ZT23 (in reference to the light phase, vertical transparent-white bars, right panel). Upon release into constant darkness, locomotor behavior immediately became arrhythmic, which is consistent with a severe loss of clock function (van der Horst et al., 1999). (B) In contrast to SCN-lesioned mice, residual circadian rhythmicities in odor-presented cry1/cry2 double knockout mice (n = 12) were not significantly higher than in mutant mice that received air only (Wilcoxon signed-rank test, p > 0.05; n = 12). This was true for both conditions, during and after odor/air presentations. Displayed are medians (black lines) and individual values.
Discussion
Odors report the availability or presence of essential environmental factors, for example, food and predators, and are ubiquitous constituents of an organism’s surroundings. Hence, it is conceivable that odor is able to influence one of the fundamental properties of most organisms: the endogenous rhythm regulation. Nevertheless, most studies investigating the effects of environmental factors on mammalian daily rhythms neglect variations in the olfactory environment, thereby missing out on a potential synchronizing stimulus. For example, odors from entrained conspecifics partially entrained circadian behavior of female Octodon degus (Governale and Lee, 2001). We systematically studied the effects of periodic odor presentations in mice and showed that periodic odor administration results in a robust lengthening of the free-running behavioral period in 3 different mouse strains and leads to reemergence of circadian rhythmicity in SCN-lesioned mice. Both findings are consistent with odor functioning as a circadian time cue, in particular when the major circadian pacemaker, the SCN, is absent.
Odor mix compounds were selected to represent odors that mice do not usually encounter, but nonetheless, for which they carry the corresponding receptors (Zou and Buck, 2006). Although our odors are arguably not as biologically relevant as, for example, the smell of food, a predator, or conspecific urine, we specifically selected them to avoid associations with learned tasks or flight and reproductive responses that might increase general arousal (Mistlberger and Skene, 2004). These complex behaviors might obscure a potential systematic link between the sense of smell and circadian function. We reasoned that if the circadian system has evolved to respond to odors, the result might very likely be a general mechanism that applies to any odor that triggers an olfactory response. In fact, odors that are not part of the natural environment of mice, for example, cedar wood oil, have already been shown to elicit genetic responses within the circadian system (Amir et al., 1999). Although Amir et al. (1999) used a single 15-min odor pulse in constant darkness and did not see any influence on circadian behavior, daily odor presentations captured the mice’s locomotor activity rhythms and lengthened their endogenous periods toward the frequency of the external time cue. The lack of behavioral synchronization by periodic odor presentations can be accounted for by at least 3 scenarios: The first possible mechanism refers to the circadian input: Since our mice are allowed to freely move around their cages, we cannot determine the exact concentration of odor that they are exposed to during the odor presentations. As a consequence, the intensity of the olfactory responses during odor presentations may vary between days and different times of days, depending on the spatial preferences of the mice. These differences in perceived odor cue intensity may contribute to the large variabilities in behavioral phenotypes of odor-treated mice. The latter is particularly evident in SCN-lesioned mice, where some mice strongly react to odor presentations and others respond weakly (Fig. 3). A second hypothesis suggests that odor pulses do indeed affect the circadian pacemaker but that they are more effective in shifting the clock at some circadian times than at others. Because odor presentations hit the free-running circadian system at slightly different times during each cycle, it is possible that phase shifts are achieved only when odor hit at a sensitive circadian time, resulting in a mild form of synchronization, that is, slight period modulation of the free-running rhythm (Pittendrigh and Daan, 1976b). But there is also a third reason that might account for the period modulation: It is possible that odor cues do not phase shift the circadian clock at any given time but rather modulate its period by directly acting on general arousal centers (“masking”). However, we did not observe immediate odor effects on the locomotor activity levels (Fig. 1, A-C) that would be indicative of masking. Nevertheless, we cannot definitely exclude that repeated odor presentations alter the animals’ endogenous period, similar to what has been described for other external factors (e.g., running wheel in cage; Weisgerber et al., 1997).
The variability in odor-induced responses may contribute to the fact that single odor presentations did not result in behavioral effects (data not shown) and, consequently, are not an appropriate measure to determine whether odor functions as a zeitgeber. This is consistent with what has been observed for single presentations of other weak zeitgebers, for example, social cues (reviewed in Mistlberger and Skene, 2004). In contrast to that, strong zeitgebers (e.g., light) are able to fully synchronize behavior and exert significant shifts of the circadian rhythm following just a single stimulus presentation (Pittendrigh and Daan, 1976b). Because of the nondetectable effects of single odor stimuli, we reverted to an entrainment paradigm with repeated odor presentations and achieved a period modulation of circadian behavior. Although period modulation is certainly not entrainment, the fact that period lengthening persisted for at least 7 days poststimulation suggests that odor was able to sustainably influence aspects of core clock function rather than merely drive behavior by activation of general arousal centers (Mistlberger and Skene, 2004). Persistence of period modulation is reminiscent of aftereffects or transients, which occur in rhythms that had previously been phase shifted or entrained by a zeitgeber (Pittendrigh and Daan, 1976a) but also in rhythms that are subject to a change in certain constant environmental conditions (Aschoff, 1960). Although we cannot reliably distinguish between aftereffects and transients in our results (due to the short duration of the recording after termination of odor presentations), both are manifestations of a significant impact on the circadian oscillator (Pittendrigh and Daan, 1976a), supporting our hypothesis of odor as a time cue. However, further evidence might be obtained from experiments with periodic odor presentations varying from 24 h (different odor T-cycles), which will allow us to evaluate whether odor-induced period aftereffects are similar to those observed for light entrainment (Aton et al., 2004). The discrepancy between odor-induced effects in C57Bl/6 and BALB/c mice (C57Bl/6 mice showed significant period lengthening beyond periodic odor administration, whereas BALB/c mice did not) is puzzling but appears inconclusive in the light of the comparatively small number of BALB/c mice (n = 6), the resulting low power of the statistical analysis, and the small numbers of recording days after odor available for period measurements. Assuming that this discrepancy is indeed significant, it might very well, at least in part, be due to mouse strain-specific differences in odor perception or odor responsiveness.
Which general mechanisms underlie odor-modulated circadian behaviors? At least 3 scenarios are conceivable: (1) the olfactory epithelium in the mouse’s nasal cavity transmits olfactory signals to the OB modulating the OB clock, which, in turn, feeds back on the SCN clock adjusting circadian behavior; (2) olfactory timing signals are transmitted to the SCN and/or to yet unknown central nervous structures which, besides the SCN, regulate circadian behavior; or (3) a combination of both. Our results do not allow for a definite distinction between these hypotheses. The lengthening of the circadian period and simultaneous increase in rhythm strength by odor presentations could be achieved by odor acting via the SCN, or via an extra-SCN oscillator, or both. However, the synergistic effect of odor and light that we and others (Governale and Lee, 2001; Amir et al., 1999; Granados-Fuentes et al., 2006) have reported supports Hypothesis 1, because it suggests that photic input alters the SCN’s sensitivity for odor presentations and, hence, indicates that the SCN plays a major role in odor entrainment. In contrast, the data from SCN-lesioned mice are in favor of Hypothesis 2, since they suggest that odor-induced effects on circadian behavior are even stronger when the SCN is absent. This is all but impossible since there are extra-SCN brain areas that respond to odor presentations with a change in clock gene expression (Pantazopoulos et al., 2011) and that are able to control circadian locomotor behavior under certain conditions, for example, the food-entrainable or the methamphetamine-sensitive oscillators (Honma et al., 1988; Mistlberger, 1994). The “odor-sensitive” structure might even be the same structure as one or the other mentioned above. In contrast to the food-entrainable oscillator that is able to control behavior in the presence of an entraining light-dark cycle (Stokkan et al., 2001), odor presentations did not affect circadian behavior during light-dark (Fig. 1), suggesting that the odor-sensitive structure is usually dominated by a light-dark-entrained SCN. Therefore, odor effects may be even more pronounced when the SCN are absent or weakened, which was underlined by the reemergence of robust circadian rhythmicity in response to odor presentations in some SCN-lesioned mice (Fig. 3, B and C). The fact that reemerged circadian rhythms initially assume a rather stable phase relationship to the odor presentations (i.e., the phases of the reemerged rhythms occur at the same times with respect to the odor presentations) supports the idea that odor is indeed acting as a weak entraining stimulus in the absence of the SCN. However, given the large variabilities of odor-induced responses in SCN-lesioned mice (Fig. 3) and the low number of mice with reemerged rhythms distinct enough to detect a phase (n = 2), this conclusion might be premature.
A second trait that perhaps distinguishes the odor-sensitive structure from the methamphetamine-sensitive oscillator is its dependence on circadian clock function: Although the present study could not demonstrate odor effects on the behavior of mice lacking a functional circadian clock (Cry1/Cry2 double knockouts), clock knockout animals have been demonstrated to show reemergence of behavioral rhythmicity in response to methamphetamine treatment (Mohawk et al., 2009). However, whether the food-entrainable oscillator depends on canonical clock function is still under debate (Feillet et al., 2006; Fuller et al., 2008; Storch and Weitz, 2009). Furthermore, our data are consistent with previous studies reporting odor-induced effects on the activity of the autonomous nervous system in mice, which depend on the presence of a functional circadian clock (Tanida et al., 2006; Tanida et al., 2008). In line with that, clock genes are also required for the autonomous peripheral oscillator controlling olfactory rhythms in insects (Tanoue et al., 2004; Zhou et al., 2005) and for the generation of circadian sensitivity in mammals (Granados-Fuentes et al., 2011).
In conclusion, we demonstrate that odor influences circadian behavior via an SCN-independent but clock gene–dependent pathway. The latter suggests that a clock outside the SCN mediates this effect. Given that the system processing olfactory information contains an autonomous circadian oscillator using canonical clock genes, it appears likely that this clock might actually reside within the olfactory system.
Ultimately, the present study supports the general hypothesis of odor as a potential time cue for the circadian clock. These results will shed new light on the evolutionary basis of circadian regulation and will help us to understand the design of the mammalian circadian system. Furthermore, they will open up new opportunities for noninvasive pharmacological treatments of clock-related diseases.
Footnotes
Acknowledgements
The authors thank Raik Paulat from the Medical-Technical Laboratories of the Charité-Universitätsmedizin Berlin, who helped to construct and built the time-controlled odor delivery system, and Dr. Erik Herzog for helpful discussions of the manuscript. The present study was supported by the Deutsche Forschungsgemeinschaft (grant AB139/4-1 to U.A.) and the Bernstein Center for Computational Neuroscience, Berlin. Work in A.K.’s laboratory is further supported by the Deutsche Forschungsgemeinschaft and the 6th EU framework program EUCLOCK.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
