Abstract
Overlapping genetic influences have been implicated in diurnal preference and subjective sleep quality. Our overall aim was to examine overlapping concurrent and longitudinal genetic and environmental effects on diurnal preference and sleep quality over ~5 years. Behavioral genetic analyses were performed on data from the longitudinal British G1219 study of young adult twins and nontwin siblings. A total of 1556 twins and siblings provided data on diurnal preference (Morningness-Eveningness Questionnaire) and sleep quality (Pittsburgh Sleep Quality Index) at time 1 (mean age = 20.30 years, SD = 1.76; 62% female), and 862 participated at time 2 (mean age = 25.30 years, SD = 1.81; 66% female). Preference for eveningness was associated with poorer sleep quality at both time points (r = 0.25 [95% confidence intervals {CIs} = 0.20-0.30] and r = 0.21 [CI = 0.15-0.28]). There was substantial overlap in the genetic influences on diurnal preference and sleep quality individually, across time (genetic correlations [rAs]: 0.64 [95% CI = 0.59-0.67] and 0.48 [95% CI = 0.42-.053]). There were moderate genetic correlations between diurnal preference and sleep quality concurrently and longitudinally (rAs = 0.29-0.60). Nonshared environmental overlap was substantially smaller for all cross-phenotype associations (nonshared environmental correlations (rEs) = -0.02 to 0.08). All concurrent and longitudinal associations within and between phenotypes were largely accounted for by genetic factors (explaining between 60% and 100% of the associations). All shared environmental effects were nonsignificant. Nonshared environmental influences played a smaller role on the associations between phenotypes (explaining between -0.06% and 40% of the associations). These results suggest that to some extent, similar genes contribute to the stability of diurnal preference and sleep quality throughout young adulthood but also that different genes play a part over this relatively short time frame. While there was evidence of genetic overlap between phenotypes concurrently and longitudinally, the possible emergence of new genetic factors (or decline of previously associated factors) suggests that molecular genetic studies focusing on young adults should consider more tightly specified age groups, given that genetic effects may be time specific.
The leading theory of sleep and wakefulness posits that the timing of our sleep-wake system is controlled by 2 processes. One is circadian and the other homeostatic (Borbély, 1982). The circadian system maintains 24-h daily physiological rhythms by a self-sustained oscillator located in the suprachiasmatic nucleus of the hypothalamus. The circadian system is synchronized to external time cues and influences the timing of sleep. It is possible to obtain a crude index of individual differences in circadian rhythms by subjectively assessing preferences for the timing of sleep-wake activities. Diurnal preference has repeatedly been assessed using the Morningness-Eveningness Questionnaire (MEQ; Horne and Östberg, 1976), which provides a subjective account of one’s tendency toward morning or evening activity. Diurnal preference as assessed by the MEQ has been shown to appropriately differentiate morning and evening types in terms of the timing of the circadian phase markers of melatonin, core body temperature, and subjective sleepiness (Lack et al., 2009). The homeostatic process refers to the rise in sleep drive, which accumulates with wakefulness and dissipates during sleep (Borbély and Achermann, 2011). Slow-wave sleep (SWS), often considered the “refreshing” stage of sleep, is used as a marker of homeostatic sleep pressure. Although it may be difficult to obtain a subjective measure of sleep homeostasis, assessment of the subjective quality of one’s sleep could provide an indirect measure of SWS and, by proxy, of the efficiency of the homeostatic drive. The Pittsburgh Sleep Quality Index (PSQI: Buysse et al., 1989) assesses 7 components of sleep quality pertaining to subjective sleep quality, sleep onset latency, sleep duration, sleep efficiency, sleep disturbances, use of sleep medication, and daytime dysfunction. While indices of the circadian system and homeostatic drive may be considered in some ways to be independent, it is possible that they are interlinked in terms of the behavior they dictate as well as their underlying mechanisms. For example, it is possible that diurnal preference could be considered to be an index of the homeostatic drive; that, a faster buildup of sleep propensity could drive people toward a morning preference whereby they go to bed and arise earlier. Not surprisingly, these subjective assessments of diurnal preference and sleep quality are often associated (Barclay et al., 2010a; Megdal and Schernhammer, 2007; Merikanto et al., 2012; Soehner et al., 2011; Tzischinsky and Shochat, 2011). In these studies, eveningness (relative to morningness) is typically associated with various indices of poor sleep quality.
Behavioral genetic studies have repeatedly demonstrated that genetic influences account for about half the variability in diurnal preference (Barclay et al., 2010a; Drennan et al., 1992; Hur et al., 1998; Koskenvuo et al., 2007; Vink et al., 2001) and about 30% to 40% in sleep quality (Barclay et al., 2010a, 2010b; Genderson et al., 2013; Heath et al., 1990; Partinen et al., 1983). Analyses of the current data set have demonstrated that the association between diurnal preference and sleep quality is largely influenced by shared (also referred to as common; overlapping) genetic factors (Barclay et al., 2010a). However, little is known about the stability of this genetic overlap over time.
The organization of sleep and the circadian system change across developmental periods (Bliwise, 2005). The association between diurnal preference and sleep quality, as well as the contribution of genetic and environmental influences on this association, may also change over particular developmental periods. Accumulating evidence suggests that genetic and environmental influences on diurnal preference change with age (Barclay et al., 2014; Hur, 2007; Jones et al., 2007). A handful of studies have focused on the stability of the genetic and environmental influences on sleep quality over time (Barclay et al., 2015; Gregory et al., 2009), yet we know relatively little about the association between diurnal preference and sleep quality over time. It is important to address this question during young adulthood when physiology continues to develop and social responsibilities tend to increase (Arnett, 2000; Dahl and Lewin, 2002), factors that can affect both sleep timing and quality.
The current study assessed associations between diurnal preference and sleep quality concurrently and longitudinally in a sample of young adult twins and siblings from the G1219 study (for a description of this study, see McAdams et al., 2013). We examined 1) stability of diurnal preference and sleep quality over time; 2) associations between diurnal preference and sleep quality concurrently and longitudinally; 3) stability of genetic and environmental influences on diurnal preference and sleep quality, within phenotypes, over time; and 4) the extent of genetic and environmental overlap between phenotypes concurrently and longitudinally.
Materials and Methods
Participants
The present analyses focused on wave 4 and wave 5 (later referred to as times 1 and 2 for simplicity of presentation) of the G1219 and G1219Twins longitudinal studies (for a description of the sample, see McAdams et al., 2013).
At time 1 (wave 4), a total of 1556 individuals participated. Approximately 5 years later, at time 2 (wave 5), a total of 862 individuals participated. The mean age at time 1 was 20.30 years (SD = 1.76, range = 18-27 years), and 62% of the sample were female. The mean age at time 2 was 25.30 years (SD = 1.81, range = 22-32 years), and 66% of the sample were female. Following the study design, the majority of participants were close in age within each time point (at time 1, 90% of the participants were aged 18-22 years; at time 2, 90% of the participants were aged 22-27 years), but the inclusion of siblings inevitably created some age spread. Zygosity was established through a questionnaire measure completed by mothers at waves 2 and 3 assessing physical similarities between twins (Cohen et al., 1975). The number of monozygotic (MZ), dizygotic (DZ), and sibling (Sib) individuals as well as full pairs participating at each wave and providing data on each variable is displayed in Table 1.
Means (SD) of raw diurnal preference and sleep quality scores and number of individuals (N) and full pairs (FP). a
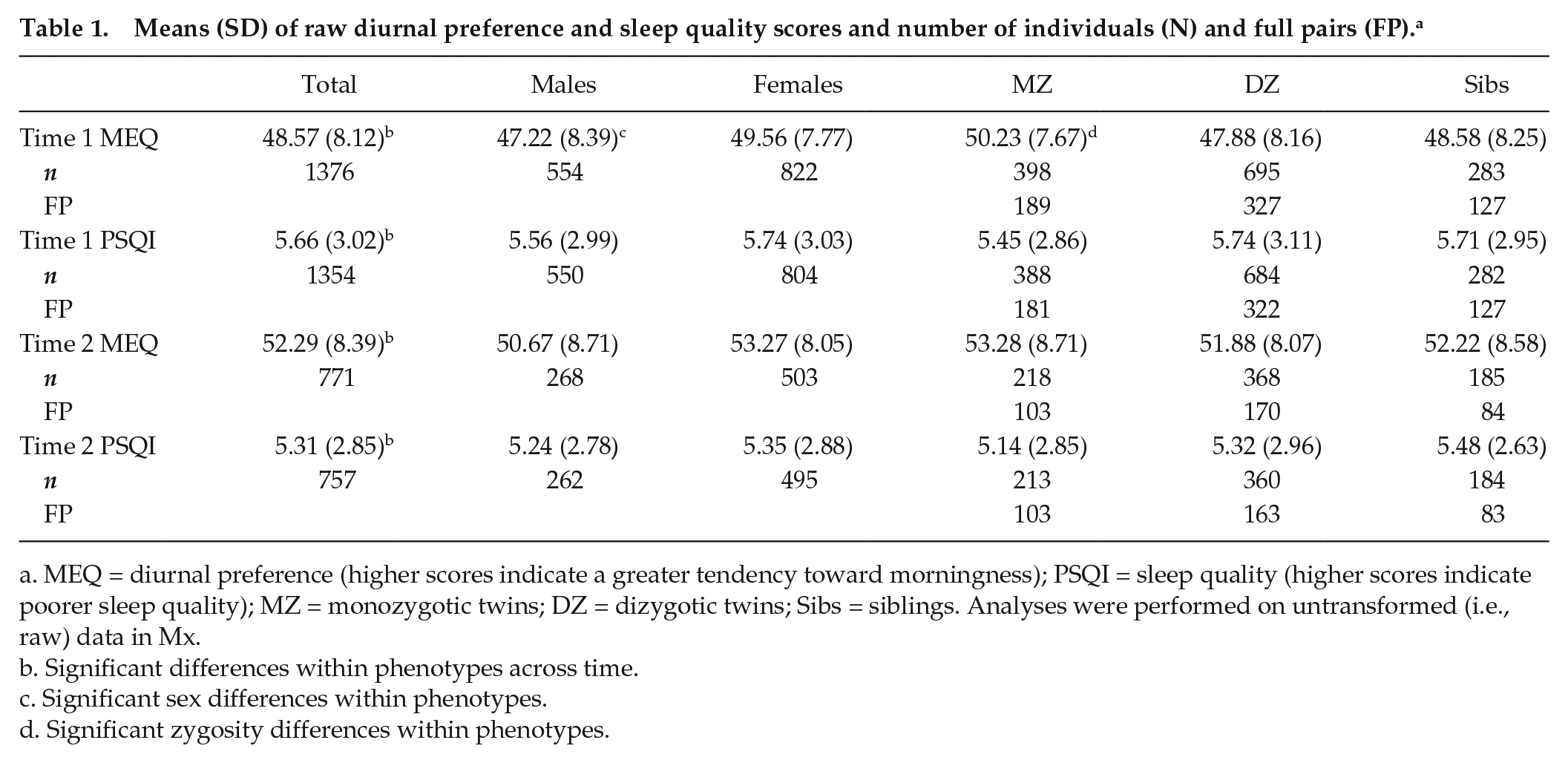
MEQ = diurnal preference (higher scores indicate a greater tendency toward morningness); PSQI = sleep quality (higher scores indicate poorer sleep quality); MZ = monozygotic twins; DZ = dizygotic twins; Sibs = siblings. Analyses were performed on untransformed (i.e., raw) data in Mx.
Significant differences within phenotypes across time.
Significant sex differences within phenotypes.
Significant zygosity differences within phenotypes.
Ethical approval for the collection of data at different waves was provided by the Research Ethics Committees of the Institute of Psychiatry, South London and Maudsley NHS Trust, and Goldsmiths, University of London. The protocol for data collection conformed to international ethical standards in accordance with the Declaration of Helsinki and Portaluppi and colleagues (2010).
Predictors of Attrition
Logistic regression, focusing on variables previously associated with attrition in this sample, demonstrated that responders at time 2 were significantly more likely than nonresponders to be female (odds ratio = 0.60, p < 0.001), have more highly educated mothers (odds ratio = 1.14, p < 0.001, where mother’s education was rated from the lowest level 0 to 8), and have a greater tendency toward morningness at time 1 (odds ratio = 1.02, p = 0.01). There were no significant differences between responders and nonresponders in terms of housing, sleep quality, or prosocial behavior at earlier waves (all p’s > 0.05).
Measures
The MEQ (Horne and Östberg, 1976) is a widely used measure of diurnal preference. It contains 19 self-report items measuring preference for the timing of daytime activities, sleeping habits, hours of peak performance, and times of “feeling best” and maximum alertness. A total score (range = 16-86) is derived in which higher scores indicate greater morningness. For the current study, this scale was reversed so that we could decompose a positive correlation for ease of interpretation for the reader. The MEQ demonstrated good internal reliability in the present sample at time 1 (Cronbach’s α = 0.78) and time 2 (α = 0.80).
The PSQI (Buysse et al., 1989) was used to examine sleep quality over the past month. The PSQI contains 18 scored items, which include both free-response questions (e.g., “During the past month, when have you usually gone to bed at night?”) and fixed-choice questions (e.g., “During the past month, how would you rate your sleep quality overall?” “Very good,” “Fairly good,” “Fairly bad,” or “Very bad”). The total score (range = 0-21) provides an overall sleep quality index, with higher scores indicating poorer quality. The PSQI demonstrated good internal reliability in the present sample at both time points (Cronbach’s α = 0.71 at both time points). Barclay et al. (2010a) provide further information on these measures.
Statistical Analyses
Data Preparation
Skew was not problematic for the MEQ or PSQI at either time point (time 1 MEQ skew = -0.16, [SE = 0.06], PSQI skew = 0.91, [SE = 0.06]; time 2 MEQ skew = -0.07, [SE = 0.08], PSQI skew = 0.93 [SE = 0.09]). Prior to analysis, the data were age and sex regressed, as has been recommended for twin modeling (McGue and Bouchard, 1984). Outliers of 3 or more standard deviations above or below the mean were omitted (n = 34 cases).
Genetic Analytic Principles Using Twin Data
Genetic models were fitted using maximum likelihood in Mx (Neale, 1997). Twin studies compare the similarity within MZ twin pairs to the similarity within DZ twin pairs (or full siblings) to estimate genetic influences on traits. Since MZ twins share 100% of their genes while DZ twins and full siblings share 50% of genetic effect on average, this information estimates the relative contribution of 3 sources of variance affecting a phenotype: additive genetic influences (A) (where alleles at a locus “add up” to influence behavior), shared environmental influences (C) (environmental influences that act to make twins within a pair similar), and nonshared environmental influences (E) (environmental influences that act to make twins within a pair different; this source of variance also incorporates error). The twin model assumes that correlations between DZ twins and siblings are relatively equal. If this assumption is upheld, parameters can be confidently estimated in genetic models by constraining these estimates to be equal between groups. If the correlation between MZ pairs is greater than that of DZ/sibling pairs, genetic influences are indicated. MZ twin correlations equivalent to DZ/sibling correlations indicate the importance of the shared environment and the extent to which MZ twin correlations are smaller than 1 indicates the magnitude of nonshared environmental factors.
Bivariate twin correlations indicate the relative sources of variance accounting for the concurrent and longitudinal phenotypic correlations. These were modeled using a correlated factors approach (see Figure 1). This model provides information on the univariate estimates and the overlap in the genetic and environmental influences on each phenotype at each time point. This information can be used to calculate the proportion of genetic and environmental influences accounting for the concurrent and longitudinal associations.
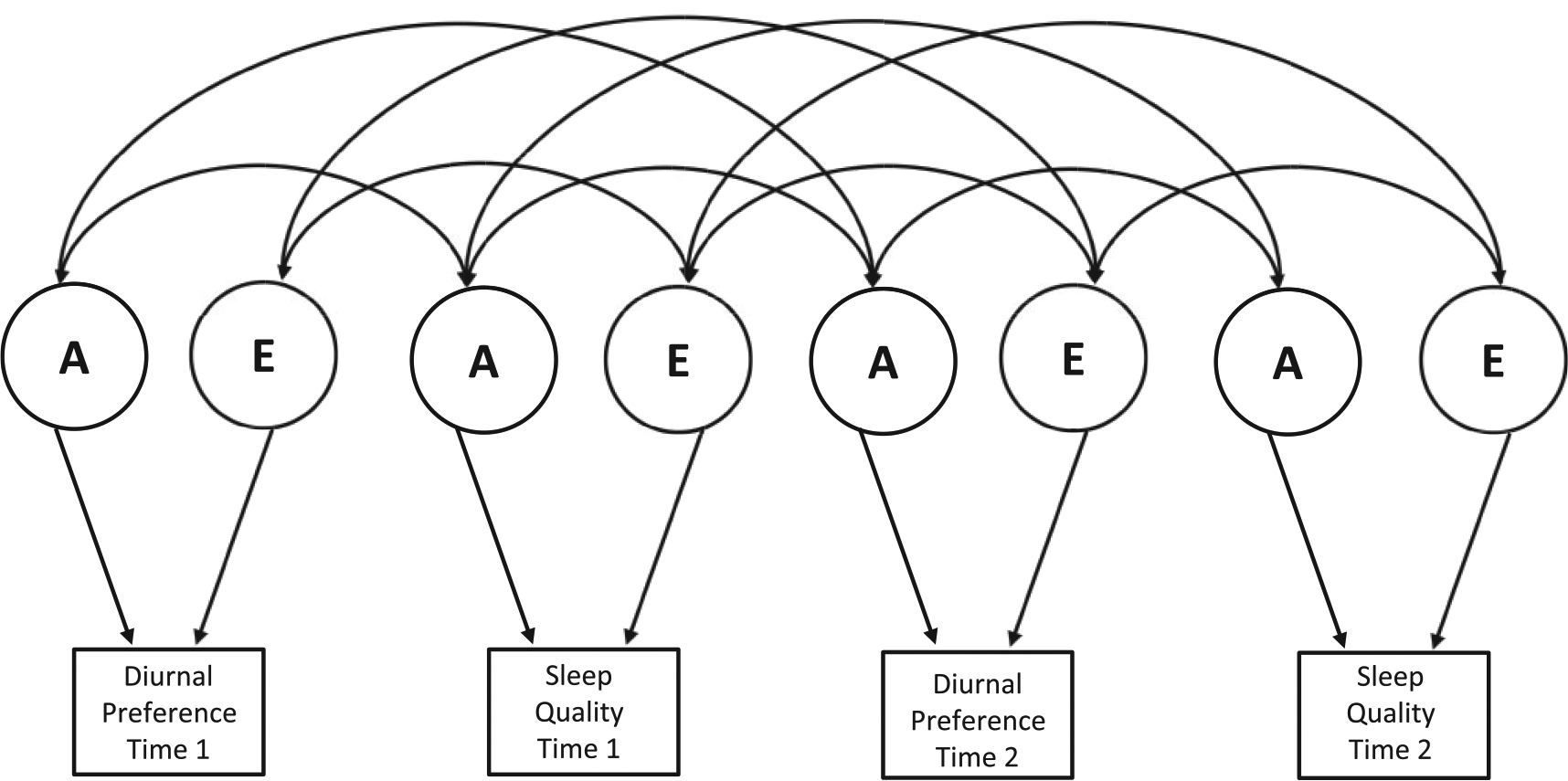
Multivariate correlated factors model. A = additive genetic influence; E = nonshared environmental influence. Figure is shown for 1 twin/sibling only.
The fit statistic provided by Mx for raw data modeling is -2LL (minus twice the log likelihood of the observations). Saturated models, which estimate the maximum number of parameters required to describe the variance-covariance matrix and means of observed variables, providing a perfect fit to the data, are first approximated to the data. The -2LL of a saturated model is then subtracted from the -2LL of the genetic model, with lower values indicating better genetic model fit. The difference is χ2 distributed; nonsignificance indicates that the genetic model does not fit the data less well than the saturated model and therefore provides a good description of the data. We first tested a scalar-sex limitation ACE model that allows quantitative sex differences. This was then followed by a homogeneity model in which male and female parameters were equated. Finally, we ran an AE homogeneity model to determine whether the C parameter could be dropped without significantly reducing model fit.
Results
Descriptives
Table 1 (and Supplementary Figures S1-S12) shows the means and standard deviations of diurnal preference and sleep quality scores from time 1 and 2, split by sex and zygosity. Compared with females, males tended toward eveningness at times 1 (fully constrained model compared with sex differences model: Δχ2 = 27.94, Δdf = 2, p < 0.001) and 2 (Δχ2 = 17.44, Δdf = 2, p < 0.001). Sleep quality did not differ between males and females. There were zygosity differences in diurnal preference (Δχ2 = 17.51, Δdf = 4, p < 0.001), where MZ twins tended toward morningness compared with DZ twins and siblings at time 1 but not time 2, and no zygosity differences in sleep quality. There was a significant increase in morningness across time (Δχ2 = 162.44, Δdf = 2, p < 0.001) and a significant improvement in sleep quality (Δχ2 = 9.79, Δdf = 2, p < 0.001).
Zero-Order Phenotypic Correlations
As previously noted (Barclay et al., 2010a), time 1 eveningness was associated with poorer sleep quality (r = 0.25 [95% confidence intervals [CIs] = 0.20-0.30]), and this was similar at time 2 (r = 0.21 [CI = 0.15-0.28]). There was substantial stability for each phenotype across time (diurnal preference: r = 0.64 [CI = 0.59-0.67]; sleep quality: r = 0.48 [CI = 0.42-0.53]). The cross-trait longitudinal correlations indicated that diurnal preference at time 1 was significantly associated with sleep quality at time 2 (r = 0.14 [CI = 0.07-0.20]). Likewise, time 1 sleep quality was significantly associated with time 2 diurnal preference (r = 0.20 [CI = 0.14-0.26]).
Univariate Twin Correlations
As shown in Table 2, all univariate MZ twin correlations were significant (range r = 0.24-0.52), as were 3 of the univariate DZ twin correlations (range r = 0.17-0.26). All univariate sibling correlations were nonsignificant (range r = 0.02-0.16).
Univariate and bivariate twin correlations (95% confidence intervals) for diurnal preference and sleep quality at time 1 and 2. a
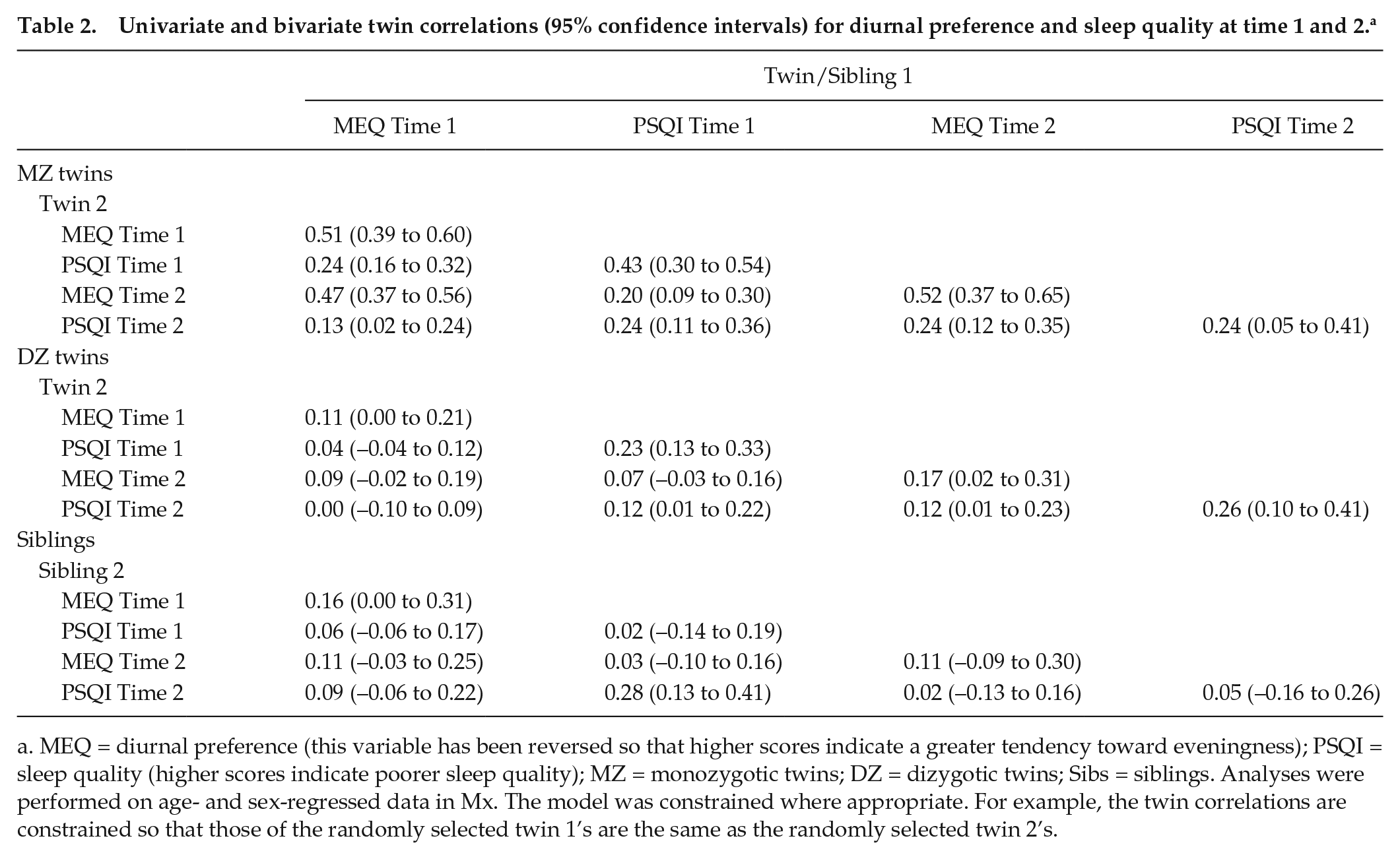
MEQ = diurnal preference (this variable has been reversed so that higher scores indicate a greater tendency toward eveningness); PSQI = sleep quality (higher scores indicate poorer sleep quality); MZ = monozygotic twins; DZ = dizygotic twins; Sibs = siblings. Analyses were performed on age- and sex-regressed data in Mx. The model was constrained where appropriate. For example, the twin correlations are constrained so that those of the randomly selected twin 1’s are the same as the randomly selected twin 2’s.
For the cross-twin within-trait correlations for diurnal preference at both time points, MZ correlations were significantly greater than the corresponding DZ correlations, suggesting the presence of genetic effects. For the cross-twin within-trait correlations for sleep quality, while the ratio of MZ:DZ twin correlations at time 1 were indicative of genetic effects, at time 2 twin correlations for MZ and DZ twins were similar, suggesting a greater influence of the environment on sleep quality at this time point. Of note, however, the sibling correlation was much smaller than the MZ twin correlation, which was more in line with expectations and highlighted possible genetic effects. Equating DZ twin and sibling correlations for sleep quality at time 2 did not significantly reduce model fit compared with the model in which MZ, DZ, and sibling correlations were free to differ (Δχ2 = 2.46, Δdf = 1, p = 0.12), indicating no significant differences in the correlations between DZ twins and siblings.
Bivariate Twin Correlations
As noted previously (Barclay et al., 2010a), the cross-twin cross-trait concurrent association between diurnal preference and sleep quality at time 1 appeared to be influenced by genetic factors (Table 2: MZ twin correlation was greater than DZ twin and sibling correlations). A similar pattern of twin correlations emerged at time 2. The stability over time in diurnal preference appeared to be influenced by genetic factors; inspection of the longitudinal cross-twin within-trait correlations showed that the MZ correlation was greater than DZ twin/sibling correlation. The longitudinal association between sleep quality at time 1 and 2, however, appeared to be influenced by both genetic and environmental (shared and nonshared) factors (MZ correlation > DZ twin correlation and similar to the sibling correlation). Equating DZ twin and sibling correlations for the association between sleep quality at time 1 and 2 did not significantly reduce model fit. For the cross-twin cross-trait associations longitudinally, genetic factors appeared to influence the association between diurnal preference at time 1 and sleep quality at time 2. Genetic factors also appeared to contribute to the association between sleep quality at time 1 and diurnal preference at time 2 (MZ correlation > DZ twin/sibling correlations).
Multivariate Correlated Factors Model
The full ACE model allowing sex differences was first run to estimate the influence of each source of variance (full scalar sex-limitation ACE model fit compared with the saturated model: Δχ2 = 335.48, Δdf = 246, p < 0.001, AIC = -156.52). Equating estimates for males and females (homogeneity model) resulted in a significant worsening of fit Δχ2 = 32.08, Δdf = 12, p < 0.001). However, a reduced AE homogeneity model (where C parameters were fixed at 0) did not fit significantly worse than the ACE homogeneity model Δχ2 = 1.86, Δdf = 10, p = 0.99, AIC = -18.14) nor the AE scalar sex-limitation model Δχ2 = 14.96, Δdf = 8, p = 0.06, AIC = -1.04). Thus, the AE homogeneity model was selected as the most parsimonious best-fitting model.
Univariate and Bivariate Estimates
The AE homogeneity multivariate correlated factors model (see Figure 1) estimates the relative contribution of genetic and environmental influences on the individual traits, and their associations, at each time point. Table 3 displays the univariate parameter estimates on the individual traits at each time point. Table 4 (top part) shows the genetic and environmental overlap between phenotypes concurrently and longitudinally. For example, there were moderate genetic correlations between diurnal preference and sleep quality concurrently at both time points (time 1: rA = 0.48 [95% CI = 0.31-0.65]; time 2: rA = 0.60 [95% CI = 0.34-0.94]). Longitudinally, the cross-trait genetic correlations were also significant, although smaller (rA’s = 0.29-0.40). The within-trait cross-time correlations demonstrated a substantial degree of stability in the genetic influences on diurnal preference and sleep quality individually, across time (rA’s: 0.90 [95% CI = 0.77-1.00] and 0.80 [95% CI = 0.57-1.00], respectively). Nonshared environmental overlap was substantially smaller for all associations (rE’s range from -0.02 to 0.41).
Univariate parameter estimates for diurnal preference and sleep quality (with 95% confidence intervals). a
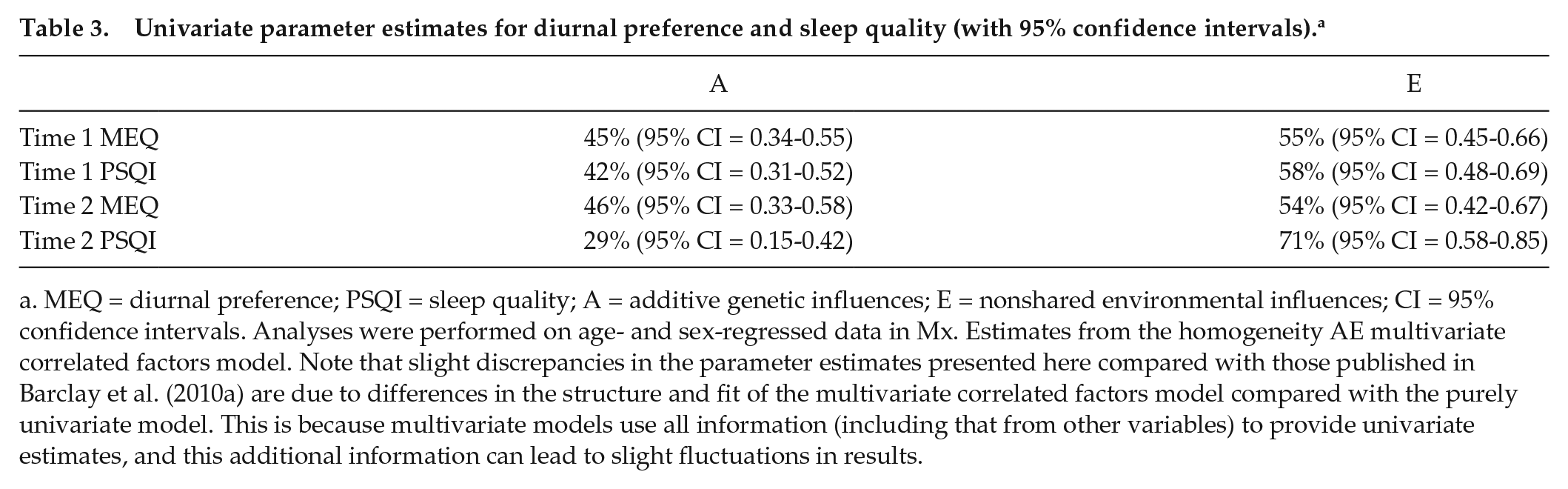
MEQ = diurnal preference; PSQI = sleep quality; A = additive genetic influences; E = nonshared environmental influences; CI = 95% confidence intervals. Analyses were performed on age- and sex-regressed data in Mx. Estimates from the homogeneity AE multivariate correlated factors model. Note that slight discrepancies in the parameter estimates presented here compared with those published in Barclay et al. (2010a) are due to differences in the structure and fit of the multivariate correlated factors model compared with the purely univariate model. This is because multivariate models use all information (including that from other variables) to provide univariate estimates, and this additional information can lead to slight fluctuations in results.
Parameter estimates (with 95% confidence intervals) from the homogeneity AE multivariate correlated factors model. a

MEQ = diurnal preference; PSQI = sleep quality; rA = additive genetic correlations; rE = nonshared environmental correlations; A = additive genetic influence; E = nonshared environmental influence. Analyses were performed on age- and sex-regressed data in Mx.
The lower part of Table 4 shows the proportions of the phenotypic associations accounted for by genetic and environmental influences. For example, the concurrent associations between diurnal preference and sleep quality at time 1 and time 2 were largely accounted for by genetic factors (A = 83%–1.05%). The longitudinal associations within and between phenotypes were also largely accounted for by genetic factors (60%–90%).
Discussion
Our results provide 5 main conclusions. First, both diurnal preference and sleep quality were relatively stable over time (i.e., both phenotypes exhibited moderate correlations across time). Second, at both time points, a preference for eveningness was associated with poorer sleep quality. Third, genetic influences contributed to both diurnal preference and sleep quality at both time points, although they were somewhat smaller for sleep quality at time 2 than time 1, with a compensatory increase in the importance of the nonshared environment. Fourth, there was moderate to substantial overlap in the genetic influences contributing to both diurnal preference and sleep quality concurrently and longitudinally. Fifth, concurrent and longitudinal associations between diurnal preference and sleep quality were largely accounted for by genetic effects, whereas the nonshared environment contributed to their differences. These results are discussed further below with reference to our initial aims set forth in our introduction.
Stability of Diurnal Preference and Sleep Quality over Time
The phenotypic correlations indicated substantial stability in diurnal preference from time 1 to time 2. This resonates with other studies that demonstrate temporal stability of diurnal preference using a range of self-report tools over short time periods (Boudebesse et al., 2013; Caci et al., 2000; Di Milia et al., 2004; Greenwood, 1994; Randler, 2009). Inspection of the means indicates a significantly greater tendency toward morningness from time 1 to time 2. Numerous studies demonstrate an age-related shift toward morningness across adulthood (Barclay et al., 2014; Carrier et al., 1997; Hur et al., 1998; Kramer et al., 1999; Paine et al., 2006; Taillard et al., 1999). Sleep quality also demonstrated some degree of longitudinal stability, although to a lesser extent—a finding that has also been observed in relation to the stability of sleep problems from early childhood through to adolescence (Gregory and O’Connor, 2002) and in relation to the stability of insomnia symptoms in adolescents over the course of 1 year (Roberts et al., 2008). Again, inspection of the means indicates that sleep quality improved, albeit marginally, across time. Although increasing age is typically associated with poorer subjective and objective sleep quality (for example, greater number of and duration of awakenings, decreased amount of SWS, and increases in lighter stage 1 and 2 sleep; Bliwise, 2005; Feinberg et al., 1967; Landolt et al., 1996; Van Cauter et al., 2000), our sample reflected young adulthood at both time points, and it is likely that declines in sleep quality are more relevant to different developmental periods (i.e., young, mid-, and late adulthood).
Associations between Diurnal Preference and Sleep Quality Concurrently and Longitudinally
As previously noted (Barclay et al., 2010a), at time 1 there was a significant association between diurnal preference and sleep quality, such that a greater tendency toward eveningness was associated with poorer sleep quality. This pattern was also demonstrated at time 2. We previously hypothesized that this association is explained by differences in the phase angle between intrinsic properties of the circadian system and wake time between morning and evening types. In essence, this means that evening types awaken closer to their body temperature nadir, when alertness is low (Barclay et al., 2010a). It is possible that low alertness upon awakening contributes to subjective feelings of having slept poorly. Our finding here of significant longitudinal associations, such that diurnal preference at time 1 predicted sleep quality at time 2 (and vice versa), suggests 2 additional possible mechanisms. The constraints of our social world impose restrictions on our sleep, forcing us to wake early to attend school/university or work. Given that evening types typically initiate sleep late, it is likely that their sleep duration is curtailed when social schedules are inflexible. Thus, a preference for eveningness at time 1 may contribute to poor sleep quality at time 2 from the accumulation of sleep debt over time. It would be beneficial for future research to address this proposition using longitudinal samples of diurnal preference and sleep quality/duration using the Munich Chronotype Questionnaire (Roenneberg et al., 2003), which accounts for differences in preference for daily activity on work and free days. Such examination would allow us to determine the extent to which the timing of sleep and its quality/duration is affected by social constraints. The converse association is a little harder to explain. Difficulty sleeping may lead one to delay bedtime in an attempt to avoid prolonged sleep-onset latency. Over time, this may result in a phase shift that delays the circadian rhythm, which is also reflected in (and maintained by) one’s subjective diurnal preference.
Stability of Genetic and Environmental Influences on Diurnal Preference and Sleep Quality, Within Phenotypes, over Time
The univariate estimates from the correlated factors model demonstrate that genetic influences accounted for about half the variability in both diurnal preference and sleep quality at time 1. These estimates have been presented previously (Barclay et al., 2010a) and are in line with estimates from numerous other studies of both phenotypes (Heath et al., 1990; Hur, 2007; Hur et al., 1998; Koskenvuo et al., 2007; Partinen et al., 1983; Vink et al., 2001). The longitudinal aspect of this study allowed us to identify the extent to which these effects contributed to stability in these phenotypes. We found that there was substantial genetic overlap for diurnal preference across time and sleep quality across time. This suggests that to a large extent, the same genes contribute to the stability of these phenotypes throughout young adulthood. A growing amount of evidence points to the importance of “clock” genes in predicting differences in the circadian rhythm as well as diurnal preference (e.g., Archer et al., 2003; Barclay et al., 2011; Carpen et al., 2005; Carpen et al., 2006; Ellis et al., 2009; Ferrante et al., 2015; Hu et al., 2016; Jones et al., 2007; Jones et al., 2016; Katzenberg et al., 1998), and particularly, those related to the serotonin system (among others) have been implicated in sleep quality, although inconsistently (Barclay et al., 2011; Barclay and Gregory, 2013; Brummett et al., 2007). Recent genome-wide association studies (GWAS) have pointed to many other genetic candidates implicated in these phenotypes (see Parsons, 2015, for a review). However, the fact that these genetic correlations were not absolute suggests that new genes may come into play (or that genes that previously were important do not continue to exert such a strong effect) during young adulthood to influence these phenotypes. Regarding the nonshared environment, overlap was substantially smaller for both phenotypes across time. Thus, while it is likely that there are some nonshared environmental factors that continue to contribute to diurnal preference over time, the majority are time specific. This is perhaps not surprising given that environmental factors that influence sleep are often likely to be transient. For example, living in a noisy household may hinder sleep, but this is unlikely to continue to affect sleep over time once the environmental factor has surpassed, which is likely in young adults who may move house or have a change in life circumstances during this time period.
Genetic and Environmental Overlap between Diurnal Preference and Sleep Quality Concurrently and Longitudinally
There was moderate to substantial overlap in the genetic influences contributing to diurnal preference and sleep quality concurrently and longitudinally, suggesting that to some extent, similar genes influence both phenotypes. These results fit with the “general genes hypothesis”—that the same genes often contribute to associated phenotypes (Eley, 1997; Lahey and Waldman, 2003; Plomin et al., 2016), highlighting that genes often have pleiotropic effects. Furthermore, genetic influences contributed to a substantial amount of the phenotypic correlations both concurrently and longitudinally. Nonshared environmental overlap between phenotypes concurrently and longitudinally was minimal if not nonexistent, highlighting that different environmental factors contribute to these phenotypes. Furthermore, environmental factors did not explain a substantial amount of the phenotypic correlations. Together, these results suggest that largely genetic factors are responsible for the associations between diurnal preference and sleep quality concurrently, and our longitudinal data point to a biological explanation of the influence of diurnal preference on later sleep quality and vice versa. In terms of our explanations for the phenotypic associations posed above, it is likely that genes indirectly contribute to the associations between these phenotypes by first influencing our behavior toward the timing of our sleep and our attributions of the sleep obtained. That said, recent data from a GWAS failed to find significant genetic overlap between a 1-item measure of diurnal preference and several sleep phenotypes including insomnia, sleep apnea, sleep need, sleeping soundly, and sweating during sleep (Hu et al., 2016). While these sleep phenotypes are likely to be associated with sleep quality, they are more specific in nature, which may explain the genetic heterogeneity for these variables and diurnal preference.
Limitations
Our findings should be considered in light of at least 4 limitations. First, our sample size was small, which reduces our power to decompose variance into genetic and environmental components. That said, twin studies focusing on sleep phenotypes (e.g., Sletten et al., 2013) and using analyses of this kind (e.g., Gregory, 2008) have used smaller samples than this one. As the first study of its kind to include measures of diurnal preference and sleep quality in a genetically informative longitudinal design, we provide a framework of initial findings that may be refined by more powerful future studies using similar designs.
Second, our short follow-up period (5 years) limits our ability to extrapolate our findings to age-related changes in the associations between diurnal preference and sleep quality over longer time frames. That said, our results provide important information about the mechanisms underlying the stability of these phenotypes within young adulthood. This is particularly important given that young adulthood is often a time of increased responsibility related to caring for and financially supporting a family, as well as developing a career, so disturbed sleep during this period may be particularly problematic and costly (Goldman-Mellor et al., 2014).
Third, unexpectedly, MZ twins exhibited a greater tendency toward eveningness than DZ twins and siblings. However, the effect sizes were small (Cohen’s d = 0.30 and 0.21 for differences between MZ vs. DZ, and MZ vs. siblings, respectively), and this finding was not replicated at time 2. In addition, responders at time 2 exhibited a greater tendency toward morningness than nonresponders. While the absolute difference in means between zygosity groups and responders versus nonresponders is worthy of consideration in future studies, this does not preclude the computation of the relative proportions of variance accounted for by genetic and environmental factors.
Fourth concerns the interdependency of the phenotypes under study. Some of the components of the PSQI, for example, sleep duration, are likely to be influenced by the combination of diurnal preference and social schedules, which together determine the circadian phase of sleep; the MEQ itself contains some items that pertain to sleep quality (i.e., “during the first half hour after having woken in the morning, how tired do you feel?”). This brings into question the extent to which diurnal preference and sleep quality can be regarded as separate phenotypes. It is possible that this interdependency explains the apparent genetic overlap observed. That said, the apparent moderate correlations, both concurrently and longitudinally, between these phenotypes argues against this point. However, the heterogeneity in the study population in both the timing and flexibility of their social schedules possibly obscures a greater association between these phenotypes that would be detectable only if social schedules were fixed.
It is further important to consider the more general limitations that are inherent in twin modeling (e.g., the equal-environments assumption, generalizability, assortative mating, zygosity determination; see Plomin et al., 2013), yet the importance of investigating etiological mechanisms using twin modeling far outweighs these limitations.
Implications
These findings show that, over time, a combination of both overlapping, and to a lesser extent different, genes appear to be important in maintaining diurnal preference and sleep quality over time. Thus, the genetic mechanisms underlying these phenotypes appear to change to some extent across this relatively short time frame. In relation to diurnal preference, the introduction of new genetic effects (or indeed the discontinuation of effects that were previously important) may reflect the circadian stabilization that occurs after the reorganization of the circadian system during adolescence (Hagenauer et al., 2009). Given that the circadian system continues to change across the life span, it is perhaps no surprise that different genes may come into play at particular developmental time points. Not only does the circadian system undergo vast changes during adolescence but so too does the homeostatic system. For example, it has repeatedly been demonstrated that SWS (a marker of homeostatic sleep pressure) declines across adolescence (Carskadon et al., 2004). It is likely that this change is at least in part genetically controlled, and so a specific set of genes may be implicated. By young adulthood, this dramatic decline in SWS may plateau, and again, different sets of genes may come into play to maintain sleep throughout this period. Not only do the present results support these explanations, but they also suggest that similar genes may be controlling the reorganization and relative stabilization of both the circadian and homeostatic systems beyond the transition from late adolescence to young adulthood.
The present findings also increase our theoretical understanding of the potential mechanisms underlying the association between a preference for eveningness and poor sleep quality. These associations were largely due to genetic factors, and a combination of similar and different genes can be implicated. While molecular genetic studies are making some headway in determining the specific genes underlying both diurnal preference and sleep quality, results are inconsistent (Barclay and Gregory, 2013; Gehrman et al., 2011; Parsons, 2015; Parsons et al., 2014). Discrepancies between studies may be due to the etiological factors contributing to these phenotypes changing over time. Demonstrating that the stability of these phenotypes over time is partially due to unique genetic and environmental influences has the potential to direct future molecular genetic studies by considering the possibility that some sources of influence may be time specific.
Footnotes
Declaration of Conflicting Interests
The authors declare no conflicts of interests.
Funding
Waves 1 to 3 of the G1219 study were supported by grants from the W T Grant Foundation, the University of London Central Research fund, and a Medical Research Council Training Fellowship and Career Development Award to Thalia C. Eley. Wave 4 of the G1219 study was supported by grants from the Economic and Social Research Council (RES-000-22-2206) and the Institute of Social Psychiatry (06/07–11) to Alice M. Gregory. Nicola L. Barclay was supported by an ESRC studentship. Wave 5 was supported by funds from Goldsmiths University of London to Alice M. Gregory.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
