Abstract
Twin studies have highlighted that a large proportion of variability in chronotype is accounted for by individual-specific environmental factors (non-shared environmental influences). However, little research has aimed to identify specific non-shared environmental influences on chronotype. Although epidemiological studies have shed light on possible environmental influences on chronotype, a substantial amount of research has highlighted the importance of genetic influences on exposure toward specific environments, a process termed gene-environment correlation. It is possible that associations between the environment and chronotype are in part determined by genetics, rather than being purely environmental in origin. One way of exploring the contribution of purely non-shared environmental components on associations between chronotype and the environment is to use the monozygotic twin differences design. This design allows us to tease apart the influences of genetics and the environment to identify purely environmental components. One hundred eighty-nine monozygotic twin pairs (mean age 19.81 years, SD = 1.26, range = 18-22 years, 66.1% female) completed the Horne and Östberg Morningness-Eveningness Questionnaire as a measure of chronotype and questionnaires assessing the following candidate non-shared environmental influences: dependent and independent negative life events, educational attainment, employment status, relationship status, deviant peers, affiliation with deviant peers, general health, smoking, drug use, and alcohol use. Linear regression analyses indicated the presence of gene-environment correlation for the majority of associations between chronotype and candidate environmental influences. When controlling for genetic and shared environmental effects, within monozygotic twin-pair differences in chronotype were associated with within monozygotic twin-pair differences in dependent negative life events (β = −0.27, p < 0.001), educational attainment (β = −0.14, p < 0.05), smoking status (β = 0.22, p < 0.01), and drug use (β = −0.16, p < 0.01). These results suggest that some of the association between these variables is purely environmental in nature. The associations between the remaining environmental variables and chronotype, however, may be intertwined with underlying genetic factors. These findings add to our understanding of genetic and environmental mechanisms underlying the biological clock.
Keywords
The circadian phase and the phase angle between sleep and the light-dark cycle vary greatly between individuals (Kerkhof, 1985). This interindividual variability in circadian rhythmicity is likely to be influenced by a host of genetic and environmental influences. Twin studies have shown that biological markers of circadian rhythmicity are heritable (e.g., Linkowski et al., 1993), and an indirect measure of circadian rhythmicity from self-report, chronotype (also commonly termed diurnal preference), is about 50% heritable (see Barclay and Gregory, 2012, for a review). Chronotype refers to an individual’s tendency toward morningness or eveningness. Morning types, so-called “larks,” arise from bed early, function best in the morning, and tend to go to bed relatively early in the evening. On the other hand, evening types, so-called “owls,” find it difficult to get up early, function best in the afternoon, and go to bed late at night, often past midnight (Kleitman, 1963). A substantial body of research has focused on correlates of chronotype, finding that eveningness is associated with poor sleep quality (Barclay et al., 2010a; Tzischinsky and Shochat, 2011), irregular sleep-wake habits (Taillard et al., 1999), greater sleep debt (Taillard et al., 1999; Roenneberg et al., 2003; Taillard et al., 2004), self-reported morbidity (Taillard et al., 2001; Paine et al., 2006), mood disorders (Taillard et al., 2001; Tzischinsky and Shochat, 2011), heightened cardiovascular responses to stress (Willis et al., 2005), and several personality dimensions including novelty seeking, harm avoidance, and neuroticism (Tonetti et al., 2009; Adan et al., 2010).
While studies point to the contribution of genetic factors in the control of circadian rhythmicity/chronotype, they also point to the reality that environmental influences are equally as important. Indeed, it is well established that light is an influential zeitgeber (from the German “time-giver”) of the biological clock (Duffy and Wright, 2005), yet it is likely that individual specific influences are also associated with one’s preference for sleep-wake activity. As well as understanding genetic and environmental influences on chronotype independently, it is becoming increasingly evident that we should consider the interplay between these factors. For example, research has highlighted that genetic propensities may to some extent shape our environment, a process termed gene-environment correlation (Kendler and Baker, 2007). It is possible that environmental influences associated with chronotype are in part determined by genetics, and thus associations between chronotype and the environment may be accounted for by gene-environment correlation. In other words, the presence of gene-environment correlation would suggest that the genes influencing chronotype are shared with those influencing a common environmental factor. Thus, identifying specific environmental factors associated with chronotype, as well as disentangling the mechanisms underlying these associations, will further increase our understanding of the factors contributing to the organization of the circadian system.
Twin studies provide us with some indication of the types of environmental factors influencing a trait by partitioning variance into environmental influences that are shared (those that act so as to make individuals within a family more alike) and non-shared (those that act to make individuals within a family different). It is typically environmental influences of the non-shared kind that are important for chronotype, with the shared environment contributing very little, if anything, to this trait in adults (see Barclay and Gregory, 2012, for a review). This is a common finding in behavioral genetic work focusing on other phenotypes in adulthood and is generally considered to occur as individuals tend to seek out novel environments as they move away from the family environment (Plomin et al., 2008). However, little is known about specific non-shared environmental influences on chronotype.
One way of exploring the contribution of purely non-shared environmental components (while accounting for gene-environment correlation) on the associations between chronotype and the environment is to use the monozygotic (MZ) twin differences design. MZ twins are considered to be genetically identical, and so any differences between them in a trait of interest must be due to differences in their non-shared environment (as well as possible measurement error; see Plomin et al., 2008, for more information on the twin design). Accordingly, the MZ differences design assesses the degree of dissimilarity between MZ twins within a pair on a trait of interest and a given set of candidate environmental measures. Thus, this design allows us to tease apart the influences of genetics and the environment to identify (1) the presence of gene-environment correlation and (2) purely environmental components.
The present study explores the contribution of several candidate non-shared environmental influences on chronotype using the MZ twin differences design. We assessed dependent and independent negative life events (the former of which are those that are in part dependent on an individual’s behavior, whereas the latter are those that are beyond an individual’s control), educational attainment, employment status, relationship status, peer influences (i.e., deviant peers and affiliation with deviant peers), general health, smoking, and drug and alcohol use. These were selected either because they have previously been shown to be associated with chronotype (e.g., Randler, 2008; Beşoluk et al., 2011; Prat and Adan, 2011; Preckel et al., 2012) or are known to be associated with sleep disturbances (e.g., see Ohayon, 2002; Barclay et al., 2012), which itself is correlated with chronotype (Barclay et al., 2010a). Accordingly, it is of interest to establish whether there are associations between these environmental factors and chronotype independent of sleep quality. Furthermore, it is important to determine whether these associations are in part dependent on gene-environment correlation or are purely environmental in origin. Examination of this within the MZ twin differences design will enable us to understand more about the mechanisms underlying such associations.
Materials and Methods
Sample
Data from all complete MZ twin pairs (n = 189 complete pairs provided data on chronotype) from wave 4 of the G1219Twins study were used in analyses (mean age = 19.8 years [SD = 1.26], range = 18-22; 66.1% female). G1219 initially comprised adolescent offspring of adults from a large-scale population-based study (GENESiS; Sham et al., 2000). The G1219Twins are a random selection of live twin births born between 1985 and 1988 identified by the UK Office of National Statistics. At wave 1 of data collection (which took place between 1999 and 2002), 3640 respondents aged between 12 and 19 years participated in the study. Informed consent was obtained from parents/guardians of all adolescents younger than 16 years and from the adolescents themselves when 16 years and older. Ethical approval for different stages of this study has been provided by the Research Ethics Committees of the Institute of Psychiatry, South London and Maudsley NHS Trust, and Goldsmiths, University of London. At wave 4 (which took place in 2007 and is the focus of this current report as it is the first wave to include data on sleep), we traced participants who had taken part in wave 2/wave 3 and sent them a questionnaire booklet. A total of 1556 individuals were included in the wave 4 data set (61% of those targeted; 74% of those participating at wave 3). Zygosity was established through a questionnaire measure completed by mothers at waves 2 and 3, assessing physical similarity between twins (Cohen et al., 1975). Questionnaire methods have been shown to yield ~95% accuracy in correctly determining zygosity when validated against DNA markers (Price et al., 2000). All participants were living in the United Kingdom at the time of data collection.
Measures
Chronotype
Chronotype was assessed using the Morningness-Eveningness Questionnaire (MEQ: Horne and Östberg, 1976). The MEQ is among the most widely used measures for assessing chronotype and was adopted for use in this study. The MEQ is a 19-item self-report questionnaire that assesses individual preference in the timing of daytime activities, sleeping habits, hours of peak performance, and times of “feeling best” and maximum alertness. Responses are used to give a total score on the morningness-eveningness dimension, ranging from 16 to 86. Higher scores indicate greater “morningness,” and lower scores indicate greater “eveningness.” Scores can be used to categorize individuals as “definitely evening-type” (scores 16-30), “moderately evening-type” (scores 31-41), “neither type” (scores 42-58), “moderately morning-type” (scores 59-69), and “definitely morning-type” (scores 70-86; see Horne & Östberg, 1976).
Sleep quality
To control for sleep quality in our analyses, the Pittsburgh Sleep Quality Index (Buysse et al., 1989) was used to assess sleep quality in the past month. Further information on the use of this scale in the present sample can be found elsewhere (e.g., Barclay et al., 2010a; Barclay et al., 2012).
Dependent and independent negative life events
Negative life events were assessed using items from the List of Threatening Experiences (Brugha et al., 1985) and the Coddington Stressful Life Events Scale (Coddington, 1984). Participants are required to respond to these checklists by indicating whether or not they have experienced a particular negative life event in the past year. Twenty-one items were summed to give a score of total life events, which were further subdivided into 13 items assessing dependent negative life events (e.g., separation due to marital difficulties) and 8 items assessing independent negative life events (e.g., death of a parent). Dependent and independent negative life events were classified according to whether it is likely that their occurrence is the consequence of an individual’s behavior (Brown and Harris, 1978). This distinction between life events has been used in previous studies (Silberg et al., 2001), as well as other papers from the G1219 study (Barclay et al., 2011a). Higher scores on both of these scales indicate a greater number of negative life events experienced.
Education
Educational achievement was assessed by one question asking participants their highest UK qualification (ranging from high school to university-level qualifications). Higher scores indicated a higher level of education attained.
Employment
Participants were initially asked to indicate their employment status according to the following categories: unemployed, full-time student, employed full-time, part-time student/work, on a study break, on government benefit, or full-time parent. Participants were finally categorized as “employed full-time” versus “full-time student” as the number of cases in the other categories were considered too small and so were not included in analyses.
Relationship status
Participants were initially asked to indicate their relationship status according to one of the following categories: married, living with partner, engaged, living with partner and engaged, going steady, casual or single. As a large proportion of our sample fell into either the going steady (33.2%) or single (48.2%) category, relationship status was finally categorized as those who were “single” versus “those in a relationship” (49.7%) at the time of assessment.
Deviant peers/affiliation with deviant peers
The presence of deviant peers was assessed by asking participants how many of their friends engaged in a number of deviant behaviors, including alcohol, tobacco, or cannabis use and whether they truanted or broke the law, as outlined by Fergusson and colleagues (2003). Responses were categorized as “none,” “one or two,” “some,” and “most/all.” Deviant peer affiliation was assessed by 3 items asking, “How likely is it that you would still stay with your friends if they were getting you in trouble (i) with your family; (ii) at work/college; (iii) with the police?” with responses ranging from “not at all likely,” “not very likely,” “fairly likely,” and “very likely” as outlined in the Edinburgh Study of Youth Transitions and Crime (Smith and McVie, 2003). Higher scores on both measures indicate greater deviance.
General health
General health was assessed by 1 item assessing subjective general health using the question, “In general, how good would you say your health is now?” with responses on a 5-point scale ranging from 1 = excellent to 5 = poor, as outlined by Ware and Sherbourne (1992). General health has reliably been measured in this way in numerous studies (e.g., see Troxel et al., 2009).
Smoking
Smoking status was assessed by asking participants whether they smoke, with responses categorized as “yes,” “no,” or “used to.” Participants responding “yes” to this question were then required to indicate how many cigarettes they smoke per day by responding to the statement, “On the days that I smoke, I smoke…” by checking one of the following responses: “1 to 5 cigarettes,” “6 to 10 cigarettes,” “11 to 15 cigarettes,” “16 to 20 cigarettes,” or “more than 20 cigarettes.”
Drug use
Drug use was assessed by responses to the statement, “I use drugs (other than alcohol and nicotine) for non-medical purposes.” Participants were required to check one of the following responses: “not true,” “somewhat true,” “very true.”
Alcohol use
Alcohol use was assessed by 1 question asking whether participants drink alcohol, with responses categorized as “yes” or “no” (variable labeled “alcohol use”). Participants indicating that they do drink were then required to respond to the following question: “When you have an alcoholic drink, how many drinks do you have?” by checking one of the following responses ranging from 0 to 8 or more (1 drink was indicated to include the following: half pint of beer/lager, 1 glass of wine, 1 glass of spirits, or an alcopop, e.g., Bacardi Breezer, Smirnoff Ice; variable labeled “number of alcoholic drinks”).
Analyses
First, a series of univariate linear regression analyses were run in STATA (Stata, 2002) to assess the contribution of each of the absolute environmental measures on absolute chronotype scores. Second, a series of univariate linear regression analyses were run using MZ twin difference scores for each of the environmental measures to predict MZ twin differences in chronotype. The MZ twin difference scores were calculated by subtracting twin 1’s score from that of twin 2’s (note that in each MZ twin pair, one twin was randomly assigned as “twin 1” and the co-twin as “twin 2”). Because for these analyses we assume that monozygotic twins share 100% of their genetic material as well as 100% of their shared environment, any differences between them must be accounted for by the non-shared environment (including measurement error). Thus, the MZ twin difference measure provides an unbiased estimate of twin discrepancy due to the non-shared environment (including measurement error; Rovine, 1994). The MZ differences analyses, using a measure of twin discrepancy, thus control for the effects of genetics and the shared environment (as for the purposes of this design, it is assumed that MZ twins do not differ because of genetic or shared environmental reasons). All regression analyses controlled for the effects of age, sex, and sleep quality, given their associations with chronotype. All regression analyses took into account clustering within the sample due to the inclusion of MZ twins, and thus non-independence of observations, by using the Robust(cluster) command in STATA.
Results
The mean chronotype score was 50.23 (SD = 7.74), with scores ranging from 24 to 74. The frequency distribution of total chronotype scores for the total sample is presented in Figure 1. There were significant sex differences in chronotype, t(376) = −4.18, p < 0.01. Males scored significantly lower (M = 47.96), indicating a greater preference for eveningness, than females (M = 51.40). The frequency distribution of the MZ twin difference scores for chronotype is shown in Figure 2. The mean of the MZ difference scores generally approximates zero since the random assignment of twins ensures that cases where twin 1 scores higher than twin 2 are canceled out by cases in which twin 2 scores higher than twin 1. What is important is the distribution of these scores. Clustering around zero demonstrates similarity between twins, whereas deviations from zero demonstrate the presence of differential chronotype scores due to the non-shared environment (Liang and Eley, 2005). In terms of MEQ categories (see Horne and Östberg, 1976), difference scores >±1 to ±10 equate to a maximum difference between twins within a pair of definite versus moderate (i.e., one twin may be a definite morning type, whereas their co-twin is a moderate morning type) or moderate versus neither type chronotypes. Difference scores greater than ±10 to ±20 equate to a maximum within twin pair difference between definite versus neither or moderate morning versus moderate evening type. Difference scores greater than 40 would differentiate between definite morning versus definite evening types. The frequency distribution of difference scores demonstrates that although the majority of the twins score within 10 points of their co-twin, there are cases in which differences are even larger, such that one twin may be more morning oriented compared with their co-twin, who is more evening oriented.
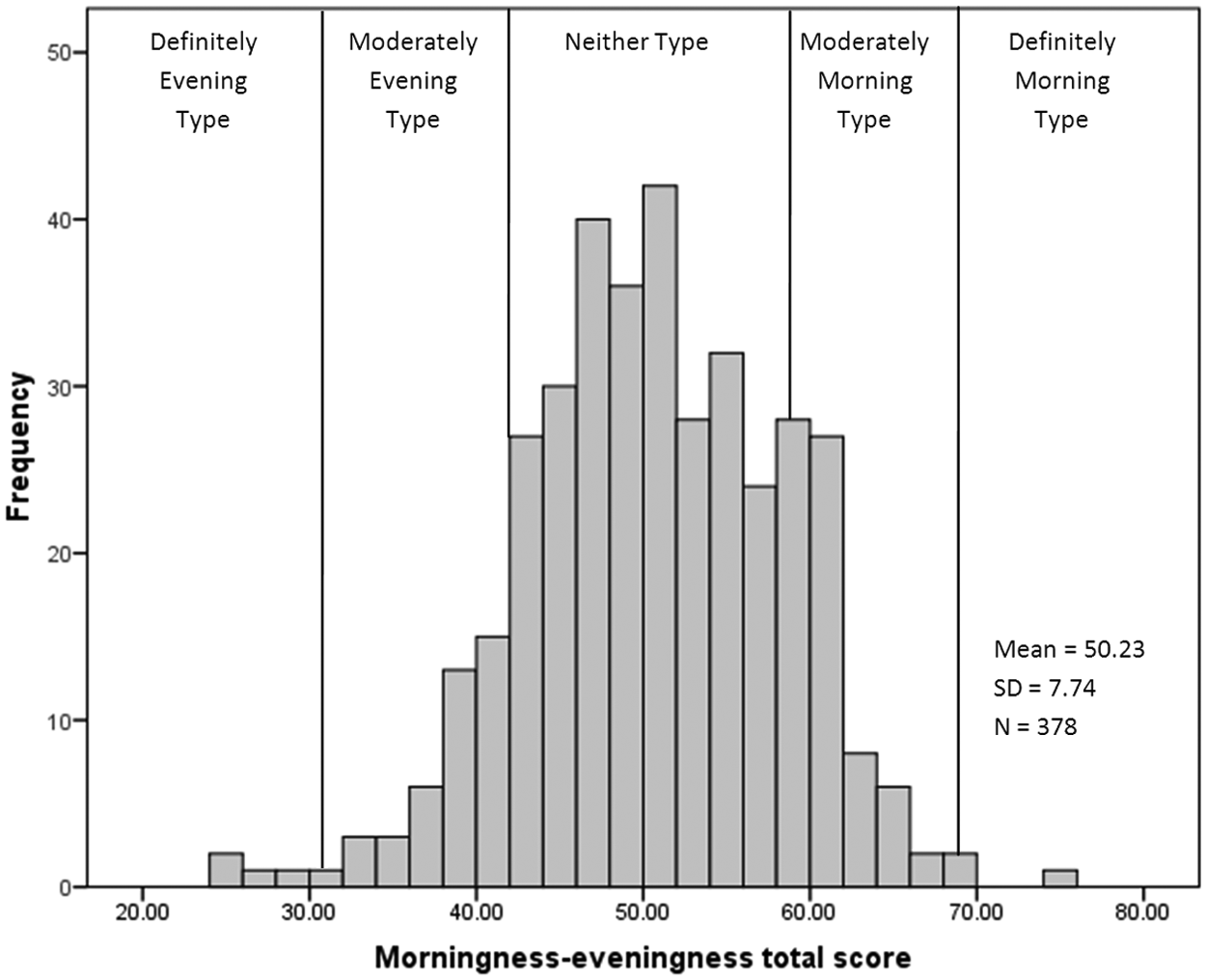
Distribution of chronotype scores in the total MZ twin sample from the morningness-eveningness questionnaire. Scores ranged from 24 to 74. Higher scores indicate greater morningness. Vertical lines separate the chronotype categories as follows: “definitely evening type” (scores 16-30), “moderately evening type” (scores 31-41), “neither type” (scores 42-58), “moderately morning type (scores 59-69), and “definitely morning type” (scores 70-86). See Horne and Östberg (1976).
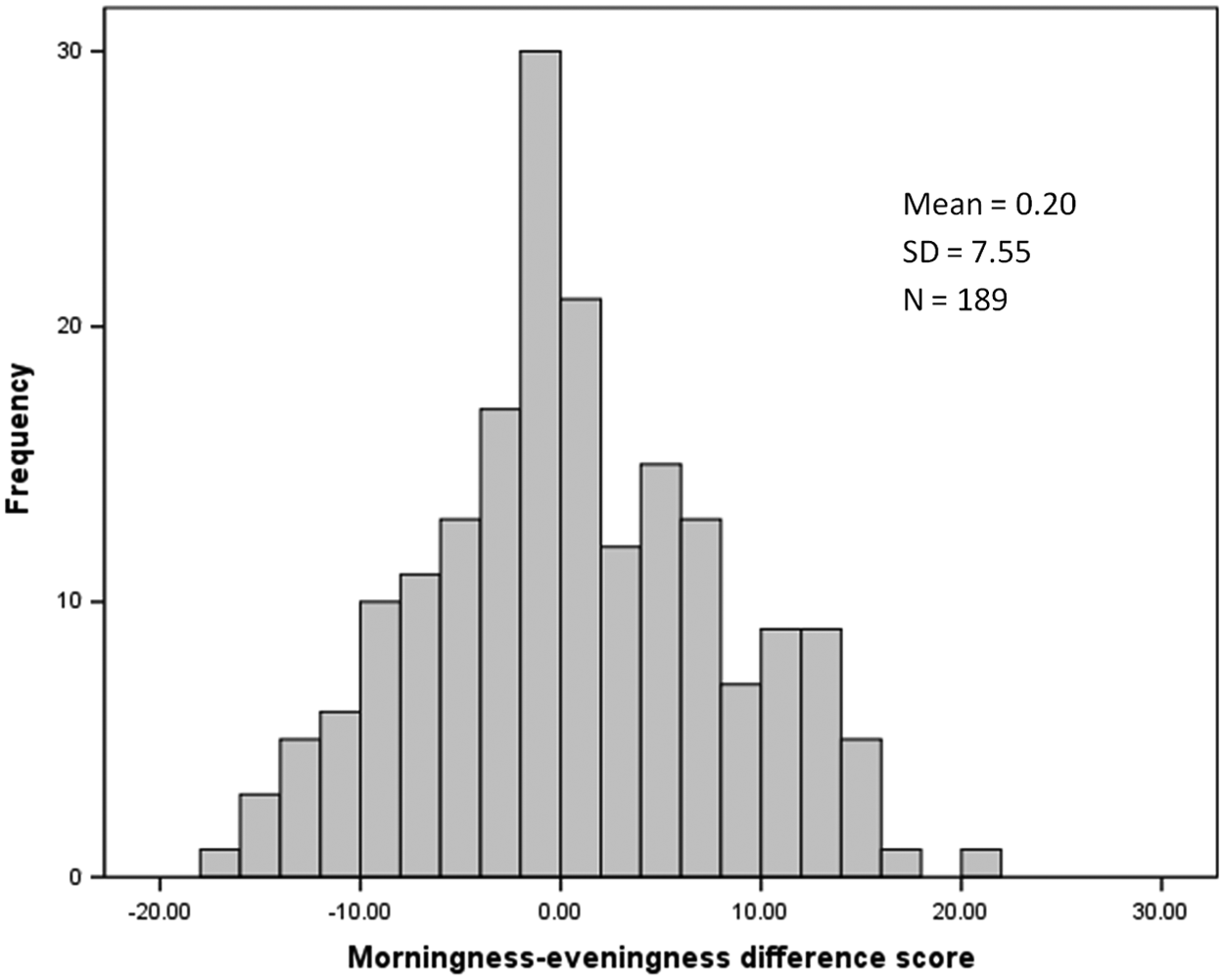
Distribution of within MZ twin pair difference scores for chronotype. Scores of zero indicate similarity of chronotype scores within twin pairs. Deviations from zero indicate dissimilarity between twins, suggesting the influence of the non-shared environment (or measurement error).
The results of the multiple univariate linear regression analyses are shown in Table 1. Column a shows the results on absolute chronotype scores. There were significant associations between a preference for eveningness and dependent negative life events, poorer educational attainment, the presence of deviant peers, poorer general health, smoking status, drug and alcohol use, and number of alcoholic drinks typically consumed when drinking. Column b shows the results of the MZ differences analyses, which control for the influence of both genetic and shared environmental effects, using twin difference scores on the environmental measures to correlate with twin differences in chronotype. In these analyses, the majority of the environmental influences were reduced to nonsignificance, indicating that their associations with chronotype are largely mediated by gene-environment correlations. However, MZ differences in dependent negative life events, educational attainment, smoking status, and drug use remained significantly associated with chronotype in comparison with the absolute measures analyses. These results indicate a purely non-shared environmental component to these associations independent of genetic/shared environmental factors. Of note, after applying a Bonferroni correction to account for multiple testing, only the association between dependent negative life events and chronotype in the MZ differences analysis remained significant.
Univariate Linear Regression Analyses Predicting (a) Absolute Chronotype Scores and (b) Monozygotic Difference Scores to Control for Genetic Vulnerability, from the Environmental Measures
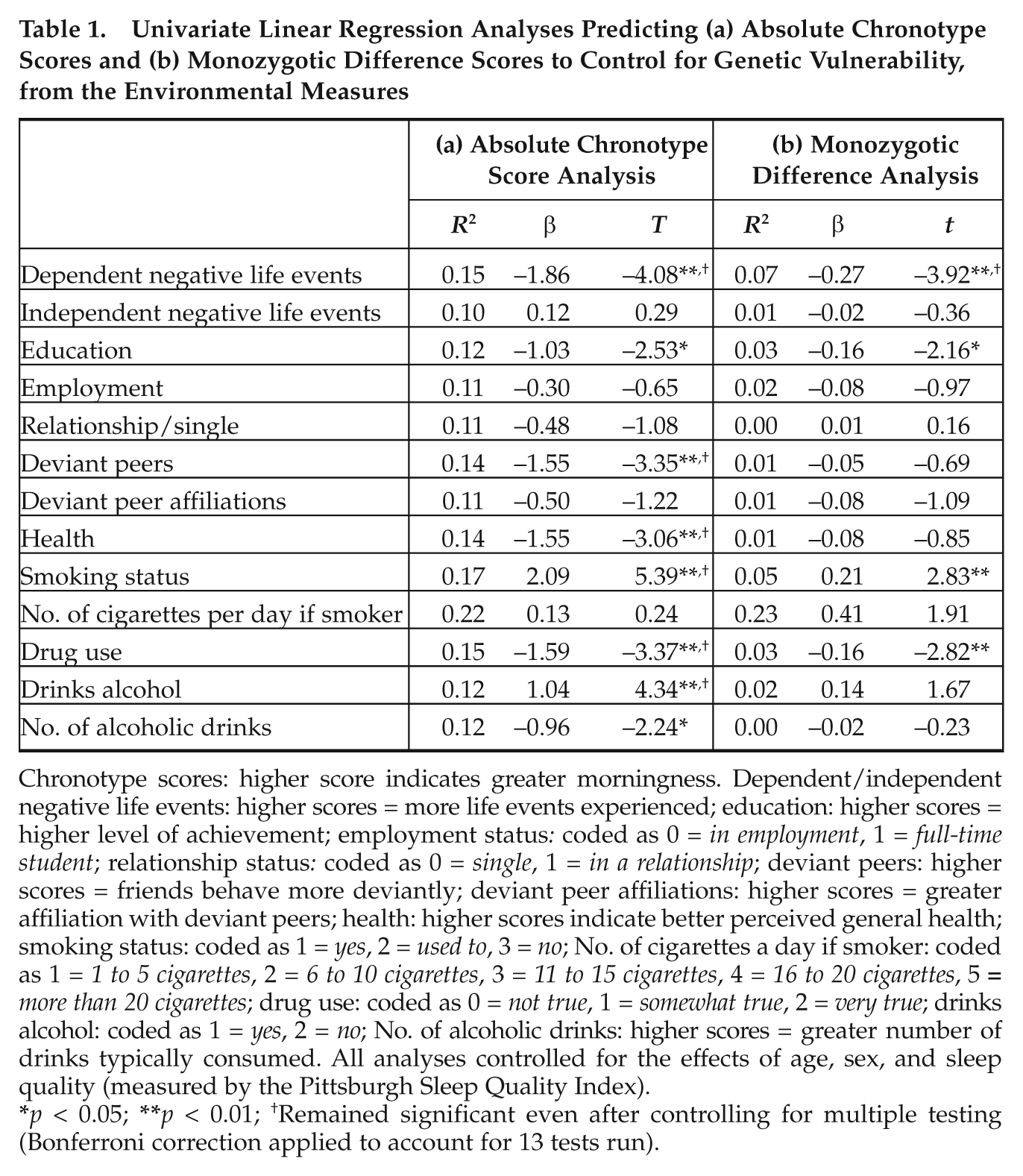
Chronotype scores: higher score indicates greater morningness. Dependent/independent negative life events: higher scores = more life events experienced; education: higher scores = higher level of achievement; employment status: coded as 0 = in employment, 1 = full-time student; relationship status: coded as 0 = single, 1 = in a relationship; deviant peers: higher scores = friends behave more deviantly; deviant peer affiliations: higher scores = greater affiliation with deviant peers; health: higher scores indicate better perceived general health; smoking status: coded as 1 = yes, 2 = used to, 3 = no; No. of cigarettes a day if smoker: coded as 1 = 1 to 5 cigarettes, 2 = 6 to 10 cigarettes, 3 = 11 to 15 cigarettes, 4 = 16 to 20 cigarettes, 5
p < 0.05; **p < 0.01; †Remained significant even after controlling for multiple testing (Bonferroni correction applied to account for 13 tests run).
Discussion
This study reveals 4 main results worthy of discussion. First, the distribution of chronotype scores showed wide variation between individuals but with clustering around neither type. This distribution is in line with results of other studies of similarly aged individuals (Baehr et al., 2000). Inspection of the distribution of chronotype difference scores indicated that the majority of twins within a pair exhibited similar chronotypes. Similarity between twins on chronotype implies the possible importance of genetic factors contributing to the phenotype (which is especially likely to be the case as previous twin research supports this and suggests a negligible role for shared environment; see Barclay and Gregory, 2012, for a review). However, there was notable within-pair variation in chronotype scores to the extent that some pairs exhibited differences greater than 10 points. Scores greater than 10 equate to possible differences in chronotype such that one twin could be moderate morning type and their co-twin moderate evening type. As MZ twins are genetically identical, any variation in chronotype must be accounted for by environmental factors. Second, there were significant associations between chronotype and a number of environmental influences assessed. This study draws on data from a large-scale, nationally representative sample, allowing the investigation of a range of candidate environmental influences. Third, this study demonstrates that, to some extent, these associations are largely mediated by processes of gene-environment correlation. Fourth, the associations between chronotype, negative life events, educational attainment, smoking, and drug use have a purely non-shared environmental component independent of genetics (and the shared environment). We can be certain that the shared environment does not contribute to these associations, as previous studies using the twin design (including our own using the present sample) have consistently found that the shared environment accounts for little, if any, variance in chronotype (see Barclay and Gregory, 2012, for a review). These findings add to our understanding of the mechanisms underlying these associations and chronotype more generally.
Associations between Chronotype and the Candidate Environmental Influences
The linear regression analyses using absolute measures revealed several noteworthy findings. First, a tendency toward eveningness was associated with a greater number of dependent negative life events experienced in the past year. Although it seems unlikely that an evening-type personality would influence the occurrence of negative life events, it is possible that this association is mediated by other personality characteristics that are known to be associated with chronotype and greater susceptibility to negative experiences. For example, greater neuroticism, risk-taking behaviors, and externalizing behaviors are associated with eveningness (Killgore, 2007; Barclay et al., 2010b) and may be associated with relationship difficulties (which may be encompassed by several of the items included in the life events scales included here). Furthermore, it is possible that the association between negative life events and chronotype is in part mediated by poor sleep quality, given that previous research including our own has suggested associations between poor sleep quality and a preference for eveningness (Megdal and Schernhammer, 2007; Barclay et al., 2010a; Tzischinsky and Shochat, 2011) and between poor sleep quality and negative life events (Healey et al., 1981; Vahtera et al., 2007; Barclay et al., 2011a). Accordingly, it is possible that poor sleep quality interferes with decision making to the extent that negative life events are more likely to occur. However, the present analyses controlled for sleep quality, so it is unlikely to underlie the association found here. Second, there was a significant association between a preference for eveningness and lower educational attainment. This is consistent with previous research, which has demonstrated associations between eveningness and poor educational outcomes in adolescents and university students (Beşoluk et al., 2011; Preckel et al., 2012). Third, the presence of deviant peers, smoking, using recreational drugs, and drinking alcohol were all associated with a preference for eveningness. This is perhaps no surprise given that chronotype has previously been associated with health-impairing behaviors, in particular alcohol, smoking, and drug use (e.g., Adan, 1994; Randler, 2008; Urbán et al., 2011). The involvement in negative lifestyle behaviors such as these (and the associated poorer general health) is colloquially associated with evening activities, such as socializing late at night. Of note, it is interesting that smoking status, but not the number of cigarettes one typically smokes, was associated with chronotype (supporting previous work, Wittmann et al., 2006). This suggests that chronotype is associated with discrete differences in smoking behavior rather than a continuum. Although this adds to the current body of literature on these associations, it should be noted that the number of individuals who smoked and thus responded to the question tapping into the number of cigarettes smoked per day was small (n = 41), and so a lack of association may have occurred due to reduced power.
While we find significant associations between chronotype and a range of candidate environmental variables, whether these associations are environmentally mediated or whether they are influenced by internal factors (i.e., personality/genetics) has not previously been explored and is discussed below.
Gene-Environment Correlation
In all significant associations between chronotype and the “environmental” variables, there was evidence of gene-environment correlation. This is evidenced by the nonsignificant beta weights in the MZ difference analyses compared with those for the corresponding absolute measures (for all but four environmental variables, for which the beta weights were considerably smaller in the MZ differences analysis, suggesting some, but not exclusive, influence of gene-environment correlation). Thus, overlapping genetic pathways may contribute to these associations, that is, that genes influencing chronotype are to some extent shared with those influencing the environmental measures with which it is associated. An alternative explanation is that peer groups may be an important mediator of the links between chronotype and the environment, and it is possible that choice of peers is influenced by underlying personality characteristics/genetics. Several studies have highlighted the importance of genetic influences on several aspects of peer interactions (Manke et al., 1995; Iervolino et al., 2002) and affiliation with deviant peers (Gillespie et al., 2009). In the present study, however, there was no association between chronotype and affiliation with deviant peers but simply with the presence of deviant peers. Thus, it appears that although an individual may socialize with deviant peers in the evening, and indeed involve themselves in the negative lifestyle behaviors associated with eveningness, the extent to which an individual may feel inclined to stay connected to deviant peers has a component not mediated by the peer socialization environment, suggesting the importance of personality characteristics/genetics.
Specific Non-shared Environmental Influences
Dependent negative life events
The finding that MZ twin differences in dependent negative life events were associated with MZ twin differences in chronotype (and indeed this was the only association to survive the correction for multiple testing) suggests that this association is influenced by a purely environmental component that is independent of genetics (in addition to processes of gene-environment correlation). This finding makes likely the explanation that external factors may be influential in this respect. While this does not rule out the possibility that genetic factors contribute to the occurrence of dependent negative life events, it suggests that the experience of negative life events is not purely a function of an individual’s behavior but also likely to be a function of the external environment. This is interesting as our previous set of analyses focusing on sleep quality (see Barclay et al., 2012) found no purely non-shared environmental component to the association between sleep quality and dependent negative life events but rather that the association was accounted for by overlapping genetic factors. Accordingly, the results of the present study are compatible with the possibility that evening types may be more likely to be exposed to negative events that are beyond their control (although, of course, the cross-sectional nature of the study limits our ability to determine the order of effects).
Educational attainment
The association between a preference for eveningness and poorer educational attainment exhibited a purely non-shared environmental component. This finding suggests that the association is not simply a function of an individual’s capacity for achievement but that wider environmental circumstances play an important role. Indeed, it is reasonable to postulate that school/university start times could influence differential academic performance between those with different chronotypes. For example, early school start times may be particularly unfavorable for evening types given that they typically sleep less on weeknights compared with morning types and suffer greater daytime consequences (Carskadon et al., 1993; Giannotti et al., 2002).
Smoking and drug use
The purely non-shared environmental components to the associations between eveningness and smoking status and drug use suggest an influence of social experiences/lifestyle choices (i.e., one may socialize more in the evening and thus smoke/use drugs due to environmental pressures from peers), as opposed to the associations being maintained exclusively by underlying genetic factors. However, as with all of the variables demonstrating purely non-shared environmental components, processes of gene-environment correlation also contribute largely to these associations, as evidenced by the decrease in beta weights for the MZ differences analyses. Thus, the genes contributing to chronotype may be shared with those influencing the environmental exposures. In the context of smoking and drug use, this suggests possible overlapping molecular pathways involved in addiction and the circadian clock. Indeed, there is recent evidence identifying the circadian gene PER2 as a biological marker of addiction (Shumay et al., 2012). Thus, the search for molecular genetic polymorphisms involved in both phenotypes should center on common neurobiological systems.
Limitations
Of course, there are likely to be many other environmental influences that contribute to the variability in chronotype, and the present study is limited to those that we had measured. Perhaps the most important environmental factor that is likely to contribute to chronotype is light (Roenneberg et al., 2003). It may be considered that light would act as a shared environmental factor between twins, given that the light-dark cycle is identical for individuals living at the same latitude (it is likely that twins were living at similar latitudes given that the present sample were all living in the United Kingdom). However, it is more likely that its influence acts in a non-shared manner, as twins are unlikely to expose themselves to exactly the same amount of light in a given day, especially in adulthood, when twins may be living more independent lives. Unfortunately, we did not obtain information regarding the extent to which twins within a pair followed a similar lifestyle (in terms of whether they both worked outdoors or indoors, how long they spent in bright light during the day, whether they shared a bedroom and switched on/off the lights at the same times each day). It is therefore plausible that heterogeneity in light exposure may account for within-twin pair variability in chronotype. It would thus be useful for future studies using the MZ twin differences design to comprehensively examine light exposure as a candidate non-shared environmental factor on chronotype. Furthermore, the present sample consists of young adults between 18 and 22 years of age, and it is likely that the types of environmental influences individuals are exposed to change across the life span. Similarly, it is known that circadian rhythms (and hence chronotype) change with age (Duffy and Czeisler, 2002; Jones et al., 2007), as does the relative importance of genetic and environmental influences on chronotype (Barclay et al., 2011b). Accordingly, further research should characterize the environmental correlates of chronotype across the life span. It is plausible that family, work, and social commitments may contribute to one’s preference for sleep-wake timing as one moves into middle adulthood, whereas during the progression to older adulthood, these influences may become less important. In addition, the cross-sectional nature of this study limits our ability to unravel processes of cause and effect. Although it is possible that many of these associations are bidirectional, longitudinal studies assessing the associations between chronotype and the environment will enable us to disentangle these processes. One final consideration is that the conceptualization of the non-shared environment in twin studies inevitably includes measurement error, and so this point should be taken into account when interpreting the results of this study.
While we typically think of circadian rhythms as being largely controlled by biological factors and entrained by the light/dark cycle (as well as other external zeitgebers), this study brings to the forefront the importance of other environmental factors associated with our preferences for sleep-wake timing. However, this study shows that the majority of the environmental influences under investigation are in fact intertwined with genetics. Thus, it is sufficient for us to conclude that while our environmental experiences/exposures are associated with our preferences for morning/evening activity, these associations are partly driven by our internal, biological milieu.
Footnotes
Acknowledgements
Waves 1-3 of the G1219 study were supported by grants from the W T Grant Foundation, the University of London Central Research fund, and a Medical Research Council Training Fellowship and Career Development Award to Thalia C. Eley. Wave 4 was supported by grants from the Economic and Social Research Council (RES-000-22-2206) and the Institute of Social Psychiatry to Alice M. Gregory, who was supported by a Leverhulme Research Fellowship during this research.
Conflict of Interest Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
