Abstract
In fruit flies Drosophila melanogaster, environmental cycles of light and temperature are known to influence behavioral rhythms through dedicated sensory receptors. But the thermosensory pathways and molecular receptors by which thermal cycles modulate locomotor activity rhythms remain unclear. Here, we report that neurons expressing warmth-activated ion channel Drosophila Transient Receptor Potential-A1 (dTRPA1) modulate distinct aspects of the rhythmic activity/rest rhythm in a light-dependent manner. Under light/dark (LD) cycles paired with constantly warm ambient conditions, flies deficient in dTRPA1 expression are unable to phase morning and evening activity bouts appropriately. Correspondingly, we show that electrical activity of a few neurons targeted by the dTRPA1SH-GAL4 driver modulates temperature-dependent phasing of activity/rest rhythm under LD cycles. The expression of dTRPA1 also affects behavior responses to temperature cycles combined with constant dark (DD) or light (LL) conditions. We demonstrate that the mid-day “siesta” exhibited by flies under temperature cycles in DD is dependent on dTRPA1 expression in a small number of neurons that include thermosensory anterior cell neurons. Although a small subset of circadian pacemaker neurons may express dTRPA1, we show that CRY-negative dTRPA1SH-GAL4 driven neurons are critical for the suppression of mid-thermophase activity, thus enabling flies to exhibit siesta. In contrast to temperature cycles in DD, under LL, dTRPA1 is not required for exhibiting siesta but is important for phasing of evening peak. Our studies show that activity/rest rhythms are modulated in a temperature-dependent manner via signals from dTRPA1SH-GAL4 driven neurons. Taken together, these results emphasize the differential influence of thermoreceptors on rhythmic behavior in fruit flies in coordination with light inputs.
Temperature cycles can entrain circadian clocks (Busza et al., 2007; Glaser and Stanewsky, 2005; Miyasako et al., 2007; Picot et al., 2009; Yoshii et al., 2010, 2005, 2009), and dedicated thermoreceptors are likely to be involved in this behavior. In flies, thermoreceptors are present in antennae, aristae (Gallio et al., 2011; Sayeed and Benzer, 1996) and chordotonal organs (Sehadova et al., 2009). The TRP family of ion channels encompasses several thermoreceptors in Drosophilia melanogaster (Montell, 2011), among which only two have been implicated in temperature entrainment: pyrexia (Wolfgang et al., 2013) and drosophila Transient Receptor Potential A1 (dTRPA1; Lee and Montell, 2013). In addition, IR25a, an ionotropic glutamate receptor, has been shown to mediate entrainment to low-amplitude temperature cycles (Chen et al., 2015). The dTRPA1-expressing anterior cells (AC) neurons are considered internal thermosensors (Hamada et al., 2008) and are known to function in the range of 27 °C to 29 °C (Hamada et al., 2008; Tang et al., 2013). Since these temperatures are likely to be encountered by flies daily (Prabhakaran and Sheeba, 2013), we examined the role of dTRPA1 in circadian and non-circadian neurons in modulating daily rhythmic activity under temperature cycles in the context of different light conditions. Recently, a molecular pathway for temperature-dependent degradation of TIMELESS (or TIM) has been proposed to occur via a calpain protease, small optic lobes (Tataroglu et al., 2015) in circadian pacemaker neurons, defining a cell autonomous mode for temperature entrainment.
In the present study, we focused on the role of a small subset of dTRPA1-expressing neurons, targeted by the most conservative and restricted driver, dTRPA1SH-GAL4 (Hamada et al., 2008), henceforth (dTRPA1SH+ neurons), to understand the contribution of dTRPA1 ion channels in modifying behavior in response to different temperature stimuli. We corroborated our results with the use of other available dTRPA1-GAL4 drivers. We studied the circadian clock–controlled activity/rest behavior of flies under both 12:12 h light/dark (LD) and temperature cycles. Under LD cycles, flies shift locomotor activity into the dark phase under constant warm temperatures and into the light phase under constant cool temperatures (Majercak et al., 1999). We asked whether signals from clock gene expressing circadian and/or non-circadian dTRPA1 expressing neurons are needed for such changes in phase and waveform of activity rhythm under LD at constant warm temperatures. We used the dTRPA1SH-GAL4 (Hamada et al., 2008) driver to manipulate dTRPA1 expression or to alter electrical properties of dTRPA1-positive AC thermosensory neurons. Activation of dTRPA1 modulates the phase of morning and evening activity peaks under LD at constant warm ambient temperature. We also examined the activity/rest pattern of flies under 12:12 h thermal cycles (TC 29 °C:21 °C) in constant darkness (DD), where thermophase (29 °C) is within the range of activation of this ion channel, as shown previously (Hamada et al., 2008; Viswanath et al., 2003). Mutational loss of dTRPA1 function renders flies incapable of exhibiting the normal “siesta” under thermal cycles in constant darkness (DD/TC). Our studies show that dTRPA1 expression outside of the known circadian neuronal network is crucial in mediating the rhythmic siesta of flies under DD/TC. Further, we show that signals from neurons targeted by dTRPA1SH+ neurons contribute to phasing of evening activity under TC cycles in constant light (LL). Taken together, the most parsimonious explanation for the above results is that non-circadian dTRPA1SH+ neurons provide sensory inputs to circadian neurons and modulate time-of-day responses to changes in temperature.
Materials and Methods
Fly Strains
All genotypes were reared on standard cornmeal medium under LD (12:12 h) at 25 °C. dTRPA1SH-GAL4 (Hamada et al., 2008), dTRPA1-GAL448951 (Green et al., 2015), and TRPA1KI-GAL4 (Kim et al., 2010) drivers were used to target subsets of dTRPA1-expressing neurons, and Pdf-GAL4 (Renn et al., 1999), cry-GAL4-39 (Klarsfeld et al., 2004), and tim-GAL4-27 (Lear et al., 2005) were used to target subsets of circadian neurons, respectively. Overexpression of dTRPA1 was achieved using UAS-dTRPA1 (Rosenzweig et al., 2008), which is expected to express the dTRPA1-A isoform (see Suppl. Fig. S5 [Hamada et al., 2008]; also see Kang et al., 2010). UAS-Dcr2D (Bloomington Stock Centre) was combined with 2 UAS-dTRPA1RNAi transgenes (VDRC-UAS-dTRPA1T1RNAi #37249 and UAS-dTRPA1T2RNAi #37250) for RNAi knockdown of dTRPA1 in different neurons. In RNAi experiments, dTRPA1SH-GAL4/UAS-dcr-2 served as GAL4 control. Neuronal hyperexcitation was induced by driving the voltage-gated Na+ channel UAS-NaChBac1 (Nitabach et al., 2006; Sheeba et al., 2008a) with dTRPA1SH-GAL4. Apoptosis-inducing transgene UAS-hid (Zhou et al., 1997) was used for ablation of neuronal subsets (donation from Michael Rosbash, Brandeis). dTRPA1ins is a strong loss-of-function mutation (Hamada et al., 2008; Head et al., 2015). TRPA1KI-GAL4 (donated by Youngseeok Lee, Kookmin University) was created by ends-out homologous recombination, which inserted the GAL4 gene at the normal translational initiation codon and also deleted the 185-bp region encoding N-terminal residues (Kim et al., 2010), thus making it a dTRPA1 driver in a null background.
Behavioral Assays
The locomotor behavior of male flies (2 to 4 d old) was measured using drosophila activity monitors (Trikinetics, Waltham, MA) in 5-min binning intervals as described previously (Sheeba et al., 2008b) in incubators (Sanyo, Japan, and Percival Drosophila Chambers, Perry, IA).
Light/Dark Cycles
Flies were subjected to (12:12 h) LD cycles with constant temperatures of 21, 25, 27, or 30 °C (LD21, LD25, LD27, and LD30) for 6 to 7 d followed by constant darkness (DD). Free-running period of individual flies was determined using chi-square periodogram analysis (excluding the first 2 d of DD to avoid transient cycles) with Actimetrics Clocklab software (Wilmette, IL). The activity profile was generated by normalizing activity counts of individual flies in 15-min bins over total activity across 24 h and then averaging across 5 consecutive days. For a given genotype, values were averaged across flies and plotted against time of day. The phase of morning peak was determined for individual flies by identifying the time point of highest activity between ZT20–02. Wherever possible, the true morning peak was distinguished from the startle response at lights-on based on the presence of anticipatory activity before lights-on (Sheeba et al., 2010). We defined the phase of onset of evening activity as the time point at which post-siesta activity levels reached 20% of maximum daily activity (for individual flies). Phase differences in morning peak or onset of evening activity among genotypes were compared using 2-way analysis of variance (ANOVA) with genotypes and temperature as fixed factors (p < 0.05), followed by Tukey’s HSD post hoc comparisons.
Temperature Cycles
Flies were reared under LD (25 °C), and adults were kept in LL (21 °C) for 24 h to eliminate phasing effects of previous LD. Flies were then subjected to 12:12 h temperature cycles in DD (thermophase: cryophase 29:21 °C, DD/TC) for 9 to 10 d to allow for stable entrainment, followed by 10-h phase-shifted TC cycles (advance or delay) for the next 13 d, and then they were released into DD 21 °C for the next 7-10 d (or when specified, directly released into DD 21 °C without phase shift). Flies were considered to have entrained when the period of activity rhythm for 7 d in DD/TC = 24 ± 0.5 h. Activity counts binned into 15-min intervals were first averaged across 5 d for individual flies, and the mean across flies for a given genotype was plotted against time of day. Mean activity counts per 2-h bins were used to compare differences in activity levels among genotypes by performing a 2-way ANOVA with genotype and time points as fixed factors followed by Tukey’s HSD test (p < 0.05). To estimate the rate of reentrainment to shifted temperature cycles, the offset of evening activity was chosen as a phase marker, and the number of transient cycles was calculated for individual flies. The number of transients taken to reentrain to shifted TC cycles was compared across genotypes using nonparametric Kruskal-Wallis ANOVA followed by multiple comparisons of mean ranks (p < 0.05). The free-running period of individual flies was determined using chi-square periodogram analysis in Clocklab using data for at least 7 d in DD. Morning peak amplitude and evening peak phases were manually determined for individual flies subjected to TC cycles in LL (LL/TC). Statistical differences among genotypes were compared by performing ANOVA, followed by Tukey’s HSD (p < 0.05).
Immunocytochemistry
The expression pattern of neurons targeted by dTRPA1SH-GAL4 was studied by driving UAS-2xeGFP and subsequent staining of adult fly brains as described previously (Sheeba et al., 2008a). Primary antibodies used were chicken anti-GFP (1:1000; Molecular Probes), rabbit anti-PER (1:10,000, donated by Ralf Stanewsky), rat anti-TRPA1 (1:500, donated by Paul Garrity), and mouse anti-PDF (1:5000, DSHB). Secondary antibodies anti-chicken Alexa 488 (1:1500, Molecular Probes), anti-rat Alexa 488 (1:3000), anti-rabbit Alexa 546 (1:3000), anti-mouse Alexa 546 (1:3000), and anti-rabbit Alexa 633 (1:3000) from Invitrogen were used. Specimens were imaged under a Zeiss LSM 700 confocal microscope. Five to six brains were stained with each set of antibodies and scanned under the confocal microscope. Images were assembled and brightness and contrast adjusted using Zeiss LSM viewer and ZEN 2011 software. Repression of GAL4-driven expression using cry-GAL80 and Pdf-GAL80 and ablation of dTRPA1SH+ neurons by expressing UAS-hid was confirmed by GFP expression studies using Zeiss epifluorescence microscope as described previously (Das et al., 2015).
Results
Temperature-dependent Modulation of Morning and Evening Activity Bouts under LD Cycles Is Mediated by dTRPA1SH+ Neurons
Changes in ambient temperature alter the pattern of activity of flies under LD cycles (Majercak et al., 1999; Miyasako et al., 2007). Compared with their preferred temperature of 25 °C (Sayeed and Benzer, 1996), flies exposed to constant low temperature (18 °C) shift their evening activity earlier into the day, whereas at constant high temperature (29 °C), flies shift their evening activity later into the night (Majercak et al., 1999). The dTRPA1 channel enables Drosophila to choose their preferred temperature both as larvae (Rosenzweig et al., 2005, 2008) and adults (Hamada et al., 2008; Kaneko et al., 2012; Rosenzweig et al., 2005). We tested whether loss of dTRPA1 alters the waveform of activity/rest rhythm under LD at different ambient temperatures (21, 25, 27, or 30 °C, henceforth LD21, LD25, LD27, and LD30). Wild-type (w1118) flies display a bimodal activity pattern with similar phase of morning (M)-peak under LD21 and LD25 (Fig. 1A). At LD30, the M-peak is significantly advanced (Fig. 1A, black arrow; 1-way ANOVA, F3,105 = 61.1, p < 0.05). Evening activity is delayed under the warmer LD30 condition compared with the cooler LD21 and LD25 regimes (Fig. 1A, gray arrow; 1-way ANOVA, F3,103 = 128.7, p < 0.05). Strong loss-of-function mutant dTRPA1ins flies (Hamada et al., 2008; Head et al., 2015) also exhibit bimodal activity patterns; however, across temperatures, they exhibit no difference in phasing of M-peak (Fig. 1A, right; 1-way ANOVA, F3,90 = 1.98, p = 0.12). Thus, mutant dTRPA1ins flies, unlike controls, are unable to shift their M-peak into predawn under warm LD30 (Fig. 1B, top; 2-way ANOVA, F3,195 = 20.4, p < 0.05). Although dTRPA1ins flies show delayed evening (E) onset compared with w1118 under most regimes, they do not show a further delay under the warm LD30, and in contrast, they show a significantly advanced E-onset compared with w1118 (Table 1; Fig. 1B, bottom; 2-way ANOVA, F3,201 = 44.9, p < 0.05). Wild-type flies delay their E-onset by ~2 h at LD30 compared with LD25 (Fig. 1B, bottom), and lack of dTRPA1 renders flies incapable of delaying E activity under warm conditions of LD30. The wild-type responses at high temperature for LD30 are consistent with an adaptive response of flies to avoid activity during the day when temperatures are generally highest, which may account for light modulation of the temperature response.
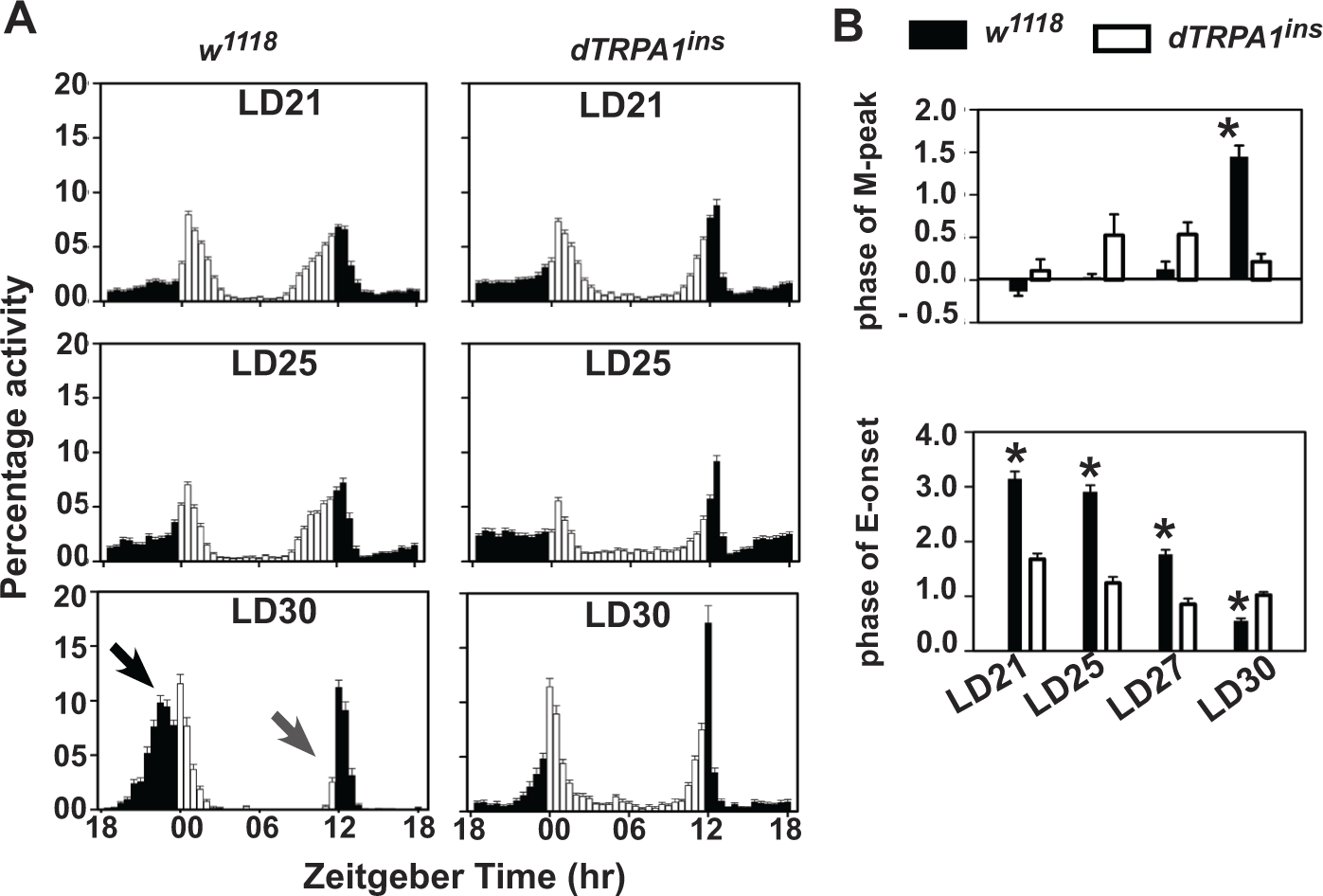
dTRPA1 neurons modulate temperature-dependent phasing of morning and evening activity under LD. (A) Wild-type (w
Phase of morning peak (A) and evening activity onset (B) of various genotypes under LD (12:12) across different ambient temperatures.
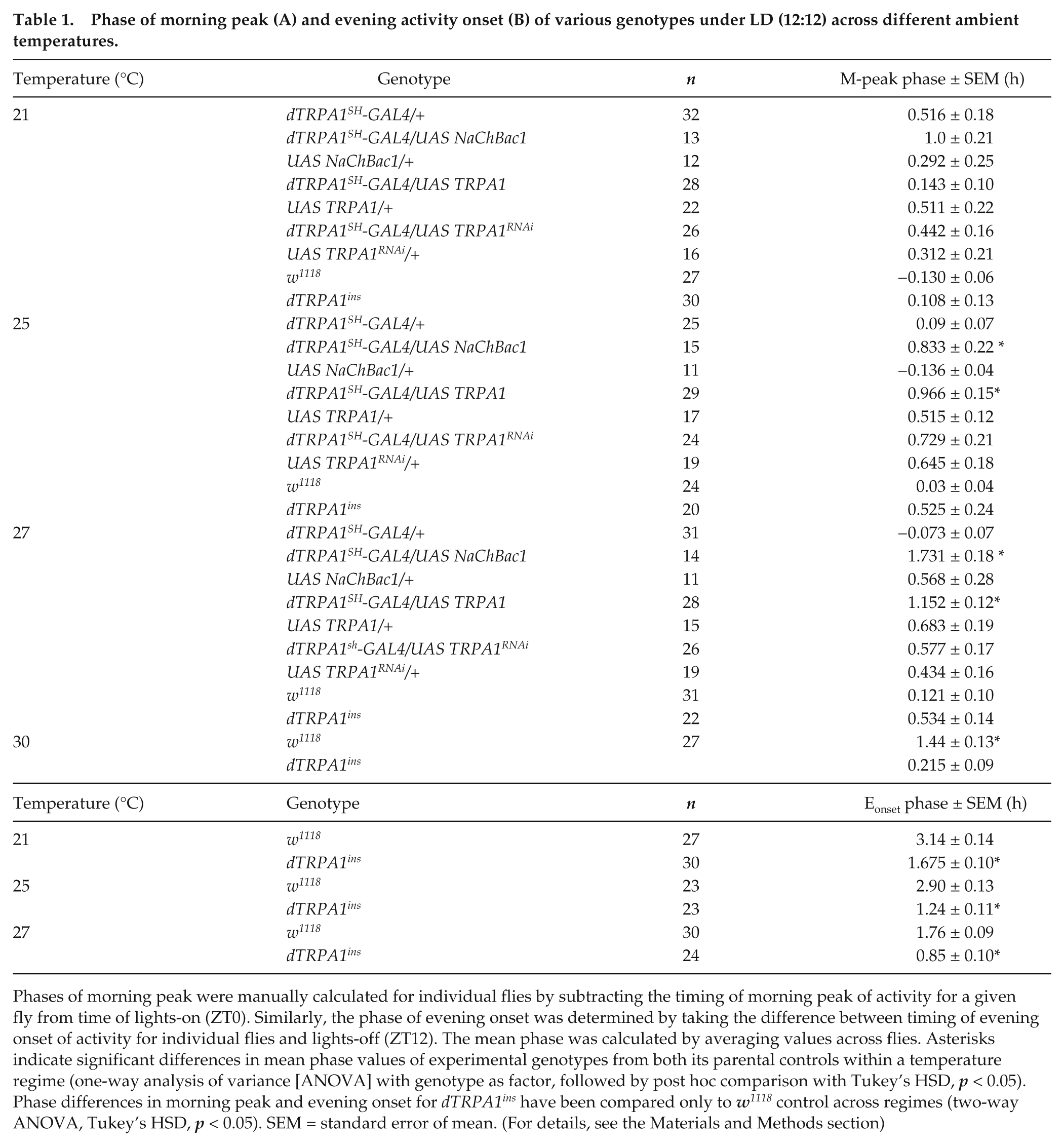
Phases of morning peak were manually calculated for individual flies by subtracting the timing of morning peak of activity for a given fly from time of lights-on (ZT0). Similarly, the phase of evening onset was determined by taking the difference between timing of evening onset of activity for individual flies and lights-off (ZT12). The mean phase was calculated by averaging values across flies. Asterisks indicate significant differences in mean phase values of experimental genotypes from both its parental controls within a temperature regime (one-way analysis of variance [ANOVA] with genotype as factor, followed by post hoc comparison with Tukey’s HSD,
We further overexpressed dTRPA1 channels using the dTRPA1SH-GAL4 driver (henceforth, dTRPA1oex). Overexpression of dTRPA1 does not significantly alter activity profiles under cool LD21, which is below the threshold temperature for dTRPA1 activation (Suppl. Fig. S1, left; Kolmogorov-Smirnov test, p > 0.1). However, under warmer temperatures of both LD25 and LD27, dTRPA1oex flies (Suppl. Fig. S1, left) show a significant advance in the M-peak compared with both parental controls (1-way ANOVA; LD25: F2,68 = 13.96; LD27: F 2,71 = 35.97; p < 0.05). Under cool LD21 conditions, dedicated thermosensors for cool temperature may override the activation of dTRPA1 neurons. We tested this by constitutive expression of UAS-NaChBac1 (dTRPA1hyperexcited). Subjecting dTRPA1hyperexcited flies to cool LD21 also does not significantly affect activity patterns (Suppl. Fig. S1, right). Interestingly, at both LD25 and LD27, electrical hyperexcitation causes an advancement of M-peak compared with controls (1-way ANOVA; LD25, F2,48 =13.13; LD27, F2,53 = 44; p < 0.05). For dTRPA1hyperexcited flies (blue curves), predawn activity appears to be broader under LD27 than under LD25 (Suppl. Fig. S1, right). Thus, artificially enhancing firing of dTRPA1SH+ neurons (dTRPA1oex or dTRPA1hyperexcited) at ambient temperatures of LD25 is sufficient to advance morning activity. This resembles w1118 at warmer conditions of LD30, showing that these neurons can modulate activity peaks. Our studies on the null mutants reveal that dTRPA1 provides temperature sensory input critical for flies to appropriately phase their activity peaks around the light/dark and dark/light transitions when ambient temperature is warm.
dTRPA1 Is Needed for Suppression of Mid-day Activity under Temperature Cycles
We subjected dTRPA1 null mutant lines to 12:12-h temperature cycles under DD (
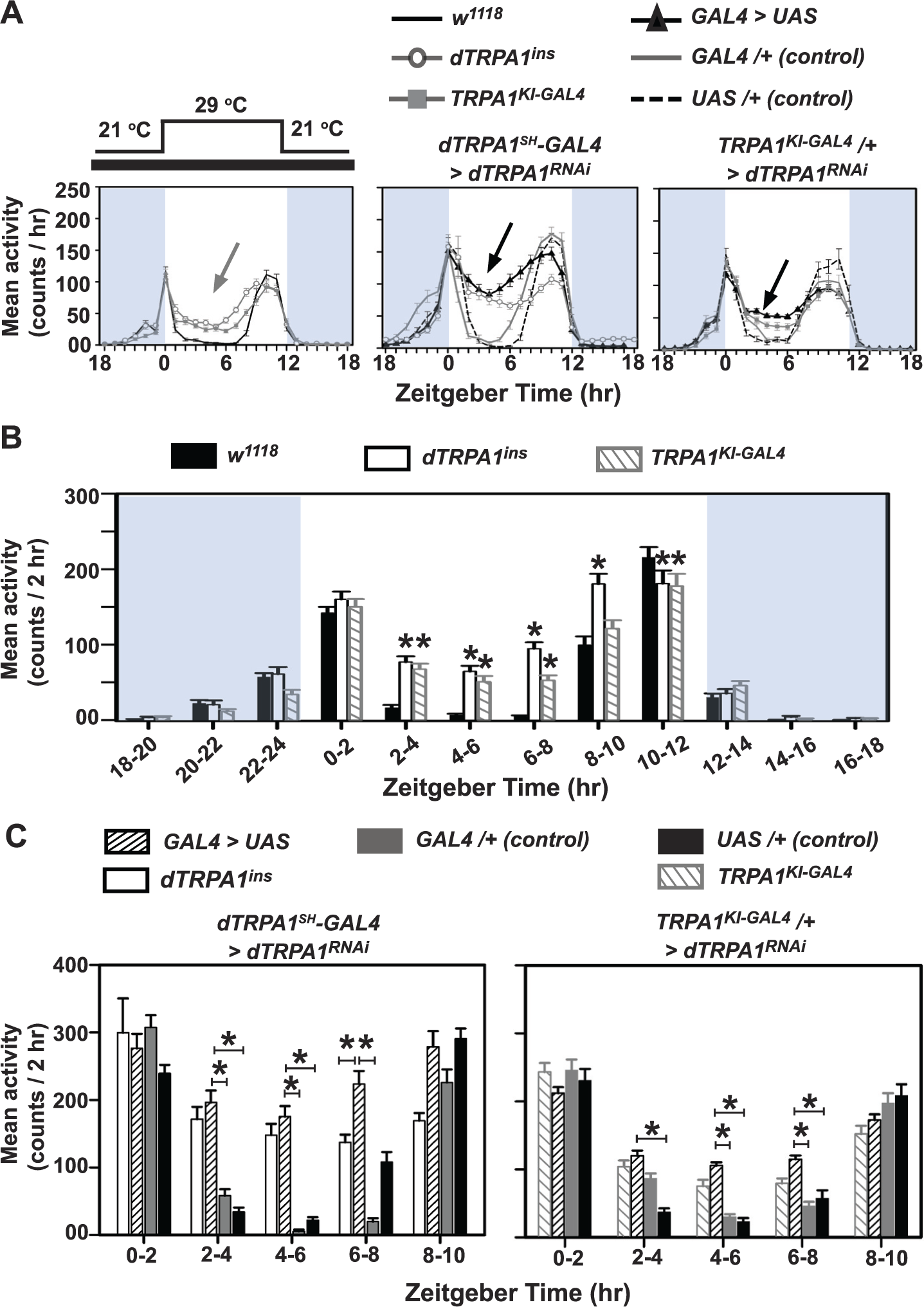
dTRPA1 is essential for consolidation of activity/rest rhythm and to inhibit mid-day activity under laboratory rectangular temperature cycles (DD/TC 29:21). (A) Average activity profiles of flies with altered levels of dTRPA1 in neurons targeted by dTRPA1
Both null mutants—dTRPA1ins and TRPA1KI-GAL4—show greater activity compared with w1118 control flies during ZT02-08 (mid-thermophase; Fig. 2B, 2-way ANOVA, F22,804 = 29.1; significant differences from w1118 within each time interval are indicated by *p ≤ 0.05). Flies with dTRPA1RNAi under dTRPA1SH-GAL4 also exhibit higher activity than controls between ZT02-08 of the thermophase (Fig. 2C, left, 2-way ANOVA, F33,912 = 13.0). During the cryophase (below the threshold activation temperature of dTRPA1), there is no detectable difference in activity levels between experimental and control genotypes (Fig. 2A, middle). Similarly, dTRPA1 down-regulation under TRPA1KI-GAL4 /+ also causes flies to exhibit significantly enhanced mid-thermophase activity compared with parental controls, between ZT04-08 (Fig. 2C, right, 2-way ANOVA, F33,984 = 7). RNAi knockdown in dTRPA1 neurons phenocopies the loss of mid-day behavioral suppression seen in the dTRPA1 null mutant flies, suggesting that dTRPA1 enables suppression of mid-day activity. Enhanced activity of dTRPA1ins and dTRPA1RNAi flies during the warm phase continued even after the 10-h phase delay in TC, although some separation between “morning” and “evening” bouts of activity was apparent during the last few days of the shifted cycles (Suppl. Fig. S2A). Further, we also knocked down dTRPA1 expression using another driver, dTRPA1-GAL448951, previously shown to target cells needed for the afternoon peak under seminatural conditions (Green et al., 2015). This driver targets a very small number of neurons (Suppl. Fig. S3A) and does not include AC neurons in most cases (7/11 brains). This does not result in a loss of siesta (Suppl. Fig. S3B). Taken together, our results suggest that dTRPA1 in AC neurons is crucial for mediating the siesta phenotype under rectangular temperature cycles.
We also subjected flies driving UAS-dTRPA1 expression under dTRPA1SH-GAL4 (henceforth, dTRPA1oex) to DD/TC. Not surprisingly, we find that they have similar activity levels compared with both parental controls (Suppl. Fig. S4A, B) as mid-day activity of controls in this condition is already close to zero (Suppl. Fig. S4B). dTRPA1oex flies differ from dTRPA1ins null flies during mid-day ZT02-06 (Suppl. Fig. S4B, right; 2-way ANOVA, F33,1358 = 25.1). Altering dTRPA1 expression, specifically in dTRPA1SH+ neurons, does not affect endogenous periodicity of activity/rest rhythm when released into DD21 following thermal cycles (Table 2), suggesting that dTRPA1-mediated changes in neuronal activity have no effect on circadian clock period. Moreover, the number of transient cycles required for reentrainment to phase-shifted temperature cycles is not significantly higher than controls in either the dTRPA1ins or dTRPA1SH-GAL4 mediated dTRPA1RNAi flies (Suppl. Fig. S2B). Similar results were obtained for phase-advanced shifts (data not shown), suggesting that signaling via dTRPA1 does not influence the rate of reentrainment to phase-shifted thermal cycles. Our results are bolstered by a recent report that also shows that dTRPA1 is not required for circadian entrainment to temperature cycles (Roessingh et al., 2015). However, alteration of dTRPA1 channel signaling disrupts the normal siesta, thus causing flies to be maladaptively active at mid-day when temperatures are ordinarily highest and flies are at higher risk for desiccation.
Percentage of flies exhibiting entrained locomotor activity rhythm to TC cycles and free-running rhythm under DD at 21 °C along with free-running period values and measure of robustness of rhythm.
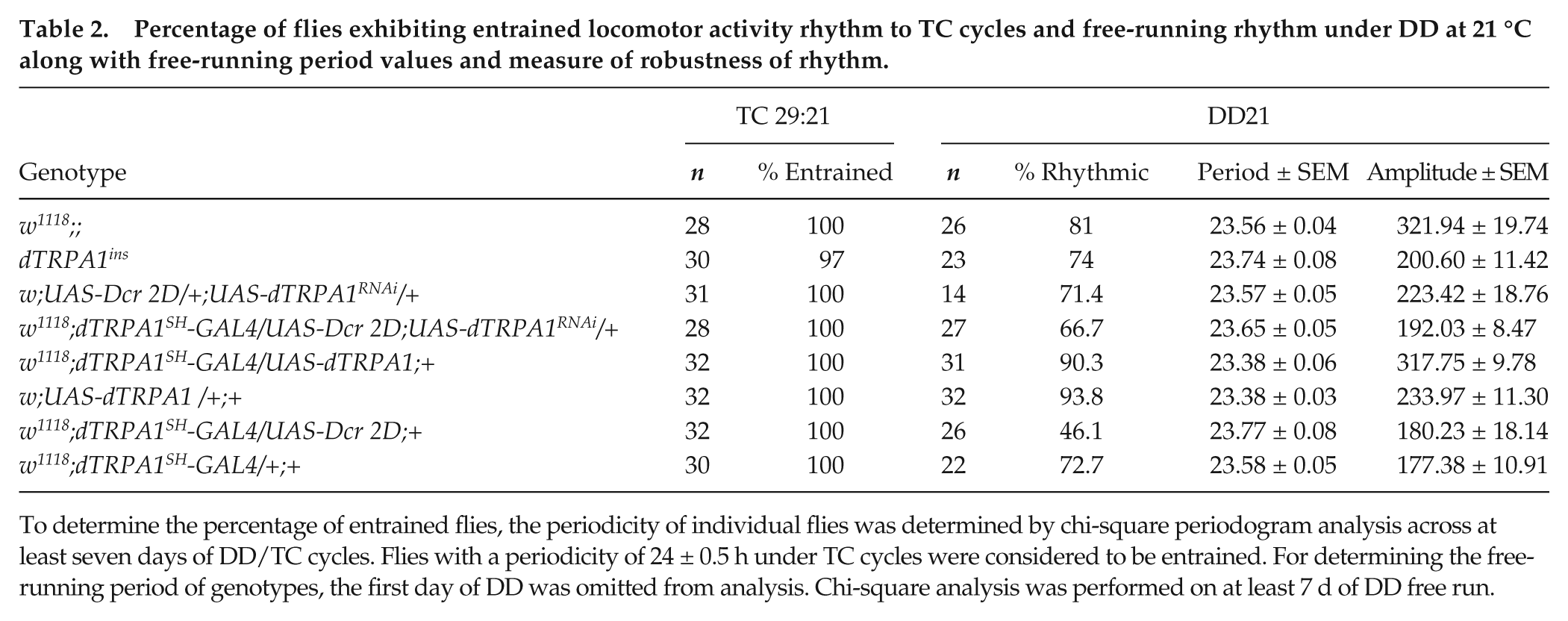
To determine the percentage of entrained flies, the periodicity of individual flies was determined by chi-square periodogram analysis across at least seven days of DD/TC cycles. Flies with a periodicity of 24 ± 0.5 h under TC cycles were considered to be entrained. For determining the free-running period of genotypes, the first day of DD was omitted from analysis. Chi-square analysis was performed on at least 7 d of DD free run.
dTRPA1 Expression in a Small Number of dTRPA1SH+ Brain Cells Is Both Necessary and Sufficient to Rescue Mid-day Inhibition of Activity under Thermal Cycles
To verify that dTRPA1SH+ neurons modulate mid-day activity, we ablated these neurons by driving ectopic expression of UAS-hid under dTRPA1SH-GAL4 driver (henceforth, dTRPA1ablated) and subjected flies to DD/TC. dTRPA1ablated flies exhibit weakened siesta especially on initial exposure to DD/TC (Fig. 3A, top-left actogram marked by asterisk; bottom-left black triangles), although a more normal siesta behavior becomes apparent after ~5 d. Thus, dTRPA1ablated flies initially phenocopy the null dTRPA1ins (Fig. 3A, bottom-left, gray open circles) and knockdown dTRPA1RNAi flies (compare with Fig. 2A, middle). Between ZT04-08, dTRPA1ablated flies exhibit significantly higher activity compared with controls (Fig. 3A, bottom-right; 2-way ANOVA, F33,1284 = 25.34). Thus, dTRPA1SH+ neurons are necessary for inhibition of mid-day activity.
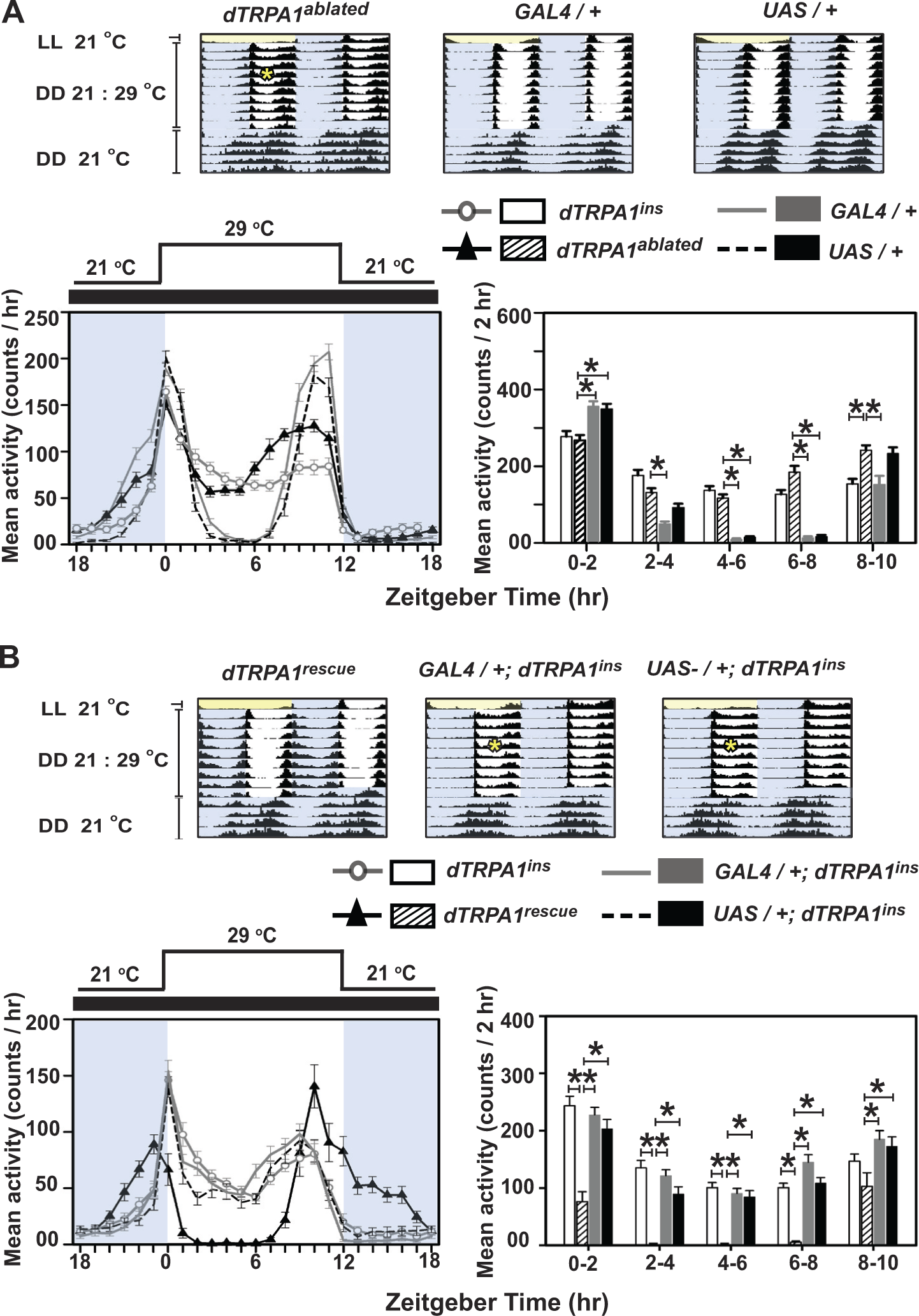
dTRPA1 function targeted by the most spatially restricted dTRPA1 driver in the brain is necessary and sufficient to inhibit mid-day activity under temperature cycles. (A, top) Average actograms of flies with ablated dTRPA1SH+ neurons (dTRPA1
To examine whether dTRPA1 expression only in the small number of dTRPA1SH+ neurons is sufficient to inhibit mid-day activity under DD/TC, we restored dTRPA1 expression in the dTRPA1SH+ neurons in the dTRPA1ins null background (dTRPA1rescue). This genetic manipulation restores the mid-day siesta. dTRPA1rescue flies exhibit wild-type behavior, as shown by clear suppression of mid-day activity in 41 of 42 flies tested (Fig. 3B, top-left actogram; Fig. 3B, bottom-left, black triangles) in contrast to genetic background control flies bearing the null mutation (dTRPA1SH-GAL4; dTRPA1ins and UAS-dTRPA1; dTRPA1ins), which fail to suppress their mid-day activity efficiently (Fig. 3B actograms, asterisks; Fig. 3B, bottom panels). However, dTRPA1rescue flies also exhibit significantly enhanced nocturnal activity and a slightly advanced and smaller morning peak (Fig. 3B, top-actograms, bottom-black triangles), which partially resembles the high temperature response seen in control flies for LD30 (Fig. 1A), not seen in either of the background controls (Fig. 3B actograms; Fig. 3B, bottom left). The increase in late-cryophase (ZT19-23) activity for dTRPA1rescue flies could be partly attributed to the effect of expressing dTRPA1 under the dTRPA1SH-GAL4 driver (compare with Suppl. Fig. S4B, blue curve), although this does not explain the apparent temperature hyperresponsive increased early-cryophase activity. Interestingly, enhanced nocturnal activity is restricted to expression of UAS-dTRPA1 by the dTRPA1SH-GAL4 driver in a null background and does not occur when expressed in a wild-type background (Suppl. Fig. S4B), once again suggesting that the driver does not completely mimic endogenous dTRPA1 expression. Further, it suggests that a combination of the absence of dTRPA1 in some cells accompanied with the overexpression in dTRPA1SH+ cells can cause enhanced nocturnal activity.
dTRPA1 Expression in the Adult Fly Brain Overlaps with Few Circadian Neurons
In contrast to the relatively broad expression pattern of the TRPA1KI-GAL4 driver (Lee, 2013; Lee and Montell, 2013), we find dTRPA1SH-GAL4–driven GFP expression in a small number of neurons, including 2 thermosensory AC neurons in each hemisphere and ~4 cells in the suboesophageal ganglion (Suppl. Fig. S5A; also see Hamada et al., 2008; Shih and Chiang, 2011). The AC neurons send dense projections to the dorsal brain in which one branch forms ipsilateral arbors while another sends contralateral projections to the opposite hemisphere (Suppl. Fig. S5A, also see Hamada et al., 2008; Shih and Chiang, 2011). A single cell in the anterior-dorsal brain in each hemisphere (Shih and Chiang, 2011), which we refer to as dorsal cell (DC; Suppl. Fig. S5A, B) and 4 to 6 cells above the superior arch (Suppl. Fig. S5A) are also seen. We also detect a pair of dorsal-lateral cells (Suppl. Fig. S5A, B, arrows) in each hemisphere and 2 cells in the accessory medulla region (Suppl. Fig. S5A, C, asterisks).
When coimmunostained with antibodies against circadian protein PERIOD and/or PDF, we find that 1 dTRPA1SH+ cell in the accessory medulla overlaps with 1 PER+PDF+ s-LNv (Fig. 4A, yellow arrow) and a second dTRPA1SH+ cell that overlaps with the PER+PDF- fifth s-LNv (Fig. 4A arrowhead; Fig. 4B-D; Table 3). This fifth s-LNv sends GFP-labeled projections to the dorsal brain that runs parallel to the PDF+ s-LNv projections (Suppl. Fig. S5C, yellow arrow) as previously described (Helfrich-Forster et al., 2007). We also find that a pair of dorsal-lateral dTRPA1SH+ cells overlaps with 2 LNd (Fig. 4A-D, 4F, white arrows; Table 3) similar to another study (Yoshii et al., 2015). Thus, we find a modest number (3-4 pairs) of canonical circadian neurons that overlap with dTRPA1SH-GAL4 driver expression (Fig. 4G; Table 3). However, we do not detect any overlap with DN1a (Yoshii et al., 2015), although we do observe consistent overlap with one sLNv (Fig. 4G). In summary, the overlap with circadian neurons with dTRPA1SH-GAL4 driver is narrower than TRPA1KI-GAL4 driver, which targets up to 12 pairs of circadian neurons (Lee, 2013).
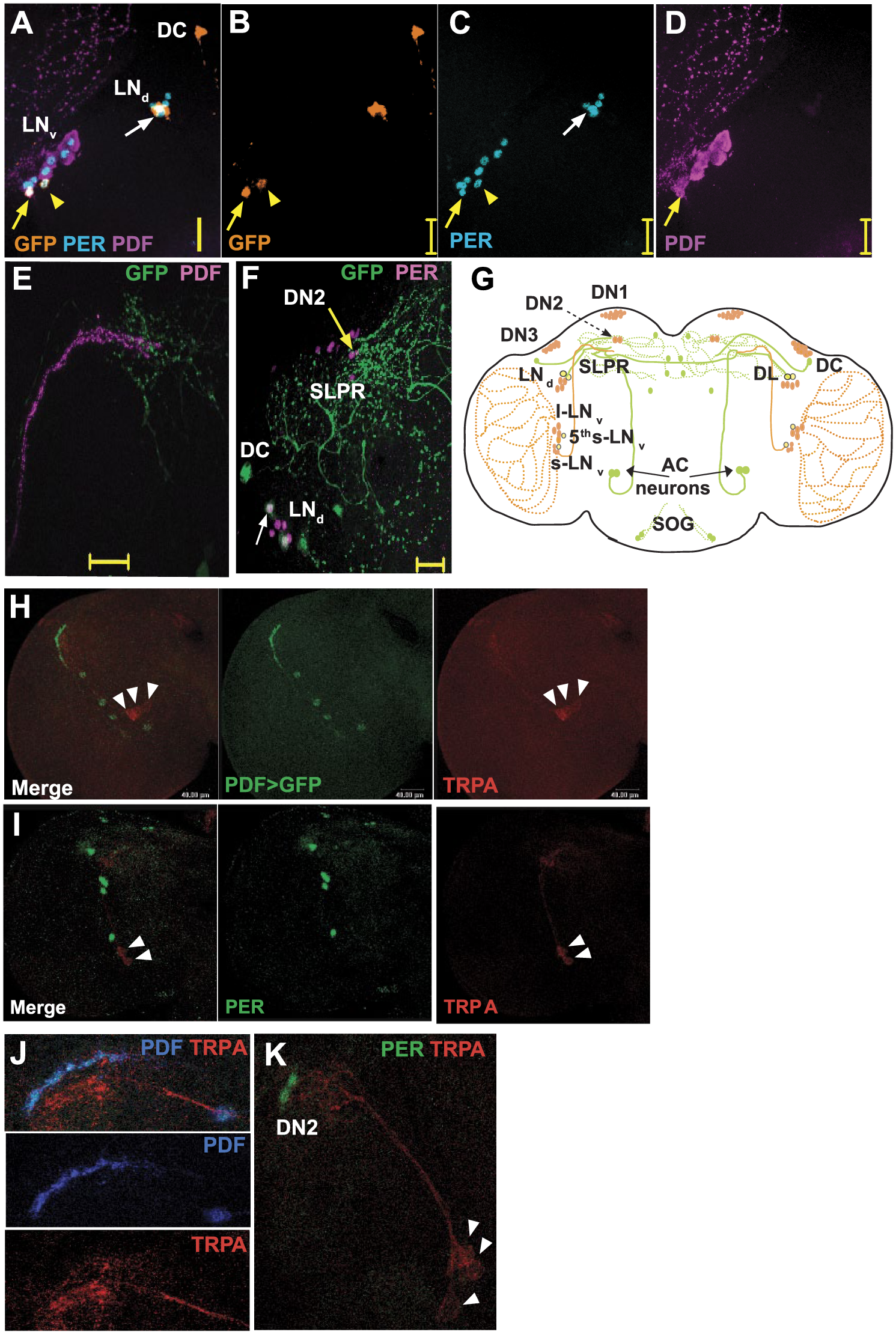
Overlap between circadian proteins and dTRPA1 expression in cell bodies and arborization fields. (A-D) Neurons targeted by a dTRPA1
Extent of overlap between dTRPA1 cells and known circadian neurons in adult fly brains.
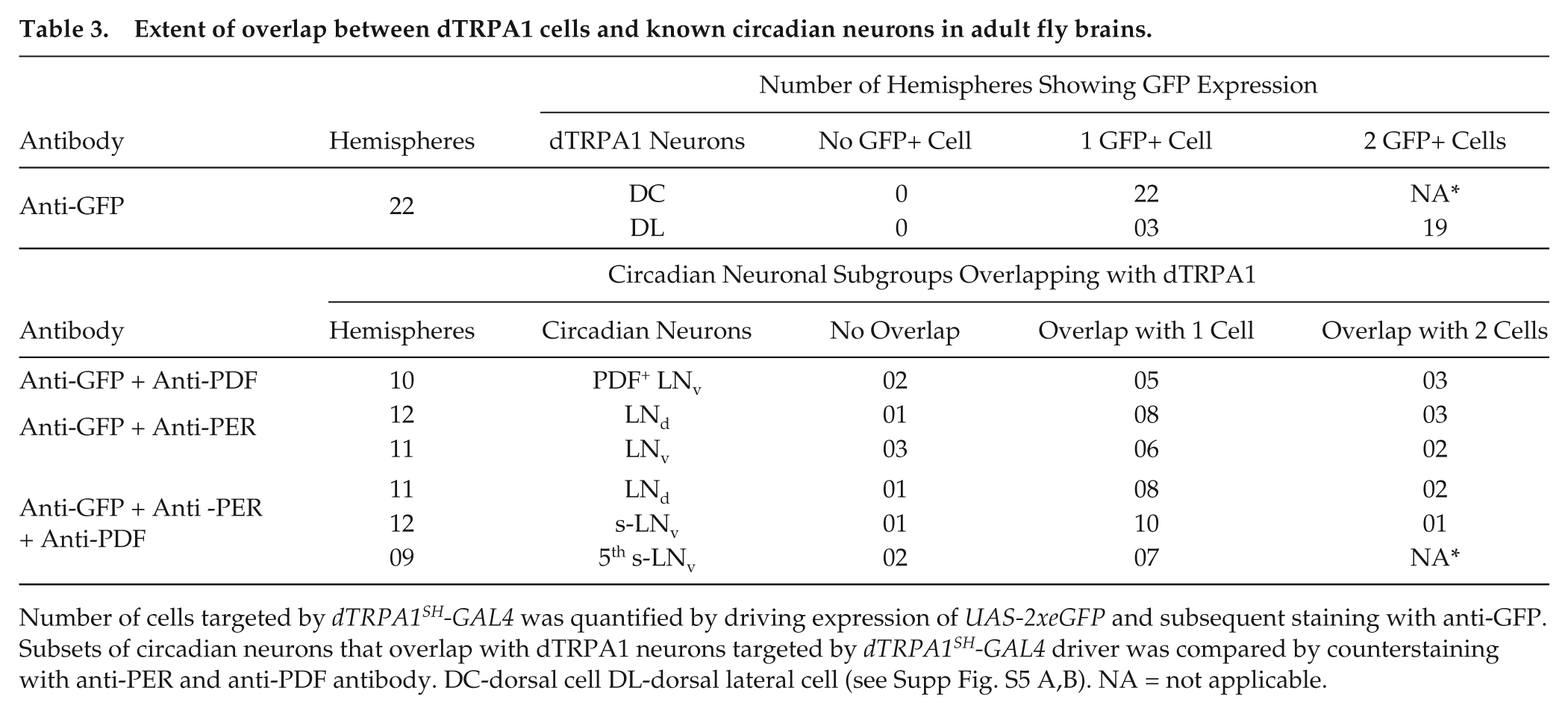
Number of cells targeted by dTRPA1SH-GAL4 was quantified by driving expression of UAS-2xeGFP and subsequent staining with anti-GFP. Subsets of circadian neurons that overlap with dTRPA1 neurons targeted by dTRPA1SH-GAL4 driver was compared by counterstaining with anti-PER and anti-PDF antibody. DC-dorsal cell DL-dorsal lateral cell (see Supp Fig. S5 A,B). NA = not applicable.
In addition, we show that the terminals of dorsal projections of s-LNv partially overlap with the dense arborizations of thermosensory AC neurons in the superior lateral protocerebrum (SLPR; Fig. 4E). Similarly, in larval brains stained with anti-TRPA1 antibody, we detect arbors of dTRPA1-expressing cells partially overlapping with dorsal projections of sLNv (Fig. 4H, J). In adult brains, the terminals of AC neurons also appear to project near the DN2 cell bodies (yellow arrow) in the SLPR (Fig. 4F). Costaining third instar larval brains with anti-TRPA1 and anti-PER (Fig. 4K) also revealed that the projections of larval dTRPA1-expressing neurons arborize in the vicinity of larval DN2. We find no overlap of dTRPA1-expressing cells with larval LNv (labeled by Pdf-GAL4>UAS-GFP; Fig. 4H) or PER+ circadian neuronal subgroups (Fig. 4I). In adult brains also, similar to a previous study (Hamada et al., 2008), we also see a pair of LC and pair of VC neurons labeled with anti-TRPA1 that do not overlap with known PER+ circadian neurons (Suppl. Fig. S5D-F). The fact that s-LNv neurons express 5HT-1B receptors (Yuan et al., 2005) and AC neurons secrete neurotransmitter serotonin (Shih and Chiang, 2011) in combination with the close proximity of s-LNv projections to terminals of AC neurons suggests the possibility of functional connectivity between the circadian and thermosensory circuits in the Drosophila brain (Fig. 4G), whereby AC neurons might communicate warm temperature signals to the circadian neurons via serotonin.
dTRPA1-dependent Suppression in Mid-thermophase Activity Is Not Mediated by Circadian Neurons Expressing dTRPA1
To test whether dTRPA1 expression in circadian neurons is sufficient to modulate mid-thermophase activity, we systematically down-regulated dTRPA1 levels in circadian neurons from wide to narrowing degrees of expression using tim-GAL4 driver, which targets all circadian neurons; cry-GAL4-39 driver, which targets LNv, LNd, and DNs (Klarsfeld et al., 2004); and pdf-GAL4 driver (Renn et al., 1999), which targets the LNv (Fig. 5). Flies with dTRPA1 down-regulation in circadian neurons Pdf-GAL4/dTRPA1RNAi or cry-GAL4-39/dTRPA1RNAi or tim-GAL4/dTRPA1RNAi show no defect in mid-day activity (Figs. 5A, B; 2-way ANOVA, followed by Tukey’s HSD, p < 0.05). RNAi knockdown under cry-GAL4-39 increases mid-day activity only between ZT6-8 (Fig. 5B, middle panel). Further, ablation of circadian neurons by expressing UAS-hid under Pdf-GAL4 or cry-GAL4-39 drivers also does not modify mid-thermophase activity under DD/TC (data not shown).
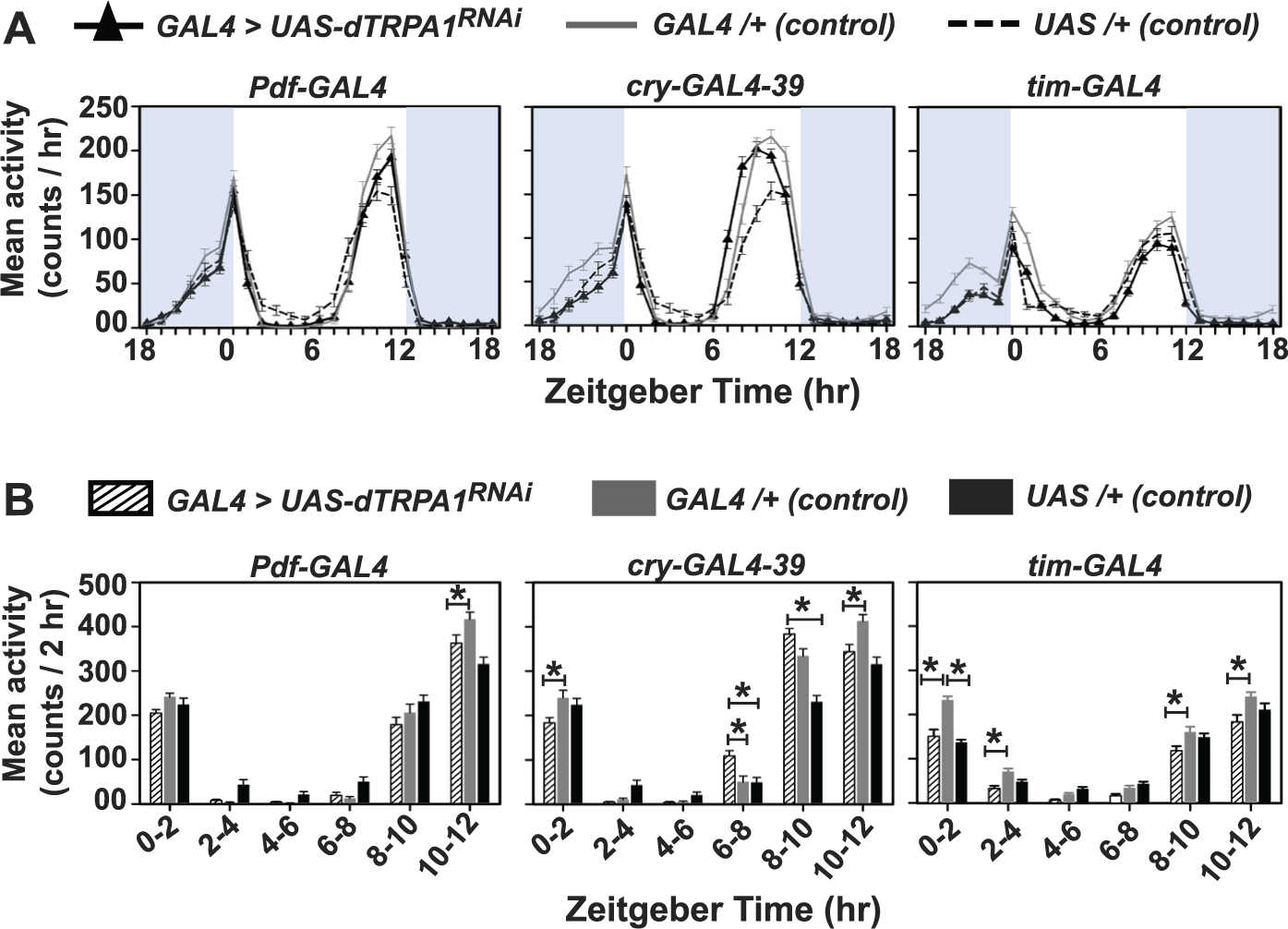
RNAi knockdown of dTRPA1 expression in circadian neuronal subsets does not affect mid-thermophase activity under temperature cycles. (A, left) Down-regulation of dTRPA1 in PDF+ neurons (Pdf-GAL4), (middle) CRY-expressing neurons (cry-GAL4-39) or (right) pan-circadian neurons targeted by tim-GAL4 does not alter mid-day activity under DD/TC. (B) Quantification of mean activity levels binned in 2-h intervals from ZT0 through ZT12 is shown for the above genotypes. dTRPA1 RNAi knockdown in neither of the circadian neuronal subgroups, PDF+ or tim-expressing cells, increase mid-day activity under DD/TC. Down-regulation of dTRPA1 in CRY+ neurons increases activity only between ZT6-8. Asterisks indicate significant differences among genotypes (2-way ANOVA, Tukey’s HSD, p < 0.05). Shaded regions indicate cryophase (21 °C). All other details are the same as in Figure 2.
Since we find that dTRPA1SH+ neurons are important for the siesta and also overlap with circadian neurons, whereas RNAi-mediated knockdown of dTRPA1 in circadian neurons alone does not impair the siesta phenotype, we attempted to verify that the non-circadian dTRPA1SH+ neurons are sufficient to induce the siesta. We knocked down dTRPA1 expression in all dTRPA1SH+ neurons except the LNv using dTRPA1SH-GAL4+Pdf-GAL80 (Suppl. Fig. S6A, B, left panels). Flies with RNAi knockdown (blue curve) display mid-thermophase activity similar to controls (Suppl. Fig. S6B, left panel; 2-way ANOVA, F55,1536 = 6.07). Similarly, dTRPA1SH-GAL4+cry-GAL80–mediated RNAi down-regulation only in CRY-negative dTRPA1SH+ neurons (Suppl. Fig. S6A, B, right panels) results in suppression of mid-day activity similar to controls (Suppl. Fig. S6A, B, right panels, 2-way ANOVA, F22,696 = 17.3) and reduced evening activity (Suppl. Fig. S6A, right panel). Unlike RNAi knockdown in dTRPA1SH+ neurons (Fig. 2A, middle, right panels), restricting RNAi knockdown to only circadian neurons expressing dTRPA1 (Fig. 5A) or to only non-circadian dTRPA1SH+ neurons (Suppl. Fig. S6A) is not sufficient to elicit enhanced mid-thermophase activity. Taken together, these results suggest that a combinatorial knockdown in both circadian and non-circadian dTRPA1SH+ neurons manifests as a loss of mid-day siesta phenotype under DD/TC, while limited knockdown in circadian neurons alone is insufficient to disrupt siesta. We also speculate that the circadian GAL80 drivers may target important dTRPA1SH+ neurons, thereby preventing RNAi knockdown in these cells, which in turn prevents enhanced mid-thermophase activity. This is also consistent with the results obtained by knocking down dTRPA1 with the TRPA1KI-GAL4 driver (Fig. 2A, right panel), which also includes a large number of circadian neurons.
Under Temperature Cycles with Constant Light, dTRPA1 Influences Both Morning and Evening Peaks of Activity
Constant light (LL) is known to disrupt circadian clocks (Konopka et al., 1989), but temperature cycles can entrain the circadian clock in LL (Glaser and Stanewsky, 2005; Sehadova et al., 2009). Although dTRPA1null flies are unable to suppress mid-day activity in the absence of light (DD/TC; Fig. 2A), this defect is not seen under LD (Fig. 1A). We asked if the continuous presence of light could modify this behavior under TC cycles. Flies were subjected to temperature cycles (12:12 h −29:21 °C) under LL of 0.1, 1, 10, or 100 lux intensity. Both w1118 and dTRPA1ins flies synchronize their activity/rest rhythm to LL/TC (Fig. 6A, B) and suppress afternoon activity in contrast to their behaviour under DD/TC. Interestingly, dTRPA1ins flies (gray open circles) exhibit a reduced M-peak amplitude (2-way ANOVA, with genotype and light intensity as factors, F3,213 = 0.7) and a ~2-h advanced evening peak compared with w1118 (black) under LL/TC across all light intensities tested (Fig. 6C; 2-way ANOVA, F3,204 = 5.12). However, restoring dTRPA1 only in dTRPA1SH+ neurons in a dTRPA1ins null background is sufficient to rescue M-peak amplitude and phasing of E-peak under LL/TC at 100 lux light intensity (Fig. 6D). E-peak occurs after ZT12 in dTRPA1rescue flies, whereas both parental controls in null background exhibit reduced M-peak (1-way ANOVA, F2,75 = 20.5) and an advanced E-peak under LL/TC (Fig. 6D, right panel, 1-way ANOVA, F2,75 = 95.8). Thus, dTRPA1 function in dTRPA1SH+ neurons is directly responsible for morning and evening bouts of activity under LL/TC. Taken together, our study suggests that thermosensitive dTRPA1 ion channels can influence distinct behaviors depending on external environmental conditions.
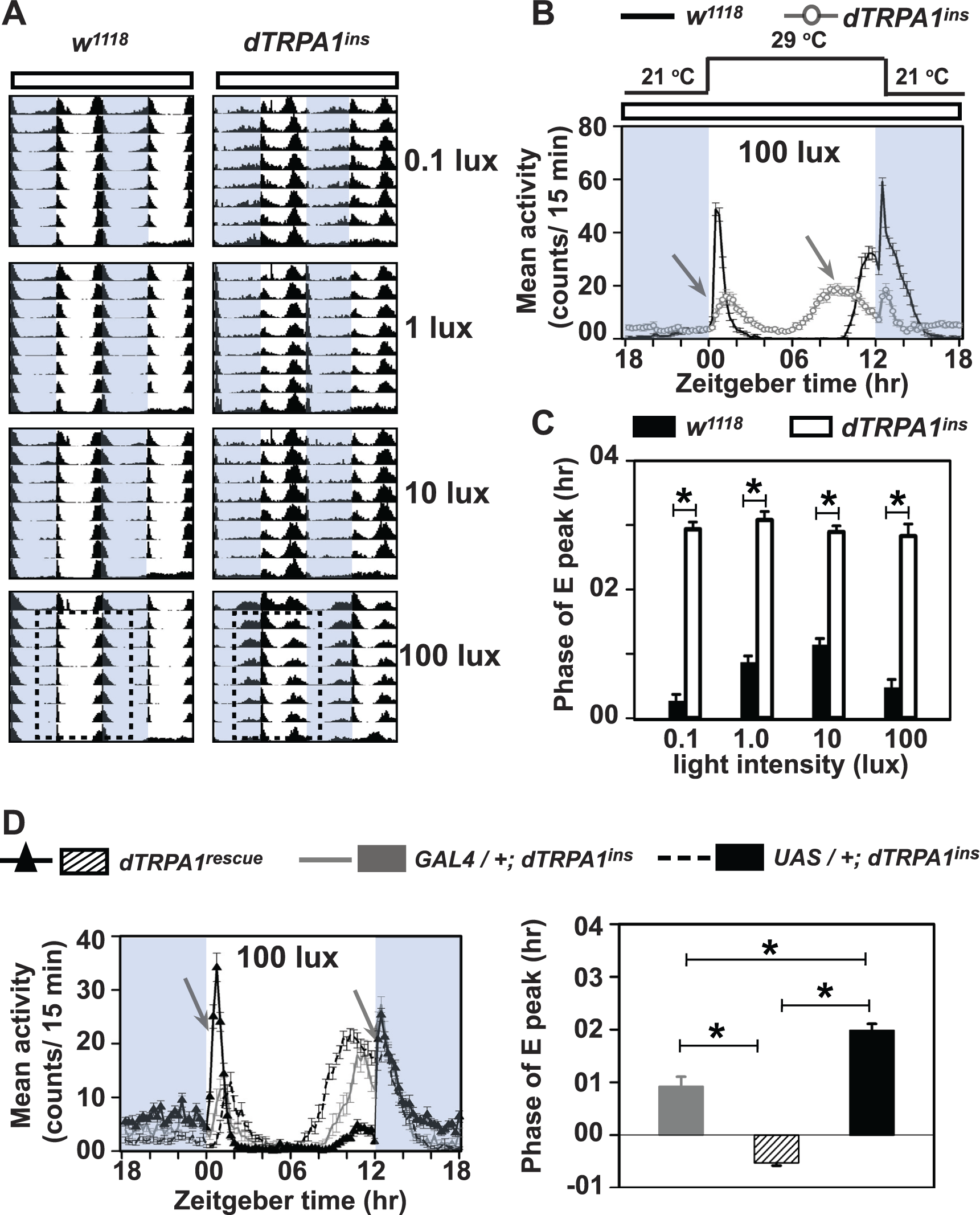
Lack of dTRPA1 modifies morning and evening peak under temperature cycles in constant light (LL/TC). (A) Average actograms of wild-type (w
Discussion
The role of dTRPA1 in thermosensation in Drosophila has been extensively studied in the past decade (Barbagallo and Garrity, 2015). But dTRPA1’s role in conveying thermal inputs to circadian clocks was suggested only recently (Lee and Montell, 2013). Their study did not conclusively identify dTRPA1 as a circadian thermosensor and left open the possibility of other unidentified thermosensors. Our study differs from that of Lee and Montell (2013) in several aspects, most importantly in their choice of asymmetric 18:6 h cycles with 29:18 °C TC cycles. We avoided using such a low cryophase temperature based on our observation that 18 °C greatly suppresses activity and contributes to masking of activity rhythms and an exaggerated startle to the morning transition rather than a gradual anticipation. While they inferred the role of dTRPA1 in entrainment based on the behavior of flies when shifted from symmetric LD cycles to an asymmetric TC, we used symmetric 12:12 h, 29:21 °C TC cycles to examine the ability of flies to shift between advanced or delayed TC cycles and did not detect any significant difference in number of transient cycles. Prior to phase shifts, we ensured that flies have entrained to the TC regime with a stable phase of entrainment and a period of 24 ± 0.5 h. Hence, we propose that the observed defects in entrainment reported by the previous study may be due to after effects of LD to TC rather than a difference in circadian entrainment to thermal cycles. Furthermore, it appears that the long-day regime does not entrain even the wild-type flies, as all genotypes appear to free run even in the presence of the TC cycles (Lee and Montell, 2013; Fig.1D-F; also see footnote in Maguire and Sehgal, 2015).
Here, we report that dTRPA1 modulates aspects of the activity/rest rhythm under LD cycles when temperatures are constantly high and in the range that can activate dTRPA1 signaling. We posit that dTRPA1ins flies are unable to sense extreme warm temperatures because of lack of signaling from dTRPA1SH+ neurons and therefore are unable to redistribute their activity into the dark phase of LD30. Inducing electrical activity of dTRPA1SH+ neurons (via UAS-NaChBac or UAS-dTRPA1) advances the phase of M-peak under LD in a temperature-dependent manner, suggesting that signals from dTRPA1SH+ neurons in the wild-type flies can shift the M-peak under warm ambient temperatures. As hyperexcitation of the dTRPA1SH+ neurons at LD21 (Suppl. Fig.S1, right) does not modify the morning and evening peaks, it appears that at cool temperatures, other thermoreceptors participate in modulation of activity patterns, while at moderate or warm temperatures, activation of dTRPA1 neurons has dominant effects on this behaviour, such that it mimics the sensation of constant warm conditions.
Our studies support the inference that when gated temperature cycles are the only time cues available, inhibitory signals due to dTRPA1 in a subset of neurons (including AC neurons) is both necessary and sufficient to prevent excessive locomotor activity during the thermophase. We hypothesize that these neurons are the temperature-sensitive cells (TS cells) proposed by a previous study to be a set of neurons distinct from the canonical circadian subsets that inhibit activity in the middle of the warm phase under temperature cycles (Busza et al., 2007). This is also consistent with our finding that down-regulation of dTRPA1 in CRY+ or TIM+ cells (including most circadian pacemaker neurons) does not abolish mid-thermophase activity. dTRPA1SH-GAL4 driven GFP expression co-localizes with PERIOD in 1-2 LNd, 1 s-LNv, and fifth s-LNv neurons (Fig. 4). Interestingly, down-regulation of dTRPA1 using the dTRPA1SH-GAL4 driver in the presence of cry-GAL80 rescues the mid-day suppression of activity. We find that effective knockdown of the channel using RNAi (resulting in phenotype similar to null mutants) occurs only with the dTRPA1SH-GAL4 or TRPA1KI-GAL4 driver, whereas the prevention of RNAi in the CRY+ subset of dTRPA1SH+ neurons enables inhibition of mid-day activity. We speculate that the cry-GAL80 driver could target neurons outside of the cry-GAL4 driver and within the target of the dTRPA1SH-GAL4 driver, and hence lack of RNAi in that subset of dTRPA1SH+ neurons suppresses mid-day activity. These results lead us to lean toward the hypothesis that the CRY-negative dTRPA1SH+ neurons primarily control suppression of mid-thermophase activity, although it is also likely that both circadian and non-circadian neurons contribute to the siesta.
It is also possible that since distal processes of AC neurons appear to partially overlap with dorsal terminals of s-LNv (Fig. 4E), the former can perceive differences in temperature via dTRPA1 ion channels and convey temporal information to s-LNv (by being active during warm temperatures and being inactive at cooler temperatures). s-LNv in turn send projections to CRY-negative DN2 neurons, and the existence of synaptic communication between s-LNv and DN2 has been suggested to be a possibility in a previous study that implicates the role of DN2 neurons in controlling temperature preference rhythm in flies during daytime (ZT1-12; Kaneko et al., 2012). AC neurons send extensive projections toward the lateral protocerebrum, a brain region that has recently been identified as an important center for processing thermal signals (Frank et al., 2015; Liu et al., 2015). AC neurons probably send thermosensory signals to higher centers in the fly brain, thus eliciting temperature-dependent behavior in flies.
In two recent reports, including one from our group, dTRPA1 expressed primarily in non-circadian neurons has been implicated in mediating the afternoon peak of activity under seminatural conditions (Das et al., 2015; Green et al., 2015). Specifically, we showed that dTRPA1SH+ cells that do not overlap with the cry-GAL4 driver are needed for the A-peak and that increased electrical activity of these neurons can enhance the amplitude of the A-peak. Our former study also showed that the A-peak can be elicited independently by a short pulse (1 h) of high temperature (greater than 29 °C), and this response is dTRPA1 mediated. This is in contrast to the behaviour elicited by these neurons if warm temperatures prevail over a 12-h duration, whereupon flies inhibit activity, suggesting that flies use different tactics to deal with a similar environmental stressor present for a brief versus a long duration. This is not surprising since the presence of very warm temperatures for a brief duration can probably be overcome by locomoting to escape that stressful environment, whereas the presence of warm cues for a longer duration could best be dealt with by minimizing locomotion and preventing desiccation. While this article was under review, another group also reported the involvement of dTRPA1 in mediating siesta under 20:29 °C temperature cycles and attributed it to reduced sleep during siesta (Roessingh et al., 2015).
We also observe that light influences dTRPA1-dependent behavioral phenotypes. Under LD, when temperatures are in the preferred range (<29 °C), dTRPA1 null mutants are able to exhibit bimodal activity/rest patterns similar to controls and under warm temperatures show phasing defects in M and E activity bouts. Under DD/TC, the M and E peaks are not significantly affected in the null mutants, whereas they are unable to suppress mid-day activity. In contrast, under LL/TC, the same mutants are able to suppress mid-day activity but show abnormal phasing of E-peak. In summary, our results suggest a crucial role for dTRPA1 receptor–mediated signaling primarily in non-circadian neurons in modifying different aspects of rhythmic behavior of flies in a temperature-dependent manner.
Footnotes
Acknowledgements
We thank other members of Holmes and Vasu labs for comments and suggestions and 2 anonymous reviewers for suggesting several improvements to a previous version of this article. We are grateful to Mark Rosenzweig for suggestions during initial experiments. We also thank Rajanna and Muniraju for assistance. This work was supported by Ramanujan Fellowship (Department of Science and Technology, India) to V.S., Council of Scientific and Industrial Research (India) fellowship to A.D., and NIH R01s GM102965 and GM107405 to T.C.H. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
