Abstract
To maintain synchrony with the environment, circadian clocks use a wide range of cycling sensory cues that provide input to the clock (zeitgebers), including environmental temperature cycles (TCs). There is some knowledge about which clock neuronal groups are important for temperature synchronization, but we currently lack knowledge on the temperature receptors and their signaling pathways that feed temperature information to the (neuronal) clock. Since TRPA1 is a well-known thermosensor that functions in a range of temperature-related behaviors, and it is potentially expressed in clock neurons, we set out to test the putative role of TRPA1 in temperature synchronization of the circadian clock. We found that flies lacking TRPA1 are still able to synchronize their behavioral activity to TCs comparable to wild-type flies, both in 16°C : 25°C and 20°C : 29°C TCs. In addition, we found that flies lacking TRPA1 show higher activity levels during the middle of the warm phase of 20°C : 29°C TCs, and we show that this TRPA1-mediated repression of locomotor activity during the “siesta” is caused by a lack of sleep. Based on these data, we conclude that the TRPA1 channel is not required for temperature synchronization in this broad temperature range but instead is required to repress activity during the warm part of the day.
Introduction
Circadian clocks are considered to be adaptive since they allow organisms to optimize the timing of physiological and behavioral activity within the environment. To maintain synchrony with the environment, circadian clocks use a wide range of cycling sensory cues that provide input to the clock, so-called zeitgebers. Light is considered to be the primary zeitgeber. However, the circadian clock is sensitive to environmental temperature cycles (TCs), in organisms ranging from bacteria to mammals (Yoshida et al., 2009; Wheeler et al., 1993; Glaser and Stanewsky, 2005; Lahiri et al., 2005; Refinetti, 2010; Aschoff and Tokura, 1986). This temperature sensitivity of the Drosophila melanogaster clock becomes apparent from the fact that flies can entrain their locomotor activity to TCs with an amplitude of just 3°C (Wheeler et al., 1993).
The molecular circadian oscillator in Drosophila is a negative translational feedback loop, in which transcription factors CLOCK (CLK) and CYCLE (CYC) form a dimer and bind E-boxes in the promoter regions of the genes period (per) and timeless (tim). The generated PERIOD (PER) and TIMELESS (TIM) proteins form a dimer and enter the nucleus to inhibit per and tim transcription by inhibiting CLK/CYC activity. The temporal delay between the activation and inhibition of per and tim transcription causes the oscillating rhythm of about 24 h. This molecular oscillator stays in synchrony with the environment because it resets in response to zeitgebers (e.g., Peschel and Helfrich-Förster, 2011).
The Drosophila brain contains ~150 clock neurons that exhibit these molecular oscillations in PER and TIM. They are divided in groups based on their anatomical location: the small and large ventral lateral neurons (s-LNv and l-LNv), the dorsal lateral neurons (LNd), the dorsal neurons 1-3 (DN1, DN2, DN3), and the lateral posterior neurons (LPN; e.g., Peschel and Helfrich-Förster, 2011). The function of the different clock neurons in temperature entrainment has been investigated either by genetic ablation of or by restoring clock function in specific neurons and measuring the resulting ability to synchronize to TCs on a molecular or behavioral level. Several studies revealed that in adult flies, the LNvs (Busza et al., 2007), DN1s (Zhang et al., 2010), DN2s (Busza et al., 2007), and LPNs (Busza et al., 2007; Yoshii et al., 2009) seem to have prominent functions in temperature synchronization. Work from our laboratory showed that a functional clock in the LNvs is sufficient for synchronization to colder TCs (16°C : 25°C), while a functional clock in the LNds and DNs is sufficient for synchronization to warmer TCs (20°C : 29°C; Gentile et al., 2013). In addition, we know that the blue light photoreceptor CRYPTOCHROME (CRY) antagonizes temperature synchronization (Gentile et al., 2013), which explains why CRY-negative (dorsal) neurons have a higher sensitivity to temperature than light (Yoshii et al., 2010).
Although we have some ideas about which subsets of the clock neurons are important for temperature synchronization, we are currently lacking knowledge on the temperature receptors and their signaling pathways that feed temperature information to the (neuronal) clock. In contrast to temperature input pathways, light input pathways to the clock are well characterized. Light resetting of the clock is primarily mediated by CRY, which acts cell autonomously in a subset of clock neurons to degrade TIM in a light-dependent manner (Emery et al., 1998; Stanewsky et al., 1998; Busza et al., 2004). In addition, the clock receives light input from peripheral photoreceptors (Helfrich-Förster et al., 2001). We hypothesize that temperature synchronization of the circadian clock is also mediated via multiple pathways, including both input from peripheral sensory organs (Sehadova et al., 2009) and cell autonomous mechanisms that mainly synchronize peripheral clocks (Glaser and Stanewsky, 2005).
Transient receptor potential (TRP) channels are nonselective cation channels associated with different sensory modalities, including thermosensation, and are therefore good candidates for acting as circadian temperature receptors (Fowler and Montell, 2013). Indeed, we recently showed that a TRP channel encoded by the pyrexia gene is required for temperature entrainment to relatively cold (16°C : 20°C) but not warm (20°C : 25°C and 25°C : 29°C) TCs (Wolfgang et al., 2013). The TRPA1-encoding gene has been implicated in larval thermotaxis (Kwon et al., 2008), adult temperature preference behavior (Hamada et al., 2008), and synchronization of locomotor behavior to TCs (Lee and Montell, 2013; Lee, 2013). The thermal threshold for TRPA1 activation when ectopically expressed in oocytes is 24°C to 29°C (Viswanath et al., 2003). At lower temperatures, indirect activation of TRPA1 has been shown in larvae, where TRPA1 acts downstream of Gq and phospholipase C (PLC) in the discrimination between 18°C and higher suboptimal temperatures (19-24°C; Kwon et al., 2008). Recently, it has been proposed that both ways of TRPA1 activation depend on a mechanical gating mechanism, in which either high temperatures above 25°C, or the PLC-dependent hydrolysis of PIP2 at lower temperatures generate cell membrane deformations that result in opening of TRPA1 by mechanical force (Liu and Montell, 2015).
Different labs have studied TRPA1 expression patterns in the brain, and in clock neurons specifically, either by immunocytochemistry with αTRPA1 antibody or by using trpA1-gal4 lines driving expression of a reporter. The αTRPA1 antibody specifically stains the temperature-sensitive anterior cell (AC) neurons (Hamada et al., 2008). Several trpA1-gal4 lines are expressed in AC neurons too, but they show broader expression patterns including weak expression in some clock neurons. Lee and Montell showed that the GAL4 knock-in trpA1GAL4 is expressed in the fifth s-LNv, 3 LNds, 2-3 DN1s, 1 DN2, 1 DN3, and all 3 LPNs (Lee and Montell, 2013; Lee, 2013). This is complemented by a recent study showing that the trpA1 promoter fusion trpA1SH-gal4 is weakly expressed in the fifth s-LNv, 3 LNds, and 1 DN1a (Yoshii et al., 2015). However, since the αTRPA1 antibody does not stain any clock neurons, it remains an open question if TRPA1 is expressed in clock neurons or not.
Here we performed the behavioral characterization of 2 trpA1 null alleles that lack all 4 known isoforms of TRPA1 (Kwon et al., 2008; Zhong et al., 2012) to test the putative role of TRPA1 in temperature entrainment (Lee and Montell, 2013). To distinguish between direct or indirect activation of TRPA1, we tested the ability of trpA1 loss-of-function mutants to synchronize to colder (16°C : 25°C) and warmer (20°C : 29°C) TCs, that fall below and above the activation threshold, respectively. We found that flies lacking TRPA1 are still able to synchronize their behavioral activity to cold and warm TCs comparable to wild-type flies. In addition, we found that loss of function of TRPA1 results in higher activity levels during the afternoon of warmer TCs and that this effect was caused by a lack of sleep. We conclude that the TRPA1 channel is not required for temperature synchronization but instead is required to repress activity during the warm part of the day.
Materials and Methods
Fly Strains and Husbandry
Fly strains and crosses were reared on standard yeast-containing fly food and were entrained to a 12 : 12 h LD cycle at constant 25°C and 60% humidity. All strains that were used carried the timeless allele ls-tim (Sandrelli et al., 2007; Peschel et al., 2006). y w; ls-tim flies were used as wild-type control strain. trpA11 flies were obtained from Bloomington (BL26504) and crossed into the y w; ls-tim background to exchange the s-tim allele that was present in the stock. trpA1W903* and Df(3L)ED4415 flies (gifts from Dan Tracey) already carried the ls-tim allele and the perSLIH mutation (Hamblen et al., 1998), which means these stocks have a different X-chromosome compared with y w; ls-tim and y w; ls-tim; trpA11 flies. However, for all behavioral and sleep experiments involving trpA1W903* and Df(3L)ED4415 (except those shown in Fig. 4), perSLIH-containing males were crossed to females homozygous for y w, and F1 males were tested. Therefore, all test and control flies carry the y w X-chromosome.
The trpA1 loss-of-function alleles trpA11 (Kwon et al., 2008) and trpA1W903* (Zhong et al., 2012) carry a deletion of the sixth trans membrane domain and a point mutation creating a stop codon in the fourth trans membrane domain, respectively, and are predicted to lead to a nonfunctional TRPA1 channel encoded by all possible transcripts. Df(3L)ED4415 is a 210-kb deficiency removing trpA1 (Ryder et al., 2007).
Locomotor Activity Recording
1 to 7 day old male flies were loaded in glass tubes, which contained food (2% agarose, 4% sucrose) on one side and were plugged with cotton wool on the other side. The glass tubes were loaded in Drosophila activity monitors (DAM2 system; Trikinetics, Waltham, MA) to record locomotor activity over the course of the behavioral experiment. During activity monitoring, environmental conditions were programmed in light- and temperature-controlled incubators (Percival). Flies were exposed to 2 different TCs, spanning cold and warm temperature ranges of 16°C : 25°C and 20°C : 29°C, respectively. Light dark (LD) and TC conditions were in all cases programmed as rectangular cycles of 12 : 12 h. Light conditions changed immediately, while the temperature inside the incubator reached the new set point within 25 to 30 min when cooling down or heating up, respectively. TCs were always in constant darkness (DD). Humidity was increased by placing a water bucket inside the incubator during experiments. During TCs, relative humidity cycled in synchrony with the TCs with 65% to 95% or 55% to 95% during 16°C : 25°C and 20°C : 29°C TCs, respectively. In constant conditions, relative humidity was 75% to 90%.
The cold TC experiment started with 3 to 5 d of LD cycles at constant temperature (22.5°C or 25°C), after which the lights were switched off for the remainder of the experiment. LD conditions were followed by 8 d of 16°C : 25°C TCs, which were 5 h advanced compared with the previous LD cycle (by shortening the final dark phase of LD by 5 h). After 8 d, flies were released to constant conditions (22.5°C or 25°C in DD). For the warm TC experiment, LD cycles at constant temperature (22.5°C or 20°C) were followed by 8 d of 20°C : 29°C TCs, which were 8 h advanced compared with the previous LD cycle (by shortening the final dark phase of LD by 8 h) and released to constant conditions (22.5°C or 20°C in DD). The different constant temperatures during the initial LD and final DD portion of the experiments had no obvious effect on the behavior, and therefore the data were pooled to generate the DD histograms showing behavior in constant conditions (Figs. 1 and 2; Suppl. Fig. S1).
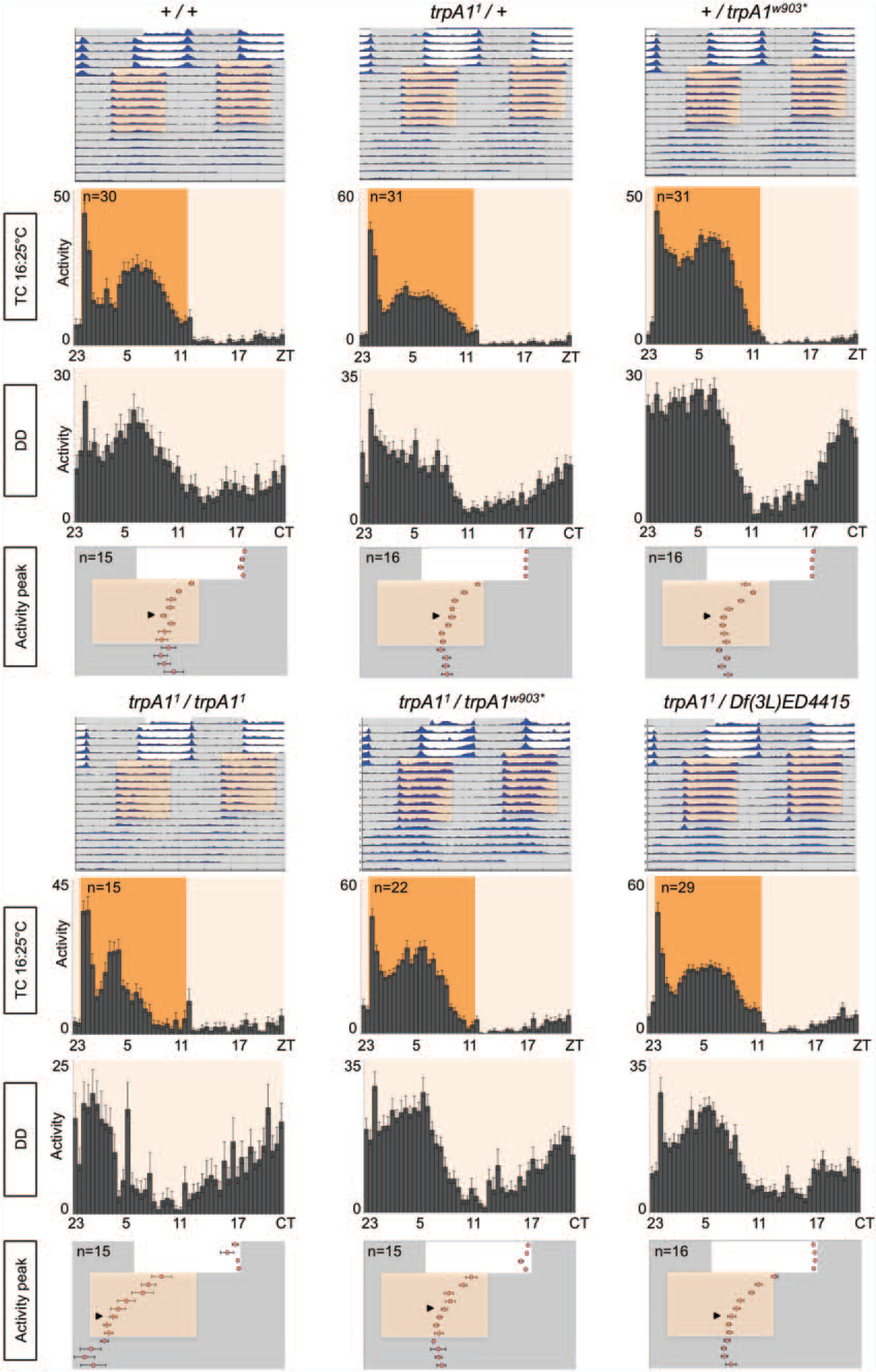
trpA1 mutants show normal behavior in 16°C : 25°C temperature cycles (TCs). Representative double-plotted actograms (top) show average locomotor activity of indicated genotypes during the cold temperature cycle experiment. Five days of LD cycles (at constant 22.5°C or 25°C) were followed by 8 d of 16°C : 25°C TCs that were 5 h advanced compared with previous LD. Average activity per genotype is plotted in blue. Histograms show the average locomotor activity during the last 4 d in TCs and the first 3 d in DD conditions. Bottom graphs show the timing (phase) of the main activity peak determined for each individual fly during the entire experiment. Red dots represent the average timing of the main activity peak. Arrowheads indicate the first day of stable phase during TCs (see the Materials and Methods section and Sehadova et al., 2009). White and gray bar or area colors indicate lights-on or lights-off, respectively. Orange/pink background shadings indicate the warm/cold phase of the TC, respectively. n numbers given for TC histograms also apply for DD histograms. Error bars indicate SEM.
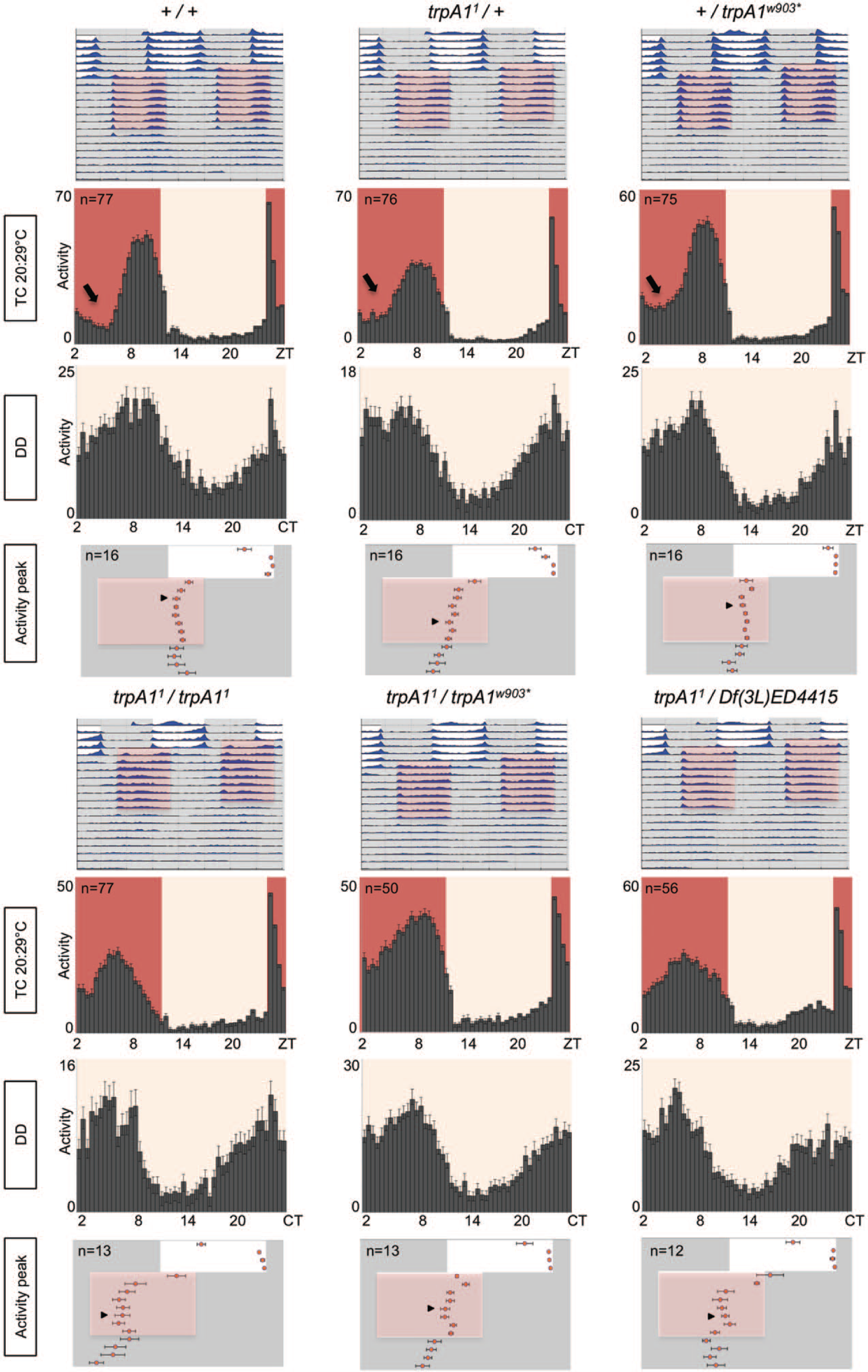
trpA1 mutants synchronize their behavior to 20°C : 29°C temperature cycles (TCs). Representative double-plotted actograms (top) show average locomotor activity of indicated genotypes during the warm temperature cycle experiment. Five days of LD cycles (at constant 22.5°C or 20°C) were followed by 8 d of 20°C : 29°C TCs that were 8 h advanced compared with previous LD. Average activity per genotype is plotted in blue. Histograms show the average locomotor activity during the last 4 d in TCs and the first 3 d in DD conditions. Bottom graphs show the timing (phase) of the main activity peak as described in Figure 2. White and gray bar or area colors indicate lights-on or lights-off, respectively. Red/pink background shadings indicate the warm/cold phase of the TC, respectively. Black arrow indicates “siesta.” n numbers given for TC histograms also apply for DD histograms. Error bars indicate SEM.
Data Collection and Analysis
Activity data were recorded every 5 min or every minute and were analyzed as total activity counts per 30 min. Actograms of locomotor activity data were generated in Matlab, using the Fly toolbox (Levine et al., 2002) and customized scripts (courtesy of L. Garcia, Polygonal Tree, London, UK). After visual inspection of individual actograms, the flies that did not survive all TC days were excluded from analysis. Microsoft Excel was used for the generation of histograms (average of last 4 d in TCs) and quantification of the timing of the main activity peak. The time of highest activity (before activity levels were going down) was manually selected as the time of the main activity peak for individual flies on each day for the entire experiment (Sehadova et al., 2009). The ZT of the activity peak was assigned according to ZT times of the initial LD cycle. If no clear activity peak could be detected, this was taken as a missing data point. Individuals with missing data points were excluded from the phase analysis but still included in the analysis of synchronization time. The number of days required for stable resynchronization to the TC was manually assigned to each individual. A fly was considered to have reached synchronization on the first day the timing of the activity peak reached its final phase. Activity profiles (line graphs) and quantification were generated using Excel. Activity was an average of the last 4 d of the TCs, like for the activity histograms. Siesta activity levels were measured during ZT 2-5.5. All bar graphs and statistical analysis were generated using GraphPad Prism 6.
Data from the behavioral experiments were used for sleep analysis. As previously described, activity data were recorded every minute (using the DAM2 system) and sleep was defined as a period of inactivity of at least 5 min (Shaw et al., 2000). Graphs with sleep profiles were generated using pySolo (Gilestro and Cirelli, 2009). Quantification of the sleep data was performed using a customized script in Excel (courtesy of Kofan Chen). The amount of sleep was measured in minutes per day. Sleep measures were an average of the last 4 d of the TCs, like for activity histograms, or of the last 3 d of LD cycles. Siesta sleep was measured during ZT 3-7.5. All bar graphs and statistical analysis were generated using GraphPad Prism 6.
Results
trpA1 Loss-of-Function Mutants Show Normal Behavior in 16°C : 25°C TCs
To investigate the potential role of the TRPA1 channel in regulating locomotor activity of adult Drosophila during LD and TCs, we exposed control flies and trpA1 loss-of-function mutants to 12 h : 12 h LD cycles at constant temperature followed by 12 h : 12 h TCs (16°C : 25°C) in DD, which were phase advanced by 5 h compared with the initial LD cycle. After the TCs, flies were released to DD at constant temperature (22.5°C or 25°C). During the initial LD cycles, wild-type flies showed bimodal crepuscular behavior, with the typical increase of locomotor activity before the light/dark transitions in the morning and evening (Wheeler et al., 1993; Fig. 1). Exposure to the phase-advanced TCs resulted in a phase advance of the evening activity peak, which after 4 to 5 d occurred roughly in the middle of the warm phase (Fig. 1; Yoshii et al., 2009; Gentile et al., 2013; Lee, 2013). After the final release into constant conditions, wild-type flies maintained the activity phase established during the TCs, indicating that their circadian clock had been stably synchronized to the fluctuating temperatures (Fig. 1; Gentile et al., 2013).
Flies lacking the TRPA1 channel showed similar behavior to wild-type flies under LD, TC, and constant conditions (Fig. 1). trpA11/trpA11 and trpA11/Df(3L)ED4415 flies needed 1 extra day to synchronize their activity to the phase-advanced TCs compared with control flies (Fig. 1; Suppl. Fig. S1), but this difference was not significant (Fig. 3A). Moreover, another combination of TRPA1 null alleles (trpA11/trpA1w903*) was indistinguishable from wild type (Figs. 1, 3A). Homozygous trpA11 flies also exhibited an earlier phase of the main activity peak at the end of the TCs compared with wild-type flies (Figs. 1, 3B). The activity peak is very sharp and occurs about 3 h after the activity that coincides with the temperature step up. This behavior is not observed in any of the other genotypes (including trpA11/Df(3L)ED4415), which all show a broader activity peak occurring 3 to 5 h later than in homozygous trpA11 flies (Figs. 1, 3B). We therefore conclude that the subtle behavioral differences associated with the trpA11 allele (phase advance and slower resynchronization) are not caused by loss of TRPA1 function.
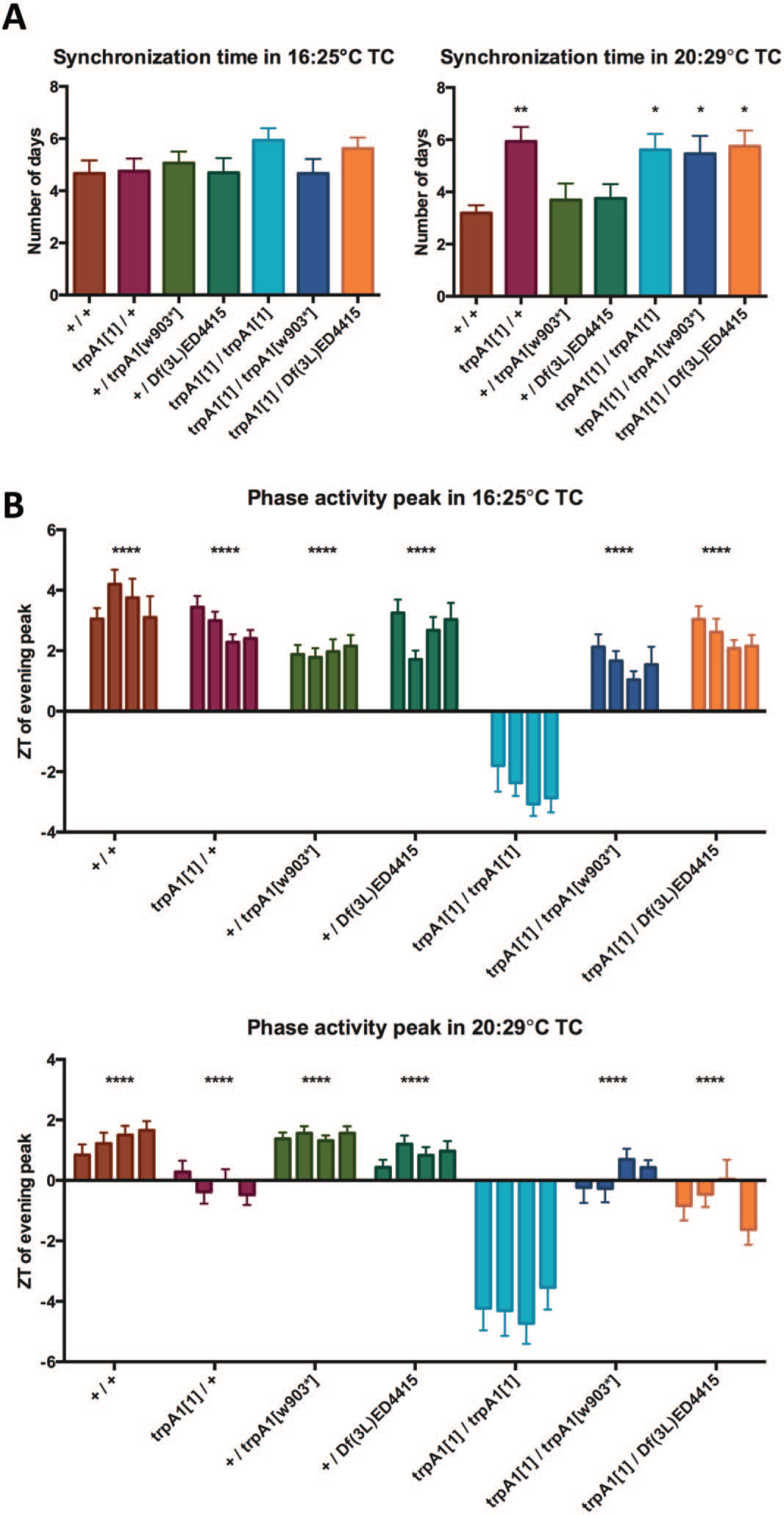
Quantification of synchronization time and phase of the main activity peak during cold and warm temperature cycles (TCs). (A) Bar graphs show the average number of days required for stable resynchronization for indicated genotypes. Error bars indicate SEM. *Significantly different from wild type when groups were compared in 1-way analysis of variance (ANOVA) and Dunnett’s multiple comparison test (*p < 0.05, **p < 0.01). n numbers are the same as in Figures 2, 3, and Supplementary Figure S1 and are between 12 and 16 flies per genotype. (B) Bar graphs show the average phase of the activity peak on the last 4 d (days 5-8) of the TCs for indicated genotypes. Error bars indicate SEM. *Significantly different from trpA11/trpA11 when genotype means were compared in 2-way ANOVA (Time × Genotype) and Dunnett’s multiple comparison test (****p < 0.0001). n numbers are between 10 and 16 flies per genotype.
trpA1 Loss-of-Function Mutants Synchronize Their Behavior to 20°C : 29°C TCs
Because the thermal threshold for TRPA1 activation is ~27°C (Viswanath et al., 2003), we also performed behavioral characterization during warmer TCs (20°C : 29°C). Control flies and trpA1 loss-of-function mutants were exposed to a similar resynchronization paradigm as described above for 16°C : 25°C TCs, with the exception that the TC was phase advanced by 8 h compared with the initial LD regime. In these conditions, wild-type flies required only 3 d to synchronize to the TCs, and their main activity peak was confined to the second part of the warm phase (Figs. 2, 3A) (Gentile et al., 2013). Flies lacking the TRPA1 channel again showed very similar behavior to wild-type flies and synchronized their activity to the phase-advanced TCs (Fig. 2). Quantifying the number of days required for stable resynchronization revealed that all genotypes containing the trpA11 allele need about 2 more days compared with wild type and trpA1w903*/+ as well as Df(3L)ED4415/+ controls (Figs. 2, 3; Suppl. Fig. S1). Since trpA11/+ flies require the same amount of time to synchronize as the 3 trpA1 loss-of-function genotypes, we conclude that TRPA1 is not required for synchronization to 20°C : 29°C TCs (Figs. 2, 3A).
Similar to what we observed at the colder TCs, homozygous trpA11/trpA11 flies also showed an advanced phase of the main activity peak at the end of the TCs (Figs. 2, 3B). Again, this drastic phase advance was not observed in any of the other genotypes (including trpA11/trpA1w903* loss-of-function flies), indicating that TRPA1 is not required for establishing a normal phase during entrainment to TCs (Figs. 2, 3B; Suppl. Fig. S1).
To further demonstrate that flies lacking the TRPA1 channel can properly synchronize to the shifted TCs, we tested trpA1 mutant flies that also carried the period-lengthening allele perSLIH (Hamblen et al., 1998). If TRPA1 were important for temperature entrainment of the circadian clock, one would expect the flies to free-run with a long period during TC conditions. We observed that flies with any combination of trpA11, trpA1w903*, and Df(3L)ED4415 alleles in the perSLIH background confined their activity to the warm phase of the TC, whereas they showed the expected long-period rhythms during constant conditions (Fig. 4), further indicating that that TRPA1 is not required for temperature entrainment to 20°C : 29°C TCs. Because perSLIH is a temperature-compensation defective period allele (dubbed
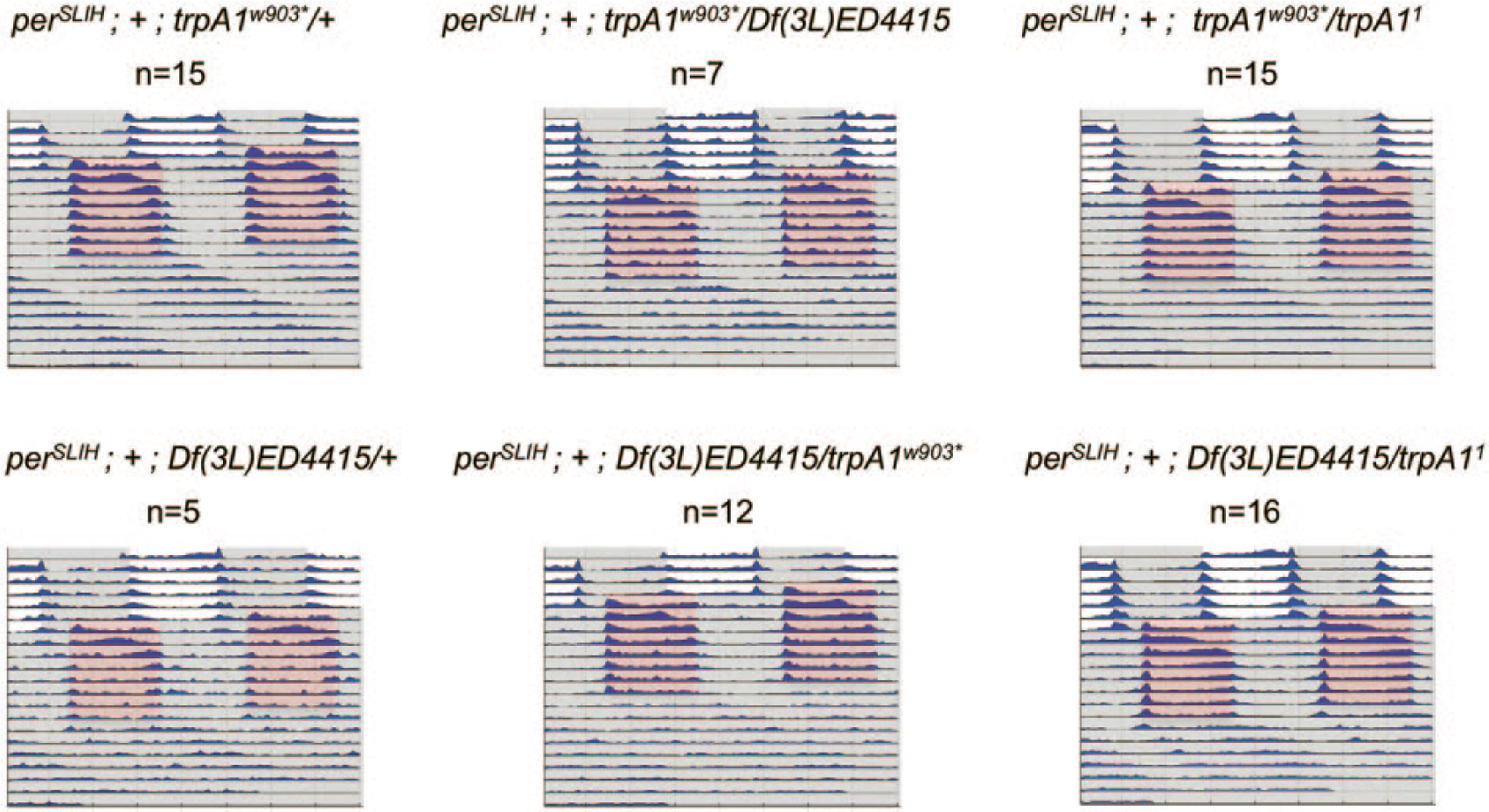
trpA1 mutants that also carry the perSLIH mutation show normal temperature entrainment behavior in 20°C : 29°C temperature cycles (TCs). Representative double-plotted actograms show average locomotor activity of indicated genotypes during the warm temperature cycle protocol. Five days of LD cycles were followed by 8 d of 20°C : 29°C TCs that were 8 h advanced compared with previous LD. Average activity per genotype is plotted in blue. White/gray and red/gray background shadings indicate the light/dark phase and the warm/cold phase, respectively.
TRPA1 Is Required for Siesta Behavior in 20°C : 29°C TCs
We observed that in genotypes lacking a functional copy of trpA1, activity during the middle of the warm phase never dropped to trough levels as it typically does in wild-type flies (“siesta”; Fig. 2, black arrow in histograms pointing to wild-type siesta). To quantify this effect, we determined the total activity levels during times when wild-type flies showed very little activity (ZT2 to ZT5.5) during the last 4 d of the TCs (Fig. 5A). Flies mutant for trpA1 (trpA11/trpA11, trpA11/trpA1w903*, and trpA11/Df(3L)ED4415) indeed had significantly higher activity levels compared with wild-type and genetic control flies during the normal trough times (Figs. 5B; Suppl. Fig. S2). This suggests that TRPA1 is required for repressing locomotor activity in the middle of the warm phase of warmer (20°C : 29°C) TCs.
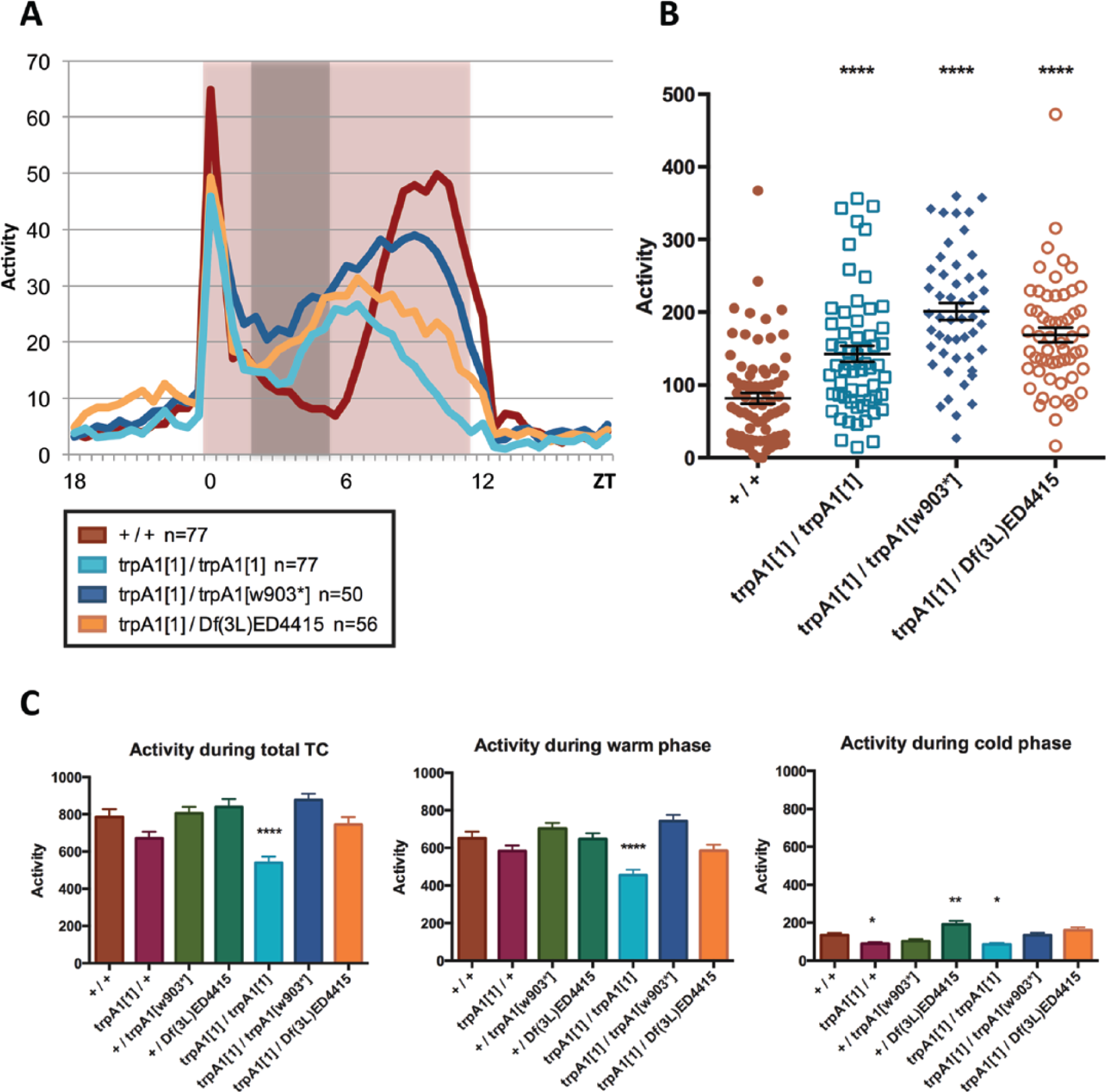
trpA1 mutants have higher activity levels during siesta in 20°C : 29°C temperature cycles (TCs). (A) Average activity profiles of indicated genotypes during the last 4 d in 20°C : 29 °C TCs (overlay of TC histograms in Fig. 3). Red/white background shadings indicate warm/cold phases. Gray bar (ZT 2-5.5) indicates part of the cycle that is used to quantify activity levels during siesta. (B) Total activity during siesta period (gray bar: ZT 2-5.5) for indicated genotypes. Dots represent individual flies. Error bars represent SEM. *Significantly different from wild type when all groups (mutant and control genotypes) were compared with each other in one-way analysis of variance (ANOVA) and Tukey’s multiple comparisons test (****p < 0.0001). (C) Total activity during the complete TC, warm phase, and cold phase. *Significantly different from wild type when groups were compared in one-way ANOVA and Dunnett’s multiple comparison test (*p < 0.05, **p < 0.01, ****p < 0.0001). n numbers apply to all figure panels: +/+ n = 77, trpA11/+ n = 76, +/trpA1w903* n = 75, +/Df(3L)ED4415 n = 74, trpA11/trpA11 n = 77, trpA11/trpA1w903* n = 50, trpA11/Df(3L)ED4415 n = 56.
To exclude the possibility that trpA1 mutants are more active in general, we quantified total activity during the complete TC and the warm and cold phase separately (Fig. 5C). Total activity levels of trpA1 mutants and genetic controls were not significantly different from the wild-type control, except for the trpA11/trpA11 mutant, which showed lower activity levels. This is consistent with the observation that trpA11/trpA11 mutant flies showed an earlier and reduced main activity peak during TCs (Figs. 2, 5). All genotypes showed very little activity during the cold phase, resulting in total activity levels reflecting the activity levels during the warm phase.
We tried to quantify siesta behavior under 16°C : 25°C TCs as well. However, under these conditions, the evening activity peak occurred in the middle of the warm phase instead of the second half, which results in the lack of a clear siesta. This is consistent with the fact that the siesta during LD cycles is more prominent at higher temperatures (Majercak et al., 1999). We did quantify total activity during the complete TC and the warm and cold phase separately (Suppl. Fig. S3). During 16°C : 25°C TCs, total activity levels of trpA1 mutants and genetic controls were not significantly different from the wild-type control. This, together with visual observation of the histograms (Fig. 1), indicates that the general activity pattern under colder (16°C : 25°C) TCs is not affected by the loss of TRPA1. It therefore seems that the increased siesta activity levels are specific to warmer (20°C : 29°C) TCs.
TRPA1 Promotes Sleep during Warm Temperatures
Inactivity in Drosophila shares essential features of mammalian sleep, including a specific body posture, increased arousal threshold, and homeostatic regulation (Hendricks et al., 2000; Shaw et al., 2000). It was shown that sleep/wakefulness states in Drosophila coincide with a difference in general brain electrical activity and spontaneous activity and reactivity of neurons, as is true for mammals (Nitz et al., 2002; Bushey et al., 2015).
In Drosophila, sleep is defined as a period of inactivity of 5 min or longer, because flies that have been inactive for this amount of time show an increased arousal threshold (Shaw et al., 2000). When kept in LD conditions, flies sleep consistently throughout the night and less during the day, with troughs in sleep in the morning and evening (Shaw et al., 2000). It has recently been shown that low locomotor activity levels observed during siesta in LD cycles reflect a sleeplike state (Cao and Edery, 2015). To investigate if alterations in wakefulness could explain the increased siesta activity of trpA1 mutant flies, we determined the amount of sleep during 20°C : 29°C TCs for wild-type and trpA1 mutant flies.
In wild-type flies, sleep patterns during TCs closely resemble those during LD cycles, with wake periods confined around the respective environmental transitions (Fig. 6A, B; Suppl. Fig. S4A; Shaw et al., 2000). This indicates that low locomotor activity levels during the middle of the warm phase of the TC are comparable to the siesta observed under LD conditions, and this behavior therefore might reflect a sleeplike state (Cao and Edery, 2015). During LD, trpA1 mutants overall showed no consistent changes of total sleep compared with controls (Fig. 6B, C). Strikingly, whereas total sleep time was not or only weakly affected, sleep time in the middle of the warm phase was significantly reduced in all 3 trpA1 loss-of-function mutant genotypes compared with y w controls (p < 0.0001; Fig. 6A, C; Suppl. Fig. S4B, C). On average, the flies from the 3 trpA1 mutant genotypes slept about 1.5 h less during the siesta compared with controls (Fig. 6C). This indicates that the higher activity levels observed in trpA1 mutants during the warm part of 20°C : 29°C TCs are caused by a repression of sleep.
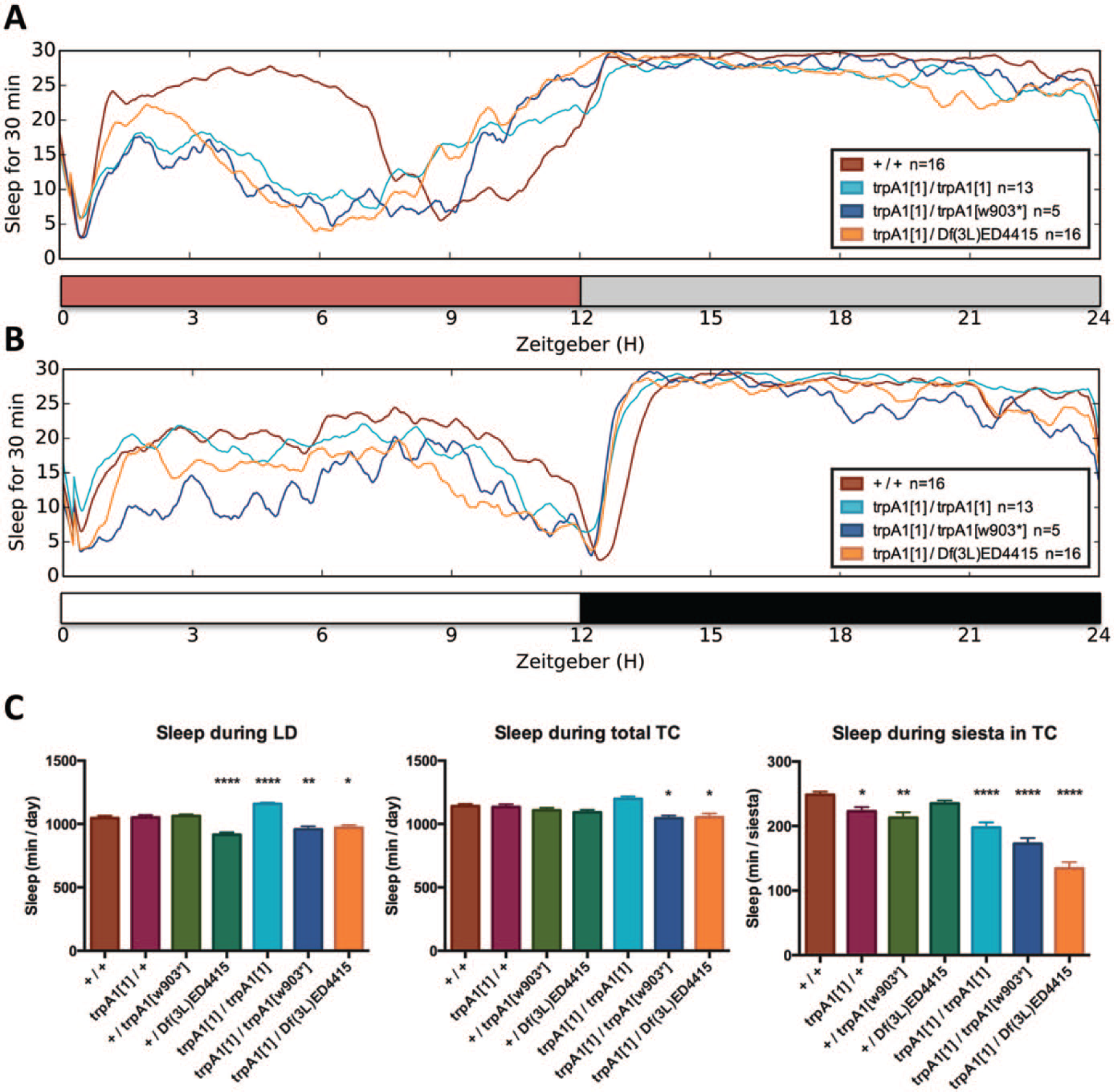
trpA1 mutants sleep less during the warm phase in 20 °C:29 °C temperature cycles (TCs). (A) Average sleep profiles of indicated genotypes during the last 4 d in 20 °C:29 °C TCs show the amount of sleep (in minutes) during each 30-min interval of the day. Red/gray bar indicates warm/cold phase (see also Suppl. Fig. S4A-C). (B) Average sleep profiles of indicated genotypes during the last 3 d in LD cycles show the amount of sleep during each 30-min interval. White/black bar indicates light/dark phase. (C) Quantification of average amount of sleep in minutes per day during the last 4 d of LD and TCs and in minutes per siesta during ZT3-7.5 of the last 4 d of TCs. *Significantly different from wild type when groups were compared in 1-way analysis of variance and Dunnett’s multiple comparison test (*p < 0.05, **p < 0.01, ****p < 0.0001). n numbers apply to all 3 panels: +/+ n = 48, trpA11/+ n = 46, +/trpA1w903* n = 43, +/Df(3L)ED4415 n = 45, trpA11/trpA11 n = 44, trpA11/trpA1w903* n = 30, trpA11/Df(3L)ED4415 n = 32.
Discussion
We performed behavioral characterization of 2 trpA1 null alleles and found that TRPA1 is not required for behavioral synchronization to TCs (16°C : 25°C and 20°C : 29°C in DD; Figs. 1-4). Based on these data, we conclude that the TRPA1 channel is not required for temperature synchronization in this broad temperature range. Our results are therefore in conflict with a previous study that suggested a role for TRPA1 in temperature entrainment (Lee and Montell, 2013). In this study, temperature entrainment of 2 trpA1 loss-of-function mutants (trpA11 and trpA1GAL4) was analyzed in 18°C : 29°C and 16°C : 25°C TCs, so similar to the conditions used here (although in the warmer TC, temperature was high for 18 h and low for 6 h). In both conditions, only very subtle temperature entrainment phenotypes were observed (e.g., in the 18°C : 29°C TCs, it took the mutants 1 d longer to synchronize) in addition to a slight phase advance of the temperature-entrained activity peak. Stronger phenotypes could be observed only after genetic ablation of >50% of the clock neurons combined with homozygosity for the trpA11 mutation. Importantly, Lee and Montell (2013) did not analyze heterozygous trpA11 flies, nor did they test the 2 loss-of-function alleles in trans or over a deficiency of the locus, as we did in our study. Since we observed behavioral abnormalities in homozygous (phase advance) and heterozygous (slower resynchronization) trpA11 mutant animals, but not in the proper genetic controls, the most likely explanation for the conflicting results is the presence of genetic background factors in the trpA11 mutant stock. It is also noteworthy that the original trpA11 stock (used by Lee and Montell, 2013) carries the s-tim allele, whereas we outcrossed all the lines used in our study to the ls-tim background to rule out any potential influence of different TIM isoforms on temperature-dependent trpA1 phenotypes. The s-tim/ls-tim polymorphism is responsible for different sensitivity of the circadian clock to light (Sandrelli et al., 2007; Peschel et al., 2006), and at this point, we cannot rule out that it also influences temperature entrainment.
Although normal for temperature entrainment, we could show that trpA1 mutant flies show higher activity levels during siesta of warm (20°C : 29°C) TCs (Figs. 2, 5), suggesting that TRPA1 is required for repressing locomotor activity during unfavorable warm conditions. By performing sleep analysis, we were able to show that this repression of locomotor activity is caused by a lack of sleep (Fig. 6; Suppl. Fig. S4). Flies that are entrained to LD cycles show an advanced evening peak of activity in cooler temperatures and a delayed evening peak of activity in warmer temperatures (Majercak et al., 1999). Delay of the evening peak results in siesta behavior, which is thought to be an adaptive response, for example, to prevent desiccation during the warmer parts of the day. It has been shown that the thermal regulation of siesta is (at least partly) mediated via temperature-sensitive splicing of a 3′-terminal intron of the clock gene period (Majercak et al., 1999; Low et al., 2008, 2012). At warmer temperatures, the intron is not efficiently removed, which decreases the accumulation of mature per mRNA, which then leads to a delay in evening activity and an increased siesta.
With respect to our findings, this temperature-sensitive splicing of per mRNA is an interesting mechanism for 2 reasons. First, per splicing efficiency has been shown to affect arousal state, where inefficient splicing at warmer temperatures causes an increase in sleep during the day but not the night (Cao and Edery, 2015). This fits with our finding that trpA1 mutants sleep less during the warm portion of the day (Fig. 6). Second, it has been suggested that PLC may be the temperature sensor that acts upstream of per mRNA splicing (Collins et al., 2004; Majercak et al., 2004). Since TRPA1 has been reported to function downstream of PLC in temperature discrimination in larvae (Kwon et al., 2008), these studies highlight the possibility that phospholipase C and TRPA1 could act together in the regulation of per mRNA splicing that in turn regulates afternoon sleep in response to temperature.
Recently, a function for TRPA1 in regulating daytime activity at very warm temperatures has been reported (Green et al., 2015). Wild-type flies kept in natural or semi-natural conditions that closely mimic the natural light and temperature profiles of very hot summer days show an afternoon (A) activity peak in addition to the typical morning (M) and evening (E) activity peaks (Vanin et al., 2012; Green et al., 2015). The A peak is observed when the daily temperature reaches a maximum of 35°C, i.e., it is observed in 25°C : 35°C but not in 20°C : 30°C cycles in which both light and temperature change gradually following natural patterns (Green et al., 2015). Interestingly, this study revealed that the A peak in such simulated natural hot summer days (25°C : 35°C) depends on TRPA1, i.e., trpA11 mutants lack this A peak. While these findings seem to be at odds with the results reported here (TRPA1 is required for a siesta during warm TCs rather than an increase in activity), it is clear that the experimental conditions are quite different. While we study the function of TRPA1 in light-independent conditions (total darkness), Green et al. (2015) use changes of light and temperature. Moreover, it is difficult to compare the results between the studies, because the A peak is not observed in the temperature ranges where we identified a role for TRPA1 in repressing midday activity (20°C : 29°C). Therefore, it remains open how the function of TRPA1 in supporting A activity at very hot temperatures (35°C) is related to that of repressing midday activity at warm temperatures (29°C). Green et al. (2015) showed that for supporting A activity, TRPA1 is neither required in clock neurons (trpA1-RNAi driven with timeless-gal4 had no effect on the A-peak) nor in the temperature-sensitive AC neurons, which are known to regulate temperature preference behavior (Hamada et al., 2008; trpA1-RNAi driven with a trpA1-gal4 driver not expressed in the AC neurons still repressed the A peak). Future work will show which TRPA1-expressing neurons mediate siesta and A peak behavior and how the apparent opposing TRPA1-dependent effects on activity are generated.
Footnotes
Acknowledgements
We thank Dan Tracey and Fumika Hamada for fly stocks and sharing of unpublished results. We thank Kofan Chen for supplying the Excel script for sleep analysis. This work was supported by BBSRC grants BB/H001204 to R.S. and by the European Union FP7 Integrated Training Network INsecTIME.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
