Abstract
The suprachiasmatic nucleus (SCN) regulates daily rhythms in physiology and behavior. Previous studies suggest a critical role for neurons expressing vasoactive intestinal peptide (VIP) in coordinating rhythmicity and synchronization in the SCN. Here we examined the firing properties of VIP-expressing SCN neurons in acute brain slices. Active and passive membrane properties were measured in VIP and in non-VIP neurons during the day and at night. Current-clamp recordings revealed that both VIP and non-VIP neurons were spontaneously active, with higher firing rates during the day than at night. Average firing frequencies, however, were higher in VIP neurons (3.1 ± 0.2 Hz, day and 2.4 ± 0.2 Hz, night) than in non-VIP neurons (1.8 ± 0.2 Hz, day and 0.9 ± 0.2 Hz, night), both day and night. The waveforms of individual action potentials in VIP and non-VIP neurons were also distinct. Action potential durations (APD50) were shorter in VIP neurons (3.6 ± 0.1 ms, day and 2.9 ± 0.1 ms, night) than in non-VIP neurons (4.4 ± 0.3 ms, day and 3.5 ± 0.2 ms, night) throughout the light-dark cycle. In addition, afterhyperpolarization (AHP) amplitudes were larger in VIP neurons (21 ± 0.8 mV, day and 24.9 ± 0.9 mV, night) than in non-VIP neurons (17.2 ± 1.1 mV, day and 20.5 ± 1.2 mV, night) during the day and at night. Furthermore, significant day/night differences were observed in APD50 and AHP amplitudes in both VIP and non-VIP SCN neurons, consistent with rhythmic changes in ionic conductances that contribute to shaping the firing properties of both cell types. The higher day and night firing rates of VIP neurons likely contribute to synchronizing electrical activity in the SCN.
Keywords
The suprachiasmatic nucleus (SCN), a small, bilateral structure in the hypothalamus, contains a network of approximately 20,000 neurons that generate and synchronize circadian rhythms in physiology and behavior (Kalsbeek and Buijs, 2002; Colwell, 2011). In mammals, daily rhythms in the membrane properties of SCN neurons are observed. Input resistances and firing rates, for example, are reportedly higher during the day than at night (Inouye and Kawamura, 1979; Green and Gillette, 1982; Schaap et al., 1999; Pennartz et al., 2002). Although previous studies have shown that most SCN neurons can function as autonomous pacemakers (Welsh et al., 1995), the rhythmicity and synchrony of firing of isolated SCN neurons are reduced markedly compared to cells in an intact SCN network (Welsh et al., 1995; Herzog et al., 2004; Welsh et al., 2010), indicating that synaptic communication mediates the coherent, reliable, daily rhythms among SCN neurons. Previous studies have highlighted that between 60% and 90% of SCN neurons show circadian rhythms in electrical activity (Welsh et al., 1995; Herzog et al., 1997; Nakamura et al., 2002; Webb et al., 2009), including the vasopressinergic neurons (Schaap et al., 1999) but not the calbindin-positive neurons (Jobst and Allen, 2002). Because the intrinsic membrane properties of SCN neurons vary (Jiang et al., 1997; Pennartz et al., 1998; Kononenko and Dudek, 2004), we sought to characterize firing properties of
We chose to focus on neurons that synthesize the neuropeptide, vasoactive intestinal peptide (VIP). Located primarily in the ventral-lateral region of the SCN, VIP neurons receive direct retinal input and project to other neurons throughout the SCN (Romijn et al., 1997; An et al., 2012). These neurons release VIP in response to light in vivo and in a circadian pattern in vitro (Shinohara et al., 1995; Shinohara et al., 1998; Francl et al., 2010). VIP mRNA and protein levels in the SCN have also been shown to vary with time of day depending on age and ambient lighting conditions (Okamoto et al., 1991; Takeuchi et al., 1992; Shinohara et al., 1993; Fukuhara et al., 1994; Okamura et al., 1995; Ban et al., 1997; Kawakami et al., 1997; Kunst et al., 2015). Studies conducted on animals in which either VIP or the VIP receptor (VPAC2) was genetically eliminated demonstrated that up to 70% of neurons in the SCN become arrhythmic and desynchronized (Aton et al., 2005; Brown et al., 2007). Co-culture and stimulation studies have placed VIP as the major agent for circadian synchrony in the SCN (Brown et al., 2005; Maywood et al., 2011; Pauls et al., 2014). In addition, exogenous application of VIP has been shown to shift and entrain daily rhythms in the SCN (Watanabe et al., 2000; Reed et al., 2001; Reed et al., 2002; An et al., 2011; Kudo et al., 2013). Taken together, these observations suggest that daily release of VIP from a subset of SCN neurons coordinates circadian rhythms in the SCN.
We tested the hypothesis that the firing properties of VIP neurons are distinct from those of other neurons in the SCN. To identify VIP-expressing neurons in acute SCN slices, we took advantage of a knock-in mouse that expresses a fluorescent reporter only in VIP neurons. Targeted recordings of VIP neurons revealed that VIP neurons, on average, have significantly higher firing rates during the day and at night than non-VIP neurons.
Materials And Methods
All experiments were performed in accordance with the guidelines published in the National Institutes of Health Guide for the Care and Use of Laboratory Animals. The Animal Studies Committee at Washington University approved all procedures involving animals.
VIP-tdTomato mice were generated by crossing VIP-ires-Cre knock-in mice (stock 010908; Jackson Laboratory, Bar Harbor, ME) (Taniguchi et al., 2011) with Rosa-CAG-LSL-tdTomato-WPRE reporter mice (stock 007908; Jackson Laboratory) (Madisen et al., 2010) and maintained on the C57BL/6N background for at least 3 generations. These animals were group-housed on either a standard 12:12-h light:dark (LD) schedule (lights on at 0700 h and off at 1900 h) or a reversed LD schedule (lights on at 1900 h and off at 0700 h) and were given access to food and water ad libitum. In both cases, the ambient temperature was 22 °C and the humidity was 50%. In addition, “nighttime” animals were maintained in the reversed LD schedule for at least 14 days prior to the preparation of acute SCN slices. The light intensity for the facility with the standard LD schedule was 333 lux, and the light intensity for the facility with the reversed LD schedule was 667 lux.
All reagents were obtained from Sigma-Aldrich (St. Louis, MO) unless otherwise noted.
Immunohistochemistry
Adult (10-12 weeks) VIP-tdTomato knock-in male mice were anesthetized with 1.25% Avertin (2,2,2-tribromoethanol and tert-amyl alcohol in 0.9% NaCl; 0.025 mL/g body weight) and were transcardially perfused with ice-cold phosphate-buffered saline (PBS) followed by 4% paraformaldehyde in PBS. Brains were rapidly removed and placed in 30% sucrose for 2 days. Frozen coronal sections (30 µm) containing the SCN were cut on a cryostat and placed in a 12-well tissue culture dish. Following washing for 1 h in PBS at room temperature (RT), sections were incubated for 30 min at 37 °C in 3% Triton in PBS and subsequently blocked in PBS containing 10% bovine serum albumin (BSA) and 0.3% Triton for 1 h at RT. Sections were then incubated overnight at 4 °C in rabbit anti-VIP primary antibody (1/1000; Immunostar, Hudson, WI) in PBS with 2% BSA and 0.25% Triton. Following washing with PBS containing 0.25% Triton for 30 min at RT, sections were incubated for 2 h at RT in donkey anti-rabbit Alexa 488 secondary antibody (1/500; Jackson Immunoresearch, West Grove, PA) in PBS with 2% BSA and 0.25% Triton. Sections were washed again in PBS for 30 min, mounted, allowed to air dry overnight in the dark, and coverslipped with DABCO (1,4-Diazobicyclo-[2,2,2]-octane) mounting medium.
Fluorescence images were acquired on a Nikon A1 confocal microscope using the NIS Elements software (Nikon Instruments, Melville, NY). Z-stacks (8-10 µm), composed of 1024 × 1024 (1 µm) optical sections, were collected and projected onto a single optical slice. Cells expressing tdTomato and/or immunolabeled for VIP in every fourth section containing the SCN were counted (ImageJ; National Institutes of Health, Bethesda, MD). A total of 8 sections were used from 4 mice (2 sections/mouse). The numbers of tdTomato-expressing cells that were also VIP positive and, conversely, the number of VIP-positive neurons that express tdTomato were determined.
Preparation of Acute SCN Slices
Acute SCN slices were prepared from adult (4-12 weeks) male mice maintained in either a standard or a reversed 12:12-h LD cycle (Granados-Fuentes et al., 2012). Time referred to here is zeitgeber time (ZT); ZT0 corresponds to the time of lights on and ZT12 corresponds to lights off in the animal facility. Daytime slices were routinely prepared at ZT5 from mice maintained in the standard LD cycle, and nighttime slices were prepared at ZT15 from mice maintained in the reversed LD cycle.
For the preparation of (daytime) slices, brains were rapidly removed in the light from animals anesthetized with 1.25% Avertin (2,2,2-tribromoethanol and tert-amyl alcohol in 0.9% NaCl; 0.025 mL/g body weight) and placed in ice-cold cutting solution containing (in mM) the following: sucrose, 240; KCl, 2.5; NaH2PO4, 1.25; NaHCO3, 25; CaCl2, 0.5; and MgCl2 7, saturated with 95% O2/5% CO2. For the preparation of nighttime slices, mice in the reversed LD cycle were removed from their cages at ZT15 under infrared illumination to avoid exposure to visible light during the preparation of acute SCN slices. Animals were anesthetized with isoflorane and enucleated using a previously described procedure (Aton et al., 2004; Hattar et al., 2006). Briefly, following the induction of anesthesia and wiping the head with Betadine, gentle pressure was applied to the sides of the head. Using sterile scissors, a single cut was made through the ocular muscles and optic nerve and the eyes were rapidly removed. Cotton gauze was placed in the orbits to stop bleeding. Animals were allowed to recover from the anesthesia (for approximately 1 h) during the transport to the laboratory for the preparation of slices. At ZT16, these animals were anesthetized with 1.25% Avertin; brains were rapidly removed and placed in ice-cold cutting solution. An additional series of experiments was also completed in which animals in the normal LD cycle were anesthetized and enucleated (at ZT5) prior to transport to the laboratory for the preparation of SCN slices (at ZT6) and electrophysiological recordings.
For all experiments, coronal slices (300 µm) were cut on a Leica VT1000 S vibrating blade microtome (Leica Microsystems, Buffalo Groves, IL) and incubated in a holding chamber with oxygenated artificial cerebrospinal fluid (ACSF) containing (in mM) the following: NaCl, 125; KCl, 2.5; NaH2PO4, 1.25; NaHCO3, 25; CaCl2, 2; MgCl2, 1; and dextrose, 25 (~310 mosmol l−1), saturated with 95% O2/5% CO2, at room temperature (~25 °C) for at least 1 h before transfer to the recording chamber.
Electrophysiological Recordings
Whole-cell current-clamp recordings were obtained at ZT7-12 (daytime) or ZT19-24 (nighttime) from visually identified SCN neurons using a Nikon FN-S2N microscope equipped with differential interference contrast (DIC) optics with infrared illumination. Slices were perfused continuously with ACSF saturated with 95% O2/5% CO2 at room temperature (~25 °C). Recordings were obtained using glass pipettes (4-5 MΩ) filled with intracellular solution containing (in mM) the following: KMeSO4, 120; KCl, 20; HEPES, 10; EGTA, 0.2; NaCl, 8; Mg-ATP, 4; Tris-GTP, 0.3; and phosphocreatine, 14 (pH 7.3-7.5; ~300 mosmol l−1). Experiments were controlled and data were collected using a Multiclamp 700B patch-clamp amplifier (Molecular Devices, Sunnyvale, CA) interfaced to a Dell personal computer with a Digidata 1332 and pCLAMP 10 (Molecular Devices). Tip potentials were zeroed before recordings were obtained. For each cell, a “loose patch” cell-attached recording was first obtained, and spontaneous activity was recorded for ~1 min. A gigaOhm seal (≥2 GΩ) was then formed, and the whole-cell configuration was established. Following compensation of whole-cell membrane capacitances and series resistances, spontaneous firing was again recorded for ~1 min. Access resistances of these cells were 15 to 23 MΩ. Data were not collected if the value significantly (20%) changed during the course of the experiment. Voltage signals were acquired at 100 kHz, filtered at 10 kHz, and stored for offline analysis. Input resistances (Rin) were determined by measuring the steady-state voltage changes produced by ±5 pA current injections from a hyperpolarized membrane potential. Action potential thresholds (APTs) were determined in each cell as the point during the upstroke (depolarizing phase) of the action potential at which the second derivative of the voltage was zero. Afterhyperpolarization (AHP) amplitudes were measured in each cell as the difference between the APT for action potential generation and the most negative membrane potential. Action potential durations were measured at 50% repolarization (APD50). The interspike membrane potential was also measured in each cell to provide an estimate of the resting membrane potential. In some recordings (
The mean whole-cell capacitances of VIP cells during the day (
Data Analysis
Electrophysiological data (Table 1) were compiled and analyzed using ClampFit (version 10.2; Molecular Devices), Microsoft Excel (Microsoft Corp., Redmond, WA), Mini Analysis (version 6.0.7; Synaptosoft, Decatur, GA), Prism (version 6; GraphPad Software, La Jolla, CA), and OriginLab (version 9.1; Northampton, MA).
Resting and Active Membrane Properties of VIP and Non-VIP SCN Neurons.
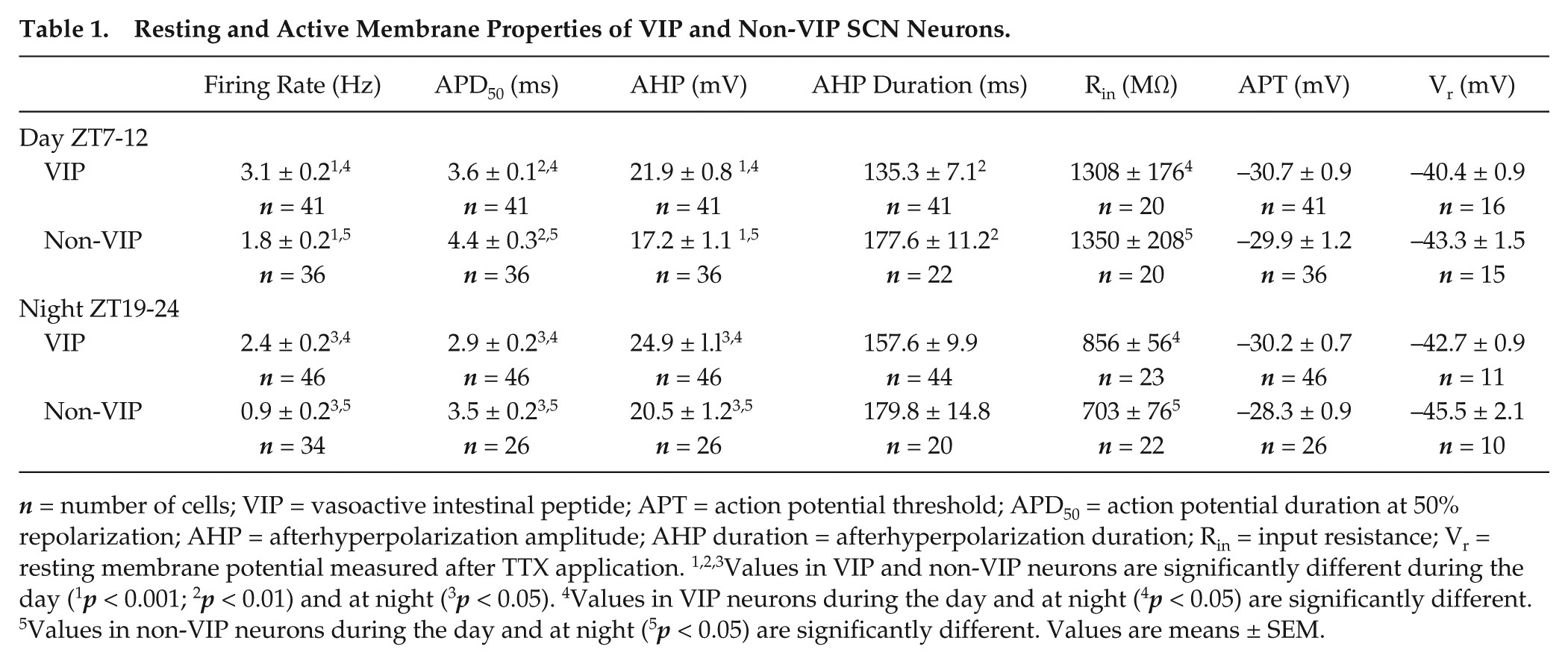
Statistics
All data are presented as means ± standard error of the mean (SEM). All data were collected from 17 slices/animals for daytime recordings and from 13 slices/animals for nighttime recordings. Statistical analyses were performed using 1-way analysis of variance (ANOVA) with Newman-Kuels post hoc pairwise comparisons or 2-sample Kolmogorov-Smirnov test. All data were analyzed using GraphPad Prism software with the exception of the cumulative distribution plots, which were analyzed using OriginLab with the 2-sample Kolmogorov-Smirnov test. Statistical significance was set at
Results
Identification of VIP Neurons in the SCN
The strategy for identification of SCN neurons in vitro is illustrated in Figure 1A. A mouse line expressing the red fluorescent protein, tdTomato (tdTom), in VIP-expressing neurons of the SCN was generated by crossing mice (VIP-Cre) expressing Cre-recombinase driven by a knock-in fragment of the VIP promoter (Taniguchi et al., 2011) with mice containing the Rosa-CAG-LSL-tdTomato-WPRE conditional floxed allele (Madisen et al., 2010). Coronal sections containing the SCN were cut from the brains of the resulting VIP-tdTom knock-in mice and stained with an anti-VIP antibody. The specificity of the anti-VIP antibody was previously validated in SCN sections from mice (VIP–/–) harboring a targeted disruption of the VIP locus: no anti-VIP immunolabeling was evident in VIP–/– SCN sections with this antibody (An et al., 2012). VIP immunolabeling and tdTom fluorescence, however, were readily detected in a subpopulation of SCN neurons from VIP-tdTom knock-in mice (Figure 1B). We found that an average of 95% of the tdTom-expressing SCN neurons also immunolabeled for VIP. Conversely, we found that all VIP-immunolabeled SCN cells expressed tdTom (Figure 1C). The VIP-tdTom knock-in mouse, therefore, reliably identifies VIP neurons in the SCN.
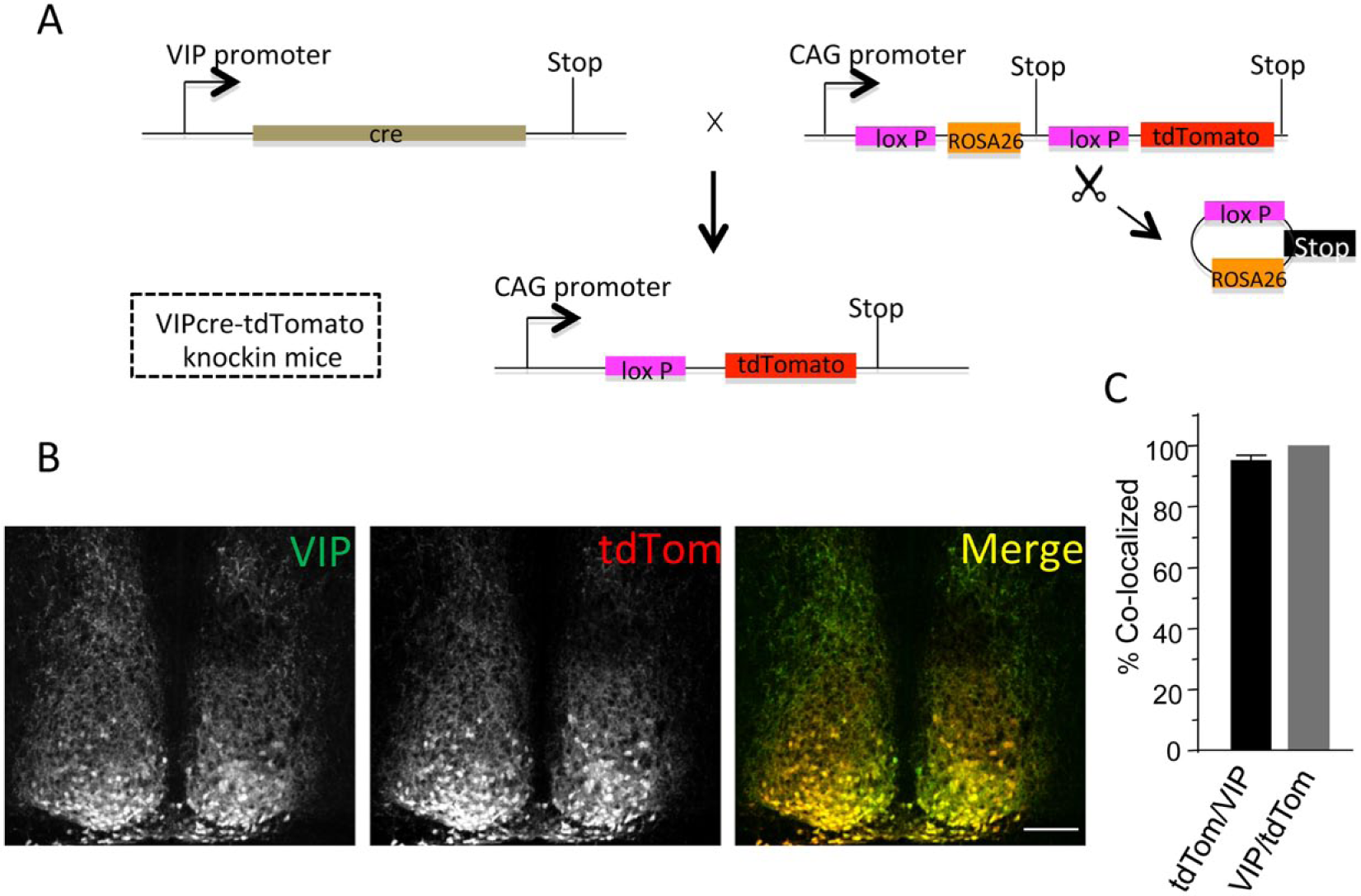
Generation and validation of tdTomato expression in vasoactive intestinal peptide (VIP) SCN neurons. (A) Schematic representation of the breeding strategy used to generate VIPcre-tdTomato knock-in mice: VIP-cre mice (Taniguchi et al., 2011) were bred with mice hemizygous for Rosa-CAG-LSL-tdTomato-WPRE (Madisen et al., 2010). In tissues expressing Cre-recombinase driven by the VIP promoter, the STOP cassette is deleted and the fluorescent tdTomato (tdTom) reporter protein is expressed. (B) Immunolabeling with an anti-VIP antibody in a representative coronal SCN section from a VIPcre-tdTomato knock-in mouse is shown. Robust VIP expression (left panel) is evident in cells that also express tdTom (middle panel). The merged image illustrates the overlap of VIP and tdTom expression (right panel); scale bar = 100 µm. (C) Cell counts revealed that nearly all of the tdTom-expressing SCN neurons were also labeled with the anti-VIP antibody (tdTom/VIP) and, in addition, that all VIP-positive neurons also express tdTom (VIP/tdTom). Values presented are means ± SEM of 1539 neurons counted in 8 sections from 4 animals.
Spontaneous Firing Properties of VIP and Non-VIP SCN Neurons Are Distinct
We visually identified VIP and non-VIP SCN neurons by the presence and absence of tdTom fluorescence, respectively (Figure 2A). In all experiments, cell-attached recording (Perkins, 2006) was obtained prior to establishing the whole-cell configuration, and spontaneous firing was recorded for 1 min. After a gigaOhm seal was formed and the whole-cell configuration was obtained, spontaneous firing was again recorded for ~1 min. Analysis of the records obtained in the cell-attached and the whole-cell configuration from the same cells revealed no significant (
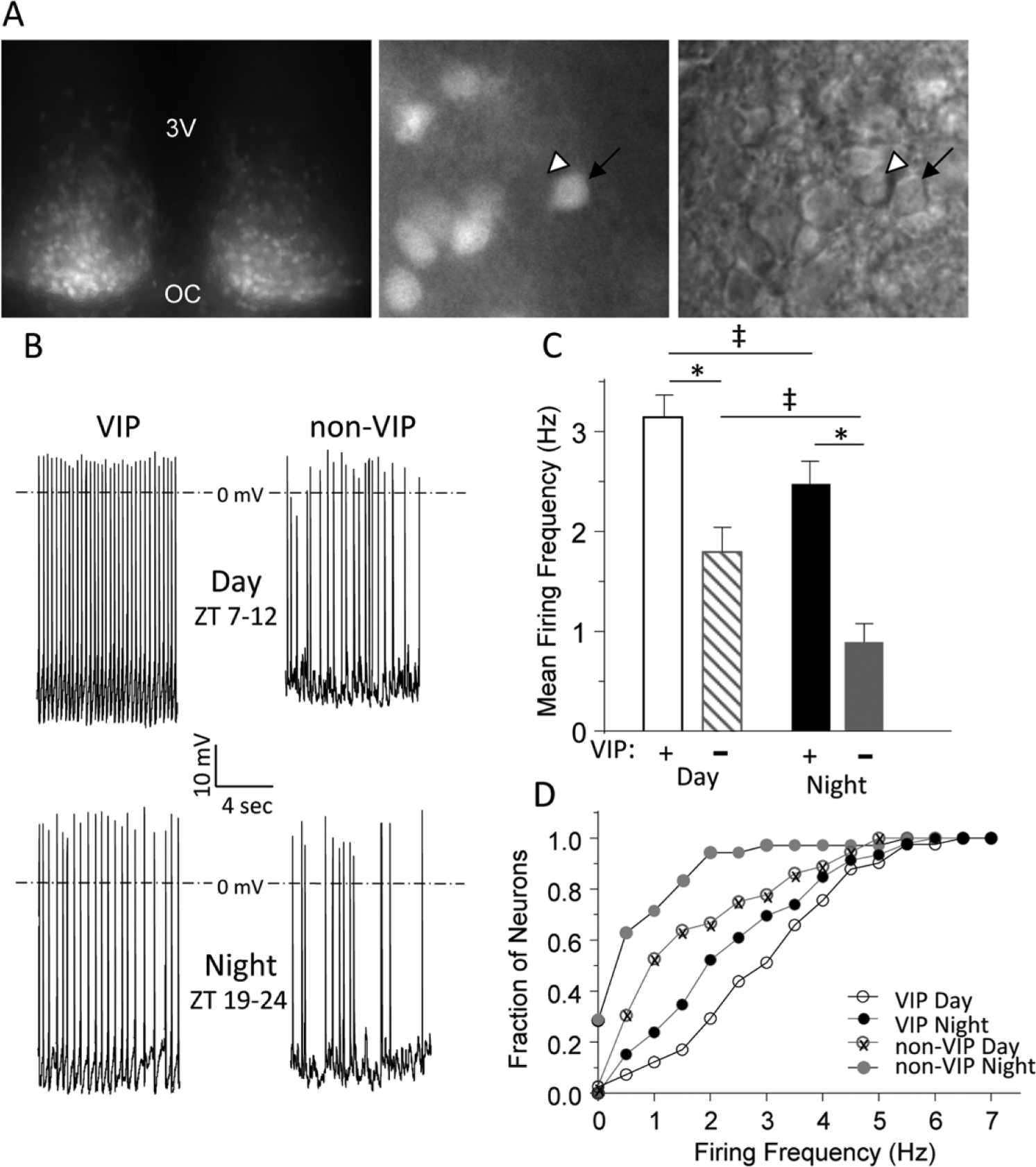
Spontaneous firing rates are higher in vasoactive intestinal peptide (VIP) than in non-VIP SCN neurons. (A) An acute SCN slice from a VIPcre-tdTomato knock-in mouse is shown. In the low-magnification fluorescence image on the left, tdTom expression is evident in both hemispheres; the third ventricle (3V) and optic chiasm (OC) are marked. In the higher magnification fluorescence image in the center, tdTom-positive neurons (arrow) are easily distinguished from nonlabeled neurons (arrowhead); the corresponding differential interference contrast (DIC) image is shown on the right. (B) Representative daytime (top) and nighttime (bottom) whole-cell current clamp recordings obtained from VIP and non-VIP neurons are shown; mean ± SEM firing frequencies are presented in (C). The mean ± SEM daytime firing rate is significantly (*
Day or night, VIP neurons had significantly (
Furthermore, whereas 90% of non-VIP neurons fired at 2 Hz or less at night, approximately 50% of VIP cells fired at similar frequencies (Figure 2D). We also found that, while 25% (8 of 34) of the non-VIP SCN neurons were electrically silent at night, none (0 of 46) of the VIP SCN neurons studied were quiescent at night (Figure 2D). This cell type–specific difference remained during the day when 65% of the non-VIP cells fired at 2 Hz or less, whereas only 30% of the VIP cells fired at 2 Hz or less (Figure 2D). Therefore, although daily rhythms in firing rates are clearly observed in both VIP and non-VIP neurons in acute SCN slices, on average, the firing rates of VIP neurons are higher than non-VIP neurons throughout the LD cycle.
Action Potential Waveforms in VIP and Non-VIP SCN Neurons Are Distinct
The waveforms of individual action potentials recorded in VIP and non-VIP SCN neurons, however, were distinct (Figures 3 and 4). During the day and at night, for example, action potentials were significantly (
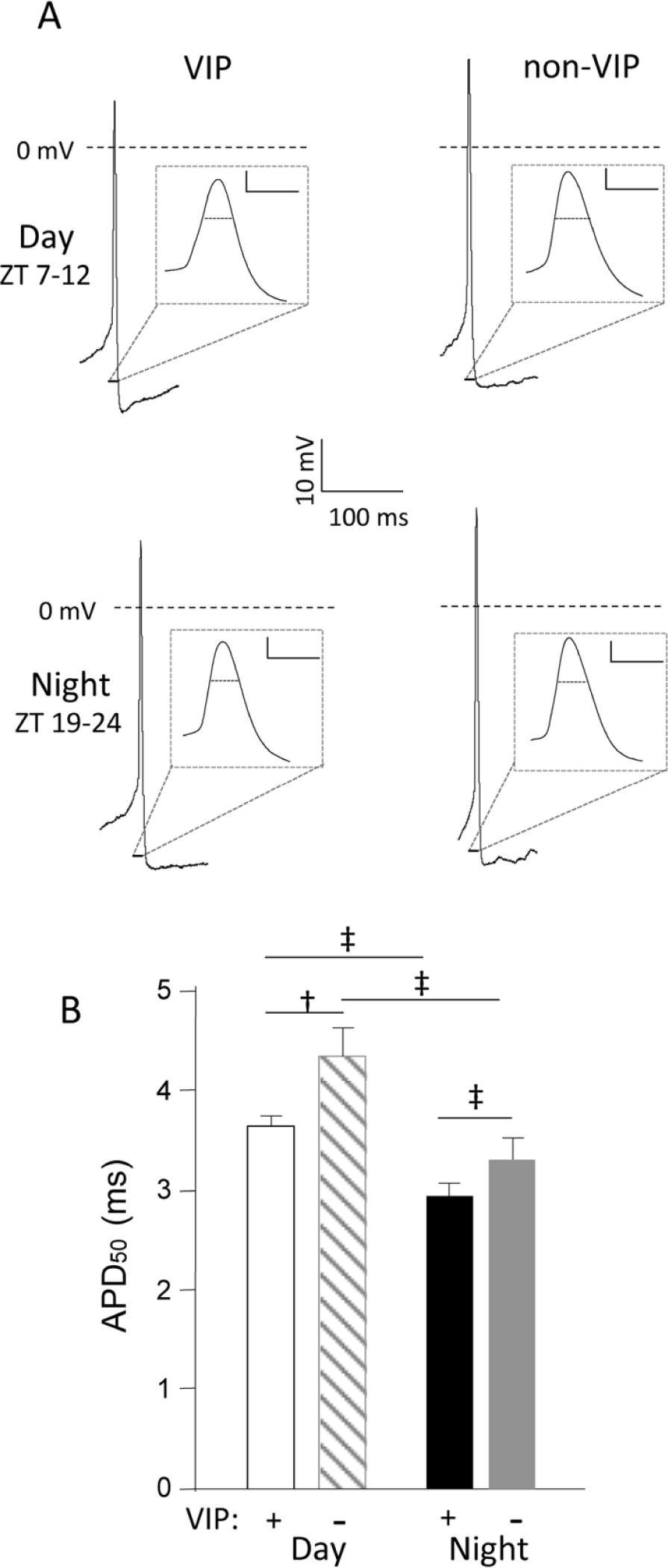
Action potentials are briefer in vasoactive intestinal peptide (VIP) than in non-VIP SCN neurons. (A) Representative action potential waveforms recorded in VIP and non-VIP SCN neurons during the day (top) or at night (bottom) are illustrated; the records are shown on an expanded time scale in the insets (scale bars = 10 mV and 5 ms). (B) For daytime recordings, the mean ± SEM action potential duration (APD50) is significantly (‡
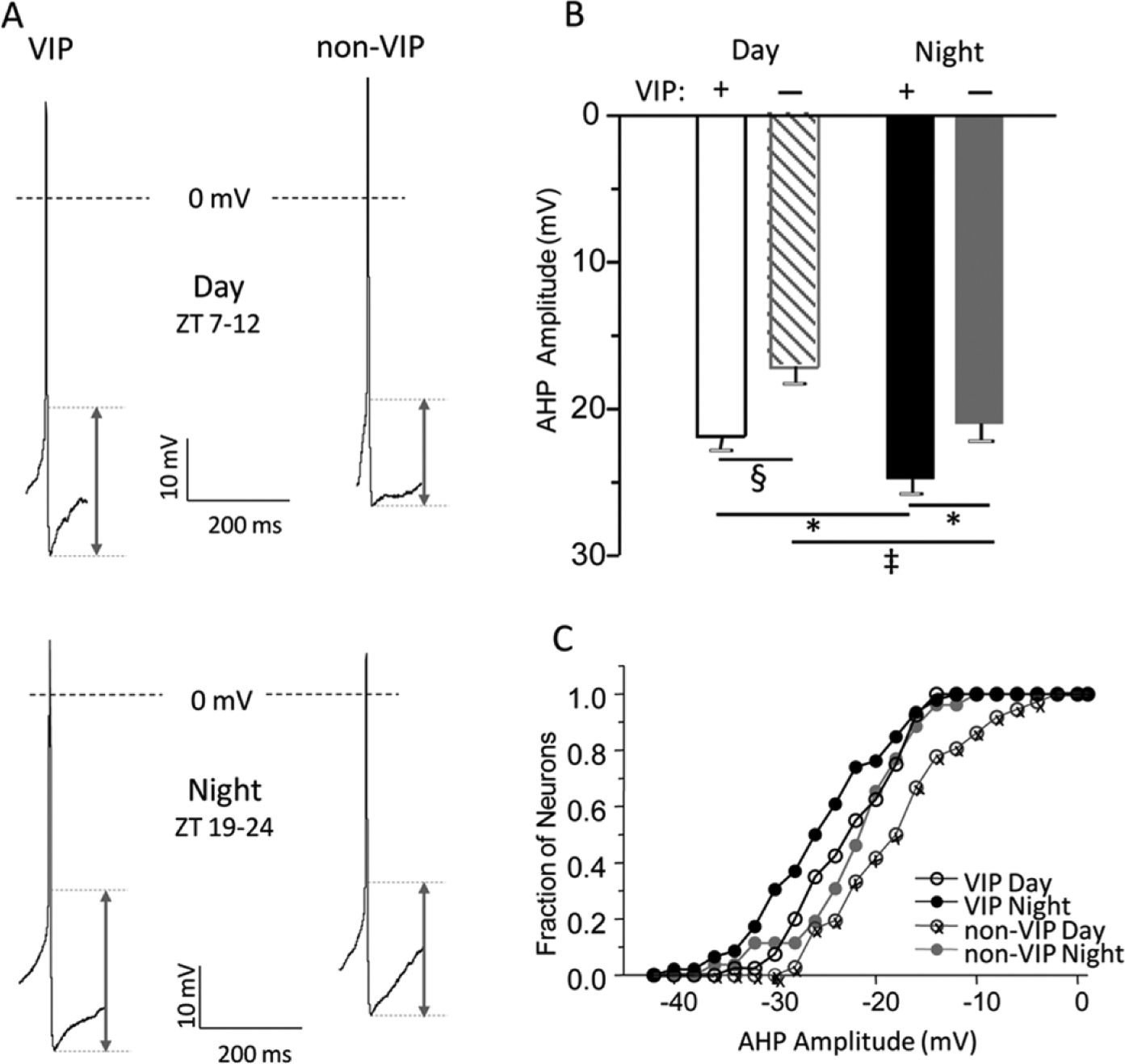
Afterhyperpolarization (AHP) amplitudes are larger in vasoactive intestinal peptide (VIP) than in non-VIP SCN neurons. (A) Representative action potential waveforms recorded in VIP and non-VIP SCN neurons during the day (top) and at night (bottom) are shown. AHP amplitudes were measured as the difference between the voltage threshold for action potential generation and the most negative membrane potential reached during the AHP. (B) The mean ± SEM AHP amplitudes measured in VIP SCN neurons (
The average AHP amplitude measured in VIP neurons was significantly (
Other parameters such as action potential thresholds (APTs) and resting membrane potentials (Vm) were not different in VIP and non-VIP neurons of the SCN either at night or during the day (Table 1). The mean input resistances (Rin) of VIP neurons and non-VIP neurons were significantly (
Discussion
The results presented here demonstrated that the repetitive firing properties of VIP-expressing neurons in the SCN differ from those of other SCN neurons throughout the LD cycle. Whereas both VIP and non-VIP neurons fire more during the day than at night, consistent with prior recordings from unidentified SCN neurons (Inouye and Kawamura, 1979; Green and Gillette, 1982; Welsh et al., 1995; Pennartz et al., 2002), the firing rates of VIP neurons were 40% higher during the day and at night than non-VIP neurons. In addition, all VIP neurons continued to fire repetitively at night, whereas only 75% of non-VIP neurons fired at night; 25% of the non-VIP neurons were electrically silent. These observations may reflect cellular heterogeneity in the population of SCN neurons classified here as “non-VIP” neurons, a hypothesis worth testing experimentally.
Intrinsic Membrane Properties of VIP SCN Neurons Are Distinct
Similar to results obtained in studies on unidentified SCN neurons (de Jeu et al., 1998; Kuhlman and McMahon, 2004, 2006; Belle et al., 2009), day-night differences in the input resistances (Rin) of VIP and non-VIP neurons were observed in the experiments presented here. In both groups of cells, Rin values were significantly higher during the day than at night, consistent with daily rhythmic changes in K+ conductance(s) (Kuhlman and McMahon, 2004, 2006). Comparisons of Rin values measured in VIP and non-VIP neurons during the day (as well as at night) revealed no significant differences, suggesting that both cell types undergo similar changes in the ionic conductance(s) mediating the daily change in firing frequencies observed.
In contrast to the similarities in Rin values, action potential waveforms in VIP and non-VIP neurons were distinct throughout the LD cycle. Action potentials recorded during the day in VIP neurons, for example, were briefer than non-VIP neurons. In addition, AHP amplitudes in VIP neurons were significantly larger than in non-VIP neurons during the day and at night. Furthermore, analysis of the mean AHP durations in VIP and non-VIP neurons revealed significantly shorter AHP durations in VIP neurons than in non-VIP neurons during the day. Shorter APD50, larger AHP amplitudes, and faster AHP durations are differences that likely contribute to the higher repetitive firing rates observed in VIP neurons. Taken together, these observations suggest that there are differences in the properties and/or the densities of the K+ currents that control the rates of action potential repolarization and AHP amplitudes in VIP compared with non-VIP SCN neurons.
As in many other types of GABAergic neurons in the central nervous system (Wang et al., 1998; Baranauskas et al., 2003), fast delayed rectifier K+ currents, encoded by voltage-gated K+ (Kv) channel pore-forming (α) submits of the Kv3 family, have been suggested to be important determinants of rapid action potential repolarization and the repetitive firing properties of SCN neurons (Itri, 2005; Kudo et al., 2011; Kudo et al., 2013). Although cell type–specific differences in the expression of Kv3-encoded fast delayed rectifier K+ currents could certainly underlie the observed differences in action potential waveforms and repetitive firing rates observed in VIP and non-VIP SCN neurons, other Kv- and/or Ca2+-dependent K+ currents (Cloues and Sather, 2003; Meredith et al., 2006; Belle et al., 2009) could also play important roles. Additional experiments, particularly voltage-clamp experiments focused on characterizing the K+ currents contributing to the control of action potential durations and the amplitudes of afterhyperpolarizations in VIP neurons and other identified cell types in the SCN, will be needed to provide further molecular insights.
Functional Implications of the Distinct Firing Properties of VIP SCN Neurons
Many studies have implicated VIP signaling in the modulation and synchronization of the rhythmic electrical activity of the SCN and behavior (Reed et al., 2001; Harmar et al., 2002; Cutler et al., 2003; Aton et al., 2005; Maywood et al., 2006; Brown et al., 2007). The findings here that the repetitive firing rates of VIP neurons are significantly higher than non-VIP neurons throughout the LD cycle are consistent with a model in which VIP-expressing neurons have a large influence on synchronizing electrical activity in the SCN. In this model, VIP-expressing neurons in the SCN play a role reminiscent of that ascribed to pigment dispersing factor (PDF)–expressing neurons in
Daily oscillations in the expression of the transcript encoding VIP, as well as in VIP protein expression and VIP release in the SCN, have been reported (Okamoto et al., 1991; Shinohara et al., 1993; Francl et al., 2010). Indeed, it has been shown that the peak expression of both VIP mRNA and protein occurs during the subjective night (Okamoto et al., 1991; Shinohara et al., 1993; Ban et al., 1997). VIP release, however, increases during the day (Francl et al., 2010), likely reflecting the daytime increase in the repetitive firing in VIP neurons. Interestingly, bath application of VIP alters the membrane and firing properties of SCN neurons for hours (Kudo et al., 2013), which may result in continued VIP release. Taken together, these observations suggest a model in which the increased daytime firing activity of VIPergic neurons leads to more VIP release, resulting in synchronization of electrical activity in the SCN. Further experiments focused on exploring this hypothesis directly will also be of interest.
Footnotes
Conflict Of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
