Abstract
Synchronizing the circadian timing system (CTS) to external light/dark cycles is crucial for homeostasis maintenance and environmental adaptation. The CTS is organized hierarchically, with the central pacemaker located in the suprachiasmatic nuclei (SCN) generating coherent oscillations that are entrained to light/dark cycles. These oscillations regulate the release of glucocorticoids by the hypothalamus-pituitary-adrenal (HPA) axis, which acts as a systemic entrainer of peripheral clocks throughout the body. The SCN adjusts its network plasticity in response to variations in photoperiod, leading to changes in the rhythmic release of glucocorticoids and ultimately impacting peripheral clocks. However, the effects of photoperiod-induced variations of glucocorticoids on the synchronization of peripheral clocks are not fully understood, and the interaction between jetlag adaption and photoperiod changes is unclear. This study presents a semi-mechanistic mathematical model to investigate how the CTS responds to changes in photoperiod. Specifically, the study focuses on the entrainment properties of a system composed of the SCN, HPA axis, and peripheral clocks. The results show that high-amplitude glucocorticoid rhythms lead to a more coherent phase distribution in the periphery. In addition, our study investigates the effect of photoperiod exposure on jetlag recovery time and phase shift, proposing different interventional strategies for eastward and westward jetlag. The findings suggest that decreasing photic exposure before jetlag during eastward traveling and after jetlag during westward traveling can accelerate jetlag readaptation. The study provides insights into the mechanisms of CTS organization and potential recovery strategies for transitions between time zones and lighting zones.
Introduction
Adapting to predictable environmental changes is regulated by the circadian timing system (CTS), a multi-scale, hierarchical, and distributed mechanism. At the single-cell level, molecular clocks are driven by positive and negative feedback loops consisting of a group of “clock” genes and proteins that generate self-sustained, autonomous oscillations with a nearly 24-h period. The endogenous molecular clock machinery is adaptable and can be synchronized to external periodic stimuli (
In mammals, light is sensed by photoreceptors in the retina, and photic information is transduced to the hypothalamic suprachiasmatic nuclei (SCN) via the intrinsically photoreceptive retinohypothalamic tract (S.-K. Chen et al., 2011). The SCN is a bilateral structure that comprises approximately 20,000 neurons; each neuron contains an autonomous circadian oscillator. Those neurons are heterogeneous. They couple with each other and function as a network, generating coherent rhythmicity through spatial and temporal coordination (Herzog, 2007). As a result, the topographically organized neurons exhibit spontaneous cyclic fluctuations in circadian clock gene expression. This rhythm is endogenous, meaning that the oscillation will be preserved even in the absence of light/dark entrainment (Webb et al., 2009). By receiving direct photic input and employing organized coupling mechanisms to reinforce its intrinsic rhythmicity, the SCN plays a role as the central pacemaker in the CTS.
Circadian regulation is astonishingly widespread: nearly half of all mammalian genes are rhythmically expressed in one or more tissues (Yan et al., 2008; Acevedo et al., 2021). Besides the SCN neurons, clock genes and proteins have been observed in cells and tissues throughout the body (such as the lung, liver, skeletal muscle, and heart), indicating the existence of endogenous clocks among the peripheral cells (Welsh et al., 2004; Yoo et al., 2004; Zhang et al., 2014). The peripheral clocks exert broad-ranging control over many physiological processes (Nahmias and Androulakis, 2021; Schlesinger et al., 2022), including steroidogenesis (H. Chen et al., 2017), glucose homeostasis (Gachon et al., 2017), and immune function (Beam et al., 2020). The peripheral cellular oscillators are heterogeneous in their intrinsic periods and, thus, must be synchronized to establish coherent tissue rhythms. While SCN exhibits inherent coupling and can maintain synchronization ex vivo for extended periods, peripheral cells display ensemble oscillations that rapidly dampen without intercellular coupling signals (Abraham et al., 2010), indicating that systemic entrainment is necessary for peripheral clock synchronization.
The light-sensitive SCN conveys photic signals through its rhythmic output, entraining the neuroendocrine system, which eventually synchronizes the peripheral clocks (Kalsbeek et al., 2010). The hypothalamic-pituitary-adrenal (HPA) axis plays an important role in coupling the oscillation between the SCN and the periphery. In the paraventricular nucleus (PVN), corticotropin-releasing hormone (CRH) is rhythmically secreted by CRH neurons, regulated by the SCN-produced neurotransmitter AVP (Kalsbeek et al., 2012). The CRH travels to the anterior pituitary, stimulating the production of adrenocorticotropic hormone (ACTH), which activates the release of glucocorticoids (CORT) from the adrenal cortex. The daily rhythmicity of plasma glucocorticoid levels functions as a systemic synchronizer that coordinates the phase of peripheral clocks by binding to the glucocorticoid-response elements (GREs) regions, activating the transcription of a number of clock genes (Yamamoto et al., 2005; Reddy et al., 2007; So et al., 2009; Cuesta et al., 2015). Glucocorticoids also exert negative feedback on both PVN and the anterior pituitary by inhibiting the secretion of CRH and ACTH, therefore indirectly regulating its own secretion (Stanojević et al., 2018; Focke and Iremonger, 2020). Entrained to the SCN, glucocorticoids display a daily peak at the habitual sleep-wake transition, with levels peaking at the onset of the active phase and declining during the inactive phase (defined as day for diurnal species and night for nocturnal species) (Oster et al., 2017).
Apart from the light/dark cycle, the SCN neurons also adapt to photoperiod changes by adjusting their ensemble phase plasticity. During short photoperiod months (winter), the SCN neurons exhibit a narrower phase distribution compared to long photoperiod months (summer) (VanderLeest et al., 2007; Meijer et al., 2010). Governed by the substantial differences in the SCN phase distribution, glucocorticoid rhythms exhibit distinct variations under photoperiod changes, with amplitudes highest during short photoperiod (winter) and lowest during long photoperiods (summer) (Amirat et al., 1980; Otsuka et al., 2012). Studies have shown that the robustness of glucocorticoid rhythmicity is strongly associated with peripheral circadian alignment (Morris et al., 2016). Therefore, it has been speculated that photoperiod changes induced by SCN and HPA rhythm will differentially regulate the synchronization of peripheral oscillators while detailed mechanisms remain to be elucidated (Pierre et al., 2018).
Optimal physiological functions occur when the central and peripheral rhythms are aligned with the external light schedules. Conversely, the misalignment between the central clock, peripheral clock, and the environment may increase the risk of disease, with the immune system, metabolism, and cardiovascular system negatively impacted (Morris et al., 2016; Olejniczak et al., 2022). Considering the complex hierarchical structure of the CTS, mathematical modeling offers advantages in accessing the physiological system at a holistic level, uncovering potential mechanisms, and quantifying properties that are experimentally challenging to identify (Androulakis, 2021). While previous theoretical studies have investigated the effects of jetlag and seasonality using relatively simplified mathematical models, and have identified the impact of intrinsic period and amplitude on jetlag recovery (VanderLeest et al., 2009; Schmal et al., 2015; Ananthasubramaniam et al., 2020; Tokuda et al., 2020), a comprehensive model that can be used to study jetlag and seasonal-related mechanisms in detail is still lacking. Furthermore, the interaction between time zone transition and lighting zone transition has been overlooked in previous research.
The current study presents a semi-mechanistic mathematical model that represents the hierarchical structure of the CTS and examines its systemic response to light-dark entrainment, annual photoperiod changes, and photoperiod-dependent jetlag responses. Focusing on peripheral clock synchronization properties, our model identified differential peripheral phase distribution under different photoperiods. By investigating the systemic circadian alterations under lighting zone and time zone transitions, our model shows that reducing photic exposure before a phase-advanced jetlag can accelerate the transition rate of the system while reducing photic exposure after a phase-delayed jetlag can also speed up the recovery rate. Furthermore, our study revealed a positive correlation between the magnitude of phase shift during jetlag and the duration of jetlag recovery, suggesting that preserving the intrinsic phase shift of the system may lead to more effective jetlag recovery. These findings shed light on the mechanisms underlying photoperiod adaptation and non-pharmacological interventions for jetlag, providing a basis for further studies and potential clinical applications.
Materials and Methods
The “Model Construction” section of the Supplementary Material (Suppl.) provides an in-depth description of the developed mathematical model that characterizes the seasonal entrainment of the HPA axis induced by the SCN organization. In brief, the hierarchical organization of the CTS is modeled at both the central and peripheral levels. As shown in Figure 1, in the central compartment, the SCN is entrained to the light/dark cycles and can adjust its network plasticity according to photoperiod changes. The SCN synchronizes the autonomous oscillations in the HPA axis, which, in turn, regulates cells in the periphery. SCN neurons can be neuroanatomically divided into two subpopulations: the ventrolateral “core” (VL) and the dorsomedial “shell” (DM). The core neurons secret vasoactive intestinal peptide (VIP), neurotensin (NT), and gastrin-releasing peptide (GRP) and receive afferent signals from the retina. The shell neurons express arginine vasopressin (AVP), angiotensin II (AII), and met-enkephalin (mENK). Neuropeptides VIP and AVP are the main synchronizers of the core and shell neuron population (Ono et al., 2021). Based on the experimentally identified structures, the SCN model in the present work consists of two ensembles of neuronal oscillators, which represent the core and the shell, respectively. At the tissue level, the neurons communicate with each other via neurotransmitters VIP and AVP (Moore et al., 2002). Both neurotransmitters are released upon the activity of PER/CRY proteins and can activate the transcription of
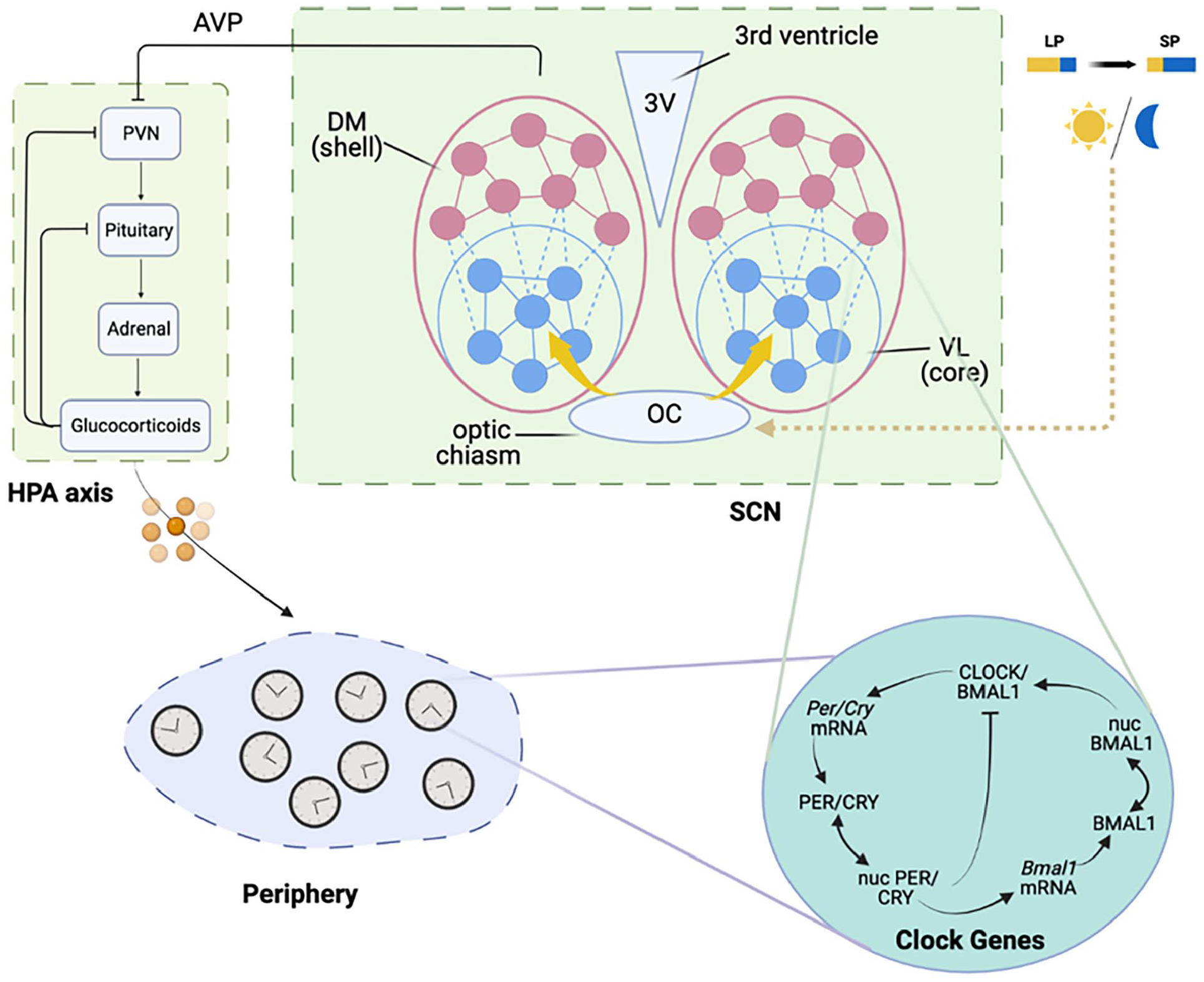
The modeled circadian timing system (CTS) is structured in three levels: the suprachiasmatic nucleus (SCN), the hypothalamic-pituitary-adrenal (HPA) axis, and the periphery. The SCN comprises of a light-sensitive core region and a light-insensitive shell region, with each neuron subpopulation consisting of mutually coupled cellular oscillators. The SCN’s coupling plasticity can change under different photoperiods. The HPA axis exhibits a negative feedback loop consisting of corticotropin-releasing hormone (CRH), adrenocorticotropic hormone (ACTH), and corticosterone (CORT). The HPA axis is entrained by the SCN via the negative regulatory role of arginine vasopressin (AVP) on CRH, as the model is based on a nocturnal species. In the peripheral compartment, cells are composed of independent oscillators that are synchronized through glucocorticoid regulation. Abbreviations: VL = ventrolateral; DM = dorsomedial; LP = long photoperiod; SP = short photoperiod; mRNA = messenger ribonucleic acid.
The SCN synchronizes the oscillations of the HPA axis through the SCN-mediated secretion of AVP in the paraventricular hypothalamic nucleus (Kalsbeek et al., 2010, 2012). To simulate the endogenous HPA oscillatory property, we used a previously developed HPA axis model (Mavroudis et al., 2012, 2014; Rao and Androulakis, 2017, 2019; Androulakis, 2021) and incorporated the synchronization signals from the SCN (Li and Androulakis, 2021, 2022). After diffusing into the peripheral cytoplasm, the glucocorticoids exert their effects by binding to glucocorticoid receptors in the periphery. The hyperphosphorylation of the GR upon its activation is considered the rate-determining step. A glucocorticoid-mediated indirect activation mechanism was used to model the phosphorylation kinetics. Binding to glucocorticoid ligand, the glucocorticoid receptor complex (FGR) translocates to the nucleus where they interact with glucocorticoid-responsive elements (GREs) promoter regions to activate glucocorticoid-responsive clock genes.
The periphery consists of a heterogeneous ensemble of cells whose free-running periods and phases vary significantly (Honma et al., 2004). To account for the heterogeneity of the periphery, we simulated cell-cell variability using Sobol sampling (
In the periphery, synchronizing heterogeneous oscillations through the entrainment of the systemic signal is essential for generating a consistent physiological rhythm (Aton and Herzog, 2005). To study the impact of systemic photoperiod-related seasonal plasticity in the synchronization state of the peripheral compartment, we evaluate the degree of synchronization among the peripheral clocks. The temporal profile synchronization
Results
Characterizing the Hierarchical Structure of the CTS and the Entrainment Properties of the Peripheral Clocks
To explore the stochastic nature of the periphery in the absence of the systemic glucocorticoid entraining signal, we calculated the phase and period distributions of peripheral cells when the coupling coefficient
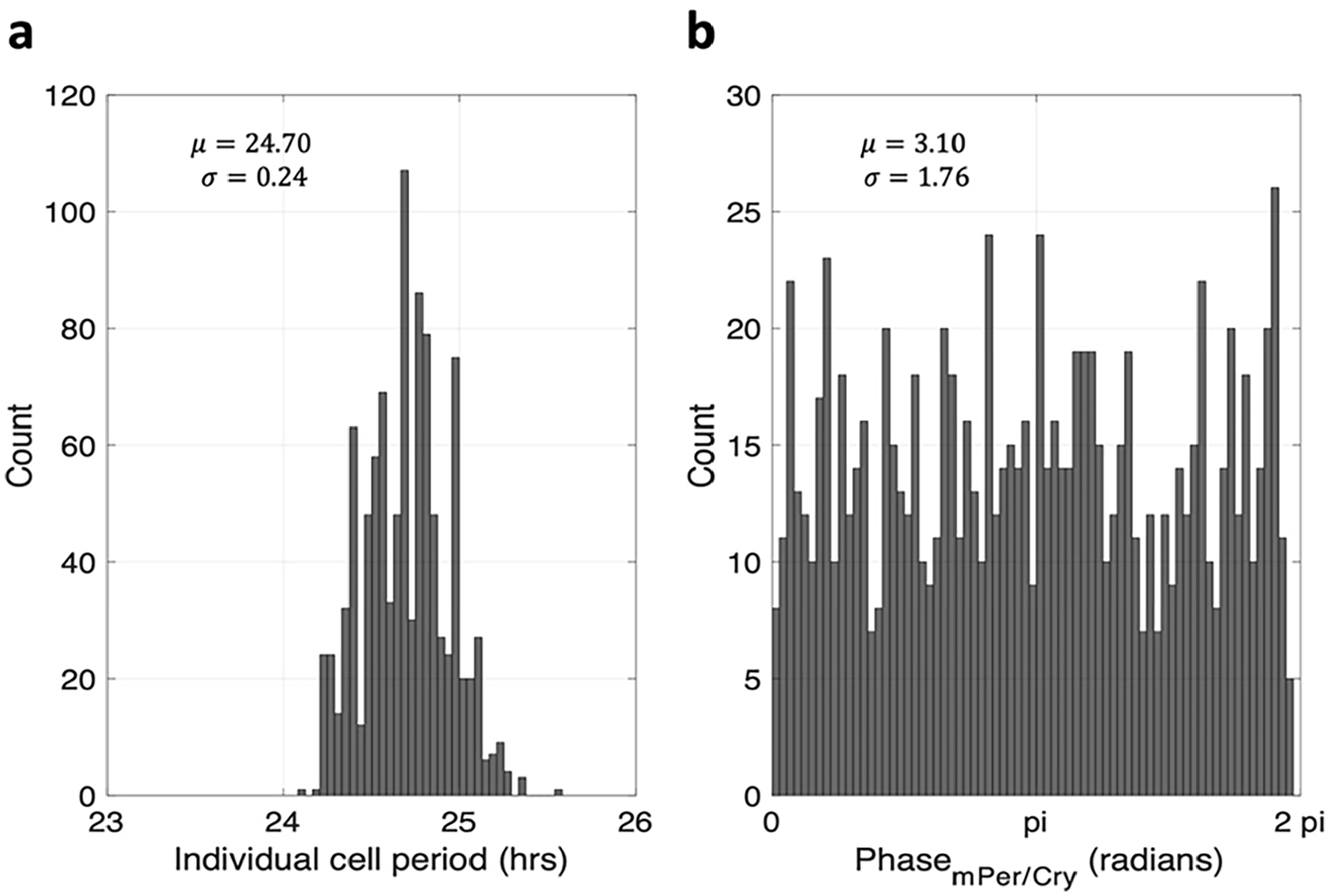
The systemic glucocorticoid signal transduced by the SCN and the HPA axis synchronizes the peripheral clocks. (a) The phase distribution and period distribution of peripheral cells in the absence of glucocorticoid entrainment. (b) The peripheral cells exhibit random phase and period distribution when not entrained by glucocorticoids. Abbreviations: SCN = suprachiasmatic nucleus; HPA = hypothalamic-pituitary-adrenal.
Regulated by the
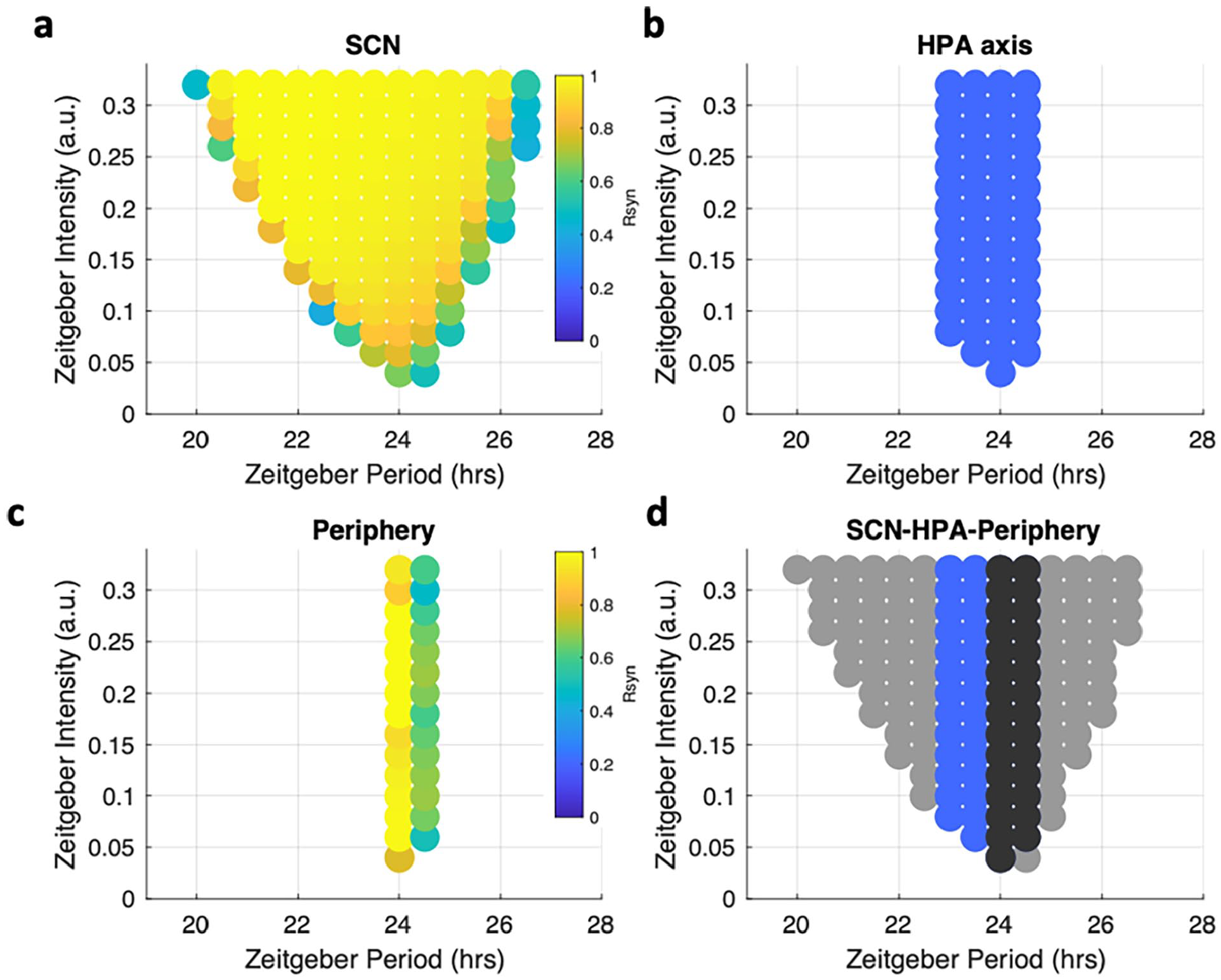
Entrainment properties of the system were identified by the “Arnold tongue” method. The entrainment range was identified in a zeitgeber period-zeitgeber intensity plane for (a) the SCN, (b) the HPA axis, (c) the peripheral clocks. (d) three Arnold tongues derived from different compartments were plotted together for comparison. Gray dots denote the SCN, blue dots denote the HPA, and black dots denote the periphery. In the SCN, the Per/Cry mRNA concentration in the light-sensitive core was used as a representative component to determine the entrainment status. In the periphery, the Per/Cry mRNA concentration was used as a representative component to determine the entrainment status. The HPA axis’ entrainment status was represented by its output glucocorticoids. An oscillation is considered to be entrained when its period equals the zeitgeber period. The color in (a) and (c) denotes the synchronization level among the SCN and peripheral ensembles, respectively. The entrainment ranges diminish as the photic signal is transduced from upstream to downstream in the circadian timing system. Abbreviations: SCN = suprachiasmatic nucleus; HPA = hypothalamic-pituitary-adrenal; mRNA = messenger ribonucleic acid.
The measured entrainment range for the SCN compartment increases with increasing
Photoperiod-Induced Variations in HPA and Their Impact on Peripheral Clock Synchronization
The photoperiod-induced variations in the HPA axis circadian dynamics are represented in Figure 4. Increasing photoperiod from
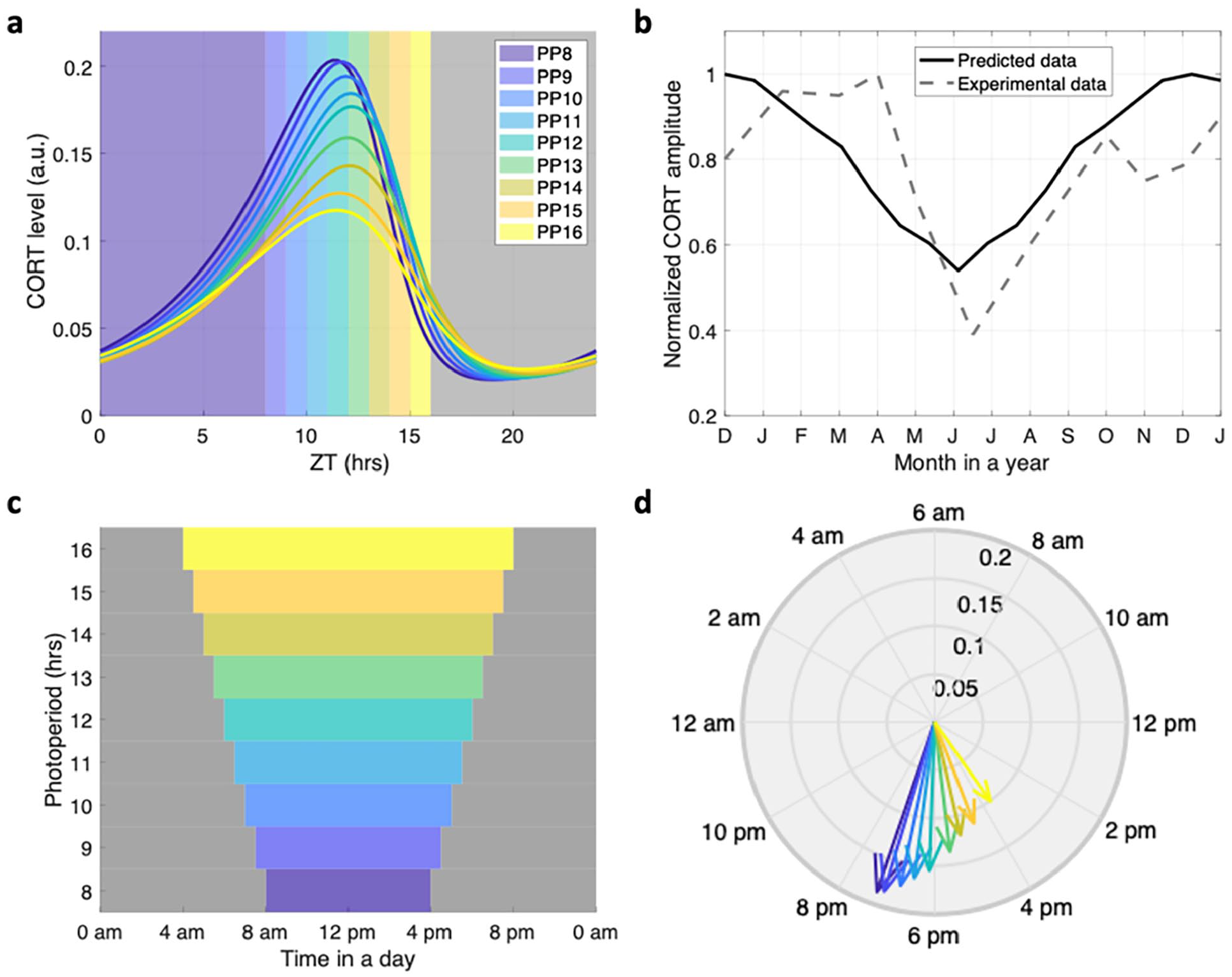
Photoperiod-induced variations in the HPA dynamics were represented by the phase and amplitude changes in glucocorticoids. (a) Circadian profiles of the glucocorticoid under different photoperiods, all light schedule starts when zeitgeber time = 0 h (ZT 0). The colorful patterns denote the light onset and offset times, and the shadow denotes darkness. (b) Annual dynamics of the HPA axis compared to experimental data. The black solid line denotes the predicted corticosterone amplitude, while the black dashed line denotes the experimentally measured corticosterone amplitude. (c) The introduced photoperiod schedule at a real-time scale. The colorful patterns denote the light onset and offset times, and the shadow denotes darkness. (d) Phase and amplitude relations of glucocorticoids under different photoperiod schedules. As the photoperiod increases, the amplitude of glucocorticoids decreases. The phases of the circadian rhythms were advanced for the long photoperiod and delayed for the short photoperiod. Abbreviations: CORT = corticosterone; PP = photoperiod; HPA = hypothalamic-pituitary-adrenal.
Entrained by glucocorticoids, the peripheral clocks exhibit photoperiod-dependent variation in their synchronization status and phase distribution. To investigate this relationship, we modeled a winter, spring, summer, and autumn light schedule with photoperiods of
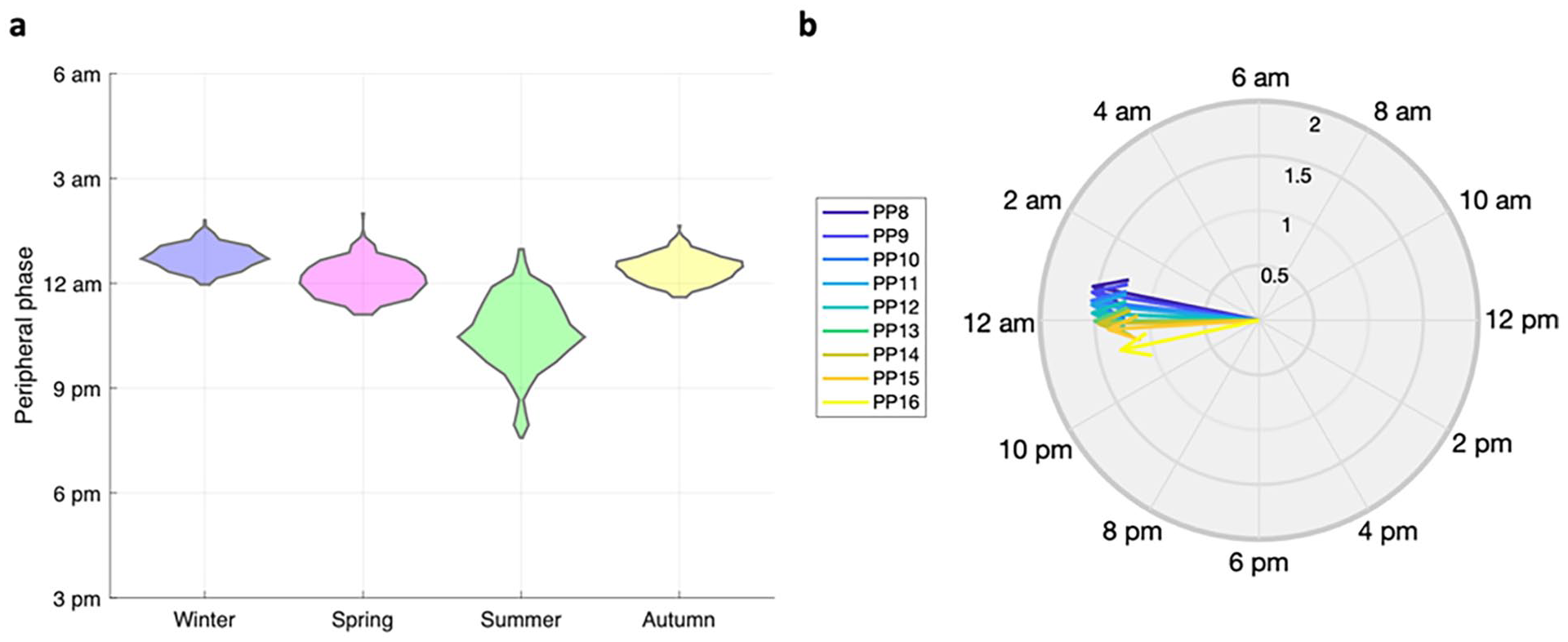
The impact of photoperiod-induced variations in the hypothalamic-pituitary-adrenal (HPA) axis on peripheral clock synchronization. (a) The phase distribution of peripheral Per/Cry mRNA under different photoperiods. The widest phase distribution occurs during the summer, in agreement with the behavior patterns of organisms in long-photoperiod months. (b) Dynamics of the peripheral Per/Cry mRNA under different photoperiod schedules. Regulated by the photoperiod-responsive glucocorticoid rhythms, the peripheral cells exhibit a higher level of amplitude during short photoperiods (SP) and a lower amplitude during long photoperiods (LP). Together, the figure illustrates that the photoperiod-induced variations in the HPA axis play a crucial role in regulating the peripheral clock. Abbreviations: PP = photoperiod; mRNA = messenger ribonucleic acid.
Impact of Photoperiod and Time Zone Changes on Jetlag Readaptation Time
Light schedule disruptions due to long-distance flights in contemporary society can result in notable circadian misalignment among different compartments of the CTS, which is associated with travel fatigue, sleep loss, and decreased physical and mental performance (Bin et al., 2020). However, the impact of photoperiod zone transitions is often overlooked in research on travel fatigue and jetlag (Wieczorek et al., 2013, 2016). We aim to investigate the consequences of both lighting zone and time zone transitions using our model.
We vary the duration of light exposure in lighting zone transition schedules, starting from short photoperiod (SP) of 8 h to long photoperiod (LP) of 16 h and vice versa, to study the effects of lighting zone transition. As shown in Figure 6a, a quick synchronization process occurs when the system moves from LP to SP, along with a phase advance at the ensemble level. Conversely, when the system transitions from SP to LP, a gradual desynchronization takes place with a phase delay, suggesting that prolonged exposure to light can cause a phase delay in the circadian clock (Figure 6b). Similarly, we subject the system to a
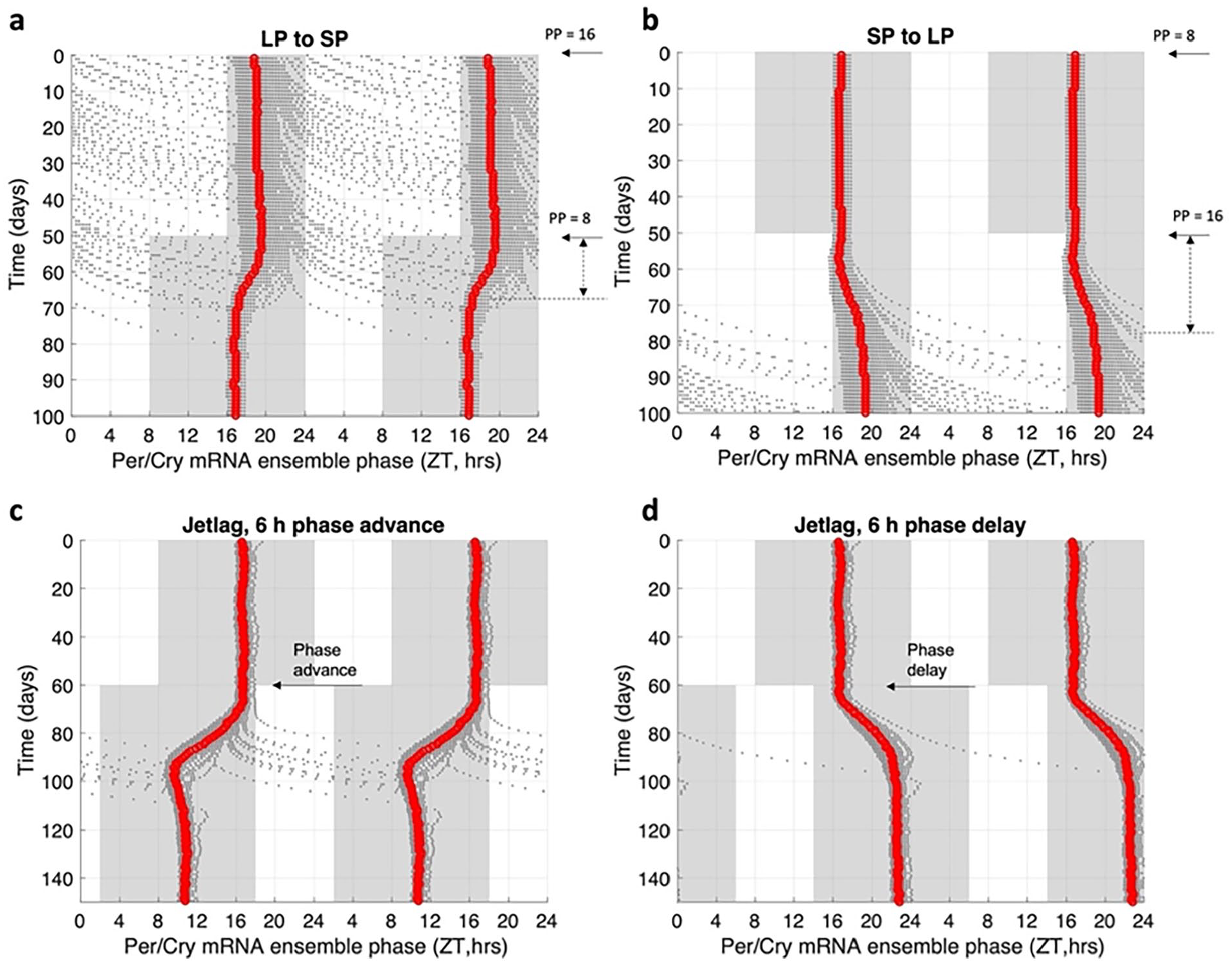
Peripheral clocks dynamics during light schedule changes. The transition of peripheral Per/Cry mRNA and protein concentrations were analyzed when (a) transitioning from long photoperiod (LP, PP = 16) to short photoperiod (SP, PP = 8) and when (b) transitioning from SP to LP. The phase transition of the peripheral ensemble upon lighting zone shifting was also examined. Results indicate that the system takes longer to transition from SP to LP compared to LP to SP, with a recovery time of 18 days for the transition from LP to SP and 27 days for the transition from SP to LP. Furthermore, a phase delay was observed when transitioning from SP to LP. (c) Desynchronization of the peripheral ensemble upon exposure to jetlag during a 6 h phase-advanced jetlag schedule and (d) a 6 h phase-delayed jetlag schedule. The photoperiod before and after jetlag remains constant at 8 h (PP = 8). The red line in panels represents the ensemble average phase calculated from Per/Cry mRNA, while the black dots represent the phase calculated from individual cells. In addition, the system exhibits a higher level of desynchronization and a longer recovery time during phase-advanced jetlag schedules. Abbreviations: ZT = zeitgeber time; mRNA = messenger ribonucleic acid.
To investigate the adaptability of the system under different photoperiod transitions across time zones, we defined the starting point A and the destination point B, each with its own photoperiods. The difference in light onset between A and B is defined as the time zone crossed. Jetlag is considered advanced if the light onset in B is earlier than that in A and delayed if occurs later. We varied factors such as photoperiods in point A (
In Figure 7, we present an example to investigate the interaction between jetlag and photoperiod transitions. We plotted the jetlag transition time and phase shift under a
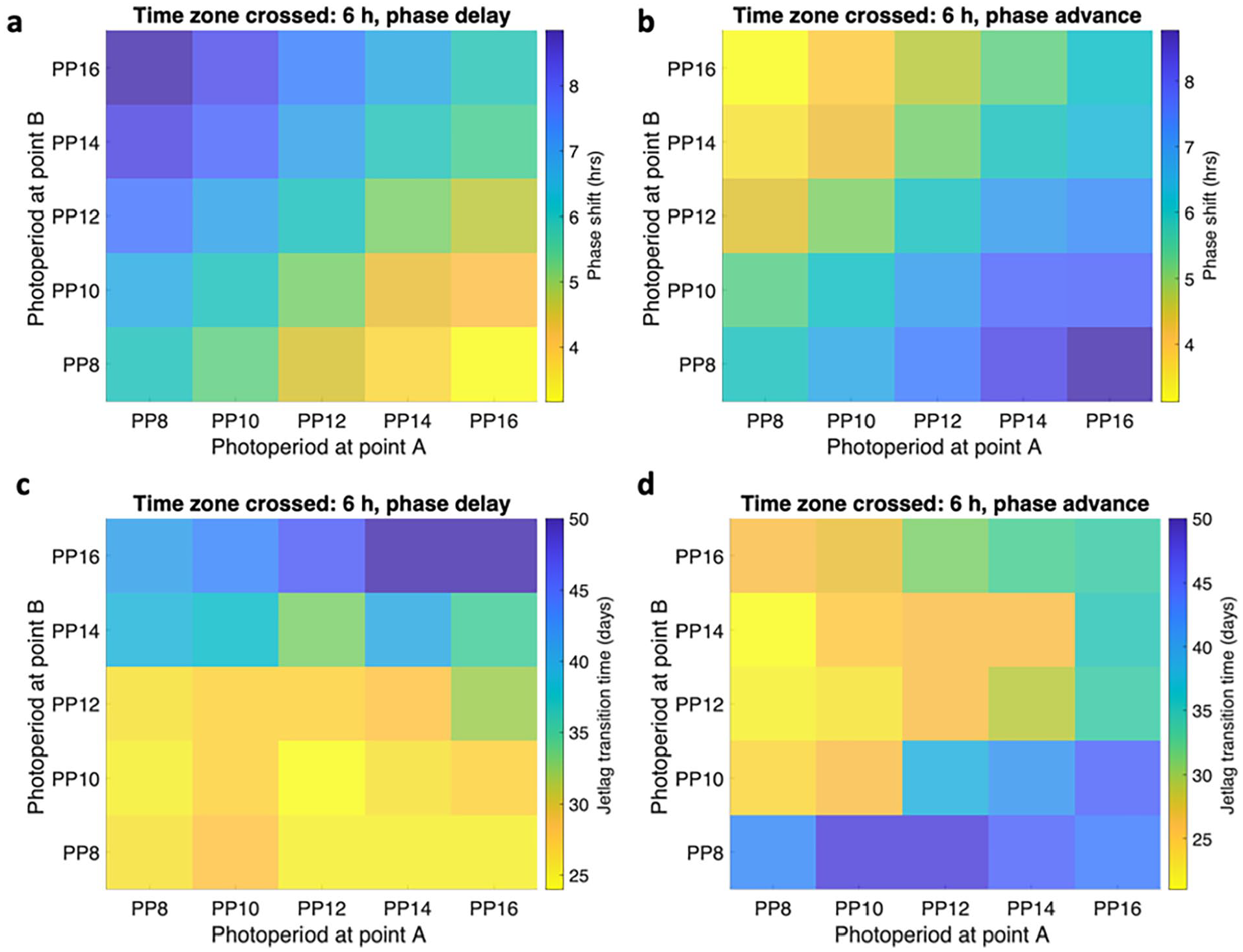
The interplay between lighting zone transition and time zone transition as demonstrated through the measurement of Per/Cry mRNA in the peripheral ensemble. Panel (a) shows the phase shift under a 6-h delayed schedule, panel (b) shows the phase shift under a 6-h advanced schedule, panel (c) shows the jetlag transition time under a 6-h delayed schedule, and panel (d) shows the jetlag transition time under a 6-h advanced schedule. The system is considered fully recovered when the peripheral ensemble peaking phase lies within a 1-h range of the new steady-state phase. The time difference between the onset of jetlag and the establishment of a new steady state is recorded as the jetlag recovery time, and the phase difference between the old steady state and the new steady state phase in the peripheral ensemble is recorded as a phase shift. An increase in phase difference is observed with an increased photoperiod exposure after jetlag during phase delay schedules, and a decrease in phase difference is observed with an increased photoperiod exposure after jetlag during phase advance schedules. Jetlag transition time exhibits a positive correlation with the phase shift. Abbreviations: PP = photoperiod; mRNA = messenger ribonucleic acid.
To quantitatively assess the impact of various light schedule factors on the systemic transition response, we analyzed the peripheral response under various jetlag schedules and conducted multiple linear regression analyses separately for the advanced and delayed cases. Models with the form of
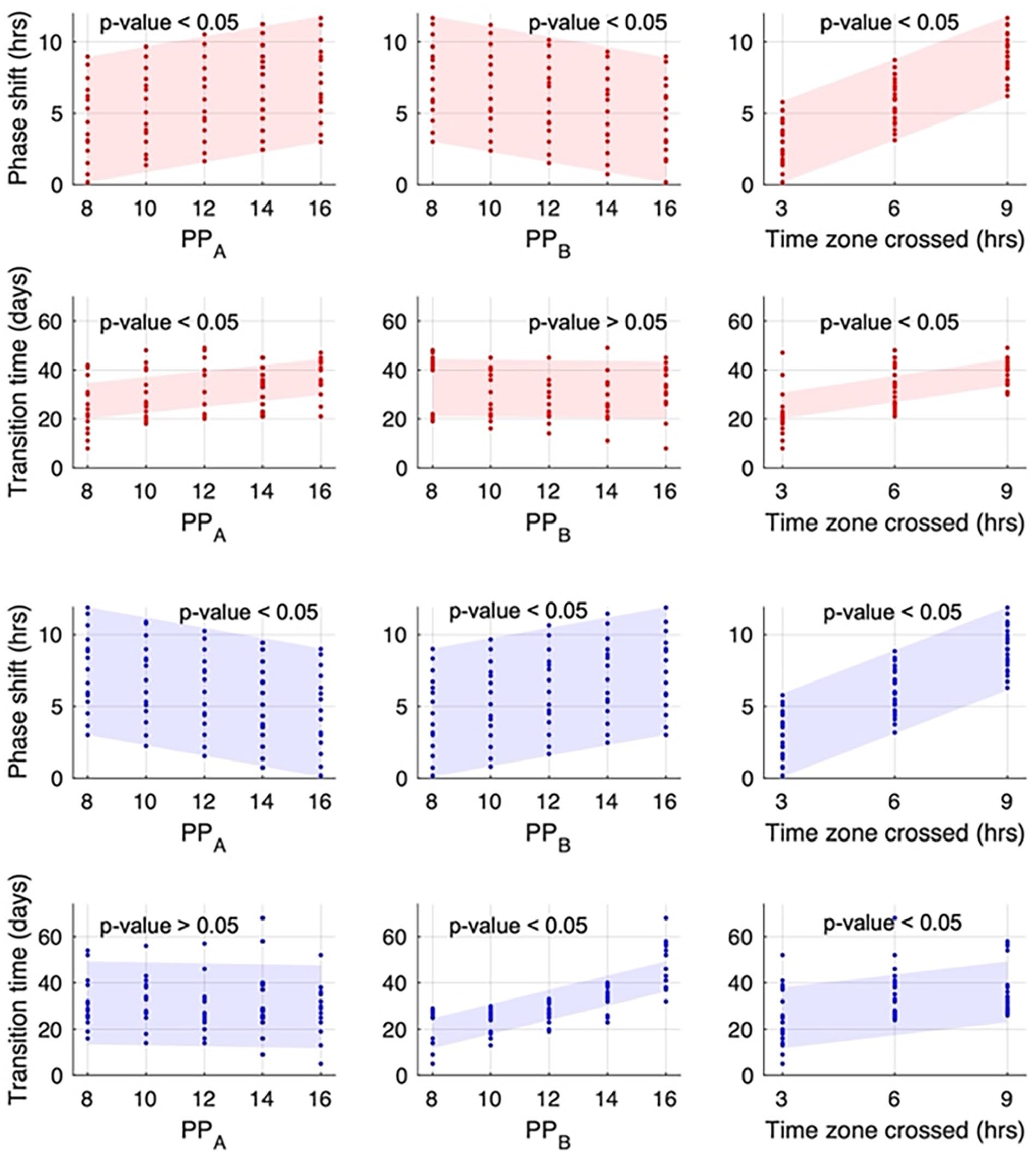
Linear regression analysis of the impact of photoperiod before and after jetlag, and time zone crossing on phase shift and jetlag transition time. Red dots represent data obtained from phase-advanced jetlag simulations, while blue dots represent data from phase-delayed schedules. Shadows represent the predicted range from the linear regression model. A linear regression model of
Light schedule factors exert different effects on the jetlag transition time depending on the direction of jetlag. In advanced schedules, both the time zone crossed and the photoperiod at point A significantly impact the jetlag transition time, while during delayed schedules, the time zone crossed and the photoperiod at point B are predicted to play a role. The jetlag transition time generally increases as the time zone crossed increases in both phase advance and phase delay schedules. In addition, our findings suggest that the jetlag transition time shows a significant decrease as the photoperiod at point A decreases in phase advance schedules, and when photoperiod at point B decreases in phase delay schedules.
Discussion
The CTS plays a crucial role in synchronizing an organism’s biological rhythms to the external light/dark cycles, facilitating successful adaptation to the environment. To gain a deeper understanding of the mechanisms underlying this hierarchically organized system, we developed a mathematical model that incorporates several key compartments of the CTS, including the SCN, the HPA axis, and the peripheral clocks. Building upon our previous work (Li and Androulakis, 2021, 2022), this model enabled us to conduct a range of novel investigations, such as characterizing hierarchical entrainment, exploring photoperiod-induced seasonal responses, and analyzing interactions between lighting zone and time zone transitions during jetlag.
Our study aimed to gain a deeper understanding of the mechanisms underlying the synchronization of peripheral clocks with external light/dark cycles. One crucial factor in this process is the circadian rhythm of glucocorticoids, which are regulated by the SCN and act as a synchronizer of peripheral clocks (Balsalobre et al., 2000; Cuesta et al., 2015). Our simulation results showed that the HPA axis, which serves as a “bridge” between the central and peripheral clocks, plays a critical role in synchronizing peripheral biological activities (Figure 2). Specifically, our analysis of the entrainment dynamics of the peripheral clocks revealed that when entrained in vivo, the downstream peripheral compartments exhibit an insensitive response when exposed to light entraining signals with extreme periods. This observation suggests a potential benefit to the organism’s homeostasis (Figure 3). It is worth noting that our findings do not contradict previous studies by Abraham et al. (2010). In their research, the entrainment ranges of both the SCN and peripheral compartments were tested under the direct entrainment of two identical entrainers. However, in our study, the SCN is directly entrained by light, whereas the downstream peripheral compartments are directly entrained by signals originating from upstream compartments. As a result, our results offer complementary insights into the entrainment dynamics of peripheral clocks in vivo, further enriching our understanding of the intrinsic homeostatic mechanisms within the CTS.
By incorporating the photoperiod-related SCN neuronal plasticity, our model exhibits responses to photoperiod variations, with glucocorticoids amplitude rising as photoperiod decreases (Figure 4). This phenomenon has also been observed experimentally in various species, where glucocorticoid levels are highest during winter months and lowest during summer months (Walker et al., 1997; Bilbo et al., 2002; Owen et al., 2005). In temperate regions with significant day length variation, photoperiod serves as a potent entrainer of immune function and physiological plasticity, which may explain some of the variability in disease incidence and severity over the year. For example, winter is generally associated with increased co-regulated proinflammatory gene expression and interleukin-6 (IL6) signaling (Dopico et al., 2015; Pierre et al., 2017). Elevated glucocorticoid levels during winter may be associated with the increased activity of numerous inflammatory immune responses (Pierre et al., 2017). Moreover, the onset of seasonal affective disorder (SAD) is often observed during winter, with symptoms reducing in summer and spring, which could lead to winter depression. Although the mechanisms of SAD are unclear, it is hypothesized that SAD is associated with delayed circadian rhythms during winter (Lewy et al., 2007). Consistent with this hypothesis, our model identified a short-photoperiod-dependent phase delay (Figures 4 and 5), suggesting that the peripheral phase delay during winter months may serve as a mechanism inducing SAD.
According to the model, varied glucocorticoid amplitude is predicted to exhibit differential synchronization effects on peripheral cells (Figure 5). Experimentally, the duration of daily activity in mammals is positively correlated with the photoperiod, with reduced activity during winter to reduce metabolic expenditure (Kenagy, 1973; Daan and Aschoff, 1975). In our model, the peripheral phase is narrowly distributed during winter while spread out during summer, which may lead to broader behavior patterns as a mechanism for adapting to longer photoperiods. It is important to acknowledge that the current model does not take into account all the factors associated with seasonal variations that may impact the periphery, such as fluctuations in temperature, variations in light intensity, changes in stress levels, and the regulatory role of the melatonin (Xu et al., 2018). However, our model highlights the potential limitations of lower amplitude glucocorticoids in synchronizing peripheral dynamics under prolonged photoperiods. In addition, it is hypothesized that high light intensity during summer months may significantly alter entrainment ability on the periphery, and further research incorporating various seasonal-related factors is expected to be conducted in the future to explore this possibility.
During jetlag, peripheral cells can become desynchronized due to the misalignment between biological clocks and social time. Our previous study reported that recovering from jetlag is correlated with an individual’s chronotype and light sensitivity, implying that bright light therapy might alleviate jetlag-associated fatigue (Li and Androulakis, 2021). However, few studies have investigated the impact of photoperiod on jetlag recovery. In this study, we sought to investigate the effect of light exposure length on jetlag recovery time and phase shift (Figures 6–8). Although the time required for the oscillatory system to transition from one state to another in our model may be longer than what is observed in certain experimental settings, our model considers a more complex network in which different compartments are interconnected, which may result in slower resynchronization times. Our goal is to provide a theoretical framework for comprehending the overall dynamics of the system rather than predicting specific quantitative outcomes. In addition, the faster synchronization rate observed in vivo may be explained by the potential synergistic effects of multiple zeitgebers under physiological conditions. Our simulation results indicate that different interventional strategies are required for eastward (phase advance) and westward (phase delay) jetlag, as significant influencing factors differ between the two. Adjusting to a phase-advanced schedule is more challenging, with a more significant positive correlation between the number of time zones crossed and jetlag transition time compared to that of a phase-delayed schedule.
Notably, our study suggests that in phase-advanced schedules, reducing light exposure before eastward traveling can mitigate jetlag symptoms, while decreasing light exposure after jetlag can accelerate recovery during phase-delayed schedules. Our results also demonstrated a complex positive correlation between intrinsic phase shift and the jetlag transition time (Suppl. Fig. S7). When the phase shift of the host is moderate (below ~10 h), a greater phase shift is likely to result in a longer jetlag transition period. However, as the phase shift increases, jetlag recovery time loses its sensitivity to respond to phase shift changes, indicating a more severe disruption of the circadian rhythm. According to these predictions, to improve jetlag recovery, it is suggested to minimize the intrinsic phase shift by decreasing the light exposure before eastward traveling and after westward traveling. Our predictions have the potential to inspire more experimental designs focused on developing non-pharmacological interventions to alleviate jetlag through optimized light exposure before and after the transition. In addition, further investigation is anticipated to explore other methods, such as adjusting feeding time and exercise (as suggested by Bae and Androulakis, 2017; Bin et al., 2019), for preserving the intrinsic phase of the system.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304231188541 – Supplemental material for The SCN-HPA-Periphery Circadian Timing System: Mathematical Modeling of Clock Synchronization and the Effects of Photoperiod on Jetlag Adaptation
Supplemental material, sj-docx-1-jbr-10.1177_07487304231188541 for The SCN-HPA-Periphery Circadian Timing System: Mathematical Modeling of Clock Synchronization and the Effects of Photoperiod on Jetlag Adaptation by Yannuo Li and Ioannis P. Androulakis in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-2-jbr-10.1177_07487304231188541 – Supplemental material for The SCN-HPA-Periphery Circadian Timing System: Mathematical Modeling of Clock Synchronization and the Effects of Photoperiod on Jetlag Adaptation
Supplemental material, sj-xlsx-2-jbr-10.1177_07487304231188541 for The SCN-HPA-Periphery Circadian Timing System: Mathematical Modeling of Clock Synchronization and the Effects of Photoperiod on Jetlag Adaptation by Yannuo Li and Ioannis P. Androulakis in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-3-jbr-10.1177_07487304231188541 – Supplemental material for The SCN-HPA-Periphery Circadian Timing System: Mathematical Modeling of Clock Synchronization and the Effects of Photoperiod on Jetlag Adaptation
Supplemental material, sj-xlsx-3-jbr-10.1177_07487304231188541 for The SCN-HPA-Periphery Circadian Timing System: Mathematical Modeling of Clock Synchronization and the Effects of Photoperiod on Jetlag Adaptation by Yannuo Li and Ioannis P. Androulakis in Journal of Biological Rhythms
Footnotes
Acknowledgements
The authors acknowledge support from NIH GM131800.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Simulations and analysis were performed in MATLAB_R2021a (The MathWorks, http://www.mathworks.com). Additional data and materials generated in this work are available in a public GitHub repository at ![]() .
.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
