Abstract
Hibernation is a dormant state of some animal species that enables them to survive harsh environmental conditions during the winter seasons. In the hibernating state, preservation of neuronal rhythmic activity at a low level is necessary for maintenance of suspended forms of behavior. As glial cells support rhythmic activity of neurons, preservation of brain function in the hibernating state implies accompanying modification of glial activity. A supportive role of glia in regulating neuronal activity is reflected through the activity of inwardly rectifying K+ channels (Kir). Therefore, we examined electrophysiological response, particularly Kir current response, of glial cells in mixture with neurons acutely isolated from active and hibernating land snail Helix pomatia. Our data show that hibernated glia have significantly lower inward current density, specific membrane conductance, and conductance density compared with active glia. The observed reduction could be attributed to the Kir currents, since the Ba2+-sensitive Kir current density was significantly lower in hibernated glia. Accordingly, a significant positive shift of the current reversal potential indicated a more depolarized state of hibernated glia. Data obtained show that modification of glial current response could be regulated by serotonin (5-HT) through an increase of cGMP as a secondary messenger, since extracellular addition of 5-HT or intracellular administration of cGMP to active glia induced a significant reduction of inward current density and thus mimicked the reduced response of hibernated glia. Lower Kir current density of hibernated glia accompanied the lower electrical activity of hibernated neurons, as revealed by a decrease in neuronal fast inward Na+ current density. Our findings reveal that glial response is reduced in the hibernating state and suggest seasonal modulation of glial activity. Maintenance of low glial activity in hibernation could be important for preservation of brain rhythmic activity and survival of the animal.
Hibernation is a natural physiological adaptation of some invertebrate and vertebrate species to meet the challenge of unfavorable environmental conditions during the winter seasons. The circannual rhythm of the hibernator is reflected by a period of dormancy characteristic of the hibernating state and period of activity (Kortner and Geiser, 2000). In the hibernating state, which lasts for several months, the animals cease movement and feeding, decrease body temperature, slow heart and breathing rate, and decrease the overall metabolic rate (Storey and Storey, 1990). In the hibernating state, brain activity is maintained at a very low but functionally responsive level (Strumwasser, 1959). In fact, at low temperature, neuronal populations of hibernating animals remain synchronized to produce coherent rhythms (Ruby, 2003) that regulate and maintain suspended forms of behavior.
Modifications in the cells of the nervous system associated with the circannual rhythm can be more easily studied in relatively simple hibernating animal species. One of them, the land snail Helix, is well suited for such studies because of the simple organization and experimental accessibility of its nervous system. When isolated, Helix nervous system cells survive well and maintain stable electrophysiological properties. Previous research has revealed that various modifications occur in the nervous system of the hibernating land snail. As indicated by the literature, modifications associated with circannual rhythm could be under the control of neurotransmitters and neuropeptides, which act as modulators of brain activity (Hiripi and Salanki, 1973; Bernocchi et al., 1998; Michaelidis et al., 2002; Hernadi et al., 2008; Pirger et al., 2010). The most important role in preparing the snail for entry into hibernation, its maintenance, and arousal has been attributed to serotonin (5-HT), since an increase in the level of this neurotransmitter has been observed in the snail brain during hibernation while a decrease in the level accompanied arousal of snails (Hiripi and Salanki, 1973; Michaelidis et al., 2002). Previous studies revealed marked modifications in neuronal physiology during hibernation. Thus, immunohistochemistry revealed an increase in the phosphorylated form of the neurofilament protein suggested to contribute to soma stability in hibernation (Vignola et al., 1995). The same study pointed to a change in Ca2+-mediated signaling, since both an increase in calmodulin immunoreactivity and a decrease in Ca2+- ATPase activity were detected in hibernated neurons. Moreover, recently obtained data indicated that the electrical activity of snail neurons was lower in hibernation. Thus, lower expression of Nav 1.8 channel and unaltered expression of Nav 1.9 channel, accompanied by reduced fast and slow Na+ currents carried through these channels, respectively, were observed in the brain of the hibernating snail (Kiss et al., 2014). Marked modifications in glia morphology have also been shown in the hibernating snail. Compared with active glia, glial processes in hibernation were reduced and formed a more regular compact sheath around the neuronal soma. Furthermore, unlike in the active state, glial processes invaded neuronal cell bodies more superficially in hibernation (Fenoglio et al., 1997). The same study also detected lower activity of alkaline phosphatase and Ca2+/Mg2+-ATPase in hibernated glia, indicating that glial activity might be modified in hibernation.
Glial cells act in partnership with neurons to preserve nervous system stability and function (Benarroch, 2005; Barres, 2008; Ransom and Ransom, 2012). Fundamental features of glial function are common to nervous systems of invertebrates and vertebrates (Lieberman et al., 1994; Prosser et al., 1994; Deitmer et al., 1999; Smit et al., 2001; Barres, 2008; Araque and Navarrete, 2010; Ng et al., 2011; Stout et al., 2014). Namely, glial cells provide trophic support for neurons, respond to neuronal firing by activation of ion channels, modify the reuptake of neurotransmitters released by neurons, and modulate rhythmic activity of neurons. In addition, a common selection of neurotransmitter receptors has been identified in invertebrate and vertebrate glia (Deitmer et al., 1999; Kettenmann and Steinhauser, 2005). Neuronal signaling is highly sensitive to the perturbation of K+ concentration in extracellular space. Glial cells are entrusted with a crucial role of supporting proper neuronal activity by tightly regulating K+ content in the neuronal microenvironment. In part, such regulation is achieved through an inwardly rectifying K+ (Kir) channel highly expressed in the membrane of both invertebrate and vertebrate glial cells (Orkand et al., 1966; Gommerat et al., 1993; Gommerat and Gola, 1994; Kofuji and Newman, 2004; Butt and Kalsi, 2006). Generally, glial Kir channels activate in response to neuronal firing and eliminate excess K+ released after neuronal activity from extracellular space. The defining electrophysiological feature of the Kir channel is its property to pass current better in the inward than the outward direction (Hibino et al., 2010). Additional important electrophysiological characteristics of this channel are its inhibition by external BaCl2 and CsCl (Ransom and Sontheimer, 1995; Gommerat and Gola, 1996).
Despite the involvement of glial cells in almost every aspect of brain function, very little is known about the electrophysiological properties of these cells in hibernation. Therefore, in the present study, we have examined the electrophysiological response of acutely isolated hibernated glial cells in mixture with neurons in a simple experimental model, the hibernating land snail Helix pomatia. Furthermore, having in mind the low electrical activity of invertebrate and vertebrate neurons in hibernation (Strumwasser, 1959; Kiss et al., 2014) and the importance of glial Kir channels in regulating the neuronal microenvironment, we have examined the Kir currents of isolated hibernated glial cells. As 5-HT is considered to have an important role in regulating the hibernation state (Hiripi and Salanki, 1973; Michaelidis et al., 2002), we have also examined its effect on glial currents. We report here reduced electrophysiological activity of hibernated glial cells.
Materials and Methods
Animals
Snail H. pomatia Linnaeus (Pulmonata: Helicidae) usually enters hibernation in late autumn and remains in a dormant state until April or May (Boss, 1974). Accordingly, adult land snails were locally collected (Belgrade, 44°49′N 20°28′E) in late October and divided into 2 groups. One group of snails was kept at a temperature of 22 °C, regularly wetted and fed, and thus maintained in an active condition. The other group of snails was placed in a dark cold chamber at a temperature of 5 °C to induce hibernation. Hibernation began in November and lasted until the beginning of May. Hibernating snails used for experiments had their apertures sealed with thick calcareous epiphragm and several thin secondary epiphragmal membranes equivalent to the hibernating state of animals in the natural environment (Boss, 1974).
Acute Dissociation of Snail Ganglion Cells
The experiments were carried out on glial cells and neurons acutely isolated from active and hibernating H. pomatia. According to previous research, cells isolated from dormant animals show stable intrinsic metabolic depression (Bishop and Brand, 2000; Guppy et al., 2000; Ross et al., 2006).
The brain–subesophageal ganglion complex was isolated according to a standard dissecting technique. Briefly, after the shell was dissected, the animal foot was pinned onto a cork-lined Petri dish in the extended position. Incision at the dorsal anterior surface of the extended snail’s foot enabled access to the ganglion complex. Enzymatic treatment followed by mechanical dissociation of the subesophageal ganglion complex was performed according to a previously described procedure (Ivic et al., 1995). Previous research has shown that an approach using only mechanical trituration of isolated leech ganglia provides for the presence of only neurons in primary culture (Meis and Deitmer, 1997). Thus, we decided to follow the procedure involving enzymatic treatment of the snail ganglion using the proteolytic enzyme pronase to separate neurons from glial cells (Balaban et al., 1990). Ganglion complex isolated from active (n = 8 animals) or hibernated (n = 6 animals) snails was placed in a solution of 0.5% pronase (protease from Streptomyces griseus, type IV) dissolved in (mM) 100 NaCl, 4 KCl, 3 MgCl2, and 7 CaCl2 and incubated in a water bath for 90 to 100 min at 32 to 34 °C. Unless otherwise noted, here and below, all chemicals were supplied by Sigma-Aldrich (Taufkirchen, Germany). We reduced the incubation time in pronase from the initial 2 h 15 min reported in previous research (Ivic et al., 1995), as we have noticed that reducing the time of incubation provides glial cells with noticeable processes and neurons with longer axons. Isolated snail brain was rinsed 3 times for 5 min with extracellular solution (ECS) composed of (in mM) 100 NaCl, 5 KCl, 5 MgCl2, 7 CaCl2, and 10 Hepes, pH 7.4 adjusted with NaOH. Next, under the dissecting microscope and using ophthalmological scissors and fine tweezers, we carefully removed the connective tissue sheath over the neuronal cell bodies, leaving the neuronal surface mostly intact. Parietal, visceral, pleural, and pedal ganglia, including neuropile, were removed, cut in fragments, and placed in ECS supplemented with 10 mM glucose. Dissociation of cells was accomplished by repeated gentle suction of ganglia with fire-polished Pasteur pipettes of decreasing tip diameter. This stage of the isolation process required certain care as insufficient trituration yielded a too small number of cells whereas excessive trituration yielded more damaged cells. Dissociated cells (200 µL of suspension) were plated on 15 mm coverslips (Manzel-Glasser, Braunschweig, Germany) coated with concanavalin A (Con A type IV). Coating was performed by incubating the coverslips in a Con A solution (1 mg of ConA per 1 mL of deionized water) for 2 h and rinsing the coverslips several times with deionized water and ECS before use. Cells were allowed to adhere to Con A–coated coverslips for 30 min (3 coverslips per animal). Electrophysiological experiments were performed within 3 h after cells adhered to the coverslips. Individual cells were imaged by a MicroQ UCMOS Camera (Toup Tek Photonics, Hangzhou, China) under the same settings as used to visualize the cells during electrophysiological experiments. Cell body diameters were measured with ToupView software (Toup Tek Photonics).
Whole-Cell Patch-Clamp Recordings
Electrophysiological recordings from glial cells and neurons dissociated from the ganglia of active and hibernated snails were obtained with the whole-cell configuration of the patch-clamp technique (Hamill et al., 1981). A coverslip with plated cells was placed in the recording chamber on the stage of an inverted Zeiss Axiovert 10 (Carl Zeiss, Jena, Germany) or Olympus BH2 upright microscope (Olympus, Center Valley, PA). Patch-clamp recordings were performed using an A-M Systems 2400 amplifier (A-M Systems, Carlsborg, WA) with Clampex 7 software (Axon Instruments, Sunnyvale, CA) or a Tecella Pico 2 amplifier with TecellaLab software (Tecella, Foothill Ranch, CA). Patch pipettes were made from borosilicate glass capillaries of 1.5 mm outer and 0.86 mm inner diameter (Science Products, Hofheim, Germany) pulled on a P-97 Flaming/Brown Micropipette Puller (Sutter Instrument, Novato, CA) just before use. Whole-cell patch-clamp recordings were conducted on freshly dissociated cells placed in ECS supplemented with 10 mM glucose of ~245 mOsm osmolarity. The intracellular solution had the following composition (in mM): 3 NaCl, 100 KCl, 1 MgCl2, 5 EGTA, and 10 Hepes, pH 7.4 adjusted with KOH with an osmolarity of ~225 mOsm. When indicated, 50 mM cyclic 3′,5′-guanosine monophosphate (cGMP) was added to the intracellular solution. Patch pipettes had a resistance of 3 to 5 MΩ. Freshly prepared inhibitors CsCl, BaCl2, and CdCl were directly added to the recording chamber at concentrations of 1 mM, 100 µM, and 50 µM, respectively. Stock solutions of serotonin creatinine sulfate (5-HT) were prepared in deionized water and kept at −20 °C. Working solutions of 5-HT were diluted in ECS on the day of the experiment. 5-HT was added directly to the recording chamber at a 5 µM concentration (Zenchenko et al., 2000). The effects of all substances were estimated 2 min after addition to the recording chamber, a time sufficient for added substances to evenly reach all parts of the recording chamber. The current responses were measured from 5 to 6 cells per active or hibernated animal. Responses from glial cells and neurons were always obtained from the same animals throughout the experiments. Membrane currents were low-pass filtered at 2 kHz and digitized at 10 kHz using a Digidata 1200 interface (Axon Instruments) or a Tecela Pico2 amplifier with integrated digitizer (Tecella). Membrane capacitance and series resistance were measured directly from the amplifiers, with an upper limit for series resistance less than 25 MΩ. All recordings were obtained at room temperature (22 °C). Before applying voltage step protocols, the cells were voltage clamped at a holding potential of −50 mV. Whole-cell currents were evoked by stepping the holding potential from −150 to +70 mV in 20 mV increments. The current reversal potential (Erev) of active and hibernated glial cells was determined from the tail current protocol by stepping the holding potential of −50 mV to a prepulse potential of −150 mV for 100 ms and then to a series of test potentials ranging from −100 to −30 mV in 10 mV increments. After a prepulse that evokes inward currents, channels close quickly but not instantaneously and make tail currents visible. The Erev was determined as the intersection of the tail current density-voltage curve with the voltage axis.
Currents evoked by voltage step protocols were analyzed in Clampfit software (Axon Instruments). Only cells with a stable recording time of minimum 4 min were included in the analysis of voltage-activated currents, as the others were considered to be damaged during the dissociation procedure. The amplitudes of evoked steady-state currents were measured 25 ms before the end of the 300 ms potential step relative to the currents at the holding potential. The amplitudes of tail currents were measured 6 ms after the onset of the test pulses. Amplitudes of the fast voltage-sensitive inward currents were measured at the current peaks at the −50, −30, −10, +10, +30, and +50 mV voltage steps. Current density was calculated by dividing the current amplitude by the membrane capacitance. Membrane conductances were calculated from the slope of the linear fit of the inward current component between −130 and −90 mV of the current–voltage curves obtained with the step protocol. The values of membrane conductances were divided by membrane capacitance values to obtain specific membrane conductances independent of cell size. Cs+- and Ba2+-sensitive currents were obtained by digitally subtracting current traces after block from control traces using Clampfit software (Axon Instruments).
Statistical Analysis of Data
Data were plotted and analyzed using SigmaPlot software (Systat Software Inc., San Jose, CA). All data are given as the mean ± SEM, with n indicating the number of cells examined. Student’s t test and paired t test for dependent samples were used to evaluate the differences between the groups of data that passed the normal distribution test. Mann-Whitney test was used to evaluate the differences between the groups of data that did not pass the normal distribution test. One-way analysis of variance was used to analyze the difference between more than 2 groups of data. The difference between compared groups was considered significant for p values less than 0.05.
Results
Characterization of Glial Cells
As there are no data on the physiological properties of isolated snail glial cells, we began the study by characterizing glial cells in mixture with neurons acutely isolated from active snails. Following the isolation procedure, dissociated snail ganglion cells exhibited well-defined morphologies characteristic of glia and neurons (Tauc, 1967; Fenoglio et al., 1997; Pantazis et al., 2006; Banks et al., 2009). Namely, isolated glial cells had small spindle-shaped cell bodies tapering at each pole into long fine processes (Fig. 1A, left panel), while large neurons were unipolar and bore marked axonal processes (Fig. 1A, middle panel). The cell body diameter of glial cells (n = 13) was 5 times smaller and significantly different (p < 0.001) from that of neurons (n = 15; Fig. 1A, right panel). In response to the same voltage protocol, whole-cell current responses of glial cells and neurons were clearly distinct. Examples presented in Figure 1B illustrate that glial cells exhibit noticeable inward currents of slightly higher amplitudes than outward currents, while neurons display prominent outward currents (Fig. 1B, right panel). In addition, glial cells uniformly show an absence of the fast voltage-sensitive inward currents typically observed in neurons (Fig. 1B, inset). The current amplitude-voltage profile of glial cells (n = 22) was weakly inwardly rectifying and noticeably different from that of neurons characterized by outward rectification (n = 22; Fig. 1B, lower panel). To further ascertain glial physiology, we examined the presence of Kir currents in the membrane of isolated glial cells under our experimental conditions. For that purpose, we applied CsCl and BaCl2 at concentrations that selectively block the currents mediated through these channels (Ransom and Sontheimer, 1995; Olsen et al., 2007). External 1 mM CsCl significantly diminished (p < 0.01) inward currents (Fig. 2A; n = 6), while 100 µM BaCl2 significantly reduced both inward (p < 0.01) and outward currents (p < 0.05, n = 5; Fig. 2B), evidently showing that Kir channels are expressed in the membrane of isolated active glial cells and that their current represents a prominent component of the inward current. Observed changes were in accordance with the previously described effects of Cs+ and Ba2+ on Kir currents of mammalian glia (Ransom and Sontheimer, 1995).
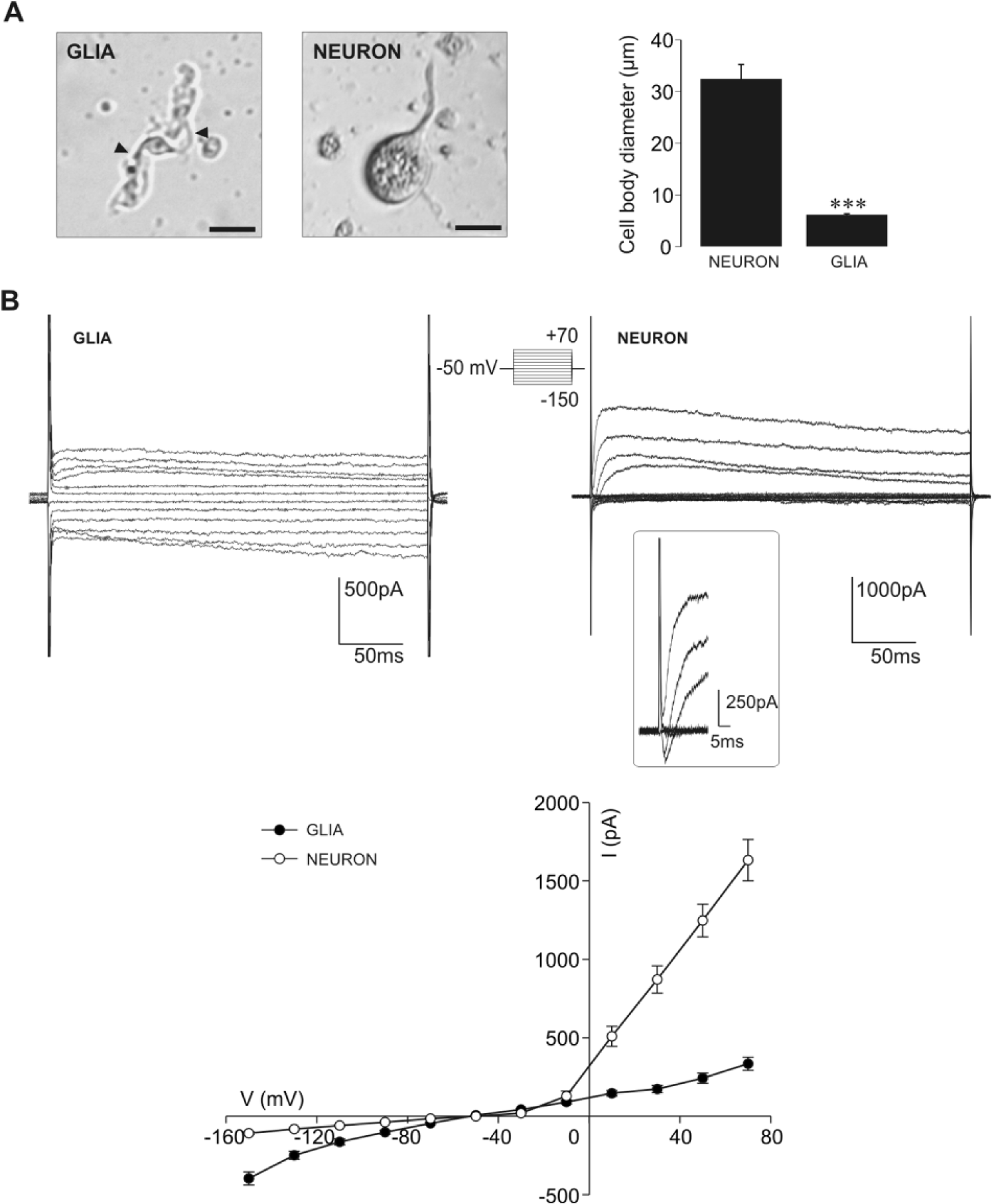
Distinct morphologies and whole-cell current responses of isolated active glial cells and neurons. (A) Images of snail glial cell and neuron illustrate prominent morphological differences (scale bars 20 µm). The small, spindle-shaped cell body of the glial cell bears 2 longer processes (depicted by arrowheads), while the large, oval neuronal cell body bears only 1 process corresponding to the axon. The mean value of the cell body diameter of glial cells (n = 13) is significantly smaller compared with that measured for neurons (n = 15, ***p < 0.001, Mann-Whitney test). (B) Examples of current recordings obtained from glial cell and neuron depict prominent differences in the whole-cell currents evoked by the same voltage protocol. Glial cells exhibit noticeable inward currents of slightly higher amplitude than outward currents. Hyperpolarizing voltage steps evoked only small inward currents in neurons. Neurons typically exhibit a fast voltage-sensitive inward current followed by a prominent outward current. Inset: fast inward currents at potentials ranging from −50 to +50 mV at a faster time base. Note the lack of the fast voltage-sensitive inward currents in the glial cell. Current-amplitude voltage curves depict weakly inwardly rectifying profile of glial cells (black circles, n = 22) and outwardly rectifying current-voltage profile of neurons (white circles, n = 22).
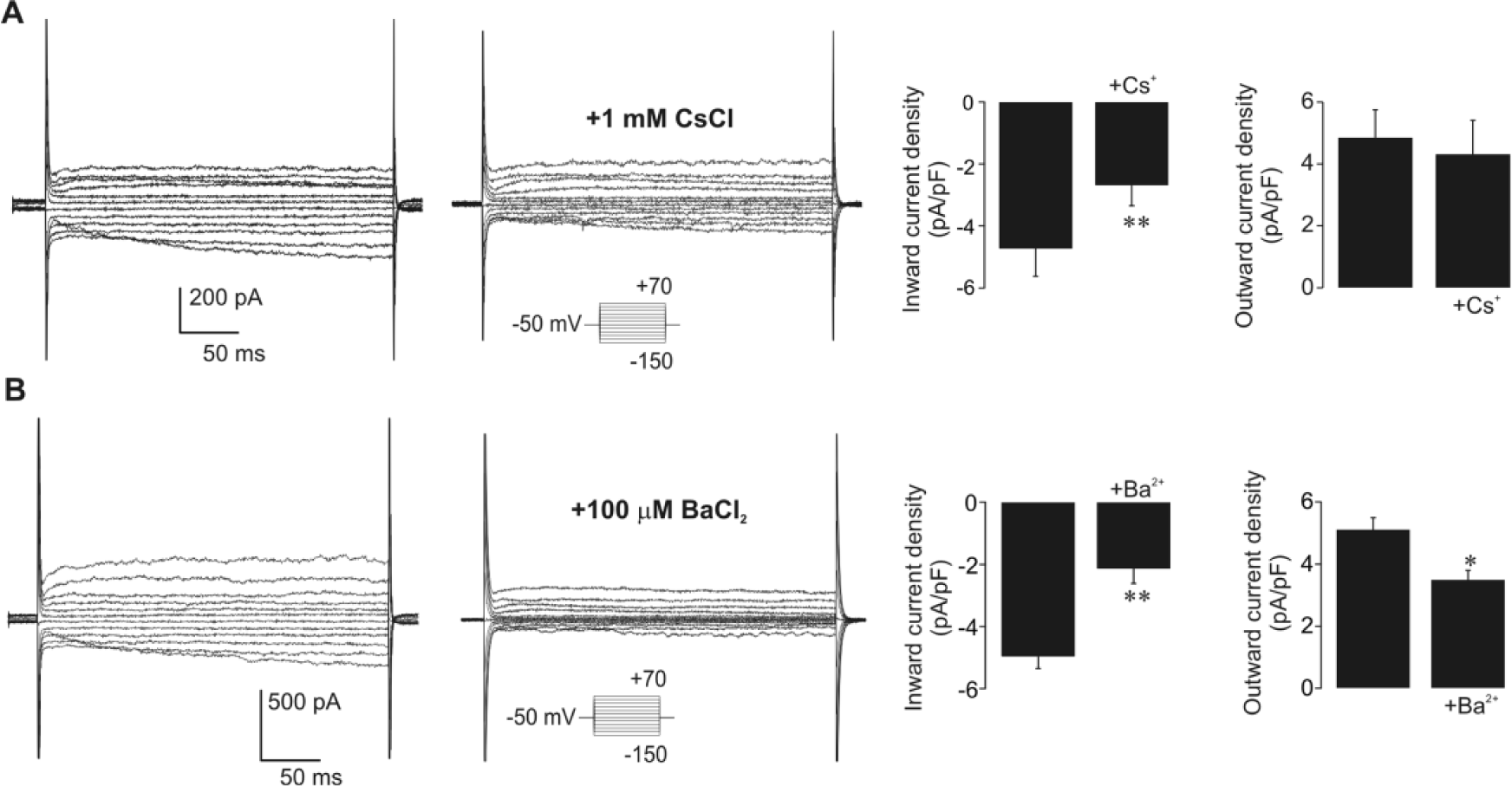
Isolated active glial cells possess prominent inwardly rectifying K+ (Kir) currents. (A) Example of voltage step current responses of glial cells without and in the presence of CsCl applied at a concentration that selectively inhibits Kir currents. In the presence of 1 mM CsCl, inward currents of glial cells were noticeably decreased. The voltage protocol is indicated in the inset. Mean data demonstrate significant diminution of inward current density in glial cells as estimated at −130 mV (**p < 0.01, paired t test, n = 6). Cs+ block was voltage dependent, as the density of outward glial currents (at +50 mV) was not significantly altered in its presence. (B) Example of voltage step current responses of glial cells without and in the presence of 100 µM BaCl2. Addition of Ba2+ at a concentration that selectively inhibits Kir induced a prominent diminution of both inward and outward currents in glial cells. Voltage protocol is indicated in the inset. Significant decrease of inward (at −130 mV) and outward (at +50 mV) current densities in the presence of Ba2+ demonstrate the less specific voltage dependence of this ion block (*p < 0.05, **p < 0.01, paired t test, n = 5).
Current Responses of Active and Hibernated Glial Cells
We next examined the current responses of hibernated glia and compared them with those obtained from active glia. Figure 3A illustrates examples of current recordings of glial cells isolated from active snails relative to glial cells isolated from hibernating snails. It can be seen that whole-cell currents of glial cells in both examined physiological states shared weakly rectifying current profiles. However, the inward currents of hibernated glial cells had a noticeably smaller amplitude than those of active glia (Fig. 3A). To limit current amplitude variation due to size differences among glial cells, we next obtained current densities of active and hibernated glial cells. By plotting current densities against the stepped membrane potentials, we observed that the differences in the current responses between active and hibernated glia were specific to the inward current, since outward current densities of hibernated glia were similar to those measured in active glia (Fig. 3B). Analysis of current responses to −130 mV step revealed that the glial inward current density was significantly lower in hibernation compared with that in the active state (p < 0.01; hibernated glia: −3.10 ± 0.36 pA/pF, n = 12; active glia: −4.96 ± 0.35 pA/pF, n = 22; Fig. 3B). The observed decrease of the inward current density was associated with a significantly lower specific membrane conductance of hibernated glial cells (p < 0.001; hibernated glia: 45.79 ± 4.79 pS/pF, n = 12; active glia: 73.23 ± 4.73 pS/pF, n = 22; Fig. 3C). With the use of 1.0 µF/cm2 as the specific capacitance of the membrane lipid bilayer (Ransom and Sontheimer, 1995), we found that conductance density is approximately 2 times reduced in the hibernating state (active: ~1.0 pS/µm2; hibernated: ~0.5 pS/µm2).
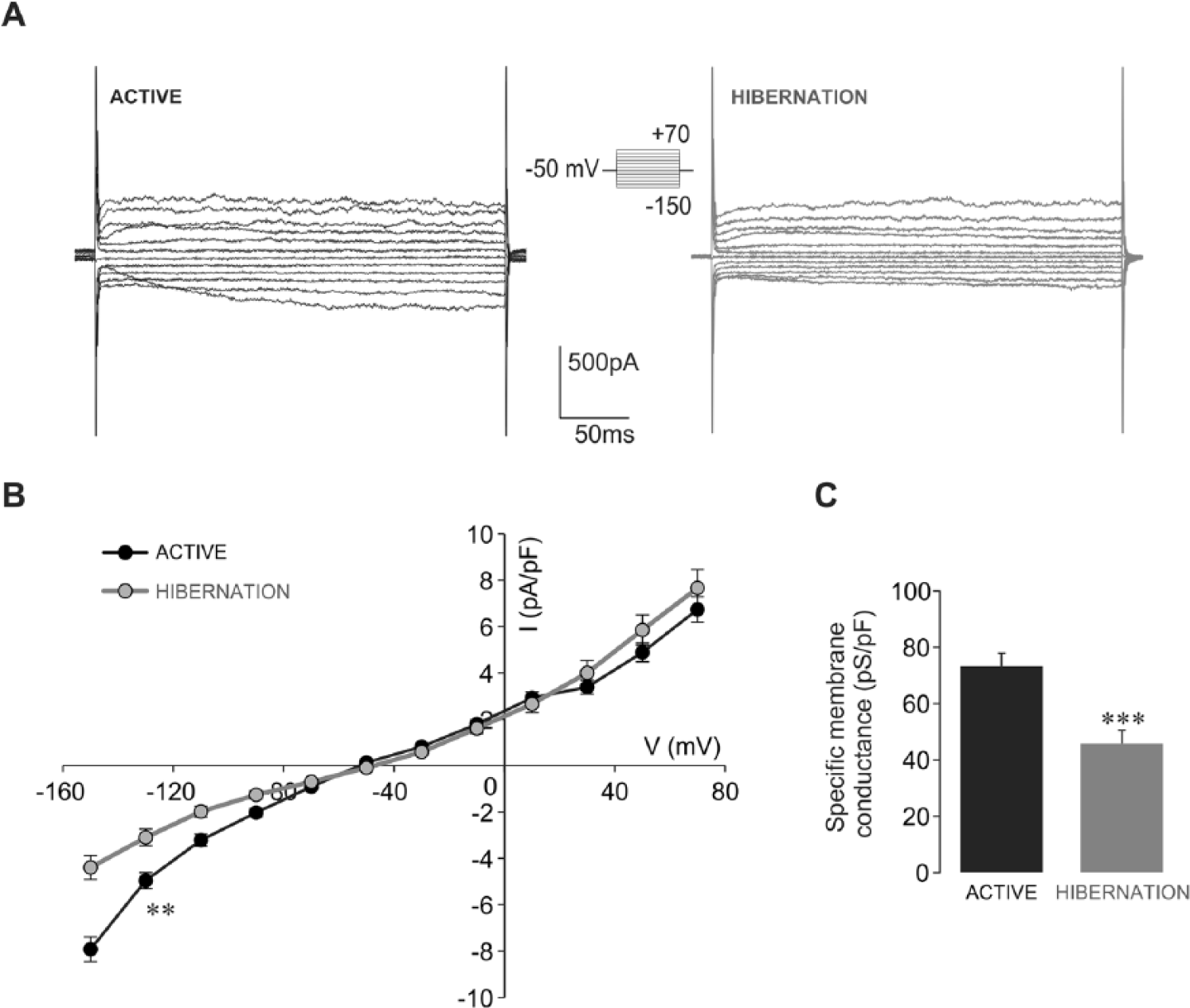
Inward current density and specific membrane conductance are reduced in isolated hibernated glial cells. (A) Examples of voltage step current responses depict reduced amplitudes of inward currents of hibernated glia relative to those of active glia (protocol shown in inset). (B) The current density-voltage relationship of active (black circles) and hibernated (gray circles) glial cells reveals that inward current density is significantly lower in hibernated glia as estimated at −130 mV (**p < 0.01, t test, hibernated: n = 12, active: n = 22). Outward current densities of hibernated glia were not significantly different from those measured from active glia. (C) Specific membrane conductance is significantly lower in hibernated glial cells (***p < 0.001, t test, hibernated: n = 12, active: n = 22).
Next, we examined if observed changes in the inward current and membrane conductance of hibernated glia could be attributed to the Kir current. Examples presented in Figure 4A depict marked differences in the amplitude of the inward component of the Ba2+-sensitive Kir current between active and hibernated glial cells. The corresponding current density-voltage relationship revealed a several-fold reduction of inward Ba2+-sensitive Kir current in hibernated glia. Analysis at the −130 mV step (Fig. 4B) showed that the observed reduction was statistically significant (p < 0.01; hibernated glia: −0.42 ± 0.12 pA/pF, n = 6; active glia: −3.09 ± 0.61 pA/pF, n = 5).
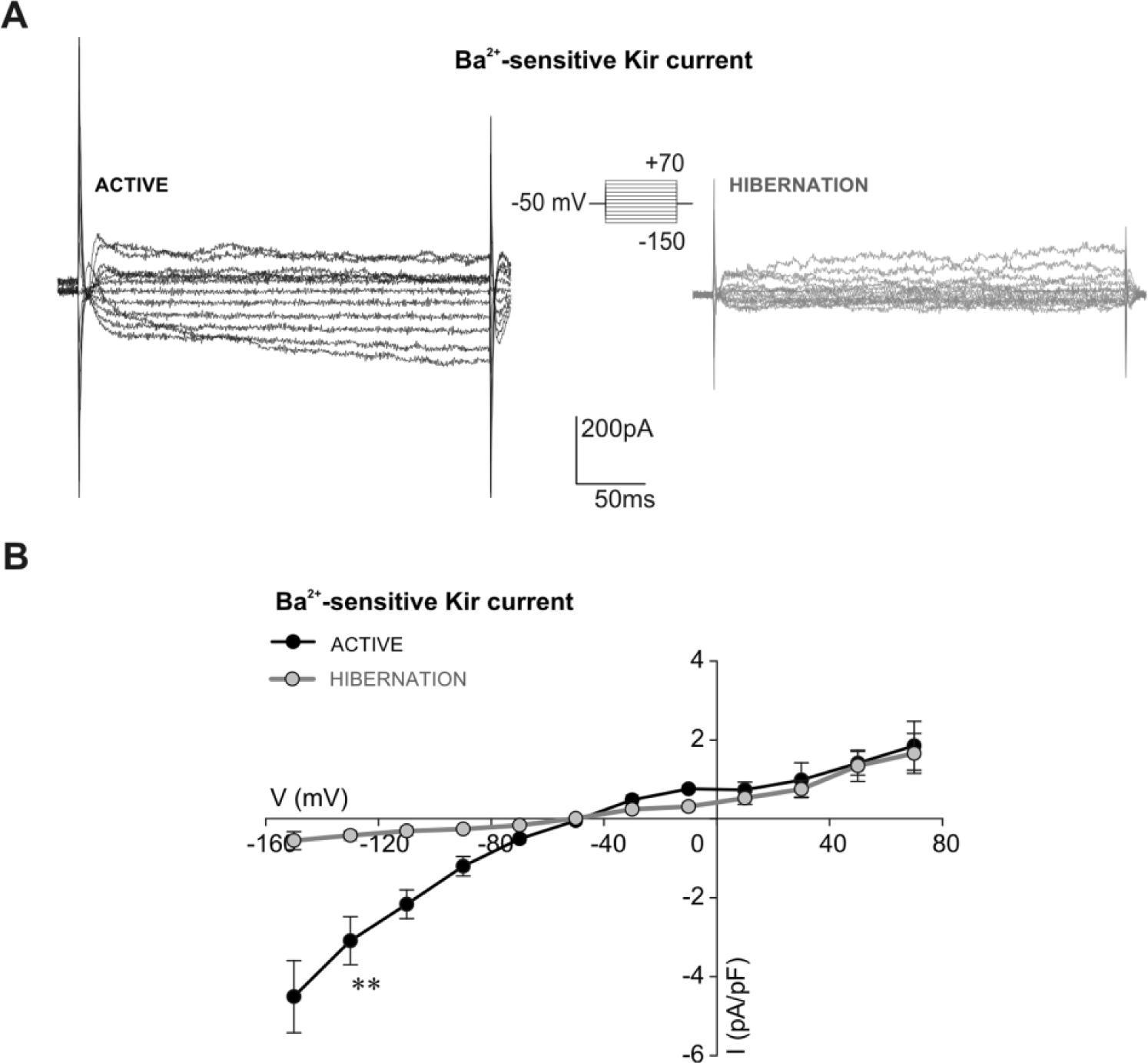
Inward component of Ba2+-sensitive Kir current is reduced in isolated hibernated glial cells. (A) Examples of Ba2+-sensitive Kir currents of active and hibernated glial cells obtained after addition of 100 µM BaCl2. Amplitudes of the inward component of the Ba2+-sensitive Kir current were noticeably smaller in hibernated glial cells. Amplitudes of the outward component of the Ba2+-sensitive Kir current were similar in active and hibernated glia. The voltage protocol is shown in the inset. (B) Current density-voltage curves show a significant difference for the inward component of the Ba2+-sensitive Kir current density between hibernated glia (gray circles, n = 6) and active glia (black circles, n = 5) at the −130 mV step (**p < 0.01, Mann-Whitney test).
Reduced Kir current density pointed to a possible change in the resting membrane potential of hibernated glia (Olsen et al., 2007). Kir channels are important for establishing the resting membrane potential of glial cells, which closely follows the K+ equilibrium potential (Ransom and Sontheimer, 1995; Olsen and Sontheimer, 2008). Furthermore, glial currents reverse direction close to the K+ equilibrium potential (Ransom and Sontheimer, 1995). Thus, Erev reflects the resting membrane potential of glia. In our experimental conditions, the determined Erev of active glia (−58.87 ± 1.14 mV, n = 8; Fig. 5) was relatively close to the K+ equilibrium potential according to the Nernst equation (−76.1 mV). However, the Erev of hibernated glia (−51.37 ± 1.15 mV, n = 8) was more positive and significantly different (p < 0.001) compared with that of active glia (Fig. 5). The positive shift of Erev indicated that hibernated glial cells rest at a more depolarized potential relative to active glia.
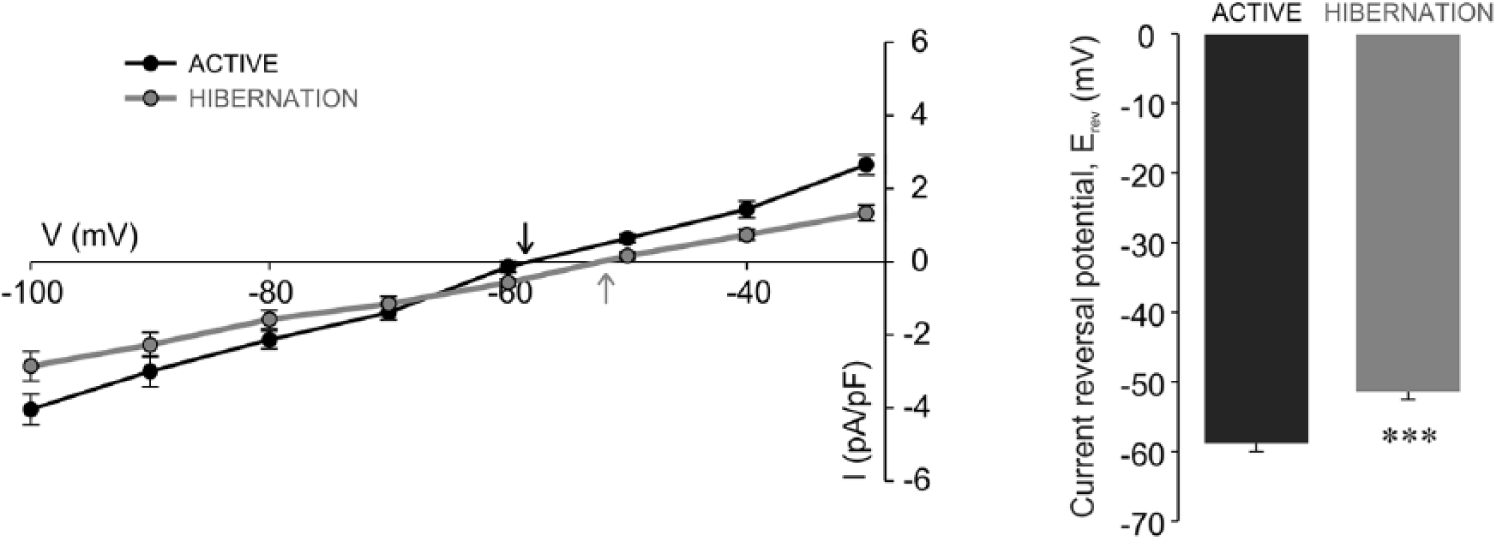
Positive shift of current reversal potential in isolated hibernated glial cells. Tail current density-voltage curves of active (black circles) and hibernated (gray circles) glial cells. The intersection of the current density-voltage curves with the voltage axis determines the current reversal potential (Erev) for active (black arrow) and hibernated (gray arrow) glia. Note the shift of Erev toward more positive values in hibernation. The Erev of hibernated glia (n = 8) is significantly different (***p < 0.001, t test) from the Erev of active glia (n = 8).
Effect of 5-HT on the Current Response of Active Glia
In the snail nervous system, 5-HT is released from varicosities that are embedded in a dense network of glial cells (Elekes, 1991). As 5-HT is considered to have an essential role in regulating the hibernation state in the land snail (Michaelidis et al., 2002), we next tested the involvement of 5-HT in the modulation of glial response that we observed in hibernation. For that purpose, the whole-cell currents of active glial cells were measured in the presence of 5-HT. Examples in Figure 6A show that addition of 5 µM 5-HT induced a prominent reduction of inward currents in active glial cells. By plotting current densities against the membrane potentials, we found that 5-HT significantly suppressed inward current density in active glia (at −130 mV, p < 0.01) while outward current densities were not significantly changed (n = 7; Fig. 6B). Further examination revealed that inward current response of active glia in the presence of 5-HT had a similar profile and amplitude as the inward current observed in hibernated glia. Thus, by comparing the inward current densities (at −130 mV), we were not able to observe any significant difference between hibernated glial cells (−3.10 ± 0.36 pA/pF, n = 12) and active glial cells in the presence of 5-HT (−3.01 ± 0.57 pA/pF, n = 7; Fig. 6C). Since the reduction of the glial inward current induced by application of 5-HT was not immediately apparent, we supposed that secondary messenger cascades could be involved in mediating the 5-HT effect. Previous findings in the snail indicated that the 5-HT effect on neuronal current is mediated by cGMP (Paupardin-Tritsch et al., 1986). Therefore, in the next series of experiments, we examined the effect of cGMP added in a pipette (cGMPin) on the inward current of active glia. Our results showed that the inward current density (at −130 mV) with cGMPin was reduced (−3.36 ± 0.25 pA/pF, n = 7) and significantly different compared with the measured inward current of active glia (Fig. 7C). The subsequent addition of 5-HT to the same cell while cGMPin (Fig. 7A–C) had no further effect (at −130 mV) on the inward current (−3.43 ± 0.19 pA/pF, n = 7). At the same time, there was no significant difference in the inward current density (at −130 mV) between active glia in the presence of 5-HT, active glia with cGMPin, and active glia in the presence of 5-HT while cGMPin (Fig. 7C). In each of these experimental conditions, the inward current densities were similar to those observed in hibernated glia and significantly lower when compared with active glia (Fig. 7C). These data indicated that 5-HT-induced reduction of the inward current of active glia is mediated by cGMP.
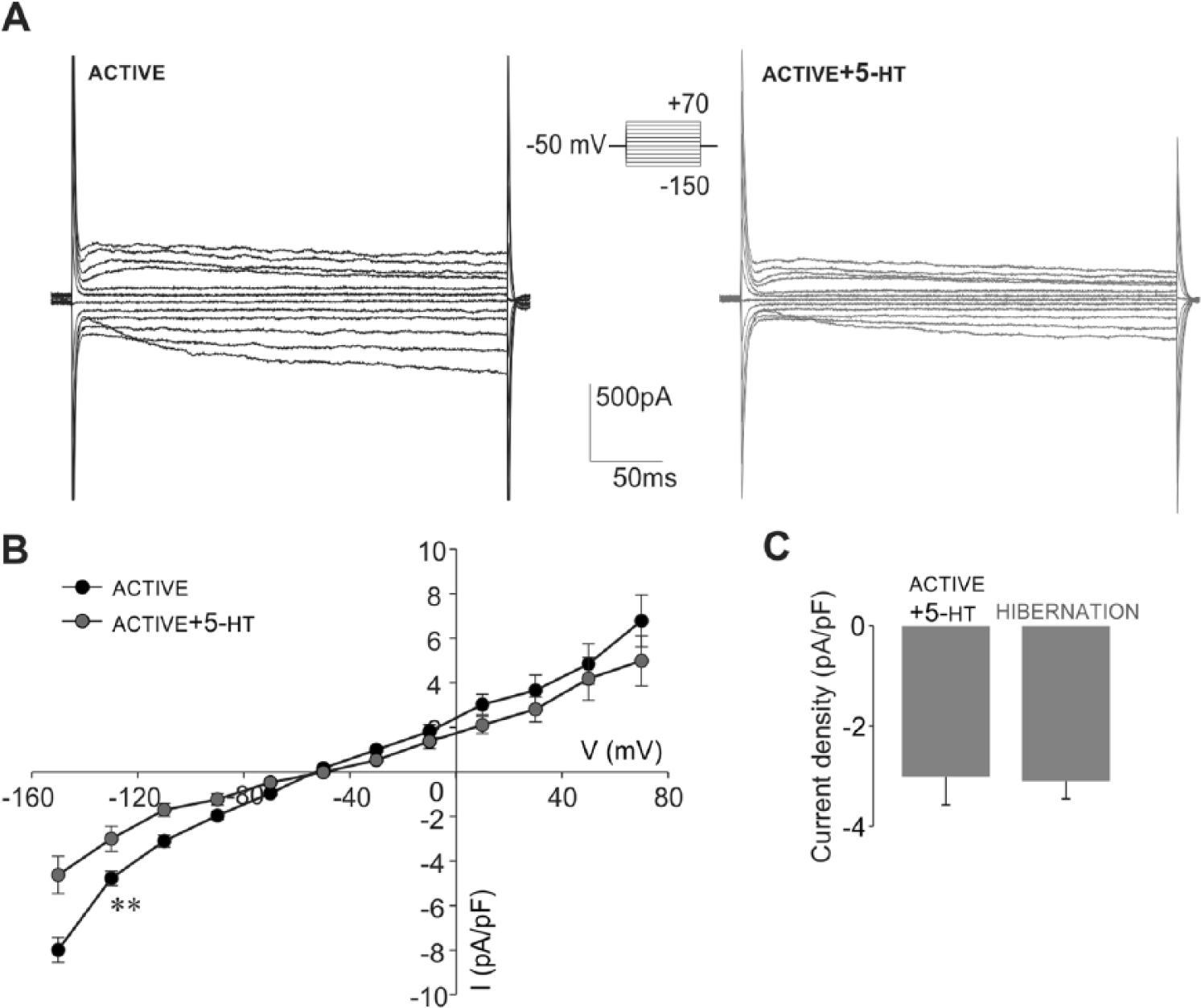
Addition of 5-HT reduces the inward currents in isolated active glial cells and mimics the current response of hibernated glia. (A) Examples of voltage step current responses of active glia before and after addition of 5 µM 5-HT (protocol shown in inset). (B) Current density-voltage curves before (black circles) and after (gray circles) addition of 5-HT revealed that 5-HT induced a significant reduction of inward current density in active glia as estimated at −130 mV (**p < 0.01, paired t test, n = 7). The change in the outward current density was not significant (estimated at +50 mV). (C) The inward current density measured from active glia in the presence of 5-HT (n = 7) is not significantly different from that of hibernated glia (n = 12) as estimated at −130 mV.
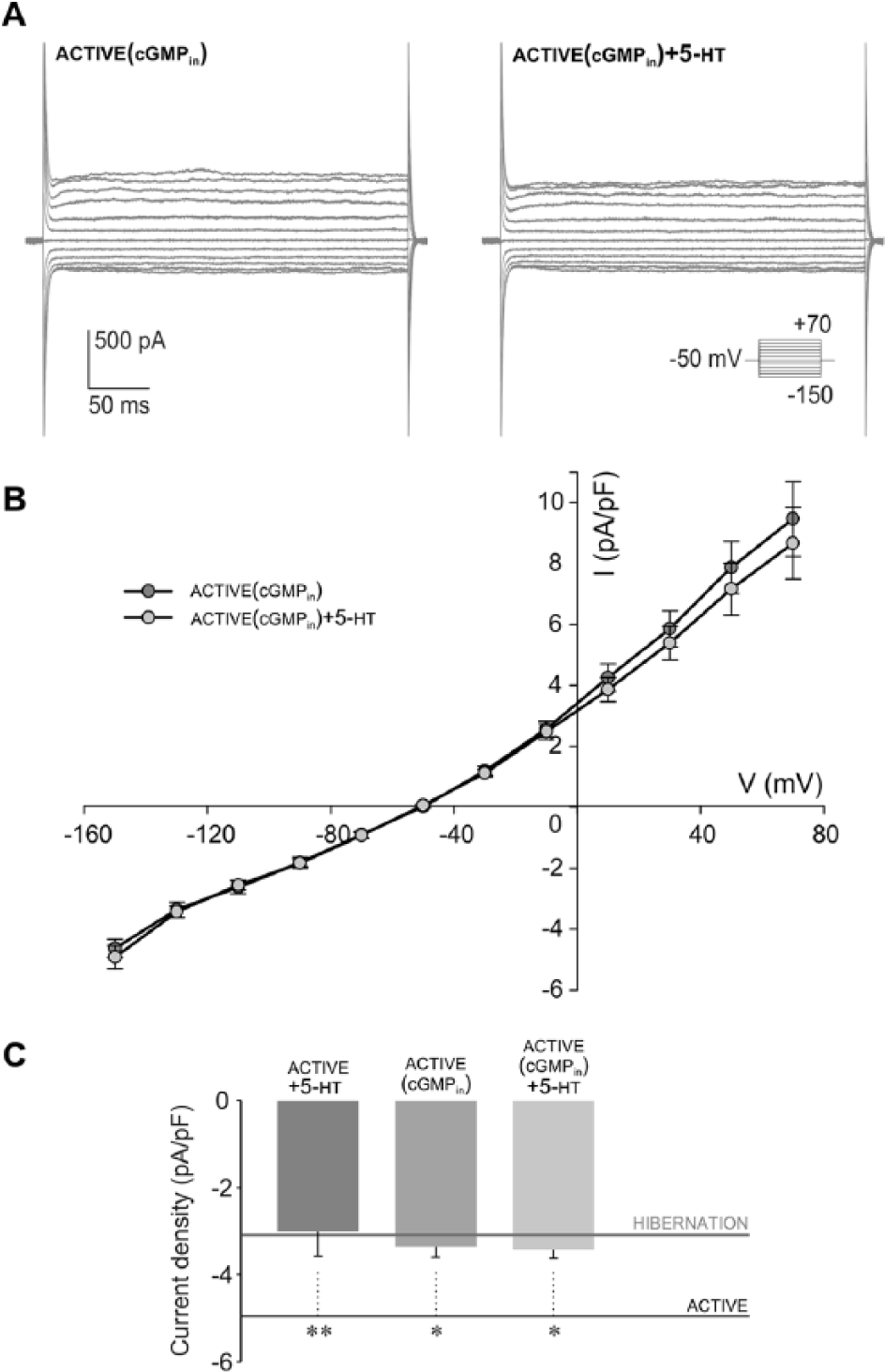
5-HT–induced reduction of inward glial current is mediated by cGMP. (A) Examples of voltage step current responses (protocol shown in inset) of active glia containing 50 mM cGMP in pipette (cGMPin), followed by extracellular addition of 5 µM 5-HT (cGMPin + 5-HT). Note that when cGMPin, subsequent 5-HT application had no effect on the inward current amplitude of active glia. (B) Current density-voltage curves of active glial cells (n = 7) reveal a similarity between the effects of cGMPin (dark gray circles) and extracellularly added 5-HT while cGMPin (cGMPin + 5-HT, light gray circles). (C) The inward current density of active glia (at −130 mV) in the presence of 5-HT (active + 5-HT, n = 7) is not significantly different from that when cGMPin (active [cGMPin], n = 7). Subsequent addition of 5-HT while cGMPin (active [cGMPin] + 5-HT, n = 7) did not cause a significant change in the inward current density (at −130 mV) of active glia. Each experimental condition caused a significant reduction of the inward current density compared with the active state (mean value at −130 mV depicted by black line, n = 22, **p < 0.01, *p < 0.05, 1-way analysis of variance, post hoc Bonferroni t test). The inward current density of active glia in each analyzed experimental condition was reduced and not significantly different compared with that measured in hibernation (mean value at −130 mV, n = 12, depicted by gray line).
Fast Inward Currents of Active and Hibernated Neurons
To examine if the lower Kir current density of hibernated glial cells is associated with lower electrical activity of neurons, we next examined the fast inward Na+ current in hibernated neurons. Current traces presented in Figure 8A depict the difference in fast inward currents between active and hibernated neurons. The corresponding peak current density-voltage plot (Fig. 8A) demonstrated that the fast inward current density of hibernated neurons (−4.56 ± 1.05 pA/pF, n = 14), analyzed at the maximum value at the +10 mV step, was significantly lower (p < 0.05) than that measured from active neurons (−11.10 ± 2.01 pA/pF, n = 22). Since fast inward currents in molluscan neurons are carried by Na+ and Ca2+ (Chen and Tsai, 2000), we next blocked Ca2+ currents by 50 µM CdCl2 and examined the neuronal Na+ currents. Current traces in Figure 8B illustrate that in the presence of Cd2+, fast inward currents of hibernated neurons have lower amplitude compared with that of active neurons. The peak current density-voltage plot (Fig. 8B) revealed that the observed change in the density of the fast inward Na+ current between isolated active and hibernated neurons measured at the +10 mV step is statistically significant (p < 0.05; active: −6.26 ± 1.48 pA/pF, n = 5; hibernated: −2.01 ± 0.80 pA/pF, n = 5). Since fast Na+ currents are responsible for neuronal firing (Catterall, 2000), obtained data indicated that snail neurons have lower electrical activity in hibernation.
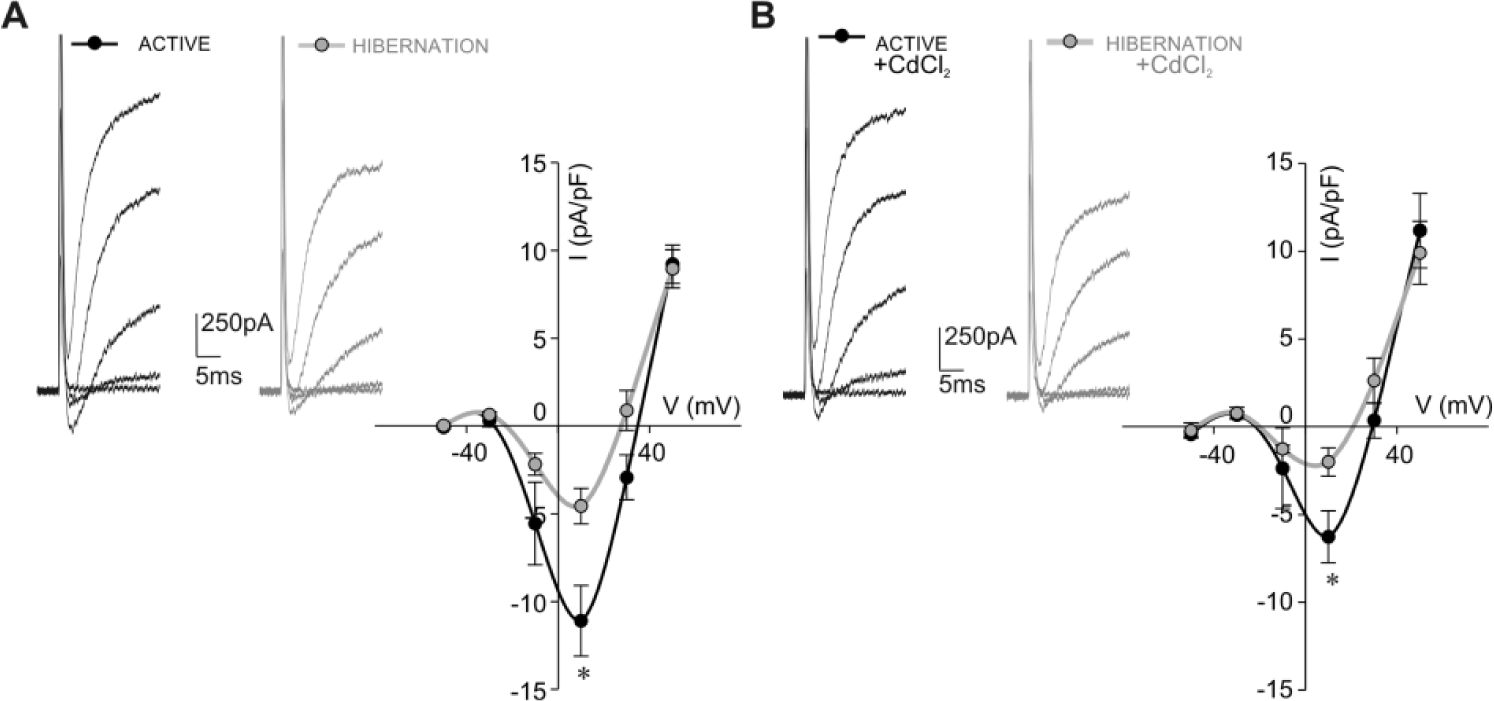
Fast inward Na+ current density is lower in isolated hibernated neurons. (A) Examples of fast voltage-sensitive inward currents of active and hibernated neurons in response to voltage steps ranging from −50 to +50 mV in 20 mV increments. Corresponding peak current density-voltage curves revealed significantly lower fast inward current density of hibernated neurons (gray circles, n = 14) compared with that of active neurons (black circles, n = 22) as estimated at the maximum current peak at +10 mV (*p < 0.05, Mann-Whitney test). (B) Examples of fast voltage-sensitive inward currents of active and hibernated neurons (at potentials from −50 to +50 mV) in the presence of 50 µM CdCl2. The peak current density-voltage plot shows that fast inward Na+ current density (at +10 mV) is significantly lower in hibernated neurons (gray circles, n = 5) compared with that measured from active neurons (black circles, n = 5, *p < 0.05, t test).
Discussion
Previous research has shown that the amplitude of cortical and subcortical neuronal activity is reduced in the brains of hibernating squirrels (Strumwasser, 1959). Furthermore, research performed on identified snail neurons that regulate feeding and avoidance behavior has revealed that these neurons exhibit reduced fast and slow Na+ currents in hibernation (Kiss et al., 2014). Likewise, the data presented in this study show that a decrease of the fast Na+ current density also occurs in a population of neurons acutely isolated from hibernating land snails. Altogether, these data point out that in the hibernating state, the electrical activity of neurons is maintained at a low level. In addition, maintenance of low neuronal activity implies that rhythmic brain activity generated by neuronal populations is preserved in hibernation. In fact, vocalization and complex motor coordination in deeply hibernating squirrels are preserved even though the amplitude of general brain wave activity is reduced by 90% (Strumwasser, 1959). According to previous research, glial cells contribute to the maintenance of neuronal rhythmic activity. Thus, dysfunction of glial cells disrupts brainstem respiratory (Hulsmann et al., 2000) and spinal cord locomotor rhythms (Baudoux and Parker, 2008). Furthermore, as previously shown, perturbation of glial metabolism in rat (Prosser et al., 1994) and glial Ca2+ signaling in Drosophila (Ng et al., 2011) disrupt the circadian rhythm of neuronal activity. Therefore, it would be reasonable to expect that the maintenance of neuronal rhythms in hibernation at a low level requires the adjustment of glial activity. Findings we present in this study show that electrophysiological activity of glial cells is modified in hibernating snails. As the glial resting membrane potential closely follows Erev (Ransom and Sontheimer, 1995), our data on increased Erev value suggest that glial cells are more depolarized in the hibernating state. This observation, based on the positive shift of Erev as determined from tail currents, needs to be interpreted with certain caution. Although the tail current protocol accurately determines the current reversal potential (Molleman, 2003), additional data on the resting membrane potential value will be needed to further assess membrane depolarization of hibernated glia. According to previous research, a more depolarized resting membrane potential can be related to a lower expression of Kir channels in glial membrane (Olsen et al., 2007; Olsen and Sontheimer, 2008). Keeping in mind that the current density is determined by the number of channels per unit of membrane (Nattel, 2008), our data on reduced Kir current density indicate that fewer Kir channels are present on the membrane of hibernated glia. In addition, reduction of the specific membrane conductance and conductance density also supports the premise that there are fewer Kir channels on the glial membrane in the hibernating state. Reduced Kir current density of hibernated glial cells could serve a lower neuronal requirement for K+ clearance (Olsen and Sontheimer, 2008). In this way, reduced glial activity is expected to provide support for the proper functioning of neurons at low levels of activity and contribute to the preservation of nervous system stability during the harsh conditions of hibernation. In fact, absence of strict regulation of K+ concentration in the neuronal microenvironment by glial Kir channels would affect various neuronal processes such as activity of ion channels and synaptic transmission and severely compromise brain function (Kofuji and Newman, 2004; Giaume et al., 2007).
Research performed on hibernating squirrels has pointed to the involvement of 5-HT in the entrance and maintenance of the hibernation state (Spafford and Pengelley, 1971; Haak et al., 1991; Popova et al., 1993). Similarly, previous data obtained in snails has also indicated that 5-HT is involved in the maintenance of the hibernation state, as an elevated level of 5-HT was detected in the brain for almost the whole period of hibernation (Michaelidis et al., 2002). Modulation of neuronal electrical activity by 5-HT has already been observed in hibernating squirrels (Zenchenko et al., 2000). Findings from this study indicate that 5-HT may be involved in maintaining the glial response at low levels in hibernating snails. Thus, addition of 5-HT to active glia reduced inward current density and mimicked the lower electrophysiological response observed in hibernated glia. Our results indicate that the mechanism of 5-HT action on inward glial current involves cGMP as a secondary messenger. Similar to the observed effect of 5-HT alone, the administration of cGMPin reduced the inward current density of active glia to the value observed in hibernation. Subsequent addition of 5-HT in the presence of cGMPin did not induce further reduction of the glial inward current density. That the effects of cGMPin and 5-HT on the inward current density of active glia were not additive suggests that 5-HT and cGMPin acted on the same ion channel through the same signaling pathway. Thus, obtained data argue that in hibernation, 5-HT stimulates formation of cGMP in glial cells. Our findings are in agreement with previous research showing that an increase in cGMP level mediates the 5-HT effect on the snail (Paupardin-Tritsch et al., 1986) and rat (Regina et al., 2003) neurons. We propose that 5-HT–induced modification of the glial current response observed in our study can be attributed to reduction of the Kir component of the inward current, since previous research has shown that 5-HT specifically inhibits currents mediated by Kir channels (Karschin et al., 1994). Our results suggest that 5-HT acted directly on 5-HT receptors known to be present on the glial membrane (Deitmer et al., 1999; Kettenmann and Steinhauser, 2005). In molluscan neurons, 6 subtypes of 5-HT receptors have been pharmacologically and electrophysiologically described (Gerschenfeld and Paupardin-Tritsch, 1974). In addition, the functional expression of molluscan 5-HT receptor subtypes appears to be correlated with the annual rhythm of the animal. Thus, previous research on identified neurons of active and hibernating Helix lucorum has shown that the composition of 5-HT receptor subtypes is changed in hibernation (Sakharov and Korobtsov, 1976). Therefore, investigating the composition of 5-HT receptor subtypes in glial cells in active and hibernating states and the effect of 5-HT antagonists merits further research. At this stage, we can only hypothesize that inhibiting 5-HT receptors in hibernated glia will restore the inward current to the value characteristic of active glia.
The present study complements previous data on morphological modifications of hibernated glia (Fenoglio et al., 1997) and further demonstrates that electrophysiological activity of H. pomatia glial cells is modified in hibernation. We propose that snail glial cells undergo circannual modulation of activity, whereby the hibernating state is characterized by lower glial activity compared with the active state. As brain stability and function depend on the neuron-glia partnership (Barres, 2008), maintenance of glial activity at a low level could be important for supporting neuronal rhythmic activity and survival of animals in the hibernating state under harsh environmental conditions.
Footnotes
Acknowledgements
This work was supported by a grant from the Ministry of Education, Science and Technological Development, Republic of Serbia (grant 173027). The authors are grateful to Sutter Instrument Company for donating the MP-285 Micromanipulator System and Tecella Company for donating the Tecella Pico 2 amplifier.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
