Abstract
The large repertoire of circadian rhythms in diverse organisms depends on oscillating central clock genes, input pathways for entrainment, and output pathways for controlling rhythmic behaviors. Stress-activated p38 MAP Kinases (p38K), although sparsely investigated in this context, show circadian rhythmicity in mammalian brains and are considered part of the circadian output machinery in
Circadian clocks are conserved molecular processes that dictate the daily behavioral rhythms of organisms and play an integral role in the ability of organisms to respond to environmental stimuli and regulate metabolism (Reppert and Weaver, 2002). In
The circadian clock dictates metabolic rates over the course of a day-night cycle (Langmesser and Albrecht, 2006). Interestingly, metabolism is closely linked with oxidative stress caused by reactive oxygen species (ROS). In
Disruptions in the circadian clock by either mutation or environmental factors can lead to a reduced lifespan in many organisms and pathological outcomes in humans such as cancer, cardiovascular disease, metabolic syndromes, and age-associated brain dysfunction (Kondratova and Kondratov, 2012), suggesting a role for circadian rhythms in aging. As organisms age, circadian and sleep rhythms deteriorate (Froy, 2011; Joshi et al., 1999; Ko et al., 2002; Rezaval et al., 2008; Zhdanova et al., 2011; Zheng et al., 2007). This age-dependent deterioration is accompanied by decreased expression of clock genes, such as
We have recently found that the stress response protein kinase p38 MAP Kinase (p38K) is a regulator of lifespan and aging in the fly (Vrailas-Mortimer et al., 2011). The activity of p38K is regulated by dual phosphorylation of the TGY motif, and in a variety of organisms the phosphorylation state of p38K cycles throughout the 24-h day (Chik et al., 2004; Hayashi et al., 2003; Pizzio et al., 2003; Vitalini et al., 2007). In
Materials and Methods
Fly Stocks, Rearing, and Transgenics
Flies were raised at 25 °C in a 12-h:12-h light-dark (LD) cycle on standard cornmeal molasses media. p38KbΔ45, p38KbΔ25, Δp38Ka, UAS-p38Kb wt, UAS-p38Kb ala2, UAS-p38Kb KD3-FLAG, UAS-p38Kb KD8-FLAG, and p38Kb-GAL4 were as described previously (Vrailas-Mortimer et al., 2011). We obtained
Measurement of Circadian Rhythms
For analysis of p38K mutants, either male or virgin female flies 1 to 2 days old were placed individually in
Western Immunoblot Assay
Flies were reared in a 12-h:12-h LD cycle and collected during the second day of entrainment at ZT1, ZT7, ZT13, and ZT19 or placed in constant darkness and collected during the second day of constant darkness at CT1, CT7, CT13, and CT19. Thirty heads from male flies were homogenized in EB1 as described by Chiu et al. (2011) and in 2× Laemmli buffer. Equivalent amounts of each sample were loaded on a 4% gel. Proteins were transferred to PDVF membranes using the manufacturer’s protocols. Antibodies used were rabbit anti-PER (a gift from A. Sehgal, 1:40,000) and rabbit anti-SERCA (Sanyal et al., 2005 1:10,000). Blots were developed using enhanced chemiluminescence (ECL Plus; Pierce/Thermo Scientific) and scanned using a flatbed scanner. All immunoblots were performed in triplicate.
Co-Immunoprecipitation
Flies were reared in a 12-h:12-h LD cycle and collected at ZT1. We expressed 2 copies of p38Kb KD (UAS-p38Kb KD3-FLAG and UAS-p38Kb KD8-FLAG) in clock neurons using Cry-GAL4. Cry-GAL4 outcrossed to
Immunohistochemistry
For analysis of p38Kb expression, flies were reared in a 12-h:12-h LD cycle and collected at ZT2 for PDF costaining and ZT16 for PER costaining. In Lk6 expression analysis, flies were reared in a 12-h:12-h LD cycle and then reared in constant darkness and collected on the second day at CT1, CT7, CT13, and CT19. Flies collected at various time points were fixed in 4% paraformaldehyde at 4 °C for 24 h. Fixed brains were dissected in phosphate-buffered saline; permeabilized in phosphate-buffered saline plus 0.1% Triton X-100 (PBT) for 30 min; blocked in 0.15% Triton X-100, 2% BSA, and 5% NGS (block) for 1 h; and stained overnight in primary antibody solution in block. All washes were carried out in PBT. After secondary antibody staining in block, brains were mounted in Vectashield (Vector Laboratories) and imaged with a Zeiss 510 laser scanning confocal microscope. Lk6 stainings were imaged on the same settings for all genotypes at all time points. Antibodies used were rabbit anti-PER (gift from Amita Sehgal), mouse anti-PDF (DSHB), rabbit anti-Lk6 (gift from Jordan Raff), rabbit anti-βgal (Gibco), and mouse anti-βgal (Gibco).
Results
p38Kb Is Expressed in Clock Neurons
There are two p38K homologs in
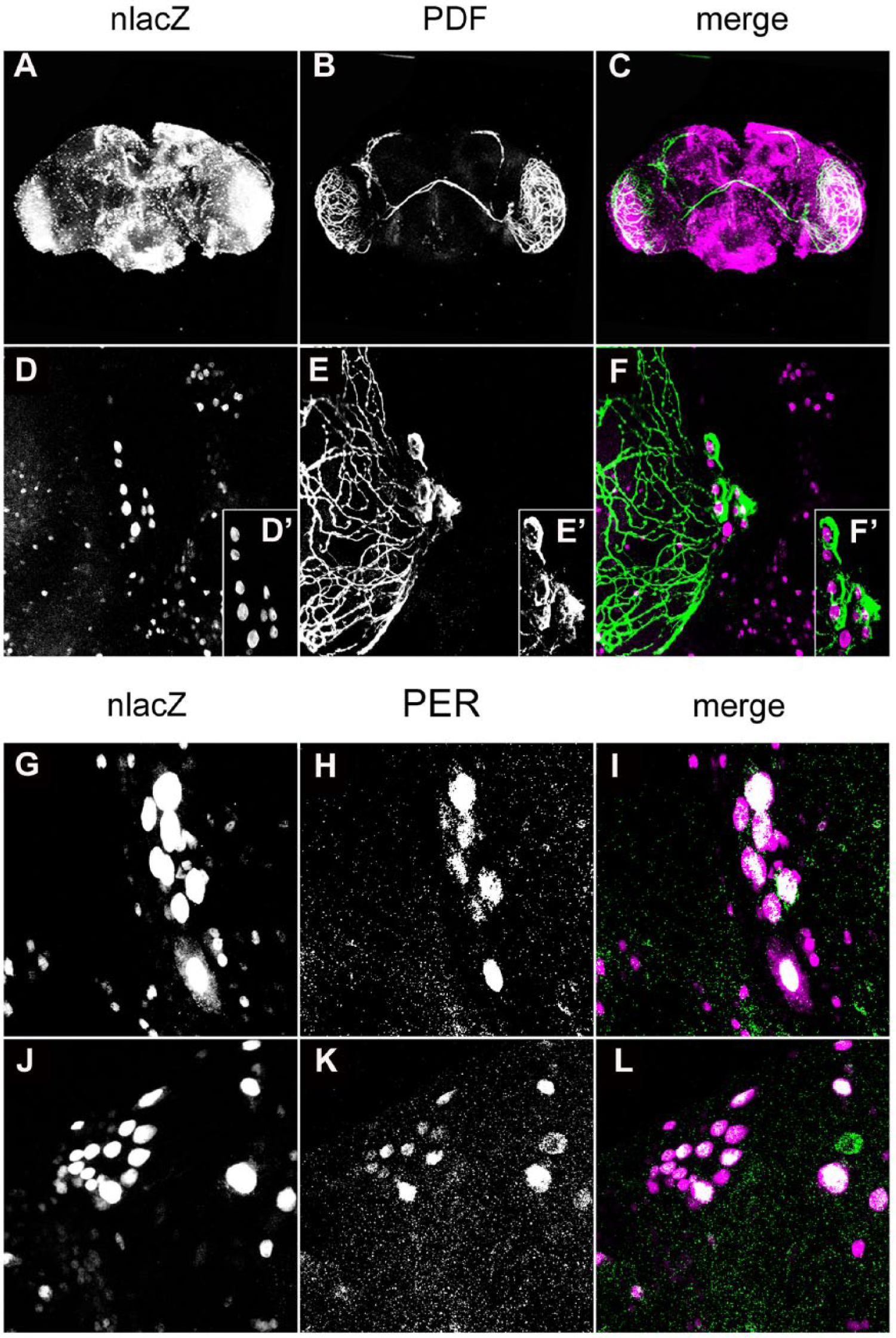
p38K is expressed in clock neurons in adult
p38K Mutants Have Aberrant Circadian Rhythms
Since p38Ka and p38Kb are expressed in clock neurons (Fig. 1 and Kadener et al., 2009; Kula-Eversole et al., 2010; Nagoshi et al., 2010; Wijnen et al., 2006), we tested whether p38K signaling plays a role in regulating circadian rhythms using a locomotor-based assay in which the animals display a rhythmic locomotor behavior when entrained to a 12-h:12-h LD cycle. We first measured locomotor behavior rhythms in LD in single null mutants of either p38Ka (Δp38Ka) or p38Kb (p38KbΔ45) as well as a viable DKO combination (p38K DKO) of a null allele of p38Ka (Δp38Ka) and a hypomorphic allele of p38Kb (p38KbΔ25) and found no effect on locomotor behavior (data not shown). Therefore, we tested whether loss of p38K under free-running conditions (constant darkness [DD]) affects circadian locomotor behavior. We find that loss of either p38Ka or p38Kb does not affect normal circadian rhythms in young animals. These findings are consistent with the recent results from Dusik et al. (2014). Interestingly, we find that a significantly larger proportion of p38K DKO animals are arrhythmic (Fig. 2, A and C; Table 1; and Suppl. Fig. S1), while animals that are not arrhythmic display normal circadian rhythms. This dichotomy between normal and abnormal rhythms is not entirely unexpected as the p38K DKO phenotypes are partially penetrant (Vrailas-Mortimer et al., 2011). In addition, we find that 32% of p38K DKO animals also display ultradian rhythms compared with 4% of wild-type controls (Fig. 3).
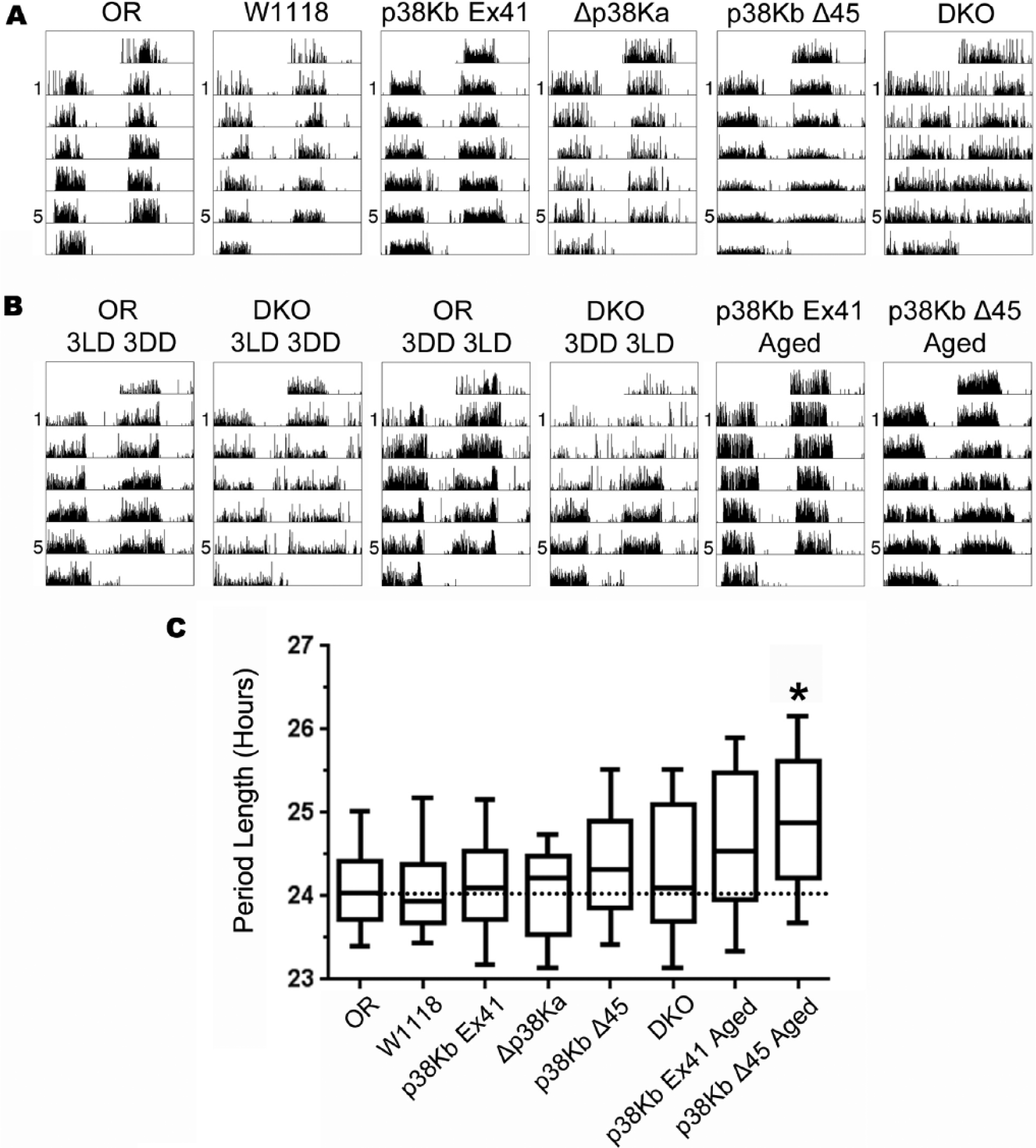
p38K mutants have longer free-running rhythms. (A) p38K single mutants exhibit normal circadian rhythms. However, loss of both p38K genes (DKO) results in mostly arrhythmic animals. (B) p38K DKO circadian defect is not a result of age-dependent locomotor dysfunction. In addition, aged p38Kb single mutants (p38Kb Δ45) display an increase in period compared with age-matched controls (p38Kb Ex41). (C) Box and whisker plots of young and aged animals. No significant difference in tau for young mutants and controls was determined by ANOVA. Aged p38Kb mutants have a significant increase in period length compared with age-matched controls (p38Kb Ex41) using Student’s
Loss of p38K results in increased arrhythmicity and increased tau with age.
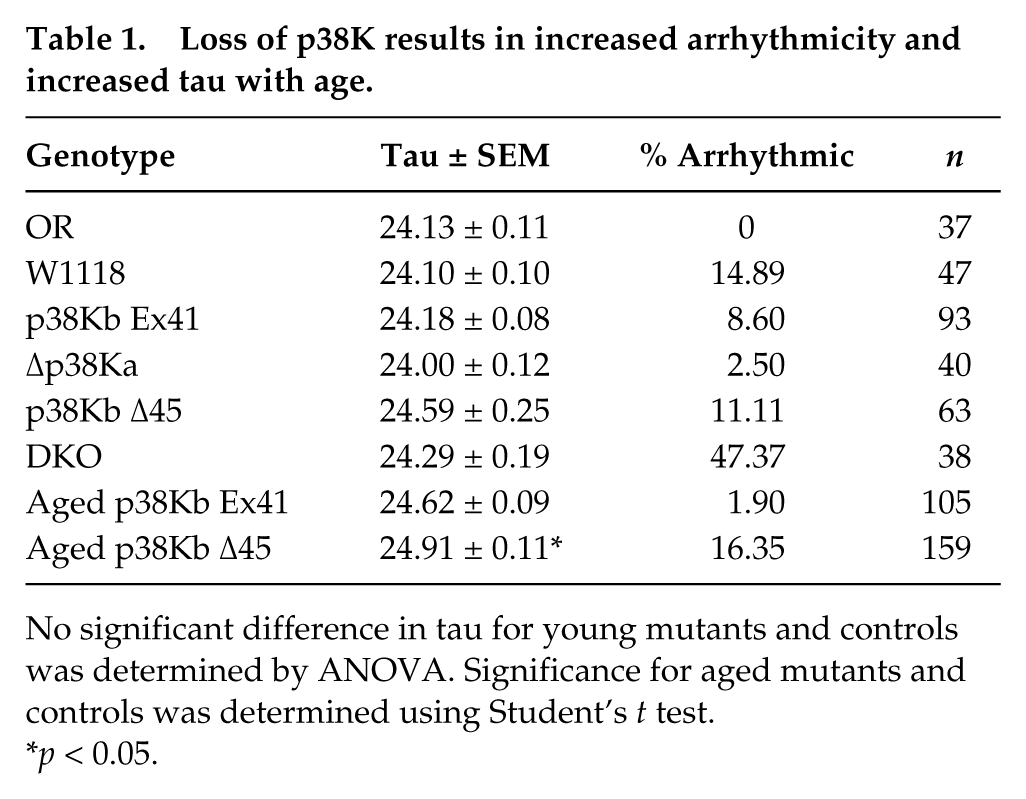
No significant difference in tau for young mutants and controls was determined by ANOVA. Significance for aged mutants and controls was determined using Student’s
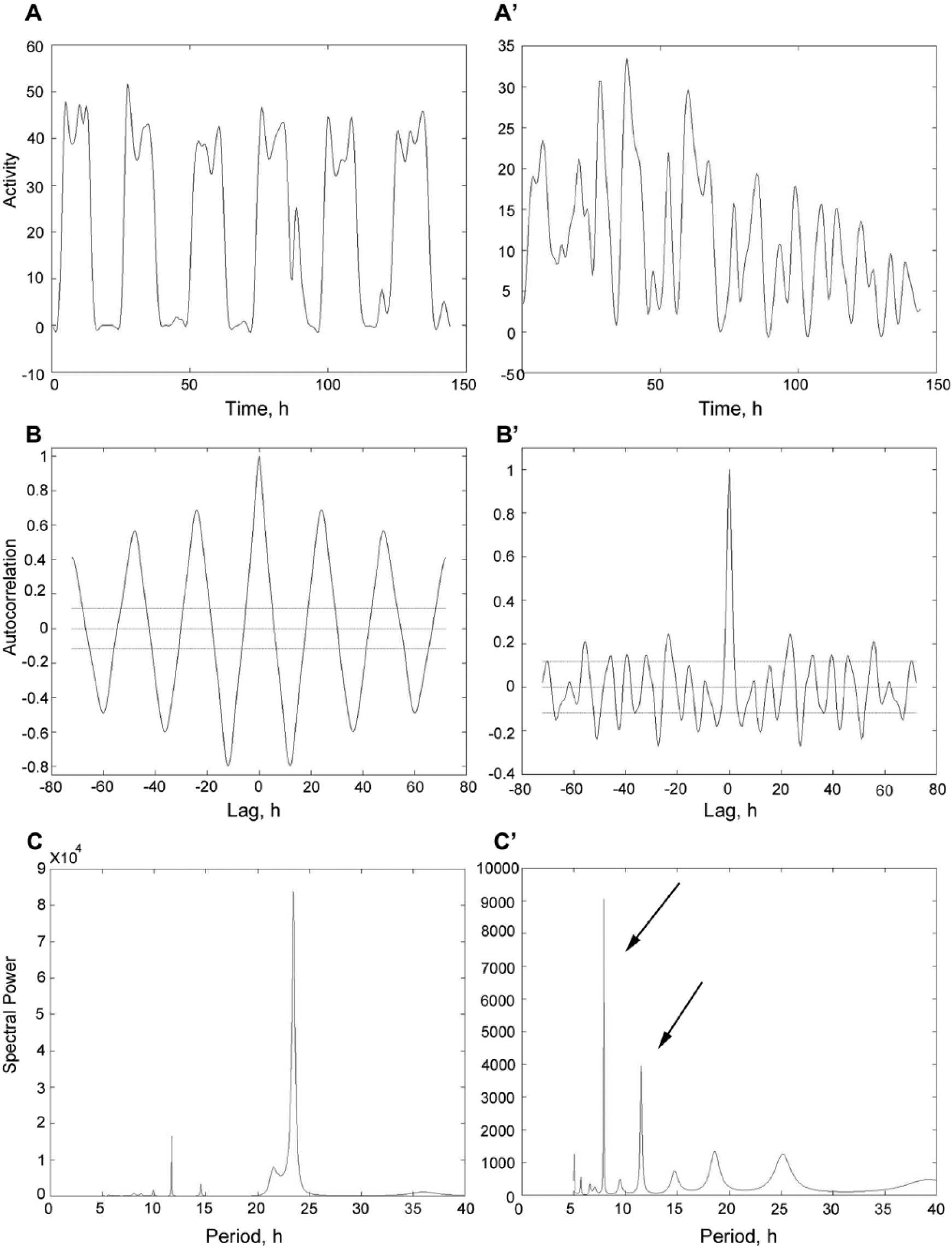
p38K mutants display ultradian rhythms. p38K DKO and precise excision animals were reared in 12-h:12-h light-dark cycle (LD) and then placed in constant darkness for 6 days. (A and A′) actograms, (B and B′) autocorrelograms, and (C and C′) MESA spectrum. (A-C) p38Kb Ex41 animals show predominantly normal circadian rhythms as depicted here. (A′-C′) In contrast, spectra of p38K DKO animals commonly reveal the appearance of ultradian rhythms (black arrows) as in this example. Compare C′ with C.
Since we have previously found that the p38K DKO animals have an age-dependent locomotor defect (Vrailas-Mortimer et al., 2011), we wanted to confirm that the increase in arrhythmicity in the p38K DKO animals is not the result of a locomotor deficit. As a control, animals were reared in a 12-h:12-h LD cycle and then transferred to constant darkness for 3 days (3LD, 3DD). Consistent with our earlier observation, these animals continue to display arrhythmic behavior only in DD. Since the 3DD comes at an age when p38K DKO animals display locomotor behavior defects, we placed LD-entrained p38K DKO animals in constant darkness for 3 days followed by a return to a 12-h:12-h LD cycle (3DD 3LD). Again, the p38K DKO animals were arrhythmic in DD, but locomotor behavior was restored when they were returned to LD (Fig. 2B), suggesting that p38K signaling is required for normal free-running circadian rhythms and any behavioral defects are not a consequence of impaired locomotor function. As we have found that p38Kb single mutants have several age-dependent phenotypes (Vrailas-Mortimer et al., 2011), we tested whether loss of p38Kb leads to an age-dependent loss of circadian rhythms. We found that aged p38Kb mutants have a modest but significant increase in tau of 24.9 h compared with a precise excision control (p38Kb Ex41) of 24.6 h (Fig. 2, B and C; Table 1; and Suppl. Fig. S1).
Optimal p38Kb Signaling Is Required in Clock Neurons for Maintaining Normal Circadian Rhythms
Because global knockout of p38Ka and p38Kb can lead to pleiotropic effects, we decided to test the role of p38Kb specifically in clock neurons. Therefore, we overexpressed 2 different dominant negative forms of p38Kb, both of which we have previously found to inhibit p38Kb signaling in vivo (Vrailas-Mortimer et al., 2011). The first is a phospho-null form of p38Kb (p38Kb ala2) in which the critical Thr and Tyr residues of the TGY phosphorylation motif have been mutated to Ala. Although p38Kb ala2 can interact with upstream kinases, it cannot be phosphorylated, thus preventing activation of p38Kb. Reminiscent of p38KbΔ45 mutants, overexpression of p38Kb ala2 in clock neurons using either a Pdf 0.5-GAL4 or a Cry-GAL4 driver results in a significant increase in arrhythmicity and tau in free-running conditions of 25.9 h and 25.2 h, respectively (Figs. 4 and 5, Tables 2 and 3, and Suppl. Figs. S2 and S3). Furthermore, overexpression of a second dominant negative construct, a kinase dead version of p38Kb (p38Kb KD) that cannot phosphorylate downstream targets, also resulted in an increased arrhythmicity and period under free-running conditions when used in a single copy (p38Kb KD3 [weak line] or p38Kb KD8 [strong line, data not shown]) or when used in 2 copies (p38Kb KD3 and p38Kb KD8, 25.3 h for expression with Pdf 0.5-GAL4 and 25.5 h for expression with Cry-GAL4) (Figs. 4 and 5, Tables 2 and 3, and Suppl. Figs. S2 and S3). These results further confirm that p38K signaling influences circadian rhythms and mirror recent findings that RNAi knockdown of p38Kb or expression of a different p38Kb dominant negative transgene (in which only 1 of the phosphorylation sites is mutated) in clock neurons also results in period lengthening under free-running conditions (Dusik et al., 2014).
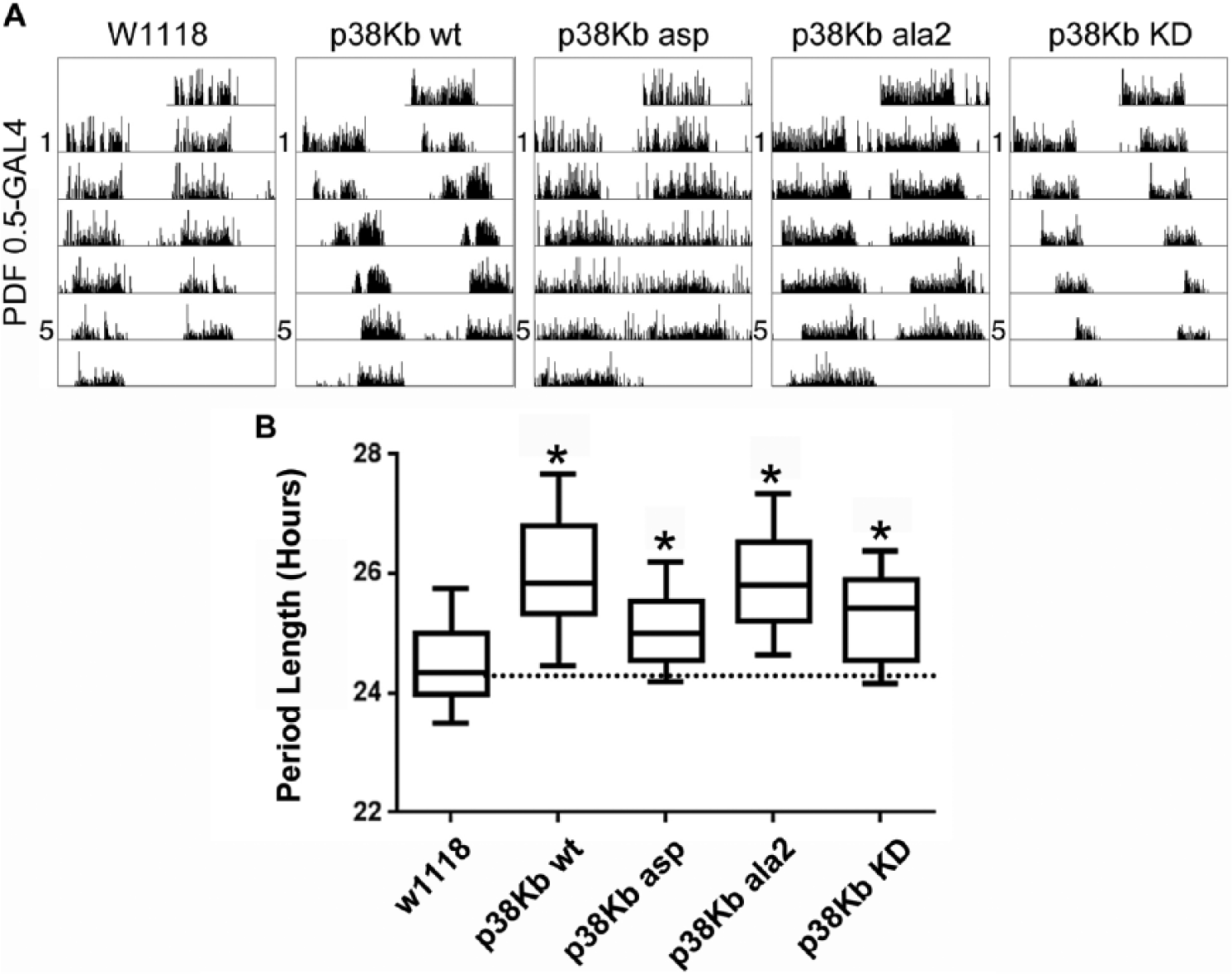
Either p38Kb activation or inhibition in clock neurons results in arrhythmic flies and lengthens the free-running rhythm. (A) Both activation (p38Kb wt and p38Kb asp) and inhibition (p38Kb ala2 and p38Kb KD) of p38Kb signaling in
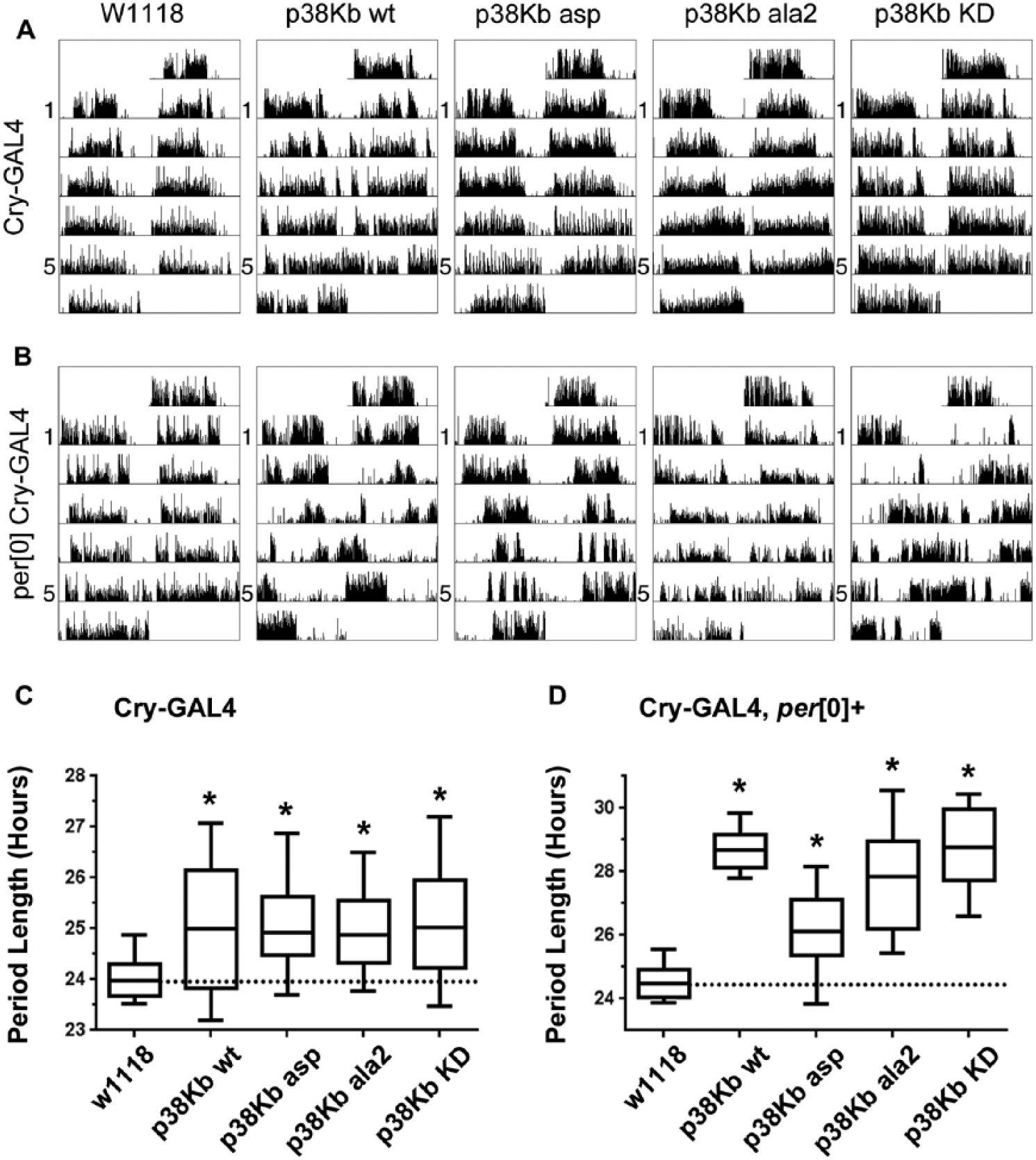
Reduced PER strongly enhances p38K-dependent circadian rhythm phenotypes. (A) Both activation (p38Kb wt and p38Kb asp) and inhibition (p38Kb ala2 and p38Kb KD) of p38K signaling in
Tau and arrhythmicity of p38K expression in Pdf-expressing clock neurons.
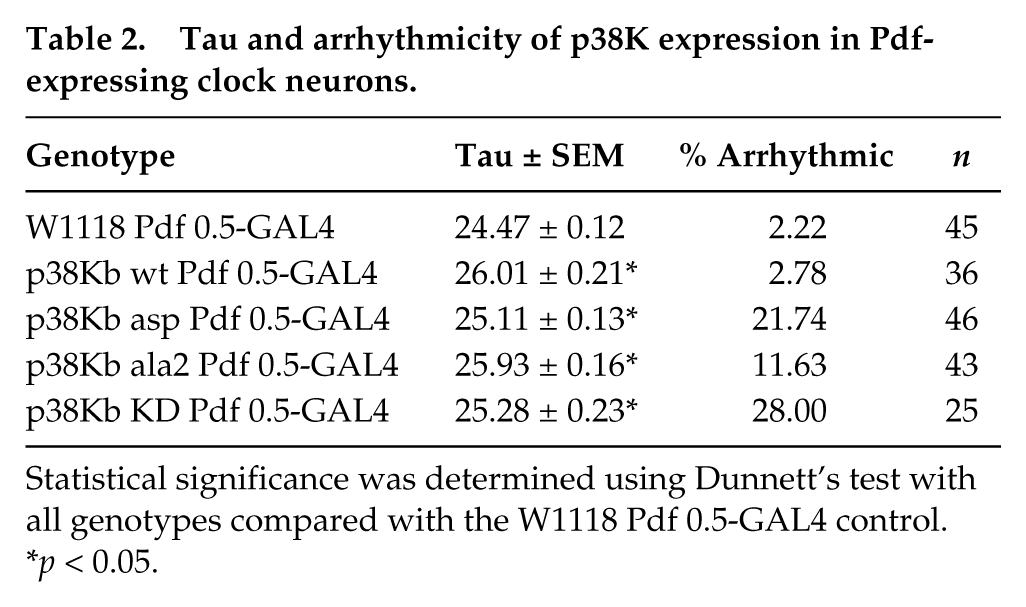
Statistical significance was determined using Dunnett’s test with all genotypes compared with the W1118 Pdf 0.5-GAL4 control.
Aberrant p38K signaling in
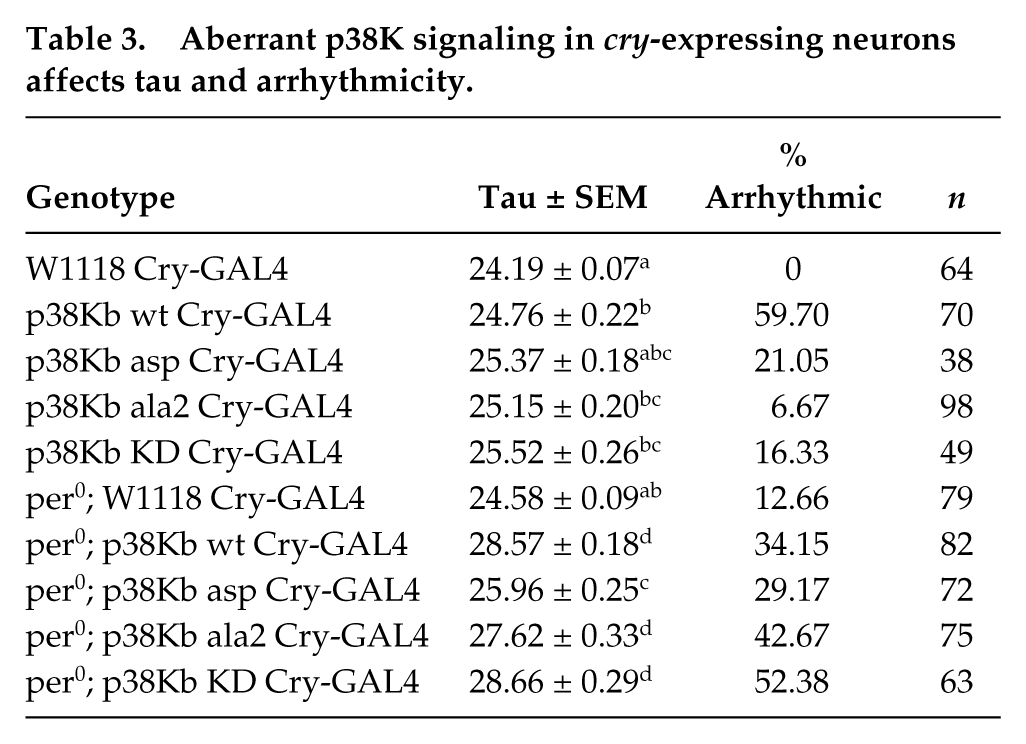
Significance between groups was determined by ANOVA followed by Tukey’s HSD. Significance groups are indicated by superscript letters.
Paradoxically, overexpression of wild-type (p38Kb wt) in clock neurons in free-running conditions also results in an increased tau as well as an increase in the number of arrhythmic animals (Figs. 4 and 5, Tables 2 and 3, and Suppl. Figs. S2 and S3), which also was observed by Dusik et al. (2014). As the phosphorylation of p38Kb is important for its downstream functions, we tested whether increasing the amount of activated p38Kb would result in a further increase in period length or arrhythmicity. We found that overexpression of a p38Kb phospho-mimic (p38Kb asp, in which the Thr and Tyr residues of the TGY motif are mutated to Asp) in PDF positive neurons under free-running conditions leads to a further increase in the number of arrhythmic animals compared with p38Kb wt overexpression (Fig. 4, Table 2, and Suppl. Fig. S2). However, p38Kb asp expression using the Cry-GAL4, which is more widely expressed, results in a further increase in period length compared with p38Kb wt overexpression (Fig. 5, Table 3, and Suppl. Fig. S2).
Our observations are contrary to the expectation that p38Kb signaling would act in a bidirectional manner, with overexpression and inhibition of p38Kb resulting in opposite phenotypes. In fact, our results point to a situation where normal clock function requires optimal p38Kb signaling. Accordingly, different magnitudes or spatial patterns of p38Kb activation may result in different outcomes on circadian rhythms, an idea consistent with the recent observation that the levels of p38K phosphorylation cycle in specific clock neurons (Dusik et al., 2014).
p38Kb-Mediated Regulation of Circadian Rhythm Is Sensitive to Levels of Per
Since aberrant p38Kb signaling leads to an increase in tau only in free-running conditions, much like loss of the core clock component
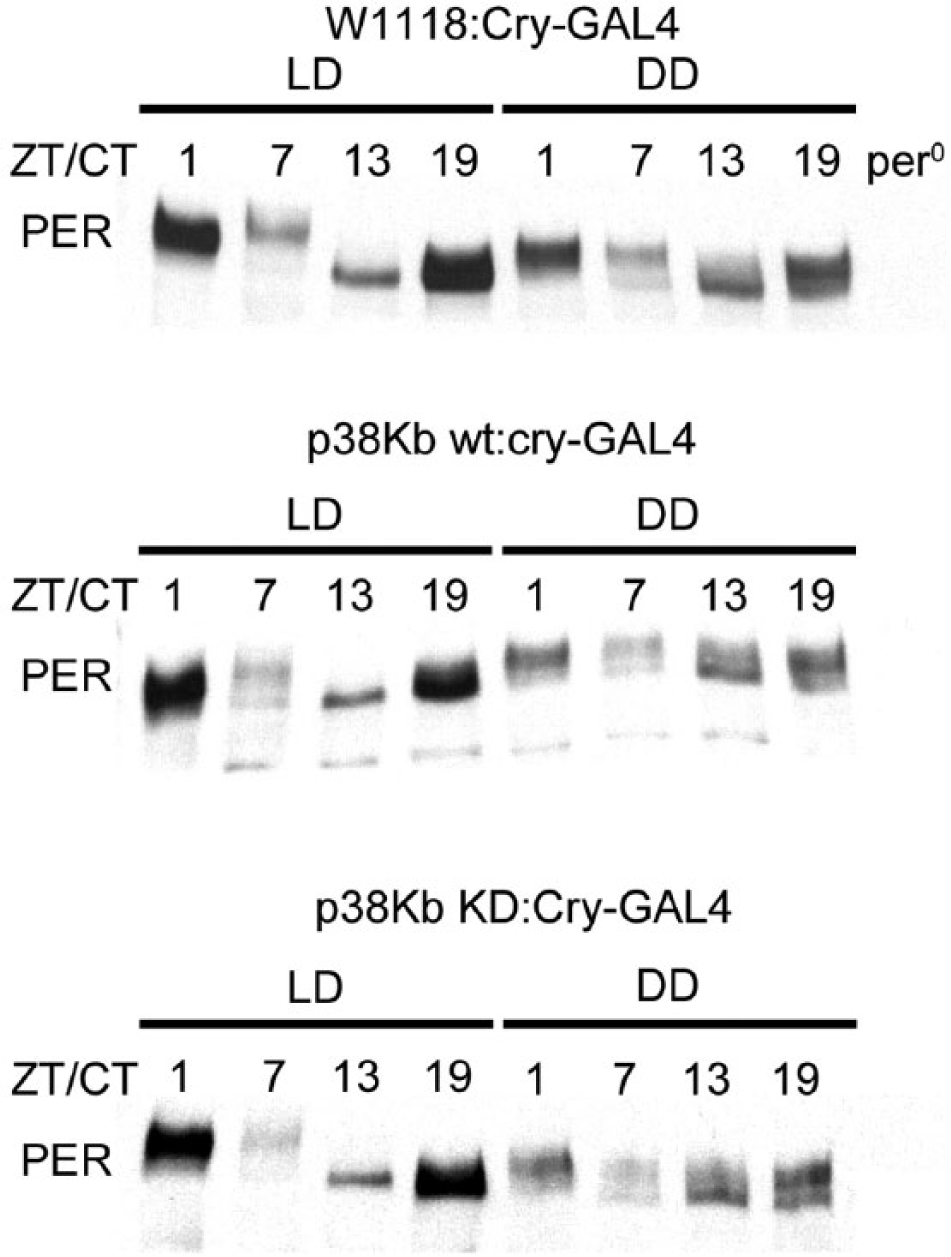
PER cycling under free-running conditions is not regulated by p38Kb. Animals were entrained in a 12-h:12-h light-dark cycle (LD) and collected on the second day of constant darkness (DD). Perturbations in p38Kb do not affect the cycling of PER hyperphosphorylation state throughout the 24-h clock. However, inhibition of p38Kb results in a decrease in overall PER levels.
To determine whether PER might be a target of p38Kb in vivo, we used the Cry-GAL4 to drive clock neuron expression of p38Kb KD, which is FLAG tagged. The protein product of the p38Kb KD mutant gene is thought to be able to bind to downstream targets but has reduced efficiency in subsequent release (Robinson et al., 1996), thus providing a unique opportunity to observe potentially transient interactions. We immunoprecipitated p38Kb KD from whole heads collected at ZT1, when PER levels are high, and assayed for binding to PER. Although p38Kb was recently shown to phosphorylate PER in vitro (Dusik et al., 2014), we were unable to detect PER binding to p38Kb KD in vivo. It may be that the p38Kb-PER interaction is too transient for us to detect even using the p38Kb KD. Another possibility is that p38Kb-mediated phosphorylation of PER is tightly regulated temporally and spatially. For example, p38Kb may only phosphorylate PER at night when PER is cytoplasmic rather than nuclear as it is at ZT1 (Curtin et al., 1995).
p38Kb Genetically Interacts with Mef2 to Regulate Circadian Rhythms
As p38 MAPKs have a number of downstream targets, p38Kb may be regulating other factors that give rise to circadian locomotor behaviors. We recently found that p38Kb acts through the transcription factor Mef2, a bona fide p38K target, to regulate aging and oxidative stress (Vrailas-Mortimer et al., 2011). Mef2 is transcriptionally regulated by the CLK/CYC complex (Sivachenko et al., 2013) and expressed in both the l-LNv and s-LNv neurons (Blanchard et al., 2010). Expression of a dominant negative Mef2 (Mef2 EnR) in which the transcriptional activation domain has been replaced with the Engrailed transcriptional repression domain results in arrhythmia, as reported previously (Fig. 7 and Table 4, Blanchard et al., 2010). We find that overexpression of p38Kb wt in the Mef2 EnR background rescues the arrhythmia phenotype from 84% to 53% arrhythmic (Fig. 7 and Table 4). Furthermore, p38Kb wt overexpression alone exhibits an increased tau, which is further magnified by inhibition of Mef2 (24.8 h and 26.6 h, respectively (Fig. 7 and Table 4), similar to what we observe when removing a single copy of
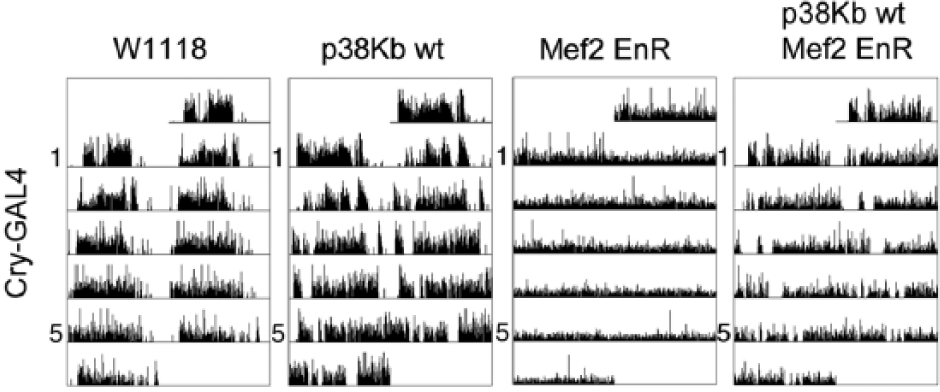
p38Kb genetically interacts with Mef2 to regulate circadian locomotor behaviors. Expression of dominant negative Mef2 (Mef2 EnR) driven by Cry-GAL4 results in arrhythmia, which is partially rescued by p38Kb wt overexpression.
p38Kb genetically interacts with Mef2.
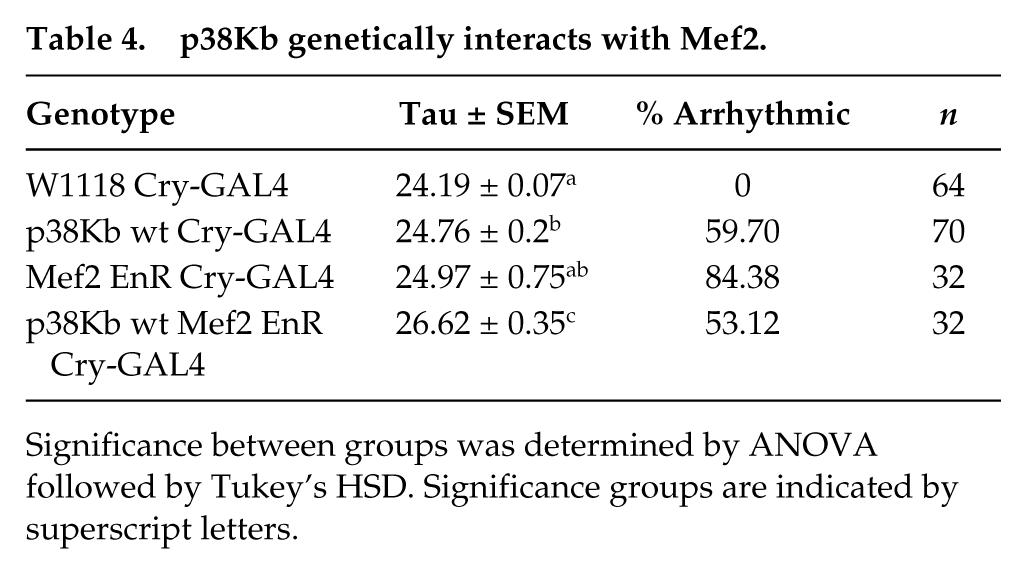
Significance between groups was determined by ANOVA followed by Tukey’s HSD. Significance groups are indicated by superscript letters.
p38Kb Signaling Affects MNK/Lk6 Circadian Cycling
In mammalian systems, another downstream target of p38K is the MAPK interacting kinase MNK/Lk6, which phosphorylates the AKT downstream target eIF4E. Upregulation of the AKT pathway leads to an increased period length in flies (Zheng and Sehgal, 2010), and
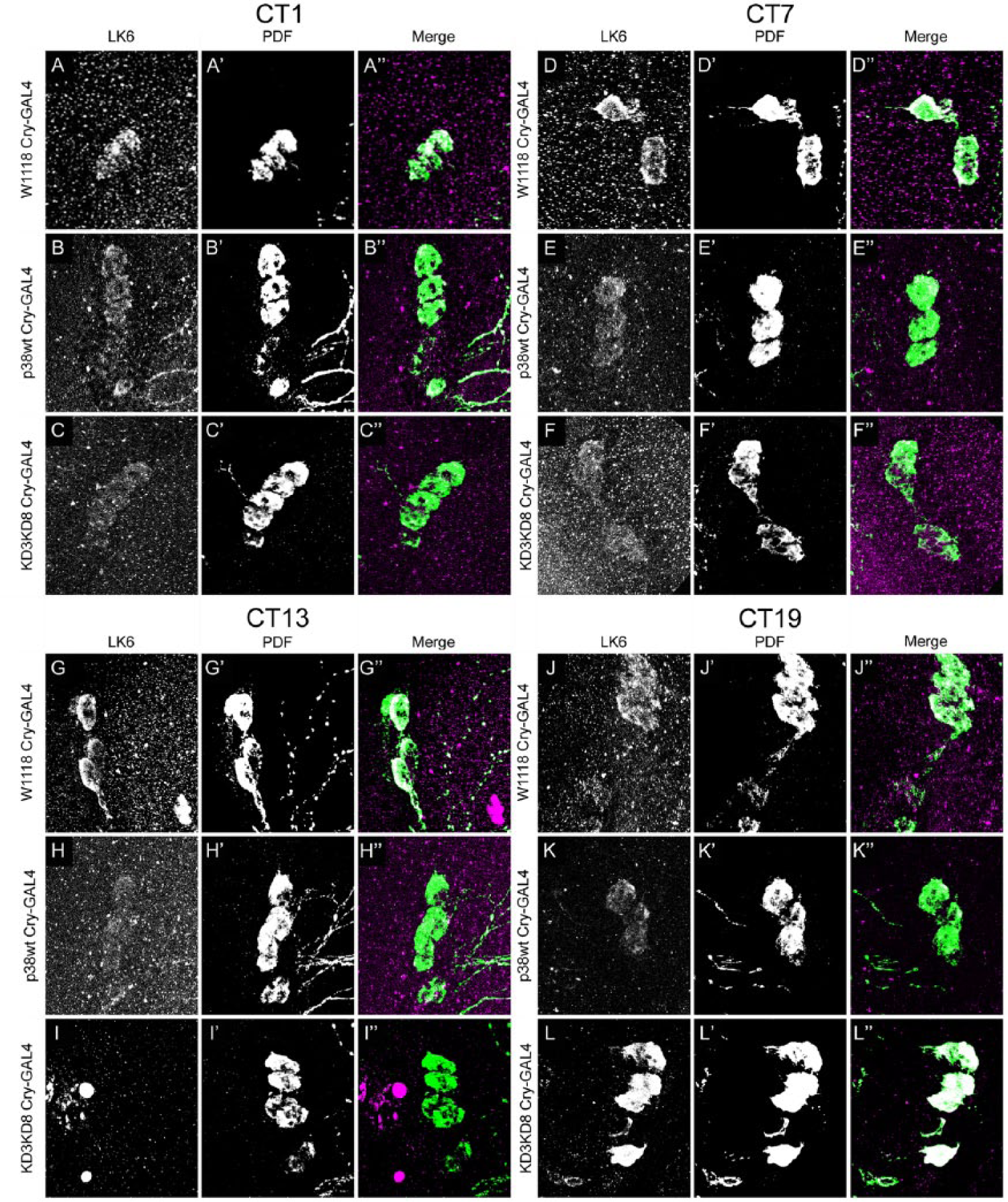
p38Kb regulates Lk6 expression in clock neurons throughout the clock. Animals were entrained in a 12-h:12-h light-dark (LD) cycle and then placed in constant darkness (DD) and collected during the second day of DD. Lk6 is expressed in PDF positive l-LNv clock neurons (A, D, G, and J). Lk6 expression is decreased at CT13 when p38Kb is overexpressed (compare H with G). Inhibition of p38Kb results in decreased Lk6 expression at CT13 (compare I with G) but increased expression at CT19 (compare L with J). Merged images are LK6 (magenta) and PDF (green), and white shows colocalization.
Discussion
The ability of an organism to properly respond to its environment is, in part, influenced by circadian rhythms (Bartok et al., 2013; Kondratov et al., 2006; Krishnan et al., 2008; Zheng et al., 2007), suggesting that stress response pathways may play an important role in maintaining these rhythms. Interestingly, manipulating aging genes in clock neurons, which are tightly linked to stress response, negatively affects circadian rhythms (Zheng et al., 2007). Therefore, other genes linked to stress and aging, such as the stress-activated protein kinase p38K, may also play roles in regulating circadian rhythms. Our data suggest that p38Kb contributes to maintaining a proper circadian clock with either inhibition or promiscuous activation of p38Kb leading to aberrant rhythms. Flies have two p38K genes, p38Ka and p38Kb, that have differential levels of expression in the fly brain, with p38Kb as the predominant species (Vrailas-Mortimer et al., 2011). Furthermore, we find that p38Kb is expressed in clock neurons and that loss of either gene alone at a young age does not influence circadian locomotor behavior. However, we have previously found that p38Kb mutants have many age-dependent phenotypes (Vrailas-Mortimer et al., 2011), and consistent with these observations, we find that aged p38Kb mutant animals have a minor but significant period lengthening in free-running conditions. In younger animals, p38Kb-dependent phenotypes require much stronger knockdown, perhaps due to the ubiquitous and high level of p38Kb expression in the brain. Indeed, we notice a large percentage of arrhythmic flies when both p38Ka and p38Kb levels are reduced (p38K DKO). Additionally, significant period lengthening is observed when p38Kb is inhibited in clock neurons using the Cry-GAL4 or Pdf 0.5-GAL4 driver lines in conjunction with either a phospho-null (p38Kb ala2) or a kinase-dead version of p38Kb (p38Kb KD). These results also confirm that p38Kb perturbation in clock neurons is sufficient to precipitate circadian phenotypes, which was recently observed by Dusik et al. (2014). Interestingly, Dusik et al. (2014) also observed that knockdown of p38Ka results in period lengthening. MAPKs are known to homodimerize, which is important for cytoplasmic functions as well as nuclear translocation (Khokhlatchev et al., 1998). The p38Ka and p38Kb amino acid sequences are 78% identical and have 94% sequence similarity and have been found to co-Immunoprecipitate (co-IP) with each other (Belozerov et al., 2014; Guruharsha et al., 2011). Therefore, p38Ka and p38Kb likely act as both homodimers and heterodimers, which may have specific functions. The phospho-null or kinase dead forms of p38Kb may inhibit p38Kb homodimer and heterodimer functions but may also block the formation of p38Ka homodimers, which normally would be able to compensate for the lack of p38Kb homodimers and heterodimers. RNAi knockdown of either p38Kb or p38Ka also leads to period lengthening (Dusik et al., 2014), which is not too surprising as the mRNA coding sequences of p38Kb and p38Ka are 73% identical, and these RNAi lines target regions in common for p38Ka and p38Kb. Thus, p38Ka and p38Kb may have compensatory roles in regulating circadian rhythm, and inhibition of both proteins is necessary to elicit a phenotype.
Activation of p38Kb in clock neurons, achieved through the expression of either wild-type p38Kb or a phospho-mimic p38Kb (p38Kb asp), has the same effect as p38Kb inhibition in extending the length of the circadian period. This seemingly contradicts the simple expectation that p38Kb overexpression might shorten period length. Therefore, we cannot assume a simplistic model of p38Kb in the regulation of circadian rhythms. One idea for why bidirectional changes in p38Kb produce the same phenotype (period lengthening) is that in both cases, oscillations in either p38Kb expression or activity are dampened. Another possibility is that p38Kb is required for the regulation of a downstream target that is under the control of a cycling mechanism.
As p38Kb is a regulator of the stress response and loss of p38Kb results in increased oxidative stress and reduced lifespan (Vrailas-Mortimer et al., 2011), it is possible that p38Kb perturbations adversely affect the health of clock neurons. However, the number of PDF positive neurons is not reduced following p38Kb inhibition (data not shown). Since we observe p38Kb-dependent phenotypes only in complete darkness, we hypothesized that p38Kb may affect the core clock machinery, in particular PER, since
Although p38Kb may be regulating PER, p38 MAPKs have a variety of downstream targets that may also contribute to maintaining circadian rhythms. One such downstream target is Mef2, which, like p38Kb, results in period length extension and increased arrhythmia when overexpressed or inhibited (Blanchard et al., 2010). We find that p38Kb genetically interacts with Mef2 in regulating circadian locomotor behavior, with overexpression of p38Kb wt rescuing the arrhythmia observed by Mef2 inhibition. Although Mef2 has been shown to cycle in the s-LNv neurons (Blanchard et al., 2010), the Mef2 phosphorylation state in these cells is not known. In addition, Mef2 is also expressed in the l-LNv neurons, although Mef2 cycling in these neurons has yet to be examined. One possibility is that manipulating p38Kb in the s-LNvs interferes with p38Ka activity in these neurons, resulting in dysregulation of Mef2, although Mef2 has yet to be shown as a downstream target of p38Ka. Alternatively, p38Kb may regulate Mef2 in the l-LNv neurons, which are hypothesized to integrate multiple circadian inputs and transmit this information to multiple outputs including the s-LNv neurons (reviewed in Helfrich-Forster, 2005). As the relationship between the l-LNv and s-LNv neurons is not fully understood, further insights may be gained by observing how manipulating signaling cascades in the l-LNv neurons influences the s-LNv neurons.
Another downstream target of p38K is the MAPK interacting kinase MNK/Lk6, the transcription of which oscillates in a circadian fashion and is regulated by CLK/CYC (Claridge-Chang et al., 2001; Dubruille et al., 2009). We find that Lk6 protein is indeed expressed in clock neurons and upon p38Kb inhibition shows aberrant expression compared with clock neurons in control brains. This indicates that p38Kb perturbations can affect MNK/Lk6 function in clock neurons, although it remains to be seen whether this is a direct effect or occurs indirectly through, for instance, the CLK/CYC complex. Overall, these results suggest that p38Kb may be regulating multiple signals that contribute to circadian rhythm locomotor behavior. If p38Kb is functioning as a nodal point to integrate several upstream signaling inputs as well as relaying signaling events to a large number of downstream effectors in clock neurons, then the level of synchrony that is required to maintain a normal circadian rhythm must be exquisite. Studying these multiple p38Kb-dependent signaling pathways and their oscillations simultaneously is a prerequisite for understanding the complexity and plasticity in this system.
Footnotes
Acknowledgements
The authors thank Justin Blau and Amita Sehgal for useful discussions and the gift of Pdf 0.5-GAL4 and UAS-Mef2 EnR and anti-PER antibodies, respectively. The authors also thank Verena Dusik and Charlotte Helfrich-Forster for sharing their unpublished results and useful comments. This study was supported by grants from the Brain and Behavior Research Foundation (formerly the National Alliance for Research on Schizophrenia and Depression) and by National Institutes of Health Grants 1R03DA027979 and 5R21MH091520 to S.S., a Center for Behavioral Neuroscience NSF postdoctoral fellowship, a PD-CERC T32 ES012870 fellowship, and start-up funds from the University of Denver to A.D.V.M. All research materials are available upon request.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
