Abstract
Rhythmic events in the female reproductive system depend on the coordinated and synchronized activity of multiple neuroendocrine and endocrine tissues. This coordination is facilitated by the timing of gene expression and cellular physiology at each level of the hypothalamo-pituitary-ovarian (HPO) axis, including the basal hypothalamus and forebrain, the pituitary gland, and the ovary. Central to this pathway is the primary circadian pacemaker in the suprachiasmatic nucleus (SCN) that, through its myriad outputs, provides a temporal framework for gonadotropin release and ovulation. The heart of the timing system, a transcription-based oscillator, imparts SCN pacemaker cells and a company of peripheral tissues with the capacity for daily oscillations of gene expression and cellular physiology. Although the SCN sits comfortably at the helm, peripheral oscillators (such as the ovary) have undefined but potentially critical roles. Each cell type of the ovary, including theca cells, granulosa cells, and oocytes, harbor a molecular clock implicated in the processes of follicular growth, steroid hormone synthesis, and ovulation. The ovarian clock is influenced by the reproductive cycle and diseases that perturb the cycle and/or follicular growth can disrupt the timing of clock gene expression in the ovary. Chronodisruption is known to negatively affect reproductive function and fertility in both rodent models and women exposed to shiftwork schedules. Thus, influencing clock function in the HPO axis with chronobiotics may represent a novel avenue for the treatment of common fertility disorders, particularly those resulting from chronic circadian disruption.
Circadian Timing System in Mammals
The circadian timing system regulates daily rhythms of gene expression, hormone secretion, metabolism, and behavior (Albrecht, 2012; Marcheva et al., 2013). Circadian rhythms are generated at the molecular level by the rhythmic transcription of circadian clock genes. Most differentiated mammalian cells host an autoregulatory transcriptional-translational feedback loop of clock gene transcription factors (Albrecht, 2012; Mohawk et al., 2012). The transcriptional enhancers BMAL1 and CLOCK are central to the oscillator, with CLOCK displaying histone acetyl transferase activity at target promoters (Etchegaray et al., 2003; Hirayama et al., 2007; Nader et al., 2009). Together, the BMAL1:CLOCK complex binds to E-box (CACGTG) sequences in target promoters and drives transcription of the period (per 1-3) and cryptochrome genes (cry1 and cry2). After some critical posttranslational modifications, including phosphorylation by Casein kinases, the PER and CRY proteins translocate to the nucleus, wherein they repress the activity of the BMAL1:CLOCK enhancer complex and suppress their own transcription. The inherent delay in nuclear translocation of the repressor complex, dictated by parallel phosphorylation and protein degradation pathways, underlies the near 24-h timing of the oscillator. In addition to the core loop, a secondary loop of interlocking transcriptional regulators including the repressor REV-ERBα and the enhancer retinoic acid-like orphan receptor α(RORα) provides stability of function by regulating BMAL1 expression. The core clock genes also control output genes or clock-controlled genes (CCGs). CCGs are the effector molecules of the clock, mediating its temporal control over cell-type–specific gene expression and physiology (Hamada et al., 2004; Kennaway et al., 2003).
In mammals, the timing system has been described as a multioscillator hierarchy of coordinated and synchronized cell and tissue clocks (Menaker et al., 2013)). At the top of this hierarchy is the central pacemaker located in the suprachiasmatic nucleus (SCN) of the basal hypothalamus. In addition to maintaining pacemaker function in the SCN, the molecular clock also regulates the timing of gene expression and physiology in peripheral tissues and extra-SCN brain regions (Abe et al., 2002; Guilding et al., 2009; Menaker et al., 2013; Yamazaki et al., 2000; Yoo et al., 2004). It is now widely believed, with minimal exceptions, that the majority of mammalian cells are autonomous or semi-autonomous oscillators. This arrangement offers a unique set of challenges for the organism. Clearly, synchronization among central and peripheral oscillators (e.g., SCN, liver, pituitary gland, etc.) is a fundamental property of physiological homeostasis and a defining feature of the timing system (Buijs et al., 2001; Guo et al., 2005; Menaker et al., 2013; Mohawk et al., 2012). The responsibility for this coordination falls largely on the timing of neural efflux from the SCN carried directly through peripheral nerves and indirectly through control of hypothalamic neuroendocrine-releasing factors (e.g., GnRH; Albrecht, 2012; Menaker et al., 2013). Internal circadian organization is broadly defined as the coordinated and synchronized timing of central and peripheral clocks as well as the timing of each oscillator relative to the external (and internal) environment. A decline in circadian organization (circadian disruption or chronodisruption, as seen during chronic jet lag, rotating shift work, etc.) can lead to health issues including prediabetes, cancer, and cardiovascular disease (Evans and Davidson, 2013).
Circadian Timing System in Endocrine Physiology: From Gonadotroph to β-Cell
Some of the earliest evidence for circadian clock function in the mammalian endocrine system was recorded well before the SCN was identified as the locus of the neural pacemaker (Moore and Eichler, 1972; Stephan and Zucker, 1972). In the mid-20th century, John Everett and Charles Sawyer established the role of a neural oscillator or “timer” in the temporal control of the ovulatory surge of luteinizing hormone (LH) secretion. They found that blocking the LH surge with a timed injection of anesthetic in the early afternoon on the day of ovulation (proestrus), a treatment that globally silenced neuronal activity, prevented the rise in LH (and thus ovulation) and delayed it 24 h (Everett and Sawyer, 1950; Sawyer et al., 1949). That is, rather than occurring several hours later when the effects of sedation waned, ovulation was delayed until the afternoon of the following day. These key data supported the intimate relationship between the circadian timing system and the hypothalamic-pituitary-gonadal (HPG) axis. Subsequent mechanistic studies solidified the SCN as the critical mediator of timing in neuroendocrine physiology (Guo et al., 2005; Hastings et al., 2007; Kalsbeek and Fliers, 2013). The SCN regulates rhythms of hormone secretion via a complex interplay of neuroendocrine, endocrine, and autonomic timing cues (Buijs et al., 2003a; Buijs et al., 2003b; Guo et al., 2005; Hastings et al., 2007; Kriegsfeld and Silver, 2006). Outside the brain, the molecular oscillator has been localized to each endocrine tissue, although functional significance in each hormone regulatory system is poorly understood. Well before it was determined that peripheral tissues were composed of cell-based oscillators, it was postulated that tissue autonomous clocks could contribute to the timing of hormone synthesis and secretion (Andrews, 1971; Andrews and Folk, 1964; Ungar and Halberg, 1962). Since the cloning of the first mammalian circadian clock gene (King et al., 1997) and the determination that clock genes are global regulators of cellular function (Albrecht, 2012), considerable evidence has accumulated linking the molecular clock to endocrine physiology (Bass and Takahashi, 2010; Huang et al., 2011; Kalsbeek et al., 2012; Prasai et al., 2011; Sellix and Menaker, 2011; Tonsfeldt and Chappell, 2012; Williams and Kriegsfeld, 2012). These discoveries have stimulated interest in the impact of chronodisruption in all its forms on endocrine physiology (Bass and Takahashi, 2010; Mahoney, 2010). The hypothalamo-pituitary-ovarian (HPO) axis represents an excellent model system for investigating the impact of chronodisruption on physiology. The coordinated activity of neural, neuroendocrine, and endocrine oscillators is critical for HPO axis function (Fig. 1). Synchronization depends on communication among the various oscillators through direct (autonomic nervous cues) and indirect (neuroendocrine releasing factors, hormones) timing cues. Further, feedback from circulating hormones may act to modulate the phase and amplitude of tissue sensitivity to neuroendocrine and endocrine cues. By more closely exploring the activity of the clock and its target genes in endocrine tissues, novel functional relationships may emerge, providing insight into the connection between clock function and physiology. A clearer understanding of these relationships may eventually allow us to exploit targeted manipulation of the clock with chronobiotics, paving the way for new and exciting means of treatment for complex and devastating fertility disorders.
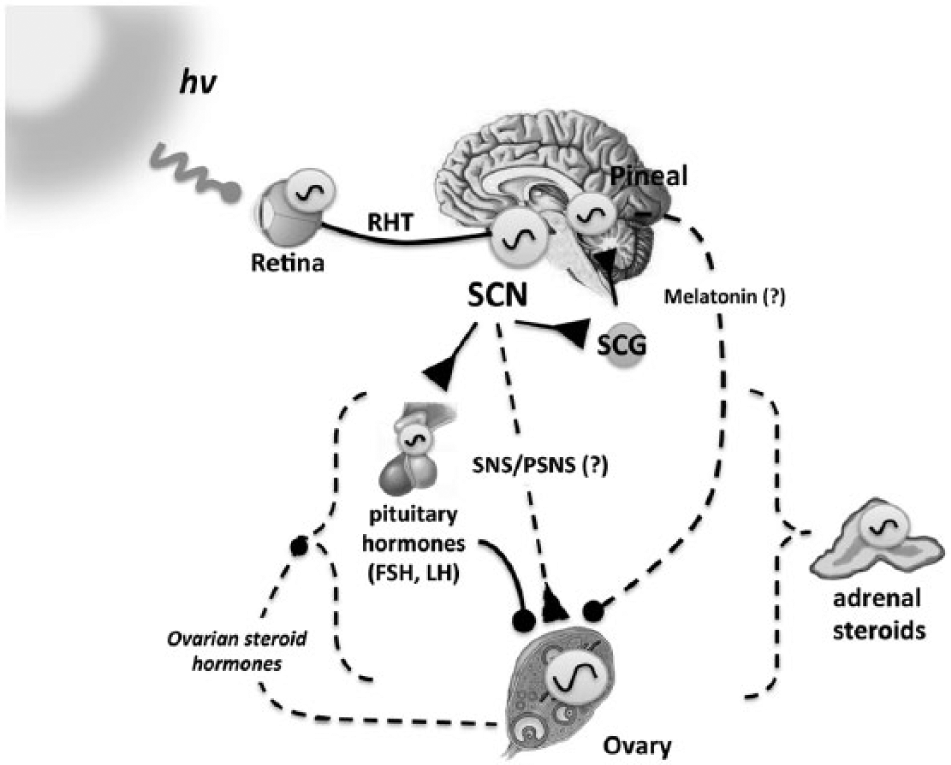
The circadian timing system in the hypothalamo-pituitary-ovarian (HPO) axis. A schematic diagram of the HPO axis indicating the integration of the circadian timing system in HPO axis function. The ovary shows phasic sensitivity to gonadotropins (LH and FSH that may depend on the timing of the ovarian clock and/or numerous timing cues of central and peripheral origin. Time of day or “photic” cues are passed to the central pacemaker in the SCN via the RHT. The SCN then conveys this information to the pineal via the SCG and the pituitary indirectly via the activity of releasing hormone neurons in the basal hypothalamus and forebrain. The SCN may also modulate the timing of the ovarian clock more directly via autonomic nervous cues (SNS, PSNS). Cells of the ovarian follicle also express melatonin receptors, and melatonin could indirectly modulate the timing of the ovarian clock. Adrenal glucocorticoids (e.g., corticosterone) may also act to synchronize the timing of the molecular clock in the pituitary and ovary. Ovarian steroid feedback affects the timing of clock gene expression in several tissues, including the uterus, pituitary, and ovary, and could further gate the response to gonadotropins. Nervous signals are indicated by lines ending in triangles, while lines ending in solid dots indicate hormonal pathways. hv indicates light, RHT, retinohypothalamic tract; SNS/PSNS, sympathetic nervous system, parasympathetic nervous system; FSH, follicle-stimulating hormone; LH, luteinizing hormone; SCN, suprachiasmatic nucleus; SCG, superior cervical ganglion.
Diversity and Complexity of Ovarian Clock Function
Ovarian clocks show significant phylogenetic diversity, having been detected in mammals, non-mammalian vertebrates, and some invertebrates (Sellix and Menaker, 2010). The molecular clock mechanism has been described in the ovary of the silkworm, moth, frog, and fruit fly, although the functional significance for fertility remains unclear (Kubo et al., 2010; Sellix and Menaker, 2010; Takeuchi et al., 2014). Limited evidence suggests that the clock may regulate fertility in drosophila, though core clock genes are not rhythmically expressed in the fly ovary (Beaver et al., 2003; Kotwica et al., 2009; Rush et al., 2006). Cells of the drosophila ovary lack functional CRY, which serves as a photoreceptor in drosophila responsible for the light-dependent degradation of timeless (Rush et al., 2006). Regardless, daily rhythms of oviposition (egg laying) are well described in drosophila and persist in mutant flies with a period closely matching that of locomotor activity (McCabe and Birley, 1998). These rhythms can be entrained by temperature cycles and are influenced almost equally by both temperature and light (Kannan et al., 2012). Flies lacking functional period or timeless gene expression do have reproductive deficits marked by irregular oocyte maturation and reduced fecundity (Beaver et al., 2003; TM et al., 2008). Surprisingly, flies lacking pigment-dispersing factor or functional ventral lateral neurons, known to be critical components of the drosophila timing system, continue to display daily rhythms of egg-laying (Howlader et al., 2006; Howlader and Sharma, 2006). Thus, while the location of the endogenous pacemaker for egg-laying rhythms in flies remains to be determined, evidence strongly suggests it is located outside of the ovary.
More is known regarding ovarian clock function in nonmammalian vertebrates, including domestic hens and quail (Nakao et al., 2004; Nakao et al., 2007). It is clear that the circadian clock plays a role in the timing of oviposition and egg laying in birds (Underwood et al., 1997). Nakao and colleagues explored the functional significance of the clock in avian follicles, linking it to transcriptional regulation of steroidogenic enzymes within granulosa cells (GCs; Nakao et al., 2004; Nakao et al., 2007). Given the dearth of evidence (nonmammalian vertebrates) and the general ambiguity (invertebrates) regarding ovarian clock function in these species, this discussion will be limited to the evidence for clock function in the mammalian ovary.
Circadian Clock Function in the Mammalian Ovary
The female reproductive system in mammals exhibits elegant and precise rhythmicity, normally tightly entrained to the light:dark cycle by photoperiodic cues originating in the SCN (see Boden and Kennaway, 2006; Kennaway, 2005). In mammals, circulating gonadotropin LH and follicle-stimulating hormone (FSH) levels oscillate with a diurnal rhythm marked by significant afternoon “surges” on the day of ovulation (Bronson and Vom Saal, 1979; Goldman, 1999; Legan and Karsch, 1975; Moenter et al., 2003). In rodents, the preovulatory LH surge occurs in the late afternoon/early evening on proestrus (the third day of the 4- to 5-d reproductive cycle; Bronson and Vom Saal, 1979; Everett and Sawyer, 1950). By all accounts, this afternoon LH surge is necessary and sufficient for ovulation, luteinization, and subsequent formation of the corpus luteum (Karsch et al., 1997; Moenter et al., 2003). The ovarian follicle is subdivided both anatomically and functionally into the mesenchyme-derived theca cells (TCs), cuboidal androgen- and estrogen-producing cells that line the outside of the follicle, and epithelial granulosa cells (GCs) that line the inside of the follicle, surround the oocyte, and primarily synthesize estrogen (Kacsoh, 2000; Leung and Adashi, 2004). Following follicular rupture, both the TCs and GCs undergo luteinization, resulting in increased vascularization of the GC layer and a transition to increased production of progesterone (Leung and Adashi, 2004). The process of luteinization results in considerable changes in the pattern of steroidogenic enzyme expression in both TCs and GCs (Espey and Richards, 2006). Normal patterns of steroid hormone production, maturation of the follicle, and ovulation depend on the synergistic interaction between these two cell types.
The timing of ovulation, limited to a temporal window on the afternoon of proestrus, depends on the timing of the LH surge (Karsch et al., 1997; Kriegsfeld and Silver, 2006; Moenter et al., 2003). This dogma holds that the ovary simply acts as a vessel for development of the oocyte, responding to threshold levels of LH with initiation of an ovulatory program (Espey and Richards, 2006). However, it has been suggested that the LH surge must arrive at the appropriate time of day to be effective (Sellix et al., 2010). Our experiments reveal that delivery of exogenous LH (equine) to rats previously treated with a selective GnRH receptor antagonist (cetrotide) was effective at inducing ovulation only when the LH was provided during the dark phase (Sellix et al., 2010). We have also seen nearly identical responses to ovine FSH, which is also known to stimulate ovulation through cross-talk between FSH and LH receptors (Galway et al., 1990). This rhythm of ovulatory responsiveness to LH persisted in constant darkness, suggesting that it is gated by the timing system (Sellix et al., 2010). It is possible that SCN-dependent cues of either a humoral (e.g., adrenal steroids) or neural (autonomic nerves) nature play some role in setting this window of sensitivity to gonadotropins. It is equally possible that the ovary contains a circadian oscillator that dictates rhythmic expression of key players in the ovulatory response pathway. This hypothesis challenges the notion that peripheral oscillators in the HPG axis are passive with regard to the timing of key reproductive events. This notion has been extended to other peripheral endocrine organs, including the pituitary, endocrine pancreas, and adrenal gland (Marcheva et al., 2010; Resuehr et al., 2007; Yoder et al., 2014), indicating that the circadian clocks in peripheral endocrine organs may play an active role in the timing of sensitivity to various activating and inhibiting factors. As with these other endocrine organs, recent discoveries have shed additional light on the functional significance of the clock in the mammalian ovary.
Rhythms of clock gene expression have been reported in the intact ovary (Fahrenkrug et al., 2006; He et al., 2007a, 2007b; Karman and Tischkau, 2006; Tischkau et al., 2011) and isolated GCs (Chen et al., 2013a; Chen et al., 2013b; Chu et al., 2012; Chu et al., 2011; He et al., 2007b; Yoshikawa et al., 2009) from both rats and mice. Clock gene expression has also been reported in oocytes and preimplantation embryos from mice (Amano et al., 2009; Hamatani et al., 2004; Johnson et al., 2002), rabbits, and cattle (Amano et al., 2010). Rhythms of clock gene expression were first independently reported in the rat ovary (Fahrenkrug et al., 2006; Karman and Tischkau, 2006). Gonadotropin exposure in vivo induced cyclic expression of bmal1 and per2 mRNA in the ovaries of hypophysectomized prepubertal rats (Karman and Tischkau, 2006). Further, diurnal rhythms of per1 and per2 expression were observed that persisted across the 4- to 5-d estrus cycle (Fahrenkrug et al., 2006). Rhythms of clock gene expression were also detected in large preantral follicles, small antral follicles, Graafian follicles, and corpora lutea. The physiological significance of rhythmic clock gene expression in these cells is poorly defined. It is possible that the ovarian clock modulates the timing of ovulation by regulating the expression of gonadotropin receptors and/or regulates the timing and amplitude of steroid and peptide hormone secretion. It has been suggested that the timing of ovulation may depend on rhythmic sensitivity of the ovary to gonadotropins (Sellix, 2013; Sellix et al., 2010). While these data indicate that the clock in the ovary could drive rhythms of sensitivity to LH, there is little evidence supporting the cell autonomous nature of this sensitivity in follicular cells. We will focus on the mechanism whereby the clock in each different cell type of the ovary contributes to ovarian physiology (summarized in Fig. 2).
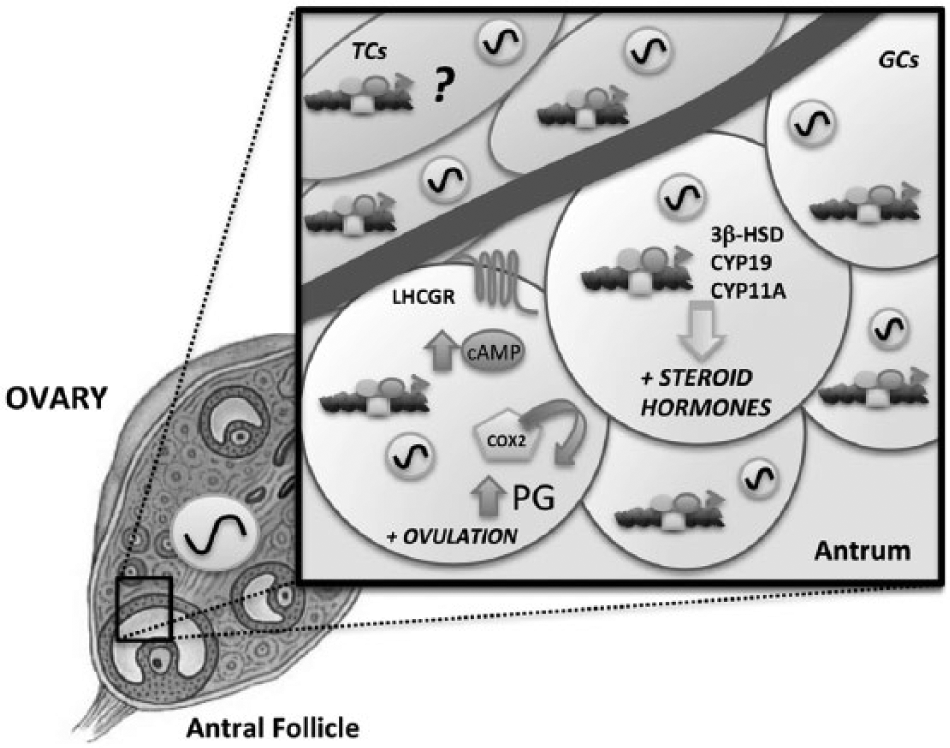
Circadian clock function in ovarian follicles. In granulosa cells (GCs), the clock (BMAL1:CLOCK heterodimer indicated as circles binding to gene promoters) regulates the timing and amplitude of several gene products associated with the ovulatory response to gonadotropins, including the LH receptor (LHCGR) and prostaglandin synthase (COX2). The clock in GCs also drives rhythms of enzymes critical for steroid hormone biosynthesis including steroidogenic acute regulatory protein (StAR), 3β-hydroxysteroid dehydrogenase (3β-HSD), 11β-hydroxylase (CYP11A), and aromatase (CYP19). Although clock genes are rhythmically expressed in theca cells (TCs), there is currently no evidence supporting cell-type–specific regulation of CCG expression in these cells. StAR, steroidogenic acute regulatory protein; 3β-HSD, 3β-hydroxysteroid dehydrogenase; CYP11A, 11β-hydroxylase; CYP19, aromatase; LHCGR, luteinizing hormone/chorionic gonadotropin receptor; COX2, cyclooxygenase-2.
Circadian Clock Function in Ovarian GCs
Most of what we currently know regarding clock function in the mammalian ovary relates to the timing of gene expression in mature or luteinized GCs from rats and mice. It has been reported that rhythms of clock gene expression are limited to mature GCs and luteal cells (Chu et al., 2012; Chu et al., 2011; He et al., 2007a, 2007b). It is well known that the LH surge induces a significant change in gene expression within the GCs lining the preovulatory follicle (Espey and Richards, 2002; Espey et al., 2003; Richards, 2005). However, it remains to be seen if these LH-responsive genes oscillate with a circadian rhythm in the absence of the surge. Several candidate genes associated with the response of GCs to LH can be considered CCGs, including prostaglandin synthase, LH receptor, steroidogenic enzymes, gap junction proteins, and non–clock gene transcription factors (Fig. 2; Chen et al., 2013a; Chen et al., 2013b; Sellix and Menaker, 2010).
A significant step in the response of the GC to LH is the increase in the level of prostanoids, including prostaglandin E2 (PGE2) and PGF2α (Sirois et al., 2004). Reports indicate that the expression of cyclooxygenase-2 (COX2), the rate-limiting enzyme for prostaglandin synthsis, is regulated by the BMAL1::CLOCK complex through E-boxes in the COX2 promoter (Sirois et al., 2004). These data indicate that successful follicular rupture, a process dependent on prostanoid signaling, may require proper transcriptional control by the circadian clock. Another likely candidate is liver receptor homolog 1 (LRH-1), which was first identified as an orphan nuclear receptor in the liver (Nitta et al., 1999). In the ovary, lrh-1 expression is limited to the GC layer (Liu et al., 2003). In the liver, LRH-1 binds to the core circadian clock protein CLOCK (Oiwa et al., 2007) and acts synergistically to drive CLOCK-BMAL1–mediated transcription (Oiwa et al., 2007). Whether LRH-1 has the same activity in ovarian cells is currently unknown. In the ovary, LRH-1 controls steroid hormone biosynthesis in GCs through direct activation of cytochrome P450 side chain cleavage (cyp11a1) transcription following luteinization (Kim et al., 2005). It has been suggested that regular lrh-1 expression in mouse GCs is critical for ovulation (Duggavathi et al., 2008). Whether disruption of molecular clock function in GCs results from, or is linked to, abnormal lrh-1 or cox2 gene expression is unknown. Nevertheless, the reduced fecundity associated with lrh-1 deficiency and the link between lrh-1 or cox2 transcription and clock gene expression make it likely that disruption of the oscillator in the ovary will have detrimental effects on fertility.
Treatment with PGE2 in vivo has been shown to phase shift the rhythm of per1, d-element binding protein (dbp), and rev-erbα mRNA expression in the heart, liver, and kidney (Tsuchiya et al., 2005). Most recently, it was revealed that luteinized GCs do in fact have robust circadian rhythms of ptgs2 and LH receptor (lhcgr) gene expression that are disrupted and in some cases abolished following attenuation of bmal1 expression (Chen et al., 2013b). These data indicate that rhythms of lhcgr expression are directly regulated by the BMAL1:CLOCK enhancer complex binding to E-box motifs in the lhcgr promoter. Further, these data suggest that an increase in COX2 and LHCGR expression and/or PG secretory activity preceding the arrival of the LH surge may facilitate the responsiveness of GCs to LH and the subsequent transition to a functional corpus luteum. Additional evidence for a link between the circadian clock and PG signaling was recently discovered in the bovine uterus (Isayama et al., in press). The expression of both reverbα and ptgs2 in bovine uterine stromal cells (USCs) was significantly down-regulated following treatment with bmal1 small interfering RNA (siRNA). Further, production of PGF2α was reduced in USCs following exposure to bmal1 siRNA. Finally, levels of ptgs2 mRNA in both USC and uterine epithelial cells were increased following treatment with the synthetic REVERBα antagonist SR8278, suggesting that REVERBα directly suppresses ptgs2 gene expression in uterine tissue, most likely through binding at rev-erb response elements in the ptgs2 promoter. This evidence supports the assertion that a similar activity of REVERBα controls the expression of ptgs2 in GCs. It was recently suggested that the appearance of rhythmic clock gene expression in maturing follicles depends on FSH-induced expression of gap junctions in GCs (Chen et al., 2013a). According to these reports, disruption of intercellular communication via gap junction blockers reduces the amplitude and lengthens the period of PER2 expression in rat GCs (Chen et al., 2013a), the suggestion being that gonadotropin-dependent communication among GCs could play a critical role in the appearance and/or maintenance of CCG expression in the follicle. Together, these data indicate that multiple core clock genes both directly or indirectly modulate the timing and amplitude of CCG expression in GCs.
Circadian Clock Function in Ovarian TCs
Because of the difficulty in isolating purified populations, evidence for cell autonomous clock function in ovarian TCs and interstitial (stromal) cells has been limited to histological analyses (Fahrenkrug et al., 2006; Gras et al., 2012; Karman and Tischkau, 2006). While the clock genes are rhythmically expressed in these cells, evidence for a clock-controlled program of cell-type–specific gene expression in theca and stromal cells remains elusive. Given TCs’ critical involvement in ovarian steroid production and their established role in both follicular growth and ovulation, it is imperative that we better define the role of the circadian clock in TCs going forward. The effects of circadian disruption on TCs may have considerable implications for fertility, particularly as it relates to the impact of chronodisruption on androgen synthesis and secretion from these cells (Sen and Hammes, 2010).
Circadian Clock Function in the Oocyte
Clock genes are expressed in mouse, rabbit, and bovine oocytes (Amano et al., 2009; Amano et al., 2010; Hamatani et al., 2004; Johnson et al., 2002). In mice, clock gene mRNA fluctuates as a function of developmental stage (Amano et al., 2009). However, it has been suggested that, rather than driving daily rhythms of gene expression, they are part of the maternal program responsible for normal oocyte maturation. The increased levels of cry1 mRNA expression in germinal vesicle oocytes suggests that it plays a role, along with clock and bmal1, in suppressing CCG expression during oocyte maturation and early embryonic development. In support of this hypothesis, it was determined that knockdown of cry1 mRNA expression with siRNA leads to slight inhibition of meiosis progression in murine oocytes (Amano et al., 2009). Together, these data support an oscillator-independent role for the clock genes in the development of the mature oocyte.
It was recently reported that mice with a conditional knockout of the BMAL1 locus in steroidogenic cells (steroidogenic-factor 1-Cre; Bmal1flx/flx) show severe deficits in implantation success and compromised progesterone secretion (Liu et al.,2014). Notably, these mice showed normal estrous cycles, rhythms of behavior and number of corpora lutea when compared with littermate controls. Normal implantation was rescued when WT ovaries were transplanted to SF1-Cre;BMAL1-/- mice and implantation rates were reduced when conversely, KO ovaries were transplanted into WT hosts. These data strongly suggest that BMAL1 deletion in steroidogenic cells of the ovary disrupts implantation due to abnormal progesterone secretion. These effects are not due to disruption of the endometrial clock, as the SF1-Cre transgene used was reportedly not expressed in uterine tissue. This construct did result in deletion of BMAL1 in the brain, adrenal gland and pituitary. Nonetheless, these data support the hypothesis that the ovarian clock plays a role in fertility, linked in large part to the control of ovarian steroid hormone synthesis.
Circadian Clock in the Ovary: Role in Steroidogenesis
Evidence from rodents (Alvarez et al., 2008; Chen et al., 2013a; Chen et al., 2013b; Ratajczak et al., 2009) and birds (Nakao et al., 2007) suggest that the circadian clock plays a significant role in the amplitude and timing of steroid hormone synthesis in the ovary. Circadian rhythms of steroidogenic acute regulatory protein (StAR), 3β-hydroxysteroid dehydrogenase, 11β-hydroxylase, and aromatase (cyp19) have been observed in mature GCs (Chen et al., 2013a; Chen et al., 2013b). These rhythms are altered or abolished following clock disruption (Chen et al., 2013b). Further, bmal1–/– mice have abnormally low levels of progesterone secretion due to reduced StAR expression (Ratajczak et al., 2009). These data suggest that chronodisruption alone may lead to abnormal patterns and/or levels of ovarian steroid hormone synthesis and secretion or exacerbate the already negative effects of endocrine pathology. It was recently reported that mice with a conditional knockout of the BMAL1 locus in steroidogenic cells (steroidogenic-factor 1-Cre; Bmal1flx/flx) show severe deficits in implantation success and compromised progesterone secretion (Liu et al., 2014). Notably, these mice showed normal estrous cycles, rhythms of behavior and number of corpora lutea when compared with littermate controls. Normal implantation was rescued when WT ovaries were transplanted to SF1-Cre;BMAL1-/- mice and implantation rates were reduced when conversely, KO ovaries were transplanted into WT hosts. These data strongly suggest that BMAL1 deletion in steroidogenic cells of the ovary disrupts implantation due to abnormal progesterone secretion. These effects are not due to disruption of the endometrial clock, as the SF1-Cre transgene used was reportedly not expressed in uterine tissue. This construct did result in deletion of BMAL1 in the brain, adrenal gland and pituitary. Nonetheless, these data support the hypothesis that the ovarian clock plays a role in fertility, linked in large part to the control of ovarian steroid hormone synthesis.
Is the Ovarian Clock Required for Normal Reproductive Function?
The cells of the ovarian follicle display clear phasic sensitivity to gonadotropins, suggesting the activity of a circadian oscillator in the GC, TC, or both (Yoshikawa et al., 2009). Data showing rhythmic expression of the LH receptor gene in GCs also support this notion (Chen et al., 2013b). Further, our data suggest that the timing of ovulation depends not only on the rhythm of LH secretion, itself driven by the SCN, but also on the timing of ovarian sensitivity to gonadotropins potentially set by the ovarian clock (Sellix et al., 2010). While the location and contribution to physiological function of the oscillator remain a subject of conjecture, considerable evidence suggests that it is located in the follicle. Evidence to either refute or confirm this hypothesis has yet to materialize. Extrinsic inputs to the ovary, including adrenal glucocorticoids and sympathetic nervous cues, might also contribute to the timing of ovarian sensitivity. It is thus equally plausible that the SCN coordinates the timing of ovulation independently of, or in partnership with, the ovarian clock through non–gonadotropin-dependent signaling.
Impaired fertility and fecundity have been observed in behaviorally arrhythmic clock mutant mice (Alvarez et al., 2008; Boden et al., 2010; Dolatshad et al., 2006; Miller et al., 2004; Ratajczak et al., 2009). However, these animals are still able to ovulate and produce fertilizable ova (Ratajczak et al., 2009). While these data suggest that mutations that globally disrupt the clock have negative effects on fertility, they do not address the specific role of the clock in ovarian follicles. Lack of an effect of a global mutation on ovulation may result from developmental compensation or redundancy within the core clock (as in Clock-null mice, see Debruyne et al., 2006). Studies using targeted knockout of clock gene expression in specific follicular cells, as was recently reported in the pituitary (Chu et al. 2013), should shed some additional light on clock function in specific compartments of the developing follicle. Overall, the evidence for autonomous clock function in follicular cells supports a role for the clock in ovarian steroid synthesis, follicular growth, and ovulation. Identifying a link between the timing of the clock, the timing of ovarian sensitivity to LH, and the expression of CCGs such as lrh-1, ptgs2, and gap junctions may lead to improved understanding of the molecular bases of infertility.
Ovarian Clock Function and Fertility Disorders: New Discoveries and Future Promise
The timing of clock gene expression in the rat and mouse ovary is clearly influenced by the developmental stage of the follicle (Gras et al., 2012). Further, it has been suggested that the ovarian clock is influenced by the varying titer of serum steroid hormones across the cycle (Murphy et al., 2013; Nakamura et al., 2010). Limited evidence supports a modest feedback effect of estrogen and progesterone on circadian rhythms of behavior (Royston et al., 2014; Takahashi and Menaker, 1980). Ovariectomy in rats and hamsters lengthens the free-running period of locomotor activity, an effect that can be reversed following estrogen replacement (Albers, 1981; Morin et al., 1977). Although steroid receptors are present in SCN pacemaker neurons, steroid hormones at physiologically relevant concentrations do not appear to affect the timing of clock gene expression in the SCN (Murphy et al., 2013; Nakamura et al., 2008). However, estradiol can modulate the timing of clock gene expression in other regions, including areas of the basal forebrain that send dense projections to the SCN, raising the possibility of indirect control of behavior by steroid hormone feedback (Perrin et al., 2006). While the means of steroid feedback remain unresolved, recent evidence points to estrogen receptor subtype–specific effects on rhythms of locomotor activity (Royston et al., 2014). Activated steroid receptors may act downstream of the molecular clock, a possibility supported by the fact that estrogen can alter the expression of gap junctions in SCN neurons (Shinohara et al., 2001). SCN pacemaker neurons certainly express steroid hormone receptors, including estrogen, progestin, and androgen receptors (Iwahana et al., 2008; Karatsoreos et al., 2011; Vida et al., 2008). It is worth noting that androgens can affect the period of locomotor activity, and gonadectomy alters the timing and amplitude of clock gene expression in SCN neurons (Daan et al., 1975; Karatsoreos et al., 2011; Karatsoreos et al., 2007). The effects of ovarian steroid hormones on peripheral clock function are less ambiguous, with clear effects of steroids on rhythms of clock gene expression in tissues including the uterus, liver, and ovary (He et al., 2007c; Hirao et al., 2011; Nakamura et al., 2005; Nakamura et al., 2010). Regardless of the mechanism, the timing system is clearly affected by steroid hormones, with tissue-specific effects apparent in both central and peripheral oscillators.
While the feedback effects of ovarian steroids on central clock function remain to be clarified, recent evidence suggests that irregular patterns of hormone secretion can globally influence the timing system. A common feature of fertility disorders such as polycystic ovarian syndrome (PCOS) is disruption of neuroendocrine and endocrine function leading to irregular patterns and timing of ovarian steroid hormone secretion (Franks and Hardy, 2010; Nisenblat and Norman, 2009; Padmanabhan and Veiga-Lopez, 2011). Altered timing of clock gene expression in peripheral tissues such as the ovary may occur as a result of, or be partially responsible for, the hormonal imbalances associated with these disorders (Amaral et al., 2014). As an example, an irregularly high serum androgen level during sexual development is a common clinical feature of PCOS (Shi and Vine, 2012). Excess androgen, or hyperandrogenemia, leads to a dramatic change in the timing system, particularly the timing of clock gene expression in the ovarian follicle and liver (Sellix et al., 2013). These data suggest that reproductive endocrinopathies, particularly those arising due to developmental programming by abnormal steroid environments (e.g., PCOS), can alter the timing of the clock in tissues associated with both reproductive and metabolic function. This effect on the timing of ovarian clock gene expression may compound the negative effects of excess androgen on key components of the steroid biosynthetic and ovulatory response pathways. It is possible that treatment with clock-modulating compounds or chronobiotics, such as the REVERBα ligands or cryptochrome modulators (Chen et al., 2012), could be used as novel strategies for improving reproductive success in individuals suffering from this and other fertility disorders. Moreover, treatment with melatonin or melatonin receptor agonists commonly used for the treatment of insomnia or non–24-h sleep disorder might also improve internal circadian organization and resolve reproductive and metabolic dysfunction (Cardinali et al., 2011; Pai and Majumdar, in press). Other environmental manipulations such as precisely timed exercise or caloric restriction that are known to alter circadian organization may also improve HPO axis function (Mendoza et al., 2012; Schroeder et al., 2012). It remains to be seen whether manipulation of the timing system by any means, either alone or in conjunction with other therapies, represents a viable approach for addressing reproductive dysfunction in women.
Conclusion
The timing of endocrine rhythms is a pervasive aspect of mammalian reproductive physiology, critical for life on a rotating planet. Although we have made great strides in our understanding of the timing system’s role in endocrinology, we have seen only a glimpse of its potential function. The depth of integration, at the cellular and molecular level, remains to be determined for the majority of hormones and hormone regulatory pathways. It is apparent that the timing system plays a significant role in ovarian physiology, participating in the processes of ovulation, steroid hormone synthesis, and oocyte maturation. While we certainly know a great deal, many critical questions remain. How does the clock regulate the response of the ovary to hormones and neural cues, particularly at the level of each ovarian cell type? When is the molecular clock actually “turned on” in developing follicular cells? What is the mechanism regulating the ontogeny of the clock in ovarian cells? And, lastly, how might adjusting the timing of the clock in ovarian follicles, or altering the synchrony between the ovarian clock and other tissues of the HPO axis, improve physiological function and/or diminish the risk of endocrine pathophysiology? As we face a growing epidemic of infertility due to genetic disease and environmental factors such as obesity, diabetes, and endocrine-disrupting compounds, the information we gain from the study of clock function in these tissues may provide the key to identifying new and effective treatments. Further, answering these questions may yield additional insights into the study of stem cells, developmental biology, and cancer biology.
Footnotes
Acknowledgements
The author wishes to thank Ms. Amanda Mereness for proofreading and assistance with the preparation of the manuscript.
