Abstract
The cockroach Rhyparobia (Leucophaea) maderae expresses a circadian rhythm in pheromone-dependent mating activity that peaks at the late day/early night. In contrast, the circadian rhythm in olfactory sensitivity of the Madeira cockroach is at its minimum during this time. Until now, the reasons for this obvious discrepancy in phase were not understood. Previously, it was shown that cyclic nucleotides modulate olfactory sensitivity in a zeitgeber time (ZT)–dependent manner. In moths’ olfactory receptor neurons, adapting pheromone concentrations elevate cGMP levels, which decrease pheromone sensitivity. In contrast, cAMP elevations sensitized pheromone responses. Thus, with immunoassay kits, it was determined whether cAMP and cGMP baseline levels vary in a ZT-dependent manner in antennal lysates of female R. maderae, revealing underlying circadian rhythms in olfactory sensitivity. Furthermore, it was examined whether adapting pheromone exposure elevates cGMP levels in cockroach antennae, possibly overshadowing underlying circadian rhythms in sensitivity via sensory adaptation. It was shown for the first time that cAMP and cGMP baseline levels oscillate in antiphase in a ZT-dependent manner in an insect’s antenna, with the maximum in cAMP concentrations coinciding with maximal mating activity during the late day. Moreover, the cAMP baseline level oscillation expressed a circadian rhythm since it persisted under constant darkness in contrast to cGMP baseline levels. Furthermore, while excess exposure to male pheromones increased cGMP and decreased cAMP baseline levels, the stress hormone octopamine increased adenylyl cyclase activity at all ZTs tested. Therefore, it is suggested that cyclic nucleotide–dependent modulation of olfactory sensitivity due to olfactory overstimulation and stress could be responsible for previously measured phase discrepancies between rhythms in mating behavior and pheromone sensitivity.
The cockroach Rhyparobia maderae (Leucophaea maderae) is an established model organism for circadian rhythm research (Homberg et al., 2003; Page, 1982). Its major circadian timekeeping center, the accessory medulla, which is located in the optic lobes and controls circadian locomotor activity rhythms, was intensely investigated (Colwell and Page, 1990; Homberg et al., 2003; Nishiitsutsuji-Uwo and Pittendrigh, 1968; Page, 1982, 2009; Reischig and Stengl, 2003; Roberts, 1974; Sokolove, 1975; Stengl and Homberg, 1994). Also, circadian rhythms of mating activity were observed in R. maderae, with maxima at the late day at zeitgeber time (ZT) 8 to 10 and the beginning of the night at ZT 12 to 14 (Rymer et al., 2007). The zeitgeber employed was the light-dark (L/D) cycle with the light phase at ZT 0 to 12 and the dark phase at ZT 12 to 24. The cellular and molecular mechanisms of these behavioral rhythms, however, remained elusive. Mating activity in cockroaches is controlled via volatile pheromones released by males to attract females (Farine et al., 2007; Sreng, 1993). In addition, contact pheromones in the male’s cuticle appear to guide the decision of female cockroaches to engage in copulation or not (Sreng, 1984, 1993). To determine whether circadian rhythms in pheromone detection might underlie circadian mating activity rhythms, extracellular recordings (electroantennograms [EAGs]) of the summed activity of antennal cells in response to pheromones were obtained (Rymer et al., 2007; Saifullah and Page, 2009). Surprisingly, the maximal amplitudes of the pheromone- and food odor–dependent EAGs as well as odor-dependent action potential frequencies of olfactory receptor neurons (ORNs) obtained in single sensillum recordings were at the early day (Page and Koelling, 2003; Saifullah and Page, 2009). In contrast to expectations, the minimal amplitudes of antennal parameters indicating minima of olfactory sensitivity were at the late day during the peak of mating activity and the time of best olfactory learning of food sources (Decker et al., 2007; Rymer et al., 2007). In contrast to cockroach data, in moths, circadian rhythms in odor sensitivity coordinate circadian rhythms in mating with phase-locked maxima of both rhythms during the activity phase and minima during the moths’ rest phase (Linn et al., 1996; Rosen et al., 2003; Stengl, 2010).
The reasons for this obvious discrepancy between the phase of the circadian rhythms in the antenna and the behavior of the Madeira cockroach are not understood. Apparently, unknown mechanisms adapt (desensitize) odor detection during the late day and/or sensitize odor detection during the resting phase at the beginning of the day. Thereby, underlying antennal rhythms in pheromone sensitivity might be masked and phase shifted. In moth ORNs, changes in cyclic nucleotide levels modulate odor detection in the antenna (Stengl, 2010). In different species, the stress hormone octopamine (OA) sensitized pheromone detection and pheromone-dependent behavior ZT- and cAMP-dependently (Farooqui, 2007; Flecke et al., 2006; Flecke et al., 2010; Flecke and Stengl, 2009; Linn et al., 1992; Linn and Roelofs, 1986; Pophof, 2002; Zhukovskaya and Kapitsky, 2006). In contrast, elevations in cGMP occurred after adapting odor exposure (Boekhoff et al., 1993; Stengl et al., 2001; Ziegelberger et al., 1990). In addition, perfusion with cGMP analogs decreased the response to pheromones in the hawk moth in a ZT-dependent manner (Flecke et al., 2006; Redkozubov, 2000). Therefore, antennal cAMP concentration increase sensitize while cGMP increase adapt odor detection in insects. However, it remained unknown whether circadian rhythms of cyclic nucleotides occur in insect antennae. Therefore, here with immunoassays, it was examined whether in antennae of the Madeira cockroach, cyclic nucleotide levels are controlled by a circadian pacemaker. In addition, it was determined whether the stress hormone OA and/or excess male pheromones in crowded colonies change cyclic nucleotide levels and thus have the potential to override endogenous circadian rhythms of odor sensitivity.
Materials and Methods
Animals
The experiments were performed with adult female cockroaches that were taken from laboratory colonies. They were reared under a 12:12-hour L/D photoperiod at 50% relative humidity and 25 °C room temperature. Dried dog food, vegetables, and water were provided ad libitum. For the adaptation (sensory desensitization) experiments, 2 sets of data were compared to determine the effect of excess male pheromones on cyclic nucleotide concentrations in female antennae. The first set of female antennae was obtained by collecting females from laboratory cockroach colonies kept under conditions of an equal ratio of males to females. The second batch of female antennae was collected from colonies with a large excess of male cockroaches to expose females to adapting concentrations of male pheromones.
Preparation and Quantification of Cyclic Nucleotides
For sample collections, 10 female cockroaches each were taken out of laboratory colonies at either ZT 2, 5, 10, 14, or 20. They were stored temporarily in dark vessels to reduce stress and allow for calming down. Then, quickly, the animals were shock-frozen in liquid nitrogen by rapidly emptying the vessel into a dewar. To examine whether cyclic nucleotide concentrations oscillate under constant darkness (DD), females were kept at ZT 20, isolated in vessels, and shock-frozen 2, 5, 10, 14, and 20 hours after the last L/D cycle. Frozen antennae were ground in a mortar, transferred into cups, and mixed with homogenization buffer (20 antennae/400 µL). The homogenization buffer contained 0.05% sodium cholate, 200 mM sodium chloride (NaCl), 2 mM magnesium chloride (MgCl2), 10 mM ethylene glycol-bis(2-aminoethylether)-N,N,N’,N’-tetraacetic acid (EGTA), 50 mM 3-(N-morpholino) propanesulfonic acid (MOPS), and 1 mM DL-1,4-dithiothreitol (DTT) at pH 7.0. After mixing 3 times for 10 seconds, samples were centrifuged at 900 g for 5 minutes at 4 °C. Then, 50 µL of the supernatant was transferred into 200 µL of incubation buffer at 37 °C. Protein determination with 5 µL of homogenate in duplicate was performed with Bradford assays (Bradford, 1976). The incubation buffers contained 0.05% sodium cholate, 200 mM NaCl, 5 mM MgCl2, 90 µM CaCl2, 1 mM 1,2-bis(o-aminophenoxy)ethane-N,N,N’,N’-tetraacetic acid (BAPTA), 50 mM MOPS, 1 mM DTT, 1 mM adenosine triphosphate, 4 µM guanosine triphosphate, and 1 mM 3-isobutyl-1-methylxanthine (IBMX) at pH 7.0 (modified after Vogl et al., 2000). IBMX was necessary to prevent degradation of cyclic nucleotides. The OA buffer contained 50 µM of the stress hormone OA. Therefore, the resulting OA concentration in the mixture of 50 µL of homogenate and 200 µL of OA buffer was 20% lower. The incubation lasted 10 minutes and was stopped by adding 100 µL of 7% perchloric acid solution. This step was followed by mixing the contents and centrifuging at 900 g for 15 minutes at 4 °C. All proteins were denatured by perchloric acid. To neutralize the liquid phase, 200 µL of each incubation solution was mixed 3 times for 10 seconds with 50 µL of 10 nM ethylenediaminetetraacetic acid (EDTA) solution and 250 µL of chloroform/trioctylamine solution (1:1) and then centrifuged at 500 g for 5 minutes at 4 °C. Supernatant was taken for quantifying cAMP and cGMP concentrations. Commercially available immunoassay kits for determining cAMP and cGMP concentrations were used (TRK500, TRK432, GE Healthcare, Chalfont St. Giles, UK; 581001, 581021, Cayman Chemical, Ann Arbor, MI). Because of the different weight of antennae per samples, the cyclic nucleotide concentrations were normalized by calculating a quotient of cyclic nucleotide concentration by corresponding protein concentration. For n = 1, 20 antennae were sampled. To control intracellular Ca2+ concentrations for all assays, 10 nM free Ca2+ concentrations were used in buffers (for calculation: Webmaxc Standard software, Stanford University, Stanford, CA).
Data Analysis of Cyclic Nucleotide Experiments
Before data were statistically evaluated, the distribution of the data was analyzed by using the Shapiro-Wilk test and the Kolmogorov-Smirnov-Lilliefors test. Since the results of the normality tests showed no normal distribution, the data were analyzed by using nonparametric tests. In the experiments, 5 different ZTs were compared. Here, the Kruskal-Wallis test, which compares more than 2 unpaired nonparametric groups for difference in the population, was used, followed by the Mann-Whitney test including the Bonferroni correction. This correction is necessary to address the problem of multiple comparisons; thus, the significance level of α = 0.05 is reduced to α = 0.01. To determine the effect of OA on adenylyl cyclases (ACs), OA and OA-free buffers were used. For achieving directly comparable results, tested buffers were applied to samples from 1 homogenate. Thus, effects can be proved even if variation is high. Here, the Wilcoxon test for paired samples was used. Most results are presented in box plots, including the median (horizontal band near the middle of the box), the quartiles (bottom of box: 25th percentile, median of the lower 50% of the data; and top of box: 75th percentile, median of the upper 50% of the data), and the whiskers (2 lines outside the box: 10th/90th percentiles, 80% of all data).
Results
Circadian mating activity rhythms and circadian rhythms in pheromone sensitivity of the Madeira cockroach appeared to be phase-shifted via unknown mechanisms. We hypothesized that synchronized circadian rhythms of antennal sensitivity and behavior are masked stimulus-dependently. Masking stimuli were suggested to be adapting pheromone stimulation and the stress hormone OA, which couples to ACs. Since adapting pheromone stimulation elevates antennal cGMP levels while OA sensitizes apparently cAMP-dependently (Stengl, 2010), with immunoassay kits, cAMP and cGMP concentrations were quantified in antennal lysates of adult female R. maderae. Experiments were performed at different ZTs and at DD (circadian times [CTs]) to distinguish stimulus-dependent changes in cyclic nucleotides from endogenous circadian rhythms. Furthermore, it was examined whether the stress hormone OA or adapting pheromone concentrations change antennal cyclic nucleotide levels stimulus-dependently.
Cyclic Nucleotide Concentrations Change in a ZT-Dependent Manner in Antennal Lysates
Female antennae collected under raring conditions of about equal ratios of males to females expressed ZT-dependent changes in cyclic nucleotide concentrations (Fig. 1A-C). The cGMP level expressed its minimum shortly before and at the beginning of the mating phase at ZT 5 and 10 (Fig. 1A, 1C). Thus, there was a highly significant difference between the cGMP concentrations at ZT 2, 5, 10, 14, and 20 (Kruskal-Wallis test, α = 0.001, p < 0.001). At ZT 2 (n = 11), the median was 15.1 pmol/mg, at ZT 5 (n = 12), it was 6.9 pmol/mg, at ZT 10 (n = 14), it was 6.8 pmol/mg, at ZT 14 (n = 11), it was 18.5 pmol/mg, and at ZT 20 (n = 9), it was 16.3 pmol/mg. Therefore, the cGMP levels at ZT 5 were significantly reduced compared to ZT 2 (Mann-Whitney test including Bonferroni correction, α = 0.0002, p < 0.0002) and compared to ZT 14 and ZT 20 (Mann-Whitney test including Bonferroni correction, α = 0.01, p < 0.01). Furthermore, the cGMP level at ZT 10 was highly significantly reduced compared to ZT 2 and ZT 14 (Mann-Whitney test including Bonferroni correction, α = 0.0002, p < 0.0002) and significantly reduced compared to ZT 20 (Mann-Whitney test including Bonferroni correction, α = 0.01, p < 0.01).
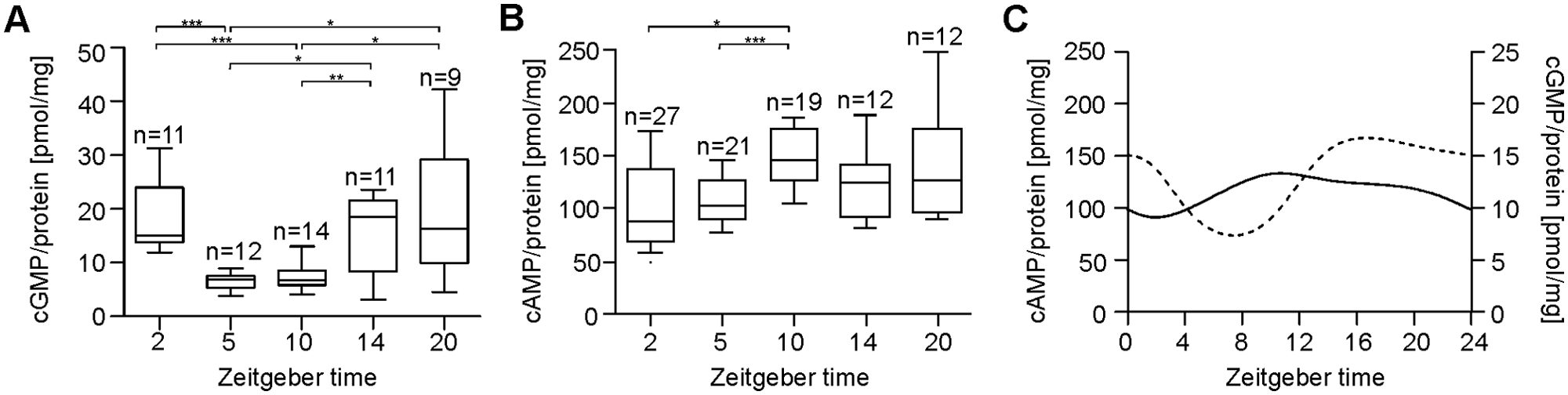
ZT-dependent changes in cyclic nucleotide concentrations under conditions of about an equal ratio of males to females. (A) Antennal cGMP levels of female Madeira cockroaches are significantly reduced at ZT 5 and 10 compared to ZT 2, 14, and 20 (Mann-Whitney test including Bonferroni correction, α = 0.01, p < 0.01). (B) The cAMP levels are significantly lower at ZT 2 and 5 as compared to ZT 10 (Mann-Whitney test including Bonferroni correction, α = 0.01, p < 0.01). (C) Cyclic nucleotide oscillations are presented as a B-spline curve by calculating the medians at each time investigated. The solid line represents cAMP baseline levels (left); the dashed line represents cGMP baseline levels (right). The incubation buffer contained 10 nM Ca2+ (n = 1: 20 female antennae).
In addition, cAMP concentrations also changed in a ZT-dependent manner with maximum values at ZT 10 (Kruskal-Wallis test, α = 0.01, p < 0.01). The medians at ZT 2 (87.2 pmol/mg; n = 27) and ZT 10 (145.1 pmol/mg; n = 19) significantly differed (Mann-Whitney test including Bonferroni correction, α = 0.01, p < 0.01) as well as at ZT 5 (103.9 pmol/mg; n = 21) and ZT 10 (Mann-Whitney test including Bonferroni correction, α = 0.0002, p < 0.0002). Thus, the cAMP level was significantly higher during the mating phase of the cockroaches at the late day as compared to the resting phase at the beginning and middle of the day (Fig. 1B, 1C). The medians at ZT 14 (124.9 pmol/mg; n = 12) and ZT 20 (126.1 pmol/mg; n = 12) did not differ from other medians at other ZTs.
The cAMP Baseline Concentration Oscillation Persists under DD in Contrast to cGMP Baseline Concentration Changes
Female antennae collected on the first day of DD expressed rhythms in cAMP baseline levels (Fig. 2A, 2C), whereas the cGMP baseline levels decreased progressively (Fig. 2B, 2C). There was a significant difference between the cAMP baseline levels at CT 2, 5, 10, 14, and 20 (Kruskal-Wallis test, α = 0.05, p < 0.05). At CT 2 (n = 13), the cAMP median was 108.2 pmol/mg and increased until CT 10 (132.6 pmol/mg; n = 13). Then, the cAMP baseline levels decreased until CT 20 (81.0 pmol/mg; n = 13). This decrease was significant (Mann-Whitney test including Bonferroni correction, α = 0.01, p < 0.01) (Fig. 2A). Interestingly, a continuous decrease in cGMP baseline level concentrations was observed. Moreover, this time-dependent decrease was highly significant (Kruskal-Wallis test, α = 0.001, p < 0.001). Thereby, the highest concentration of cGMP was detected at CT 2 (n = 8) with 1.0 pmol/mg, whereas at CT 20 (n = 9), the lowest cGMP concentration (0.3 pmol/mg) could be detected. This effect was highly significant (Mann-Whitney test including Bonferroni correction, α = 0.0002, p < 0.0002) (Fig. 2B).
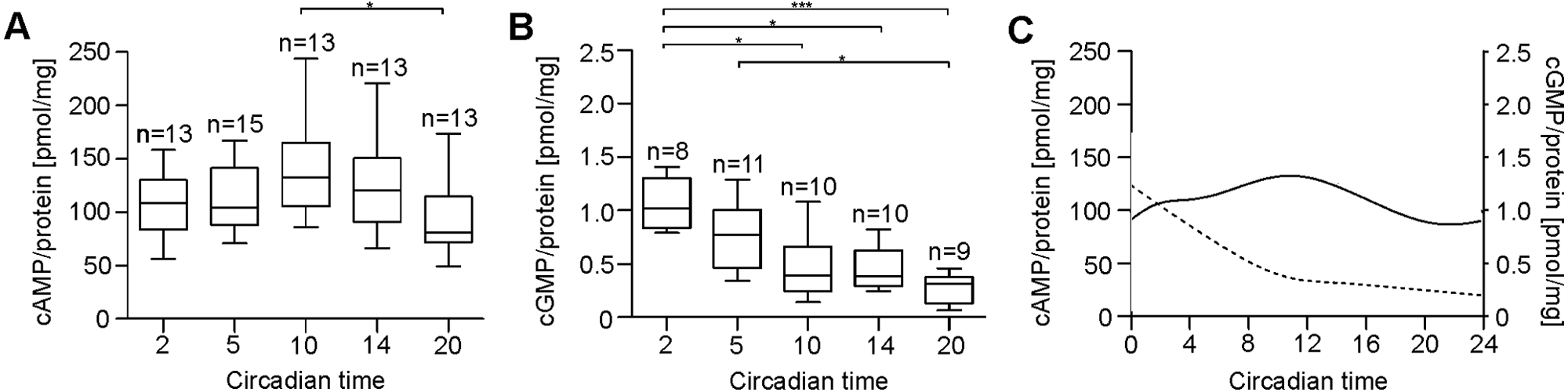
Changes in cyclic nucleotide concentrations under DD. (A) In DD, antennal cAMP levels are significantly elevated at CT 10 as compared to CT 20 (Mann-Whitney test including Bonferroni correction, α = 0.01, p < 0.01). (B) The cGMP level is significantly reduced at CT 10, 14, and 20 as compared to CT 2 (Mann-Whitney test including Bonferroni correction, α = 0.01, p < 0.01). Furthermore, the cGMP level at CT 5 significantly differed from CT 20 (Mann-Whitney test including Bonferroni correction, α = 0.01, p < 0.01). (C) Additionally, cyclic nucleotide oscillations are presented as a B-spline curve by calculating the medians at each time investigated. The solid line represents cAMP baseline levels (left); the dashed line represents cGMP baseline levels (right).
Cyclic Nucleotide Concentrations in Antennal Lysates of Female Cockroaches Depend on Male-to-Female Ratios of Cockroach Colonies
In the second set of experiments, stimulus-dependent changes in cyclic nucleotide levels were examined. Since adapting pheromone stimulation increases antennal cGMP levels in moths (Stengl et al., 2001), antennae were collected from cockroach colonies with a strong surplus of males to increase male pheromone concentrations to adapting levels. As expected, cGMP concentrations in female antennae significantly increased both at ZT 5 and 10 (ZT 5, set 1: n = 12, set 2: n = 16; Mann-Whitney test, α = 0.001, p < 0.001) (ZT 10, set 1: n = 14, set 2: n = 8; Mann-Whitney test, α = 0.001, p < 0.001) (Fig. 3A, 3B). In contrast, a strong surplus of males decreased antennal cAMP concentrations at ZT 5 (set 1: n = 21, set 2: n = 13; Mann-Whitney test, α = 0.001, p < 0.001) (Fig. 3C) but not at ZT 10 (set 1: n = 19, set 2: n = 4) (Fig. 3D).
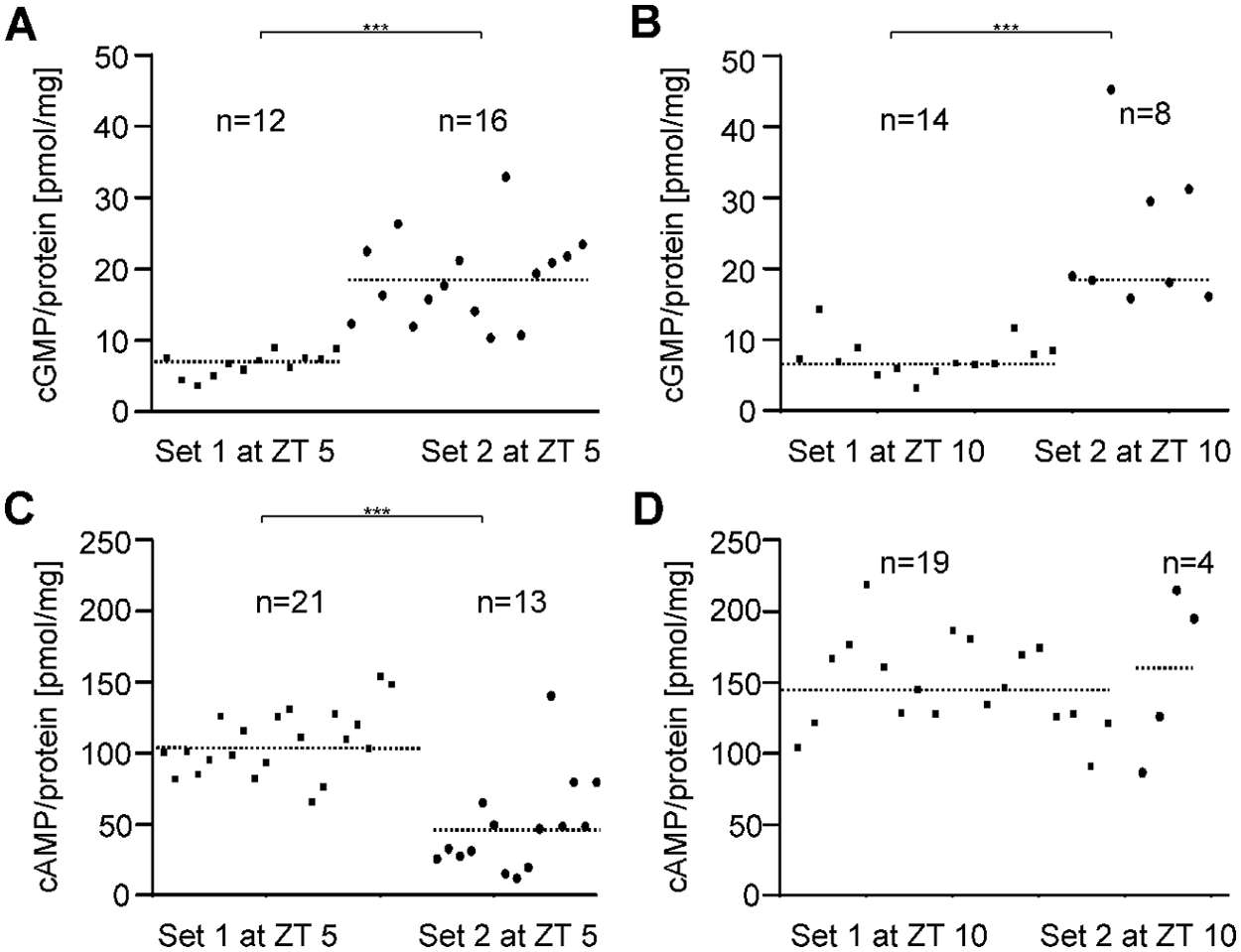
Increased exposure to male pheromones due to high male-to-female ratios in laboratory colonies affects baseline levels of cyclic nucleotides. Two data sets were collected: set 1 at conditions of about an equal ratio of males to females, and set 2 at conditions of elevated male pheromone exposure caused by an excess of courting males. (A, B) The cGMP concentrations significantly increased under conditions of strong and prolonged pheromone exposure at both ZT 5 and 10 (Mann-Whitney test, α = 0.001, p < 0.001). (C, D) In contrast, cAMP levels decreased significantly at ZT 5 (Mann-Whitney test, α = 0.001, p < 0.001), while no statistical difference was found at ZT 10. The incubation buffer contained 10 nM Ca2+ (n = 1: 20 female antennae).
The ZT Dependence of cGMP Concentrations Is Lost in Antennal Lysates of Female Cockroaches Raised with an Excess of Males
Female antennae collected from colonies with a strong surplus of males did not express a significant difference in the cGMP concentrations at different ZTs (Kruskal-Wallis test, α = 0.05, p > 0.05) (Fig. 4A). Medians at ZT 2 (14.0 pmol/mg; n = 4), 5 (18.5 pmol/mg; n = 16), 10 (18.7 pmol/mg; n = 8), 14 (12.9 pmol/mg; n = 8), and 20 (15.6 pmol/mg; n = 8) did not differ significantly (Fig. 4A). However, the highest cGMP concentrations were detected at ZT 5 and 10. The cAMP concentrations collected at ZT 5 (31.8 pmol/mg; n = 13) were highly significantly decreased (Mann-Whitney test, α = 0.01, p < 0.01) (Fig. 4B) compared to ZT 10 (160.4 pmol/mg; n = 4).
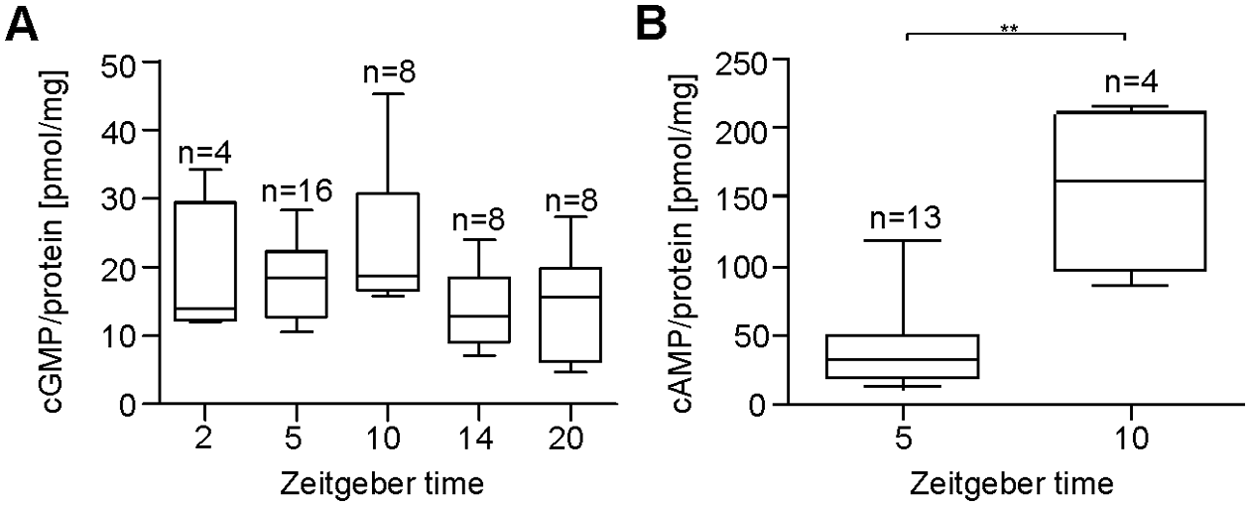
An increase in the male-to-female ratio overshadows previously measured ZT-dependent rhythms in cGMP but not in cAMP concentrations. (A) At different ZTs, no significant differences in cGMP baseline levels are detectable under conditions of excess male pheromones due to increased levels at ZT 5 and 10. (B) However, the cAMP baseline level is reduced highly significantly further at ZT 5 as compared to its maximum level at ZT 10 (Mann-Whitney test, α = 0.01, p < 0.01). The incubation buffer contained 10 nM Ca2+ (n = 1: 20 female antennae).
OA Elevates cAMP Levels Both at Rest as Well as at the Peak Time of Mating Activity
To search for stress-dependent changes in cyclic nucleotide levels, the incubation buffer containing the stress hormone OA (40 µM OA; 10 nM free Ca2+ concentration) was employed (Fig. 5A, 5B). In the OA buffer, AC activity significantly increased at the resting phase at ZT 5 (n = 11) as well as during the maximum of mating activity at ZT 10 (n = 10) to about the same maximal values (Wilcoxon test, α = 0.05, p < 0.05). With OA, the medians of the cAMP concentrations increased from 46.5 pmol/mg to 235.5 pmol/mg at ZT 5 and from 172.2 pmol/mg to 234.3 pmol/mg at ZT 10. Accordingly, the OA-dependent increase in cAMP concentration at ZT 5 was significantly stronger than at ZT 10.
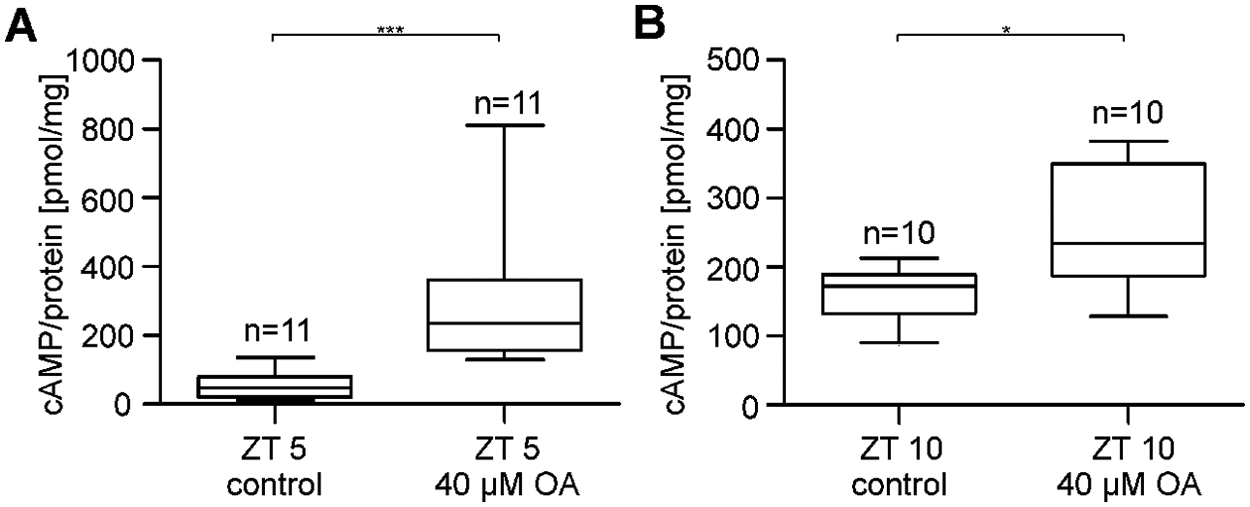
OA increased cAMP concentrations in antennal lysates of R. maderae at both rest and activity phase. (A, B) The OA-dependent increase of antennal cAMP concentrations is significant at ZT 5 and 10 (Wilcoxon test for paired samples, α = 0.05, p < 0.05). The incubation buffers contained either no (control) or 40 µM OA (n = 1: 20 female antennae).
Discussion
Previously, a puzzling phase difference was found between circadian rhythms in pheromone-dependent behavior and antennal pheromone sensitivity (Decker et al., 2007; Farine et al., 2007; Page and Koelling, 2003; Rymer et al., 2007; Saifullah and Page, 2009; Sreng, 1993). Thus, we hypothesized that sensory adaptation due to prolonged pheromone stimulation and/or stress-dependent sensitization might modulate antennal pheromone detection and override (mask) underlying phase-coupled circadian rhythms. Since in moths, cyclic nucleotide levels modulate olfactory sensitivity in a ZT-dependent manner (Flecke et al., 2006; Flecke et al., 2010; Villet, 1978), here, it was examined whether in antennae of the Madeira cockroach, cyclic nucleotide concentrations vary in a ZT-dependent manner and additionally depend on stress and adapting pheromone exposure. Indeed, for the first time, cyclic nucleotide concentrations in insect antennae were shown to differ ZT-dependently in phase with previously observed circadian rhythms of odor-dependent behavior under nonadapting conditions (Decker et al., 2007; Rymer et al., 2007). Furthermore, first evidence was provided that the stress hormone OA increased cAMP concentrations in insect antennae at all ZTs because OA elevated cAMP levels both at rest as well as at the peak time of mating activity. In contrast, exposure to excess male pheromones decreased cAMP levels and increased cGMP concentrations in a ZT-dependent manner. Thus, these findings suggest that the previously observed discrepancy between the phases of circadian rhythms in olfactory sensitivity and olfactory-dependent behavior might be due to masking of underlying rhythms caused by pheromone overstimulation in crowded cockroach colonies (Decker et al., 2007; Ha and Smith, 2009; Knadler and Page, 2009; Page and Koelling, 2003; Rymer et al., 2007; Saifullah and Page, 2009; Stengl, 2010) (Fig. 6).
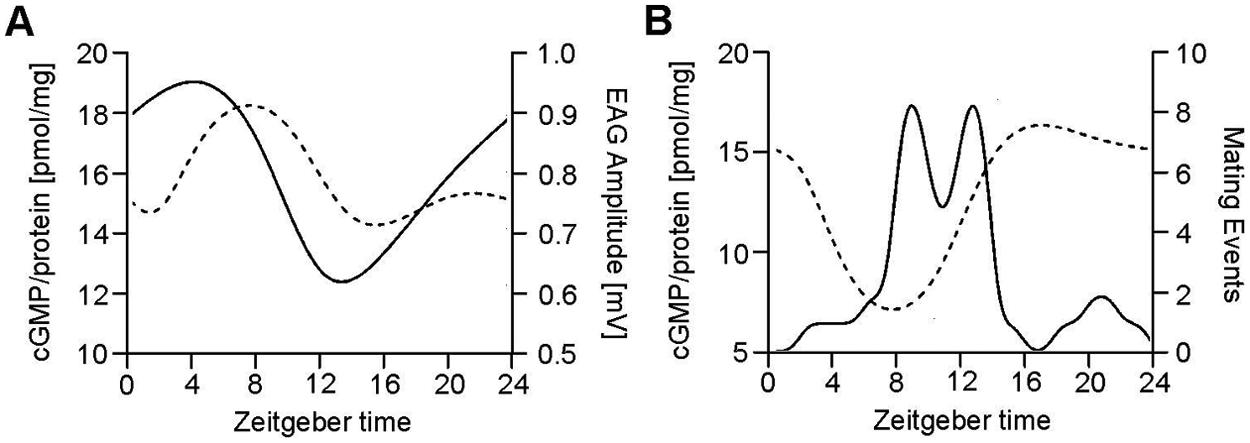
Correlation of minima in cGMP concentrations with a previously reported increase in olfactory sensitivity and pheromone-dependent mating activity. (A) Antennal cGMP data of female Madeira cockroaches collected under conditions of excess males presented as a dashed B-spline curve by calculating the medians at each time investigated (left) compared to single olfactory sensillum EAG amplitudes (right, solid line) (modified after Saifullah and Page, 2009). (B) Antennal cGMP data collected from female cockroaches at conditions of an equal ratio of males to females presented as a dashed B-spline curve by calculating the medians at each time point investigated (left) compared to cockroaches’ mating behavior (right, solid line) (modified after Rymer et al., 2007).
The Role of cAMP in Insect Olfactory Transduction
Olfactory transduction cascades in insects are still under lively debate. Most electrophysiological studies were performed on the signal transduction cascades underlying pheromone transduction in moths, while little is known about odor transduction in cockroaches (Stengl, 2010; Ha and Smith, 2009). In contrast to cockroaches (Sreng, 1993), female moths attract their mates over long distances with the release of a species-specific sex pheromone bouquet. The males detect the pheromones with specialized ORNs, which pair-wise innervate long trichoid sensilla on the males’ antennae (Sanes and Hildebrand, 1976). The pheromones bind to 7TM receptors, which appear to be inversely inserted into the dendritic membranes and which form heteromultimers with a highly conserved coreceptor termed Orco (Nakagawa and Vosshall, 2009; Vosshall and Hansson, 2011). There is no consensus yet whether pheromone transduction involves a sole ionotropic and/or G protein–coupled signal transduction cascade (Nakagawa and Vosshall, 2009; Stengl, 2010; Vosshall and Hansson, 2011). While in the fruit fly, it is assumed by some authors that odor application causes increases in cAMP concentrations (Deng et al., 2011; Wicher et al., 2008), in moths, there is evidence from multiple studies for a G protein–activated phospholipase Cβ cascade causing increases in IP3, DAG, and intracellular Ca2+, which follow in respective gating of second messenger–dependent ion channels (Stengl, 2010). Different signal transduction cascades allow ORNs to detect pheromone concentrations over at least 4 log-units via changes in intracellular Ca2+ and in cyclic nucleotide concentrations. This was confirmed via different electrophysiological studies showing that changes in cyclic nucleotide levels shift odor sensitivity in a ZT-dependent manner via gating of ion channels in ORNs (Dolzer et al., 2008; Flecke et al., 2006; Flecke et al., 2010; Krannich and Stengl, 2008).
The second-messenger levels in the ORNs are not only determined via pheromone receptors but also via hormone receptors such as OA receptors, which reflect the internal state of the moth (Stengl, 2010). Since the stress hormone OA expresses circadian rhythms in the hemolymph with maxima during the moths’ mating phase at the late night (Lehman, 1990; Linn and Roelofs, 1986; Stengl, 2010), it might be responsible for the observed circadian rhythms of olfactory sensitivity (Flecke and Stengl, 2009; Zhukovskaya and Kapitsky, 2006). The finding that OA actions were partly mimicked via cAMP perfusion in pheromone-sensitive sensilla supported this hypothesis (Flecke et al., 2010; Flecke and Stengl, 2009). In addition, the results of this study are consistent with this assumption because OA increased cAMP concentrations. However, since OA increased cAMP levels at all ZTs tested, it is likely that in addition to hemolymph-born OA, there must be an additional OA release mechanism in the cockroach antenna that acutely responds to acute stress stimuli. This stimulus-dependent OA release and resulting cAMP increases could then mask observed ZT-dependent rhythms in cAMP concentrations caused via OA concentration changes in the hemolymph (Stengl, 2010). Whether cAMP sensitizes pheromone transduction in the cockroach at the early day as it does in the moth and whether OA expresses circadian oscillations in the hemolymph of the Madeira cockroach still remain to be examined. In addition, it remains to be examined whether the activity of antennal ACs is Ca2+ dependent and under circadian regulation since adapting pheromone concentrations cause increases in Ca2+ (Stengl, 2010), which apparently affected cAMP levels differently at ZT 5 and 10.
The Role of cGMP in Insect Olfactory Transduction
In contrast to cAMP, perfusion of moth trichoid sensilla with cGMP caused adaptation of the pheromone response (Flecke et al., 2006). Furthermore, adapting odor concentrations increased cGMP levels in antennal homogenates of different insects (Boekhoff et al., 1993; Ziegelberger et al., 1990). These antennal cGMP increases took place in pheromone-dependent moth receptor neurons and supporting cells of trichoid sensilla as was shown in immunocytochemical studies combined with minute-long pheromone stimulation (Stengl et al., 2001). The current study found for the first time that cGMP concentrations change in a ZT-dependent manner in cockroach antennae. In addition, evidence was provided that cAMP and cGMP concentration changes are inversely correlated. However, the respective mechanisms are not known. Since cAMP and cGMP levels were affected in opposite ways via excess males in the cockroach colonies, this mechanism could be caused by excessive stimulation of pheromone receptors. Male cockroaches produce sex pheromones as well as contact pheromones, and thus, it is very likely that the surplus of males over females in the insect colonies resulted in overstimulation of females via adapting male pheromone concentrations. Therefore, it is likely that adapting pheromone stimuli mask underlying cGMP rhythms in the Madeira cockroach possibly via changes in intracellular free Ca2+ concentrations. Whether there is a circadian rhythm in cGMP concentrations in cockroach ORNs regulated via a molecular clockwork in the receptor neurons themselves, as suggested in other insects, remains to be studied (Merlin et al., 2007; Schuckel et al., 2007; Tanoue et al., 2004).
To summarize, this study provides the first evidence for ZT-dependent antagonistic rhythms of cyclic nucleotides modulating odor sensitivity in cockroach antennae to orchestrate odor-dependent behavior. Possibly, antagonistic rhythms in cAMP and cGMP reflect a general ancient signaling mechanism coupled to eukaryotic cilia (Johnson and Leroux, 2010). Furthermore, circadian changes in cyclic nucleotides were also found in the suprachiasmatic nucleus, the circadian clock of mammals (O’Neill and Reddy, 2012; Prosser and Gillette, 1991; Prosser et al., 1989). In our study, maximum antennal cAMP levels and thus maximum pheromone sensitivity are in phase with maximal pheromone-dependent mating activity. In addition, acute external stimuli such as adapting pheromone concentrations as well as the internal state of the animal, which is related via hormones, appear to be able to mask underlying phase-coupled rhythms of sense organ sensitivity and behavior. Future experiments will test whether changes in intracellular free Ca2+ concentrations inversely couple circadian rhythms in cAMP and cGMP concentrations in different insect antennae as a general scheme of homeostasis.
Footnotes
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
