Abstract
The endogenous circadian clock is entrained by external cues, mainly the light-dark cycle received by photopigments located in the retina. The authors investigated (1) the effect of aging on the synchronization of the rest-activity rhythm and (2) the physiological basis of light photoreception in the gray mouse lemur, a nocturnal Malagasy primate. Old individuals were tested at different irradiance levels under 3 different light wavelengths previously shown to trigger maximal response in young adults. Investigators analyzed animal activity and temperature waveforms and used 2 reference treatments, strong white light and continuous darkness. The investigation revealed manifold effects of aging picturing a general loss of responsiveness to light and age-related changes in activity and temperature. In addition, the investigation shows that photoentrainment is a continuous process. Short wavelengths (430 nm) are more efficient than longer wavelengths (470-540 nm) at low light levels at dawn and across all light levels at dusk. This suggests an implication of S-cones, differential at dawn and dusk. This results, surprising for several reasons, calls for further investigation. The study brings an interesting contribution to the understanding of the physiological processes underlying synchronization to light.
Keywords
Synchronization of the circadian clock—mainly by photoentrainment—is crucial for animal physiology to be optimized to cyclic variations of the environment. In vertebrates, photoentrainment primarily relies on reception of light cues by eye photopigments—retinal ganglion cells, cones, and rods—which interplay in complex and poorly known mechanisms (Altimus et al., 2010; Doyle and Menaker, 2007; Lall et al., 2010). Aging has been suggested to impair behavioral and physiological functioning, alter synchronization, and aggravate age-related health problems in humans (review in Turner and Mainster, 2008). Yet, the mechanisms whereby aging affects synchronization largely remain to be studied.
We set out to investigate the retinal bases of photoentrainment and the potential impacts of aging on synchronization in a prosimian, the gray mouse lemur (
Here, we exposed old adults to different combinations of light intensity and wavelength and compared locomotor and temperature responses to reference treatments. We analyzed these patterns to decipher refined mechanisms of light reception. Given that we used the same protocol as in young adults (Perret et al., 2010), we could test aging effects. We anticipated aging to cause a loss of responsiveness to light (e.g., in humans; Turner and Mainster, 2008) and to alter behavioral and physiological parameters (Aujard et al., 2006). Although aging has been studied from many perspectives in the mouse lemur (Languille et al., 2011), this study is the first to adopt a holistic behavioral point of view anticipated to provide a comprehensive view of aging impact and to explore refined processes of light photoentrainment.
Materials and Methods
Animal selection
We tested 13 healthy old females—5 to 7 years old—from the captive breeding colony of the UMR CNRS/MNHN in Brunoy (for description of the colony and housing conditions, see Perret et al., 2010). We performed ocular examinations before and after pupil dilatation using slit lamp microscope and indirect ophthalmoscopy. Different ocular conditions were diagnosed: no visible ocular alterations (38%), bilateral sclerosis (31%), incipient lens opacity (31%). All alterations are commonly encountered in old mouse lemurs (Beltran et al., 2007). Although we anticipated that these alterations would have no effect on synchronization, we included ocular lesion (absence/presence) as a factor in analyses.
In mouse lemurs, short photoperiod (light phrase ≤12 h) rapidly induces activity reduction, pronounced fattening, daily torpor, and complete sexual rest (Perret and Aujard, 2001). By contrast, long photoperiod induces increased activity, reduced torpor, and sexual activation, which blurs activity and temperature waveforms, thus interfering with the estimation of the parameters characterizing animal response to light. We thus exposed animals to short LD 12:12 photoperiod (ensuring no interference with the parameters studied) 1 month before and during the whole experiment. Animals were housed in individual cages with a nestbox. They were placed in light-insulated ventilated chambers to control for light and temperature (for testing conditions, see Perret et al., 2010).
Experiment
We exposed animals to different light treatments and compared their locomotor activity and body temperature profiles to 2 reference treatments.
We recorded animal activity and temperature using telemetry. Animals were implanted with small transmitters that recorded data points every 10 min during the experiments (technical characteristics, implant process detailed in Perret et al., 2010). We analyzed profiles over the last 7-day period of each test using ClockLab software (Actimetrics, Evanston, IL).
We first exposed a focal animal to LD 12:12 white light reference treatment (WL) by using a strong fluorescent daylight tube (2.6 106 nmol·m-2·sec-1, corresponding to 15.7 1014 photons·cm-2·sec-1) to get full synchronization (described in Perret et al., 2010). The experiment consisted of exposing a focal individual to a sequence of decreasing light intensities for a specific wavelength. After exposure to the lowest intensity, the animal was submitted to continuous darkness LD 0:24 (DD) to record free-running pattern (described in Perret et al., 2010). All animals were synchronized under WL and in free-run under DD. To avoid any possible entrainment of circadian rhythms by external cues, food was distributed 3 times a week at random during the daily resting period.
Each test of specific intensity and wavelength lasted at least 10 days to ensure that animal response was homogeneous over a period of 7 days indispensable for data analysis. We tested the same range of intensities as in young adults: 320, 45, 13, and 6 nmol·m-2·sec-1 (values measured at the nestbox entrance), which corresponded to 53 1010, 7.5 1010, 2.16 1010, and 1 1010 photons·cm-2·sec-1 (Perret et al., 2010). We tested only the 3 peak wavelengths identified as most efficient at triggering synchronization, 431, 474, and 542 nm, using nonmonochromatic LED lights (spectra and LED characteristics in Perret et al., 2010).
We obtained data on 83 tests. Of the 13 animals, we tested 6 for the 3 wavelengths, 1 for 2 wavelengths, and 6 for 1 wavelength only. As in Perret et al. (2010), we used the rapid increase or decrease in activity and body temperature to delineate nocturnal activity onset and offset in all tests performed, and we extracted the following parameters:
τ: mean period, length of time between consecutive activity onsets, in minutes
ψ
ψ
We analyzed individual patterns by normalizing data to the period observed in free-running periods.
Statistics
We tested potential aging effects by analyzing the parameters extracted from individual profiles. We pooled data obtained for old adults (13 animals) and young adults (34 animals, data from Perret et al., 2010) for the 3 intermediate wavelengths. Concerning young adults, we retained males and females as no sex effect had been observed on synchronization (Perret et al., 2010). We used generalized linear mixed models suited to repeated observations on the same individuals and sample sizes varying across treatments (Bolker et al., 2009) and selected the best model using a maximum likelihood approach and minimization of AIC. Factor significance was tested using Wald
We then analyzed the variations of synchronization. We considered that a synchronization pattern comprised 2 synchronization events, each in response to a light transition (light-dark and dark-light) and each scored by 0/1 for absence/presence of synchr onization. Three patterns had been observed in young adults: full synchronization (11), partial synchronization with advanced phasing to light off (01), no synchronization (00) (Perret et al., 2010). We considered not the pattern but the event as the variable to explain. As fixed effects, we tested the effects of light transition, age (young/old), ocular lesion, light intensity and dominant wavelength, and free-running period (factor affecting synchronization; e.g., in Duffy et al., 2001). We used the best model to compute the predicted synchronization expected under a light treatment. This value could be interpreted as the probability of synchronization for an animal or the proportion of animals synchronized within a population.
We analyzed the variations of the period in nonsynchronized animals (including DD light treat-ment) to question the continuity of their response between continuous darkness and light.
We analyzed synchronization to tested light treatments in old animals only. The highly balanced design allowed us to test the relative wavelength efficiency at different light intensities independently and try to decipher the retinal bases of photoentrainment.
We tested potential aging effects on animal behavior by analyzing the variations of activity and temperature parameters in reference treatments.
Results
Aging Effects on Synchronization
Considering all animals, the variations of synchronization were best explained by age, light intensity, light transition, and free-running period (Table 1, first column). All 3 intermediate wavelengths were similarly efficient at triggering synchronization in adults and ocular lesion did not influence synchr onization. Whatever their age, animals were better synchronized with increasing light intensity and for dark-light than light-dark transition (Intensity and Light transition effects, respectively).
Variations of synchronization in all animals, in old animals only, and variations of period in nonsynchronized animals
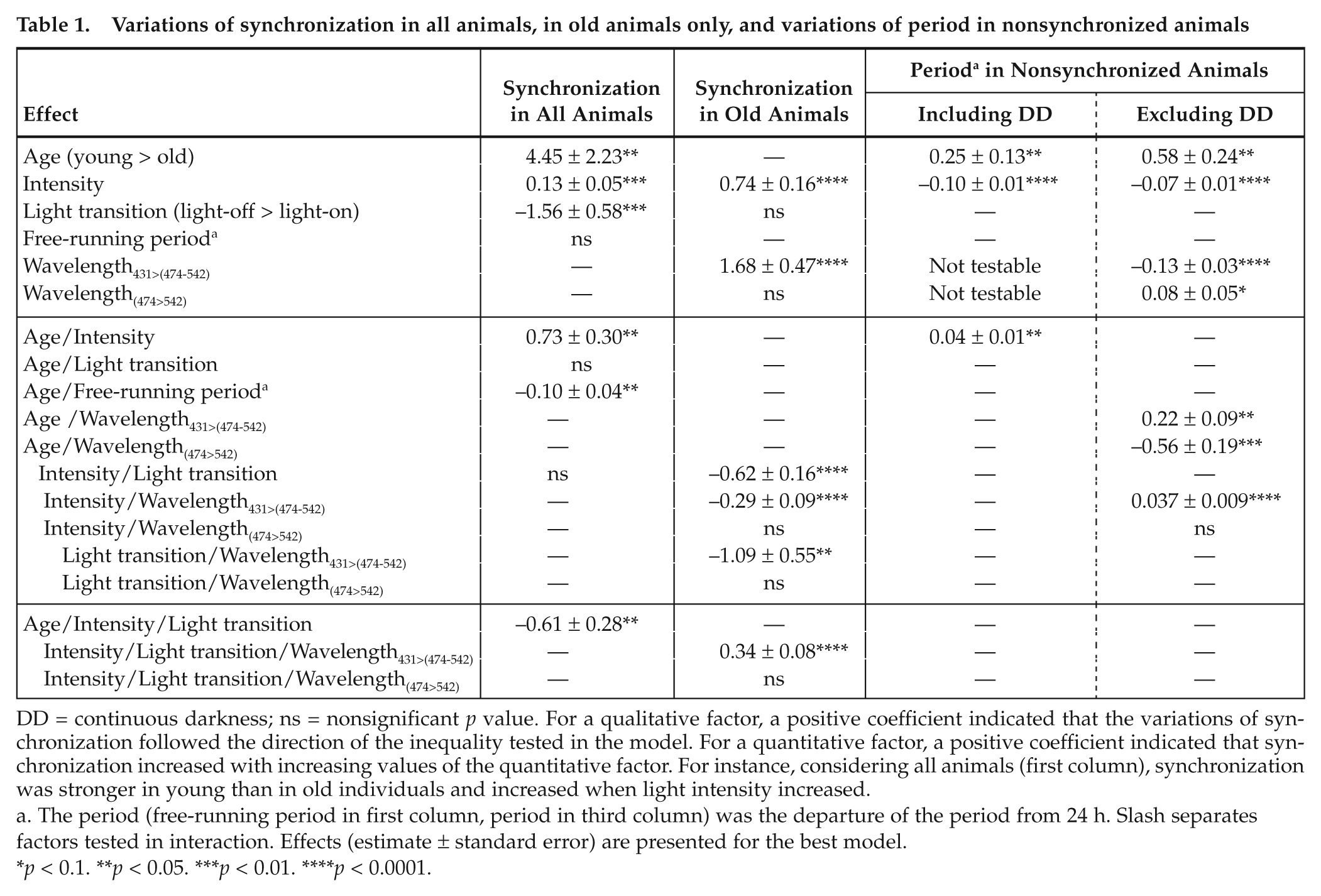
DD = continuous darkness; ns = nonsignificant
The period (free-running period in first column, period in third column) was the departure of the period from 24 h. Slash separates factors tested in interaction. Effects (estimate ± standard error) are presented for the best model.
Aging effects were manifold and pictured a general loss of responsiveness in old animals (Age effect). The positive effect of light intensity on synchronization was less pronounced in old adults (Age × Intensity effect). As light intensity increased, the difference in synchronization between dark-light and light-dark became weaker in young but remained strong in old adults (Age × Intensity × Light transition effect, Figure 1). This revealed that at relatively high light intensities, old adults still showed partial synchronization with advanced phasing to light-off while young adults were fully synchronized. Finally, in young adults, animals with shorter free-running periods were less synchronized than animals with free-running periods closer to 24 h; this effect became weaker in old animals (Age × Free-running period effect).
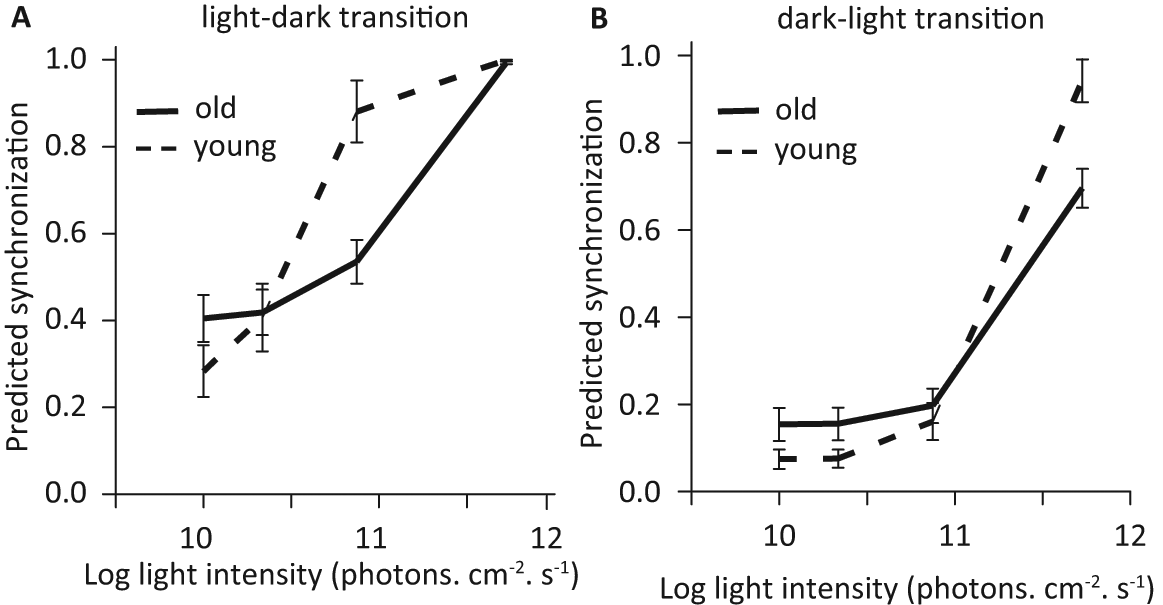
Predicted synchronization (mean and standard error) in relation to age (young, dashed line; old, plain line), light intensity, and light transition (A, light-dark; B, dark-light). Values predicted by the best model represent the proportion of synchronized animals in a population or the probability that an animal get synchronized.
Variation of Period in Nonsynchronized Animals
Including animals in DD, we found that the period increased with age, with increasing light intensity, and more strongly in old adults (Figure 2, Table 1, third column). Excluding DD conditions, we found similar effects and we could test the additional effect of wavelengths (Table 1, fourth column). Shorter wavelengths were more efficient at generating a longer period (Wavelength effect), but this effect faded with increasing light intensity (Intensity × Wavelength effect). The higher efficiency of shorter wavelengths was stronger in old than young adults (Age × Wavelength effects).
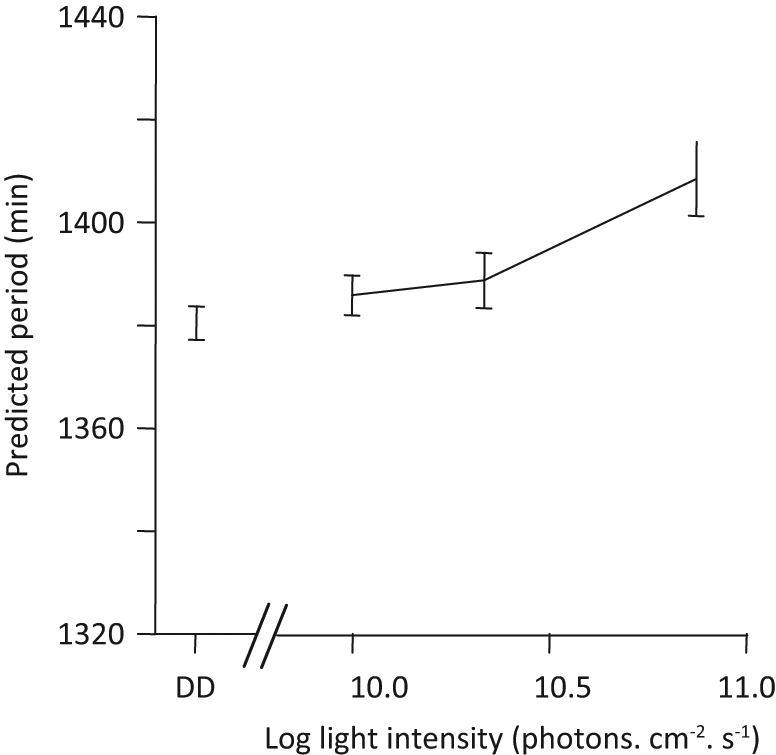
Predicted circadian period (mean and standard error) in relation to light intensity (in log units) in nonsynchronized animals. On this graph, age categories are pooled, and DD represents continuous darkness. Values are predicted by the best model (see methods for detail).
Bases of Photoentrainment
Considering only old animals, synchronization varied with the interactions between light intensity, dominant wavelength, and light transition but not with free-running period or ocular lesion (Table 1, second column). Again, synchronization increased with light intensity and was higher for dark-light than light-dark (Intensity and Light transition effects), which reflected the transitional partial synchronization pattern between no synchronization and full synchronization as light intensity increased.
More interestingly, old animals were better synchronized at 431 nm than at 474 or 542 nm. At dawn and for low light intensities, shorter wavelengths (431 nm) were more efficient than longer wavelengths (Light transition × Intensity × Wavelength effect). Longer wavelengths (474 nm and 542 nm) became more efficient than 431 nm when light intensity increased (Figure 3D). By contrast, at dusk, wavelength efficiency was comparable at all light intensity levels, with short wavelengths slightly more efficient (Figure 3C). A higher efficiency of 431 nm compared to longer wavelengths was compatible only with S-cone pattern response, not with that of any other photoreceptor (Figure 3A, 3B). By contrast, the higher efficiency of longer wavelengths (474 and 542 nm) did not coincide with the response pattern of S-cones.
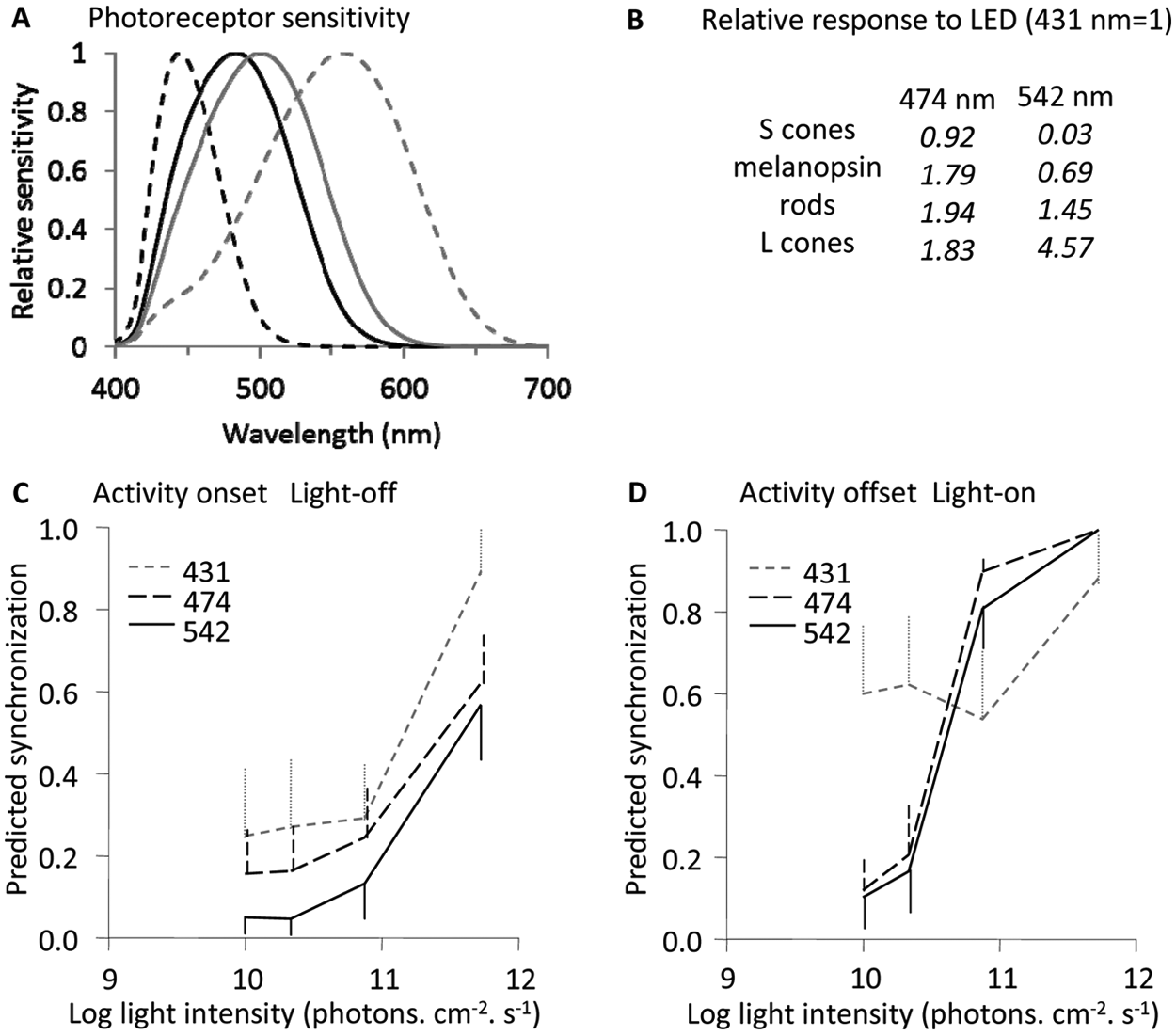
Variations of synchronization to light in relation to wavelength. (A) Relative sensitivity of photoreceptors in the mouse lemur. From left to right, peaks are for S-cones (dashed black line), melanopsin (plain black line), rod (plain gray line), and L-cones (dashed gray line). Curves were normalized for a maximal absorption of 1. (B) Photoreceptor relative responses to LED lights, with response to LED 431 nm taken as reference (response = 1). (C, D) Predicted synchronization (mean and standard error) in relation to light intensity and dominant wavelength (dotted, 431 nm; dashed, 474 nm; plain line, 542 nm) for light-dark transition (C) and dark-light transition (D). Standard errors are represented only on 1 side of the curves, either up or down for clarity reasons.
Aging Effects on Locomotor Activity and Body Temperature
Independently of light treatment, old animals showed better phasing (lower ψonset and ψoffset values) and shorter resting phase; they were more active during the first and last hours of night and reached a lower body temperature during their torpor than young animals (Table 2). With similar values in WL as young animals, old animals maintained a longer period and lower ψ values under free-running conditions. Similarly active in DD as young animals, old animals were more active in WL (Table 2). The higher proportional activity before light-on in WL than in DD shown in all animals was more pronounced in young animals. Old animals showed a lower body temperature in DD than WL while it was the contrary for young animals (Table 2).
Variations of locomotor and temperature parameters
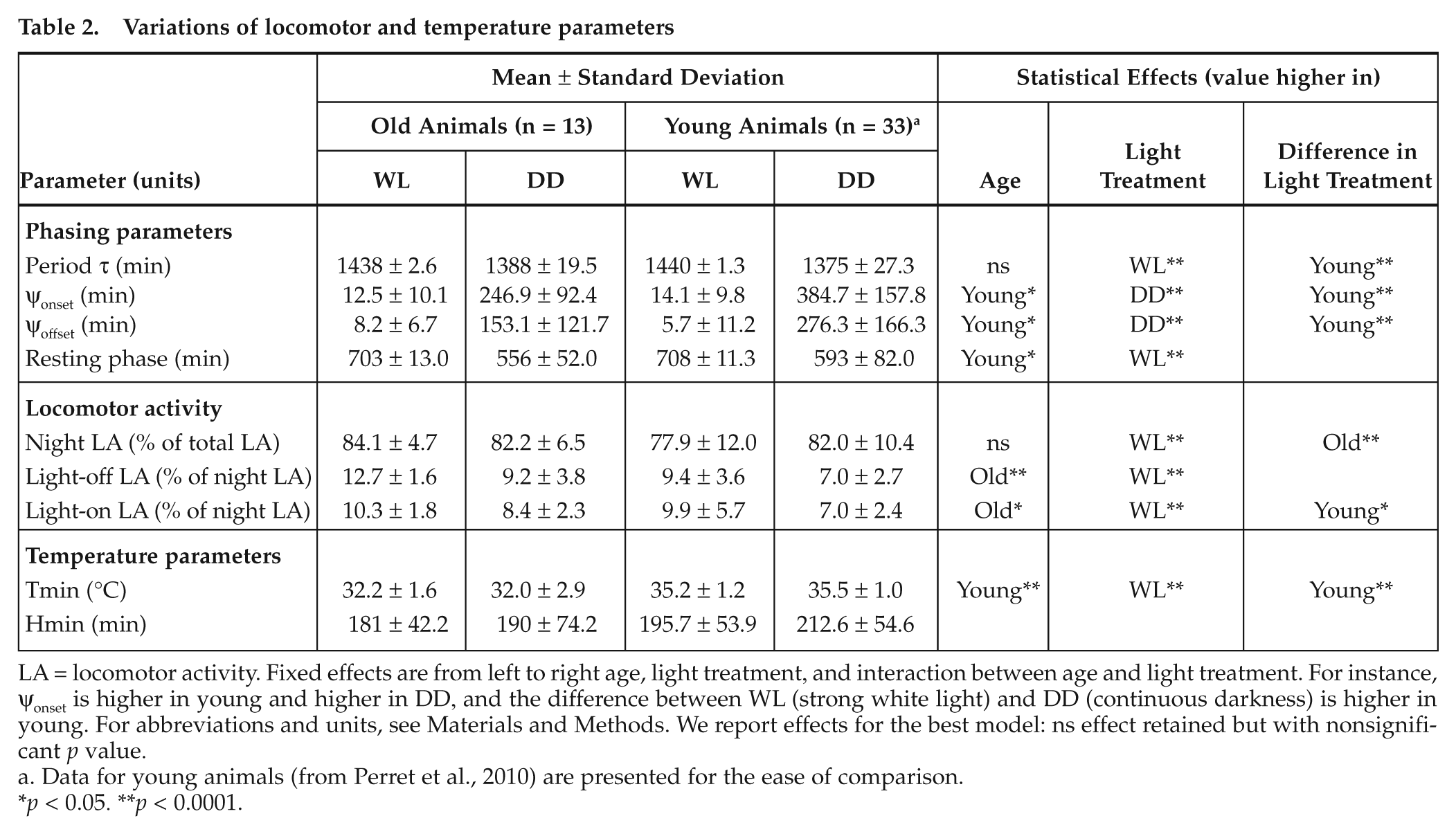
LA = locomotor activity. Fixed effects are from left to right age, light treatment, and interaction between age and light treatment. For instance, ψonset is higher in young and higher in DD, and the difference between WL (strong white light) and DD (continuous darkness) is higher in young. For abbreviations and units, see Materials and Methods. We report effects for the best model: ns effect retained but with nonsignificant
Data for young animals (from Perret et al., 2010) are presented for the ease of comparison.
Discussion
Aging Affects Synchronization
As in young adults (Perret et al., 2010), old adults show a synchronization increase with light intensity, stronger for dark-light than light-dark transition. Yet, we observe manifold aging effects of synchronization which depict a general loss of sensitivity to light. Old lemurs exhibit a 0.3 to 0.5 log unit loss in sensitivity at dusk and dawn, respectively. In humans, elderly people require 10 times (1 log unit) brighter illumination to enjoy the same photoentrainment as young people (Turner and Mainster 2008). Numerous causes have been suggested and tested: reduction in pupil size, increase of short wavelengths lens filtering and subsequent loss of efficiency of short wavelengths (Revell and Skene 2010), photoreceptors lost or misshapen (rods more affected than cones), slowing in rhodopsin regeneration, and dysfunctions in neural pathways (reviewed in Jackson and Owsley, 2000; Werner, 2005). Many mechanisms (if not all) may apply to lemurs, and their relative relevance remains to be studied.
Age-related changes are visible from activity and temperature profiles. Old animals show an increase of activity during the resting phase and a loss of responsiveness to light, as observed in rodents or humans (Dijk and Lockley, 2002; Duffy et al., 1998; Weinert and Weinert, 2003). Old animals have similar temporal patterns in body temperature as young adults, except that they reach lower body temperature, as previously found (review in Languille et al., 2011).
Individuals with shorter free-running period require more intensity to be synchronized. This directly results from the oscillatory properties of the endogenous clock, which causes entrainment to occur earlier for shorter circadian periods (Pittendrigh and Daan, 1976; Roenneberg et al., 2003). Such interindividual differences would explain morningness and eveningness in humans (Duffy et al., 1998) and maybe the decreased precision in the onset of activity phase or the difficulty of resynchronization in old individuals (Weinert and Weinert, 2003; Zee et al., 1992). The dependency of synchronization on the free-running period is more pronounced in young than in old adults, probably masked by the overall loss of responsiveness in old adults.
Physiological Basis of Light Photoreception
Our study brings out 2 substantial findings. First, light photoreception is likely a continuous process rather than a switch-off (no synchronization/synchronization to a light transition). At light levels ineffective at entraining the circadian pacemaker, animals show a progressive change in their period and thus a continuous response to light cues. Shorter wavelengths are more efficient at producing these changes. Responses likely depend on the number and wavelength of the photons received. Similar results have already been obtained in Syrian hamsters exposed to continuous low intensity (e.g. Boulos et al., 2002; Evans et al., 2007).
Second, wavelength relative efficiency at triggering synchronization depends on light intensity but also on light transition. For dark-light transition (dark-adapted conditions), maximal efficiency shifts from short (431 nm) to longer wavelengths (470-540 nm) as light intensity increases, while for light-dark transition (light-adapted conditions), short wave lengths are always the most efficient across the entire range of intensities tested.
A higher efficiency of shorter wavelengths relative to longer wavelengths is only compatible with a contribution of S-cones, the only photoreceptor yielding a lower response at 474 and 542 nm compared to 431 nm. This argument stems on the comparison of photoreceptor response through 2 methods: (1) we considered the absorption function of the photoreceptors, and (2) we computed the photons captured by photoreceptors when viewing the LEDs tested (spectra in Perret et al., 2010). Both methods yielded similar results as LEDs, although nonmonochromatic, and had a distinctive dominant wavelength.
This finding is weakly supported by the general knowledge on photoreceptors. First, rods (most studied for synchronization) play a major role in synchronization to light across a large range of intensities (Altimus et al., 2010; Lall et al., 2010). Long-wavelength sensitive cones are also involved but less heavily (Dkhissi-Benyahya et al., 2007; Lall et al., 2010). A role of S-cones, although suggested (e.g. Altimus et al., 2010; Dacey et al., 2005; Lall et al., 2010; Provencio and Foster, 1995), remains to be elucidated (Lall et al., 2010). Second, S-cones are rare in mouse lemur retina (less than 0.2% of the total cone population; Dkhissi-Benyahya et al., 2001), which likely hinders their potential role in synchronization. Although dusk and dawn light undergoes more dramatic changes in blue and red than other wavelengths (Endler 1993), which may enhance the interest of having a short-wavelength based synchronization mechanism, it is difficult to see what role, if any, S-cones may have and how this would work. Indeed, cones are less responsive to light than rods. If any, cone contribution should be restricted to light-adapted conditions, as found for L-cones (Lall et al., 2010), but it should not extend to dark-adapted conditions, as we reveal here. Fourth, aging in humans, but also in lemurs, causes a short wavelength lens filtering, which decreases the efficiency of such wavelength range (references in Turner et al., 2010; Werner, 2005). Hence, short wavelengths (431 nm) should be less efficient and their relative contribution should decrease with aging. Further work is needed to ascertain or infirm our findings.
Finally, our results support the idea that E and M oscillators are served by different photoreceptors. This idea has already been suggested for bats, for which photoentrainment may be mediated by S-cones at dusk and M-cones at dawn (Joshi and Vanlalnghaka, 2005). Identification of the mechanisms at stake requires further investigation: experiments on young adults with greater sample size and fully repeated design, and retinal examination of old adults to track any age-related changes in photoreceptor and RGC populations.
Our results concerning aging effects may have limited ecological implications. First, due to a short median survival in the wild (Lutermann et al., 2006), only a reduced proportion of animals may be concerned with aging problems. Second, this loss of responsiveness causes old adults to require 2.5-30 lux for synchronization, values similar to what other Malagasy forest primates require (Erkert, 2008). Forest light decreases from 50 to 1 lux in less than 15 minutes at dusk lux (Pariente, 1980), which largely encompasses old adults’ physiological limitations. The interest of this study rests more on the description of the primary steps of photoentrainment than on potential ecological impacts of variations in photoentrainment ability. Studying age-related dysfunctions helps to understand synchronization itself, as it helps to decipher a large range of physiological processes, such as endocrine systems, thermore gulation, sensorial, cerebral, and cognitive functions (review in Languille et al., 2011).
Footnotes
Acknowledgements
We are grateful to Sabine Chahory (Maison-Alfort National Veterinary School, Ophthalmology Unit) for ocular examinations and to Roger Botalla for light setup. This research was supported by ANR BLAN-0132-01. All experiments were carried out in accordance with the European Communities Council Directive (86/609/EEC).
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
