Abstract
Mating success depends on many factors, but first of all, a male and a female need to meet at the same place and time. The circadian clock is an endogenous system regulating activity and sex-related behaviors in animals. We studied bumble bees (Bombus terrestris) in which the influence of circadian rhythms on sexual behavior has been little explored. We characterized circadian rhythms in adult emergence and locomotor activity under different illumination regimes for males and gynes (unmated queens). We developed a method to monitor adult emergence from the pupal cocoon and found no circadian rhythms in this behavior for either males or gynes. These results are not consistent with the hypothesis that the circadian clock regulates emergence from the pupa in this species. Consistent with this premise, we found that both gynes and males do not show circadian rhythms in locomotor activity during the first 3 days after pupal emergence, but shortly after developed robust circadian rhythms that are readily shifted by a phase delay in illumination regime. We conclude that the bumble bees do not need strong rhythms in adult emergence and during early adult life in their protected and regulated nest environment, but do need strong activity rhythms for timing flights and mating-related behaviors. Next, we tested the hypothesis that the locomotor activity of males and gynes have a similar phase, which may improve mating success. We found that both males and gynes have strong endogenous circadian rhythms that are entrained by the illumination regime, but males show rhythms at an earlier age, their rhythms are stronger, and their phase is slightly advanced relative to that of gynes. An earlier phase may be advantageous to males competing to mate a receptive gyne. Our results are consistent with the hypothesis that sex-related variations in circadian rhythms is shaped by sexual selection.
Circadian rhythms organize the behavior and physiology of organisms along with predicted 24-h environmental oscillations on our planet. These rhythms are generated by endogenous cell-autonomous molecular clockwork that is entrained by ecologically relevant temporal cues such as changes in light intensity (reviewed by Saunders, 2002; Ashton et al., 2022), temperature (reviewed by Tomioka and Matsumoto, 2010), food availability (reviewed by Boulos and Terman, 1980; la Fleur et al., 2024), and the social environment (reviewed by Eban-Rothschild and Bloch, 2012; Siehler et al., 2021). The core circadian system influences physiology and behavior by means of molecular and neuronal output pathways (reviewed by Tomioka et al., 2012). Insects have provided important model systems for research on the molecular bases and ecological contexts of circadian rhythms. Studies with insects have explored how circadian clocks influence functionally significant behaviors such as emergence from the pupa (Pittendrigh, 1954; Saunders, 2002), oviposition (Howlader and Sharma, 2006), mating (Sakai and Ishida, 2001; Wang et al., 2021), locomotor activity (Petersen et al., 1988), temperature preference (Moore and Rankin, 1993), and social organization (Fuchikawa et al., 2016).
Given that courtship, mating behavior, or both, show circadian rhythms in many species, it is reasonable to assume that sexual selection may influenced circadian phenotypes. However, the role of timing as a sexually selected trait has received relatively little attention. Successful courtship and mating require receptive males and females to meet in the same place and time. Precise timing of mating activities is also important for minimizing the mate-searching interval which is costly in terms of time, energy, and increased predation risk (reviewed by Hughes et al., 2012). Thus, sexually mature and receptive adults are predicted to have a functional and properly entrained circadian system influencing sexual behavior or courtship. Indeed, there are sex differences in daily rhythms of mammals, including humans, many of which are regulated by gonadal hormones in a sex-dependent manner (i.e. only one sex responds; reviewed by Joye and Evans, 2022). There is also evidence for daily rhythms in sexual behaviors and receptivity in insects such as moths and flies (reviewed by Sakai and Ishida, 2001; Groot, 2014). Some studies further established causal links between circadian clocks and the release of sex pheromones for mating time and courtship behavior in Drosophila (Kyriacou and Hall, 1980, Sakai and Ishida, 2001; Fujii et al., 2007) and mosquitos (Wang et al., 2021). The premise that the timing of courtship is adaptive is supported by studies with Great Tits in which males with earlier onset of activity and morning singing increased fitness by having more extra-pair mating compared with later singing males (reviewed by Hau et al., 2017). Similarly, in European ground squirrels, males show earlier activity onset compared to females. Being early may be advantageous in male–male competition for territories and receptive females (Everts et al., 2004).
In social insects such as honey bees, bumble bees, and ants, circadian processes are commonly influenced by social interactions (reviewed by Eban-Rothschild and Bloch, 2012). For example, gynes show strong circadian rhythms that are necessary for timing their mating flights, but mated queens are active around the clock with no diurnal rhythms (Free et al., 1992; Eban-Rothschild et al., 2011; Shpigler et al., 2022). There is also evidence that social synchronization of activity rhythms is particularly strong in social honey bees (Fuchikawa et al., 2016; Beer et al., 2016; Siehler et al., 2021). Many social insect colonies dwell in nests that are protected from direct light and other environmental fluctuations, which may reduce the need to precisely anticipate change in ambient conditions. Consistent with this notion, newly emerged honey bee and bumble bee workers are active around the clock with no overt circadian rhythms, but after a few days develop clear circadian rhythms in locomotor activity (Spangler, 1972; Moore et al., 1998; Yerushalmi et al., 2006; Meshi and Bloch, 2007; Beer and Helfrich-Förster, 2020). This pattern contrasts with solitary bees such as Megachile Rotundata and Osmia bicornis that typically emerge from the pupa with circadian rhythms that help them synchronize their physiology and behavior with the ambient conditions (Beer et al., 2019).
To start testing whether there is sexual dimorphism in circadian rhythms in bumble bees, we focused on two of the best-studied rhythms in insects, locomotor activity and the time of pupal emergence. Using Bombus terrestris, the best-studied bumble bee, we addressed the following hypotheses: (1) Pupal emergence is not gated by the circadian clock in bumble bee sexuals that emerge into an environmentally regulated and protected nest environment. (2) Gynes and males develop circadian rhythms in locomotor activity after few days post eclosion. (3) Gynes and males show strong and synchronized activity rhythms under different illumination regimes. (4) There are differences between circadian rhythms of males and gynes. Our results are consistent with these four hypotheses.
Materials and Methods
Bees
Bombus terrestris colonies, consisting of a queen, 10 to 15 workers, and brood at various stages of development, were purchased from BioBee Biological Systems Ltd. Kibbutz Sde Eliyahu, Israel. Each colony was housed in a wooden nest box (21 × 21 × 12 cm) with a transparent Plexiglas front well and cover. All the colonies were kept in the same environmental rearing chamber (29°C ± 1°C; 55% ± 10% RH) in constant darkness at the Bee Research Facility at the Edmond J. Safra Campus of the Hebrew University of Jerusalem, Givat Ram, Jerusalem. The colonies were fed ad libitum with commercial sugar syrup and honey bee collected pollen (purchased from BioBee Biological Systems Ltd). We fed and collected bees under dim red light (Energizer headlights with red filters) that bumble bees do not see well (Peitsch et al., 1992).
We collected the focal bees for Experiments 1 and 3, which were conducted successively, from the same four source (“donor”) colonies. Two other colonies (at around 5 weeks after first worker emergence) were used as donor colonies for Experiment 2. These colonies were kept under constant darkness until they contained male and gyne pupae. We measured the length of the marginal cell of the right front wing as an index for body size (Yerushalmi et al., 2006). We used these measurements to differentiate gynes from worker bees.
Experiment 1. The Temporal Organization of Adult Emergence From the Pupa
We conducted four trials (trials 1–4, 08/2020, 11/2020, 12/2020, and 01/2021, respectively) in which we monitored the time of adult emergence for males and gynes. The experimental outline is shown in Supplementary Fig. S1A. We kept four donor colonies at the age of about 7 to 8 weeks after the date of the first worker emergence in an environmental chamber (29°C ± 1°C; 55% ± 10% RH). Colonies at this stage typically cease producing workers and have male and gyne pupae. When males and gynes had started to emerge in these colonies, we changed the illumination regime to 12 h light:12 h dark (LD) in order to entrain the colonies to a similar phase. The lights (IP65 led flood lights, 50 W, white, ~700 lux) were placed in the room such that all colonies received similar illumination intensity (determined using HOBO data loggers placed in different parts of the room). After a week of entrainment (on day 8), we collected male and gyne focal pupae. We used forceps and gloves to avoid touching the pupae with our hands. Gyne pupae are larger than male and worker pupae, allowing us to visually differentiate them. Male pupae are similar in size to that of large workers, but their size is typically more uniform. Each pupa was placed individually in a small Petri dish (35 mm diameter), besides the first trial in which we placed 6 to 8 pupae (male and gyne separately) together in larger Petri dishes (90 mm diameter). The walls of the gyne cages were elevated to allow sufficient space for their emergence and movement (Suppl. Fig. S1B). We entered the monitoring chamber only for refilling the humidifiers with fresh water once every 2 days at randomly selected hours. This procedure was important for minimizing the risk of entrainment by entering the room or our activity.
We used time-lapse video recording to monitor the cages with the pupae using four IP (Network) Dome 2.8 mm, HIKVISION cameras (Suppl. Fig. S1C). The cameras were set for recording an image once every 10 min. We downloaded the time-lapse video records using the Ethernet settings of the camera system at the end of the monitoring session. Each video footage included a specific timestamp allowing us to identify the date and time in which it was taken (Suppl. Fig. S1D). This system enabled us to monitor up to 100 pupae simultaneously. The emergence data for date and time was analyzed for the period spanning over the first 4 days starting at the emergence of the first bee.
Experiment 2. Development of Circadian Rhythms in Locomotor Activity
We kept two colonies in an environmental chamber (29°C ± 1°C; 55% ± 10% RH) under constant darkness. We started the experiment when the colonies were around 5 weeks post first worker emergence. We entered the chamber once every 2 to 3 days with red-filtered headlights for feeding and refilling the humidifiers. We observed these colonies until we detected male or gyne pupae which we then collected, placed inside a lightproof dark box, and transferred to the locomotor activity monitoring chamber. The monitoring chamber was illuminated with constant dim red light (Edison Federal EFEF 1AE1 Far [Cherry] Red LED; maximum and minimum wavelengths were 750 and 730, respectively) for camera recording during the monitoring period. We placed each pupa individually in a monitoring cage made of a modified Petri dish (diameter of 90 mm) and provisioned all cages with ad libitum sugar syrup and chickpea-sized pollen-paste balls. We monitored the newly emerging bees for about 20 days after the day of pupal emergence.
Experiment 3. The Influence of Illumination Regime on Circadian Rhythms in Locomotor Activity
We kept four large colonies, at about 7 to 8 weeks after the emergence of the first worker and containing approximately 100 workers each, in an environmental chamber (29°C ± 1°C; 55% ± 10% RH) under constant darkness for a week. We then switched the illumination regime to 12 hrs light:12 hrs dark (LD, light on from 08:00 to 20:00) for 8 days (see experimental outline in Suppl. Fig. S2). During this period, we entered the climate chamber once every 2 to 3 days for feeding and refilling the humidifiers at randomly selected hours (see Exp. 2). To reach the desired sample size, we collected newly emerged male and gyne bees over three consecutive days: the third, fourth, and fifth days of the entrainment session, such that the bees were entrained individually for 5, 4, and 3 days, respectively (Suppl. Fig. S2). We placed each male and gyne individually in a modified Petri dish (same cages as in Exp.2) and housed all the cages in the locomotor activity monitoring chamber.
We monitored locomotor activity under a light–dark illumination regime (LD, 12 h:12 h; light on from 08:00 to 20:00) during days 2 to 6. We then delayed the light phase by 4 h (“Shifted-LD,” 12 h:12 h; light on from 12:00 to 24:00) during days 6 to 10. At 10:00 of the next morning, during which the room was dark, we turned off the lights and kept the bees under constant darkness (DD) until day 14. We turned the lights on again at 10:00 on the next day and kept the bees under constant light (LL) for the last 4 days (days 15–18) of the experiment. We repeated this experiment twice, each repetition with bees from different source colonies.
Monitoring Locomotor Activity
Locomotor activity was measured as the number of pixels between two successive frames on the camera field of view. We used the ClockLab data acquisition system (Actimetrics Inc., Evanston, IL, United States) to record bee location automatically at a frequency of 1 Hz. The data acquisition system includes four cameras (Sentech STC-MB33USB mini-USB video cameras with Computer TZ32910CS-IR lenses) and an image acquisition board (USB-6501 interface). Each camera covered a single tray on which we placed 30 monitoring cages. We could monitor up to 116 bees in each trial with the remaining four cages, one on each tray, left vacant and used as a control recording background noise.
Analyses of Circadian Rhythms
We used the ClockLab circadian analyses software (Actimetrics, USA) to analyze circadian rhythms in all experiments. We generated double-plotted actograms to visually assess the phase and strength of circadian rhythms. We used periodogram analysis to quantitatively assess the strength of circadian rhythms. The index for rhythm strength was the “Power” which is calculated as the height of the periodogram peak above the p = 0.01 significance threshold for a period at the circadian range.
In Experiment 1, we first converted the emergence data into a text file with the count of emergence events per hour in a format that fits the ClockLab software. We used this approach also for the emergence data in Experiment 2 (Suppl. Fig. S3). We generated actograms with 60-min bins for visual inspection and performed χ2 periodogram analyses.
In Experiment 2, we analyzed sliding 4-day windows starting at the day of emergence. We performed χ2 periodogram analyses with 10-min activity bins and a free-running period between 20 and 28 h as described by Yerushalmi et al. (2006). If the power value was below the p = 0.01 threshold line, it was assigned a zero value. The onset of rhythmicity was defined as the first day of the first 4-day window in which the Power value was statistically significant.
In Experiment 3, χ2 periodogram analysis was performed as in Experiment 2, but separately for each illumination regime: Days 2 to 6 in LD, days 6 to 10 in shifted-LD, days 10 to 14 in DD (dark: dark), and days 14 to 18 in LL (light:light). We used double-plot actograms to assess the time of onset, offset, and acrophase of the activity phase for each day. We used the time of the morning onset as an index for the phase because it was typically clearer and easier to calculate compared to the offset. It should be noted, however, that analyses with the three phase indices were overall similar. The time of the onset or offset was defined as at least three consecutive 10-min bins each with activity reaching at least 10% of the maximum activity per bin during this day separated by a period of at least 5 h of reduced activity between the offset and the following onset (following Fuchikawa et al., 2016). We calculated phase shift as the phase difference between the morning onset before and after the shift in LD. Regression lines were fitted to activity onsets of the 4 days of each light regime, and we measured the difference between the extension of the regression line of the LD phase and the extension of the regression line of the shifted-LD phase on the first day of DD (Suppl. Fig. S4). We used the time difference between the morning onset of the last day of LD (day 4) and lights-on time (8:00) to calculate the phase angle, which describes the temporal relation between the internal circadian clock and external zeitgebers (time cues) (Suppl. Fig. S4).
Statistical Analyses
We used GraphPad Prism version 5.00, with a p-value of α < 0.05 for all the analyses. For each set of data, we first assessed whether the data were normally distributed using the Kolmogorov–Smirnov test. We used unpaired t-tests and analysis of variance (ANOVA) to compare two or more groups for data that meet the assumptions of parametric statistics, respectively, or the nonparametric Mann–Whitney or Kruskal–Wallis tests, respectively, for data not meeting the assumptions of parametric statistics.
Results
Experiment 1. The Temporal Organization of Adult Emergence From the Pupa
To explore the temporal organization of adult emergence, we placed male and gyne pupae that we collected from four donor colonies entrained under LD, and recorded the time of emergence under constant conditions. We recorded emergence time for 167 bees in four independent trials (Suppl. Table S1), that overall produced similar results. Both males and gynes emerge from the pupa around the 24-h cycle with no circadian or other apparent, rhythms (Figure 1).
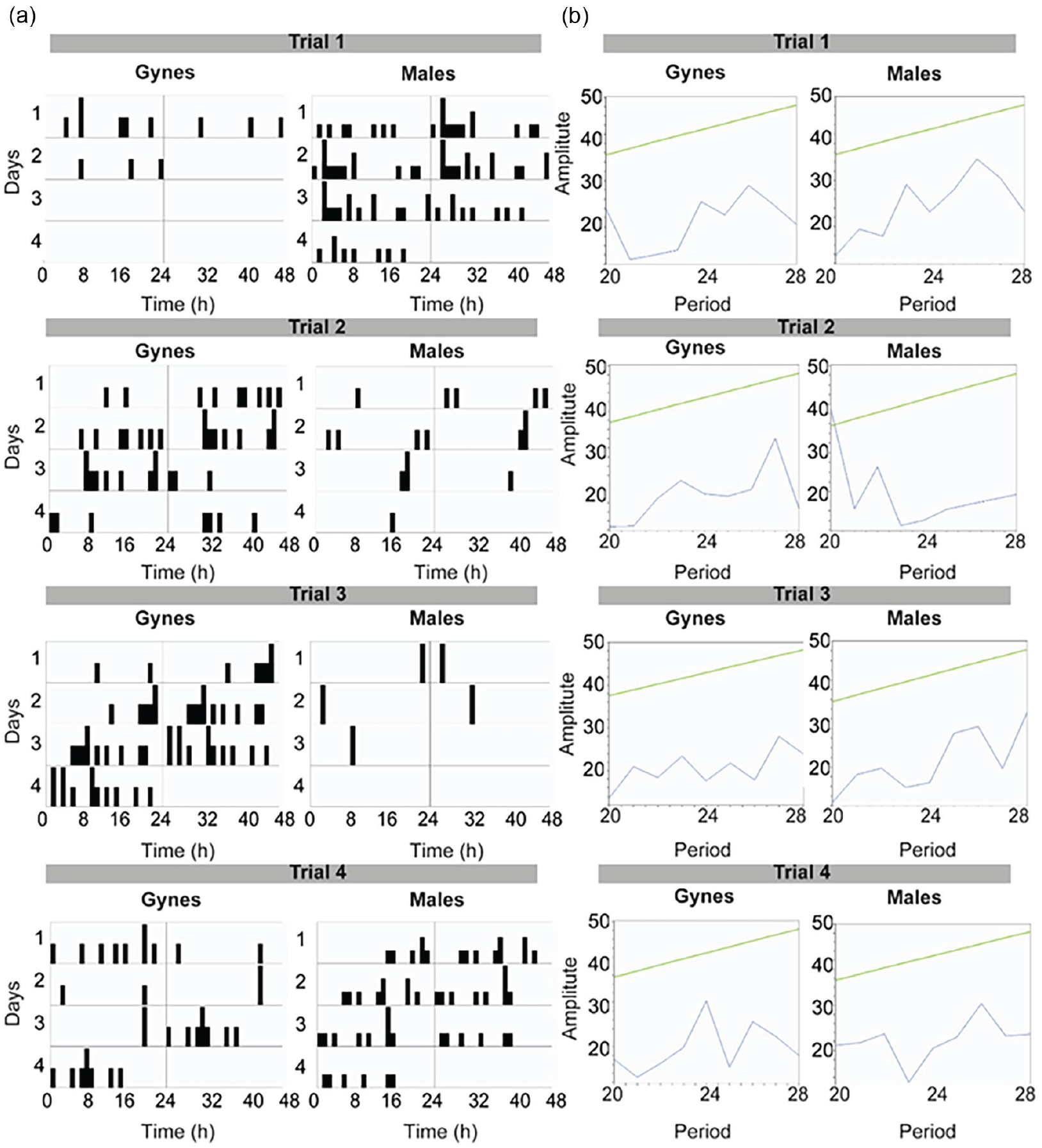
The temporal organization of pupal emergence for males and gynes. (a) Double-plot actograms showing emergence time over the first 4 days of pupal emergence in four independent trials. Each bar in the actogram corresponds to the number of bees emerging from pupae in a 60-min interval. (b) Chi-square periodogram analyses for the data presented in (a). The straight diagonal (green in the online version) line depicts the p = 0.01 significance threshold.
Experiment 2. Development of Circadian Rhythms in Locomotor Activity
We placed pupae individually in monitoring cages and recorded their locomotor activity under constant darkness starting from the day of pupal emergence. This procedure allowed us to estimate both the time of pupal emergence and the day in which each emerging adult bee showed circadian rhythms for the first time. Altogether, we placed 92 pupae in monitoring cages, 19 of which did not emerge during the monitoring session. Additional nine males and seven gynes died during the monitoring sessions and were not included in our analyses. Locomotor activity was analyzed for 35 males and 22 gynes monitored under constant darkness for 25 days. The day of emergence from the pupa varied among bees which is expected given that the pupae collected for this experiment were at different stages of development. Figure 2a shows representative double-plot actograms for a male and a gyne. As can be seen in these actograms, the bees do not show circadian rhythms in locomotor activity during the first days post-emergence, but later typically develop clear circadian rhythms. The free-running period varied among individuals (data not shown) and for many individuals also over time (Figure 2a). All males (100%) and 50% of the gynes showed significant circadian rhythms on Day 4, and all the bees (100%) showed significant circadian rhythm in locomotor activity on Day 5 (Figure 2b). The time (hours) elapsed between the time of pupal emergence until the morning onset on the first day in which the bee showed statistically significant circadian rhythm was shorter for males relative to gynes (Unpaired t-test, p < 0.001; Figure 2c). Given that we could estimate the time of pupal emergence for each bee (red arrow in Figure 2a), we also assessed the temporal organization of pupal emergence for bees in this experiment. These analyses showed that both males and gynes emerge from the pupa around the clock with no circadian rhythms (Suppl. Fig. S3), consistent with the findings of Experiment 1 in which emergence time was assessed using a different methodology.
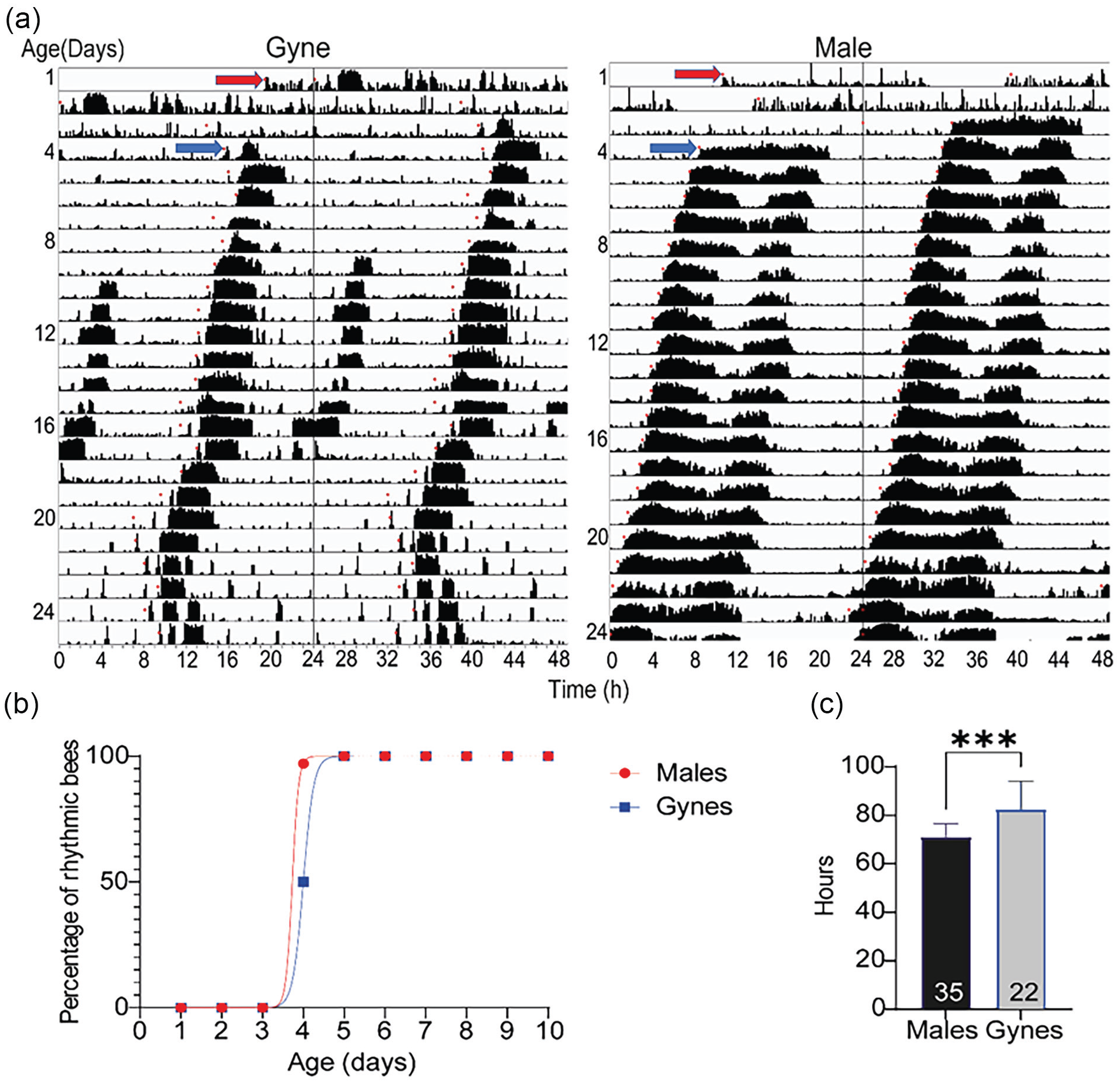
Development of circadian rhythms in locomotor activity for males and gynes. (a) Representative double-plot actograms showing circadian rhythms in locomotor activity from the day of emergence for a gyne and a male. The height of the bars within each day corresponds to the amount of locomotor activity per 10 min. The first arrow (red filling in the online version) points to the estimated time of emergence from the pupal cocoon and the second arrow (blue filling in the online version) points to the time in which each bee showed clear circadian rhythms for the first time.
Experiment 3. The Influence of Illumination Regime on Circadian Rhythms in Locomotor Activity
In order to explore the influence of lighting regime on circadian rhythms, we collected newly emerged male and gyne bees, placed each of them individually in a monitoring cage, and recorded their locomotor activity under four different illumination regimes (LD, shifted LD, DD, and LL). We performed two independent trials and altogether analyzed locomotor activity for 173 bees, each monitored individually. Most of the males (98%, 45 out of 46) in Trial 1, and all the males (100%, 11 out of 11) in Trial 2 showed robust circadian rhythms in constant light (LL). The proportion of individuals showing circadian rhythms in LL was slightly lower for gynes (Trial 1: 60%, 21 out of 35; Trial 2: 89%, 16 out of 18; Figure 3a). Males showed stronger circadian rhythms compared to gynes under all tested light regimes (unpaired t-test;
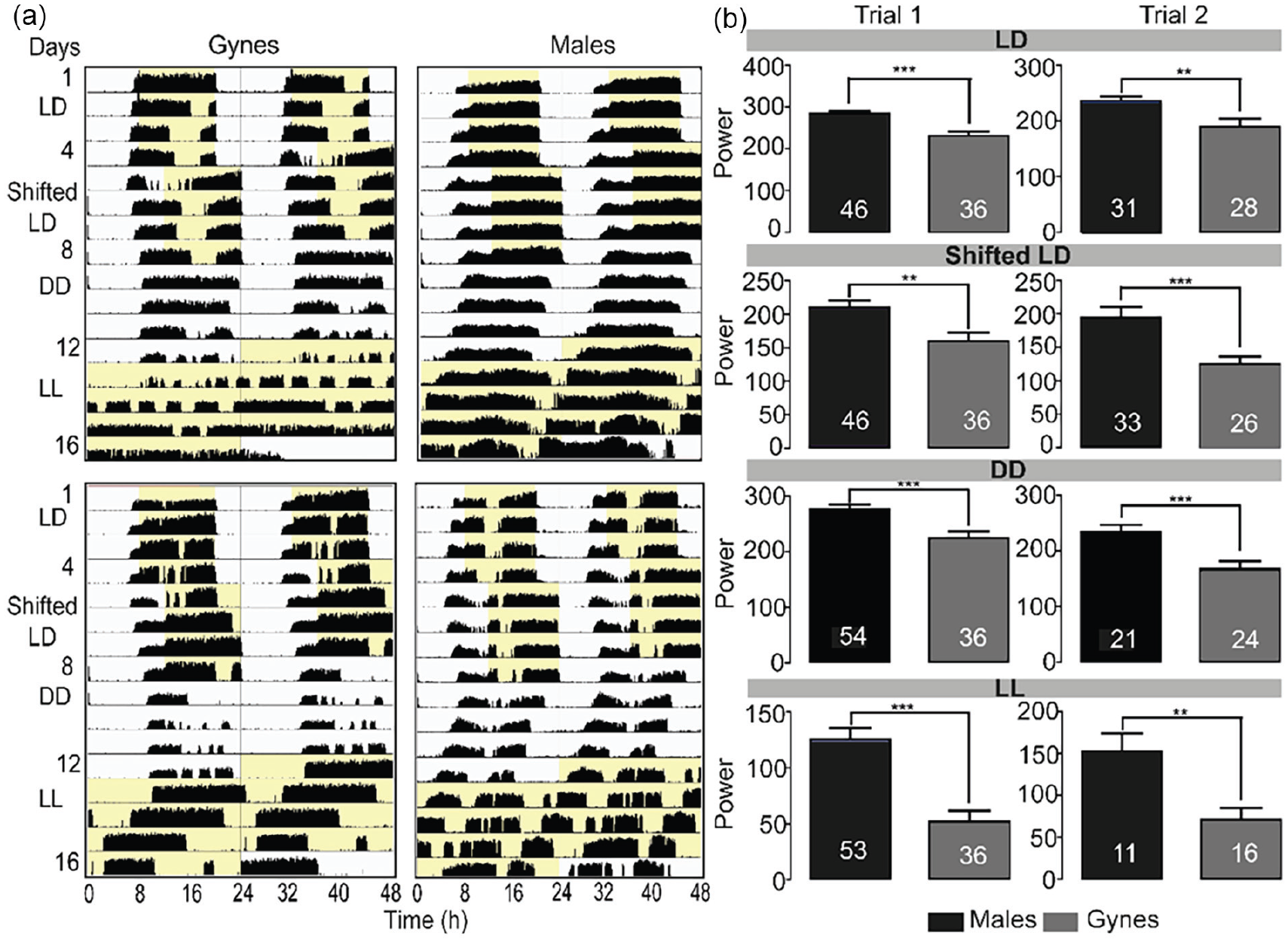
Locomotor activity of males and gynes under changing illumination regimes.
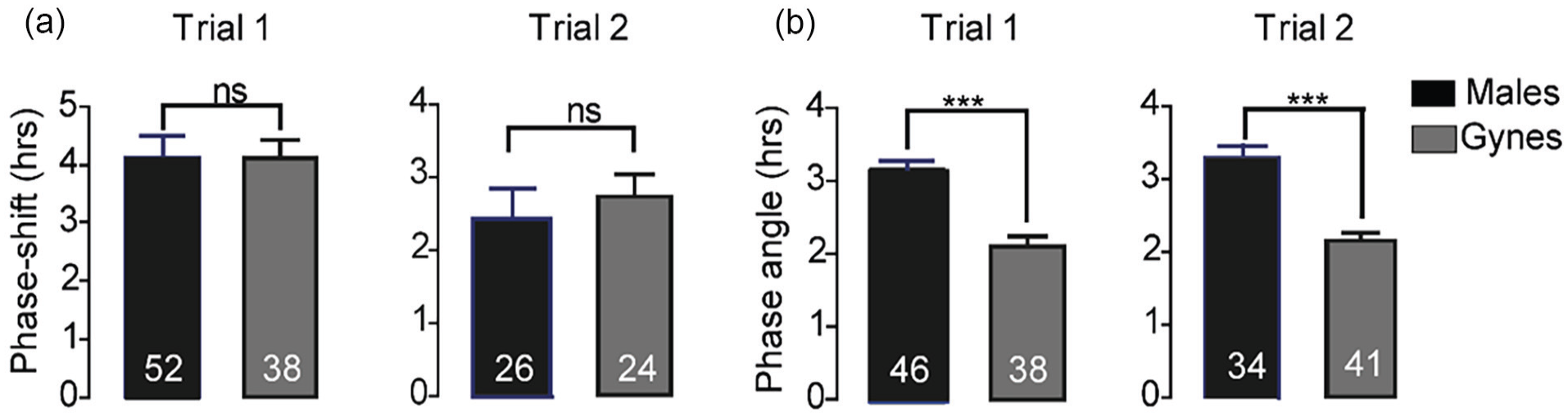
Both sexes showed strong circadian rhythms in locomotor activity, which were effectively and similarly shifted in response to a 4-h delay in the phase of the LD illumination regime in both trials (comparing males to gynes:
Bumble bee males and gynes showed longer free-running period (tau, estimated with the periodogram analysis, see Methods) in constant darkness relative to constant light (Figure 5; paired t-test;
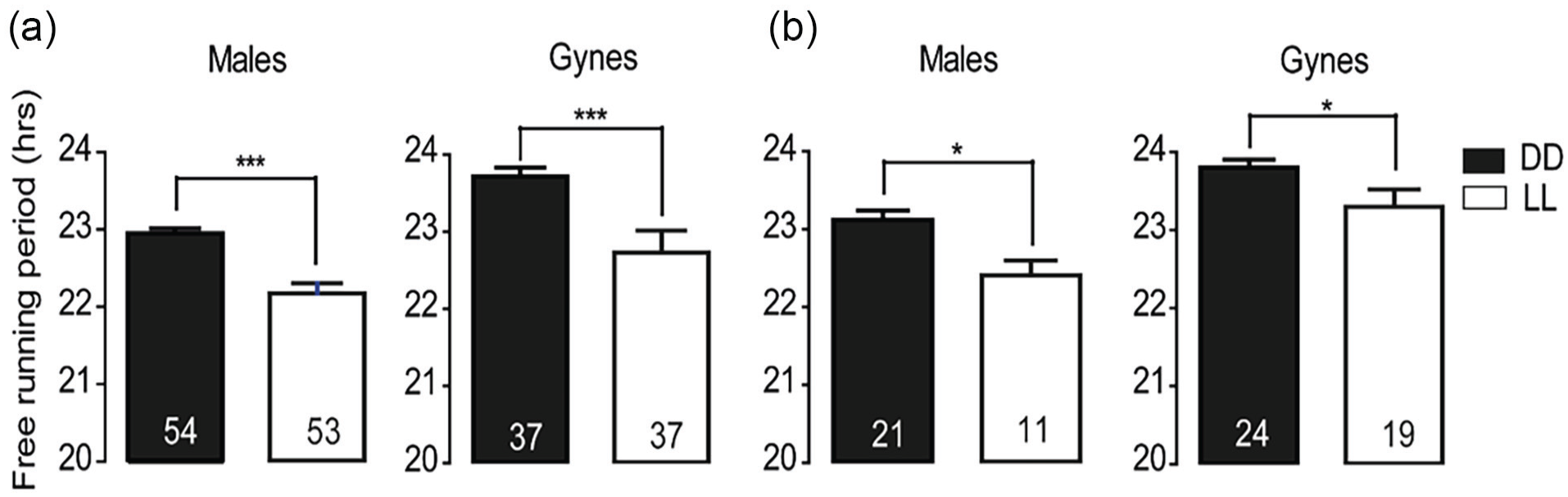
The Free Running Period of circadian rhythms in locomotor activity for males and gynes under constant light and constant dark illumination regimes. Both males and gynes showed a longer free-running period under constant darkness. (a) Trial 1 and (b) Trial 2. Asterisks indicate significant p values (paired t-test, *p < 0.05; ***p < 0.001).
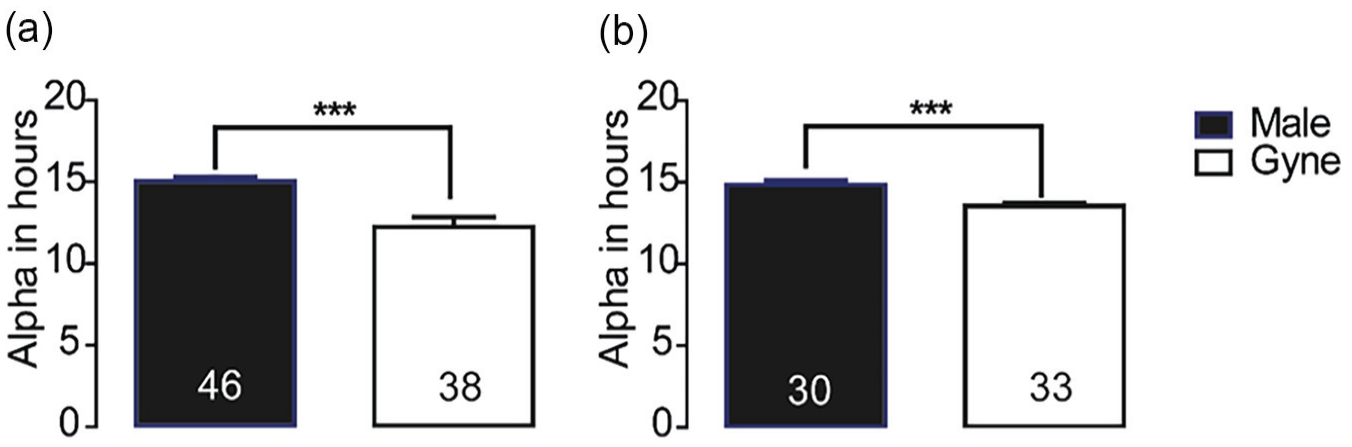
The interval between the onset and offset of the daily bout of activity (α) was longer for males compared with gynes (Mann–Whitney test, p < 0.0001 in both trials; Figure 6).
Discussion
Receptive males and females need to precisely coordinate their time of sexual receptivity and mating activity in order to successfully reproduce. Consistent with the notion that the circadian system is involved in sexual coordination, we found that male and gyne bumble bees develop strong circadian rhythms allowing them to precisely synchronize their daily activity. It is also noted that we identified several consistent differences between the two sexes. Males have stronger rhythms with an overall earlier phase and a longer Alpha. On the contrary, the time of emergence from the pupal cocoon was not gated by circadian clocks. We suggest that bumble bees do not need strong rhythms in adult emergence and during their early life because they emerge into a protected and regulated hive environment, but later need strong activity rhythms for timing foraging or mating-related behaviors.
The finding that gyne and male pupal emergence are not gated by the circadian clock contrasts with findings for a wide variety of insects in which emergence typically occurs at a definite time of day. It was shown in fruit flies and some other holometabolous insects that light cycles can entrain emergence rhythms consistent with having a fully functional clock gating the time of emergence (Brady, 1988; Sehgal et al., 1992). In Drosophila, mutations in the clock gene Period disrupt eclosion rhythms, causally linking clock genes to the regulation of eclosion timing (Konopka and Benzer, 1971). A rhythmic emergence also depends on the coupling between the central clock located in the brain and a peripheral clock located in the prothoracic glands producing the molting hormone and ecdysone. Molting hormone titers vary with a circadian rhythm and regulates eclosion behavior (Mark et al., 2021). It is assumed that gated emergence is important for reducing risks such as dehydration and predation (Daan, 1981). Workers of some social insects such as honey bees, bumble bees, and ants show a different pattern: they do not show circadian activity rhythms during their first days post eclosion and/or emerge from the pupa around the clock with no circadian rhythms (Eban-Rothschild and Bloch, 2012; Fujioka et al., 2017). Circadian rhythms in these behaviors are perhaps not needed because these social insects emerge into a tightly regulated nest environment in which they are protected from daily environmental changes and predation risks. Indeed, solitary bees that need to cope with environmental changes shortly after eclosion, emerge with clear rhythms that are influenced by temperature and light (Tweedy and Stephen, 1970; Yocum et al., 2016; Bennett et al., 2018; Beer et al., 2019). However, some social ants, such as queenless Diacamma sp., show rhythmic eclosion, which may be attributed to evidence that shortly after eclosion they spend substantial time aboveground (Fuchikawa et al., 2014). Our study extends the finding for bumble bees showing that gynes and males emerge with no circadian rhythms.
Consistent with the lack of circadian gating of emergence time, newly emerged gynes and males did not show circadian rhythms in locomotor activity during their first days as adults. These findings support and extend an earlier report for bumble bee gynes (Eban-Rothschild et al., 2011) and are consistent with the findings that circadian rhythms in locomotor activity of newly emerged honey bee drones and gynes are also typically weak or absent (Spangler, 1972; Harano et al., 2007). Both males and gynes developed circadian rhythms in their activity after several days, consistent with a similar ontogeny of circadian rhythms in locomotor activity in bumble bee and honey bee workers (reviewed by Eban-Rothschild and Bloch, 2012).
We found that both males and gynes showed strong internal circadian rhythms in locomotor activity which are readily entrained by light:dark cycles. The development of circadian rhythms in bumble bee gynes depends on their age and reproductive phase. These findings are consistent with earlier studies with honey bees in which virgin gynes emerged with no rhythms, later developed strong rhythms, but switched to activity around the clock with attenuated or no circadian rhythms when starting to lay eggs (Harano et al., 2007; Eban-Rothschild et al., 2011; Shpigler et al., 2022). Circadian rhythmicity is also similarly associated with the reproductive state in several ant species (Sharma et al., 2004). Males developed rhythms (100% on Day 4) slightly earlier than gynes (50% on Day 4, 50% on Day 5) along with their sexual maturity at 6 to 9 days after emergence and the time they are ready to conduct their mating flights (Yoon et al., 2016).
Interestingly, by contrast to studies with Drosophila and other insects (Emery et al., 2000) most of the males and gyne bumble bees showed robust circadian rhythms under constant light illumination in our experiments. A hypothesis that is worth studying in the future is that insects such as bees, beetles, and bugs that do not encode the light-sensitive Drosophila-type Cryptochrome (Cry-d; Rubin et al., 2006; Deppisch et al., 2023) show stronger rhythms under constant light compared to insects such as flies and butterflies that encode Cry-d, whereas Drosophila having only photosensitive CRY, and monarch butterflies, and mosquitos having both photosensitive CRY and mammalian CRY show arrhythmicity in their activity under continuous light conditions (linden bug, Kaniewska et al., 2020; beetle, Reshma et al., 2024; Drosophila, Emery et al., 2000; monarch butterfly, Liams et al., 2024; mosquito Au et al., 2022).
The slight differences in the ontogeny of circadian rhythms were accompanied by several additional sex-related differences in circadian rhythms. Males typically had an advanced activity phase and showed stronger circadian rhythms relative to gynes. We speculate that these differences may be functionally significant because they improve male reproductive success by allowing them to anticipate meetings with receptive females. This premise is consistent with studies on sex differences in other insects. For example, in Manduca sexta and Hyles lineata hawkmoths, female flight activity peaked in advance of males, which in turn timed their flight activity to periods of female sexual receptivity (Broadhead et al., 2017). In another moth Spodoptera littoralis, the release of the female sex-pheromone is circadianly regulated and was shown to effectively entrain male activity, providing the best evidence available so far for sexual selection acting on sociosexual communication in insects (Ghosh et al., 2024). This study also reports several sex-related differences. For example, females show shorter circadian period, which contrasts with our findings for the bumble bee. In Anopheles gambiae, males have an earlier flight activity peak than females, which is consistent with their aggregation in swarms to which females later enter (Rund et al., 2012). A study with Great tits in which fitness indices were explicitly measured indicated that males that are active earlier in the morning are preferred by females over later active males (Greives et al., 2015). Reproductively active male European ground squirrels initiate their aboveground activity earlier than females (Everts et al., 2004); in this system, it was suggested that this pattern can be advantageous for males by improving their success in male–male competition over territories and receptive females (reviewed by Hau et al., 2017; Helm et al., 2017). The stronger and faster male rhythms in bumble bees may be related to their mating strategy in which males wait for females at a meeting point, whereas in moths, males respond to the female sex attractants. Additional known mating strategies of bumble bee males include the deposition of sex-attractant scent marks on objects like twigs, lurking for receptive gynes at the nest entrance, and therefore, an early phase and strong rhythms may be advantageous (Valterová et al., 2019).
Many animal species perform sex-related activities such as courtship or mating at a definite time of day. Some of the best-studied species in this regard are moths in which successful mating requires temporal synchronization of both general activity and sexual communication (Silvegren et al., 2005; Groot, 2014). In honey bees and ants, receptive males and gynes synchronize their mating flights to species-specific times of the day, which is thought to reflect a mechanism evolved to reduce unproductive interactions between related sympatric species (Hölldobler and Wilson, 1990; McCluskey, 1992; Koeniger et al., 1996). Temporal isolation between closely related species is also considered a mechanism for speciation. For example, there is evidence for variation in the time of sexual activities between strains of Drosophila and Nasonia (Sakai and Ishida, 2001; Bertossa et al., 2013).
Taken together, our results suggest that circadian rhythms in bumble bee males and gynes were shaped by diverse evolutionary processes including sexual selection and their complex social lifestyle in which the brood and young adults are protected from external fluctuations in predation risk and abiotic environmental cycle. Our study also adds to the growing number of studies indicating that even basic physiological processes such as those generating circadian rhythms differ to some extent between males and females and therefore studies of only one sex are not sufficient.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304241283863 – Supplemental material for Sex-Related Variation in Circadian Rhythms in the Bumble Bee Bombus Terrestris
Supplemental material, sj-docx-1-jbr-10.1177_07487304241283863 for Sex-Related Variation in Circadian Rhythms in the Bumble Bee Bombus Terrestris by Ozlem Gonulkirmaz-Cancalar and Guy Bloch in Journal of Biological Rhythms
Footnotes
Acknowledgements
The authors thank Oded Shertzer for his help during the experimental setup and Almog Yaniv for his help with the camera setup. This project was supported by the European Union’s Horizon 2020 research and innovation program (grant no: 765937-CINCHRON) and grants from US–Israel Binational Agricultural Research and Development Fund (grant no. BARD; IS-5077-18 R to GB), and the US-Israel Binational Science Foundation (grant no. BSF; 2017188 to GB).
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
NOTE
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
