Abstract
Daily rhythms generated by endogenous circadian mechanisms and synchronized to the light-dark cycle have been implicated in the timing of birth in a wide variety of species. Although chronodisruption (e.g., shift work or clock gene mutations) is associated with poor reproductive outcomes, little is known about circadian timing during pregnancy. This study tested whether daily rhythms change during full-term pregnancies in mice and women. We compared running wheel activity continuously in both nonpregnant (
In many organisms, including humans, reproduction is influenced by the circadian system. Circadian clocks in the brain and tissues generate daily rhythms in sleep/wake, metabolism, hormone secretion, locomotor activity, and many other behaviors. These clocks synchronize (entrain) to daily environmental stimuli such as the light-dark cycle (Takahashi et al., 2008). For example, normal pregnancy requires a functional circadian system (Rosenwasser et al., 1987; Germain et al., 1993; Honnebier and Nathanielsz, 1994; Scribner and Wynne-Edwards, 1994; Ducsay, 1996; McElhinny et al., 1997; McGregor et al., 1999; Gonzalez-Mariscal et al., 2013; Miller and Takahashi, 2014; Perez et al., 2015), and circadian disruption can perturb pregnancy outcomes (Mahoney, 2010; Bonzini et al., 2011; Lin et al., 2011; Gamble et al., 2013; Amaral et al., 2014; Vilches et al., 2014; Fernandez et al., 2016). In addition, in women, spontaneous membrane rupture and the onset of labor most commonly occur between midnight and 0400 h (Cooperstock et al., 1987). Similarly, rodents are most likely to deliver pups around dawn (Reppert et al., 1987; Viswanathan and Davis, 1992). For example, mice with mutations in the circadian genes
An important aspect of circadian regulation is an individual’s chronotype, the stable phase relationship (in hours) between daily biological and environmental events (Roenneberg et al., 2007). Those who rise early and have an early midpoint of sleep (“larks”) have an early chronotype, whereas those who rise late and have a late midpoint of sleep (“owls”) have a late chronotype. Chronotype depends on age, sex, and genetics (Roenneberg et al., 2004; Patke et al., 2017; Chong, 2018), and misalignment between an individual’s chronotype and the external environment (chronodisruption or circadian misalignment) can negatively affect physiology and behavior (Reppert and Weaver, 2002; Erren and Reiter, 2009; Johnston et al., 2016). For example, chronodisruption can result when an individual with a late chronotype is forced to wake early.
In addition to being affected by the circadian system, pregnancy appears to influence the circadian regulation of behaviors, as pregnant women often report and are treated for sleep disturbances (reviewed in Lee, 1998; Sahota et al., 2003; McLafferty et al., 2018). Indeed, such daily disturbances have been detected in women of all ages, parities, races/ethnicities, and income levels and across all months of pregnancy (Mindell et al., 2015; Reid et al., 2017). In addition, pregnancy has variable effects on the time of peak daily expression of some clock genes in the rat brain (Schrader et al., 2010, 2011). However, whether pregnancy affects chronotype has not been addressed. Here, we examined the impact of normal pregnancy on circadian rhythms in both mice and women.
Materials And Methods
Mice, Housing, and Mating
All mice (C57BL/6NJ, aged 3-6 months; Jackson Laboratories, Bar Harbor, ME) were maintained individually in a cage with a running wheel on a 12 h:12 h light:dark cycle (2.4 ± 0.5 × 1018 photons/s*m2; General Electric). Food and water were available at all times in the temperature- and humidity-controlled Danforth Animal Facility at Washington University in St. Louis. All animal procedures were approved by the Washington University Institutional Animal Care and Use Committee and followed National Institutes of Health guidelines. Females were paired with a male for 2 h; those that developed a copulatory plug were considered pregnant. We used an infrared camera (Dlink, Taipei, Taiwan) to determine the time of delivery of the first pup. Mice that completed their pregnancies were included in the analysis.
Locomotor Activity in Mice
We assessed locomotor activity of nonpregnant and pregnant females by measuring wheel-running data in 6-min bins (Clocklab, Actimetrics, Evanston, IL). Daily onset of activity was defined as the first time when activity was counted for at least 1 h after at least 4 h of inactivity (Clocklab software, Actimetrics). Chronotype data were statistically analyzed in Prism (GraphPad 7.02, La Jolla, CA) by using either an independent Student
Human Subject Recruitment
Institutional Review Board approval was obtained from the Washington University School of Medicine Human Research Protection Office (IRB 201410060, approved October 20, 2014). Women planning pregnancy were recruited from the local community (
Demographic information of study participants (

Actigraphy in Women
We used the MotionWatch 8 actigraphy watch (CamNtech Ltd, Cambridge, UK) to detect wrist movements every minute. We obtained recordings from women before conception and throughout pregnancy. Trimesters were defined as gestational weeks 4 through 13 (trimester 1), 14 through 27 (trimester 2), and 28 through delivery (trimester 3). Data were analyzed with a custom R script (http://www.R-project.org/). Briefly, total daily activity was calculated as the sum of all accelerometer counts each day. Sleep onsets were determined from the actigraphy profile of each 24 h (1200 h to 1200 h the next day). We calculated the instantaneous frequency of motion counts as the inverse of the time between each wrist movement. Based on the distribution of instantaneous frequency counts, we identified the time of sleep onset. We excluded weekends and days with more than 13 h of inactive time and included only patients with at least 7 days of usable data per trimester. A custom R script was used to correct for Daylight Saving Time, and paired Wilcoxon rank-sum tests were used for statistical analyses.
Results
Chronotype Advances during Pregnancy in Mice and Women
To determine whether daily rhythms change during pregnancy, we continuously monitored running wheel activity of C57BL/6NJ mice. Nonpregnant dams (
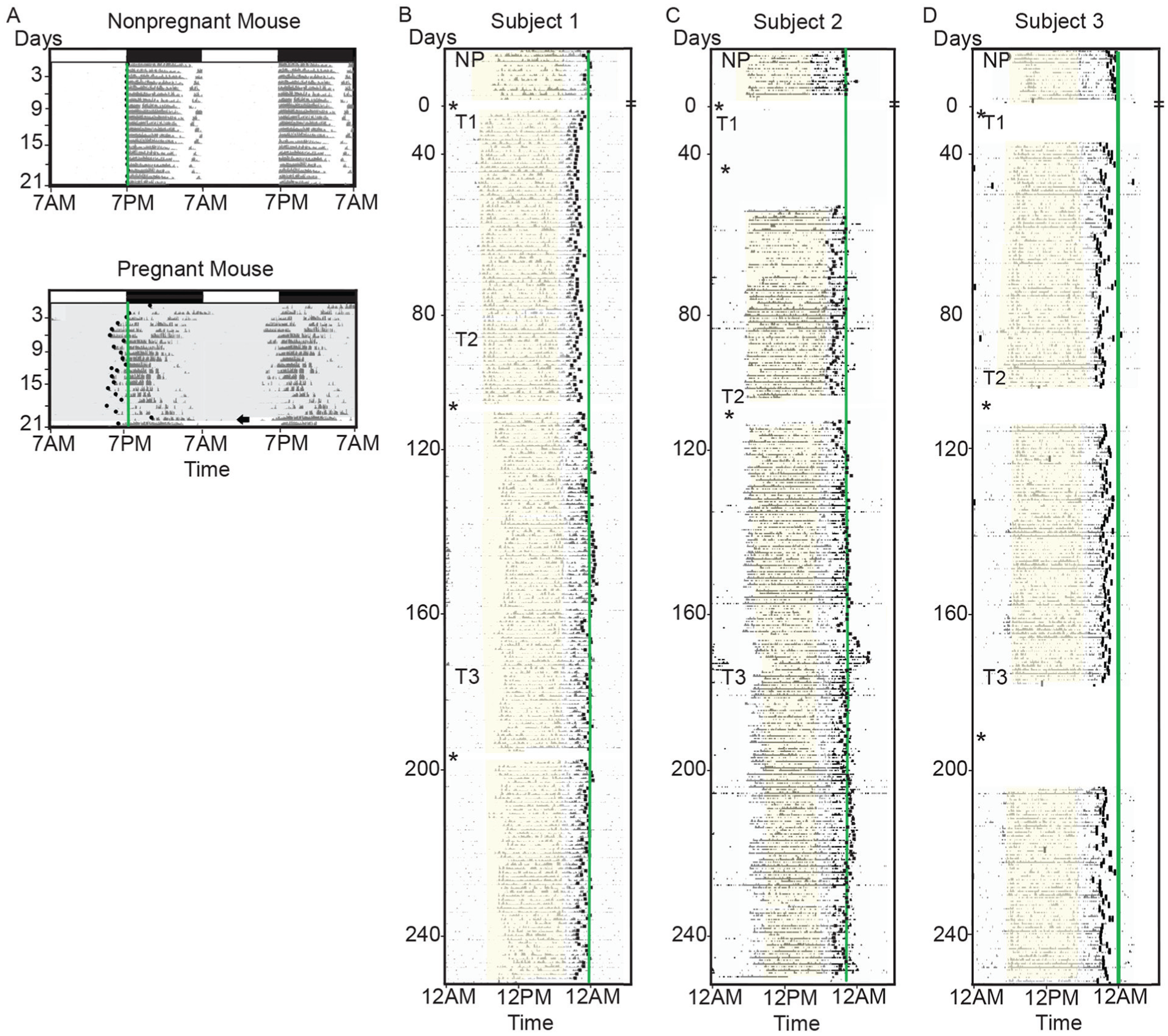
Pregnancy shifts daily locomotor activity patterns in women and mice. (A) Representative double-plotted actograms from 2 female mice recorded in a light-dark cycle as indicated by the white and black bars. (B-C) Representative single-plotted actograms of normal (subject 1), early (subject 2), and late (subject 3) chronotype women for 2 weeks while nonpregnant (NP) and throughout their first, second, and third trimesters (sunrise to sunset, yellow shading).
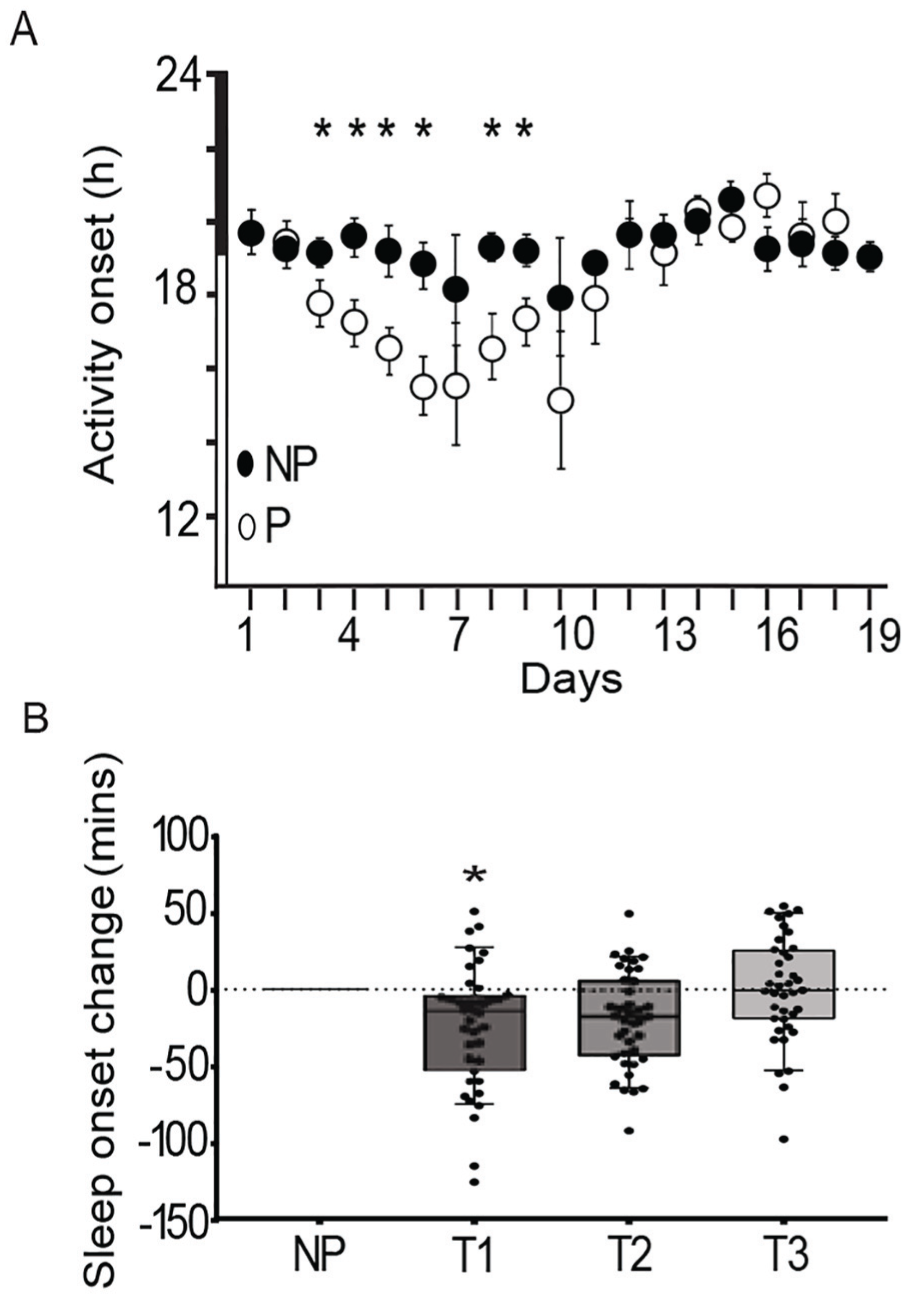
The daily onset of activity in mice and sleep in women becomes earlier during early pregnancy. (A) The daily activity onset advanced in mice in early pregnancy (open circles, mean ± SEM,
To determine the effects of pregnancy on circadian timing in women, we analyzed locomotor wrist actigraphy data from 39 women planning pregnancy. Women who conceived were followed throughout their entire pregnancy. We analyzed the data as the change in sleep onset to account for the individual differences in the baseline chronotype of the participants. Similar to the chronotype change in pregnant mice, the timing of sleep onset in women became earlier during the first (T1) and second (T2) trimesters compared to the sleep onset before pregnancy (
Measures of sleep and wrist activity (median, quartile 1, quartile 3) before and during pregnancy in women (

Superscripted letters indicate differences between prepregnancyA, first trimesterB, second trimesterC, and third trimesterD (Wilcoxon rank-sum test,
Seasonal distribution of sleep onset (mean vector, 95% confidence interval [CI]) during pregnancy in women (
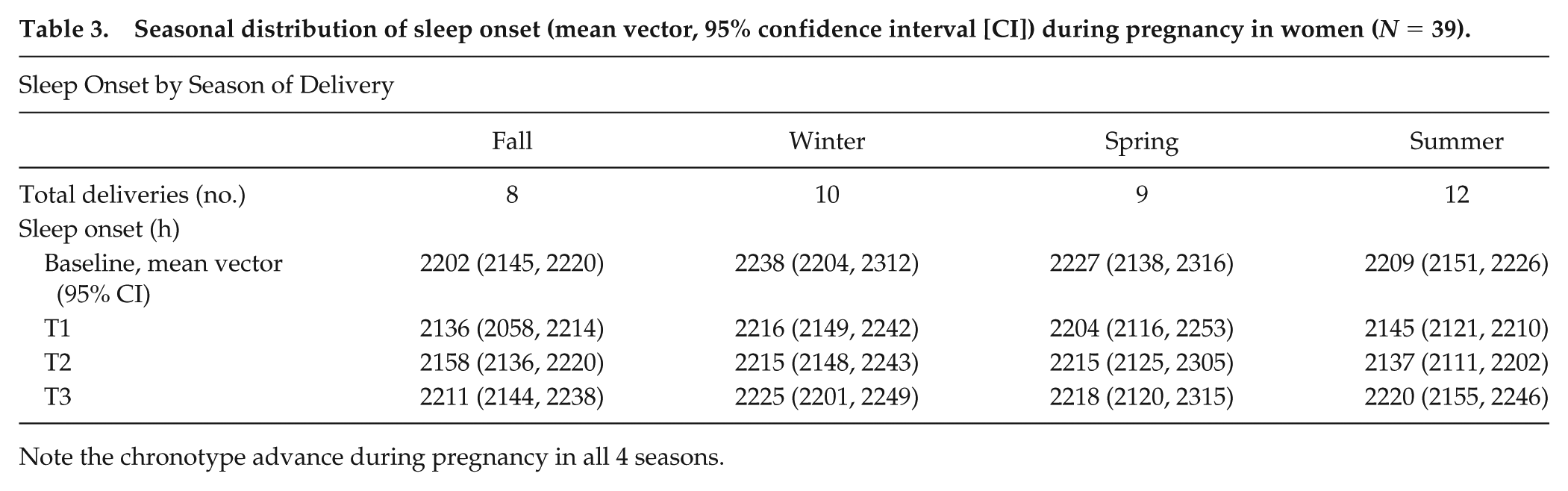
Note the chronotype advance during pregnancy in all 4 seasons.
Locomotor Activity Declines in Mice and Women during Pregnancy
Shifts in chronotype could reflect changes in circadian phase or in the amount of locomotor activity. Because pregnancy is often accompanied by reduced overall movement (Richards, 1966; Albers et al., 1981; Rosenwasser et al., 1987; Scribner and Wynne-Edwards, 1994; Kimura et al., 1996; Poudevigne and O’Connor, 2006), we measured total daily activity throughout pregnancy. Mice showed significantly reduced levels of locomotor activity beginning on day 12 of pregnancy (Fig. 3A; repeated-measures 2-way ANOVA,
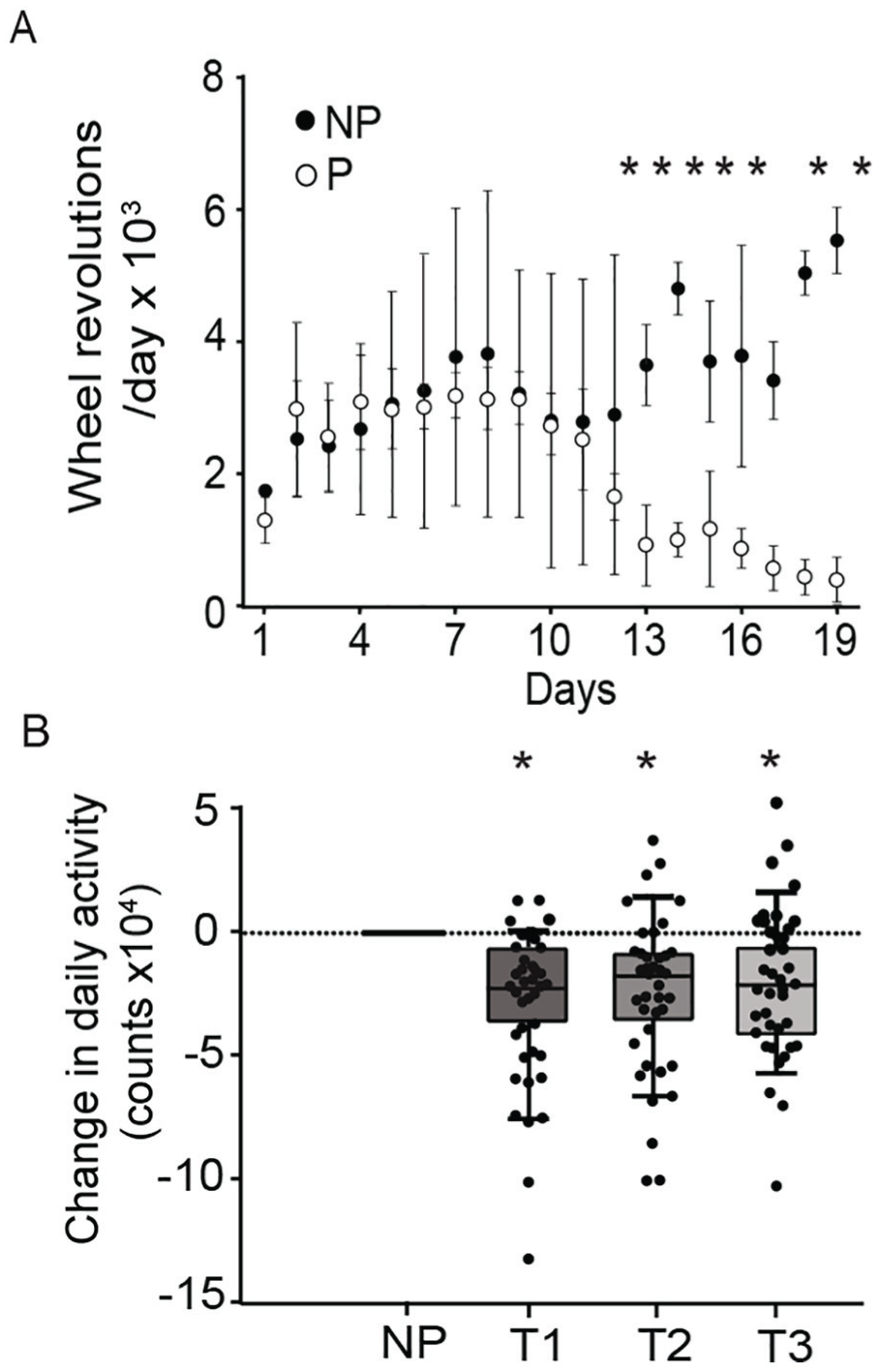
Total activity diminishes across pregnancy in women and mice. (A) Pregnant (P) mice (
Discussion
Our results indicate that chronotype and activity levels change during pregnancy in both women and mice. Despite differences in baseline chronotype of the study participants, or the time that women wake and sleep, women showed lower activity throughout pregnancy and advanced sleep onset during their first 2 trimesters. Their advanced chronotype returned to the prepregnancy state during the third trimester. In mice, a normal pregnancy also was characterized by 3 phases: 1) an advance in the onset of daily activity by pregnancy day 3, 2) a return of activity onset characteristic of the prepregnant stage around pregnancy day 10, and 3) a decrease in levels of locomotor activity beginning at pregnancy day 12 and lasting until delivery.
Women differed from mice in several ways in our study. Although chronotype advanced during pregnancy in both women and mice, this was manifested in daily sleep onset in women and locomotor activity onset in mice. Sleep onset rather than sleep offset (activity onset) was used as a surrogate for chronotype as social constraints of work (e.g., causing women to use alarm clocks), and familial relationships may have masked the effects of pregnancy on women’s timing of sleep offset. In addition, women, but not mice, showed decreased locomotion starting early in pregnancy. By categorizing data from women by trimester, we may have diminished the ability to detect events in early pregnancy that we were able to resolve with daily measures from inbred mice. Regardless, actigraphy provides an inexpensive and noninvasive method to continuously monitor chronotype during pregnancy. It is intriguing that we find similar phenotypes monitoring wrist actigraphy in women and wheel running in mice. Suppression of general activity during pregnancy has been reported in women using questionnaires, pedometry, and actigraphy (Shima, 1990; Sternfeld, 1997; Lindseth and Vari, 2005; Cioffi et al., 2010; Harrison et al., 2011; Nascimento et al., 2015; Oliveira et al., 2017) and in mice using general activity and wheel running (Richards, 1966; Terada, 1974; Albers et al., 1981; Rosenwasser et al., 1987; Scribner and Wynne-Edwards, 1994; Ducsay, 1996). We conclude that noninvasive methods such as actigraphy can be used to study how chronotype disruption, including shift work, might affect reproductive and delivery outcomes.
The mechanisms underlying pregnancy-induced changes in chronotype are unknown, but the similar responses in women and mice suggest a conserved mechanism. Two candidates for consideration are ovarian steroid hormones and metabolism. Shifts in chronotype at the time of puberty have been attributed to sex hormones in rats, common degus, and macaques (Golub et al., 2002; Hagenauer et al., 2009; Hagenauer et al., 2011a, 2011b; Hagenauer and Lee, 2012) and could underlie the pregnancy-induced shifts in daily rhythms. In rats, high levels of estrogen during the proestrus stage advance the timing of daily onset of activity and increase daily wheel running (Albers et al., 1981). In contrast, progesterone can delay activity onset during estrus and lengthen the period of locomotor activity (Albers et al., 1981). Thus, the coordinated increases in estrogen and/or progesterone during early pregnancy in mice and women may underlie the observed changes in chronotype.
Changes in maternal metabolism over pregnancy coincide with alterations in the progesterone/estradiol ratio and or progesterone/estradiol receptor status and may contribute to the findings presented here (Jones and Astwood, 1942; Hahn and Hays, 1963; Romero et al., 1988; Maclin et al., 1990; Potgieter et al., 1995). For example, during pregnancy, maternal basal and postprandial glucose metabolism adapts to meet maternal and fetal demands (Lain and Catalano, 2007). Our observations of the normal advances in chronotype during pregnancy may explain why studies have shown that late chronotype pregnant women are about 2.5 times more likely to develop gestational diabetes in their second trimester than those who have an earlier chronotype (Facco et al., 2017). Future studies should investigate whether these changes in chronotype reflect changes in circadian timing. Mice will serve as a powerful model to investigate the genes, molecules, and cells underlying circadian changes during pregnancy.
Although some studies report increased sleep duration during pregnancy, others report insomnia in mice and women at term (Driver and Shapiro, 1992; Hertz et al., 1992; Brunner et al., 1994; Kimura et al., 1996; Reichner, 2015; Hashmi et al., 2016; Reid et al., 2017). Our findings that activity levels decreased during late pregnancy in mice and throughout pregnancy in women are consistent with a large body of literature (Rosenwasser et al., 1987; Scribner and Wynne-Edwards, 1994; Kimura et al., 1996; Poudevigne and O’Connor, 2005, 2006) and suggest that a common mechanism modulates sleep/wake cycles during pregnancy in the 2 species. These data also indicate temporally separated changes in the regulation of daily activity and physiology by circadian (time of day gating) and sleep homeostatic (sleep debt accumulated during wakefulness) processes. Future studies assessing changes in other measures of the sleep homeostat including sleep fragmentation throughout pregnancy would be informative. Taken together, these results indicate that pregnancy induces an advance of the circadian system that resolves prior to term delivery and is separable from the processes controlling sleep duration.
Footnotes
Acknowledgements
The authors thank Drs. Deborah Frank and Antonina Frolova and members of the Herzog and England labs for helpful discussions and comments on drafts of the manuscript. This work was supported, in part, by the March of Dimes Prematurity Research Center at Washington University. Dr. Carmel A. Martin-Fairey was supported, in part, by T32 HD049305 and F32 HD093269-01.
Author Contributions
CAM-F designed and conducted experiments, analyzed and interpreted data, and wrote the article. PZ designed and conducted experiments, analyzed and interpreted data, and wrote the article. LW conducted experiments, analyzed and interpreted data, and wrote the article. TR designed and conducted experiments, analyzed and interpreted data, and wrote the article. JF designed and interpreted data and wrote the article. XM interpreted data. RM conducted experiments and analyzed and interpreted data. ESJ designed and conducted experiments, analyzed and interpreted data, and wrote the article. SKE designed experiments, analyzed and interpreted data, and wrote the article. EDH designed experiments, analyzed and interpreted data, and wrote the article.
Conflict Of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
