Abstract
In the fruit fly Drosophila melanogaster, social interactions especially among heterosexual couples have been shown to have significant impact on the circadian timing system. Olfaction plays a major role in such interactions; however, we do not know yet specifically which receptor(s) are involved. Further, the role of circadian clock neurons in the rhythmic regulation of such sociosexual interactions (SSIs) is not fully understood. Here, we report the results of our study in which we assayed the locomotor activity and sleep-wake behaviors of male-male (MM), female-female (FF), and male-female (MF) couples from several wild-type and mutant strains of Drosophila with an aim to identify specific olfactory receptor(s) and circadian clock neurons involved in the rhythmic regulation of SSI. The results indicate that Or47b receptor neurons are necessary for SSI, as ablation or silencing of these neurons has a severe impact on SSI. Further, the neuropeptide pigment dispersing factor (PDF) and PDF-positive ventral lateral (LNv) clock neurons appear to be dispensable for the regulation of SSI; however, dorsal neurons may be involved.
Circadian clocks time a multitude of physiological and behavioral processes in a wide variety of organisms ranging from bacteria to humans. These clocks use several time cues to keep track of time in their local environment, among which social cues are of special importance. In the fruit fly Drosophila melanogaster, social interactions especially among heterosexuals have been shown to have significant effect on the circadian timing system (Fujii et al., 2007). Such interactions are rhythmic and occur predominantly in the night, resulting in intense locomotor activity involving stereotypic behaviors such as chasing, avoiding, singing, dancing, licking, and copulation (Hall, 1994). In Drosophila, core circadian clock genes have been implicated in the regulation of rhythms in some sociosexual behaviors such as courtship and mating (Wheeler et al., 1991; Tauber et al., 2003). Although we know a fair deal about the regulation of locomotor activity and adult emergence rhythms in Drosophila, our understanding of the mechanisms underlying reproduction-related rhythms such as mating and egg laying is still in its infancy (Howlader et al., 2006). Circadian clocks are known to regulate interpulse intervals in the courtship song of Drosophila (Kyriacou and Hall, 1980), an important ritual in courtship and mating (Hall, 1994). Not only the song but also the process of courtship itself is rhythmic, at least in some species of Drosophila (D. melanogaster, D. yakuba, and D. kikkawai), with a peak occurring just before lights-on (Hardeland, 1972; Loher and Zervas, 1979). While the mating rhythm in Drosophila is believed to be primarily dictated by females (Sakai and Ishida, 2001), a recent study showed that nocturnal sex drive (NSD), estimated in terms of close association between males and females, and quantified as enhanced nighttime activity is rhythmic, is dictated primarily by males (Fujii et al., 2007). NSD requires intact olfactory ability because in flies with loss-of-function mutation in a widely expressed olfactory receptor Or83b (Larsson et al., 2004), it is severely compromised (Fujii et al., 2007). Although there is no doubt about the involvement of olfaction in sociosexual interactions (SSIs) between males and females (Levine et al., 2002; Fujii et al., 2007; Krupp et al., 2008), specific olfactory receptors involved are still unknown. In D. melanogaster, several pheromone receptors reside in the trichoid sensillae (Vosshall and Stocker, 2007), of which 4—Or65a, Or88a, Or47b, and Or67d—are thought to be critical for sociosexual communications between males and females (van der Goes van Naters and Carlson, 2007).
In Drosophila, circadian clocks regulate oscillations of genes responsible for vision, learning and memory, synaptic transmission, olfaction, locomotion, detoxification, metabolic stress, metabolism, mating, and egg laying and at the molecular level consist of transcription-translational feedback loops involving mRNA and proteins of core clock genes such as period (per), timeless (tim), Clock (Clk), and cycle (cyc) (Allada and Chung, 2010). The circadian organization is multioscillatory; central pacemaker cells located in the fly brain and peripheral oscillators in various organs work in tandem to regulate rhythmicity in behavior and physiology (Plautz et al., 1997; Giebultowicz, 2000). The brain pacemakers in D. melanogaster comprise at least 6 groups of clock neurons including pigment dispersing factor (PDF), positive small and large ventral lateral neurons (sLNv and ILNv), PDF-negative fifth sLNv, dorsal lateral neurons (LNd), 3 groups of dorsal neurons (DN1, DN2, and DN3), and lateral posterior neurons (LPN) (Kaneko and Hall, 2000; Hall, 2003; Helfrich-Förster, 2003, 2004; Sheeba et al., 2008). Electrical silencing or genetic ablation of PDF-positive neurons results in arrhythmicity under constant dark (DD) conditions (Renn et al., 1999; Blanchardon et al., 2001; Nitabach et al., 2002).
Here, we report the results of our study aimed at identifying specific olfactory receptor neuron(s) and clock neurons involved in SSIs between males and females of D. melanogaster. The results of our study suggest that the circadian neurotransmitter PDF and the PDF-positive LNv neurons are not required for SSI and that such interactions are primarily male driven and mediated by olfactory receptor neurons: Or47b and its circadian clock system.
Materials and Methods
Fly Strains
Fly strains used in our study were Canton S (CS), 2 white eye strains (w and w1118), Oregon R (OR), yellow white (yw), Or83b0, Or65aGAL4, Or47bGAL4, Or88aGAL4, Or67dGAL4, Gr5aGAL4, pdfGAL4, cryGAL4, pdf0, UASKir2.1, UASdti, UASdORKC1, and UASdORKNC1. The receptor Or83b is a widely expressed olfactory receptor, which acts as a coreceptor of most olfactory receptors, and flies with Or83b loss-of-function mutation (Or83b0) have abnormal behavioral and physiological responses to many odors (Larsson et al., 2004; Vosshall and Stocker, 2007). The olfactory receptors Or65a, Or47b, Or88a, and Or67d expressed in the trichoid sensillae are known to be involved in social communication between males and females (Vosshall and Stocker, 2007; van der Goes van Naters and Carlson, 2007). Or67d is strongly activated by the pheromone 11-cis-vacennyl acetate (cVA), and Or67dGAL4 knock-in mutants were obtained by replacing an open reading frame of Or67d by a yeast transcriptional activator (GAL4) (Kurtovic et al., 2007). Gr5a receptor neurons are sugar responsive, regulate feeding and food storage (Vosshall and Stocker, 2007), and are known to possess circadian oscillators (Chatterjee et al., 2010). pdfGAL4 targets the expression of the gene of interest in large and small LNv neurons (Renn et al., 1999), whereas 4.0 cryGAL4 targets a much larger subset of neurons comprising some sLNv, lLNv, LNd, DN1, DN2, and most DN3 neurons (Hao et al., 2008). DTI encodes the diphtheria toxin, whose expression disrupts protein synthesis in the target cells, resulting in its ablation (Thum et al., 2006). The Drosophila open rectifier K+ (dORKC1) encodes the potassium channel, which acts as a K+-selective hole in the cell membrane and affects its resting membrane potential, while dORKNC1 is nonconducting, and its expression does not significantly lower the firing properties of the cell (Nitabach et al., 2002).
Measurement and Data Analysis
Different strains of D. melanogaster were maintained as preadults and subsequently as adults under 12:12-hour light/dark (LD) cycles with 500-lux white fluorescent light during the light phase and dim red light (λ > 650 nm) during the dark phase. Virgin flies were segregated as male-male (MM), female-female (FF), and male-female (MF) pairs and introduced into activity tubes of 7-mm diameter and 80-mm length for locomotor activity recording. The locomotor activity tubes had corn-meal food at one end and a cotton plug at the other. These tubes were placed in the DAM system (Trikinetics, Waltham, MA), and locomotor activity data were collected and stored in 5-minute bins. Food in the activity vials was changed every day at arbitrary times during the daytime to avoid any possible disturbance due to larval movement and physiological effects of the presence of eggs and larvae. To avoid any bias of disturbance, activity vials of same-sex couples were also changed in parallel. This was done by placing a fresh activity tube against the mouth of the tube containing flies and by gently letting the flies slide into the fresh tube. The activity data were analyzed using CLOCKLAB software from Actimetrics (Wilmette, IL). We used locomotor activity and sleep as readouts for SSI between males and females. A couple was considered to be asleep only when no activity count was registered for a duration of 5 minutes (Shaw et al., 2000). The activity and sleep profiles of the couples were computed by averaging raw activity data collected over a period of 24 hours, using data from the first 5 days of recording. In addition, we compared the total, day, and night activity and sleep of the MM, FF, and MF social groups by summing up total activity counts and sleep episodes (in minutes) during the entire day (daytime + nighttime), light (daytime), and dark (nighttime) phases of 12:12-hour LD cycles.
Plotting Activity and Sleep Data
For visual comparison of activity patterns, actograms, plots of activity with time of the day along the abscissa and day in a chronological manner along the ordinate, were drawn. The actograms show average activity data of couples, double plotted for the ease of visual identification of activity/rest patterns. In a double-plotted actogram, activity data for the second day are replotted on the second half of the first line (24-48 hours), those of the third day are replotted on the second half of the second line (24-48 hours), and so on. Therefore, activity data in the row indicated as day 1 include data for the first and second cycles, day 2 for second and third cycles, day 3 for third and fourth cycles, and so on. Because these are normalized activity counts, the replotted patterns are not exactly identical but are similar. The activity waveforms of couples were plotted using raw activity data counts collated in hourly bins and averaged across all couples during the first 5 days of recording. Sleep profile was generated by averaging sleep data in minutes (in 30-minute bins) across all couples and plotting it as a function of the time of day.
Statistical Analysis
The activity and sleep data of couples were analyzed using 2- or 3-way analyses of variance (ANOVA) with phase (day and night), social group (MM, FF, and MF), and/or strain (different genotypes specified) as fixed factors. Statistically significant interactions were subjected to post hoc multiple comparisons using the Tukey test, where p < 0.05 was considered as a statistical level of significance. Statistical analysis of data was implemented using STATISTICA for Windows Release 5.0B (StatSoft, 1995).
Results
MF Couples of Wild-Type CS Flies Display Enhanced Daytime and Nighttime Activity and Loss of Daytime and Nighttime Sleep
We find that in wild-type CS flies, MF couples display enhanced activity and reduced sleep compared to MM or FF couples (Fig. 1A-C). Two-way ANOVA of the activity data showed a significant effect of social group (p < 0.0001) and social group × phase interaction (p < 0.001). Post hoc multiple comparisons using the Tukey test revealed that daytime and nighttime activities of MF couples are significantly higher than MM or FF couples (p < 0.0001 for all 4 pair-wise comparisons) (Fig. 1B, 1C). Two-way ANOVA on the sleep data showed a significant effect of social group (p < 0.0001), phase (p < 0.0001), and social group × phase interaction (p < 0.0001). Post hoc multiple comparisons revealed that the total sleep of MF couples is reduced (p < 0.0001) compared to MM or FF couples. During nights, MF couples sleep significantly less compared to MM (p < 0.0001) or FF couples (p < 0.0001), while during the day, they sleep significantly less than MM (p < 0.0001) but as much as FF couples (p > 0.05) (Fig. 1D, 1E).

SSI in wild-type CS flies. Double plots of actograms (A) show higher nighttime activity in MF couples (n = 27) compared to MM (n = 28) or FF (n = 28) couples under 12:12-hour LD cycles. The actograms were averaged across all couples. Time of day (0-48 hours) is plotted along the abscissa and number of days in a chronological manner along the ordinate. The light and dark bars above the actograms (A) indicate light and dark phases of LD cycles. In double-plotted actograms, activity data on line 1 (day 1) include data for first and second cycles; day 2, second and third cycles; day 3, third and fourth cycles; and so on. The average activity profiles of MM, FF, and MF couples plotted using raw activity data as a function of time show that MF couples exhibit greater activity compared to MM or FF couples (B). The total daytime and nighttime activities of MM, FF, and MF couples were calculated by summing up activity counts during the light and dark phases of LD cycles (C). The average sleep profiles of MM, FF, and MF couples plotted using raw sleep data (in minutes of sleep per 30 minutes) as a function of time show that MF couples exhibit less sleep compared to MM or FF couples (D). The total, daytime, and nighttime sleep of MM, FF, and MF couples was calculated by summing up sleep durations (in minutes) during the whole day (light + dark, 24 hours), light (12 hours), and dark (12 hours) phases, respectively, of LD cycles (E). Clear and gray-shaded areas in the activity and sleep profiles (B, D) indicate day and night under LD cycles. Asterisks on the MM and FF bars indicate significant differences from the corresponding MF bars at p < 0.05.
Because it is entirely possible that mere mathematical addition of activity counts of 2 individual flies may result in unique patterns in the final activity profile of the pair, we obtained activity data of solitary CS males and females and then pooled the data from 2 males, 2 females, or 1 male and 1 female to generate cumulative activity data of MF (n = 16), MM (n = 16), and FF (n = 16) couples (data not shown). Analysis of the simulated pooled activity data revealed that the activity profile and daytime and nighttime activities of MF couples do not differ statistically from MM or FF couples (p > 0.05), suggesting that mathematically pooling time series data of males and females into different social groups does not alter either the levels or profiles of activity.
The activity and sleep of 4 other wild-type strains (yellow white [yw], white eye [w], w1118, and Oregon R) of Drosophila also showed a similar increase in activity and loss of sleep in MF couples relative to MM or FF couples (Suppl. Fig. S1). These results suggest that MF couples of wild-type flies display SSI-mediated circadian phenotype.
SSI Is Olfaction Mediated
To study the role of olfaction in SSI, we examined locomotor activity and sleep of 3 social groups (MM, FF, and MF) of flies with loss-of-function mutation in a widely expressed olfactory receptor Or83b (Or83b0). Because it is likely that reduction, if any, of the SSI phenotype in MF couples may be due to genetic background effect, we compared the activity and sleep of Or83b0 flies with that of its genetic background w1118 (Fig. 2 and Suppl. Fig. S1). In contrast to the wild-type background strain (w1118), activity of MF couples in Or83b0 flies is similar to MM couples during daytime and nighttime but greater than FF couples during nights (Fig. 2A, 2B and Suppl. Fig. S1), which implies that olfaction is essential for SSI. Three-way ANOVA on the activity data showed a significant effect of strain (p < 0.0001), social group (p < 0.0001), strain × social group (p < 0.005), social group × phase (p < 0.0001), and strain × social group × phase interactions (p < 0.0001). Post hoc multiple comparisons revealed that in Or83b0 flies, daytime as well as nighttime activities of MF couples are similar to MM couples (p > 0.05) but greater than FF couples during night (p < 0.0005) (Fig. 2B). Activity of the control (w1118) MF couples is significantly greater than MM or FF couples during daytime (p < 0.005) and nighttime (p < 0.0001) (Fig. 2 and Suppl. Fig. S1). Nighttime activity of w1118 MF couples is greater than that of Or83b0 (p < 0.05) (Fig. 2 and Suppl. Fig. S1). Three-way ANOVA on the sleep data showed a significant effect of social group (p < 0.0001), phase (p < 0.0001), strain × social group (p < 0.0001), social group × phase (p < 0.0001), strain × phase (p < 0.0001), and strain × social group × phase interaction (p < 0.0001). While total, daytime, and nighttime sleep in Or83b0 MF couples is similar to MM couples (p > 0.05) (Fig. 2C, 2D), their total and daytime sleep is similar to FF couples (p > 0.05), but nighttime sleep is lower than FF couples (p < 0.005) (Fig. 2C, 2D). The total, daytime, and nighttime sleep of w1118 MF couples is significantly less than MM (p < 0.0001) or FF couples (p < 0.0001) (Suppl. Fig. S1). Total and nighttime sleep of Or83b0 MF couples is lower than those of w1118 (p < 0.05) (Fig. 2 and Suppl. Fig. S1).
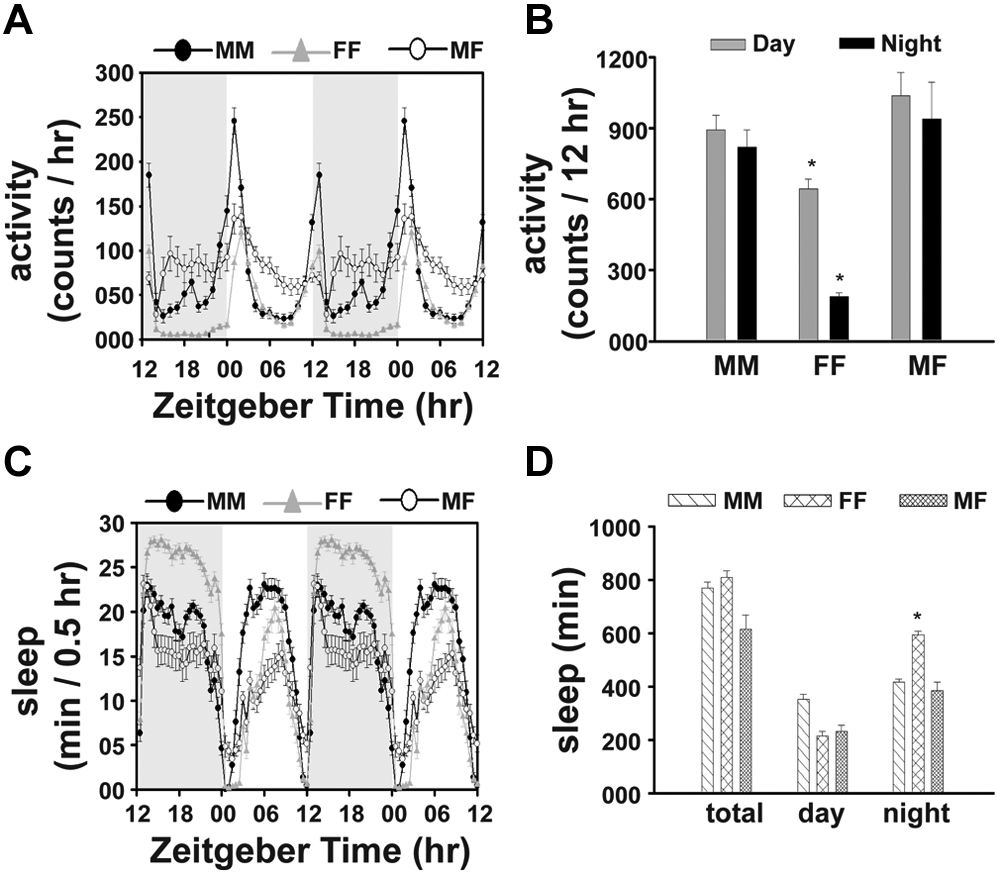
SSI is olfaction mediated. The activity data (A, B) of Or83b0 flies indicate that SSIs are disrupted because daytime and nighttime activities of MF couples (n = 28) are not different from MM couples (n = 28), although higher than FF couples (n = 28). The daytime and nighttime sleep of MF couples is also not different from MM couples, although their nighttime sleep is less than FF couples (C, D). All other details are the same as in Figure 1.
Reduced SSI in Or47b Manipulated MF Couples
While Or83b is distributed in almost all olfactory receptor neurons (ORNs), a smaller number of ORNs express the receptors Or47b, Or65a, Or88a, and Or67d. Among these, we found that ablation of Or47b neurons results in the most dramatic disruptions in SSI (Fig. 3 and Suppl. Figs. S3-S5). Three-way ANOVA on the activity data of Or47b-ablated flies (Or47bGal4/UASdti) and its 2 genetic controls (Or47bGal4 and UASdti) showed a significant effect of strain (p < 0.0001), social group (p < 0.0001), phase (p < 0.0001), strain × social group (p < 0.0001), social group × phase (p < 0.0001), and strain × social group × phase interaction (p < 0.0002). In the Or47b-ablated flies, daytime and nighttime activities of MF couples do not differ from MM or FF couples (p > 0.05), while daytime and nighttime activities of MF couples of controls (Or47bGal4 and UASdti) are significantly higher than MM or FF couples (p < 0.0001) (Fig. 3A, 3B). Three-way ANOVA on the sleep data of Or47b-ablated flies (Or47bGal4/UASdti) and its 2 genetic controls (Or47bGal4 and UASdti) showed a significant effect of strain (p < 0.0001), social group (p < 0.0001), phase (p < 0.0001), strain × social group (p < 0.0001), strain × phase (p < 0.05), social group × phase (p < 0.0001), and strain × social group × phase interaction (p < 0.0002). The total, daytime, and nighttime sleep of Or47b-ablated MF couples is similar to MM or FF couples (p > 0.05) (Fig. 3C, 3D). The total as well as nighttime sleep in control MF couples is lower than MM or FF couples (p < 0.0001), while their daytime sleep is significantly lower than MM couples (p < 0.0001) but similar to FF couples (p > 0.05) (Fig. 3C, 3D). This indicates that the differences in activity and sleep in Or47b-manipulated flies are due to the lack of Or47b receptor neurons and not due to some leaky expression of driver or UAS controls. These results suggest that Or47b ORNs are necessary for SSI. This is further confirmed in flies with electrically silenced Or47b neurons (Suppl. Fig. S2). The MF couples of Or47b-silenced flies show similar nighttime and marginally increased daytime activities compared to MM couples (p = 0.02 for MM couples; p > 0.05 for FF couples). Nighttime activity of control MF couples is significantly higher than MM or FF couples (p < 0.005) (Suppl. Fig. S2). Daytime activity of control MF couples is similar to FF couples (p > 0.05) but is greater than MM couples (p < 0.005) (Suppl. Fig. S2).

SSI is reduced in Or47b-ablated flies. The activity data (A, B) show that daytime and nighttime activities of MF couples (n = 24) of Or47b-ablated flies (Or47bGAL4/UASdti) are not different from MM (n = 24) or FF (n = 24) couples, while those of the control MF couples, Or47bGAL4 (n = 24) and UASdti (n = 16), are significantly greater than their respective MM or FF couples. Daytime sleep of MF couples of ablated flies is similar to MM or FF couples, while that of MF couples of controls is lower than MM but similar to FF couples (C, D). The nighttime sleep of MF couples of ablated flies is similar to MM or FF couples, while that of controls is lower than MM or FF couples (C, D). All other details are the same as in Figure 1.
Manipulation of other ORNs (Or65a, Or88a, and Or76d) involved in MF SSI has little or no effect on SSI-mediated circadian phenotype (Suppl. Figs. S3-S5). Furthermore, ablation of gustatory receptor neuron Gr5a, known for its regulation of feeding and food storage, has little or no effect on SSI (Suppl. Fig. S6).
SSI Is Male Driven
MF couples consisting of Or47b-ablated males and intact females (M−F+) are less active and sleep more in the night compared to couples with intact males and intact (M+F+) or ablated females (M+F−) (Fig. 4A, 4B), which suggests that intact olfactory ability in males is essential for SSI. Two-way ANOVA on the activity data of the 3 types of MF couples showed a significant effect of social group (p < 0.005) and social group × phase interaction (p < 0.006). Post hoc multiple comparison revealed that nighttime activity of M−F+ couples is significantly lower than M+F− or M+F+ couples (p < 0.0005), while there is no difference in their daytime activity (p > 0.05) (Fig. 4A, 4B). Two-way ANOVA on the sleep data showed a significant effect of social group (p < 0.0001), phase (p < 0.0001), and social group × phase interaction (p < 0.005). Post hoc multiple comparison revealed that total and nighttime sleep of M−F+ couples is significantly greater than M+F+ (p < 0.0005) or M+F− couples (p < 0.0005), while their daytime sleep does not differ (p > 0.05) (Fig. 4C, 4D).
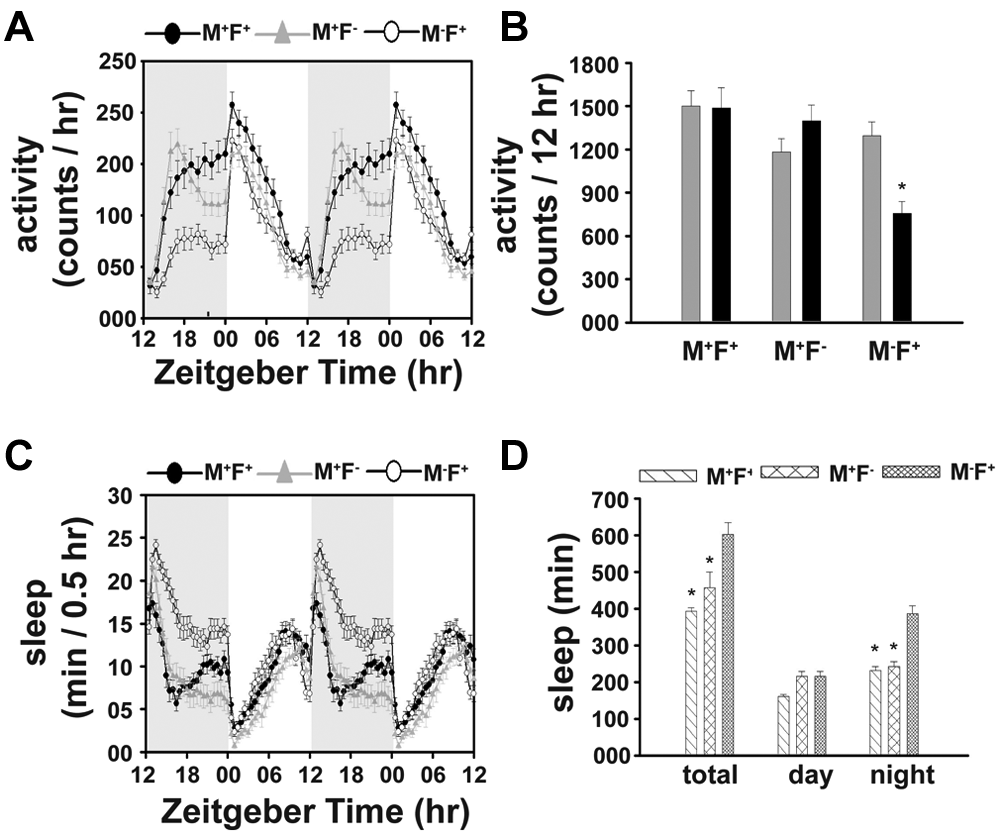
Olfactory ability in males is necessary for SSI. The activity data (A, B) show greater nighttime activity in MF couples (n = 24) consisting of intact (Or47bGAL4) males in the company of either intact or ablated females (M+F+ or M+F−) compared to couples (n = 24) with ablated males in combination with intact females (M−F+), while their daytime activity does not differ. Asterisks on the M−F+ bars indicate significant differences from the corresponding M+F+ or M+F− bars at p < 0.05 (B). Correspondingly, the nighttime sleep of M−F+ couples is lower than M+F− or M+F+ couples (C, D). All other details are the same as in Figure 1.
SSI-Mediated Nighttime Activity in MF Couples Free-Runs in DD
To find out whether SSI is controlled by circadian clocks, we studied the locomotor activity behavior of MF, MM, and FF couples of CS flies under DD conditions (Suppl. Fig. S7). The locomotor activity behavior of all 3 couples (MM, FF, and MF) is rhythmic and free-runs with a circadian period of 23.60 ± 0.54 hours (mean ± SD), 23.72 ± 0.38 hours, and 23.91 ± 0.48 hours, respectively (Suppl. Fig. S7). The activity of MM and FF couples is primarily restricted to subjective day, while that of MF couples spreads well into the subjective night. The average activity during subjective nights of MF couples (43.87% ± 3.31%) is significantly higher than MM (29.39% ± 1.85%) or FF (27.40% ± 1.95%) couples (p < 0.0005) (Suppl. Fig. S7). The average activity during subjective days of MF couples (56.12% ± 3.30%) is significantly lower (p < 0.0005) than MM (70.60% ± 1.85%) or FF couples (72.60% ± 1.95%) (Suppl. Fig. S7).
Silencing of CRYPTOCHROME (CRY)–Positive Clock Neurons Severely Affects SSI
We found that neither PDF nor PDF-positive neurons are necessary for the expression of SSI-mediated circadian phenotype (Suppl. Figs. S8 and S9). Next, we examined other clock neurons in the circadian network using 4.0 cryGAL4 driver. Three-way ANOVA on the activity data of cryGal4/UASdORKC, cryGal4/UASdORKNC, cryGal4, UASd ORKC, and UASdORKNC flies showed a significant effect of strain (p < 0.0001), social group (p < 0.0001), phase (p < 0.0001), strain × social group (p < 0.0001), and social group × phase interaction (p < 0.05). Nighttime activity of MF couples of all control groups showed an increase (p < 0.005) relative to MM or FF couples, which is associated with a decrease in sleep (p < 0.0001). Nighttime activity and sleep of CRY-silenced MF couples are similar to MM couples (p > 0.5) but are different than FF couples (p < 0.0005, data not shown). Daytime activity and sleep of silenced MF couples are similar to MM or FF couples (p > 0.5). Loss of SSI in MF couples is due to silencing of CRY-positive clock neurons and not due to some leaky expression of driver or UAS control because daytime and nighttime activities of MF couples of electrically silenced CRY-positive flies (cryGAL4/UASdORKC1) are less active (p < 0.0001) compared to MF couples of control cryGAL4/UASdORKNC1, cryGAL4, UASdORKC1/+, or UASdORKNC1/+ flies (Fig. 5A, 5B). Total, daytime, and nighttime sleep of silenced MF couples is significantly higher than couples of all control strains (p < 0.0001) (Fig. 5C, 5D). These results indicate that flies with electrically silenced CRY-positive neurons display reduced SSI.
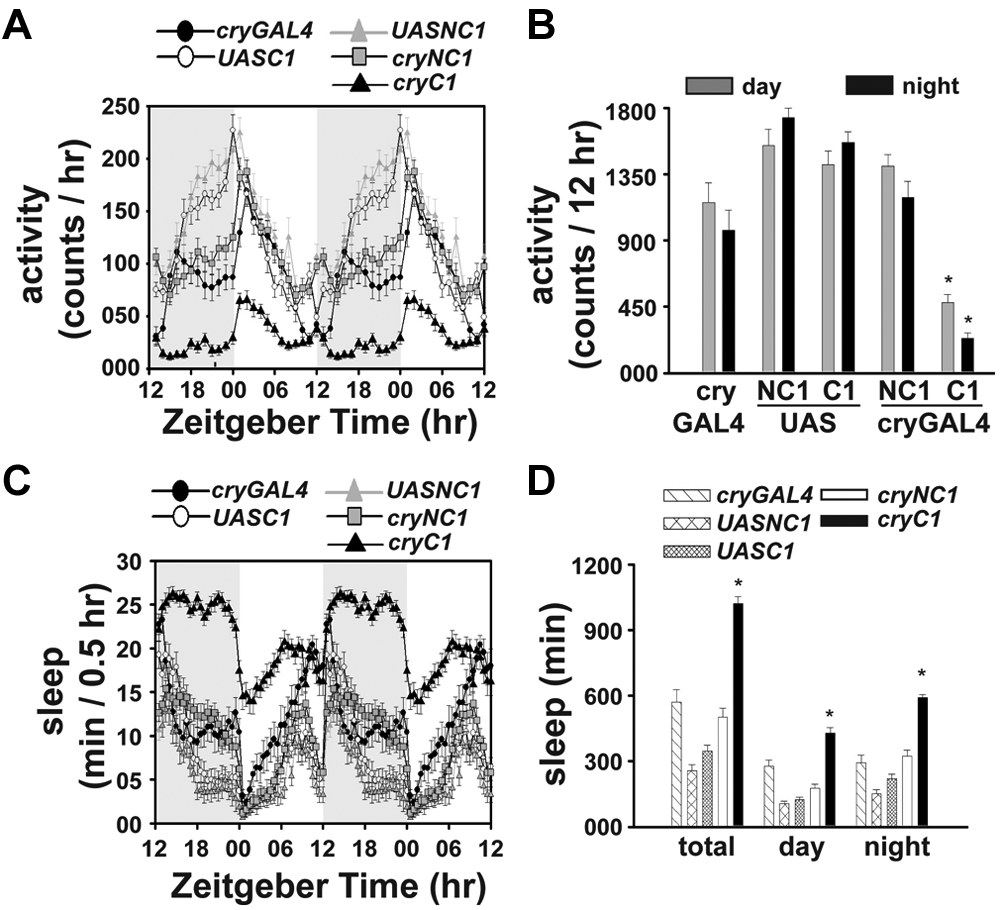
CRY-positive neurons are essential for SSI. The activity data (A, B) show lower nighttime activity in MF couples with electrically silenced CRY-positive neurons (n = 17) compared to controls (crydORKNC1: n = 20; crydORKC1: n = 20; cryGAL4: n = 16; UASdORKC1: n = 16; and UASdORKNC1: n = 16). The nighttime activity of CRY-silenced MF couples is significantly lower and nighttime sleep is greater than controls (C, D). Asterisks on the crydORKC1 (cryC1) bars indicate significant differences from the corresponding MF bars at p < 0.05 (B, D). All other details are the same as in Figure 1.
Discussion
Our results provide evidence for enhanced activity in MF couples through the evaluation of locomotor activity and sleep levels in pair-wise interacting flies. Given that the amount of activity and courtship are known to be positively correlated, this implies that heterosexual couples are involved in courtship at night (Hall, 1994), which is consistent with the fact that Drosophila courtship follows a daily rhythm with a distinct peak at night (Hardeland, 1972; Fujii et al., 2007). Enhanced activity and loss of sleep in hetero sexual couples are not artifacts of mathematical summation of male and female data because the mere addition of activity of solitary males and females yielded activity data that did not differ from the sum of activity of 2 same-sex individuals. SSI-mediated circadian phenotype persists in MF couples even after being housed together for 10 days, which suggests that SSI does not depend upon the actual act of mating; rather, it reflects courtship-associated locomotor activity. However, in our present study, it was not possible to find out which individual in the MF couple is active and which is not. In a separate study where the activity of males and females was individually recorded following SSI, we found that males display SSI-mediated circadian phenotypes, while females show an overall reduction in activity (Shahnaz Rehman Lone and Vijay Kumar Sharma, unpublished observations). The results of our study suggest that olfaction plays a critical role in SSI (Fig. 2); however, other sensory modalities may also be involved. Our results are in agreement with several previous studies, which showed that social interactions in Drosophila are olfaction mediated (Levine et al., 2002; Fujii et al., 2007; Krupp et al., 2008). Although it was shown previously that olfaction plays a key role in SSI, it was not clear which olfactory receptor(s) are involved in the communication of social signals for these interactions. The olfactory receptor Or67d, which is known to be involved in the reception of male-specific pheromone 11-cis-vaccenyl acetate (cVA) (Kurtovic et al., 2007), does not seem to be required for SSI (Suppl. Fig. S5). Although ablation of 2 other ORNs, Or65a (Suppl. Fig. S3) and Or88a (Suppl. Fig. S4), does not alter the sleep component of the SSI phenotype, it causes a measurable impact on activity, suggesting that these receptors may have some role to play in SSI. Manipulation of Or47b neurons is sufficient in blocking SSI (Fig. 3 and Suppl. Fig. S2), which suggests that Or47b ORNs are at the core of the olfactory signaling cascade involved in the communication of social signals during SSI. Reduction of SSI in Or47b-manipulated flies is associated with the loss of Or47b neurons and not due to any genetic artifact carried over from the parental strains because MF couples of parental control strains (Or47bGAL4 and UASdti) display a normal SSI-mediated circadian phenotype (Fig. 3). By driving the expression of GFP, we confirmed that the field of expression of the Or47b driver line in our experiment (data not shown) is similar to that reported previously (Fishilevich and Vosshall, 2005). We also confirmed the efficiency of the UAS lines (UASdti or UASdORKC1/NC1) by crossing them with either elavGAL4 or 4.0 cryGAL4 driver and then checking for pre-adult survivorship and the presence or absence of rhythmic behavior. As expected, ablated flies did not survive beyond the pre-adult stage, and silenced flies were arrhythmic under DD, quite unlike their unsilenced controls. SSI appears to be dictated by males because MF couples with Or47b-ablated males display a reduced SSI-mediated circadian phenotype compared to MF couples with wild-type males (Fig. 4). However, SSI may not be exclusively male driven because MF couples consisting of manipulated males continue to show some amount of nocturnal activity.
The locomotor activity behavior of MF couples is rhythmic under LD cycles, with activity spread over most parts of the night and first half of the day, while that of MM or FF couples is mostly restricted during the day. Under DD, the activity of all 3 types of couples follows circadian rhythmicity; activity of MF couples is confined to subjective night and the early part of subjective day, while that of same-sex couples is confined mostly to subjective day (Suppl. Fig. S7). The circadian neurotransmitter PDF and the PDF-positive neurons are not required for SSI because MF couples of flies with loss-of-function mutation in the circadian neurotransmitter PDF (pdf0) or with electrically silenced PDF-positive clock neurons exhibit a normal SSI-mediated circadian phenotype (Suppl. Figs. S8 and S9). This suggests that neither the circadian neurotransmitter PDF nor the PDF-positive neurons are required for the regulation of SSI, at least not under LD conditions. They may, however, become prominent for the sustenance of SSI-mediated circadian phenotype in DD. These results are consistent with the findings of a recent study (Hamasaka et al., 2010), which showed that neuropeptide F–positive LNd and PDF-positive clock cells are not necessary for hetero sexual interactions in Drosophila. Although it is likely that the evening activity peak of pdf0 and PDF-silenced flies is phase advanced, which in turn would have resulted in an increase of nighttime activity, a careful analysis of the activity data revealed that an increase in activity occurs throughout the night and is not merely restricted to the transition zone. While it appears that PDF-positive clock neurons may not be involved in SSI, CRY-positive neurons may have some role as CRY-manipulated flies show a significant reduction in SSI-mediated circadian phenotype. This was consistently seen in flies where CRY-positive neurons were manipulated either by electrically silencing CRY neurons by the expression of the voltage-gated dORKC1 channels (Fig. 5) or Kir2.1 channels (data not shown) or by the expression of the PER protein in the CRY neurons (data not shown). However, it is likely that reduced SSI in flies with electrically silenced CRY-positive neurons could be due to silencing of peripheral oscillators harboring CRY and not due to that of core clock neurons in the circadian network. While this work was in prepa ration, an article by Fujii and Amrein (2010) appeared, demonstrating that PDF-positive neurons regulate SSI, a result that is in striking contrast to the findings of our study. The only way we can explain this is that in the Fujii and Amrein (2010) study, courtship ratio was used to estimate SSI, whereas we used amount of activity and sleep. Courtship ratio index is estimated in terms of the male’s proximity to the female over a 48-hour period divided by its proximity during a 4-hour period during dusk, and hence, this method does not account for the general activity of males and females. Moreover, in our study, we measured locomotor activity without accounting for whether males and females were in close proximity. Nevertheless, the results of our study are consistent with one of the results of Fujii and Amrein (2010), where the authors found no detectable effect of loss of clock function in the PDF-expressing neurons (pdfGAL4/UASCYCΔ) on courtship ratio.
In summary, the results of our study suggest that MF couples of several wild-type strains of flies display an SSI-mediated increase in activity and sleep loss compared to MM and FF couples. This behavior involves olfactory means of communication between males and females, in which males play a greater role. Among the olfactory receptors, Or47b receptor neurons are necessary for the regulation of SSI; however, Or65a and Or88a ORNs may also participate, albeit as second fiddles. Other sensory modalities and ORNs may also be a part of the SSI mechanisms but only in a secondary role. SSI is clock controlled; while the circadian neurotransmitter PDF and functional clocks in LNv are not necessary for the expression of its circadian phenotype, clocks in the peripheral Or47b ORNs and DN neurons seem to be critical.
Footnotes
Acknowledgements
The authors thank Barry Dickson, Todd Holmes, and Amita Sehgal for generous gifts of fly stocks; Vijayraghavan and (the late) Veronica Rodrigues for permitting them to use their fly stock facility; Gajendra for generously supplying them with fly stocks; Nisha and Pankaj for helping them during the experiments; Sheeba Vasu for helpful comments on the article; and JNCASR for financial support. They thank the 3 anonymous reviewers for carefully reading the article and suggesting improvements.
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
