Abstract
Sleep is a fundamental biological rhythm involving the interaction of numerous brain structures and diverse neurotransmitter systems. The primary measures used to define sleep are the electroencephalogram (EEG) and electromyogram (EMG). However, EEG-based methods are often unsuitable for use in high-throughput screens as they are time-intensive and involve invasive surgery. As such, the dissection of sleep mechanisms and the discovery of novel drugs that modulate sleep would benefit greatly from further development of rapid behavioral assays to assess sleep in animal models. Here is described an automated noninvasive approach to evaluate sleep duration, latency, and fragmentation using video tracking of mice in their home cage. This approach provides a high correlation with EEG/EMG measures under both baseline conditions and following administration of pharmacological agents. Moreover, the dose-dependent effects of sedatives, stimulants, and light can be readily detected. This approach is robust yet relatively inexpensive to implement and can be easily incorporated into ongoing screening programs to provide a powerful first-pass screen for assessing sleep and allied behaviors.
Sleep is perhaps the most familiar biological rhythm, arising from multiple brain structures and neurotransmitter systems. This coordinated neural activity in turn drives alternating patterns of behavior characterized by changes in rest and activity, body posture, and responsiveness to stimuli (Tobler 1995). However, despite decades of research, many of the mechanisms underlying this complex behavior remain poorly understood (Tafti and Franken 2002; Winrow et al., 2009). Although sleep and wakefulness exhibit differences in both behavioral and neural parameters, the primary measures used to define sleep in mammals have focused upon neural surrogates. Electroencephalogram (EEG) and electromyogram (EMG) describe sleep on the basis of rapid eye movement (REM) and non-REM (NREM) states. This gold standard approach of classifying sleep involves the implantation of cranial electrodes from which recordings are made via either tethered- or telemetry-based systems, enabling assessment of sleep structure, and the analysis of EEG power spectra for different sleep-wake states (Lo et al., 2004; Weiergraber et al., 2005). While sleep-wake behaviors have been shown to persist in the absence of the neocortex (Vanderwolf et al., 1978), EEG analysis has undoubtedly led to important advances in our understanding of sleep (e.g., Chemelli et al., 1999; Cirelli et al., 2004; Franken et al., 2001; Maret et al., 2005; Tobler et al., 1996). However, the application of EEG recordings to high-throughput studies has been limited (Winrow et al., 2009). In addition, the need for surgical implantation of electrodes makes the EEG/EMG approach time-consuming, invasive, often expensive to implement, and generally unsuitable for programs involving screening large numbers of animals. As a preliminary screen, the use of simple behavioral definitions of sleep including duration, latency, and number of sleep episodes (fragmentation) would provide a valuable first-pass evaluation of sleep-wake behavior, prior to in-depth EEG studies.
Several attempts to address the requirement for a first-pass screen for sleep-wake behavior have been developed. Storch et al. (2004) used small magnets implanted subcutaneously in mice, and activity was determined by the movement of the magnet relative to a sensor beneath the cage. Although this approach produced a reliable low-resolution measure of activity-rest patterns, it still necessitated surgical intervention. A noninvasive approach was developed by Flores et al. (2007) based upon monitoring behavioral activity through piezoelectric sensors positioned on the cage floor (Megens et al., 1987). Sleep states were classified using a novel pattern recognition algorithm to identify regular motions associated with respiration when the animal assumed a specific sleep posture, whereas wake was characterized by large-amplitude spikes in the piezoelectric signal (Flores et al., 2007). This system was able to distinguish sleep from wake with up to a 0.95 correlation when compared with EEG analysis. Although this approach has been refined for high-throughput screens (Donohue et al., 2008), the requirement of specialized equipment and custom software may limit the application of this method. A major advance in the area of high-throughput sleep scoring was made by Pack and colleagues in 2007, who used video monitoring combined with an object recognition algorithm to evaluate periods of sustained immobility as a surrogate of EEG/EMG-defined sleep (Pack et al., 2007). As many sleep labs routinely use video recordings to assist in sleep scoring, this approach builds upon infrastructure that is often already in place and can also be used to evaluate other related behaviors. Importantly, Pack et al. identified that periods of continuous inactivity 40 sec or longer were highly correlated with sleep in mice. Monitoring over a 24-h period, the investigators showed that there was a 92% agreement with tethered EEG/EMG recordings (Pack et al., 2007).
Although the studies described above provide strong evidence that high-throughput behavioral assays can accurately identify EEG/EMG definitions of sleep, these methods have not become widely adopted for first-pass sleep-wake assessment. One explanation for this may be the need for proprietary software, making these methods inaccessible. Moreover, these techniques have not been validated for assessing sleep-wake behavior under different experimental paradigms (e.g., pharmacological and environmental manipulation). In recent years there have been major advances in the development of image-analysis software for a broad range of commercial and scientific tasks, including the assessment of multiple behaviors in mice (Crawley 2008). As a result, high-throughput assessment of complex behaviors from video data using commercially available behavioral software offers an ideal solution for preliminary assessment of sleep-wake behavior.
Here we describe a simple but robust automated technique to evaluate sleep-wake behavior in mice based upon video monitoring with subsequent analysis using commercially available software. This approach provides an extremely high agreement (correlation coefficient >0.94) with EEG/EMG recordings under both baseline conditions and following administration of pharmacological agents. The flexibility of this method is illustrated by showing the dose-dependent effects of sedatives, stimulants, and light on sleep-wake behaviors. Research on biological rhythms typically focuses on locomotor activity as a readily measured circadian behavior. However, the absence of locomotor activity is not equivalent to sleep, and the lack of accessible assays can result in circadian research failing to address this fundamental biological rhythm. The approach described here provides a powerful first-pass screen for assessing sleep-wake behaviors, providing a valuable additional component to circadian phenotyping screens.
Methods
Animals
Adult male C57BL/6 mice (3-9 months old, Harlan, UK) were used throughout the video-tracking and sleep validation studies. Mice were entrained under a standard 12:12 h light-dark cycle for a minimum of 14 days and were given at least 72 h to habituate to a home cage prior to the start of all studies. Under these conditions, light onset was designated zeitgeber time (ZT) 0 and dark onset as ZT12. Ambient temperature was maintained at 22 ± 2 °C, and food and water were available ad libitum. All studies were carried out under Home Office license and in accordance with the Animal (Scientific Procedures) Act 1986, UK.
Video Equipment and Experimental Setup
For all studies mice were individually housed in plastic cages (25 x 30 cm) and isolated from possible confounding external stimuli using a system of light-tight ventilated chambers (manufactured by Lanview, Portsmouth, UK, and ventilated with 120-mm axial fans, 4412 F/2GLL-515, ebm-papst, Chelmsford UK). Light was provided using a custom overhead fiber optic light source system (Schott AG, Mainz, Germany) with light levels ~400 lux when measured at the bottom of the cage (Figure 1A). A 12-channel randomized fiberoptic cable (Schott AG, Mainz, Germany) was used to provide an even distribution of light over the cage area and was necessary to prevent interference with telemetry receivers caused by fluorescent lighting. Light-dependent resistors connected to a ClockLab interface (Actimetrics, Wilmette, IL) were used to record the light-dark conditions within the light-tight chamber at all times. Miniature near infrared (NIR) video cameras (Sentient Mini-night vision CCTV camera, Maplin, UK) were mounted above each cage to permit the recording of mice under either light or dark (NIR) conditions. Cameras were fitted with a wide angle lens (130°, 2.8 mm, Open Twenty Four Seven Ltd, UK) to enable the whole cage area to be viewed. Cages were positioned approximately 30 cm below the camera, which was centered over the area containing the mouse. In some circumstances, glare from the NIR LEDs in the cameras occurred from the cage bars, and this was reduced by use of tracing paper as a diffuser. Cameras were connected to a 16-channel digital hard-drive recorder (Model VXM4B-16, Videcon, PLC), and video data were recorded at 25 frames per second with a resolution of 704 x 576 pixels. To maintain video tracking throughout the study, mice were prevented from entering an area hidden to the camera using custom-made perspex blocks (23 12 × 3.2 cm, Aquarius Plastics, Surrey, UK). Video tracking was performed using ANY-maze (Version 4.5, Stoelting, US). Full details of analysis protocols and software settings are provided as supplementary information.
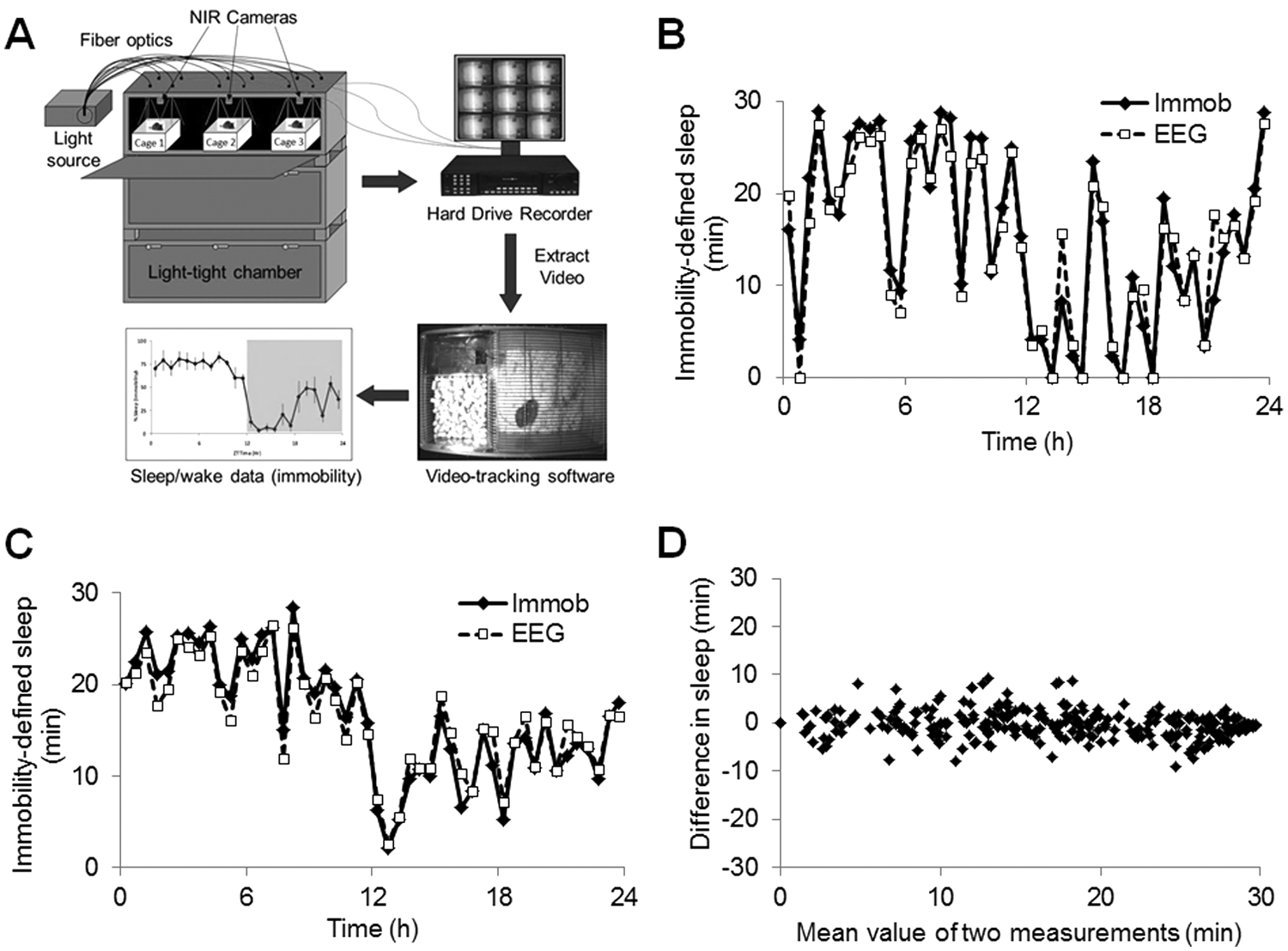
Validation of immobility-defined sleep versus EEG/EMG-defined sleep. (A) Experimental setup for the noninvasive assessment of sleep using digital video analysis. Mice were housed in light-tight chambers with light provided by a halogen light source through fiber optic cables. Miniature NIR cameras were mounted above each individual cage and were connected to a digital hard-drive recorder and monitor. Video data were extracted from the hard-drive recorder and analyzed offline using commercially available video-tracking software (ANY-maze, Stoelting). (B) Comparison of sleep assessed by digital video analysis (“Immob”) compared with EEG/EMG-defined sleep (“EEG”) across a 24-h baseline period under a 12:12 h light-dark cycle for an individual mouse. (C) Average immobility-defined sleep determined using digital video analysis and EEG/EMG in 6 mice. (D) Bland-Altman plot displaying the mean difference (min) between the 2 methods used to assess sleep using the individual 30 min values from B. The estimated bias across the 24-h period was +0.24 min with a standard deviation of 2.93 min. The Bland-Altman 95% limits of agreement were −5.50 to +5.98 min. Data points were analyzed in 30-min periods to more accurately represent the polyphasic nature of sleep in mice. A similar high correlation was observed when analyzed in 1-h periods. In all cases sleep, per 30-min interval is shown and sensitivity was set at 95%.
EEG/EMG Recording and Analysis
Mice anaesthetized under isoflurane (3-5% for induction, 1-3% for maintenance) were implanted with EEG/EMG telemetry transmitters (TL11M2-F-20, EET, Data Sciences International, St. Paul, MN) as described previously (Lupi et al., 2008). Mice were allowed at least 3 weeks to recover before studies were undertaken. All implanted mice remained healthy and gave clear EEG/EMG recordings allowing the discrimination of sleep-wake stages. EEG and EMG data were transmitted to a radio receiver (RPC-1, Data Sciences International) placed underneath each cage. Signals were then routed via a data exchange matrix to a PC running Dataquest A.R.T. software (Version 3.01). The EEG and EMG data were continuously sampled using DSI Dataquest Gold acquisition software at 500 Hz, with a 100-Hz filter cutoff. EEG and EMG signals were band-pass filtered (0.5-35 Hz for EEG and 80-100 Hz for EMG), and sleep-wake stages were scored offline as wakefulness, NREM sleep, and REM sleep in 10-sec epochs using a semiautomated approach. The sleep scoring procedure consisted of an initial automated step using SleepSign software (Kissei Comtec, Nagano, Japan) followed by a review of all epochs by an experienced sleep scorer. Total sleep for the recording period was determined by the addition of all NREM and REM epochs.
Assessment of Baseline Sleep Determined by Simultaneous EEG/EMG and Video Analysis
Mice implanted with EEG/EMG electrodes were recorded simultaneously using video cameras in their home cage. Once mice were entrained and acclimatized to the cage environment, EEG, EMG, and video data were recorded for a 24-h period (under a 12:12 h light-dark cycle). Offline analysis of the video data determined the optimum immobility detection settings. In addition to the duration of immobility required to accurately identify sleep (as described by Pack et al., 2007), the definition of immobility also depends upon the percentage of the area of the tracked animal that is required to be immobile. If this is set too low, false positives will occur when the animal is stationary but not asleep, for example, when grooming or feeding. If this is set too high, false negatives will occur due to small movements associated with respiration and muscle twitches during sleep. To address this issue of sensitivity, the immobility sensitivity setting within the software was adjusted in increments from 50% to 100% and compared the total amounts of sleep derived from EEG/EMG classification. The baseline 24-h period was analyzed in both 1-h and 30-min segments to determine the correlation of the method at higher and lower resolutions.
Effect of Zolpidem on Immobility-defined Sleep in Comparison with EEG/EMG Analysis
Three different doses of zolpidem (10, 5, or 1 mg/kg i.p., Tocris, Bristol, UK) dissolved in physiological saline (containing 0.3% Tween 80, Sigma-Aldrich) or vehicle were administered to 3 separate groups of mice (n = 6 for each dose/vehicle) at ZT14 (2 h after lights off). Each dose of zolpidem was administered together with an independent vehicle, and all mice were dosed using a randomized design with each mouse receiving a single injection. All drug solutions were freshly prepared on the day of the experiment and injected intraperitoneally at a volume of 10 mL/kg body weight with administration typically completed in 5 min for 6 mice. Upon dosing, mice were immediately returned to their home cage. Video data were analyzed for a 1-h baseline period and up to 5 h post administration. In a separate validation study, mice (n = 4) implanted with EEG/EMG electrodes were administered either zolpidem (10 mg/kg, i.p.) or vehicle at ZT14. Simultaneous EEG, EMG, and video data were recorded for a period of 6 h including 1 h prior to drug administration. A time course of sleep duration for both EEG/EMG- and video-determined sleep was then calculated at 10-min intervals to provide a high-resolution measure of sleep induction. For all studies involving sleep latency, sleep onset was defined as the time from the point of injection to the period of 2 min of continuous immobility.
Effect of Caffeine on Immobility-defined Sleep
Mice were administered either caffeine (15 mg/kg, dissolved in physiological saline, n = 6, Tocris, Bristol UK) or vehicle (0.9% w/v NaCl, n = 6) intraperitoneally at ZT0 (lights onset). Upon dosing, mice were returned to their home cage, and video data were analyzed for a 1-h predrug baseline period for up to 5 h post administration. The effect of caffeine on immobility-defined sleep onset and duration together with the number of immobile episodes was then determined.
Acute Effects of Light on Immobility-defined Sleep
To evaluate the robustness of our methods further, we assessed the dose-dependent effects of light on sleep during the habitual wake period. Mice (n = 6) were entrained to a 12:12 h light-dark cycle and administered 3 different 2-h light pulses of increasing intensity (0.7, 70, and 700 lux) in a fully randomized order at ZT14. These were performed on different experimental days with a minimum of 6 days between light pulse exposures. Video data were analyzed 1.5 h prior to light administration for a total period of 4.5 h.
Statistical Analysis
Analysis of agreement was performed using the Bland-Altman statistical method (Bland and Altman, 1986). The 24-h baseline sleep-wake period determined by digital video and EEG/EMG analysis was divided into 24 x 1-h segments. To enable a higher resolution analysis of the data, this baseline period was also analyzed in 48 × 30-min segments to more accurately represent the polyphasic nature of sleep in rodents. We determined the mean bias by calculating the difference between estimates of sleep based on immobility and that defined by EEG/EMG analysis. Immobility-defined sleep onset latency data were analyzed using a Student t test with p < 0.05 considered statistically significant. Time course analysis and immobile episodes were assessed using a 2-way ANOVA (factors: time and treatment). Light pulse data were analyzed for the effect of light intensity on immobility-defined sleep during the first 45 min of light exposure using a 1-way ANOVA. Where applicable, Bonferroni post hoc tests were used.
Results
Baseline Sleep: EEG/EMG versus Video Analysis
A high level of agreement was achieved in the assessment of sleep using digital video analysis compared with simultaneous EEG/EMG-defined sleep in individual mice (Figure 1B) and for the average values derived from 6 mice across a 24-h baseline period (Figure 1C). Initially the data were compared using hourly values for sleep across the 24-h baseline (data not shown). The average EEG/EMG determined sleep per 1-h interval across a 24-h period was 32.71 ± 13.48 min. By comparison, sleep estimated by digital video analysis was 33.19 ± 15.41 min, resulting in a bias of +0.48 min (95% confidence interval +3.41 to −4.37 min, correlation coefficient 0.94). Using this approach with a temporal resolution of 30 min demonstrated that digital video analysis still provided a remarkable fit (correlation coefficient 0.94) compared with EEG/EMG recordings under baseline conditions (Figure 1B and 1C). When comparing 2 methods of measurement, the difference between the approaches may depend on the magnitude, producing a bias at high or low values. As such, we used the method of Bland and Altman (1986) to compare these 2 methods (Figure 1D). Using this approach, the estimated bias over 24 h was 0.24 min. The Bland-Altman 95% limits of agreement were −5.50 to +5.98 min. The sensitivity of immobility scoring by ANY-maze (Stoelting) was also evaluated by comparing sleep estimated by this method to that determined using EEG/EMG analysis while varying the immobility sensitivity value from 50% to 100%. We identified that the optimal sensitivity value producing the smallest bias was 95% giving a correlation of 0.94 (Supplementary Figure S1).
Effects of Zolpidem: EEG/EMG versus Video Analysis
Pharmacologically induced sleep using the hypnotic zolpidem was compared with simultaneous EEG/EMG and video analysis. For these studies, the recording period and subsequent analysis was undertaken using higher resolution (10-min intervals). There was excellent agreement with video versus EEG/EMG-defined sleep even at this resolution (Figure 2). Again, Bland-Altman statistical analysis demonstrated no systematic bias (+0.06 for zolpidem, +0.15 min for vehicle).
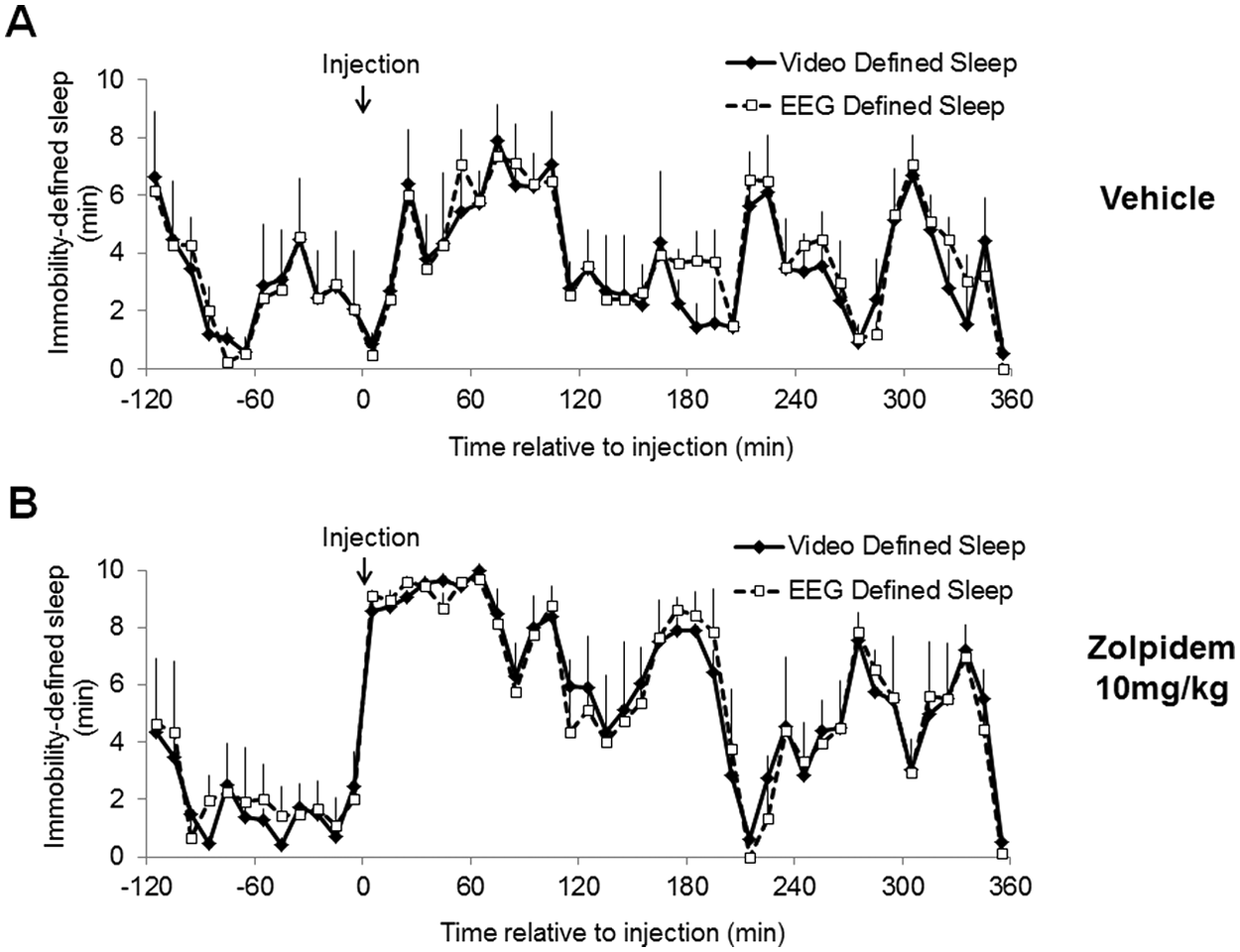
Comparison of sleep assessed using digital video analysis with EEG/EMG-defined sleep after administration of (A) vehicle (0.9% w/v NaCl) or (B) zolpidem (10 mg/kg). Treatments were administered at ZT14. Data points were analyzed in 10-min periods and represent mean values ± SEM, n = 4. Using the Bland-Altman method to assess agreement for postinjection values after zolpidem treatment resulted in a bias of +0.06 min with a standard deviation of 1.25 min (2.52 to −2.40 min, correlation coefficient 0.94). For vehicle treatment the bias was +0.15 min with a standard deviation of 1.02 min (Bland-Altman limits of agreement: 2.15 to −1.85 min, correlation coefficient 0.95). In all cases, sleep per 10-min interval is shown and sensitivity was set at 95%.
Dose Effect of Zolpidem on Sleep
We determined the sensitivity of the video analysis approach in estimating sleep by evaluating the changes in sleep after 3 different doses of zolpidem. Doses of 10 mg/kg and 5 mg/kg produced significant increases in sleep duration (Figure 3A and B). Vehicle administration also produced a smaller increase in sleep, most likely due to the effects of handling/injection. While 1 mg/kg did not produce a significant increase in sleep duration when analyzed in 1-h bins, it is clear from an analysis of the entire time course that 1 mg/kg zolpidem does increase immobility-defined sleep, although to a much smaller extent (to around 75%) and for a shorter period (~30 min post administration) compared with higher doses (Figure 3C). Using 30-min intervals, 1 mg/kg clearly shows a significant increase in sleep duration when compared with vehicle (unpaired Student t test, p < 0.001). Moreover, the effect of 1 mg/kg zolpidem is immediately evident from analysis of immobility-defined sleep onset latency, which was highly significant with all doses (Table 1). Interestingly, the higher doses (10 and 5 mg/kg) both led to a second period of increased immobility-defined sleep approximately 140 min post administration, which was not evident with 1 mg/kg. All 3 doses of zolpidem increased the number of immobile episodes during the first hour post administration (Supplementary Figure S2). Both the 5 and 10 mg/kg doses also significantly increased immobile episodes 2 to 3 h post injection.
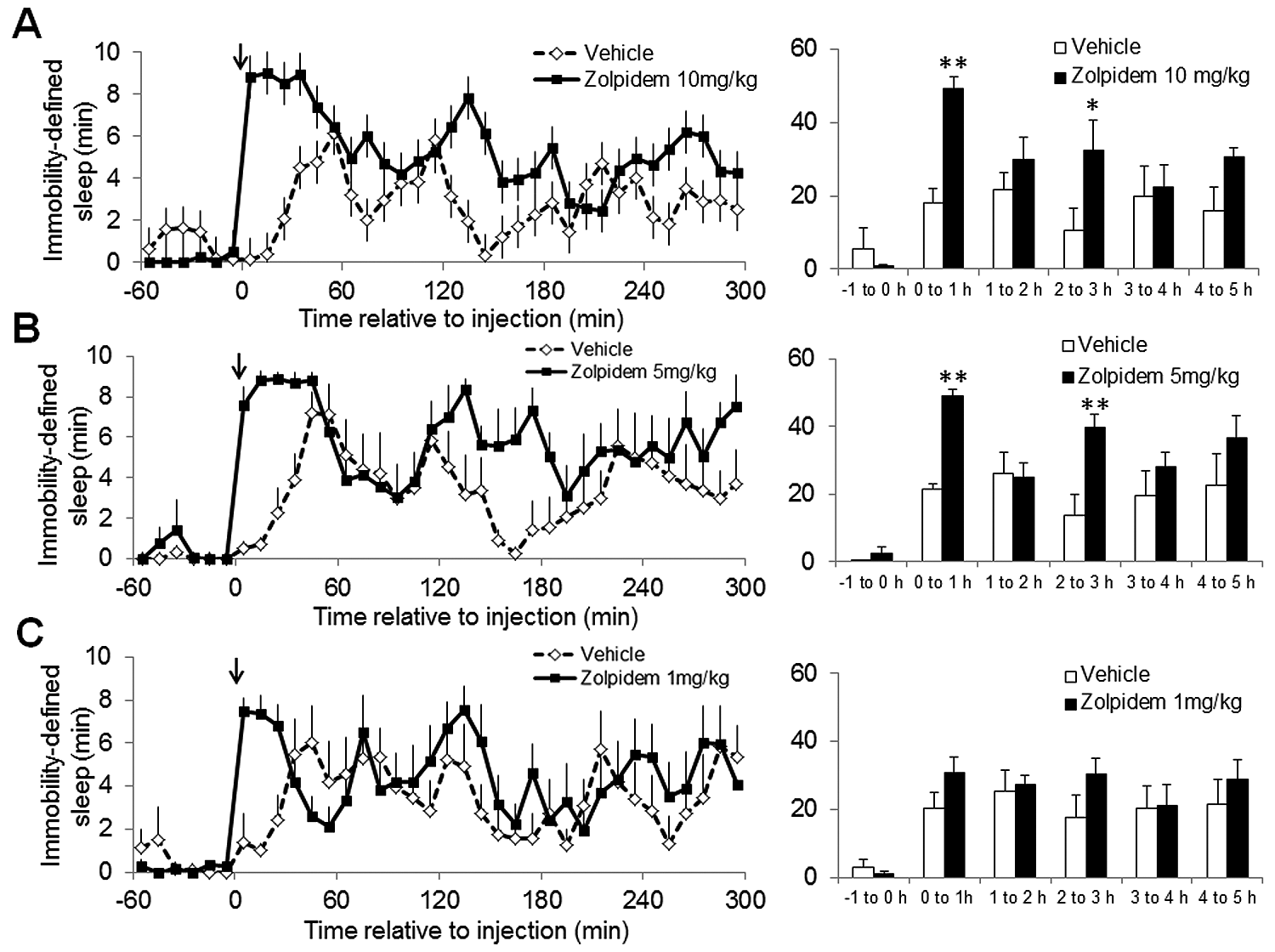
Evaluation of the effect of 3 doses of zolpidem (solid lines) or vehicle (dashed lines) on immobility-defined sleep determined using digital video analysis. Zolpidem was dosed i.p. at (A) 10 mg/kg, (B) 5 mg/kg, and (C) 1 mg/kg at ZT14. Zolpidem dosed at 10 and 5 mg/kg resulted in a significant increase in the amount of immobility-defined sleep (to around 90%) compared with vehicle (F1,10 = 22.2, p < 0.001 at 10 mg/kg; F1,10 = 11.8, p < 0.01 at 5 mg/kg). In addition, there were significant changes in sleep duration over the time course studied with all doses tested (F5,50 = 6.1, p < 0.001 at 10 mg/kg; F5,50 = 11.7, p < 0.001 at 5 mg/kg; F5,50 = 7.7, p < 0.001 at 1 mg/kg). At 10 mg/kg and 5 mg/kg there was also a significant interaction, with differences in sleep over the time course being dependent upon the vehicle or zolpidem treatment (F5,50 = 2.6, p < 0.05 at 10 mg/kg; F5,50 = 3.1, p < 0.05 at 5 mg/kg). A dose of 1 mg/kg zolpidem did not produce a significant effect on sleep duration from the vehicle. The solid black arrows indicate the time of drug/vehicle administration. Data points represent mean vales ± SEM for 6 mice. Bar graphs depict the total duration of immobility-defined sleep in hourly intervals: *p < 0.05 and **p < 0.01. Note: A separate vehicle condition was used for each of the doses of zolpidem. In all cases, sleep per 10-min interval is shown and sensitivity was set at 95%.
Dose-dependent effects of zolpidem on immobility-defined sleep onset latency.
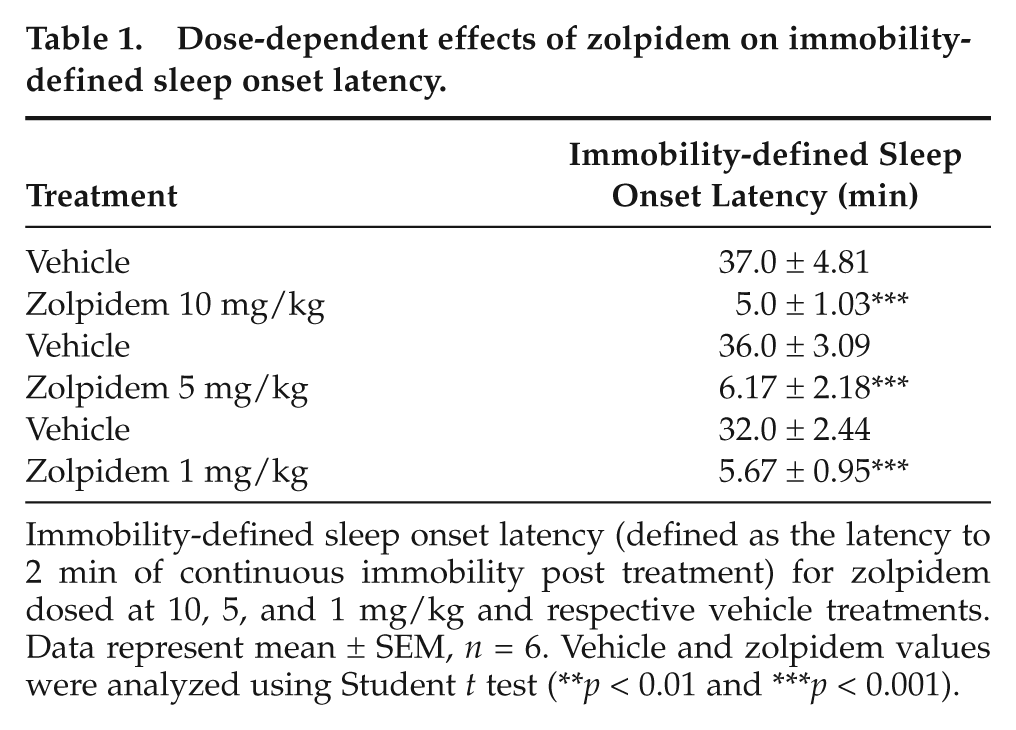
Immobility-defined sleep onset latency (defined as the latency to 2 min of continuous immobility post treatment) for zolpidem dosed at 10, 5, and 1 mg/kg and respective vehicle treatments. Data represent mean ± SEM, n = 6. Vehicle and zolpidem values were analyzed using Student t test (**p < 0.01 and ***p < 0.001).
Effect of Caffeine on Sleep
The effects of caffeine (15 mg/kg) on immobility-defined sleep were also determined using digital video analysis. There was a significant change in sleep duration over the time course studied (F35,360 = 6.0, p < 0.001), as would be expected for the beginning of the light period (Figure 4A). In addition, mice administered caffeine exhibited a significant reduction in the amount of sleep during the first 120 min post administration (F1,360 = 56.8, p < 0.001). The amount of sleep did not return to levels comparable to the vehicle-treated mice until ~180 min post caffeine administration (Figure 4A). There was a significant interaction of treatment and time, with caffeine and vehicle producing different temporal effects (F35,360 = 3.4, p < 0.001). Caffeine was also demonstrated to significantly decrease the number of immobile episodes (F1,10 = 5.0, p < 0.05) for up to 2 h post administration (Figure 4B) and significantly delayed immobility-defined sleep latency (111.17 ± 24.35 min, unpaired Student t test p < 0.01) compared with mice administered vehicle (19.67 ± 1.89 min) (Figure 4C).
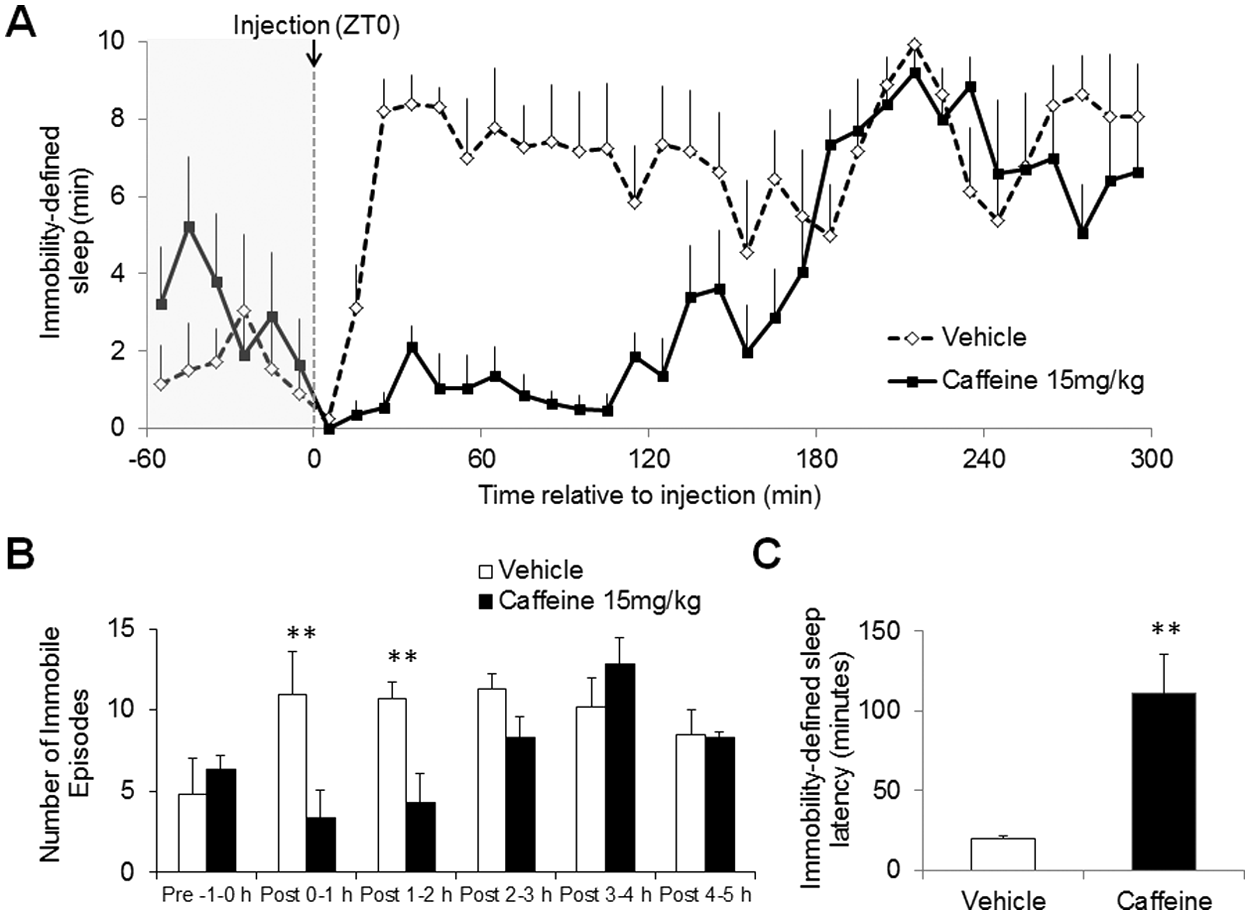
Effect of caffeine (15 mg/kg) administered at ZT0 on immobility-defined sleep assessed using digital video analysis. (A) Time course of drug administration displaying vehicle (open circles) and caffeine (closed circles) dosed i.p. at ZT0 (10-min resolution). There was a significant change in sleep duration over the time course studied (F35,360 = 6.0, p < 0.001), due to the onset of the light phase. In addition, mice administered caffeine exhibited a significant reduction in the amount of sleep during the first 120 min post administration (F1,360 = 56.8, p < 0.001). There was a significant effect of caffeine from 25 min to 105 min (post hoc Bonferroni test p < 0.05). The amount of sleep did not return to levels comparable to the vehicle-treated mice until ~180 min post caffeine administration. (B) The number of immobile/sleep episodes represented as hourly values. (C) The latency to the first immobile period (2 min of continuous immobility). **p < 0.01 vehicle vs. caffeine. Data points represent mean values ± SEM, n = 6. In all cases, sleep per 10-min interval is shown and sensitivity was set at 95%.
Effect of Light on Sleep
Light pulses of increasing intensity produced a dose-dependent increase in immobility-defined sleep in mice during the habitual sleep period (Figure 5). A 1-way ANOVA was used to test for the acute effects of light on total immobility-defined sleep across the first 45 min of light exposure. This identified a strong intensity dependent sleep-promoting response to light (F2,15 = 26.56, p < 0.001). Decreasing light intensity from 700 to 70 lux did not reduce the initial peak of immobility-defined sleep induction but did reduce the duration of the sleep-producing effect. The 0.7-lux light pulse resulted in a relatively small transient increase in immobility-defined sleep at approximately 20 min into the light exposure with a second small increase in sleep induction occurring at 60 min. For all 3 light intensities there was a gradual decrease in immobility-defined sleep at approximately 70 min until the end of the light pulse.
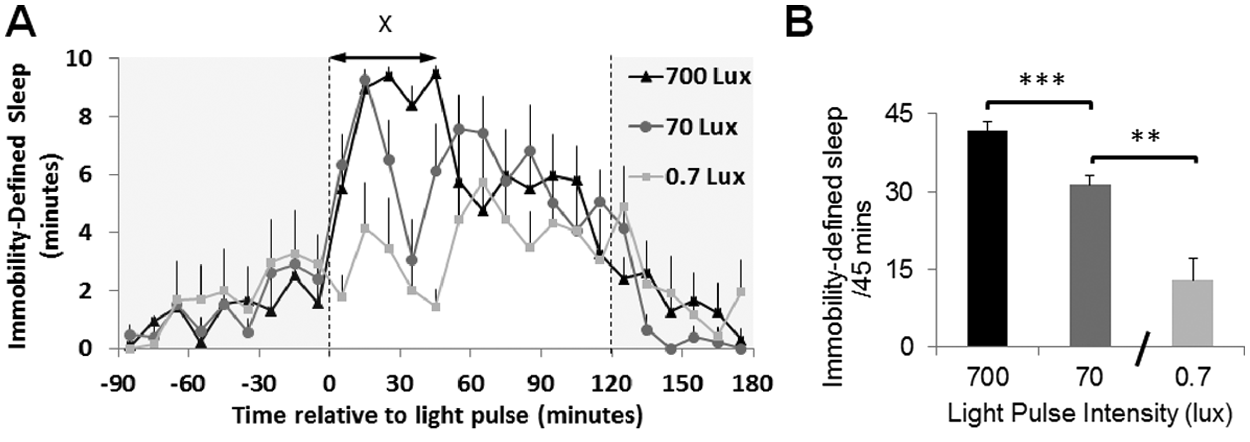
Evaluation of the effect of 3 different intensity light pulses on immobility-defined sleep assessed using digital video analysis. (A) Time course of a 2-h light pulse administered at ZT14 including the baseline and postlight pulse periods for 0.7, 70, and 700 lux light. Data points are plotted in 10-min intervals as mean values ± SEM, n = 6. One-way ANOVA was used to test for the acute effects of light on total immobility-defined sleep across the first 45 min of light exposure. This identified a strong intensity dependent sleep-promoting response to light (F2,15 = 26.56, p < 0.001). (B) Bar graph representing the total amount of immobility-defined sleep for the 3 different light intensities in the first 45 min of light exposure depicted by X on A. Data were compared using a 1-way ANOVA with a Bonferroni post hoc test. ***p < 0.001 700 lux vs. 0.7 lux and **p < 0.01 70 lux vs. 0.7 lux. In all cases, sleep per 10-min interval is shown and sensitivity was set at 95%.
Discussion
Despite the dramatic changes in sleep and wakefulness over the 24-h cycle, research on biological rhythms has often focused on locomotor activity rather than sleep-wake timing as a behavioral output of the circadian system. Here we describe a high-throughput system that can provide a rapid yet robust assessment of sleep-wake behavior in mice. This approach was compared with simultaneous EEG/EMG determined sleep under baseline conditions and following administration of the sedative-hypnotic zolpidem. Defining sleep as a period of extended immobility (>40 sec) during which 95% or more of the area of the animal is stationary, we obtained a 0.94 correlation with simultaneous EEG/EMG-defined sleep (Figure 1C) with no significant systematic bias. This method is able to determine not only total sleep duration per time interval but also the sleep onset latency and number of immobile episodes. Rather than a gross neural correlate of sleep, this approach relies upon the pronounced differences in behavior that define sleep. A key advantage of this automated system is that immobility detection uses commercially available software to automatically determine a minimum duration of inactivity, enabling the simultaneous off-line analysis of up to 16 animals (per computer). As well as using the 40 sec or more of immobility identified by Pack and colleagues (2007), we also determined the percentage area of the mouse required to remain immobile for the accurate determination of sleep. By varying the sensitivity of this component of immobility detection and comparing this to EEG/EMG-defined sleep, we identified that the optimum sensitivity setting was 95% (Supplementary Figure S1). Using a sensitivity setting greater than 95% resulted in an underestimation of sleep, whereas sensitivity settings below 95% resulted in an overestimation of sleep. Using a 95% sensitivity criteria over a 24-h cycle (12:12 h light-dark), the estimated bias of the method compared with EEG/EMG analysis was just +0.48 min/h (Supplementary Table S1).
As well as evaluating sleep under baseline conditions, we assessed the suitability of this technique in measuring the effects of pharmacological compounds on sleep. Following the administration of zolpidem, a non-benzodiazepine hypnotic, immobility-defined sleep gave a high correlation with EEG/EMG-defined sleep (0.94). The bias was again small, just +0.06 min (SD, 1.25 min) when compared against sleep assessed by EEG/EMG (Figure 2). This agreement was striking particularly as the data were analyzed at a higher resolution of 10-min intervals. To evaluate dose-dependent effects, 3 doses of zolpidem (10, 5, and 1 mg/kg) were measured using video tracking, each in a different group of animals. This produced highly consistent sleep-wake profiles, demonstrating the reproducibility of the system (Figure 3). The method was also able to differentiate between the effects of relatively high and low doses of zolpidem on sleep. The higher doses of zolpidem (5 and 10 mg/kg) differed considerably from the 1 mg/kg dose in terms of the initial amount of immobility-defined sleep during the first hour and the number of immobile episodes and sleep amount 2 to 3 h post injection. Only very subtle differences were evident between the effects of 5 and 10 mg/kg zolpidem on immobility-defined sleep, suggesting these doses produced near-saturating responses. Previous studies assessing the effects of zolpidem on sleep have used EEG analysis; however, such studies typically administer zolpidem in the light period when sleep pressure is high in nocturnal animals (Alexandre et al., 2008; Kopp et al., 2004), making direct comparisons difficult. Despite this, Alexandre et al. (2008) reported no difference between the effects 10, 5, and 2 mg/kg zolpidem on NREM sleep during the first 2 h post injection, with all doses resulting in a significant increase in sleep induction compared with vehicle.
The effects of the caffeine on sleep-wake behavior in mice at the beginning of the habitual rest period (ZT0) were also studied (Figure 4). Mice were administered caffeine at a dose of 15 mg/kg, approximately equivalent to a human consuming 6 cups of strong coffee prior to sleep. Video analysis was very effective in detecting a significant increase in wakefulness for up to 2 h post caffeine administration. A previous study using EEG-defined sleep and administering an identical dose of caffeine reported a comparable duration of wakefulness (Huang et al., 2005). Collectively, the data emphasize the validity of using video monitoring to assess the impact of drugs on the regulation of sleep.
Changes in environmental light exposure are an additional factor known to modulate sleep-wake behavior either indirectly through the entrainment of circadian rhythms or directly in the form of acute sleep-wake transitions. Previous studies in nocturnal rodents have demonstrated that light exposure increases sleep while darkness promotes wakefulness (Benca et al., 1998; Borbely et al., 1975). In the current study we demonstrated that this video-tracking approach could differentiate between the effects of dim and bright light on sleep induction during the habitual dark period and detect the intensity-dependent effects of light on sleep throughout the duration of the light pulse (Figure 5). Moreover, the data also demonstrate that this method is sensitive enough to discriminate between the effects of different light intensities on sleep.
All surrogate measures of sleep have inherent limitations. The primary limitation of this approach is that it is unable to determine sleep architecture (REM vs. NREM sleep). As such, we would envisage that this method is used as a first-pass assessment of sleep-wake behavior, particularly when screening large numbers of transgenic or mutant animals or large numbers of pharmacological compounds. This would then enable targeted selection of optimum candidates for detailed EEG/EMG assessment. Moreover, it is conceivable that the use of immobility (40 sec or more, 95% sensitivity) may be suboptimal under certain conditions. In the present study we administered zolpidem, which exhibits preferential affinity for the alpha-1 GABA(A) receptor subtype, which is associated with sedative rather than muscle relaxant properties (Crestani et al., 2000). Clearly, agents that have a strong effect on muscle tone such as high doses of GABAergic compounds may result in a degree of muscle relaxation that might be incorrectly scored as immobility-defined sleep. In addition, these studies are based on C57BL/6 mice, which display relatively clear bouts of activity and inactivity. Future studies would be required to determine sleep-wake behavior in different strains of mice (Franken et al., 1999).
The system we describe here provides a robust high-throughput screen for sleep-wake behavior that can be incorporated easily into any existing phenotyping test battery. It provides an ideal first-pass screen to evaluate transgenic/mutant mice or the effects of novel compounds prior to more focused studies using EEG/EMG. An additional advantage of using video tracking to assess sleep is that it enables the simultaneous evaluation of additional behavioral repertoires associated with changes in sleep and wakefulness. These include parameters such as the distance traveled or the time spent in certain areas of the cage, which can provide additional data on anxiety or behavioral inhibition. An example of the extended data available over 24 h is shown in Supplementary Figure S3. This degree of flexibility may be particularly valuable as part of behavioral phenotyping programs, in that it enables archived data to be subjected to post hoc data mining to evaluate other behaviors of interest.
EEG/EMG studies require surgical experience and an understanding of EEG data analysis. The methods described here provide a more accessible approach for assessing basic sleep-wake behavior. Low-cost miniature infrared cameras and hard-drive storage make the initial setup costs under £7000—considerably lower than EEG/EMG telemetry (excluding costs of light-tight chambers). Once the hardware is in place, the costs of screening are minimal—just that required for housing animals. While such an approach cannot replace the requirement for EEG/EMG in sleep research, it provides a valuable additional tool for sleep assessment in transgenic or mutant mice or in the preliminary stages of drug development. As part of a test battery, the use of video monitoring to assess sleep-wake timing in addition to wheel-running provides additional valuable data on this fundamental biological rhythm that is often overlooked in circadian research.
A substantial advantage of the use of video tracking is that it is an entirely noninvasive method. This provides a major welfare advantage against EEG. Moreover, in certain conditions the need for a tether or implanted telemetry device can potentially affect the behavior of the animal, particularly in mice, where the implant size is typically disproportionately large compared with body mass (Leon et al., 2004). The advantage of a noninvasive approach is particularly evident when evaluating sleep in mouse models of disease, as animals may be too frail to undergo invasive procedures or data may be confounded due to transgenics showing different responses to surgery or postoperative recovery rather than a real difference in sleep-wake behavior.
Conclusions
Here we describe a low-cost and high-throughput assay to assess sleep-wake behavior in mice, based on digital video tracking using commercially available behavioral phenotyping software. This approach is noninvasive and markedly inexpensive yet provides an extremely high correlation with simultaneous EEG/EMG assessment of sleep. While it is currently not possible to evaluate REM/NREM sleep using this approach, it provides an ideal preliminary screen for an assessment of sleep duration, sleep fragmentation, and sleep latency in response to genetic modification, environmental manipulation, or compound administration. The provision of additional high-throughput tools for circadian and sleep research should improve our understanding of the genetic mechanisms involved in sleep-wake regulation as well as facilitate drug discovery in this area.
Footnotes
Acknowledgements
We thank Chris Lloyd (ANY-maze) for technical assistance with the video-tracking software and members of our research group for encouragement and advice throughout. We also thank Dr Silvia Gatti-MacArthur (Hoffman-La Roche) for feedback. This work was supported by a Wellcome Trust Programme Grant to RGF/SNP and EUCLOCK (FP6 IP) to R.G.F. S.P.F. was supported by a Knoop Junior Research Fellowship (St Cross, Oxford). S.I.H.G. is supported by Hoffman-La Roche.
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
