Abstract
Polymeric textile materials, now, find extensive application in modern society than previously imagined, particularly in protective clothing. The application of these polymeric materials has been restricted by their flammability and contribution to fire risk. Flame retarding compounds and inherent flame-retardant materials have been introduced to reduce and even halt the flammability of polymers. However, over time, flame retardants tend to leach out of materials. The exposure of polymer textile to varying heat intensities may trigger polymer and flame retardant’s volatilization and thermal degradation. Firefighters may be exposed to toxic chemicals through the volatilization of flame-retardant compounds from the bunker gear. Flame retardant’s volatilization is associated with thermal degradation of the flame-retardant chemical compounds in the textile material. This review focuses on volatilization of flame retardants from protective textile materials resulting from exposure to heat and seeks to provide the necessary understanding about the release of flame retardants from flame-retardant textiles, particularly firefighting garments.
Introduction
Polymeric materials are ubiquitous and have wide application in modern society. For example, the firefighter protective clothing is fabricated from high-performance thermoplastic polymers. However, the limit to the use of most polymeric materials has been attributed to their flammability and fire risk. Polymeric materials, in their pure form, are extremely flammable and are often prepared with a combination of additives (flame retardants) to improve performance under adverse conditions, such as extreme temperature and chemical exposure, processability, durability and strength. Flame retarding polymers and/or the use of flame retardants in polymers has provided a critical solution to the usage and flammability challenges related to textiles used in fabricating protective garments used in high-risk environments such as firefighting, military, aerospace and energy sector. Initially, these textiles were chemically treated to retard flames; later, inherently retarding textiles became popular in flame retarding textiles, particularly in firefighting. 1 The chemically treated flame-retardant textiles are either mixed with flame retarding chemicals during production (reactive) or surface coated after production (additive).1,2 However, the use of flame retardants, particularly halogenated flame retardants, have been reported to be potentially harmful.2,3
Until recently, flame retardation of polymers has commonly been achieved by the incorporation of flame retardants, particularly halogenated flame retardants. In the past few decades, researchers have devoted studies in search of environmentally friendly, stable and cost-effective flame retardants for polymers including the use of inherently flame-retardant materials. 3 Inherently flame-retardant fibres – for example, aramid (e.g. Nomex®, Kevlar®), polyamide-amide (e.g. Kermel), polybenzimi-dazole (e.g. polybenzimidaxazole (PBI)) and polyimide (e.g. P84) – have been increasingly used.1,4 These fibres exhibit a very high thermal stability (decomposition temperature > 450°C) and a good flame retardancy, characterised by high Limiting Oxygen Index (LOI), low Heat Release Rate (HRR) and relatively low smoke obscuration, as compared to the ordinary engineering polymers (such as nylon or polyesters). 5 However, in a recent study, toxic brominated flame retardants (BFRs) were reported from bunker gear made from inherent flame retardants (Nomex®, Kevlar®). 3 Further research is still needed to further quantify these BFRs and more importantly identify their source in the garment.
The firefighting profession is well-known for exposing firefighters to extremely dangerous environments and a wide range of hazards, such as open flames, extreme hot conditions, smoke, toxic chemicals and hazardous substances, such as volatile organic compounds (VOCs) and semi-volatile organic compounds (SVOCs), among others. Some of the toxic chemicals that firefighters are frequently exposed to during firefighting, such as polyaromatic hydrocarbons, are known to be possible or probable carcinogens.6,7 Hence, firefighters are at high risk of developing cancers compared to the public. For example, firefighters have been reported to be at high risk of a number of cancers such as prostate, multiple myeloma, brain, testicular, colon, rectum, stomach, buccal cavity and pharynx, leukaemia, malignant melanoma, skin and non-Hodgkin’s lymphoma.6,8 Furthermore, thousands of firefighters are injured yearly and some are even killed during fire incidents globally. For example, in the United States, 69,400 firefighters were reported to be injured and 97 died in various fire incidents in 2012, and over 60,000 injuries in 2019 occurred in the line of duty.1,9 In China, over 300 firefighters are injured or disabled because of the protective gear failure under extreme fire conditions. 10 Firefighter’s protective gear provides a shield and acts as the first line of defence against flames, heat, smoke, steam and hazardous chemicals.11,12 More so, at times the gear may negatively affect the firefighter’s performance, health and safety. 13 The protective gear includes bunker gear, safety boots, helmet, safety goggles, hood and gloves. However, bunker garments protect most parts of the body, including all extremities.
The objective of this study is to review the literature on the volatilization of flame retardants from the firefighter protective garment to form a knowledge base for characterising firefighter exposure. Moreover, the work explores the literature gap in the volatilization of the commonly used toxic BFRs, particularly under thermal conditions experienced at fire incidents. In the following chapters, firefighter bunker gear is discussed with emphasis on the textile materials and flame retardants used in fabrication. Then the study further discusses literature on the common exposures (thermal and chemical-flame retardants) that firefighters encounter while in operation. Finally, the volatilization of SVOCs, particularly the BFRs from the garment due to thermal exposure is reviewed.
Bunker gear
Bunker gear is an integral part of firefighter protection, particularly thermal protection, which is achieved mainly by using a multi-layered and/or thick textile material made of durable textile fibres and flame retardants. Textile fibres, as the prime key component of the gear, require constant development to produce superior performance protective clothing. 1 Bunker gear comprises three different layers: an outer shell (outer layer), a moisture barrier (middle layer) and a thermal liner (inner layer). The outer shell provides a tough, durable first-line of defence against heat, flame and abrasion.14–17 A typical firefighter bunker gear is shown in Figure 1.

Typical firefighter bunker gear.
Most of bunker gear’s outer shell fabrics are constructed of but not limited to meta- and para-aramids activities (Nomex® and Kevlar®), PBI, polybenzobisoxazole (PBO), melamine (BASOFIL), polysulfonamide (PSA) and/or combinations of these blends. The thermal liner provides insulation against heat penetration while the moisture barrier prevents water and other firefighting-related liquids from entering the gear, keeping the thermal liner dry and ultimately the firefighter’s body protected.9,16 The multi-layer system is designed to increase the overall thickness and consequently improves the protection against low-level radiant heat exposure.9,18 Firefighter protective clothing has recently received growing attention because of the extreme thermal exposures commonly exposed to and the need to provide firefighters protection in those conditions.
Firefighting gear has since evolved from the use of rubber coat that extended past the knees and roll-on boots and flame-retardant textiles to the latest garments made from superior performing fibres such as Kevlar® and Nomex® as the backbone of bunker gear. Today’s firefighter protective clothing is primarily based on years of field experience and research studies. The development and incorporation of high-performance fibres in the textile industry have significantly improved the manufacture of protective clothing, particularly for hostile thermal environments. However, the protective gear has been associated with negative effects on firefighters such as limited mobility and impeding firefighter job performance, heat strain and exposure to toxic BFRs.3,15,19–21 Some of the recent issues related to the protective garments can be attributed to the same technological advancement.
In the past decade, there has been great interest in the research of firefighter gear thermal performance and the thermal environments experienced by firefighters.1,3,9,11,14,16,22–24 However, much of the firefighter protective clothing studies have focused on conditions firefighters find themselves in extreme conditions, such as being engulfed in fire. 25 These conditions generally result in life-threatening injuries and/or death. Little appears to have been done to address other conditions out of the extreme conditions, such as the low and medium thermal conditions. This is interesting, since most burn injuries occur outside the extreme and life-threatening fire conditions. A number of these studies involving firefighter protective garment have modelled the heat transfer through the firefighter garment to predict thermal skin burns, and data indicate that most second-degree burn injuries are sustained on the arms, shoulder and neck. 26 Thermal conditions experienced by firefighters are extraordinary and requires continuous researching and developing to ensure continued superior firefighter protection. Improved firefighter gear can minimise the risk of firefighter injury. 9 However, the presence of flame retardants in large quantities may pose a challenge, since it is well-known that flame retardants tend to leach out of materials with time. This review focuses on potential volatilization of flame retarding compounds from the textiles used in the firefighter protective garment, particularly under thermal conditions.
Firefighter bunker gear is manufactured and tested in accordance with the National Fire Protection Association (NFPA) 1971

FTT Cone Calorimeter.
Given the high-intensity heat flux incident on the gear during fires, the garment fabric will likely experience thermal oxidation degrading and decomposition. However, the protective clothing materials may still be thermally degraded when exposed to low-level heating during a long proximity firefighting. 23 This thermal composition could include flame-retardant volatilization.
Bunker gear and flame retardants
Most polymeric materials including firefighter protective gear, inter alia, have been mixed with flame-retardant additives in the manufacturing process to improve fire risk profile. Flame retardants are a broad group of chemicals commonly incorporated into consumer products such as textiles, plastics, wood and electronics with the intention to resist ignition and/or propagation of fire. According to Alaee et al., 2 there are more than 175 different flame retardants that are used worldwide to impart flame retardancy in materials. In recent years, textile materials used in protective clothing were combined with flame-retardant compounds to strengthen and make sure they can withstand thermal conditions. The wide range of flame retardants include phosphorus, halogen-based compounds and nanomaterials (e.g. carbon nanotubes, graphene, MXene). 29 Halogen-based flame retardants are the most commonly used flame retardants particularly the BFRs which consist of a diverse group of bromine-containing compounds. 30 However, some of the flame retardants are often mixed with other compound to create synergistic effect to achieve optimal flame retardancy efficiency. The use of BFRs owes to their efficacy in imparting flame retardancy and are commonly used in conjunction with antimony oxide (Sb2O3). The use of antimony oxide (Sb2O3) in conjunction with organo-bromine compounds, such as the tetrabromobisphenol A (TBBPA), polybrominated diphenyl ethers (PBDEs), Hexabromocyclododecane (HBCDDs) and Polybrominated biphenyl (PBBs), results in synergistic flame retarding effects.Furthermore, low quantities of BFR compounds can ensure highest fire safety. 31 PBDEs are additive flame retardants often added at 5%–30% weight of the product.32,33
While the latest technologies have led to the advances in flame retardants, the use dates back to 450 BC, where the Egyptians used alum to flame retard wood. A wide variety of flame retardants made from various mixtures are commercially available for use in consumer products. The use of flame retardants in consumer products, particularly BFRs, has been mired by controversies around the toxic effects of the chemicals. Bromine-containing flame retardants have been under scrutiny since the 1980s because of dioxin formation during the incineration of polymers. 34 BFRs have been found to be toxic to humans and the environment. The toxicological concerns around all BFRs and their use now include the synergistic antimony (III) oxide. 34 BFRs have been classified under the persistent organic pollutants (POPs) and are subject to international agreements and conventions such us the Aarhus protocol (1998) and the Stockholm Convention (SC) on POPs. 35 PBDEs were banned within the EU in August 2004.36,37 However, flame-retardant armaments continue to find application in environments such as those confronting firefighters. 38
Firefighter’s thermal exposure
Firefighters can be exposed to low-radiant heat flux conditions to high-intensity heat flux during firefighting, depending on the type and size of the fire. Furthermore, the heat flux and temperature that firefighters are exposed to may vary depending on the different job activities. 1 Firefighter’s thermal exposure provides the basic benchmark upon which the latest firefighter protective garments’ performance is based. Thermal environments during fires are commonly dictated by various factors, including the initial environment, ignition source, combination and arrangement of fuel and ventilation, to name but a few. 27 While the protective gear is tested against 84 kW m--2 heat flux, in terms of the NFPA 1971 standard, firefighters seldom face such high heat flux during firefighting. Firefighters are mainly exposed to low- to medium-range radiant heat. 39 In a recent study, three heat fluxes – 84, 12 and 5 kW m--2– were identified to represent the high-, medium- and low-heat flux intensities that fire firefighters are exposed to, respectively.40,41 The low- to medium-intensity exposures signifies the pre-flashover conditions and the high-intensities condition signifies flashover.40,41 During fire incidents, heat exposure is predominantly due to radiation (80%), though other heat transfer methods can occur. 42 Firefighters’ thermal exposure at fire incidents may also vary according to the different roles in extinguishing the fire. For example, a firefighter operating the nozzle at the end of a hose line may be exposed to a different heat flux compared to a supporting firefighter holding the hose a few metres away from the nozzle.
The thermal working conditions for firefighters are commonly described according to environment temperature and incident heat flux.27,42 However, this is the simplified manner of using the temperature and heat flux. In a recent study, firefighter exposure conditions were identified and categorised into three categories. These are routine, hazardous and emergency, and are based on the air temperature and radiant heat flux.18,43 Routine conditions are characterised by air temperature up to 50°C–60°C with radiant heat flux up to 1.4–1.6 kW m--2, hazardous conditions by air temperature from 50°C–300°C with heat flux from 1.4 to 8 kW m--2 and emergency conditions by air temperature from 300°C–1000°C with radiant heat flux from 8 to 200 kW m--2. 43 However, a fourth condition, namely critical (with temperature between 250°C and 1000°C and heat flux of 10–100 kW m--2), has been identified elsewhere. 27 Table 1 indicates similar data from other previous studies conducted on the categories.
Notably, Table 1 only indicates radiant heat flux but the convective and conductive heat fluxes, or the net heat flux could be higher. Firefighters typically work for 10–20 minutes in the routine environment, 1–10 minutes in the hazardous environment and <1 minute in the emergency environment. 1 A recent study has reported that firefighters usually enter burning structures couple of times with each entry taking about 10–20 min. 19 The radiant heat intensity during domestic fires may reach as high as between 40 and 200 kW m--2 for large fuelled fires. 45 Firefighters fighting a room fire have been reported to be exposed to heat fluxes up to 12.5 kW m--2 and 300°C temperature for a couple of seconds.1,46 Interestingly, the bunker gear shell layer used in a 1971 compliant suite typically do not exhibit thermal damage until they reach temperatures above 300°C. 27 In a study conducted by Rossi, 42 measurements during training indicated that firefighters are typically exposed to 5–10 kW m--2 heat flux inside a building. However, McGuire quoted by Rossi, 42 and Song et al. 18 have reported even higher heat fluxes of > 50 kW m--2 outside of burning buildings. Interestingly, exposure to low-level thermal conditions, 5–20 kW m--2, has been identified as the most common thermal hazard that can result in skin burn injuries. However, according to the ISO 6942 standard low- to medium-range radiant heat in fire conditions lies between 10 and 40 kW m--2. 39
It has been reported that the human skin starts experiencing pain around 44°C and second-degree burns occurs at approximately 55°C. 47 However, firefighter skin burn injuries have been reported at skin temperature of 42°C. 48 Firefighter thermal exposure categories provide a basic understanding of environments and risks firefighters maybe exposed to during fire incidents. The thermal classes illustrate discrepancies between experts regarding the criteria for the different levels. 27 Hence these classes remain ambiguous among researchers. 1 Firefighter exposure during firefighting activities is mainly in the routine and hazardous thermal condition and less frequent in the emergency condition. Understanding the fire conditions frequently encountered by firefighters is crucial to preventing firefighter injuries.
During a house fire, the flow of hot gases from a doorway or through a window may well be above 400°C and may travel some distance down the corridor. 25 For example, data showed that radiant heat flux from a window or door opening can be more than 50 kW m--2 at a distance of 6 metre from the building. 25 In another study, the hallway just outside the burn rooms, the temperatures at the ceiling and 1.5 m level were well above 600°C, with heat flux values at 1.5 m at 22–28 kW m--2, but firefighters operating in the crawling position would have significantly reduced ambient temperatures (∼415°C) and heat flux (∼11 kW m--2). 24 Moreover, in a recent study, it has been reported that the temperature of firefighters’ bunker gear outer shell, in a typical fire, may reach up to the order of 1000 K. 23 Exposure to flashover fire conditions, though not common, include the observation of upper layer temperature of ∼600°C or radiant heat flux at floor level of ∼20 kW m--2.49,50 Some of firefighting garments have shown to lose their strength even at lower heat flux of 10 kW m--2. 9 McCarthy and Di Marzo 47 also observed that with intense heat flux (20 kW m--2) firefighter protective gear experiences degradation even at extreme short durations as 30-second exposure. However, aramid fibres, which are the main material for bunker gear, are reported to start failing to provide thermal protection when exposed to 600–800°C due to their decomposition behaviour. 10 Furthermore, the impact of repeated thermal exposure of the bunker gear has often been studied. 9 However, no literature could be found so far on the protective gear’s flame-retardant behaviour as a result of exposure to heat and more so, the volatilization of the flame retardants.
Firefighter exposure to flame retardants
Flame retardants have the ability to leach out or even degrade from consumer materials during the material’s lifespan. Many people are exposed to flame retardants through textiles such as curtains, furniture, mattresses and even apparels in their homes and workplaces, daily. 51 More so, firefighters are exposed to these consumer products during fire conditions, where they may even be readily undergoing thermal pyrolysis and/or decomposition. The firefighting environment pre-exposes firefighters to a wide range of hazards such as polycyclic aromatic hydrocarbons (PAHs), VOCs, hydrogen cyanide (HCN), and several other organic and inorganic compounds that are common toxic substances in fire smoke.52,53 A number of studies on chemicals used to impart special fire safety attributes in textiles, such as flame retardants, have shown links to adverse health effects such as asthma and cancer.51,53
Firefighters are required to make use of self-contained breathing apparatus (SCBA) for respiratory protection against the inhalation of smoke and other airborne toxicants that result from combustion. However, firefighters may still be exposed to combustion by-products even with the wearing the SCBA throughout firefighting operations.53,54 In a study by Stubbings and Harrad, 55 human exposure to BFRs was reported to occur predominantly via indoor dust and food ingestion with a minor contribution from inhalation. Poutasse et al. 56 in quoting the works of Weschler and Nazaroff (2012), however, reported that estimates indicate that human uptake of SVOCs by skin may be large, potentially equal to or exceeding intake via inhalation. 57 For firefighters, absorption through the skin could provide another route of exposure to flame retardants and smoke toxins other than inhalation and ingestion. 53
The volatilization of flame retardants from consumer products such as the one reported by Ortuño et al. 58 produces high yields of toxic furans. Moreover, during firefighting, BFRs embedded in the bunker gear may be volatilizing due to thermal exposure, adding to firefighter exposure burden. Firefighter bunker gear may, under thermal exposure, also be a source of exposure to toxic chemicals, particularly the flame retardant compounds embedded in the gear itself. However, data on the exposure of firefighters to flame retardants from textiles is scarce.3,51 The available data on the exposure of firefighters to toxic flame retardants on bunker gear are based on the contamination of the gear during fire operations.7,52,53,59,60
There is still the need for research into understanding bunker gear, and impact of the used textile material to enable characterisation of compounds volatilizing from inherent flame retardants, particularly Kevlar® and Nomex®, under thermal exposure may provide in-depth knowledge on the fibres. A number of studies reported on thermal decomposition of some of the common inherent flame retardants such as meta-aramid fibres, 61 pyrolysis of Kevlar 62 and PBI. 5 However, the studies stop short of reporting on the presence of toxic flame retardants, including BFRs.
The leading cause of death among firefighters has changed, over the past 30 years, from cardiac-related to cancer. 22 Furthermore, a number of epidemiology studies conducted continue to find elevated risks of several cancers in firefighters. 52 Daniels et al. 63 reported on increased mortality and incidence risk for all cancers; mesothelioma; and cancers of the oesophagus, intestines, lung, kidney and oral cavity, as well as an elevated risk for prostate and bladder cancer among younger firefighters. Moreover, many firefighters (70%) have been predicted to die from cancer than the general population. 22 Fire deaths are commonly the result of toxic products of combustion rather than the thermal danger. The International Agency for Research on Cancer (IARC) has concluded that the firefighter occupational exposures should be classified carcinogenic to humans given the limited research and evidence.7,52,56 However, literature related to firefighters’ exposure to the toxic PAH released during fires remains limited. 6 A suspected critical route that calls for further scrutiny is the volatilization of toxic flame retardants in the bunker gear of firefighters.
Volatilization
Bunker gear exposed to prolonged severe fire conditions undergo three heating phases. The initial phase is marked by warming up of the material, followed by the second phase marked by onset of thermal property changes and the third phase is characterised by chemical and thermal degradation that includes volatilization of the fabric.
48
Firefighters’ frequent exposure to thermal conditions during firefighting may likely contribute to the accelerated thermal degradation and possibly thermal decomposition of flame retardants in the protective garment. For example, Ortuño et al.
56
reported the volatilization of some BFRs from flame retarded high-impact polystyrene in the back cover of a television set when heated to temperatures between 50°C and 250°C. Furthermore, the degradation of polymers has the potential to generate toxic and/or carcinogenic gases.
64
The incineration of the BFR laden products provided information on the emission of the toxic polybrominated dibenzo-
As described, inter alia, most of the latest bunker gear fabrics are fabricated of the common inherent flame retardants but not limited to meta- and para-aramids activities (Nomex® and Kevlar®), PBI, PBO, BASOFIL and/or combinations of these blends. The use of these inherent flame retardants owes to their superior performance under thermal stress, which is the common threat to firefighter safety during firefighting and their ability to replace textiles that are treated with flame retardants. For example, aramid fibres such as Kevlar® and Nomex® have been reported to start degrading at temperatures around 548.1°C and 423.7°C in air, respectively. 4 The degradation temperature of PBO starts at 610°C and PBI at 500°C in air. 5 The use of high-performance fibres in bunker gear, such as Kevlar®, Nomex® and PBO, among others, has been extensively reported in literature for the past decade. While it is generally accepted that firefighter bunker gear is fabricated from these inherent flame retardants, however, data from a recent study indicate the presence of flame BFRs’ traces in the firefighting garments (in all three layers of the protective garments). 3 However, more work still needs to be done to further characterise the presence of flame retardants in firefighter bunker gear. Similar to firefighter bunker gear, BFR traces have been reported in other consumer products such as textiles used to make curtains,66–68 carpet,66,67,69 car interior foam, 67 upholstery textiles,69,70 and camping tents 71 despite restrictions on BFRs.
In the past two decades, considerable amount of research has been conducted around the emission of organic pollutants into the environment from different consumer materials. Most work has been devoted to analysing pyrolysis of electronic scrap. 72 To date, only a few has reported on the pyrolysis of textile materials, pyrolytic gases and their respective pyrolysis temperatures. 64 To understand the volatilization of flame retardants, it is important to first understand the pyrolysis process of textile materials. Pyrolysis is the initial step in any combustion process. Figure 3 depicts a typical degradation of a fibre under thermal exposure.
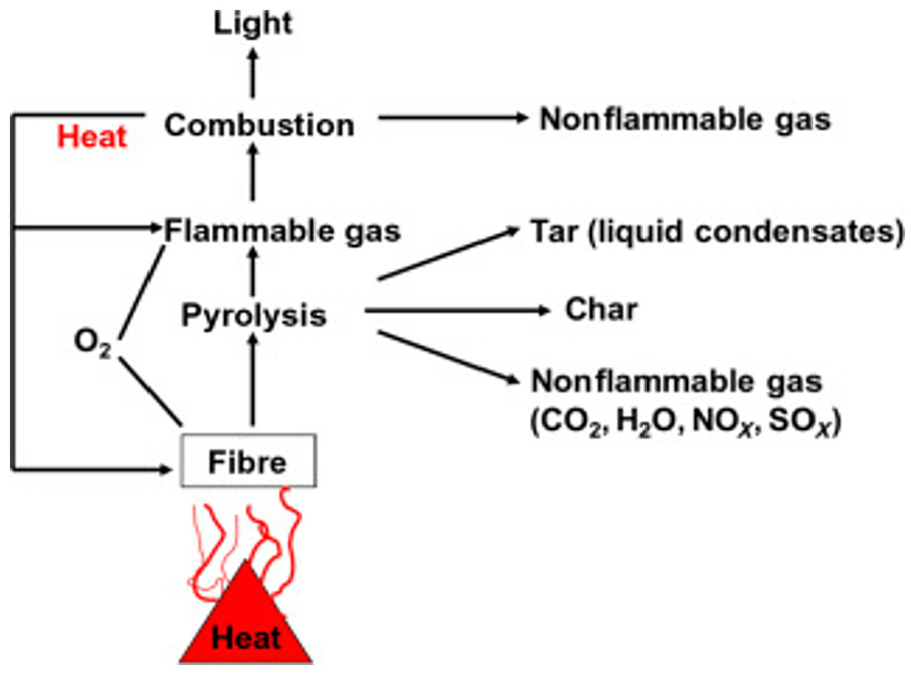
Typical thermal degradation process. 73
The exposure of a fibre to sufficient heat triggers the pyrolysis process. The process of pyrolysis is coupled with the emission of gases and other compounds, particularly volatile and semi-volatile compounds. It has evidently been reported that exposure to heat of some polymer materials such as plastic causes the volatilization of flame retardants. 56 Similarly, the constant exposure of thermal protective clothing to sufficient heat flux may trigger the pyrolysis and therefore the volatilization of flame-retardant compounds. Thermal decomposition of flame retardant as a result of exposure to extreme thermal conditions in firefighting may cause emission problems and result in both firefighter exposure and environmental pollution. Moreover, the emission of some flame retardants such as PBDEs and HBCDDs from textiles has been reported at temperatures as low as between 20°C and 80°C. 74 As a result, the volatilization of these flame retardants from bunker gear may well start at lower temperatures than initially thought. Interestingly, thermal degradation of the protective gear fabrics starts well before visible change on the material. 39 In the recent work by Horn et al., 75 firefighter bunker gear has been reported to lose strength with an increase in the washing cycles. This may further be accompanied by the accelerated leaching of the flame retardant from the garment.
According to the mechanism by which flame retardants function, it is clear that some flame retardants are degradative in their functioning. A recent study reported that the combustion of brominated and chlorinated compounds is the main source of dioxins. 56 The use of BFRs is further complicated by their potential degradation to ecotoxicological congeners in the case of PBDEs by exposure to UV. 34 When subjected to heat, BFR containing materials degrade readily with potential to form brominated product of incomplete combustion. 65 Experimental studies have reported thermal degradation of some BFRs, under pyrolytic and oxidative environments, to take place in the temperature range of 400°C–900°C (Altarawneh et al., 2019); however, temperature range as low as 280°C–900°C has been reported elsewhere. 65
Temperature is one of the important factors that influence the environmental fate of BFRs. In a study by Marsanich et al., 76 results have confirmed that the processing of TBBA at temperatures above the boiling point (180°C) may cause the release of TBBPA into the environment due to the evaporation phenomena.In the same study, pyrolysis experiments were carried out on TBBPA + Sb2O3 using a small furnace, whereby a quartz wool filter was placed at the end of the reaction tube and maintained at 100°C by an electric ribbon heater, to collect condensable phases vaporised from the sample. It has been further indicated that the degradation of TBBPA starts above 200°C 76 and takes place especially around 225°C.65,77 Rzyman et al. 77 reported that the mechanism of decomposition of pure TBBPA during thermal treatment differs from its decomposition in the presence of Sb2O3. In the differential scanning calometry analysis, the maximum peak of bromination was reported at 280°C. In another study, decomposition of TBBA was effected using the TGA, and the identification and quantification were carried out using the Gas Chromatography-Mass Spectrometry (GC/MS). 76 HBCDD thermal degradation has been widely shown to start around 230°C, and the most important gaseous product formed is hydrogen bromide (HBr).74,78 However, Barontini et al. 77 reported the process to happen between 240°C and 270°C. In plastics, hydrobromic acid (HBr) has to be released as the product of BFRs pyrolysis. 77 It is, however, important to note that thermal exposure alone is not likely the primary contributor to textile thermal degradation and/or decomposition. Other reasons of degradation of the protective textiles, such as repetitive washes, have been reported elsewhere. 75
In addition to the use of flame retardants, firefighter garment’s middle layer (moisture barrier) is commonly treated with water-resistant finishes, such as polyfluoroalkyl substances (PFASs), to ensure maximum protection against liquid penetration. Commercially available water-resistant finishes include fluoropolymer compositions sold under the names Milease®, Teflon®, Capstone®, Zepel® and Repearl®, among others. 79 Water or moisture penetration of garments compromises the thermal performance and increases the weight of the garments. Like flame retardants, interestingly, emerging research into water-resistant finishes has indicated their potential adverse health effects to humans. Moreover, PFASs have been recently detected in new or unused firefighting bunker gear, indicating that PFASs are embedded in the protective gear’s moisture barrier.22,80 In a recent study, the textiles used for firefighter gear were found to containing high levels of total fluorine (up to 2%), and PFASs were detected and measured on both new and used firefighter gear. 22 Because PFASs are shed from the textiles used in turnout gear and are more mobile, they represent another viable exposure source for firefighters that requires special attention. 22 Like flame retardants, water-repellent PFASs in the gear may be released into the environment, which may pose further exposure risk to the firefighter using the gear.
Conclusion
Volatilization of volatile and semi-volatile compounds from textiles continues to be a risk problem, particularly under the fire conditions. Currently there is general understanding on the thermal environments experienced by firefighters during fire conditions. However, there is no consensus in literature on the three thermal categories, with varying heat flux and temperature. There is a need to understand the behaviour of flame retarding compounds incorporated into the firefighter garment under a common firefighting thermal environment. Before pollution and exposure control measures can be taken, the emission of flame retardants from flame-retardant textile should be well understood and estimated. The available literature does not provide much insights on the volatilization of semi-volatile compounds from the garment. To the best of our knowledge, there is no study that has monitored the volatilization of flame retardants, particularly BFRs, from firefighter garments. However, data of other toxic substances added to bunker gear, such as PFAS, are increasingly becoming available.It is clearly evident that firefighters are a unique group, more so, that they have an elevated risk of cancers than the general population. Quantification of flame retardants and monitoring of their volatilization could provide the much-needed insight knowledge and the contribution to firefighter cancer burden.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This Study was supported with funds from the Tshwane University of Technology Material Composites Niche Area.

