Abstract
Objectives
An emerging literature has assessed cognition or imaging markers of brain health in in older age bipolar disorder (OABD). In this context, we conducted the first longitudinal study (to our knowledge) that assessed the relationship among cognition, mood symptoms, and imaging markers of brain health in OABD.
Methods
99 participants with OABD were enrolled, underwent baseline assessment, and were followed annually for up to 3 years. They completed comprehensive assessments that included evaluation of general medical status, vascular disease burden, mental status, cognitive performance. A subset of participants (n = 58) completed magnetic resonance imaging (MRI) at one or two time-points, yielding three measures of brain health: gray matter volume, fractional anisotropy (FA), and burden of white matter hyperintensities (WMH).
Results
Group-based trajectory modelling (GBTM) of overall cognitive performance revealed two groups: a group with higher cognitive performance (63 of 99, 63.6%) and a group with lower cognitive performance (36 of 99, 36.4%). GBTM also revealed two groups based on each of the three imaging markers of brain health. The higher cognitive performance group was associated with the groups with higher measure of total gray matter or higher FA. We found no relationship between the cognitive groups and level of mood symptoms during longitudinal follow-up or WMH burden.
Conclusions
In this first longitudinal study of cognition, mood symptoms, and markers of brain health in OABD, cognitive performance was related to brain health and not to mood symptoms over a follow-up of up to three years. Almost two-thirds of participants with OABD had cognitive performance comparable to older adults without OABD. Larger future studies will need to replicate and extend these findings.
Plain Language Summary Title
A Longitudinal Study of Cognition, Mood, and Brain Health in Older Adults with Bipolar Disorder
This study examined cognitive performance, mood symptoms, and brain health markers in 99 adults with bipolar disorder ages 50 years and older who were assessed annually for up to 3 years. Brain imaging was available in 58 of the participants. While previous research has suggested that repeated bipolar mood episodes might cause brain changes and cognitive decline (“neuroprogression”), this study found that most older adults with bipolar disorder maintained relatively normal cognitive performance. The study findings suggest that significant cognitive decline may occur only in a subset of patients with bipolar disorder as they age. To our knowledge, this is the first longitudinal study that examined the relationship between cognition, mood symptoms, and brain imaging markers together in older adults with bipolar disorder.
Introduction
Bipolar disorder (BD) is one of the leading causes of disability in developed countries. 1 Several studies of mixed-aged patients with BD have shown cognitive dysfunction persists after the resolution of mood symptoms 2 and this dysfunction is an important determinant of disability and functional impairment. 3 Patients with older age bipolar disorder (OABD), commonly defined as BD in patients 50 years and older, 4 are at increased risk for cognitive decline, caregiver burden, and institutionalization.4–6
Some data suggest that mood episodes can be harmful to brain health and contribute to structural brain changes7–9 and neuroprogression10,11 attributed to mechanisms similar to those implicated in neurodegenerative illness such as Alzheimer's disease 12 or vascular dementias. 13 In younger and middle-aged adults with BD, this model of neuroprogression has led to a staging approach that link mood episodes and trajectories to cognitive decline and disability. 14 However, the validity of this model has been intensely debated, and central to this controversy is the methodological challenge of inferring longitudinal disease trajectories from cross-sectional studies.15,16
Cross-sectional studies of OABD have emphasized the heterogeneity of cognitive performance in OABD. 3 The Global Aging and Geriatric Experiments in Bipolar Disorder (GAGE-BD) 17 group of investigators published the largest cross-sectional study in which they pooled and harmonized international data from >1300 individuals with BD. 18 They analyzed the global cognitive performance of 509 patients with OABD and 153 healthy comparators (HC) who were at least 50 years old. In contrast to a previous meta-analysis that identified consistently worse cognitive performance in OABD than HC, 3 they found that patients with OABD had global cognitive performance comparable to the HC. The authors cautioned that their sample included participants who were not highly symptomatic and potentially in better general health than might be seen outside of research studies.
Smaller cross-sectional studies have identified three cognitive groups in OABD.19,20 One cross-sectional study used cluster analysis in 138 participants with OABD (mean [SD] age: 59.5 [7.2]), and identified a large group with preserved cognition comprising 42% of the participants, another large group with mildly impaired group (46%), and a small moderately to severely impaired group (12%). 19 In another study, 66 participants with OABD (mean [SD] age: 63.7 [8.0]) were found to belong to three groups, each comprising about a third of the participants: one without clinically significant cognitive deficits; one with selective deficits; and one with global deficits. 20 There was no relationship between group and measure of clinical severity (e.g., history of psychosis or hospitalization).
The limited number of cross-sectional neuroimaging studies in OABD have reported reduced cortical and subcortical gray matter and an increased burden of white matter hyperintensities (WMH).6,21 Compared to HC, one study found that adults with BD (n = 103, ages 40–79) showed lower cortical thickness in dorsal and ventral frontal, parahippocampal, insular and posterior regions, and lower gray matter volume in hippocampus, amygdala, thalamus, pallidum, and putamen. 22 Another study analyzed associations among age, diagnosis, and cognition with cortical thickness and fractional anisotropy (FA) in participants with BD or schizophrenia, and HC, in those younger (<50 years) and older (≥50 years) to identify whether they exhibited accelerated brain aging. 23 They found that participants with OABD exhibited no evidence of accelerated brain aging, but there was support for an early-life, neurodevelopmental, non-progressive mechanism.
A handful of studies have examined cognitive function longitudinally in OABD using different designs.24–28 Their results have been mixed, showing cognitive trajectories consistent with either normal or accelerated aging. We are aware of only one study that followed 15 patients with OABD and 15 HC to investigate whether cognitive decline and structural brain changes are accelerated in OABD. 29 In this study, OABD patients demonstrated significantly lower performances in processing speed and episodic memory than HC. However, there were no differences observed between the two groups in the mean trajectory of cognitive changes during the 2-year follow-up. Further, longitudinal gray matter and white matter changes did not differ between OABD patients and HC. These findings do not support that BD is associated with accelerated cognitive decline and brain aging. This small study did not directly correlate cognitive trajectories with brain changes or with mood symptoms. Thus, to our knowledge, no longitudinal studies have examined the relationship among cognition, mood symptoms, and markers of brain health in OABD.
In this context, we conducted a longitudinal study focused on assessing the relationship among cognition mood, and neuroimaging markers of structural brain integrity in OABD. We hypothesized that lower mood symptoms would be associated with better cognitive performance. We also hypothesized that markers of better brain health (i.e., higher total gray matter, higher FA, and lower burden of WMH) would be associated with better cognitive performance. In addition, based on prior work showing cross-sectionally that lithium was associated with better cognitive performance in OABD, 30 we conducted exploratory analyses to examine the potential neuroprotective effects of lithium or divalproex.
Methods
The details of the study methods, including neuroimaging procedures, have been previously published. 30 Briefly, from January 1, 2010, through December 31, 2012, 209 participants signed consent; of these, 99 participants with BD and 36 mentally HC met all eligibility criteria. These individuals underwent comprehensive assessments at baseline and annually for up to 3 years. The assessments included evaluation of cognitive performance, 25 psychiatric status (see below), general medical status (Cumulative Illness Rating Scale-Geriatric [CIRS-G]), 31 and vascular disease burden (Framingham Stroke Risk Profile [FSRP]). 32 A subset of participants (BD, n = 58) completed neuroimaging at one (baseline) or two time-points (baseline and termination) assessing total gray matter volume, WMH, and FA. All participants were assessed when they were clinically stable (see below) to minimize the effects of mood disturbances on cognitive performance. Neuroimaging was completed within 3 months of cognitive assessment.
Eligibility Criteria
Participants with BD were: age 50 years or older; with a diagnosis of BD I or BD II based on the DSM-IV criteria 33 and the SCID-IV 34 ; euthymic for at least 4 weeks preceding cognitive assessment, as indicated by scores of 10 or less on both the 17-item Hamilton Rating Scale for Depression (HRSD) 35 and the Young Mania Rating Scale (YMRS) 36 ; able to understand and speak English fluently; with a corrected visual ability to read newspaper headlines and hearing capacity adequate to respond to a raised conversational voice. They did not meet the diagnostic criteria for substance abuse or dependence within the past 12 months and had no diagnoses of dementia or neurologic disorder affecting the central nervous system (e.g., Parkinson's disease, traumatic brain injury, or multiple sclerosis); in addition, they had not been treated with electro-convulsive therapy (ECT) within the past 6 months. The HC met the same eligibility criteria except that they did not meet the diagnostic criteria for any mental disorders except for a specific phobia.
Cognitive Assessment
The cognitive battery comprised 21 well-established and validated individual tests. We normalized raw scores for all individual cognitive tests using the baseline scores of the mentally HC (i.e., comparators’ performance on any test has a mean of 0 and a standard deviation of 1) and created an overall cognitive score for each participant by taking a mean of each normalized individual test. 25
Clinical Assessment
The Life Chart Method37,38 was used every 6 months to gather information retrospectively about hospitalizations, depressive or manic episodes, positive life events, and the timing of those occurrences over the past year, and to rate the severity of depressive and manic symptoms during each of the preceding 6 months on a scale of 0 to 5. For instance, a severity of 1 for depressive symptoms indicates the presence of “mild core features of depression or moderate concomitant symptoms. No clear significant impairment of functioning.” And a severity of 4 for manic symptoms indicates the presence of a “manic episode either with psychosis or requiring hospitalization.” 37 Additionally, obtainable health records were reviewed, and all participants (and available significant others) were asked about physical conditions, medications, and hospitalizations. This information was also used to complete the FSRP.
Statistical Analysis
We employed group-based trajectory modelling (GBTM), 39 a statistical approach aimed at identifying subgroups of individual trajectories summarized by polynomial functions of time and providing a probability of membership in each subgroup. We used GBTM (rather than alternative approaches to analyze our data) because it can identify clusters of individuals following the same trajectory rather than forcing individuals into predetermined categories. Further, it can handle missing data, incorporate covariates, and test developmental hypotheses (e.g., neuroprogression). 40
GBTM was used to model the trajectories of cognitive performance, severity of depressive or manic symptoms, and gray matter, WMH, FA. GBTM were fit using SAS (version 9.4) Proc Traj with maximum likelihood estimation and handling missing data with a missing at random assumption. The highest probability group assignments were extracted from the best fitting GBTM, and group membership was used as an independent variable in subsequent analyses. Fit was compared among 2-, 3-, and 4-group models through Bayesian information criterion (BIC). Logistic regression models were then used to examine the association between membership in trajectory groups controlling for FSRP. Due to our sample size, we did not distinguish between early- and late-onset BD in the analyses.
Results
Baseline characteristics of all participants are presented in Table 1.
Baseline Demographics and Medical/Vascular Burden of Patients With Older age Bipolar Disorder and Controls.
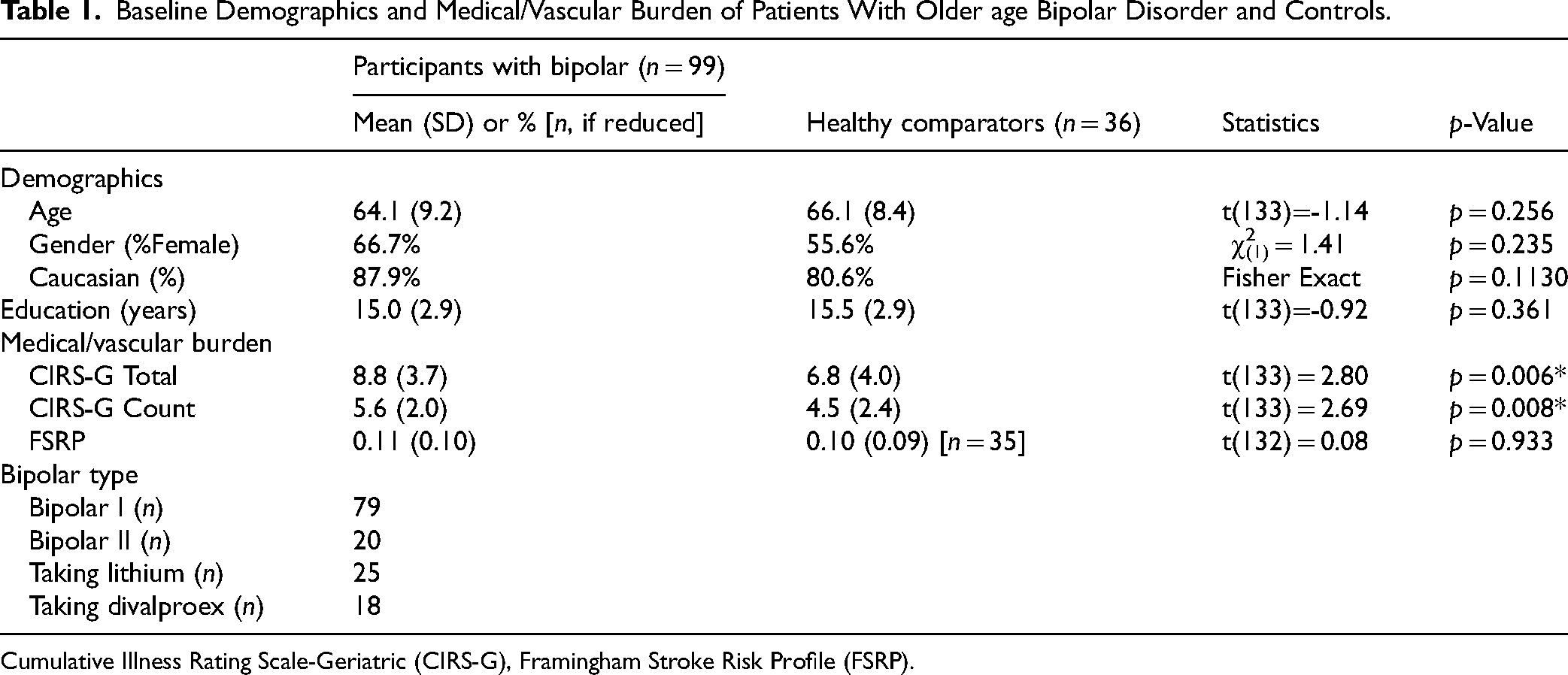
Cumulative Illness Rating Scale-Geriatric (CIRS-G), Framingham Stroke Risk Profile (FSRP).
Cognitive Performance
GBTM revealed two groups of global cognitive performance: (i) a group comprising 63 participants (63.6%) with higher and stable cognitive performance; and (ii) a group comprising 36 (36.4%) participants with lower and stable cognitive performance (Figure 1).
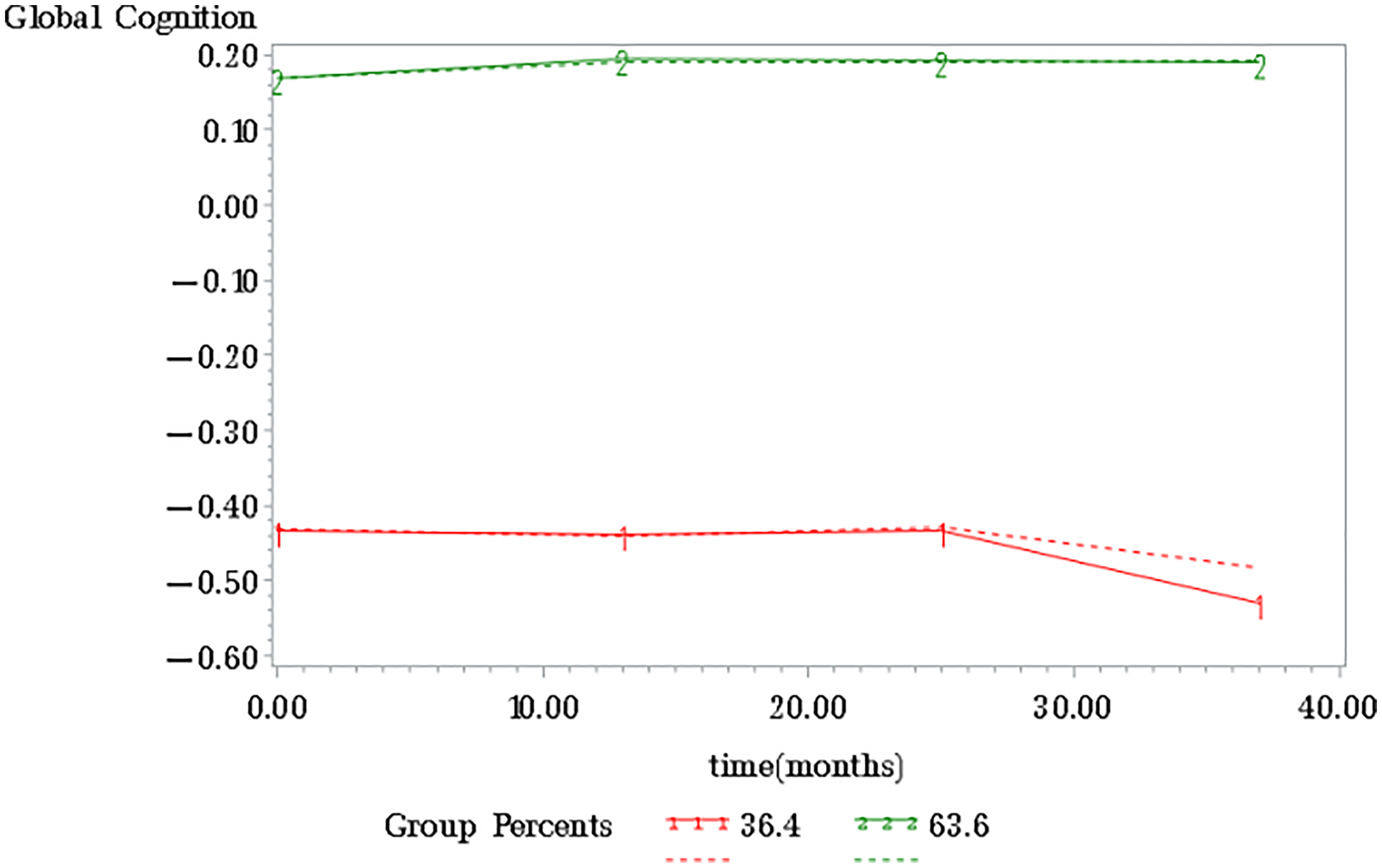
Group-based trajectory modelling of global cognition versus time. Expected (dashed lines) versus observed (solid line) trajectories.
Mood Symptoms
Even though most participants had no or few mood episodes and were minimally symptomatic between these episodes, GBTM revealed three groups based on severity of depressive symptoms: (i) a group comprising 52 participants (56.5%) with almost no depressive symptoms; (ii) a group comprising 29 participants (31.5%) with mild depressive symptoms; and (iii) a group comprising 11 participants (12.0%) with moderate depressive symptoms (Figure 2A). Similarly, GBTM revealed two groups based on severity of hypomanic/manic symptoms: (i) a group comprising 63 participants (68.5%) with almost no hypomanic/manic symptoms; and (ii) a group comprising 29 participants (31.5%) with mild hypomanic/manic symptoms (Figure 2B).
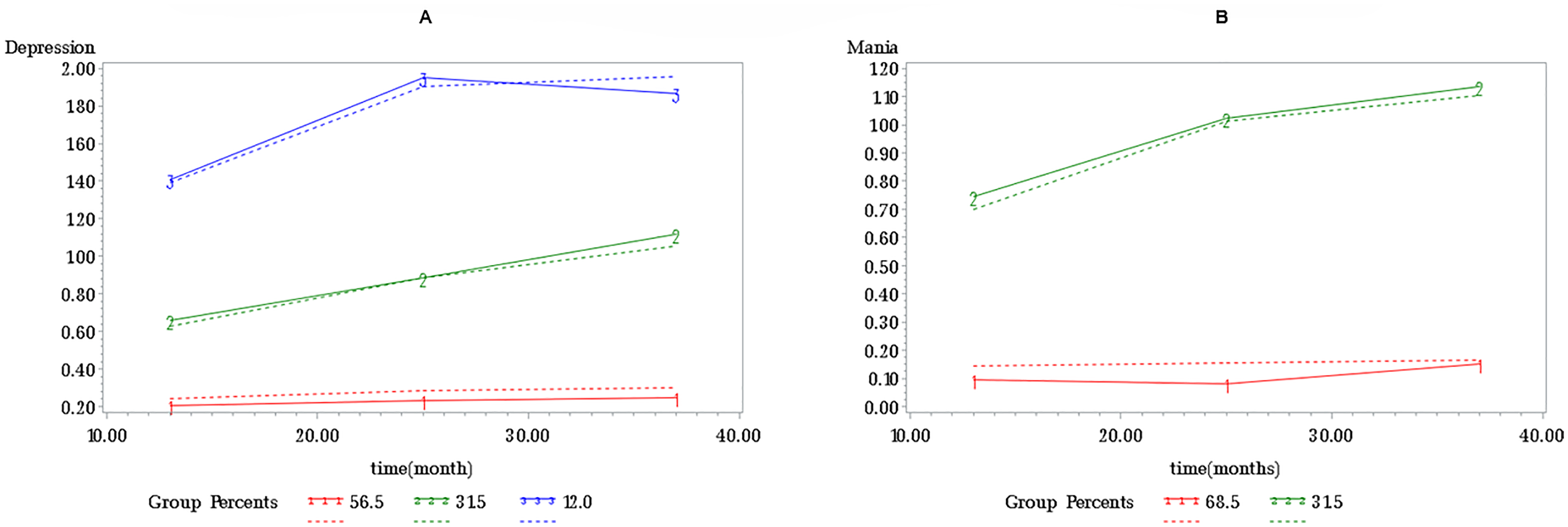
Group-based trajectory modelling of (A) depressive symptoms versus time and (B) mania symptoms versus time. Expected (dashed lines) versus observed (solid line) trajectories. Scores can range from 0 (no significant core features of depression or mania) to 5 (major depressive or manic episode with extreme functional disability).
Imaging Markers of Brain Health
Finally, GBTM revealed two groups based on each of the three imaging markers of brain health: (i-a) a group comprising 37 participants (63.8%) with higher gray matter volume, and (i-b) a group comprising 21 participants (36.2%) with lower gray matter (Figure 3A); (ii-a) a group comprising 38 participants (65.5%) with higher FA, and (ii-b) a group comprising 20 participants (34.5%) with lower FA (Figure 3B); and (iii-a) a group comprising 50 participants (86.2%) with lower WMH burden, and (iii-b) a group comprising 8 participants (13.8%) with higher WMH burden (Figure 3C).
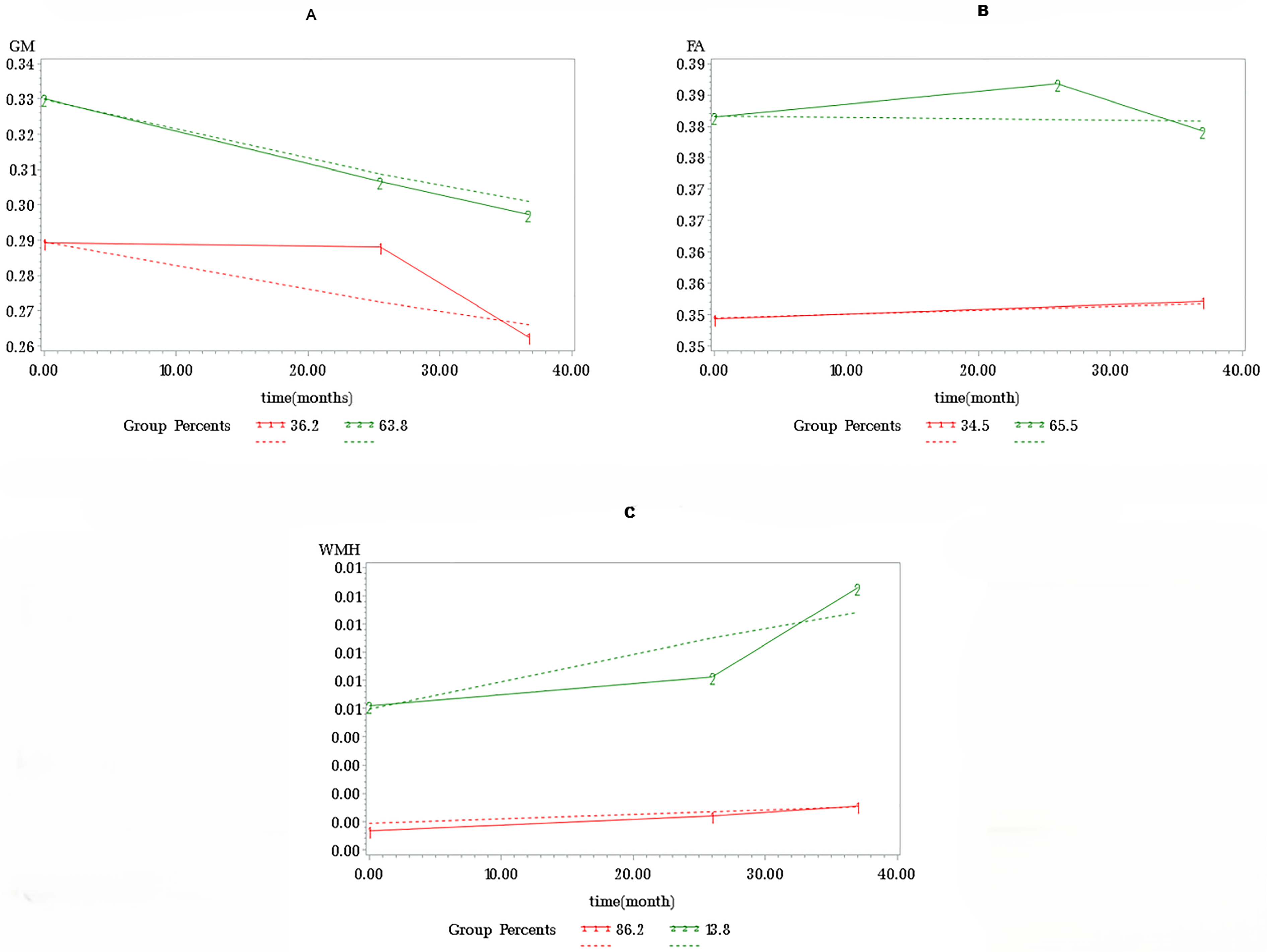
Group-based trajectory modelling of (A) gray matter volumes (GM) (mm3) versus time, (B) mean fractional anisotropy (FA) versus time, and (C) white matter hyperintensity (WMH) burden (mm3) versus time. Expected (dashed lines) versus observed (solid line) trajectories.
Associations Between Groups
Being in the higher cognitive performance group was associated with higher gray matter volume and higher FA but not associated with WMH (Table 2). There was no relationship between cognitive performance group and severity of mood symptoms (Table 2). Similarly, our exploratory analysis found no relationship between cognitive performance and being treated with lithium or divalproex (Table 2).
Group-Based Trajectory Modelling of Longitudinal Global Cognitive Performance in Relation to Indices of Brain Health, Mood, and Divalproex or Lithium Exposure.

Gray matter (GM), fractional anisotropy (FA), white matter hyperintensity burden (WMH).
Discussion
Key Findings
Using GBTM in older adults with BD followed for up to 3 years, we found cognitive performance was related to imaging markers of brain health (total gray matter and FA). We found no relationship between cognitive performance and WMH, level of mood symptoms, or exposure to lithium or divalproex. Roughly two-thirds of the participants with OABD had global cognitive performance comparable to that of comparators. Our longitudinal findings extend findings from prior cross-sectional studies examining the heterogeneity of cognitive performance in older adults with BD.19,20 They also add to the results of another single longitudinal study in OABD that found no difference in trajectory of cognitive performance or brain structures over two years between 15 patients with OABD and 15 HC. 29 To our knowledge, no previous published study has tried to integrate longitudinal cognitive performance, mood, and neuroimaging data. Broadly, our findings do not support widespread neuroprogression in patients with OABD. However, the neuroprogression model may apply to a subset of patients with OABD.
Comparison With Previous Studies
Our longitudinal analysis identified two cognitive groups. This is congruent with the results of two previous cross-sectional studies discussed in the Introduction.19,20 These studies identified three cognitive groups, two of which had better cognitive performance and approximate our group with higher cognitive performance. Also, there was no relationship between groups and measure of clinical severity (e.g., history of psychosis or hospitalization) in the study that assessed for it, 20 congruent with the absence of relationship between cognition and mood symptoms in our data. Similarly, there were no difference in cognitive impairment between patients with OABD and comparators in the large GAGE-BD study. 18
Taken together, our longitudinal findings and those of previous cross-sectional studies confirm the heterogeneity of cognitive performance observed in patients with OABD with most patients having relatively good cognitive performance.30,41 While some patients with BD who achieve older age have impaired cognitive performance, our findings and those from others3,18–20 do not support that OABD is typically associated with neuroprogression. However, it is possible that the preponderance of patients with OABD and relatively good cognitive performance is due to a survivorship bias.6,41
As expected, the groups defined based on cognitive performance were associated with the groups defined based on imaging markers of brain health, except for WMH burden, so that participants with better brain health tended have better cognition. Congruent with our findings, a clear relationship between cognition and brain structures has been reported in a systematic review of 51 studies conducted in older patients with a remitted major depressive disorder. 42 Further, reductions in cortical thickness, independent of diagnosis of BD or schizophrenia, was associated with cognitive performance in a cross-sectional analysis across older and younger patients, although FA was not. 23
We did not find a relationship between overall cognitive performance and WMH burden. This finding may be surprising since WMH burden (a marker of cerebrovascular disease) is associated with worse cognitive performance in healthy older or demented adults. 43 However, this finding extends and confirms a similar finding in one of our previous cross-sectional study of OABD (n = 27), in which WMH burden was associated with cognition in the group of HC but not in participants with OABD. 44 We speculate that this lack of association between WMH burden and cognition in OABD could be due to a variety of factors, such as survivorship bias, neuroprotective effects of lithium or other psychotropics, or biased samples of patients with BD enrolling in research in an academic medical center.
Contrary to our hypotheses, there was no significant association between the two cognitive groups and the groups defined based on depressive or manic/hypomanic symptoms. However, this may have been due to the relative stability of these mood symptoms in our participants. Similarly, our exploratory analysis did not identify a relationship between cognition and concurrent treatment with lithium or divalproex. This is in contrast with the baseline cross-sectional analysis in the same sample in which treatment with lithium was associated better markers of brain health and in turn better markers of brain health were associated with better cognition. 30 However, this cross-sectional analysis considered life-long exposure to lithium, as opposed to lithium treatment over a period of at most 3 years in the longitudinal follow-up. These differing results suggest that a long exposure to lithium may be needed to demonstrate its neuroprotective effect.
Clinical Implications and Mechanistic Interpretations
Clinically, our findings suggest that patients with OABD should have assessment of cognitive performance, and it should be a separate domain of intervention from mood. Mechanistically, that WMH burden was not associated with cognitive performance suggests that cognitive decline is more likely related to potentially longstanding neuroprogressive processes in a subset of participants, and that interventions focused on vascular disease to help with cognition are likely to be insufficient.
Limitations
Limitations of our study include that it was observational, and treatment was naturalistic. Further, the generalizability of the findings to the broader population of patients with BD is limited by a relatively small number of participants with limited diversity being treated at an academic medical center. Additionally, our participants were relatively young and without extensive medical burden. And while age of onset and duration of BD illness have been examined in neuroimaging studies of BD,21,22 this was not a focus of our analysis. Notwithstanding these limitations, our results support the conduct of larger and longer studies to better understand the complex relationship among aging, brain health, mood symptoms, and cognition in OABD. Given the challenges associated with recruiting, assessing, and retaining these patients, it is unlikely that such a study can be conducted at any single clinical center. However, it could be done if multiple centers collaborate on such a project (Bodenstein et al., under review). To that end, we have deposited this study's data with the GAGE-BD Project for future investigations. 17
Conclusion
In our study examining cognitive performance, mood symptoms, and markers of brain health in patients with OABD, we found most patients had generally normal cognitive performance for their age. Brain health markers (total gray matter and FA) were associated with cognitive performance, but there was no relationship between cognition and mood symptoms, WMH burden, or exposure to lithium or divalproex. Our findings suggest that neuroprogression may occur only in a subset of patients with OABD.
Footnotes
Acknowledgments
The authors thank Ms. Michelle Zmuda for the recruitment of control subjects and coordination of all neuropsychological assessments. AGG acknowledges the use of Anthropic's Claude AI assistant for help with editing and proofreading in the preparation of this manuscript.
Data Access
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The following disclosures within the past 36 months are reported in connection with the manuscript:
Dr. Lopez has served as a consultant for Novo Nordisk. All other authors report no financial relationships with commercial interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by the National Institutes of Health (grant numbers AG005133-28, MH076079, MH084921, MH90333).
